Enhanced UV-Reflection Facilitated a Shift in the Pollination System of the Red Poppy, Papaver rhoeas (Papaveraceae)
Abstract
1. Introduction
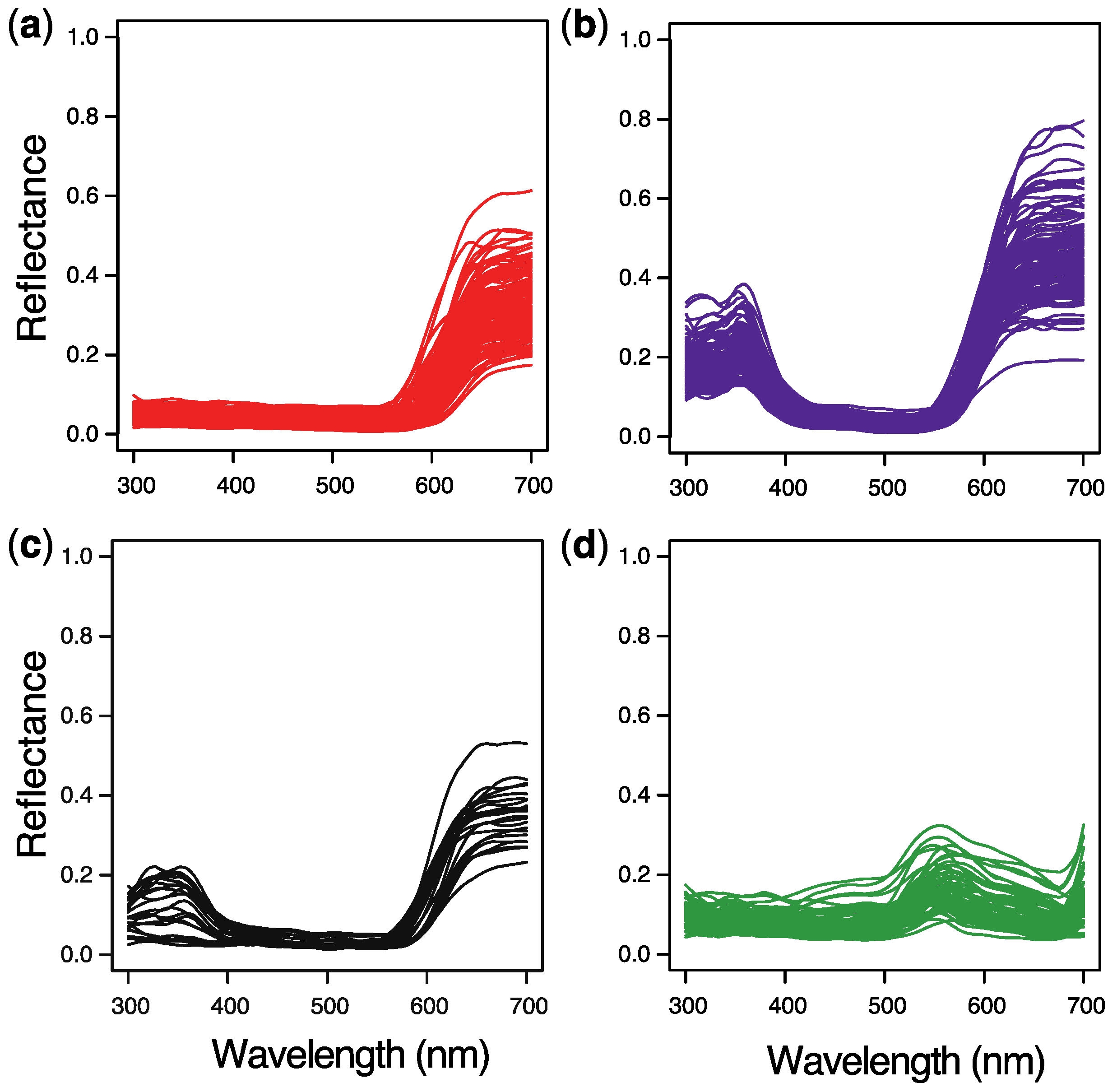
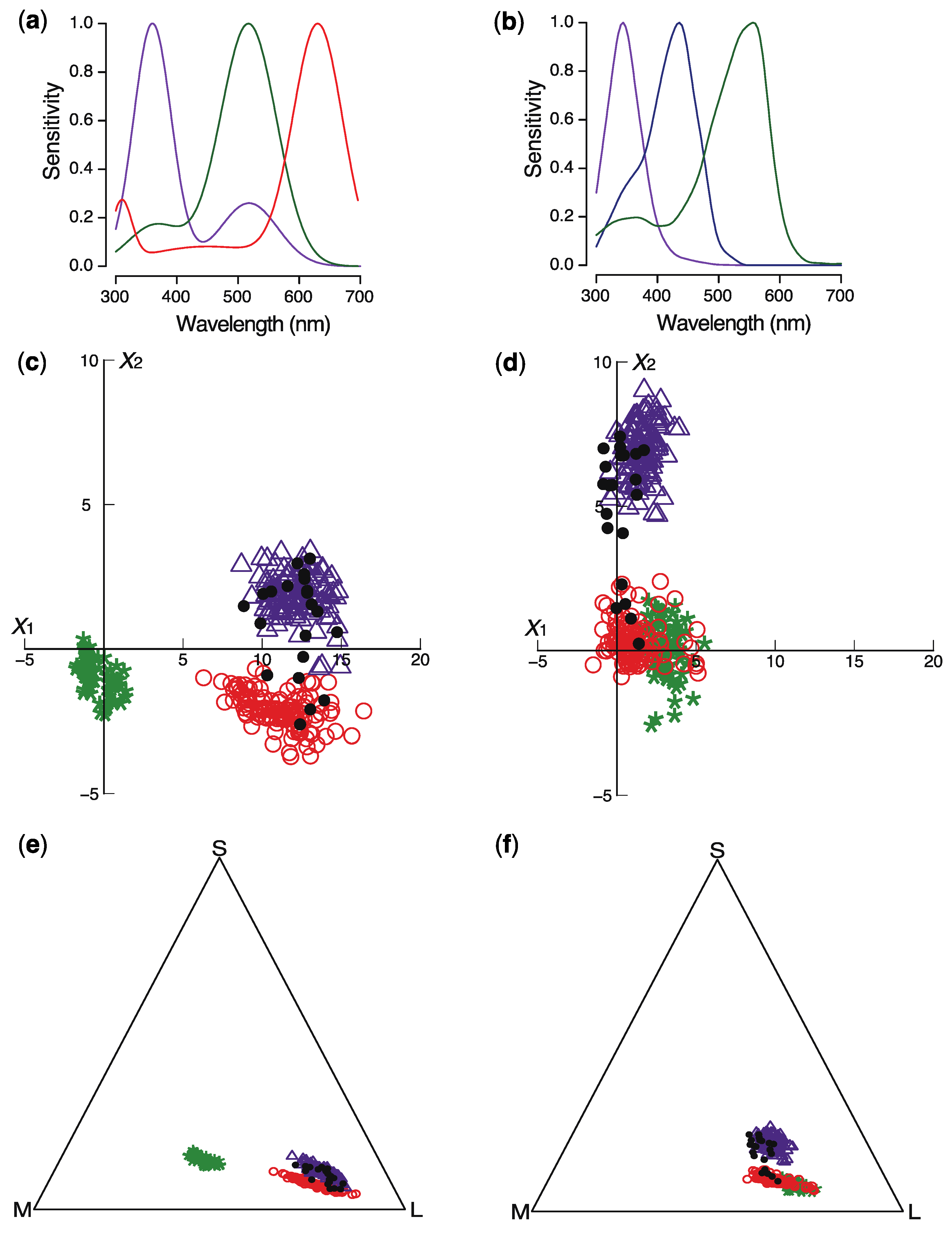
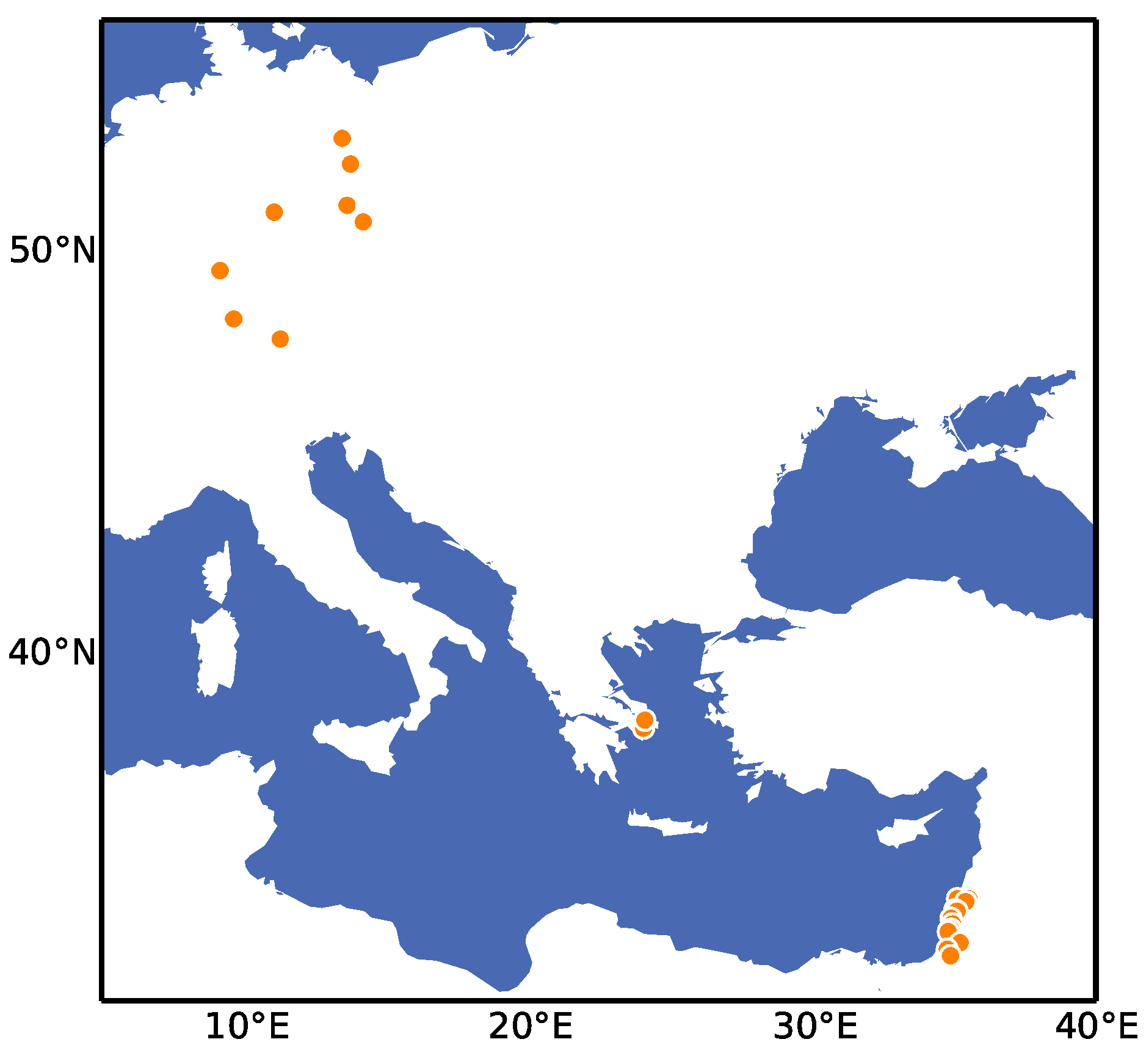
2. Results
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Photography
4.3. Spectral Reflectance Measurements
4.4. Modeling Insect Color Perception
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Parker, I.M. Pollinator limitation of Cytisus scoparius (Scotch broom), an invasive exotic shrub. Ecology 1997, 78, 1457–1470. [Google Scholar] [CrossRef]
- Larson, K.C.; Fowler, S.P.; Walker, J.C. Lack of pollinators limits fruit set in the exotic Lonicera japonica. Am. Midl. Nat. 2002, 148, 54–60. [Google Scholar] [CrossRef]
- Baker, H.G. The evolution of weeds. Annu. Rev. Ecol. Syst. 1974, 5, 1–24. [Google Scholar] [CrossRef]
- Lloyd, D.G. Self- and cross-fertilization in plants. II. The selection of self- fertilization. Int. J. Plant. Sci. 1992, 153, 370–380. [Google Scholar] [CrossRef]
- Morgan, M.T.; Wilson, W.G. Self-fertilization and the escape from pollen limitation in variable pollination environments. Evolution 2005, 59, 1143–1148. [Google Scholar] [CrossRef]
- Eckert, C.G.; Kalisz, S.; Geber, M.A.; Sargent, R.; Elle, E.; Cheptou, P.O.; Goodwillie, C.; Johnston, M.O.; Kelly, J.K.; Moeller, D.A.; et al. Plant mating systems in a changing world. Trends Ecol. Evol. 2010, 25, 35–43. [Google Scholar] [CrossRef]
- Gabra, S. Papaver species and opium through the ages. Bull. Inst. Egypte 1954, 37, 39–56. [Google Scholar]
- Kadereit, J.W. Some suggestions on the geographical origin of the central, west and north European synanthropic species of Papaver L. Bot. J. Linn. Soc. 1990, 103, 221–231. [Google Scholar] [CrossRef]
- Colledge, S.; Conolly, J.; Shennan, S. Archaeobotanical Evidence for the Spread of Farming in the Eastern Mediterranean. Curr. Anthropol. 2004, 45, S35–S58. [Google Scholar] [CrossRef]
- Willerding, U. Zur Geschichte der Unkräuter Mitteleuropas; Wachholtz: Neumünster, Germany, 1986. [Google Scholar]
