Analysis of Grape Production in the Face of Climate Change
Abstract
:1. Introduction
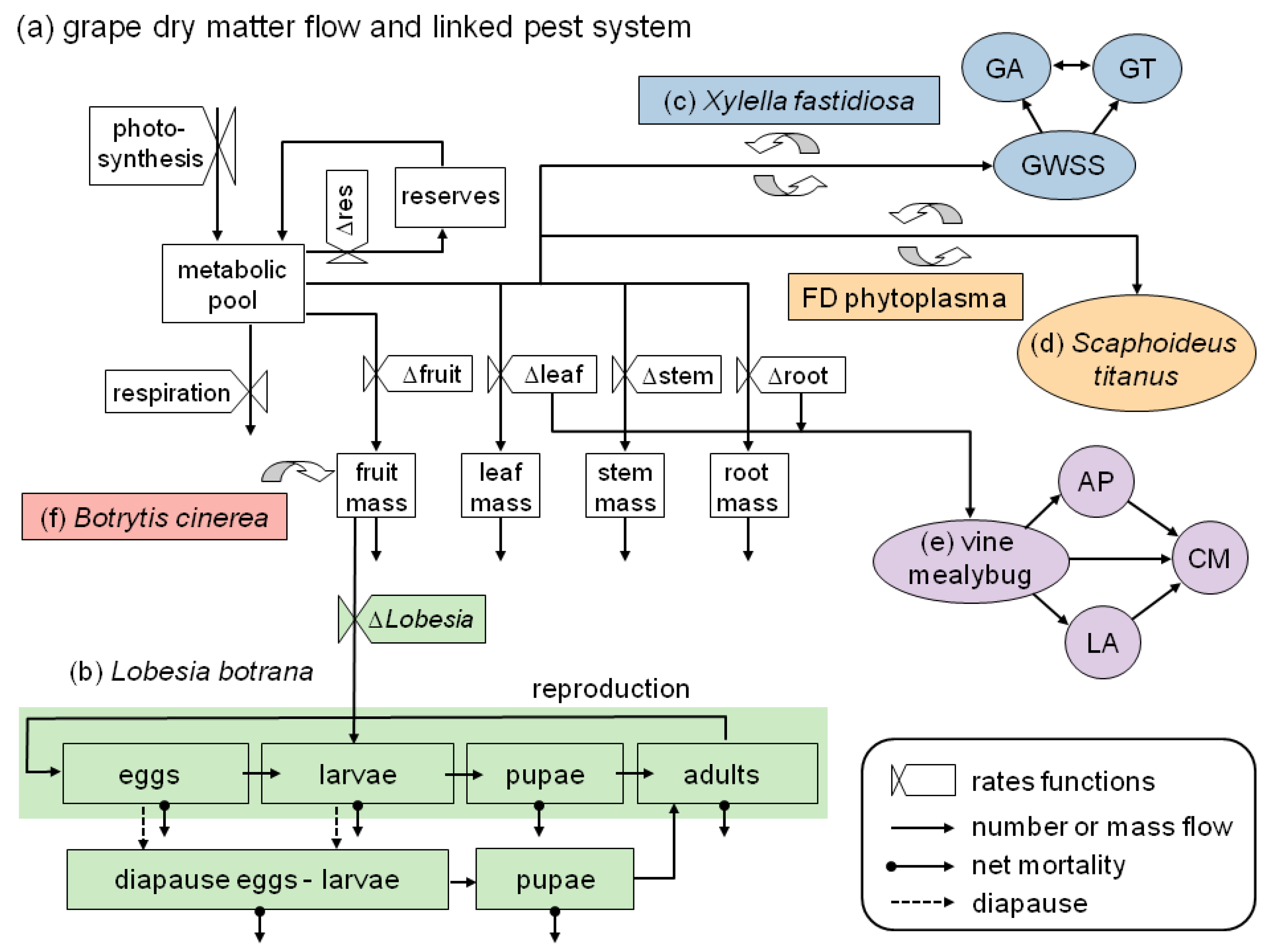
- It illustrates the ecological and geographic complexity involved in assessing the bioeconomics of grape production under climate change, including an expanded overview on ongoing and prospective work in PBDM analysis of the pest/vector/disease complex of grape (Figure 1);
- It pinpoints key ecological differences that drive different levels of management and external input intensity in olive and grape, the two major perennial traditional cropping systems of Mediterranean agriculture (Section 2);
- It provides a broad overview on how PBDMs in a GIS context can be used to explore mechanistically otherwise mostly