Risk of Transverse Myelitis Following Dengue Infection: A Systematic Review of the Literature
Abstract
:1. Introduction
2. Methods
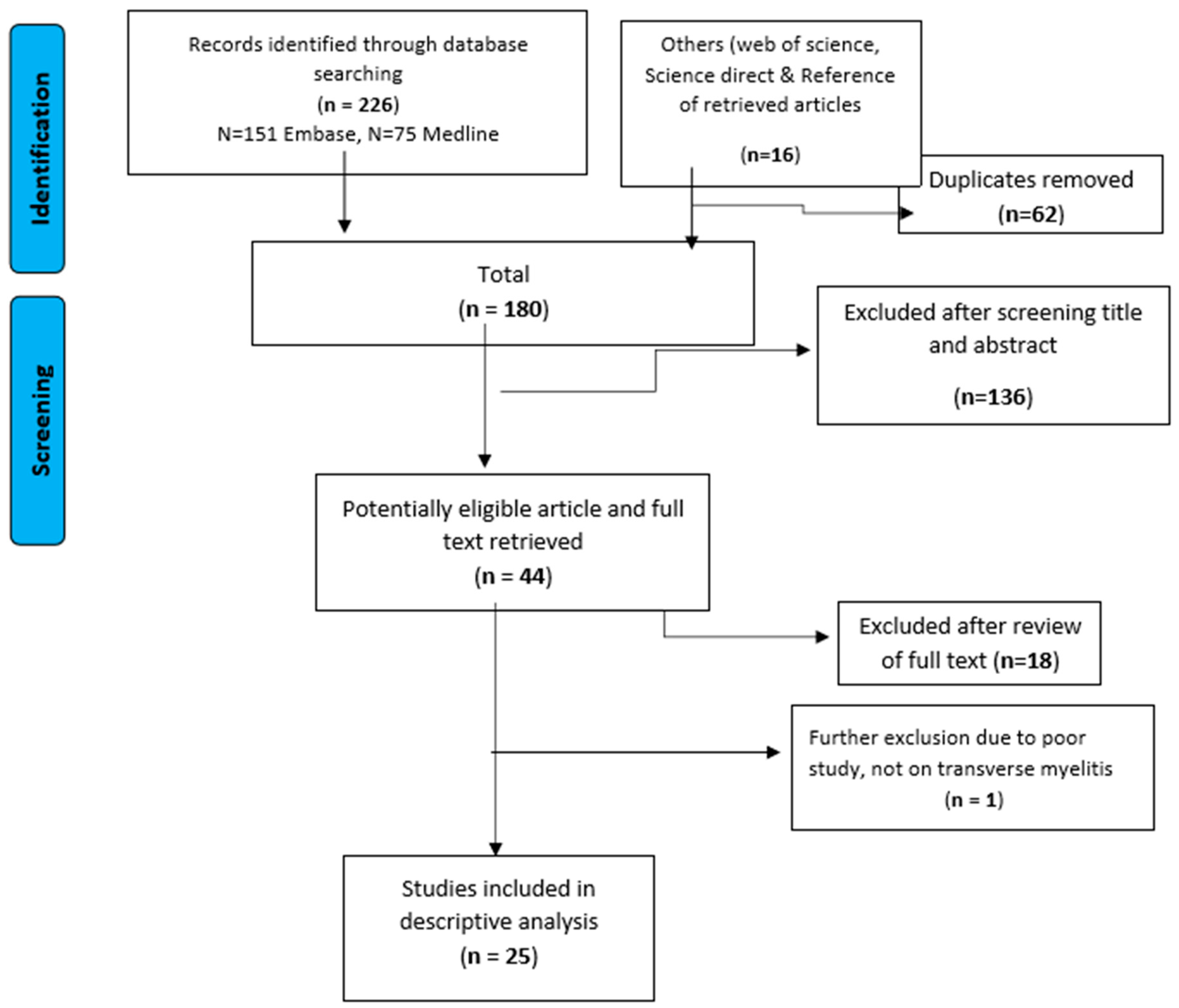
2.1. Data Search and Selection
2.2. Study Selection
2.3. Quality Assessment and Data Extraction
2.4. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Category | Search Terms |
|---|---|
| Transverse Myelitis | “transverse myelitis” or “myelitis” or “TM” or “*Myelitis, Transverse/” |
| Dengue Virus | “DEN” or “DHF” or “*Severe Dengue/” or “*Dengue Virus/* |
| Search | Results |
|---|---|
| Transverse Myelitis Category | 146,970 |
| Dengue Virus Category | 83,794 |
| Transverse Myelitis AND Dengue Virus Category (ENG) | 226 |
| (Additional publications from contacts) | 16 |
| Duplicates Removal (62) | 180 |
| Limits Applied and Studies Removed (136) | 44 |
| Title and Abstract Exclusion (18) | 26 |
| Full Text Exclusion (1) | 25 |
References
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organisation. Dengue Guidelines for Diagnosis, Treatment, Prevention and Control Treatment, Prevention and Control Treatment, Prevention and Control. 2009. Available online: http://www.who.int/tdr/publications/documents/dengue-diagnosis.pdf (accessed on 26 April 2018).
- Carod-Artal, F.J.; Wichmann, O.; Farrar, J.; Gascón, J. Neurological complications of dengue virus infection. Lancet Neurol. 2013, 12, 906–919. [Google Scholar] [CrossRef]
- Hendarto, S.K.; Hadinegoro, S.R. Dengue encephalopathy. Acta Paediatr. Jpn. 1992, 34, 50–57. [Google Scholar] [CrossRef]
- Kho, L.K.; Sumarmo; Wulur, H.; Jahja, E.C.; Gubler, D.J. Dengue hemorrhagic fever accompanied by encephalopathy in Jakarta. Southeast Asian J. Trop. Med. Public Health 1981, 12, 83–86. [Google Scholar] [PubMed]
- Nimmannitya, S.; Thisyakorn, U.; Hemsrichart, V. Dengue haemorrhagic fever with unusual manifestations. Southeast Asian J. Trop. Med. Public Health 1987, 18, 398–406. [Google Scholar]