- Zohary, D.; Hopf, M. Domestication of Plants in the Old World: The Origin and Spread of Cultivated Plants in West Asia, Europe, and the Nile Valley, 2nd ed.; Clarendon Press: Oxford, UK, 1993. [Google Scholar]
- Godwin, H. The History of the British Flora: A Factual Basis for Phytogeography, 2nd ed.; Cambridge University Press: Cambridge, UK, 1975. [Google Scholar]
- McNaughton, I.H.; Harper, J.L. The comparative biology of closely related species living in the same area I. External breeding barriers between Papaver species. New Phytol. 1960, 59, 15–26. [Google Scholar] [CrossRef]
- Dafni, A.; Bernhardt, P.; Shmida, A.; Ivri, Y.; Greenbaum, S. Red bowl-shaped flowers: Convergence for beetle pollination in the Mediterranean region. Isr. J. Bot. 1990, 39, 81–92. [Google Scholar]
- Dobson, H.E.M.; Groth, I.; Bergström, G. Pollen advertisement: Chemical contrasts between whole-flower and pollen odors. Am. J. Bot. 1996, 83, 877–885. [Google Scholar] [CrossRef]
- Proctor, M.; Yeo, P. The Pollination of Flowers; Collins: London, UK, 1973. [Google Scholar]
- Lawrence, M.J. Genetics of self-incompatibility in Papaver rhoeas. Proc. R. Soc. B 1975, 188, 275–285. [Google Scholar] [CrossRef]
- Foote, H.C.; Ride, J.P.; Franklin-Tong, V.E.; Walker, E.A.; Lawrence, M.J.; Franklin, F.C. Cloning and expression of a distinctive class of self-incompatibility (S) gene from Papaver rhoeas L. Proc. Natl. Acad. Sci. USA 1994, 91, 2265. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, M.J.; de Graaf, B.H.J.; Hadjiosif, N.; Perry, R.M.; Poulter, N.S.; Osman, K.; Vatovec, S.; Harper, A.; Franklin, F.C.H.; Franklin-Tong, V.E. Identification of the pollen self-incompatibility determinant in Papaver rhoeas. Nature 2009, 459, 992. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Harms, J.; Vorobyev, M.; Schorn, J.; Shmida, A.; Keasar, T.; Homberg, U.; Schmeling, F.; Menzel, R. Evidence of red sensitive photoreceptors in Pygopleurus israelitus (Glaphyridae: Coleoptera) and its implications for beetle pollination in the southeast Mediterranean. J. Comp. Physiol. A 2012, 198, 451–463. [Google Scholar] [CrossRef]