intractable complex problems such as crop-pests interactions that lie at the interface between global change and biological systems (i.e., global change biology) based on the paradigm of ecological analogies (Section 3);
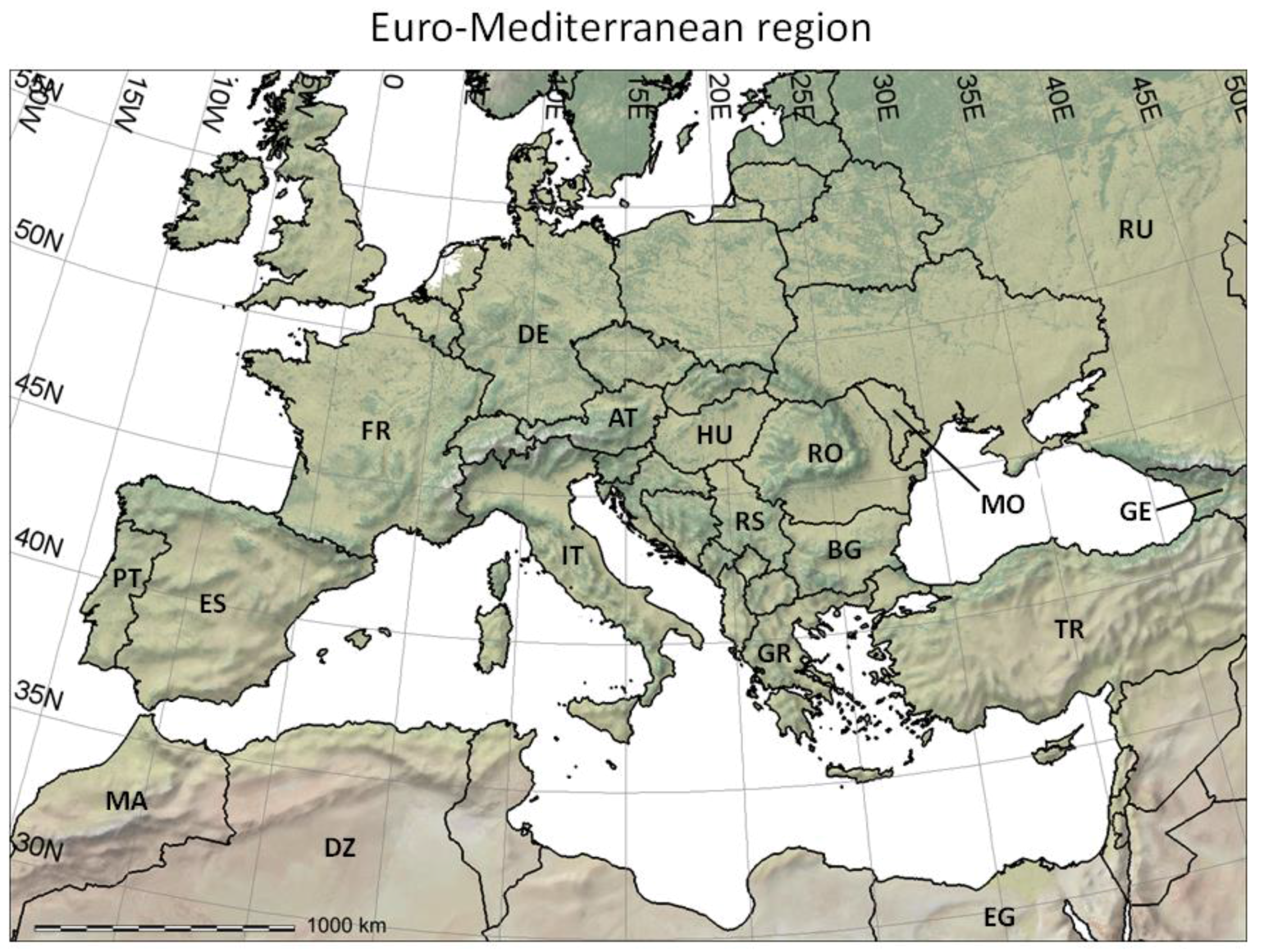
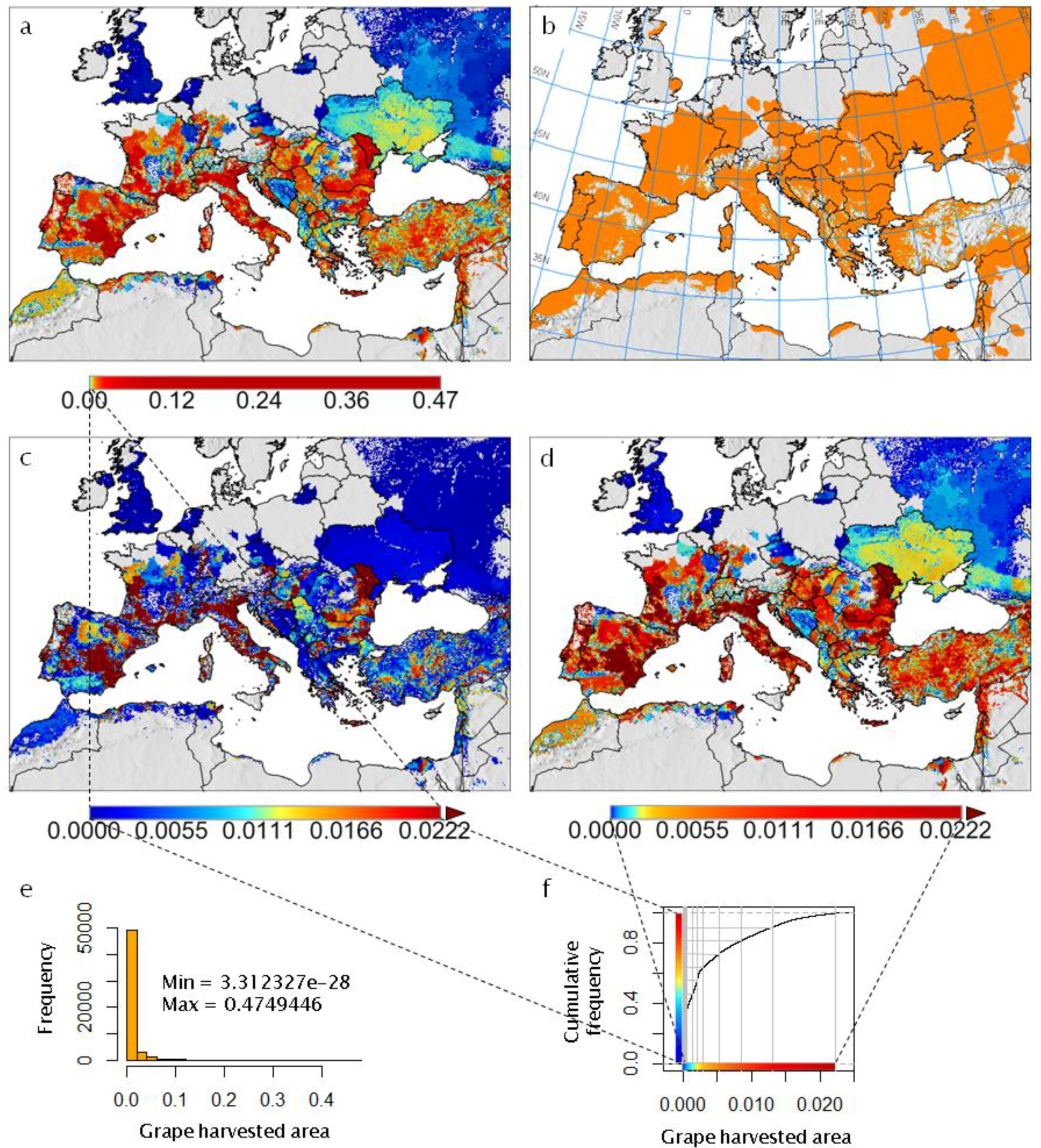
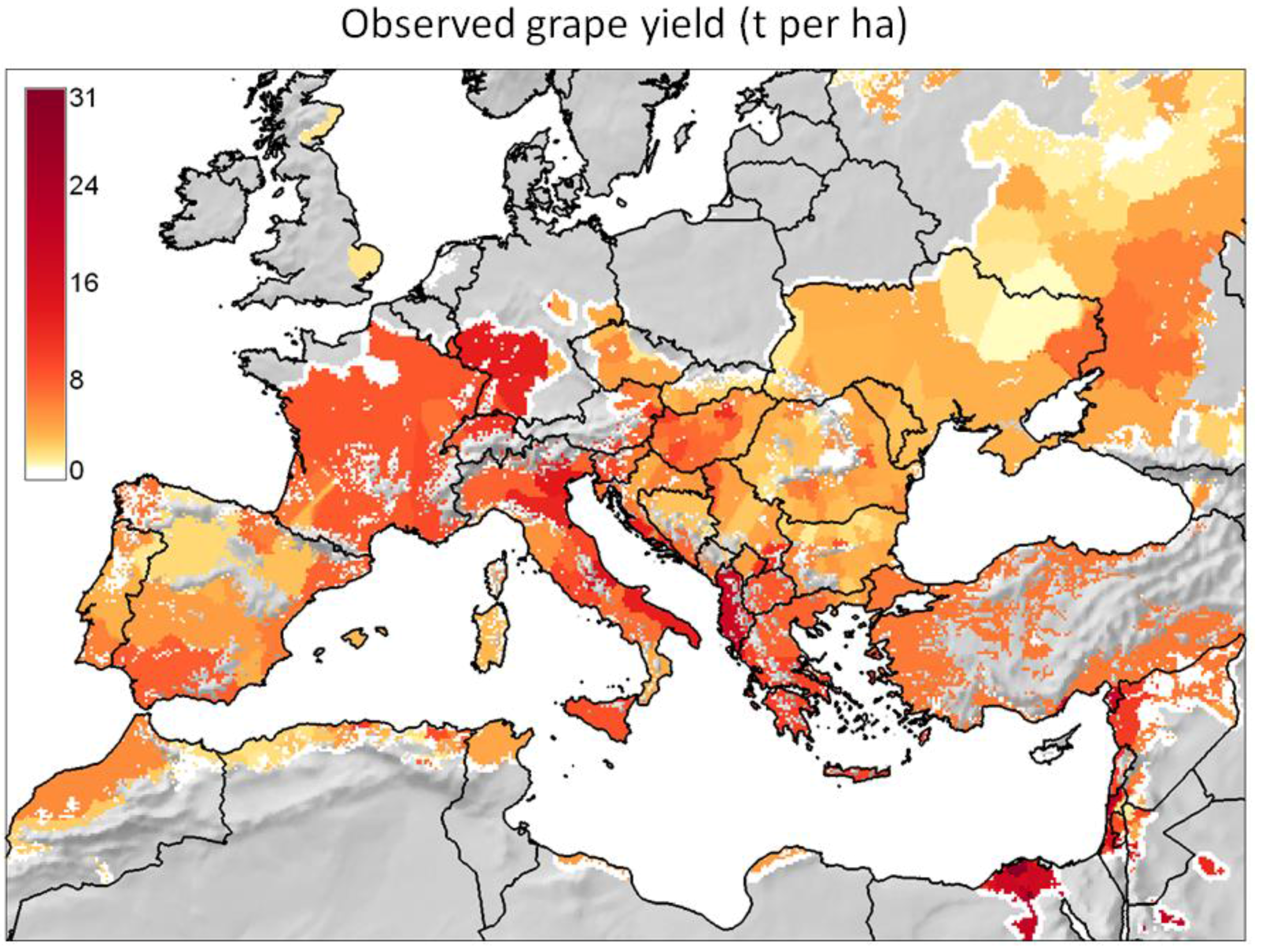
- It reviews the GIS context for PBDMs by illustrating how GRASS GIS [14] can be linked to the free software environment for statistical computing and graphics R [15] to analyze (Figure 2 and Figure 3) and assess (Figure 4) the observed geographic distribution of grape (or any other crop) production in the Euro-Mediterranean region (or any other region including globally);
- For each of the major grape growing countries of the Euro-Mediterranean region (Figure 2) it shows the probability distribution of changes in grape yield and grapevine moth infestation (Figure 5 and Figure 6), as well as the fraction of grape growing area in each country where these changes are expected to be positive or negative (Table 1 and Table 2);
- It ranks the 18 major Euro-Mediterranean grape growing countries in terms of the following bioeconomic measures of climate risk: (a) mean climate change impact on grape yield and grapevine moth infestation (Figure 5 and Figure 6); (b) relative share of the grape growing area in each country where grape yield and grapevine moth infestation are expected to be negative or positive (Table 1 and Table 2).