- Seet, R.C.S.; Lim, E.C.H.; Wilder-Smith, E.P.V. Acute transverse myelitis following dengue virus infection. J. Clin. Virol. 2006, 35, 310–312. [Google Scholar] [CrossRef]
- Li, G.H.; Ning, Z.J.; Liu, Y.M.; Li, X.H. Neurological Manifestations of Dengue Infection. Front. Cell Infect. Microbioogyl. 2017, 7, 449. [Google Scholar] [CrossRef]
- De Sousa, A.M.; Alvarenga, M.P.; Alvarenga, R.M.P. A cluster of transverse myelitis following dengue virus infection in the brazilian Amazon region. Trop. Med. Health 2014, 42, 115–120. [Google Scholar] [CrossRef]
- Larik, A.; Chiong, Y.; Lee, L.C.; Ng, Y.S. Longitudinally extensive transverse myelitis associated with dengue fever. BMJ Case Rep. 2012, 2012, bcr1220115378. [Google Scholar] [CrossRef]
- Singh, P.; Joseph, B. Paraplegia in a patient with dengue. Neurol. India 2010, 58, 962. [Google Scholar] [CrossRef]
- Puccioni-Sohler, M.; Soares, C.N.; Papaiz-Alvarenga, R.; Castro, M.J.C.; Faria, L.C.; Peralta, J.M. Neurologic dengue manifestations associated with intrathecal specific immune response. Neurology 2009, 73, 1413–1417. [Google Scholar] [CrossRef]
- Kunishige, M.; Mitsui, T.; Tan, B.H.; Leong, H.N.; Takasaki, T.; Kurane, I.; Mihara, A.; Matsumoto, T. Preferential gray matter involvement in dengue myelitis. Neurology 2004, 63, 1980–1981. [Google Scholar] [CrossRef] [PubMed]
- Leão, R.N.; Oikawa, T.; Rosa, E.S.; Yamaki, J.T.; Rodrigues, S.G.; Vasconcelos, H.B.; Sousa, M.R.; Tsukimata, J.K.; Azevedo, R.S.; Vasconcelos, P.F. Isolation of dengue 2 virus from a patient with central nervous system involvement (transverse myelitis). Rev. Soc. Bras. Med. Trop. 2002, 35, 401–404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chanthamat, N.; Sathirapanya, P. Acute transverse myelitis associated with dengue viral infection. J. Spinal Cord. Med. 2010, 33, 425–427. [Google Scholar] [CrossRef]
- PRISMA. The PRISMA Statement. Available online: http://www.prisma-statement.org/statement.htm (accessed on 1 October 2018).
