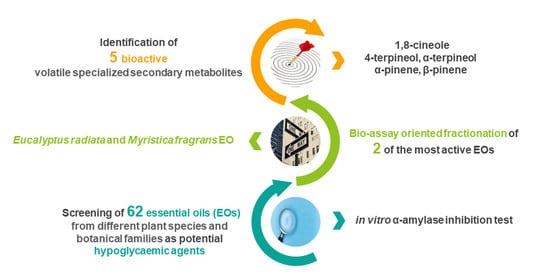
Bio-Guided Fractionation Driven by In Vitro α-Amylase Inhibition Assays of Essential Oils Bearing Specialized Metabolites with Potential Hypoglycemic Activity
Abstract
1. Introduction
2. Results and Discussion
2.1. Optimization of the In Vitro Enzymatic Test
2.2. In Vitro α-Amylase Inhibition Test
2.3. Essential Oil Composition
2.4. Bio-Guided Assay Fractionation of Eucalyptus radiata and Myristica fragrans
2.5. Data Precision
3. Materials and Methods
3.1. Reagents
3.2. In Vitro α-Amylase Inhibition Test
3.3. Maltose Calibration Curve
3.4. Flash Column Chromatography
3.5. Analysis Conditions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Diabetes Association. Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 2014, 37, S81–S90. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.J. New Approaches to the pharmacotherapy of diabetes. In Textbook of Diabetes; Pickup, J., William, G., Eds.; Blackwell Science Ltd.: Oxford, UK, 2003; pp. 73.1–73.21. [Google Scholar]
- Fujisawa, T.; Ikegami, H.; Inoue, K.; Kawabata, Y.; Ogihara, T. Effect of two α-glucosidase inhibitors, voglibose and acarbose, on postprandial hyperglycemia correlates with subjective abdominal symptoms. Metabolism 2005, 54, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Ríos, J.; Francini, F.; Schinella, G. Natural Products for the Treatment of Type 2 Diabetes Mellitus. Planta Med. 2015, 81, 975–994. [Google Scholar] [CrossRef] [PubMed]
- Bedekar, A.; Shah, K.; Koffas, M. Natural products for type II diabetes treatment. Adv. Appl. Microbiol. 2010, 71, 21–73. [Google Scholar] [PubMed]
- Nelson-Dooley, C.; Della-Fera, M.; Hamrick, M.; Baile, C. Novel Treatments for Obesity and Osteoporosis: Targeting Apoptotic Pathways in Adipocytes. Curr. Med. Chem. 2005, 12, 2215–2225. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Ueda, T.; Oki, T.; Sugita, K.; Terahara, N.; Matsumoto, K. α-glucosidase inhibitory action of natural acylated anthocyanins. 1. Survey of natural pigments with potent inhibitory activity. J. Agric. Food Chem. 2001, 49, 1948–1951. [Google Scholar] [CrossRef] [PubMed]
- Can Başer, K.H.; Buchbauer, G. Handbook of Essential Oils; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Goerg, K.J.; Spilker, T. Effect of peppermint oil and caraway oil on gastrointestinal motility in healthy volunteers: A pharmacodynamic study using simultaneous determination of gastric and gall-bladder emptying and orocaecal transit time. Aliment Pharmacol. Ther. 2003, 17, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Cappello, G.; Spezzaferro, M.; Grossi, L.; Manzoli, L.; Marzio, L. Peppermint oil (Mintoil®) in the treatment of irritable bowel syndrome: A prospective double blind placebo-controlled randomized trial. Dig. Liver Dis. 2007, 39, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Kenia, P.; Houghton, T.; Beardsmore, C. Does inhaling menthol affect nasal patency or cough? Pediatr. Pulmonol. 