- Peitsch, D.; Fietz, A.; Hertel, H.; Desouza, J.; Ventura, D.F.; Menzel, R. The spectral input systems of hymenopteran insects and their receptor-based color vision. J. Comp. Physiol. A 1992, 170, 23–40. [Google Scholar] [CrossRef]
- Martínez-Harms, J.; Palacios, A.G.; Márquez, N.; Estay, P.; Arroyo, M.T.K.; Mpodozis, J. Can red flowers be conspicuous to bees? Bombus dahlbomii and South American temperate forest flowers as a case in point. J. Exp. Biol. 2010, 213, 564. [Google Scholar] [CrossRef]
- Streinzer, M.; Roth, N.; Paulus, H.F.; Spaethe, J. Color preference and spatial distribution of glaphyrid beetles suggest a key role in the maintenance of the color polymorphism in the peacock anemone (Anemone pavonina, Ranunculaceae) in Northern Greece. J. Comp. Physiol. A 2019, 205, 735–743. [Google Scholar] [CrossRef]
- Lotmar, R. Neue Untersuchungen über den Farbensinn der Bienen, mit besonderer Berücksichtigung des Ultravioletts. Z. Vergl. Physiol. 1933, 19, 673–723. [Google Scholar] [CrossRef]
- Kugler, H. Hummeln und die UV-Reflexion an Kronblättern. Naturwissenschaften 1947, 34, 315–316. [Google Scholar] [CrossRef]
- Daumer, K. Blumenfarben, wie sie die Bienen sehen. Z. Vergl. Physiol. 1958, 41, 49–110. [Google Scholar] [CrossRef]
- Lunau, K. Interspecific diversity and uniformity of flower colour patterns as cues for learned discrimination and innate detection of flowers. Experientia 1993, 49, 1002–1010. [Google Scholar] [CrossRef]
- Van der Kooi, C.J.; Stavenga, D.G. Vividly coloured poppy flowers due to dense pigmentation and strong scattering in thin petals. J. Comp. Physiol. A 2019, 205, 363–372. [Google Scholar] [CrossRef]
- Chittka, L.; Waser, N.M. Why red flowers are not invisible to bees. Isr. J. Plant. Sci. 1997, 45, 169–183. [Google Scholar] [CrossRef]
- Sakai, A.K.; Allendorf, F.W.; Holt, J.S.; Lodge, D.M.; Molofsky, J.; With, K.A.; Baughman, S.; Cabin, R.J.; Cohen, J.E.; Ellstrand, N.C.; et al. The population biology of invasive species. Annu. Rev. Ecol. Syst. 2001, 32, 305–332. [Google Scholar] [CrossRef]
- Stockwell, C.A.; Hendry, A.P.; Kinnison, M.T. Contemporary evolution meets conservation biology. Trends Ecol. Evol. 2003, 18, 94–101. [Google Scholar] [CrossRef]