- Singh, M. Spinal epidural hematoma with myelitis and brainstem hemorrhage: An unusual complication of dengue fever. Neurol. India 2013, 61, 541–543. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, M. Dengue infection with multi-organ involvement. Scand. J. Infect. Dis. 2011, 43, 316–318. [Google Scholar] [CrossRef]
- Fong, C.Y. Longitudinal extensive transverse myelitis with cervical epidural haematoma following dengue virus infection. Eur. J. Paediatr. Neurol. 2016, 20, 449–453. [Google Scholar] [CrossRef]
- Gupta, M. Acute disseminated encephalomyelitis associated with dengue infection: A case report with literature review. J. Neurol. Sci. 2013, 335, 216–218. [Google Scholar] [CrossRef]
- Wasay, M. Encephalitis and myelitis associated with dengue viral infection clinical and neuroimaging features. Clin. Neurol. Neurosurg. 2008, 110, 635–640. [Google Scholar] [CrossRef]
- Samanta, M. Unique neurological manifestations of dengue virus in pediatric population: A case series. J. Trop. Pediatr. 2012, 58, 398–401. [Google Scholar] [CrossRef]
- Misra, U.K.; Kalita, J.; Mani, V.E.; Chauhan, P.S.; Kumar, P. Central nervous system and muscle involvement in dengue patients: A study from a tertiary care center. J. Clin. Virol. 2015, 72, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Sahu, R. Neurologic complications in dengue virus infection: A prospective cohort study. Neurology 2014, 83, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Soares, C.N. Dengue infection: Neurological manifestations and cerebrospinal fluid (CSF) analysis. J. Neurol. Sci. 2006, 249, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Weeratunga, P.N. Neurological manifestations of dengue: A cross sectional study. Travel Med. Infect. Dis. 2014, 12, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.W.; Divakar, G.T.; Lo, Y.L.; Yip, C.W. Poster session 2, Monday 10 September. Eur. J. Neurol. 2012, 19, 648. [Google Scholar]
- Tomar, L.R.; Mannar, V.; Pruthi, S.; Aggarwal, A. An Unusual Presentation of Dengue Fever: Association with Longitudinal Extensive Transverse Myelitis. Permanente J. 2015, 19, 133. [Google Scholar]
- Mo, Z.; Dong, Y.; Chen, X.; Yao, H.; Zhang, B. Acute transverse myelitis and subacute thyroiditis associated with dengue viral infection: A case report and literature review. Exp. Ther. Med. 2016, 12, 2331–2335. [Google Scholar] [CrossRef] [Green Version]
- De Oliveira Mota, M.T.; Estofolete, C.F.; Zini, N.; Terzian, A.C.; Gongora, D.V.; Maia, I.L.; Nogueira, M.L. Transverse Myelitis as an Unusual Complication of Dengue Fever. Am. J. Trop. Med. Hyg. 2017, 96, 380–381. [Google Scholar] [CrossRef]
- De Sousa, A.M.; Puccioni-Sohler, M.; Alvarenga, M.P.; Alvarenga, R.M.; Borges, A.D.; Adorno, L.F. Post-dengue neuromyelitis optica: Case report of a Japanese-descendent Brazilian child. J. Infect. Chemother. 2006, 12, 396–398. [Google Scholar] [CrossRef]
- Renganathan, A.; Ng, W.K.; Tan, C.T. Transverse myelitis in association with dengue infection. Neurol. J. Southeast Asia 1996, 1, 61–63. [Google Scholar]
