The Role of Grafting in the Resistance of Tomato to Viruses
Abstract
1. Introduction
2. Why Grafting?
3. Screening of Solanum spp. Germplasm
3.1. Results with TSWV
3.2. Results with CMV
3.3. Results with PVY
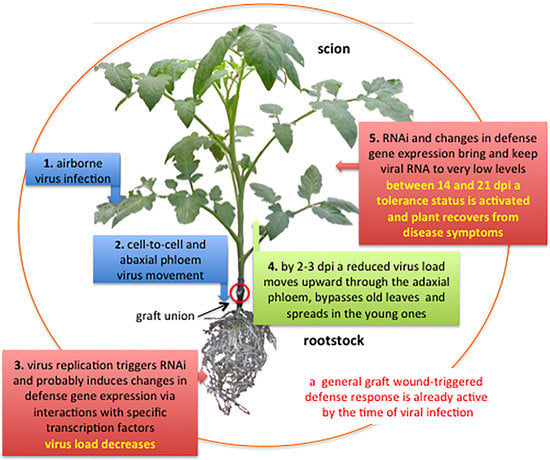
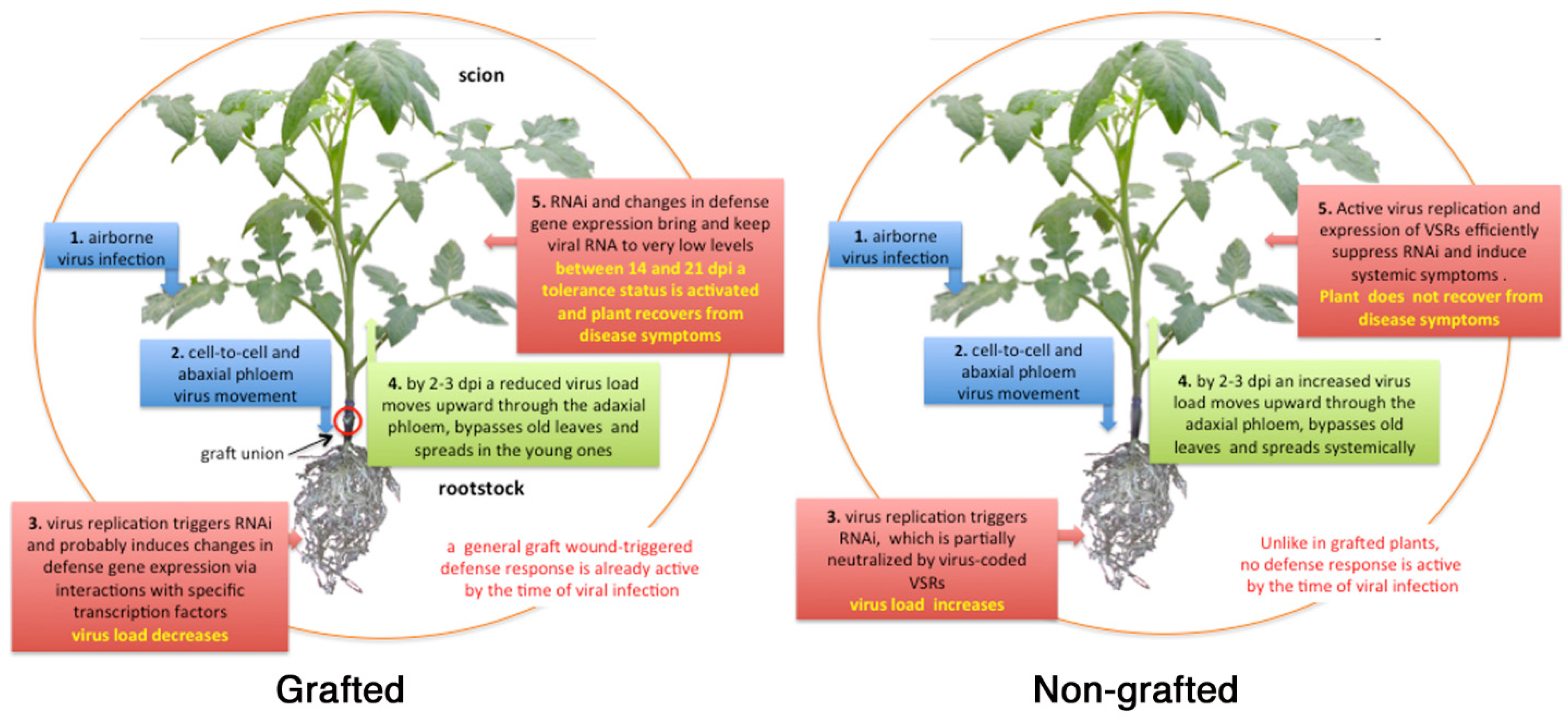
4. Proposed Models
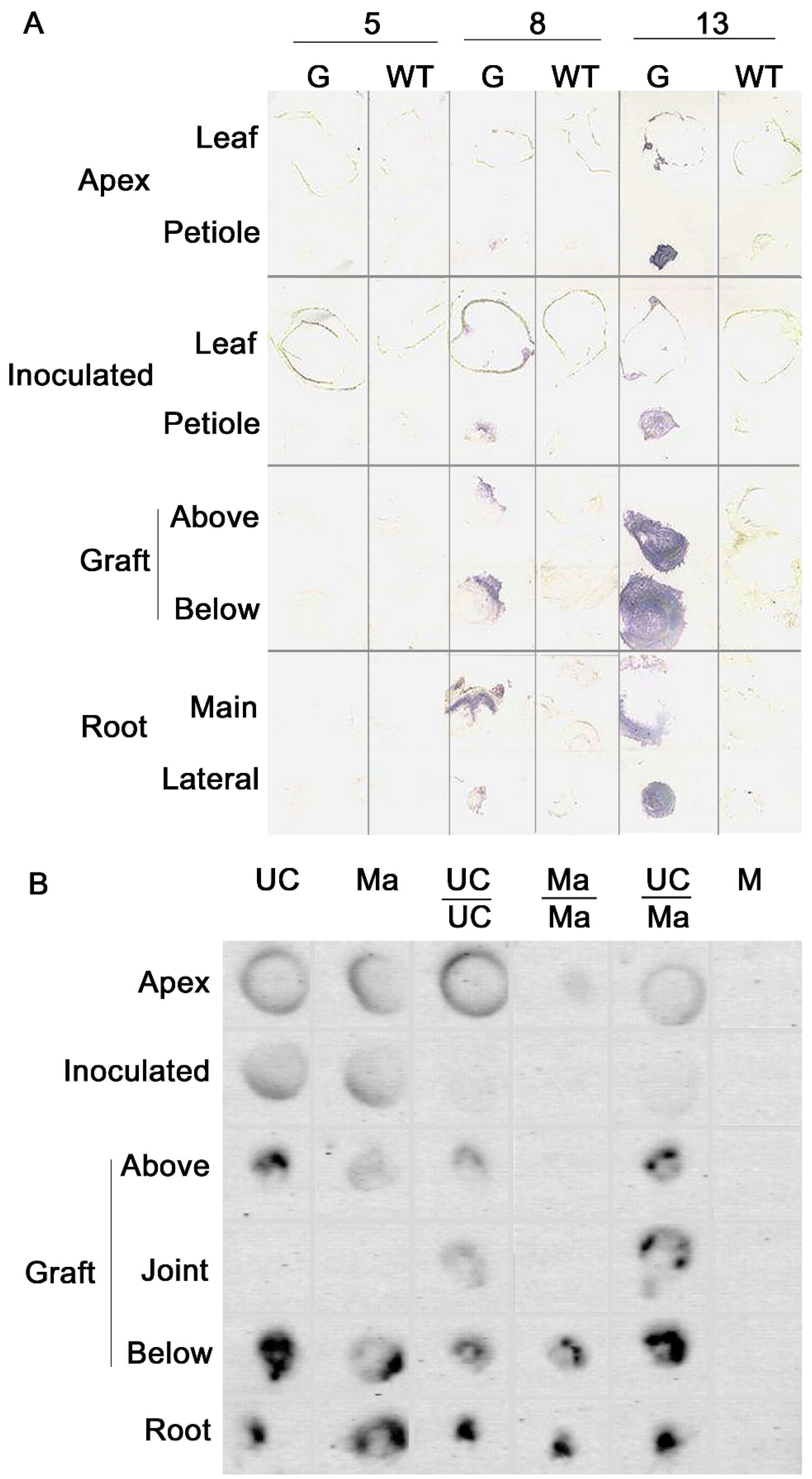
4.1. Virus Movement in Grafted Plants
4.2. RNA Interference
4.3. Response to Graft Wound and Viral Infection
4.4. Oxidative Stress and Antioxidant Enzymes
4.5. Resilience and Tolerance
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Xu, C.; Sun, X.; Taylor, A.; Jiao, C.; Xu, Y.; Cai, X.; Wang, X.; Ge, C.; Pan, G.; Wang, Q.; et al. Diversity, distribution, and evolution of tomato viruses in China uncovered by small RNA sequencing. J. Virol. 2017, 91, e00173–17. [Google Scholar] [CrossRef] [PubMed]
- Scholthof, K.B.G.; Adkins, S.; Czosnek, H.; Palukaitis, P.; Jacquot, E.; Hohn, T.; Hohn, B.; Saunders, K.; Candresse, T.; Ahlquist, P.; et al. Top 10 plant viruses in molecular plant pathology. Mol. Plant Pathol. 2011, 12, 938–954. [Google Scholar] [CrossRef] [PubMed]
- García-Arenal, F.; Palukaitis, P. Cucumber mosaic virus. In Desk Encyclopedia of Plant and Fungal Virology; Mahy, B.W.J., van Regenmortel, M.H.V., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 171–176. [Google Scholar]
- Jacquemond, M. Cucumber mosaic virus. Adv. Virus Res. 2012, 84, 440–505. [Google Scholar]
- Parrella, G.; Gognalons, P.; Gebre-Selassiè, K.; Vovlas, C. An update of the host range of Tomato spotted wilt virus. J. Plant Pathol. 2003, 85, 227–264. [Google Scholar]
- Turina, M.; Tavella, L.; Ciuffo, M. Tospoviruses in the Medirranean area. Adv. Virus Res. 2012, 84, 404–439. [Google Scholar]
- Moury, B.; Verdin, E. Viruses of pepper crop in the Mediterranean basin: A remarkable stasis. Adv. Virus Res. 2012, 84, 128–163. [Google Scholar]
- Loebenstein, G.; Gaba, V. Viruses of Potato. Adv. Virus Res. 2012, 84, 210–248. [Google Scholar]
- Turina, M.; Kormelink, R.; Resende, T. Resistance to tospoviruses in vegetable crops: Epidemiological and molecular aspects. Annu. Rev. Phytopathol. 2016, 54, 347–371. [Google Scholar] [CrossRef]
- de Oliveira, A.S.; Boiteux, L.S.; Kormelink, R.; Resende, R.O. The Sw-5 gene cluster: Tomato breeding and research toward orthotospovirus disease control. Front. Plant Sci. 2018, 9, 1055. [Google Scholar] [CrossRef]
- Oliveira, R.M.; Dianese, É.C.; Lima, M.F.; Resende, R.O.; Inoue-Nagata, A.I.; Boiteux, L.S. Sources of resistance to Potato virus Yand Pepper yellow mosaic virus in Solanum (section Lycopersicon) germplasm. Eur. J. Plant Pathol. 2017, 150, 691–699. [Google Scholar] [CrossRef]