- Lambrinos, J.G. How interactions between ecology and evolution influence contemporary invasion dynamics. Ecology 2004, 85, 2061–2070. [Google Scholar] [CrossRef]
- Barrett, S.C.H.; Colautti, R.I.; Eckert, C.G. Plant reproductive systems and evolution during biological invasion. Mol. Ecol. 2008, 17, 373–383. [Google Scholar] [CrossRef]
- Richardson, D.M.; Allsopp, N.; D’Antonio, C.M.; Milton, S.J.; Rejmanek, M. Plant invasions—The role of mutualisms. Biol. Rev. 2000, 75, 65–93. [Google Scholar] [CrossRef]
- Dudek, B.; Schneider, B.; Hartmut, H.H.; Stavenga, D.G.; Martínez-Harms, J. Highly different flavonol content explains geographic variations in the UV reflecting properties of flowers of the corn poppy, Papaver rhoeas (Papaveraceae). Phytochemistry 2020. [Google Scholar] [CrossRef]
- Haribal, M.M.; Feeny, P.P. Combined roles of contact stimulant and deterrents in assessment of host-plant quality by ovipositing zebra swallowtail butterflies. J. Chem. Ecol. 2003, 29, 653–670. [Google Scholar] [CrossRef] [PubMed]
- Mallikarjuna, N.; Kranthi, K.R.; Jadhav, D.R.; Kranthi, S.; Chandra, S. Influence of foliar chemical compounds on the development of Spodoptera litura (Fab.) in interspecific derivatives of groundnut. J. Appl. Entomol. 2004, 128, 321–328. [Google Scholar] [CrossRef]
- Sosa, T.; Chaves, N.; Alias, J.C.; Escudero, J.C.; Henao, F.; Gutiérrez-Merino, C. Inhibition of mouth skeletal muscle relaxation by flavonoids of Cistus ladanifer L.: A plant defense mechanism against herbivores. J. Chem. Ecol. 2004, 30, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Hollósy, F. Effects of ultraviolet radiation on plant cells. Micron 2002, 33, 179–197. [Google Scholar] [CrossRef]
- Rozema, J.; van de Staaij, J.; Björn, L.O.; Caldwell, M. UV-B as an environmental factor in plant life: Stress and regulation. Trends Ecol. Evol. 1997, 12, 22–28. [Google Scholar] [CrossRef]
- Shirley, B.W. Flavonoid biosynthesis: ‘new’ functions for an ‘old’ pathway. Trends Plant. Sci. 1996, 1, 377–382. [Google Scholar] [CrossRef]