- Solomon, T.; Dung, N.M.; Vaughn, D.W.; Kneen, R.; Raengsakulrach, B.; Loan, H.T.; Day, N.P.; Farrar, J.; Myint, K.S.; Warrell, M.J.; et al. Neurological manifestations of dengue infection. Lancet 2000, 353, 1053–1059. [Google Scholar] [CrossRef]
- Verma, R.; Sharma, P.; Garg, R.K.; Atam, V.; Singh, M.K.; Mehrotra, H.S. Neurological complications of dengue fever: Experience from a tertiary center of north India. Ann. Indian Acad. Neurol. 2011, 14, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Brady, O.J.; Gething, P.W.; Bhatt, S.; Messina, J.P.; Brownstein, J.S.; Hoen, A.G.; Moyes, C.L.; Farlow, A.W.; Scott, T.W.; Hay, S.I. Refining the global spatial limits of dengue virus transmission by evidence-based consensus. PLoS Neglected Trop. Dis. 2012, 6, e1760. [Google Scholar] [CrossRef] [PubMed]
- Wilder-Smith, A.; Bypass, P. The elusive global burden of dengue. Lancet Infect. Dis. 2016, 16, 629–631. [Google Scholar] [CrossRef]
- Mishra, G.; Jain, A.; Prakash, O.; Prakash, S.; Kumar, R.; Garg, R.K.; Pandey, N.; Singh, M. Molecular characterization of dengue viruses circulating during 2009–2012 in Uttar Pradesh, India. J. Med. Virol. 2015, 87, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Afreen, N.; Deeba, F.; Naqvi, I.; Shareef, M.; Ahmed, A.; Broor, S.; Parveen, S. Molecular investigation of 2013 dengue fever outbreak from Delhi, India. PLOS Curr. Outbreaks 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Zhou, D.S.; Kawasaki, K.; Yasui, K. The pathogenesis of spinal cord involvement in dengue virus infection. Virch. Arch. 2003, 442, 472–481. [Google Scholar]
- Regitz-Zagrosek, V. Sex and gender differences in health: Science & Society Series on Sex and Science. EMBO Rep. 2012, 13, 596–603. [Google Scholar]
- Zandman-Goddard, G.; Peeva, E.; Rozman, Z.; Ben-Zvi, I.; Langevitz, P.; Shvartser, Y.; Amital, D.; Amital, H.; Kivity, S.; Lidar, M.; et al. Sex and gender differences in autoimmune diseases. In Sex and Gender Aspects in Clinical Medicine; Springer: London, UK, 2012; pp. 101–124. [Google Scholar]
- Lahat, E.; Pillar, G.; Ravid, S.; Barzilai, A.; Etzioni, A.; Shahar, E. Rapid recovery from transverse myelopathy in children treated with methylprednisolone. Paediatr. Neurol. 1998, 19, 279–282. [Google Scholar] [CrossRef]
| Study | Year of Study | Country | Study Design | Number of Dengue Cases | Diagnosis Method | Outcome |
|---|---|---|---|---|---|---|
| Singh et al. [17] | 2013 | India | Case Report | 1 | Dengue IgM seropositivity | 1 patient died. |
| Ghosh et al. [18] | 2011 | India | Case Report | 1 | Dengue IgM seropositivity CSF analysis | full recovery |
| Seet et al. [7] | 2006 | Singapore | Case Report | 1 | Antibody index ratio of dengue IgM:IgG | full recovery |
| Kunishige et al. [13] | 2004 | Singapore | Case Report | 1 | Dengue IgM seropositivity (CSF) | partial recovery |
| Fong CY et al. [19] | 2016 | Malaysia | Case Report | 1 | Dengue IgM seropositivity | full recovery |
| Gupta et al. [20] | 2013 | India | Case report | 1 | Dengue IgM seropositivity (ELISA) History NS1 ag assay | full recovery |