2008, 43, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Kehrl, W.; Sonnemann, U.; Dethlefsen, U. Therapy for Acute Nonpurulent Rhinosinusitis with Cineole: Results of a Double-Blind, Randomized, Placebo-Controlled Trial. Laryngoscope 2004, 114, 738–742. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, J.S. Method for Treating Gastrointestinal Disorders. US Patent USOO6420435B1, 16 July 2002. [Google Scholar]
- Kim, D.H.; Goh, H.J.; Lee, H.W.; Kim, K.S.; Kim, Y.T.; Moon, H.S.; Lee, S.W.; Park, S.Y. The effect of terpene combination on ureter calculus expulsion after extracorporeal shock wave lithotripsy. Korean J. Urol. 2014, 55, 36–40. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Romics, I.; Siller, G.; Kohnen, R.; Mavrogenis, S.; Varga, J.; Holman, E. A Special Terpene Combination (Rowatinex®) Improves Stone Clearance after Extracorporeal Shockwave Lithotripsy in Urolithiasis Patients: Results of a Placebo-Controlled Randomised Controlled Trial. Urol. Int. 2011, 86, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.K. Antidiabetic potential of plant natural products: A review. Int. J. Green Pharm. 2016, 10, S96–S113. [Google Scholar]
- Tan, X.C.; Chua, K.H.; Ram, M.R.; Kuppusamy, U.R. Monoterpenes: Novel insights into their biological effects and roles on glucose uptake and lipid metabolism in 3T3-L1 adipocytes. Food Chem. 2016, 196, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Jelenkovic, L.; Jovanovic, V.; Palic, I.; Mitic, V.; Radulovic, M. In Vitro Screening of α-Amylase Inhibition by Selected Terpenes from Essential Oils. Trop. J. Pharm. Res. 2014, 13, 1421. [Google Scholar] [CrossRef]
- Sahin Basak, S.; Candan, F. Chemical composition and In vitro antioxidant and antidiabetic activities of Eucalyptus Camaldulensis Dehnh. essential oil. J. Iran Chem. Soc. 2010, 7, 216–226. [Google Scholar] [CrossRef]
- Enzymatic Assay of Alpha-Amylase Sigma-Aldrich. Available online: https://www.sigmaaldrich.com/technical-documents/protocols/biology/enzymatic-assay-of-a-amylase.html (accessed on 20 August 2020).
- Bernfeld, P. Amylases, α and β. In Methods in Enzymology; Colowick, S.P., Kaplan, N., Eds.; Academic Press, Inc.: Cambridge, MA, USA; Harcourt Brace & Company: San Diego, CA, USA; New York, NY, USA; Boston, MA, USA; London, UK; Sydney, Australia; Tokyo, Japan; Toronto, ON, Canada, 1955; pp. 149–158. [Google Scholar]
- Rubiolo, P.; Sgorbini, B.; Liberto, E.; Cordero, C.; Bicchi, C. Essential oils and volatiles: Sample preparation and analysis. A review. Flavour. Fragr. J. 2010, 25, 282–290. [Google Scholar] [CrossRef]
- Bicchi, C.; Liberto, E.; Matteodo, M.; Sgorbini, B.; Mondello, L.; Zellner, B.; d’Acampora Costa, R.; Rubiolo, P. Quantitative analysis of essential oils: A complex task. Flavour. Fragr. J. 2008, 23, 382–391. [Google Scholar] [CrossRef]


| Species | Family | Common Name | Plant Part | % Inhibition Activity | Standard Deviation |