2. Olive vs. Grape
3. The PBDM Approach
4. The GIS Context for PBDMs
5. Climate Change Effects on Grape and Its Major Insect Pest
6. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vivier, M.A.; Pretorius, I.S. Genetically tailored grapevines for the wine industry. Trends Biotechnol. 2002, 20, 472–478. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Lagiou, P. Healthy traditional Mediterranean diet: An expression of culture, history, and lifestyle. Nutr. Rev. 1997, 55, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Grigg, D. Food consumption in the Mediterranean region. Tijdschr. Voor Econ. En Soc. Geogr. 1999, 90, 391–409. [Google Scholar] [CrossRef]
- Blondel, J. The “design” of Mediterranean landscapes: A millennial story of humans and ecological systems during the historic period. Hum. Ecol. 2006, 34, 713–729. [Google Scholar] [CrossRef]
- Medina, F.X. Mediterranean diet, culture and heritage: Challenges for a new conception. Public Health Nutr. 2009, 12, 1618–1620. [Google Scholar] [CrossRef] [PubMed]
- United Nations Educational, Scientific and Cultural Organization (UNESCO). Mediterranean Diet. Available online: https://ich.unesco.org/en/RL/mediterranean-diet-00884 (accessed on 1 March 2017).
- Ponti, L.; Gutierrez, A.P.; Altieri, M.A. Preserving the Mediterranean diet through holistic strategies for the conservation of traditional farming systems. In Biocultural Diversity in Europe; Agnoletti, M., Emanueli, F., Eds.; Environmental History; Springer: Cham, Switzerland, 2016; pp. 453–469. ISBN 978-3-319-26313-7. [Google Scholar]
- Pattara, C.; Russo, C.; Antrodicchia, V.; Cichelli, A. Carbon footprint as an instrument for enhancing food quality: Overview of the wine, olive oil and cereals sectors. J. Sci. Food Agric. 2017, 97, 396–410. [Google Scholar] [CrossRef] [PubMed]
- Alessandri, A.; De Felice, M.; Zeng, N.; Mariotti, A.; Pan, Y.; Cherchi, A.; Lee, J.-Y.; Wang, B.; Ha, K.-J.; Ruti, P.; Artale, V. Robust assessment of the expansion and retreat of Mediterranean climate in the 21st century. Sci. Rep. 2014, 4, 7211. [Google Scholar] [CrossRef] [PubMed]
- Geri, F.; Amici, V.; Rocchini, D. Human activity impact on the heterogeneity of a Mediterranean landscape. Appl. Geogr. 2010, 30, 370–379. [Google Scholar] [CrossRef]
- Hannah, L.; Roehrdanz, P.R.; Ikegami, M.; Shepard, A.V.; Shaw, M.R.; Tabor, G.; Zhi, L.; Marquet, P.A.; Hijmans, R.J. Climate change, wine, and conservation. Proc. Natl. Acad. Sci. USA 2013, 110, 6907–6912. [Google Scholar] [CrossRef] [PubMed]
- Ponti, L.; Gutierrez, A.P.; Ruti, P.M.; Dell’Aquila, A. Fine-scale ecological and economic assessment of climate change on olive in the Mediterranean Basin reveals winners and losers. Proc. Natl. Acad. Sci. USA 2014, 111, 5598–5603. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.P.; Ponti, L.; Gilioli, G.; Baumgärtner, J. Climate warming effects on grape and grapevine moth (Lobesia botrana) in the Palearctic region. Agric. For. Entomol. 2017. [Google Scholar] [CrossRef]
- Neteler, M.; Bowman, M.H.; Landa, M.; Metz, M. GRASS GIS: A multi-purpose Open Source GIS. Environ. Model. Softw. 2012, 31, 124–130. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: http://www.R-project.org (accessed on 26 June 2017).
- Kaniewski, D.; Van Campo, E.; Boiy, T.; Terral, J.-F.; Khadari, B.; Besnard, G. Primary domestication and early uses of the emblematic olive tree: Palaeobotanical, historical and molecular evidence from the Middle East. Biol. Rev. 2012, 87, 885–899. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, R.C. Recording rural landscapes and their cultural associations: Some initial results and impressions. Environ. Sci. Policy 2006, 9, 360–369. [Google Scholar] [CrossRef]
- Sirami, C.; Nespoulous, A.; Cheylan, J.-P.; Marty, P.; Hvenegaard, G.T.; Geniez, P.; Schatz, B.; Martin, J.-L. Long-term anthropogenic and ecological dynamics of a Mediterranean landscape: Impacts on multiple taxa. Landsc. Urban Plan. 2010, 96, 214–223. [Google Scholar] [CrossRef]
- Lavorel, S. Ecological diversity and resilience of Mediterranean vegetation to disturbance. Divers. Distrib. 1999, 5, 3–13. [Google Scholar] [CrossRef]
- Batáry, P.; Báldi, A.; Kleijn, D.; Tscharntke, T. Landscape-moderated biodiversity effects of agri-environmental management: A meta-analysis. Proc. R. Soc. Lond. B Biol. Sci. 2011, 278, 1894–1902. [Google Scholar] [CrossRef] [PubMed]
- Bagella, S.; Filigheddu, R.; Caria, M.C.; Girlanda, M.; Roggero, P.P. Contrasting land uses in Mediterranean agro-silvo-pastoral systems generated patchy diversity patterns of vascular plants and below-ground microorganisms. Comptes Rendus Biol. 2014, 337, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Bagella, S.; Caria, M.C.; Farris, E.; Rossetti, I.; Filigheddu, R. Traditional land uses enhanced plant biodiversity in a Mediterranean agro-silvo-pastoral system. Plant Biosyst. Int. J. Deal. Asp. Plant Biol. 2016, 150, 201–207. [Google Scholar] [CrossRef]
- Provost, C.; Pedneault, K. The organic vineyard as a balanced ecosystem: Improved organic grape management and impacts on wine quality. Sci. Hortic. 2016, 208, 43–56. [Google Scholar] [CrossRef]