| Wasay et al. [21] | 2008 | Pakistan | Case Series | 6 | Dengue IgM seropositivity | 4 patients made a full recovery, 2 patients died. |
| Samanta et al. [22] | 2012 | India | Case Series | 3 | Dengue IgM seropositivity (serum/viral/blood) | 1 patient made a full recovery, 1 patient made a partial recovery, 1 patient died. |
| Misra et al. [23] | 2015 | India | Case Study | 116 | Dengue IgM seropositivity History, Exam, NS1 antigen assay | 78 patients made a full recovery, 27 patients made a partial recovery, 11 patients died. |
| Sahu et al. [24] | 2014 | India | Cohort | 484 | Dengue IgM seropositivity | 479 patients made a full recovery, 5 patients died |
| Soars et al. [25] | 2006 | Brazil | Cross-sectional study | 13 | Dengue IgM seropositivity (blood/CSF (ELISA)) | 12 patients made a full recovery, 1 patient with encephalitis died |
| Weeratunga et al. [26] | 2014 | Sri Lanka | Cross-sectional Study | 7 | Dengue IgM seropositivity (blood/CSF) | 6 patients made a full recovery, 1 patient made a partial recovery. |
| Puccioni-Sohler et al. (Brazil) [12] | 2009 | Brazil | Retrospective study | 27 | Dengue IgM seropositivity | Partial recovery |
| Larik et al. [10] | 2012 | Singapore | Case Report | 1 | Dengue IgM seropositivity Dengue RNA | Full recovery |
| Lim et al. [27] | 2012 | Singapore | Case Report | 1 | Dengue IgM seropositivity | Partial recovery |
| Tomar et al. [28] | 2015 | India | Case Report | 1 | Dengue IgM seropositivity | Full recovery |
| Mo et al. [29] | 2016 | China | Case Report | 1 | Dengue IgM/IgG seropositivity (CSF) | Partial recovery |
| Mota et al. [30] | 2017 | Brazil | Case Report | 1 | Dengue IgM seropositivity | Partial recovery |
| Leão et al. [14] | 2000 | Brazil | Case Report | 1 | Dengue IgM seropositivity (CSF) | Full recovery |
| Miranda de Sousa A et al. [31] | 2014 | Brazil | Case Report | 1 | Dengue IgM seropositivity (CSF) | Full recovery |
| Renganathan et al. [32] | 1996 | Malaysia | Case Report | 1 | Dengue IgM seropositivity | Full recovery |
| Chanthamat et al. [15] | 2010 | Thailand | Case Report | 1 | NA | Full recovery |
| Solomon et al. [33] | 2000 | Vietnam | Prospective Study | 1675 | IgM/IgG seropositivity (CSF) | Partial recovery |
| Sousa et al. [9] | 2004 | Brazil | Retrospective Study | 51 | Dengue IgM seropositivity (CSF) | 49 patients made a full recovery, 2 patients had partial recovery |
| Verma et al. [34] | 2011 | India | Retrospective Study | 26 | Dengue IgM seropositivity | Partial recovery |
| Study | Age | Sex | Number of TM Cases | Serotype | Treatment |
|---|---|---|---|---|---|
| Singh et al. [17] | 45 | Male | 1 | - | T9-11 laminectomy Evacuation of epidural haematoma. Multiple blood and platelet transfusions. Conservative management applied. |
| Ghosh et al. [18] | 4 | 1 | - | High dose methylprednisolone. Platelet transfusion and Packed red cell Supportive therapy for hepatitis and glomerulonephritis | |
| Seet et al. [7] | 44 | Female | 1 | - | IV methylprednisolone 1 g for 5 days, Spinal MRI, Catheterisation for urinary retention, Intensive physiotherapy |