|---|---|---|---|---|---|
| Artemisia vulgaris L. | Compositae | Mugwort | Leaf/Flower | 48 | 3 |
| Cananga odorata (Lam.) Hook.f. and Thomson | Annonaceae | Ylang-ylang | Flower | no activity | - |
| Carum carvi L. | Apiaceae | Caraway | Seed | 17 | 2 |
| Cedrus atlantica (Endl.) Manetti ex Carrière | Pinaceae | Cedar wood | Wood | no activity | - |
| Chrysopogon zizanioides (L.) Roberty | Poaceae | Vetiver | Root | no activity | - |
| Cinnamomum zeylanicum Nees | Lauraceae | Cinnamon leaf | Leaf | no activity | - |
| Cinnamomum zeylanicum Nees | Lauraceae | Cinnamon bark | Bark | no activity | - |
| Cinnamomum camphora (L.) J.Presl | Lauraceae | Camphor | Wood | 20 | 2 |
| Cinnamomum cassia (L.) J.Presl | Lauraceae | Cinnamon bark | Bark | no activity | - |
| Citrus × aurantium L. | Rutaceae | Bitter orange | Fruit peel | 23 | 5 |
| Citrus × aurantium L. | Rutaceae | Neroli | Flower | 20 | 1 |
| Citrus × aurantium L. | Rutaceae | Petitgrain | Leaf | 4 | 1 |
| Citrus bergamia Risso et Poiteau | Rutaceae | Bergamot | Fruit peel | 16 | 3 |
| Citrus limon (L.) Osbeck | Rutaceae | Lemon | Fruit peel | 15 | 2 |
| Citrus medica L. | Rutaceae | Finger citron | Fruit peel | 14 | 4 |
| Citrus nobilis Lour. | Rutaceae | Mandarin | Fruit peel | no activity | - |
| Citrus paradisi Macfad. | Rutaceae | Grapefruit | Fruit peel | no activity | - |
| Citrus sinensis (L.) Osbeck | Rutaceae | Sweet orange | Fruit peel | 14 | 3 |
| Corymbia citriodora (Hook.) K.D.Hill and L.A.S.Johnson | Myrtaceae | Eucalyptus. lemon-scented | Leaf | 44 | 5 |
| Cupressus sempervirens L. | Cupressaceae | Cypress | Leaf/Twig | 17 | 1 |
| Cymbopogon martini (Roxb.) W.Watson. | Poaceae | Palmarosa | Leaf | no activity | - |
| Cymbopogon nardus (L.) Rendle | Poaceae | Citronella Ceylon | Leaf | 22 | 2 |
| Cymbopogon schoenanthus (L.) Spreng. | Poaceae | Lemongrass | Leaf | 7 | 2 |
| Elettaria cardamomum (L.) Maton | Zingiberaceae | Cardamom | Seed | 10 | 3 |
| Eucalyptus globulus Labill. | Myrtaceae | Eucalyptus | Leaf | 34 | 3 |
| Eucalyptus radiata A.Cunn. ex DC. | Myrtaceae | Leaf | 65 | 4 | |
| Foeniculum vulgare Mill. | Apiaceae | Fennel | Fruit | no activity | - |
| Gaultheria procumbens L. | Ericaceae | Wintergreen | Leaf | no activity | - |
| Hyssopus officinalis L. | Lamiaceae | Hyssop | Leaf | 18 | 1 |
| Jasminum officinale L. | Oleaceae | Jasmine | Flowers | no activity | - |
| Juniperus communis L. | Cupressaceae | Juniper berry | Fruit | 5 | 2 |
| Juniperus virginiana L. | Cupressaceae | Cedarwood | Wood | 15 | 2 |
| Laurus nobilis L. | Lauraceae | Laurel | Leaf | 50 | 4 |
| Lavandula angustifolia Mill. × L. latifolia Medik. | Lamiaceae | Lavandin | Leaf | 6 | 3 |
| Lavandula angustifolia Mill. | Lamiaceae | Lavender | Leaf | 5 | 2 |
| Matricaria chamomilla L. | Compositae | Chamomile | Flowers | 32 | 4 |
| Melaleuca alternifolia (Maiden and Betche) Cheel | Myrtaceae | Tea tree | Leaf | 14 | 2 |