- Havaux, M.; Kloppstech, K. The protective functions of carotenoid and flavonoid pigments against excess visible radiation at chilling temperature investigated in Arabidopsis npq and tt mutants. Planta 2001, 213, 953–966. [Google Scholar] [CrossRef]
- Albert, A.; Sareedenchai, V.; Heller, W.; Seidlitz, H.K.; Zidorn, C. Temperature is the key to altitudinal variation of phenolics in Arnica montana L. cv. ARBO. Oecologia 2009, 160, 1–8. [Google Scholar] [CrossRef]
- Jansen, M.A.K.; Gaba, V.; Greenberg, B.M. Higher plants and UV-B radiation: Balancing damage, repair and acclimation. Trends Plant. Sci. 1998, 3, 131–135. [Google Scholar] [CrossRef]
- Del Valle, J.C.; Buide, M.L.; Casimiro-Soriguer, I.; Whittall, J.B.; Narbona, E. On flavonoid accumulation in different plant parts: Variation patterns among individuals and populations in the shore campion (Silene littorea). Front. Plant. Sci. 2015, 6, 939. [Google Scholar] [CrossRef] [PubMed]
- Koski, M.H.; Ashman, T.-L. An altitudinal cline in UV floral pattern corresponds with a behavioral change of a generalist pollinator assemblage. Ecology 2015, 96, 3343–3353. [Google Scholar] [CrossRef] [PubMed]
- Koski, M.H.; Ashman, T.-L. Floral pigmentation patterns provide an example of Gloger’s rule in plants. Nat. Plants 2015, 1, 14007. [Google Scholar] [CrossRef] [PubMed]
- Jaakola, L.; Hohtola, A. Effect of latitude on flavonoid biosynthesis in plants. Plant. Cell Env. 2010, 33, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Seckmeyer, G.; Pissulla, D.; Glandorf, M.; Henriques, D.; Johnsen, B.; Webb, A.; Siani, A.-M.; Bais, A.; Kjeldstad, B.; Brogniez, C.; et al. Variability of UV irradiance in Europe. Photochem. Photobiol. 2008, 84, 172–179. [Google Scholar] [CrossRef]
- Darwin, C. The Effects of Cross and Self Fertilization in the Vegetable Kingdom; John Murray: London, UK, 1876. [Google Scholar]
- Grant, V. Pollination systems as isolating mechanisms in angiosperms. Evolution 1949, 3, 82–97. [Google Scholar] [CrossRef]
- Levin, D.A. The origin of reproductive isolating mechanisms in flowering plants. Taxon 1971, 20, 91–113. [Google Scholar] [CrossRef]
- Grant, V. Modes and origins of mechanical and ethological isolation in angiosperms. Proc. Natl. Acad. Sci. USA 1994, 91, 3–10. [Google Scholar] [CrossRef]