- Pappu, H.R.; Jones, R.A.C.; Jain, R.K. Global status of Tospovirus epidemics in diverse cropping systems: Successes achieved and challenges ahead. Virus Res. 2009, 141, 219–236. [Google Scholar] [CrossRef] [PubMed]
- López, C.; Aramburu, J.; Galipienso, L.; Soler, S.; Nuez, F.; Rubio, L. Evolutionary analysis of tomato Sw-5 resistance-breaking isolates of Tomato spotted wilt virus. J. Gen. Virol. 2011, 92, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Peiró, A.; Cañizares, M.C.; Rubio, L.; López, C.; Moriones, E.; Aramburu, J.; Navarro, J.S. The movement protein (NSM) of Tomato spotted wilt virus is the avirulence determinant in the tomato Sw-5 gene-based resistance. Mol. Plant Pathol. 2014, 15, 802–813. [Google Scholar] [CrossRef]
- Abad, J.; Anastasio, G.; Fraile, A.; García-Arenal, F. A search for resistance to Cucumber mosaic virus in the genus Lycopersicon. J. Plant Pathol. 2000, 82, 39–48. [Google Scholar]
- Murphy, J.F.; Sikora, E.J.; Sammons, B.; Kaniewski, W.K. Performance of transgenic tomatoes expressing cucumber mosaic virus CP gene under epidemic conditions. Hort. Sci. 1998, 33, 1032–1035. [Google Scholar] [CrossRef]
- Tomassoli, L.; Ilardi, V.; Barba, M.; Kaniewski, W. Resistance of transgenic tomato to Cucumber mosaic cucumovirus under field conditions. Mol. Breed. 1999, 5, 121–130. [Google Scholar] [CrossRef]
- Gallitelli, D.; Vovlas, C.; Martelli, G.P.; Montasser, M.S.; Tousignant, M.E.; Kaper, J.M. Satellite-mediated protection of tomato against cucumber mosaic virus: II. Field test under natural epidemic conditions in southern Italy. Plant Dis. 1991, 75, 93–95. [Google Scholar] [CrossRef]
- Sayama, H.; Sato, T.; Kominato, M.; Natsuaki, T.; Kaper, J.M. Field testing of a satellite-containing attenuated strain of cucumber mosaic virus for tomato protection in Japan. Phytopathology 1993, 83, 405–410. [Google Scholar] [CrossRef]
- Mascia, T.; Gallitelli, D. Satellites as Viral Biocontrol Agents. In Viroids and Satellites; Hadidi, A., Flores., R., Randles, J.W., Palukaitis, P., Eds.; Academic Press: London, UK, 2017; pp. 681–688. [Google Scholar]
- Aramburu, J.; Galipienso, L.; Matas, M. Characterization of potato virus Y isolates from tomato crops in northeast Spain. Eur. J. Plant Pathol. 2006, 115, 247–258. [Google Scholar] [CrossRef]
- Parrella, G.; Ruffel, S.; Moretti, A.; Morel, C.; Palloix, A.; Caranta, C. Recessive resistance genes against potyviruses are localized in collinear genomic regions of the tomato (Lycopersicon spp.) and pepper (Capsicum spp.) genomes. Theor. Appl. Genet. 2002, 105, 855–861. [Google Scholar] [CrossRef]