| Kunishige et al. [13] | 42 | Male | 1 | 1 | IV methylprednisolone Antibiotics |
| Fong CY et al. [19] | 12 | Female | 1 | - | IV methylprednisolone 30 mg/kg/day for 3 days followed by oral prednisolone IV Immunoglobulin (IVIG) 1 g/kg/day for 2 days. Intubated, 6 cycles of plasma exchange. Cervical epidural haematoma was managed conservatively |
| Gupta et al. [20] | 26 | Female | 1 | - | Methylprednisolone 1.0 mg/5 days Mechanical ventilation for 2 weeks |
| Wasay et al. [21] | 18–35 | 5 females, 1 male | 1 | - | MRI/CT +/‒ EEG observations |
| Samanta et al. [22] | Male | 1 | primary/ secondary infection | Pulsed methylprednisolone Conservative therapy | |
| Misra et al. [23] | 5–70. | 26 females, 90 males | 1 | 1, 2 and 3 | - |
| Sahu et al. [24] | 25 +/‒ 18.3 | 7 | - | Symptomatic treatment | |
| Soars et al. [25] | 11–79. | 10 female, 3 male | 2 | 1, 2 and 3 | Corticosteroids IVIG |
| Weeratunga et al. [26] | Mean: 35 | 1 female, 6 male | 2 | - | Methylprednisolone pulsed 1 g/3 days |
| Puccioni-Sohler et al. (Brazil) [12] | 22–74 | 6 females, 4 males | 3 | - | Methylprednisolone 1.0 mg/5 days. Additional Human IVIG 400 mg/kg/5 days for 1 patient |
| Larik et al. [10] | 43 | Male | 1 | - | IVIG |
| Lim et al. [27] | 43 | Male | 1 | - | IVIG |
| Tomar et al. [28] | 42 | Male | 1 | - | IV Methylprednisolone |
| Mo et al. [29] | 65 | Male | 1 | - | IV Methylprednisolone IVIG Plasma Exchange |
| Mota et al. [30] | 21 | Male | 1 | - | IV Methylprednisolone |
| Leão et al. [14] | 58 | Male | 1 | - | Ceftriaxone |
| Miranda de Sousa A et al. [31] | 11 | Female | 1 | - | IV Methylprednisolone 1g/day followed by prednisolone |
| Renganathan et al. [32] | 14 | Female | 1 | - | Symptomatic treatment |
| Chanthamat et al. [15] | 61 | Female | 1 | - | IV Methylprednisolone |
| Solomon et al. [33] | 2 | - | Symptomatic treatment | ||
| Sousa et al. [9] | Mean:34 | 26 | 3 | IV methylprednisolone for 5 days | |
| Verma et al. [34] | 8 females, 18 males | 1 | - | IV Methylprednisolone |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Badat, N.; Abdulhussein, D.; Oligbu, P.; Ojubolamo, O.; Oligbu, G. Risk of Transverse Myelitis Following Dengue Infection: A Systematic Review of the Literature. Pharmacy 2019, 7, 3. https://doi.org/10.3390/pharmacy7010003
Badat N, Abdulhussein D, Oligbu P, Ojubolamo O, Oligbu G. Risk of Transverse Myelitis Following Dengue Infection: A Systematic Review of the Literature. Pharmacy. 2019; 7(1):3. https://doi.org/10.3390/pharmacy7010003
Chicago/Turabian StyleBadat, Nafisa, Dalia Abdulhussein, Peter Oligbu, Olakunle Ojubolamo, and Godwin Oligbu. 2019. "Risk of Transverse Myelitis Following Dengue Infection: A Systematic Review of the Literature" Pharmacy 7, no. 1: 3. https://doi.org/10.3390/pharmacy7010003
APA StyleBadat, N., Abdulhussein, D., Oligbu, P., Ojubolamo, O., & Oligbu, G. (2019). Risk of Transverse Myelitis Following Dengue Infection: A Systematic Review of the Literature. Pharmacy, 7(1), 3. https://doi.org/10.3390/pharmacy7010003