| Melaleuca viridiflora Sol. ex Gaertn. | Myrtaceae | Niaouli | Leaf | 28 | 4 |
| Melissa officinalis L. | Lamiaceae | Lemon balm | Leaf | no activity | - |
| Mentha × piperita L. | Lamiaceae | Peppermint | Leaf | 33 | 3 |
| Mentha × piperita L. | Lamiaceae | Peppermint | Leaf/twig | 40 | 5 |
| Mentha arvensis L. | Lamiaceae | Mint | Leaf | 39 | 6 |
| Myristica fragrans Houtt. | Myristicaceae | Nutmeg | Seed | 59 | 4 |
| Myrtus communis L. | Myrtaceae | Myrtle | Leaf | 20 | 7 |
| Ocimum basilicum L. | Lamiaceae | Basil | Leaf | 14 | 5 |
| Origanum majorana L. | Lamiaceae | Marjoram | Leaf | 10 | 1 |
| Origanum vulgare L. | Lamiaceae | Oregano | Leaf | 9 | 4 |
| Pelargonium graveolens L’Hér. | Geraniaceae | Geranium | Leaf | 9 | 3 |
| Pimpinella anisum L. | Apiaceae | Aniseed | Fruit | 27 | 5 |
| Pinus mugo Turra | Pinaceae | Pine needle | Leaf/Twig | no activity | - |
| Pinus sylvestris L. | Pinaceae | Pine sylvestris | Leaf/Twig | no activity | - |
| Piper nigrum L. | Piperaceae | Black pepper | Fruit | no activity | - |
| Pogostemon cablin (Blanco) Benth. | Lamiaceae | Patchouli | Leaf | no activity | - |
| Rosa × damascena Herrm. | Rosaceae | Rosa | Flower | no activity | - |
| Salvia officinalis L. | Lamiaceae | Sage. Dalmatian | Leaf | 3 | 2 |
| Salvia sclarea L. | Lamiaceae | Clary sage | Leaf/Flower | no activity | - |
| Santalum album L. | Santalaceae | Sandalwood | Wood | no activity | - |
| Syzygium aromaticum (L.) Merr. and L.M.Perry | Myrtaceae | Clove oil | Leaf/Buds | no activity | - |
| Thuja occidentalis L. | Cupressaceae | Cedar leaf | Leaf | 7 | 5 |
| Thymus vulgaris L. | Lamiaceae | Thyme | Leaf | no activity | - |
| Verbena officinalis L. | Verbenaceae | Vervain | Leaf | no activity | - |
| Zingiber officinale Roscoe | Zingiberaceae | Ginger | Rhizome | no activity | - |
| % Inhibition Approach A | IC50 1 (mg mL−1) | % Inhibition Approach B | IC50 1 (mg mL−1) | % CV 2 | |
|---|---|---|---|---|---|
| Eucalyptus radiata | 65 ± 3 | 1.53 ± 0.08 | 65 ± 4 | 1.54 ± 0.11 | 0.080 |
| Myristica fragrans | 59 ± 4 | 1.70 ± 0.13 | 58 ± 5 | 1.71 ± 0.17 | 0.780 |
| Laurus nobilis | 51 ± 8 | 1.98 ± 0.32 | 51 ± 5 | 1.98 ± 0.19 | 0.120 |
| Acarbose | 56 ± 6 | 1.80 ± 0.20 | 55 ± 6 | 1.80 ± 0.22 | 0.020 |
| Species | Hydrocarbon Compounds | Oxygenated Compounds | List of the Most Abundant Components |
|---|---|---|---|
| Artemisia vulgaris L | 9.0 | 91.0 | α-Thujone (47.4), Camphor (30.0), β-Thujone (7.80), Sabinene (3.90), Camphene (3.70) |
| Carum carvi L. | 37.8 | 62.2 | Carvone (59.6), Limonene (35.4), β-Myrcene (0.700), cis-Dihydroxy carvone (0.600), trans-Dihydroxy carvone (0.200) |
| Cinnamomum camphora (L.) J.Presl | 54.8 | 45.2 | 1,8-Cineole (44.1), Limonene (17.4), p-Cymene (14.6), α-Terpinene (9.60), β-Pinene (7.60) |
| Citrus × aurantium L. (neroli) | 97.5 | 2.5 | Linalyl acetate (41.4), Linalool (28.5), Limonene (11.4), β-Pinene (7.60), trans-β-Ocimene (2.60) |