- Kang, B.C.; Yeam, I.; Frantz, J.D.; Murphy, J.F.; Jahn, M.M. The pvr1 locus in pepper encodes a translation initiation factor eIF4E that interacts with Tobacco etch virus VPg. Plant J. 2005, 41, 392–405. [Google Scholar] [CrossRef] [PubMed]
- Albacete, A.; Martinez-Andujar, C.; Martinez-Pérez, A.; Thompson, A.J.; Dodd, I.C.; Pérez-Alfocea, F. Unravelling rootstock x scion interactions to improve food security. J. Exp. Bot. 2015, 66, 2211–2226. [Google Scholar] [CrossRef] [PubMed]
- Rouphael, Y.; Kyriacou, M.C.; Colla, G. Vegetable Grafting: A Toolbox for Securing Yield Stability under Multiple Stress Conditions. Front. Plant Sci. 2018, 8, 2255. [Google Scholar] [CrossRef] [PubMed]
- Kyriacou, M.C.; Rouphael, Y. Towards a new definition of quality for fresh fruits and vegetables. Sci. Hortic. 2018, 234, 463–469. [Google Scholar] [CrossRef]
- Louws, F.J.; Rivard, C.L.; Kubota, C. Grafting fruiting vegetables to manage soilborne pathogens, foliar pathogens, arthropods and weeds. Sci. Hortic. 2010, 127, 127–146. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y.; Leonardi, C.; Bie, Z. Role of grafting in vegetable crops grown under saline conditions. Sci. Hort. 2010, 127, 147–155. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y.; Mirabelli, C.; Cardarelli, M. Nitrogenus efficiency traits of mini-watermelon in response to grafting and nitrogen-fertilization doses. J. Plant Nutr. Soil Sci. 2011, 174, 933–941. [Google Scholar] [CrossRef]
- Savvas, D.; Colla, G.; Rouphael, Y.; Schwarz, D. Amelioration of heavy metal and nutrient stress in fruit vegetables by grafting. Sci. Hortic. 2010, 127, 156–161. [Google Scholar] [CrossRef]
- Rouphael, Y.; Cardarelli, M.; Colla, G. Yield, mineral composition, water relations, and water use efficiency of grafted mini-watermelon plants under deficit irrigation. HortScience 2008, 43, 730–736. [Google Scholar] [CrossRef]
- Schwarz, D.; Rouphael, Y.; Colla, G.; Venema, J.H. Grafting as a tool to improve tolerance of vegetables to abiotic stresses: Thermal stress, water stress and organic pollutants. Sci. Hortic. 2010, 127, 162–171. [Google Scholar] [CrossRef]
- Borgognone, D.; Colla, G.; Rouphael, Y.; Cardarelli, M.; Rea, E.; Schwarz, D. (Effect of nitrogen form and nutrient solution pH on growth and mineral composition of self-grafted and grafted tomatoes. Sci. Hortic. 2013, 149, 61–69. [Google Scholar] [CrossRef]
- Gaion, L.A.; Braz, L.T.; Carvalho, R.F. Grafting in vegetable crops: A great technique for Agriculture. Int. J. Veg. Sci. 2018, 24, 85–102. [Google Scholar] [CrossRef]
- Riga, P.; Benedicto, L.; García-Flores, L.; Villaño, D.; Medina, S.; Gil-Izquierdo, A. Rootstock effect on serotonin and nutritional quality of tomatoes produced under low temperature and light conditions. J. Food Comp. Anal. 2016, 46, 50–59. [Google Scholar] [CrossRef]