- Viggiani, G. La difesa integrata dell’olivo: Attualità e prospettive. Inf. Fitopatol. 1989, 2, 23–32. [Google Scholar]
- Daane, K.M.; Johnson, M.W. Olive fruit fly: Managing an ancient pest in modern times. Annu. Rev. Entomol. 2010, 55, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Pertot, I.; Caffi, T.; Rossi, V.; Mugnai, L.; Hoffmann, C.; Grando, M.S.; Gary, C.; Lafond, D.; Duso, C.; Thiéry, D.; et al. A critical review of plant protection tools for reducing pesticide use on grapevine and new perspectives for the implementation of IPM in viticulture. Crop Prot. 2016. [Google Scholar] [CrossRef]
- Delcour, I.; Spanoghe, P.; Uyttendaele, M. Literature review: Impact of climate change on pesticide use. Food Res. Int. 2015, 68, 7–15. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Ponti, L.; Gilioli, G. Climate change effects on plant-pest-natural enemy interactions. In Handbook of Climate Change and Agroecosystems: Impacts, Adaptation, and Mitigation; Hillel, D., Rosenzweig, C., Eds.; Imperial College Press: London, UK, 2010; pp. 209–237. ISBN 978-1-84816-655-4. [Google Scholar]
- Gutierrez, A.P.; Ponti, L. Analysis of invasive insects: Links to climate change. In Invasive Species and Global Climate Change; Ziska, L.H., Dukes, J.S., Eds.; CABI Publishing: Wallingford, UK, 2014; pp. 45–61. [Google Scholar]
- Gutierrez, A.P. Applied Population Ecology: A Supply-Demand Approach; John Wiley and Sons: New York, NY, USA, 1996; ISBN 0-471-13586-0. [Google Scholar]
- Zavaleta, E.S.; Hobbs, R.J.; Mooney, H.A. Viewing invasive species removal in a whole-ecosystem context. Trends Ecol. Evol. 2001, 16, 454–459. [Google Scholar] [CrossRef]
- Hulme, P.E. Beyond control: Wider implications for the management of biological invasions. J. Appl. Ecol. 2006, 43, 835–847. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change (IPCC). Climate Change 2007: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2007; ISBN 978-0-521-70597-4. [Google Scholar]
- Intergovernmental Panel on Climate Change (IPCC). Climate change 2014: Impacts, Adaptation, and Vulnerability. Part A: Global and sectoral aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014; ISBN 978-1-107-64165-5. [Google Scholar]
- Sutherst, R.; Bourne, A. Modelling non-equilibrium distributions of invasive species: A tale of two modelling paradigms. Biol. Invasions 2009, 11, 1231–1237. [Google Scholar] [CrossRef]
- Gilman, S.E.; Urban, M.C.; Tewksbury, J.; Gilchrist, G.W.; Holt, R.D. A framework for community interactions under climate change. Trends Ecol. Evol. 2010, 25, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.P.; Ponti, L. Eradication of invasive species: Why the biology matters. Environ. Entomol. 2013, 42, 395–411. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.P.; Williams, D.W.; Kido, H. A model of grape growth and development: The mathematical structure and biological considerations. Crop Sci. 1985, 25, 721–728. [Google Scholar] [CrossRef]
- Wermelinger, B.; Baumgärtner, J.; Gutierrez, A.P. A demographic model of assimilation and allocation of carbon and nitrogen in grapevines. Ecol. Model. 1991, 53, 1–26. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Ponti, L.; Cooper, M.L.; Gilioli, G.; Baumgärtner, J.; Duso, C. Prospective analysis of the invasive potential of the European grapevine moth Lobesia botrana (Den. & Schiff.) in California. Agric. For. Entomol. 2012, 14, 225–238. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Ponti, L.; Hoddle, M.; Almeida, R.P.P.; Irvin, N.A. Geographic distribution and relative abundance of the invasive glassy-winged sharpshooter: Effects of temperature and egg parasitoids. Environ. Entomol. 2011, 40, 755–769. [Google Scholar] [CrossRef] [PubMed]
- Rigamonti, I.E.; Jermini, M.; Fuog, D.; Baumgärtner, J. Towards an improved understanding of the dynamics of vineyard-infesting Scaphoideus titanus leafhopper populations for better timing of management activities. Pest Manag. Sci. 2011, 67, 1222–1229. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.P.; Daane, K.M.; Ponti, L.; Walton, V.M.; Ellis, C.K. Prospective evaluation of the biological control of vine mealybug: Refuge effects and climate. J. Appl. Ecol. 2008, 45, 524–536. [Google Scholar] [CrossRef]
- González-Domínguez, E.; Caffi, T.; Ciliberti, N.; Rossi, V. A mechanistic model of Botrytis cinerea on grapevines that includes weather, vine growth stage, and the main infection pathways. PLoS ONE 2015, 10, e0140444. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.P.; Baumgärtner, J.U. Multitrophic level models of predator-prey energetics: I. Age-specific energetics models—Pea aphid Acyrthosiphon pisum (Homoptera: Aphididae) as an example. Can. Entomol. 1984, 116, 924–932. [Google Scholar] [CrossRef]
- Gutierrez, A.P. The physiological basis of ratio-dependent predator-prey theory: The metabolic pool model as a paradigm. Ecology 1992, 73, 1552–63. [Google Scholar] [CrossRef]