| Citrus × aurantium L. | 23.9 | 76.1 | Limonene (90.2), β -Myrcene (3.70), Linalyl acetate (1.60), α-Pinene (0.900), Sabinene (0.500) |
| Citrus × aurantium L. (Petit grain) | 7.4 | 92.6 | Linalyl acetate (56.8), Linalool (24.4), α-Terpineol (5.60), Geranyl acetate (3.40), Neryl acetate (1.80) |
| Citrus bergamia Risso et Poiteau | 52.0 | 48.0 | Linalyl acetate (34.1), Limonene (32.3), γ-Terpinene (7.80), β-Pinene (6.60), α-Pinene (1.00) |
| Citrus limon (L.) Osbeck | 97.2 | 2.8 | Limonene (71.9), β-Pinene (11.6), γ -Terpinene (7.90), α-Pinene (1.50), β-Myrcene (1.50) |
| Citrus medica L. | 72.5 | 27.5 | Limonene (54.9), Linalyl acetate (14.5), β-Pinene (9.10), Linalool (4.60), Geranial (4.60) |
| Citrus nobilis Lour. | 99.9 | 0.1 | Limonene (75.6), γ -Terpinene (14.5), α-Pinene (1.90), β-Pinene (1.10), β -Myrcene (1.00) |
| Corymbia citriodora (Hook.) K.D.Hill and L.A.S.Johnson | 1.8 | 98.2 | Citronellal (81.0), Neoisopulegol (7.10), Citronellol (6.00), Citronellyl acetate (1.50), 1,8-Cineole (0.800) |
| Cupressus sempervirens L. | 91.9 | 8.1 | α-Pinene (46.7), Δ-3-Carene (25.3), α-Terpinolene (4.20), Limonene (4.00), α-Terpinyl acetate (3.30) |
| Cymbopogon nardus (L.) Rendle | 11.6 | 88.4 | Citronellal (37.9), Geraniol (19.5), Citronellol (12.5), Limonene (7.20), Geranyl acetate (4.40) |
| Cymbopogon schoenanthus (L.) Spreng. | 7.0 | 93.0 | Geranial (38.2), Neral (32.5), Geraniol (7.30), Geranyl acetate (4.20), trans-β-Caryophyllene (2.90) |
| Elettaria cardamomum (L.) Maton | 9.2 | 90.8 | α-Terpinyl acetate (43.8), 1,8-Cineole (34.7), Linalyl acetate (6.00), Linalool (2.70), Limonene (2.20) |
| Eucalyptus globulus Labill. | 16.9 | 83.1 | 1,8-Cineole (82.1), Limonene (6.80), γ-Terpinene (3.30), p-Cymene (3.10), α-Pinene (2.20) |
| Eucalyptus radiata A.Cunn. ex DC. | 14.6 | 84.9 | 1,8-Cineole (75.1), α-Terpineol (7.6), Limonene (4.3) α-Terpinene (4.4) α-Pinene (2.7) |
| Hyssopus officinalis L. | 34.8 | 65.2 | 1,8-Cineole (39.2), α-Pinene (7.10), Isopinocamphone (6.10), Sabinene (5.80), β-Pinene (5.60) |
| Juniperus communis L. | 95.4 | 4.6 | α-Pinene (35.9), β -Myrcene (14.2), Sabinene (8.4) Limonene (8.00), β-Pinene (5.40) |
| Juniperus virginiana L. | 99.7 | 0.3 | β -Himachalene (50.8), α -Himalachene (16.0), γ-Himalachene (10.0), δ-Cadinene (2.50), α -Chamigrene (2.00) |
| Laurus nobilis L. | 20.5 | 79.5 | 1,8-Cineole (65.4), α-Terpinyl acetate (8.10), α-Pinene (6.4), Sabinene (5.10), β-Pinene (3.80) |
| Lavandula angustifolia Mill. × L. latifolia Medik. | 8.9 | 91.1 | Linalyl acetate (35.6), Linalool (27.1), Camphor (9.40), 1,8-Cineole (7.60), Borneol (3.40) |
| Lavandula angustifolia Mill. | 13.8 | 86.2 | Linalyl acetate (34.7), Linalool (27.9), trans-β-Caryophyllene (4.20), 4-Terpineol (3.70), Lavandulyl acetate (3.70) |
| Litsea cubeba (Lour.) Pers. | 17.3 | 82.7 | Geranial (42.4), Neral (34.6), Limonene (12.6), Sabinene (1.90), α-Pinene (1.20) |