- Tsaballa, A.; Athanasiadis, C.; Pasentsis, K.; Ganopoulos, I.; Nianiou-Obeidat, I.; Tsaftaris, A. Molecular studies of inheritable grafting induced changes in Pepper (Capsicum annuum) fruit shape. Sci. Hort. 2013, 149, 2–8. [Google Scholar] [CrossRef]
- Temperini, O.; Calabrese, N.; Temperini, A.; Rouphael, Y.; Tesid, R.; Lenzid, A.; Carito, A.; Colla, G. Grafting artichoke onto cardoon rootstocks: Graft compatibility, yield and Verticillium wilt incidence. Sci. Hort. 2013, 149, 22–27. [Google Scholar] [CrossRef]
- Kumar, S.; Bharti, N.; Saravaiya, S.N. Vegetable Grafting: A Surgical Approach to combat biotic and abiotic stresses: A review. Agric. Rev. 2018, 39, 1–11. [Google Scholar] [CrossRef]
- Lee, J.M.; Kubota, C.; Tsao, S.J.; Bie, Z.; Echevarria, P.H.; Morra, L.; Oda, M. Current status of vegetable grafting: Diffusion, grafting techniques, automation. Sci. Hortic. 2010, 127, 93–105. [Google Scholar] [CrossRef]
- Singh, H.; Kumar, P.; Chaudhari, S.; Edelstei, M. Tomato grafting: A global perspective. Hortscience 2017, 52, 1328–1336. [Google Scholar] [CrossRef]
- Avgelis, A.D. Watermelon necrosis caused by a strain of melon necrotic spot virus. Plant Pathol. 1989, 38, 618–622. [Google Scholar] [CrossRef]
- Gomez, J.; Cuadrado, I.; Velasco, V. Melon necrotic spot virus (MNSV) in Almeria. III. The effect of melon grafting on MNSV control. Bol. San. Veg. Plagas 1993, 19, 197–219. [Google Scholar]
- Hirai, G.; Nakazumi, H.; Yagi, R.; Nakano, M.; Shiga, Y.; Dohi, H. A new MNSV resistant melon rootstock variety “Sorachi Dai Ko 3”. Bull. Hokkaido Prefect. Agricult. Exp. Stn. 2003, 84, 47–54. [Google Scholar]
- Banttari, E.E.; Vanette, J.R. Aerosol spread of plant viruses: Potential role in disease outbreaks. Ann. N. Y. Acad. Sci. 1980, 353, 167–173. [Google Scholar] [CrossRef]
- Fillhart, R.C.; Bachand, G.D.; Castello, J.D. Airborne transmission of tomato mosaic Tobamovirus and its occurrence in red spruce in the northeastern United States. Can J. Forest Res. 1997, 27, 1176–1181. [Google Scholar] [CrossRef]
- Rivero, R.M.; Ruiz, J.M.; Romero, L. Role of grafting in horticultural plants under stress conditions. Food. Agric. Environ. 2003, 1, 70–74. [Google Scholar]
- Rivard, C.L.; Louws, F.J. Grafting to manage soilborne diseases in heirloom tomato production. Hortscience 2008, 43, 2104–2111. [Google Scholar] [CrossRef]
- Miguel, A. Use of grafted plants and IPM methods for the production of tomatoes in the Mediterranean region. In Proceedings of the International Conference Alternatives to Methyl Bromide, Lisbon, Portugal, 27–30 September 2004; Batchelor, T., Flávia, A., Eds.; European Commission: Brussels, Belgium; pp. 141–145.