- Regev, U.; Gutierrez, A.P.; Schreiber, S.J.; Zilberman, D. Biological and economic foundations of renewable resource exploitation. Ecol. Econ. 1998, 26, 227–242. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Regev, U. The bioeconomics of tritrophic systems: Applications to invasive species. Ecol. Econ. 2005, 52, 383–396. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Ponti, L. Assessing and managing the impact of climate change on invasive species: The PBDM approach. In Invasive Species and Global Climate Change; Ziska, L.H., Dukes, J.S., Eds.; CABI Publishing: Wallingford, UK, 2014; pp. 271–288. [Google Scholar]
- Pemsl, D.E.; Gutierrez, A.P.; Waibel, H. The economics of biotechnology under ecosystem disruption. Ecol. Econ. 2008, 66, 177–183. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Ponti, L.; Herren, H.R.; Baumgärtner, J.; Kenmore, P.E. Deconstructing Indian cotton: Weather, yields, and suicides. Environ. Sci. Eur. 2015, 27, 12. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Mills, N.J.; Schreiber, S.J.; Ellis, C.K. A physiologically based tritrophic perspective on bottom-up-top-down regulation of populations. Ecology 1994, 75, 2227–2242. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Gilioli, G.; Baumgärtner, J. Ecosocial consequences and policy implications of disease management in East African agropastoral systems. Proc. Natl. Acad. Sci. USA 2009, 106, 13136–13141. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.P.; Ponti, L. Bioeconomic sustainability of cellulosic biofuel production on marginal lands. Bull. Sci. Technol. Soc. 2009, 29, 213–225. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Ponti, L.; d’Oultremont, T.; Ellis, C.K. Climate change effects on poikilotherm tritrophic interactions. Clim. Chang. 2008, 87, S167–S192. [Google Scholar] [CrossRef]
- Ponti, L.; Gutierrez, A.P.; Basso, B.; Neteler, M.; Ruti, P.M.; Dell’Aquila, A.; Iannetta, M. Olive agroecosystems in the Mediterranean Basin: Multitrophic analysis of climate effects with process-based representation of soil water balance. Procedia Environ. Sci. 2013, 19, 122–131. [Google Scholar] [CrossRef]
- Neteler, M. Estimating daily Land Surface Temperatures in mountainous environments by reconstructed MODIS LST data. Remote Sens. 2010, 2, 333–351. [Google Scholar] [CrossRef]
- Metz, M.; Rocchini, D.; Neteler, M. Surface temperatures at the continental scale: Tracking changes with remote sensing at unprecedented detail. Remote Sens. 2014, 6, 3822–3840. [Google Scholar] [CrossRef]
- Artale, V.; Calmanti, S.; Carillo, A.; Dell’Aquila, A.; Hermann, M.; Pisacane, G.; Ruti, P.M.; Sannino, G.; Striglia, M.V.; Giorgi, F.; Bi, X.; Pal, J.S.; Rauscher, S. An atmosphere-ocean regional climate model for the Mediterranean area: Assessment of a present climate simulation. Clim. Dyn. 2010, 35, 721–740. [Google Scholar] [CrossRef]
- Dell’Aquila, A.; Calmanti, S.; Ruti, P.; Struglia, M.V.; Pisacane, G.; Carillo, A.; Sannino, G. Effects of seasonal cycle fluctuations in an A1B scenario over the Euro-Mediterranean region. Clim. Res. 2012, 52, 135–157. [Google Scholar] [CrossRef]
- Bivand, R. Using the R–Grass interface: Current status. OSGeo J. 2007, 1, 36–38. [Google Scholar]
- Monfreda, C.; Ramankutty, N.; Foley, J.A. Farming the planet: 2. Geographic distribution of crop areas, yields, physiological types, and net primary production in the year 2000. Glob. Biogeochem. Cycles 2008, 22, GB1022. [Google Scholar] [CrossRef]
- European Environment Agency (EEA). Corine Land Cover 2000 Raster Data. Available online: https://www.eea.europa.eu/ds_resolveuid/DAT-79-en (accessed on 20 June 2017).
- Tominski, C.; Fuchs, G.; Schumann, H. Task-driven color coding. In Proceedings of the 2008 12th International Conference Information Visualisation, London, UK, 9–11 July 2008; pp. 373–380. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO). AGRO-MAPS; FAO: Rome, Italy, 2012. [Google Scholar]
- Giorgi, F.; Bi, X. Updated regional precipitation and temperature changes for the 21st century from ensembles of recent AOGCM simulations. Geophys. Res. Lett. 2005, 32, L21715. [Google Scholar] [CrossRef]
- International Organisation of Vine and Wine (OIV). OIV Statistical Report on World Vitiviniculture; International Organisation of Vine and Wine: Paris, France, 2017. [Google Scholar]
- Bindi, M.; Fibbi, L.; Gozzini, B.; Orlandini, S.; Miglietta, F. Modelling the impact of future climate scenarios on yield and yield variability of grapevine. Clim. Res. 1996, 7, 213–224. [Google Scholar] [CrossRef]
- Fraga, H.; García de Cortázar Atauri, I.; Malheiro, A.C.; Santos, J.A. Modelling climate change impacts on viticultural yield, phenology and stress conditions in Europe. Glob. Change Biol. 2016, 22, 3774–3788. [Google Scholar] [CrossRef] [PubMed]
- Mozell, M.R.; Thach, L. The impact of climate change on the global wine industry: Challenges & solutions. Wine Econ. Policy 2014, 3, 81–89. [Google Scholar] [CrossRef]
- Moriondo, M.; Ferrise, R.; Trombi, G.; Brilli, L.; Dibari, C.; Bindi, M. Modelling olive trees and grapevines in a changing climate. Environ. Model. Softw. 2015, 72, 387–401. [Google Scholar] [CrossRef]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2009. [Google Scholar]
- Brewer, C.A. ColorBrewer: Color Advice for Maps. Available online: http://colorbrewer2.org/ (accessed on 19 June 2017).