| Matricaria chamomilla L. | 36.0 | 64.0 | α-Bisabolol oxide A (47.0), trans-β-Farnesene (24.0), α-Bisabolol oxide B (6.4), Chamazulene (2.60), Germacrene D (1.60) |
| Melaleuca alternifolia (Maiden and Betche) Cheel | 45.9 | 54.1 | 4-Terpineol (44.1), γ-Terpinene (21.1), α-Terpinene (9.40), α-Terpinolene (3.20), 1,8-Cineole (3.20) |
| Melaleuca viridiflora Sol. ex Gaertn. | 23.4 | 76.6 | 1,8-Cineole (64.9), Limonene (9.50), α-Pinene (6.20), α-Terpineol (4.20), Viridiflorol (2.40) |
| Mentha × piperita L. (leaf) | 1.1 | 98.9 | Menthol (48.2), Menthone (24.9), Isomenthone (13.3), Menthyl acetate (6.40), Neomenthol (2.00) |
| Mentha × piperita L. (Leaf/Twig) | 0.9 | 99.1 | Menthol (52.0), Menthone (23.0), Isomenthone (10.0), Menthyl acetate (4.60), Neomenthol (4.26) |
| Mentha arvensis L. | 6.6 | 93.4 | Menthol (40.2), Menthone (19.5), Isomenthone (8.00), Menthyl acetate (7.40), Neomenthol (5.20) |
| Myristica fragrans Houtt. | 85.2 | 14.8 | Sabinene (27.0), α-Pinene (23.0), β-Pinene (13.0), Limonene (10.0), 4-Terpineol (6.70) |
| Myrtus communis L. | 56.1 | 43.9 | Limonene (28.9), α-Pinene (15.1), Mirtenyl acetate (13.6), Linalool (13.50), Linalyl acetate (5.00) |
| Ocimum basilicum L | 5.1 | 94.9 | Estragole (88.4), 1,8-Cineole (3.40), α-trans-Bergamotene (2.30), trans-β-Ocimene (1.00), Linalool (0.600) |
| Origanum majorana L. | 41.3 | 58.7 | Linalool (34.0), 4-Terpineol (17.7), γ-Terpinene (10.8), α-Terpinene (7.00), Sabinene (5.80) |
| Origanum vulgare L. | 28.8 | 71.2 | Carvacrol (67.4), p-Cymene (12.2), γ-Terpinene (5.10), trans-β-Caryophyllene (4.70), Linalool (1.90) |
| Pelargonium graveolens L’Hér. | 5.1 | 94.9 | Citronellol (34.6), Geraniol (18.5), Citronellyl formate (9.50), Linalool (6.70), Isomenthone (5.10) |
| Pimpinella anisum L. | 3.7 | 96.3 | trans-Anethol (92.2), Limonene (2.1), Estragole (1.70), Foeniculin (.0700), Linalool (0.400) |
| Rosmarinus officinalis L. | 35.1 | 64.9 | 1,8-Cineole (43.3), Camphor (18.1), α-Pinene (12.8), β-Pinene (9.50), trans-β-Caryophyllene (5.90) |
| Salvia officinalis L. | 32.2 | 67.8 | α-Thujone (22.5), Camphor (18.5), 1,8-Cineole (11.4), α-Humulene (7.20), β-Thujone (6.20), |
| Syzygium aromaticum (L.) Merr. and L.M.Perry | 10.2 | 89.8 | Eugenol (82.0), trans-β-Caryophyllene (9.10), Eugenyl acetate (7.10), α-Humulene (1.10), Caryophyllene oxide (0.300) |
| # | Compounds | Eucalyptus radiata | Myristica fragrans | ||||
|---|---|---|---|---|---|---|---|
| Total EO | Hydrocarbon Fraction | Oxygenated Fraction | Total EO | Hydrocarbon Fraction | Oxygenated Fraction | ||
| 1 | α-Thujene | 0.2 | 0.3 | / | / | 0.7 | / |
| 2 | α-Pinene | 2.7 | 6.2 | / | 23.0 | 21.8 | / |
| 3 | Sabinene | 1.0 | 5.7 | / | 27.0 | 26.3 | / |
| 4 | β-Pinene | 0.6 | 4.2 | / | 13.0 | 20.4 | / |
| 5 | β-Mircene | 0.4 | 5.8 | / | 1.1 | 1.7 | / |
| 6 | α-Phellandrene | / | / | / | 1.4 | 1.6 | / |
| 7 | Δ-3-Carene | / | / | / | 0,4 | 0.6 | / |