- Schwarz, D.; Beuch, U.; Bandte, M.; Fakhro, A.; Buttner, C.; Obermeieret, C. Spread and interaction of Pepino mosaic virus (PepMV) and Pythium aphanidermatum in a closed nutrient solution recirculation system: Effects on tomato growth and yield. Plant Pathol. 2010, 59, 443–452. [Google Scholar] [CrossRef]
- Samuel, G. The movement of tobacco mosaic virus within the plant. Ann. Appl. Biol. 1934, 21, 90–111. [Google Scholar] [CrossRef]
- Hull, R. Plant Virology, 5th ed.; Academic Press: London, UK, 2014. [Google Scholar]
- Andrianifahanana, M.; Lovins, K.; Dute, R.; Sikora, E.; Murphy, J.F. Pathway for phloem-dependent movement of pepper mottle potyvirus in the stem of Capsicum annuum. Phytopathology 1997, 87, 892–898. [Google Scholar] [CrossRef]
- Leisner, S.M.; Turgeon, R.; Howell, S.H. Long distance movement of cauliflower mosaic virus in infected turnip plants. Mol. Plant Microbe Interact 1992, 5, 41–47. [Google Scholar] [CrossRef]
- Mas, P.; Pallas, V. Long-distance movement of cherry leaf roll virus in infected tobacco plants. J. Gen. Virol. 1996, 77, 531–540. [Google Scholar] [CrossRef]
- Cheng, N.H.; Lin, C.L.; Carter, S.A.; Nelson, R.S. Vascular invasion routes and systemic accumulation patterns of tobacco mosaic virus in Nicotiana benthamiana. Plant J. 2000, 23, 349–362. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.S.; Carter, S.A.; Deom, C.M.; Nelson, R.S. Tobamovirus and potyvirus accumulation in minor veins of inoculated leaves from representatives of the Solanaceae and Fabaceae. Plant Physiol. 1998, 116, 125–136. [Google Scholar] [CrossRef]
- Vuorinen, A.L.; Kelloniemia, J.; Valkonen, J.P.T. Why do viruses need phloem for systemic invasion of plants? Plant Sci. 2011, 181, 355–363. [Google Scholar] [CrossRef]
- Navarro, J.A.; Sanchez-Navarro, J.A.; Pallas, V. Key checkpoints in the movement of plant viruses through the host. Adv. Virus Res. 2019, 104, 1–64. [Google Scholar]
- Turgeon, R. The sink–source transition in leaves. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1989, 40, 119–138. [Google Scholar] [CrossRef]
- Rajamäki, M.-L.; Valkonen, J.P.T. Viral genome-linked protein (VPg) controls accumulation and phloem-loading of a potyvirus in inoculated potato leaves. Mol. Plant Microbe Interact. 2002, 15, 138–149. [Google Scholar]
- Blackman, L.M.; Boevink, P.; Cruz, S.S.; Palukaitis, P.; Oparka, K.J. The movement protein of cucumber mosaic virus traffics into sieve elements in minor veins of Nicotiana clevelandii. Plant Cell 1998, 10, 525–538. [Google Scholar] [CrossRef]
- Spanò, R.; Mascia, T.; Kormelink, R.; Gallitelli, D. Grafting on a Non-Transgenic Tolerant Tomato Variety Confers Resistance to the Infection of a Sw5- Breaking Strain of Tomato spotted wilt virus via RNA Silencing. PLoS ONE 2015, 10, e0141319. [Google Scholar] [CrossRef]
- Spanò, R.; Gallitelli, D.; Mascia, T. Grafting to manage infections of top stunting and necrogenic strains of cucucmber mosaic virus in tomato. Ann. App. Biol. 2017, 171, 393–404. [Google Scholar] [CrossRef]
- Spanò, R.; Ferrara, M.; Montemurro, C.; Mulè, G.; Gallitelli, D.; Mascia, T. Grafting alters tomato transcriptome and enhances tolerance to an airborne virus infection. Sci. Rep. 2020, 10, 2538. [Google Scholar] [CrossRef]
- Mascia, T.; Santovito, E.; Gallitelli, D.; Cillo, F. Evaluation of reference genes for quantitative reverse transcription polymerase chain reaction normalization in infected tomato plants. Mol. Plant Pathol. 2010, 11, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Finetti-Sialer, M.M.; Lanave, C.; Padula, M.; Vovlas, C.; Gallitelli, D. Occurrence of two distinct tomato spotted wilt virus subgroups in Southern Italy. J. Plant Pathol. 2002, 84, 145–152. [Google Scholar]
- Ciuffo, M.; Finetti Sialer, M.M.; Gallitelli, D.; Turina, M. First report in Italy of a resistance breaking strain of infecting tomato cultivars carrying the resistance gene. New Dis. Rep. 2005, 10, 48. [Google Scholar]
- Gallitelli, D. The ecology of cucumber mosaic virus and sustainable agriculture. Virus Res. 2000, 71, 9–21. [Google Scholar] [CrossRef]
- Aramburu, J.; Galipienso, L.; Lopez, C. Reappearance of cucumber mosaic virus isolates belonging to subgroup IB in tomato plants in north-eastern Spain. J. Phytopathol. 2007, 155, 513–518. [Google Scholar] [CrossRef]
- Mascia, T.; Finetti-Sialer, M.M.; Cillo, F.; Gallitelli, D. Biological and molecular characterization of a recombinant isolate of Potato virus Y associated with a tomato necrotic disease occurring in Italy. J. Plant Pathol. 2010, 92, 131–138. [Google Scholar]