- Ponti, L.; Gilioli, G.; Biondi, A.; Desneux, N.; Gutierrez, A.P. Physiologically based demographic models streamline identification and collection of data in evidence-based pest risk assessment. EPPO Bull. 2015, 45, 317–322. [Google Scholar] [CrossRef]
- Bournier, A. Grape insects. Annu. Rev. Entomol. 1977, 22, 355–376. [Google Scholar] [CrossRef]
- Flaherty, D.L.; Jensen, F.; Kasimatis, A.; Kido, H.; Moller, W. Grape Pest Management; Agricultural Sciences Publications, University of California: Berkeley, CA, USA, 1981. [Google Scholar]
- Ragusa, S.; Tsolakis, H. (Eds.) La Difesa Della Vite Dagli Artropodi Dannosi; Università degli Studi di Palermo: Palermo, Italy, 2006. [Google Scholar]
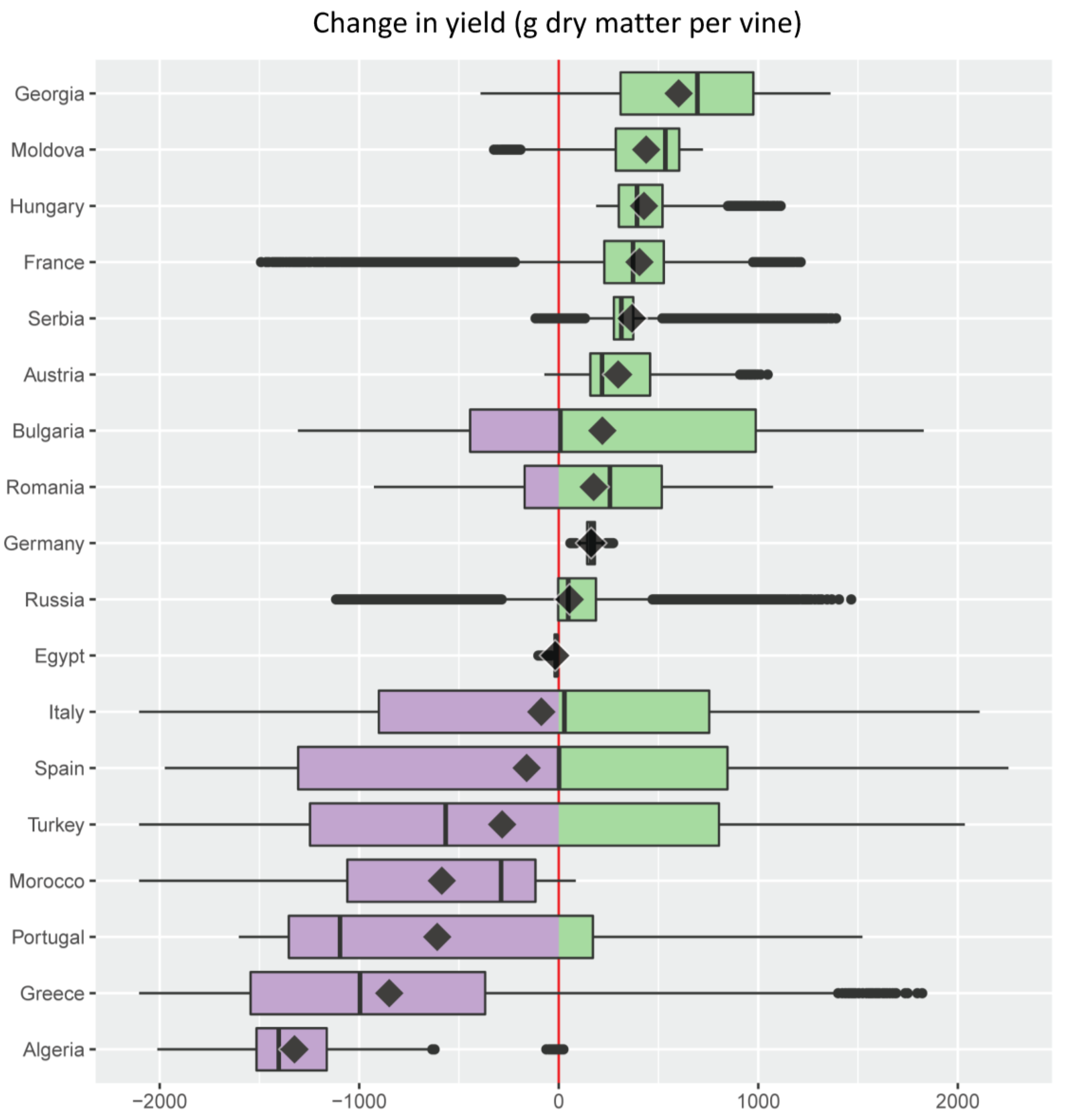
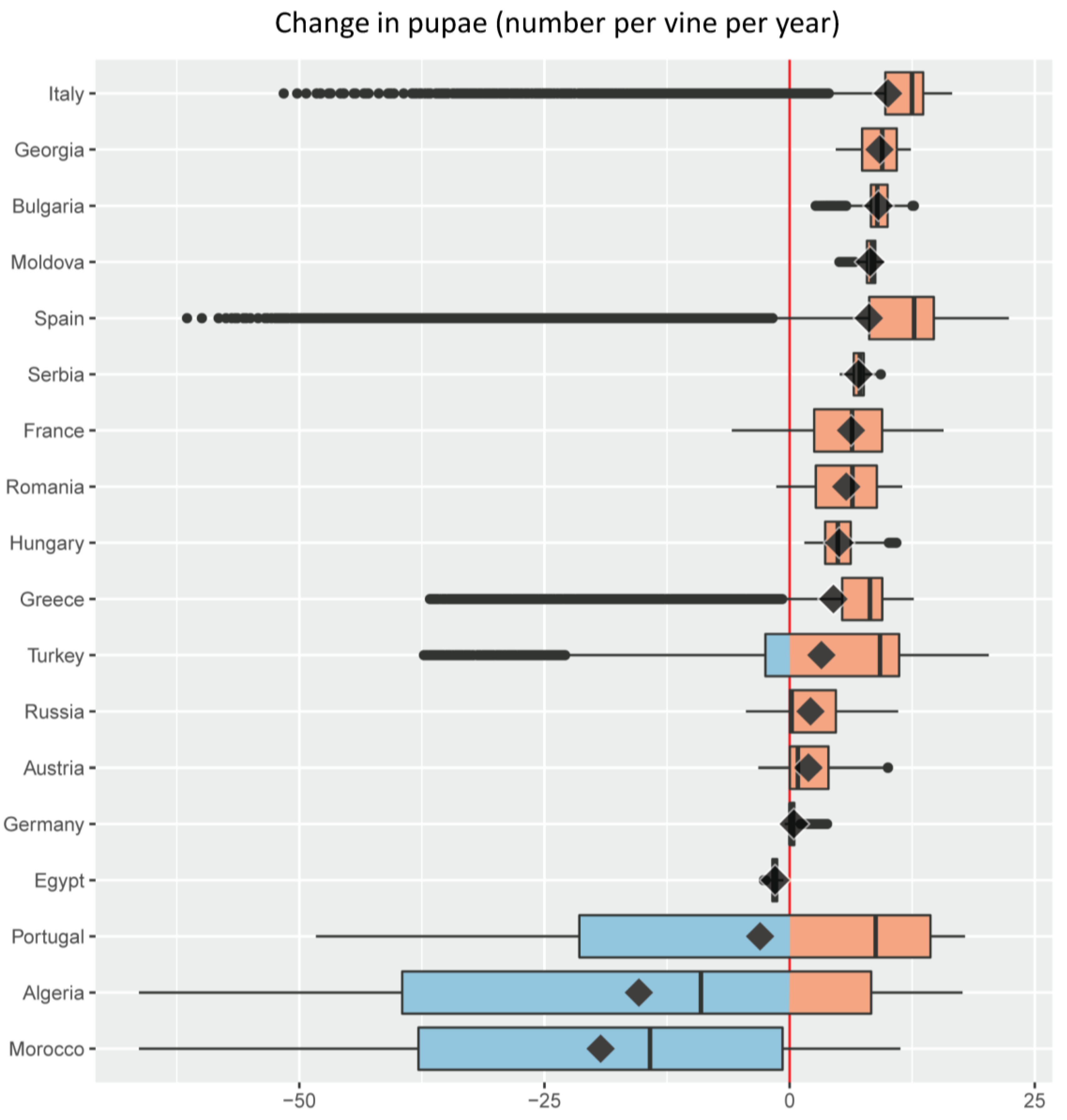
| Country | % Grape Growing Area | |
|---|---|---|
| Δ Yield < 0 | Δ Yield > 0 | |
| Germany | 0.0 | 100.0 |
| Hungary | 0.0 | 100.0 |
| Serbia | 0.5 | 99.5 |
| Austria | 2.1 | 97.9 |
| France | 2.5 | 97.5 |
| Moldova | 5.0 | 95.0 |
| Georgia | 12.0 | 88.0 |
| Romania | 26.7 | 73.3 |
| Russia | 27.3 | 72.7 |
| Italy | 49.3 | 50.7 |
| Bulgaria | 49.7 | 50.3 |
| Spain | 50.0 | 50.0 |
| Turkey | 65.2 | 34.8 |
| Portugal | 73.2 | 26.8 |
| Greece | 84.6 | 15.4 |
| Morocco | 96.9 | 3.1 |
| Algeria | 99.9 | 0.1 |
| Egypt | 100.0 | 0.0 |