- Grant, V. Isolation and hybridization between Aquilegia formosa and A. pubescens. El Aliso 1952, 2, 341–360. [Google Scholar]
- Chen, Z.; Liu, C.-Q.; Sun, H.; Niu, Y. The ultraviolet colour component enhances the attractiveness of red flowers of a bee-pollinated plant. J. Plant. Ecol. 2020, 13, 354–360. [Google Scholar] [CrossRef]
- Lunau, K.; Papiorek, S.; Eltz, T.; Sazima, M. Avoidance of achromatic colours by bees provides a private niche for hummingbirds. J. Exp. Biol. 2011, 214, 1607. [Google Scholar] [CrossRef] [PubMed]
- Koski, M.H.; Ashman, T.-L. Dissecting pollinator responses to a ubiquitous ultraviolet floral pattern in the wild. Funct. Ecol. 2014, 28, 868–877. [Google Scholar] [CrossRef]
- Mitich, L.W. Corn Poppy (Papaver rhoeas L.). Weed Technol. 2000, 14, 826–829. [Google Scholar] [CrossRef]
- Keasar, T.; Harari, A.R.; Sabatinelli, G.; Keith, D.; Dafni, A.; Shavit, O.; Zylbertal, A.; Shmida, A. Red anemone guild flowers as focal places for mating and feeding by Levant glaphyrid beetles. Bot. J. Linn. Soc. 2010, 99, 808–817. [Google Scholar] [CrossRef]
- Sabatinelli, G.; Eberle, J.; Fabrizi, S.; Ahrens, D. A molecular phylogeny of Glaphyridae (Coleoptera: Scarabaeoidea): Evolution of pollination and association with ‘Poppy guild’ flowers. Syst. Entomol. 2020. [Google Scholar] [CrossRef]
- Meyer, R.S.; Purugganan, M.D. Evolution of crop species: Genetics of domestication and diversification. Nat. Rev. Genet. 2013, 14, 840–852. [Google Scholar] [CrossRef]
- Darwin, C. The Variation of Animals and Plants under Domestication; John Murray: London, UK, 1868; Volume 2. [Google Scholar]
- Dewet, J.M.J.; Harlan, J.R. Weeds and domesticates: Evolution in man-made habitat. Econ. Bot. 1975, 29, 99–107. [Google Scholar] [CrossRef]
- Barrett, S.H. Crop mimicry in weeds. Econ. Bot. 1983, 37, 255–282. [Google Scholar] [CrossRef]
- Vorobyev, M.; Osorio, D. Receptor noise as a determinant of colour thresholds. Proc. R. Soc. B 1998, 265, 351–358. [Google Scholar] [CrossRef]