| 8 | α-Terpinene | 4.4 | / | / | 0.9 | 1.2 | / |
| 9 | p-Cimene | 0.6 | 11.3 | / | 0.7 | 2.1 | / |
| 10 | Limonene | 4.3 | 65.4 | / | 10.0 | 15.3 | / |
| 11 | 1,8-Cineole | 75.1 | / | 69.8 | 1.8 | / | 5.0 |
| 12 | trans-β-Ocimene | tr | 0.2 | / | 0.05 | 0.1 | / |
| 13 | γ-Terpinene | tr | 0.1 | / | 4.9 | 6.6 | / |
| 14 | α-Terpinolene | tr | 0.1 | / | 0.6 | 1.1 | / |
| 15 | cis-Sabinene Hydrate | / | / | / | / | / | 0.7 |
| 16 | trans-Sabinene Hydrate | / | / | / | / | / | 0.5 |
| 17 | Linalool | 0.3 | / | 0.7 | 0,2 | / | 1.6 |
| 18 | Linalyl propionate | 0.2 | / | 0.4 | / | / | / |
| 19 | 4-Terpineol | 1.0 | / | 1.7 | 6.7 | / | 45.4 |
| 20 | α-Terpineol | 7.6 | / | 16.0 | 0.2 | / | 1.7 |
| 21 | Eugenol | / | / | / | 0.5 | / | 4.5 |
| 22 | Safrole | / | / | / | 0.7 | / | 1.1 |
| 23 | Myristicin | / | / | / | 4.4 | / | 37.3 |
| 24 | Neral | 0.3 | / | 1.2 | / | / | / |
| 25 | Geranial | 0.4 | / | 1.6 | / | / | / |
| 26 | α-Terpinyl acetate | 0.3 | / | 5.0 | / | / | 1.8 |
| 27 | trans-β-Caryophyllene | / | 0.6 | / | / | 0.2 | / |
| Repeatability (n = 3) | Intermediate Precision | ||||
|---|---|---|---|---|---|
| % Inhibition | % RSD | % Inhibition * | % RSD | ||
| Acarbose | 57 | 2 | Acarbose | 59 | 5 |
| 55 | 57 | ||||
| 56 | 53 | ||||
| 57 | |||||
| 55 | |||||
| 56 | |||||
| Laurel | 54 | 8 | Laurel | 54 | 12 |
| 51 | 53 | ||||
| 46 | 52 | ||||
| 56 | |||||
| 44 | |||||
| 42 | |||||
| Nutmeg | 56 | 4 | Nutmeg | 54 | 9 |
| 61 | 63 | ||||
| 60 | 60 | ||||
| 50 | |||||
| 65 | |||||
| 60 | |||||
| Eucalyptus | 66 | 4 | Eucalyptus | 54 | 9 |
| 68 | 70 | ||||
| 62 | 60 | ||||
| 62 | |||||
| 67 | |||||
| 69 | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capetti, F.; Cagliero, C.; Marengo, A.; Bicchi, C.; Rubiolo, P.; Sgorbini, B. Bio-Guided Fractionation Driven by In Vitro α-Amylase Inhibition Assays of Essential Oils Bearing Specialized Metabolites with Potential Hypoglycemic Activity. Plants 2020, 9, 1242. https://doi.org/10.3390/plants9091242
Capetti F, Cagliero C, Marengo A, Bicchi C, Rubiolo P, Sgorbini B. Bio-Guided Fractionation Driven by In Vitro α-Amylase Inhibition Assays of Essential Oils Bearing Specialized Metabolites with Potential Hypoglycemic Activity. Plants. 2020; 9(9):1242. https://doi.org/10.3390/plants9091242
Chicago/Turabian StyleCapetti, Francesca, Cecilia Cagliero, Arianna Marengo, Carlo Bicchi, Patrizia Rubiolo, and Barbara Sgorbini. 2020. "Bio-Guided Fractionation Driven by In Vitro α-Amylase Inhibition Assays of Essential Oils Bearing Specialized Metabolites with Potential Hypoglycemic Activity" Plants 9, no. 9: 1242. https://doi.org/10.3390/plants9091242
APA StyleCapetti, F., Cagliero, C., Marengo, A., Bicchi, C., Rubiolo, P., & Sgorbini, B. (2020). Bio-Guided Fractionation Driven by In Vitro α-Amylase Inhibition Assays of Essential Oils Bearing Specialized Metabolites with Potential Hypoglycemic Activity. Plants, 9(9), 1242. https://doi.org/10.3390/plants9091242