- Ruiz, M.T.; Voinnet, O.; Baulcombe, D.C. Initiation and maintenance of virus-induced gene silencing. Plant Cell 1998, 10, 937–946. [Google Scholar] [CrossRef]
- Ding, S.W.; Voinnet, O. Antiviral immunity directed by small RNAs. Cell 2007, 130, 413–426. [Google Scholar] [CrossRef]
- Kalantidis, K.; Schumacher, H.T.; Alexi-adis, T.; Helm, J.M. RNA silencing movement in plants. Biol. Cell 2008, 100, 13–26. [Google Scholar] [CrossRef]
- Molnar, A.; Melnyk, C.; Baulcombe, D.C. Silencing signals in plants: A long journey for small RNAs. Genome Biol. 2011, 12, 215. [Google Scholar] [CrossRef]
- Mermigka, G.; Verret, F.; Kalantidis, K. RNA silencing movement in plants. J. Integr. Plant Biol. 2016, 58, 328–342. [Google Scholar] [CrossRef] [PubMed]
- Palauqui, J.C.; Elmayan, T.; Pollien, J.M.; Vaucheret, H. Systemic acquired silencing: Transgene-specific post-transcriptional silencing is transmitted by grafting from silenced stocks to non-silenced scions. EMBO J. 1997, 16, 4738–4745. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Grierson, D. Enhancement of post-transcriptional gene silencing by grafting. Plant Signal Behav. 2008, 3, 30–33. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ali, E.M.; Kobayashi, K.; Yamaoka, N.; Ishikawa, M.; Nishiguchi, M. Graft Transmission of RNA Silencing to Non-Transgenic Scions for Conferring Virus Resistance in Tobacco. PLoS ONE 2013, 8, e63257. [Google Scholar]
- Kasai, A.; Sano, T.; Harada, T. Scion on a Stock Producing siRNAs of Potato Spindle Tuber Viroid (PSTVd) Attenuates Accumulation of the Viroid. PLoS ONE 2013, 8, e57736. [Google Scholar] [CrossRef]
- Kørner, C.J.; Pitzalis, N.; Peña, E.J.; Erhardt, M.; Vazquez, F.; Heinlein, M. Crosstalk between PTGS and TGS pathways in natural antiviral immunity and disease recovery. Nat. Plants 2018, 4, 157–164. [Google Scholar] [CrossRef]
- Takeda, A.; Sugiyama, K.; Nagano, H.; Mori, M.; Kaido, M.; Mise, K.; Tsuda, S.; Okuno, T. Identification of a novel RNA silencing suppressor, NSs protein of Tomato spotted wilt virus. FEBS Lett. 2002, 532, 75–79. [Google Scholar] [CrossRef]
- Bucher, E.; Sijen, T.; de Haan, P.; Goldbach, R.; Prins, M. Negative-strand tospoviruses and tenuiviruses carry a gene for a suppressor of gene silencing at analogous genomic positions. J. Virol. 2003, 77, 1329–1336. [Google Scholar] [CrossRef]
- Hedil, M.; De Ronde, D.; Kormelink, R. Biochemical analysis of NSs from different tospoviruses. Virus Res. 2017, 242, 149–155. [Google Scholar] [CrossRef]
- Rubino, L. Biology of satellites. In Viroids and Satellites; Hadidi, A., Flores, R., Randles, J.W., Palukaitis, P., Eds.; Academic Press: London, UK, 2017; pp. 567–575. [Google Scholar]
- Cillo, F.; Finetti Sialer, M.M.; Papanice, M.A.; Gallitelli, D. Analysis of mechanisms involved in the cucumber mosaic virus satellite RNA-mediated transgenic resistance in tomato plants. Mol. Plant Microbe Interact. 2004, 17, 98–108. [Google Scholar] [CrossRef]
- Gilardi, G.; Gullino, M.L.; Garibaldi, A. Critical aspects of grafting as a possible strategy to manage soil-borne pathogens. Sci. Hortic. 2013, 149, 19–21. [Google Scholar] [CrossRef]
- Guan, W.J.; Zhao, X.; Hassell, R.; Thies, J. Defense mechanisms involved in disease resistance of grafted vegetables. Hortscience 2012, 47, 164–170. [Google Scholar] [CrossRef]
- Cookson, S.J.; Clemente Moreno, M.J.; Hevin, C.; Nyamba Mendome, L.Z.; Delrot, S.; Magnin, N.; Trossat-Magnin, C.; Ollat, N. Heterografting with nonself rootstocks induces genes involved in stress responses at the graft interface when compared with autografted controls. J. Exp. Bot. 2014, 65, 2473–2481. [Google Scholar] [CrossRef]
- Melnyk, C.W. Plant grafting: Insights into tissue regeneration. Regeneration 2016, 4, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Melnyk, C.W.; Gabel, A.; Hardcastle, T.J.; Robinson, S.; MIyashima, S.; Grosse, I.; Meyerowitz, E.M. Transcriptome dynamics at Arabidopsis graft junctions reveal an inter tissue recognition mechanism that activates vascular regeneration. Proc. Natl. Acad. Sci. USA 2019, 115, E2447–E2456. [Google Scholar] [CrossRef]
- Cheong, Y.H.; Chang, H.-S.; Gupta, R.; Wang, X.; Zhu, T.; Luan, S. Transcriptional profiling reveals novel interactions between wounding, pathogen, abiotic stress, and hormonal responses in Arabidopsis. Plant Physiol. 2002, 129, 661–677. [Google Scholar] [CrossRef]
- Heyman, J.; Canher, B.; Bisht, A.; Christiaens, F.; De Veylder, L. Emerging role of the plant ERF transcription factors in coordinating wound defense responses and repair. J. Cell Sci. 2018, 131, jcs208215. [Google Scholar] [CrossRef]