- Vorobyev, M.; Brandt, R.; Peitsch, D.; Laughlin, S.B.; Menzel, R. Colour thresholds and receptor noise: Behaviour and physiology compared. Vis. Res. 2001, 41, 639–653. [Google Scholar] [CrossRef]
- Koshitaka, H.; Kinoshita, M.; Vorobyev, M.; Arikawa, K. Tetrachromacy in a butterfly that has eight varieties of spectral receptors. Proc. R. Soc. B 2008, 275, 947–954. [Google Scholar] [CrossRef]
- Goldsmith, T.H.; Butler, B.K. The roles of receptor noise and cone oil droplets in the photopic spectral sensitivity of the budgerigar, Melopsittacus undulatus. J. Comp. Physiol. A 2003, 189, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Lind, O.; Chavez, J.; Kelber, A. The contribution of single and double cones to spectral sensitivity in budgerigars during changing light conditions. J. Comp. Physiol. A 2013, 200, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Wyszecki, G.; Stiles, W.S. Color Science: Concepts and Methods, Quantitative Data and Formulae, 2nd ed.; John Wiley & Sons: New York, NY, USA, 1982. [Google Scholar]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Harms, J.; Hadar, R.; Márquez, N.; Menzel, R.; Shmida, A.; Stavenga, D.G.; Vorobyev, M. Enhanced UV-Reflection Facilitated a Shift in the Pollination System of the Red Poppy, Papaver rhoeas (Papaveraceae). Plants 2020, 9, 927. https://doi.org/10.3390/plants9080927
Martínez-Harms J, Hadar R, Márquez N, Menzel R, Shmida A, Stavenga DG, Vorobyev M. Enhanced UV-Reflection Facilitated a Shift in the Pollination System of the Red Poppy, Papaver rhoeas (Papaveraceae). Plants. 2020; 9(8):927. https://doi.org/10.3390/plants9080927
Chicago/Turabian StyleMartínez-Harms, Jaime, Ravit Hadar, Natalia Márquez, Randolf Menzel, Avi Shmida, Doekele G. Stavenga, and Misha Vorobyev. 2020. "Enhanced UV-Reflection Facilitated a Shift in the Pollination System of the Red Poppy, Papaver rhoeas (Papaveraceae)" Plants 9, no. 8: 927. https://doi.org/10.3390/plants9080927
APA StyleMartínez-Harms, J., Hadar, R., Márquez, N., Menzel, R., Shmida, A., Stavenga, D. G., & Vorobyev, M. (2020). Enhanced UV-Reflection Facilitated a Shift in the Pollination System of the Red Poppy, Papaver rhoeas (Papaveraceae). Plants, 9(8), 927. https://doi.org/10.3390/plants9080927