| Country | % Grape Growing Area | |
|---|---|---|
| Δ Pupae < 0 | Δ Pupae > 0 | |
| Moldova | 0.0 | 100.0 |
| Hungary | 0.0 | 100.0 |
| Bulgaria | 0.0 | 100.0 |
| Serbia | 0.0 | 100.0 |
| Georgia | 0.0 | 100.0 |
| France | 3.7 | 96.3 |
| Russia | 3.8 | 96.2 |
| Romania | 4.4 | 95.6 |
| Italy | 8.0 | 92.0 |
| Spain | 11.5 | 88.5 |
| Germany | 13.0 | 87.0 |
| Greece | 16.0 | 84.0 |
| Austria | 21.0 | 79.0 |
| Turkey | 27.4 | 72.6 |
| Portugal | 41.2 | 58.8 |
| Algeria | 58.9 | 41.1 |
| Morocco | 81.3 | 18.7 |
| Egypt | 100.0 | 0.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ponti, L.; Gutierrez, A.P.; Boggia, A.; Neteler, M. Analysis of Grape Production in the Face of Climate Change. Climate 2018, 6, 20. https://doi.org/10.3390/cli6020020
Ponti L, Gutierrez AP, Boggia A, Neteler M. Analysis of Grape Production in the Face of Climate Change. Climate. 2018; 6(2):20. https://doi.org/10.3390/cli6020020
Chicago/Turabian StylePonti, Luigi, Andrew Paul Gutierrez, Antonio Boggia, and Markus Neteler. 2018. "Analysis of Grape Production in the Face of Climate Change" Climate 6, no. 2: 20. https://doi.org/10.3390/cli6020020
APA StylePonti, L., Gutierrez, A. P., Boggia, A., & Neteler, M. (2018). Analysis of Grape Production in the Face of Climate Change. Climate, 6(2), 20. https://doi.org/10.3390/cli6020020