- Xie, Z.; Nolan, T.M.; Jiang, H.; Yin, Y. AP2/ERF Transcription Factor Regulatory Networks in Hormone and Abiotic Stress Responses in Arabidopsis. Front. Plant Sci. 2019, 10, 228. [Google Scholar] [CrossRef]
- Fujimoto, S.Y.; Ohta, M.; Usui, A.; Shinshi, H.; Ohme-Takagi, M. Arabidopsis ethylene-responsive element binding factors act as transcriptional activators or repressors of GCC box-mediated gene expression. Plant Cell 2000, 12, 393–404. [Google Scholar]
- Pagnussat, G.C.; Yu, H.-J.; Quy, A.; Ngo, Q.A.; Rajani, S.; Mayalagu, S.; Johnson, C.S.; Capron, A.; Xie, L.-F.; Ye, D.; et al. Genetic and molecular identification of genes required for female gametophyte development and function. Arab. Dev. 2005, 132, 603–614. [Google Scholar] [CrossRef]
- Del Toro, F.J.; Donaire, L.; Aguilar, E.; Chung, B.-N.; Tenllado, F.; Canto, T. Potato virus Y HCPro suppression of antiviral silencing in Nicotiana benthamiana plants correlates with its ability to bind in vivo to 21- and 22- nucleotide small RNAs of viral sequence. J. Virol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Soitamo, A.J.; Jada, B.; Lehto, K. HC-Pro silencing suppressor significantly alters the gene expression profile in tobacco leaves and flowers. BMC Plant Biol. 2011, 11, 68. [Google Scholar] [CrossRef] [PubMed]
- Aloni, B.; Karni, L.; Deventurero, G.; Levin, Z.; Cohen, R.; Katzir, N.; Lotan-Pompan, M.; Edelstein, M.; Aktas, H.; Turhan, E. Physiological and biochemical changes at the rootstock-scion interface in graft combinations between Cucurbita rootstocks and a melon scion. J. Hortic. Sci. Biotech. 2008, 83, 777–783. [Google Scholar] [CrossRef]
- Xu, Q.; Guo, S.R.; Li, H.; Du, N.S.; Shu, S.; Sun, J. Physiological aspects of compatibility and incompatibility in grafted cucumber seedlings. J. Am. Soc. Hort. Sci. 2015, 140, 299–307. [Google Scholar] [CrossRef]
- Mandadi, K.K.; Sholthof, K.-B.G. Plant Immune Responses Against Viruses: How Does a Virus Cause Disease? Plant Cell 2013, 25, 1489–1505. [Google Scholar] [CrossRef]
- Sagi, M.; Davydov, O.; Orazova, S.; Yesbergenova, Z.; Ophir, R.; Stratmann, J.W.; Fluhr, R. Plant respiratory burst oxidase homologs impinge on wound responsiveness and development in Lycopersicon esculentum. Plant Cell 2004, 16, 616–628. [Google Scholar] [CrossRef]
- Rouxel, T.; Balesdent, M.-H. Avirulence Genes. In Encyclopedia of Life Sciences (ELS); John Wiley & Sons, Ltd.: Chichester, UK, 2010. [Google Scholar]
- Aloni, B.; Cohen, R.; Karni, L.; Aktas, H.; Edelstein, M. Hormonal signaling in rootstock-scion interactions. Sci. Hort. 2010, 127, 119–126. [Google Scholar] [CrossRef]
- Paudel, D.B.; Sanfaçon, H. Exploring the Diversity of Mechanisms Associated with Plant Tolerance to Virus Infection. Front. Plant Sci. 2018, 9, 1575. [Google Scholar] [CrossRef]
- Berger, S.; Sinha, A.K.; Roitsch, T. Plant physiology meets phytopathology: Plant primary metabolism and plant–pathogen interactions. J. Exp. Bot. 2017, 58, 4019–4026. [Google Scholar] [CrossRef]
- Cooper, J.I.; Jones, A.T. Responses of plants to viruses: Proposals for the use of terms. Phytopathology 1983, 73, 127–128. [Google Scholar] [CrossRef]
- Pagan, I.; Garcia-Arenal, F. Tolerance to plant pathogens: Theory and experimental evidence. Int. J. Mol. Sci. 2018, 19, 810. [Google Scholar] [CrossRef] [PubMed]

| Virus | Acronim | Transmission |
|---|---|---|
| Alfalfa mosaic virus | AMV | aphids |
| Cucumber mosaic virus | CMV | aphids |
| Parietaria mottle virus | PMoV | thrips |
| Pelargonium zonate spot virus | PZSV | pollen, thrips |
| Pepino mosaic virus | PepMoV | contact |
| Potato virus Y | ToMV | aphids |
| Tobacco mosaic virus | TMV | contact |
| Tomato mosaic virus | ToMV | contact/aerosol |
| Tomato brown fruit rugose virus | ToBFRV | contact |
| Tomato cholorosis virus | ToCV | aleyrodids |
| Tomato infectious chlorosis virus | TICV | aleyrodids |
| Tomato spotted wilt virus | TSWV | thrips |
| Tomato yellow leaf curl virus | TYLCV | aleyrodids |
| Tomato yellow leaf curl Sardinia virus | TYLCSV | aleyrodis |
| 14 * | 21 * | 21 * | 28 * | |
|---|---|---|---|---|
| Non-Grafted Plants | Symptoms | Symptoms | Viral RNA Load ** | Symptoms |
| UC82 (UC) | VN, LE | LY | 2.63 ± 0.08 a | PD |
| Manduria (Ma) | mLD | R | 0.85 ± 0.02 e | R |
| Regina (Re) | VN, LE | LY | 1.85 ± 0.01 c | PD |
| Sw5-Messapico (Me) | VN, LE | LY | 1.91 ± 0.01 c | PD |
| Sw5-Faino (Fa) | VN, LE | LY | 2.0 ± 0.03 bc | PD |
| Pullrex (Pu) | LY | D | 2.1 ± 0.04 b | PD |
| Molfettese (Mo) | mMos | R | 0.78 ± 0.02 e | R |
| Solanum integrifolium | mMos | R | 0.87 ± 0.03 e | R |
| S. nigrum | sMos, sLD | N, sLD, LE | 1.9 ± 0.09 c | N |
| S. torvum | mMos | mMos | 1.25 ± 0.06 d | mMos |
| Grafted Plants *** | Symptoms | Symptoms | Viral RNA Load ** | Symptoms |
| UC/Mo | Mos | R | 0.19 ± 0.02 c,d,e | R |
| UC/S. integrifolium | sMos, LE | sLD, LY | 0.35 ± 0.06 a | N |
| UC/Ma | Mos | R | 0.10 ± 0.06 e,f,g | R |
| UC/UC | mMos | R | 0.04 ± 0.008 f,g | R |
| Ma/Ma | mMos | R | 0.01 ± 0.003 g | R |
| Me/Mo | sMos, | sMos, LY | 0.30 ± 0.07 a,b,c,d | LY, sLD |
| Me/S. integrifolium | sMos, | sMos, LY | 0.31 ± 0.07 a,b,c | LY, sLD |
| Me/Ma | mMos | R | 0.01 ± 0.004 g | R |
| Me/Me | mMos | R | 0.02 ± 0.006 g | R |
| Fa/Mo | Mos | Mos | 0.22 ± 0.04 b,c,d,e | R |
| Fa/S. integrifolium | LE, VN | LY, VN | 0.38 ± 0.04 a | N |
| Fa/Ma | mMos | R | 0.12 ± 0.04 e,f,g | R |
| Fa/Fa | mMos | R | 0.12 ± 0.06 e,f,g | R |
| Pu/Mo | sMos | sLD, VN | 0.34 ± 0.02 a,b | N |
| Pu/S. integrifolium | LE, VN | LY, VN | 0.36 ± 0.03 a | SLD, VN |
| Pu/Ma | Mos | LE, VN | 0.18 ± 0.04 d,e | LY, VN |
| Pu/Pu | Mos | R | 0.16 ± 0.03 e,f | R |
| CMV-TTS * | 15 *** | 21 *** | 21 *** | 30 *** |
| Plants | Symptoms | Symptoms | Viral RNA Load † | Symptoms |
| UC | TS, LD | sTS | 0.16 ± 0.008 b | sTS |
| Ma | TS, LD | R | 0.25 ± 0.001 a,b | R |
| UC/Ma § | TS | R | 0.33 ± 0.021 a | R |
| UC/UC § | TS | R | 0.31 ± 0.025 a | R |
| Ma/Ma § | TS | R | 0.3 ± 0.007 a | R |
| CMV-77 ** | 15 *** | 15 *** | 21 *** | 30 *** |
| Plants | Symptoms | Viral RNA Load † | Symptoms | Symptoms |
| UC | SN, LN | 0.04 ± 0.001 c | PD | PD |
| Ma | mS | 0.33 ± 0.025 a,b | tR | PD |
| UC/Ma § | SN, LN | 0.24 ± 0.07 b | SN, LN ¥ | SN, LN |
| UC/UC § | SN, LN | 0.43 ± 0.06 a | SN, LN | SN, LN |
| Ma/Ma § | YBL | 0.03 ± 0.001 c | R | R |
| 14 * | 14 * | 21 * | 21 * | |
|---|---|---|---|---|
| Plants | Symptoms | Viral RNA Load *** | Symptoms | Viral RNA Load *** |
| UC | Mos, LD | 5.72 ± 1.34 b | sMos, sLD | 16.07 ± 1.96 a |
| Ma | mLD | 8.03 ± 0.93 b | R | 2.22 ± 0.68 c |
| UC/Ma ** | mS | 17.92 ± 1.84 a | R | 4.69 ± 1.16 bc |
| UC/UC ** | A | 13.68 ± 2.4 a | A | 6.03 ± 0.44 b |
| Ma/Ma ** | mLD | 16.07 ± 1.96 a | R | 2.39 ± 1.30 c |
| Mock | Infected | |||||
|---|---|---|---|---|---|---|
| Locus Name | Ma/Ma * | UC/UC * | UC/Ma * | Ma/Ma * | UC/UC * | UC/Ma * |
| Solyc11g072600.1 (RAP2/ERF4) *** | 3.20 ** | 3.63 | ||||
| Solyc04g080700.2 (wound-responsive family protein) | 3.57 | 3.26 | 2.55 | 2.51 | 2.86 | 2.61 |
| Solyc07g056000.2 (XTH) | 1.74 | 2.55 | ||||
| Solyc07g052980.2 (XTH) | 1.27 | 2.24 | ||||
| Solyc02g091920.2 (XTH) | −1.58 | −1.94 | −1.61 | −1.44 | ||
| Solyc09g075210.2 (AtLEA5/SAGS21) | −3.29 | −4.33 | −3.41 | −4.52 | −1.93 | −2.13 |
| Solyc06g006000.2 (MEE59) | 5.03 | 5.51 | 5.49 | 3.75 | 4.23 | 4.25 |
| Solyc01g108910.2 (MEE14) | 5.41 | 5.03 | 5.00 | 5.00 | 4.54 | 4.64 |
| Solyc01g095140.2 (LEA14, LSR3) | 2.30 | 3.42 | 2.43 | 2.21 | ||
| Locus Name | GO * | Mock | Infected | ||||
|---|---|---|---|---|---|---|---|
| Ma/Ma ** | UC/UC | UC/Ma | Ma/Ma | UC/UC | UC/Ma | ||
| Genes Involved in ROS Production | |||||||
| Solyc03g117980.2 (RbohD) *** | GO:0004601 | 2.79 † | 1.51 | 1.56 | 2.97 | 2.17 | 2.21 |
| Genes Involved in ROS Scavenging | |||||||
| Solyc01g006300.2 (POD2) | GO:0004601 GO:0006979 GO:0042744 | 3.38 | 2.45 | 2.54 | |||
| Solyc11g018800.1 (POD) | GO:0004601 GO:0006979 GO:0042744 | 4.21 | 3.10 | 2.89 | 4.05 | 4.04 | 4.18 |
| Solyc02g082760.2 (CAT2) | GO:0004096 GO:0006979 GO:0042744 | −1.76 | −2.05 | −2.22 | −1.69 | ||
| Solyc12g094620.1 (CAT2) | GO:0004096 GO:0042542 GO:0042744 | 3.88 | 2.59 | 2.05 | 1.95 | 2.12 | |
| Solyc04g025650.2 (MO1) | GO:0004497 | 1.69 | 2.13 | 2.07 | 3.32 | 3.22 | 3.12 |
| Solyc06g049080.2 (MSD1) | GO:0004784 | −1.51 | −1.48 | −1.49 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spanò, R.; Ferrara, M.; Gallitelli, D.; Mascia, T. The Role of Grafting in the Resistance of Tomato to Viruses. Plants 2020, 9, 1042. https://doi.org/10.3390/plants9081042
Spanò R, Ferrara M, Gallitelli D, Mascia T. The Role of Grafting in the Resistance of Tomato to Viruses. Plants. 2020; 9(8):1042. https://doi.org/10.3390/plants9081042
Chicago/Turabian StyleSpanò, Roberta, Massimo Ferrara, Donato Gallitelli, and Tiziana Mascia. 2020. "The Role of Grafting in the Resistance of Tomato to Viruses" Plants 9, no. 8: 1042. https://doi.org/10.3390/plants9081042
APA StyleSpanò, R., Ferrara, M., Gallitelli, D., & Mascia, T. (2020). The Role of Grafting in the Resistance of Tomato to Viruses. Plants, 9(8), 1042. https://doi.org/10.3390/plants9081042