The Ethylene Biosynthesis Genes ACS2 and ACS6 Modulate Disease Severity of Verticillium dahliae
Abstract
1. Introduction
2. Results
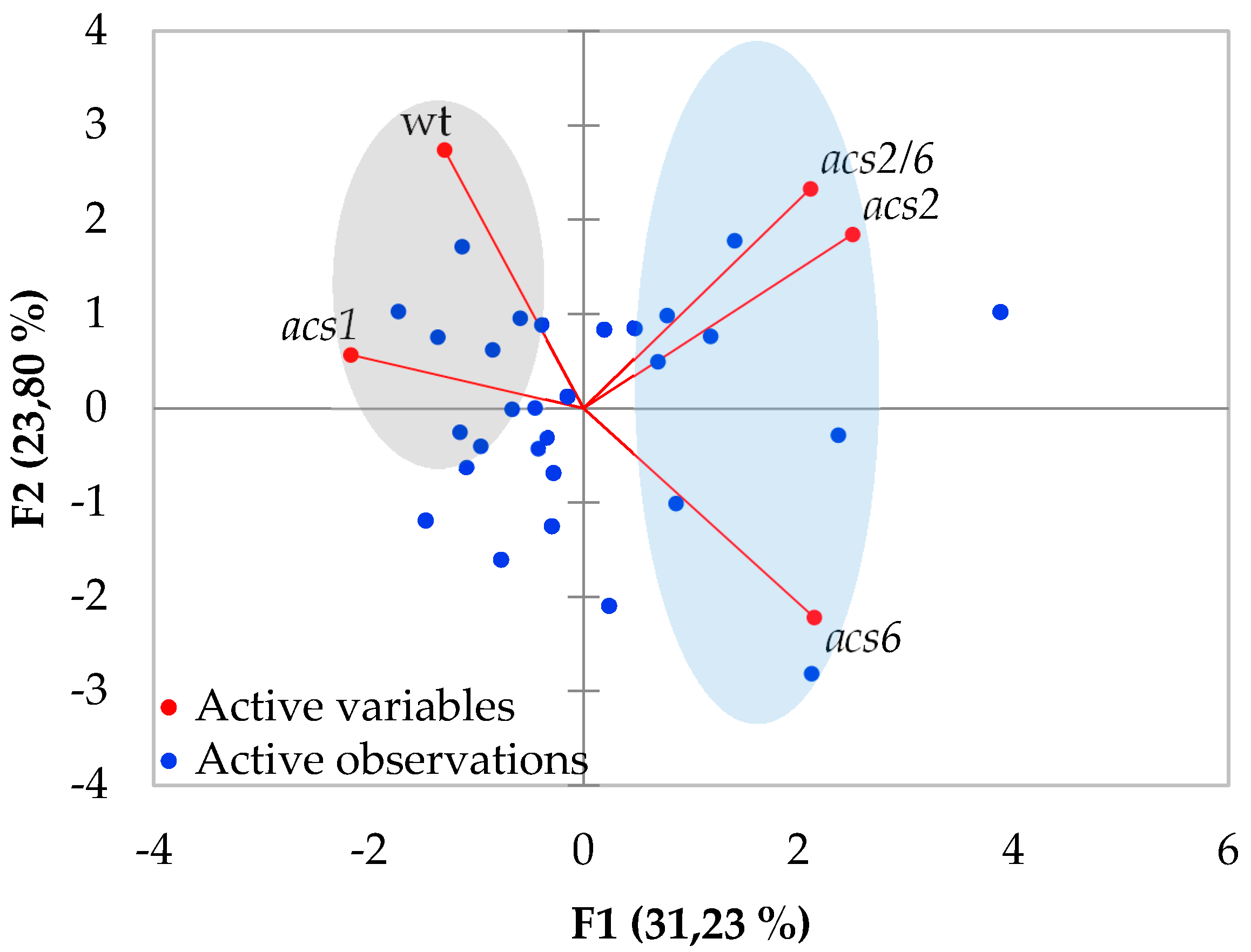
2.1. Verticillium Wilt Symptom Development
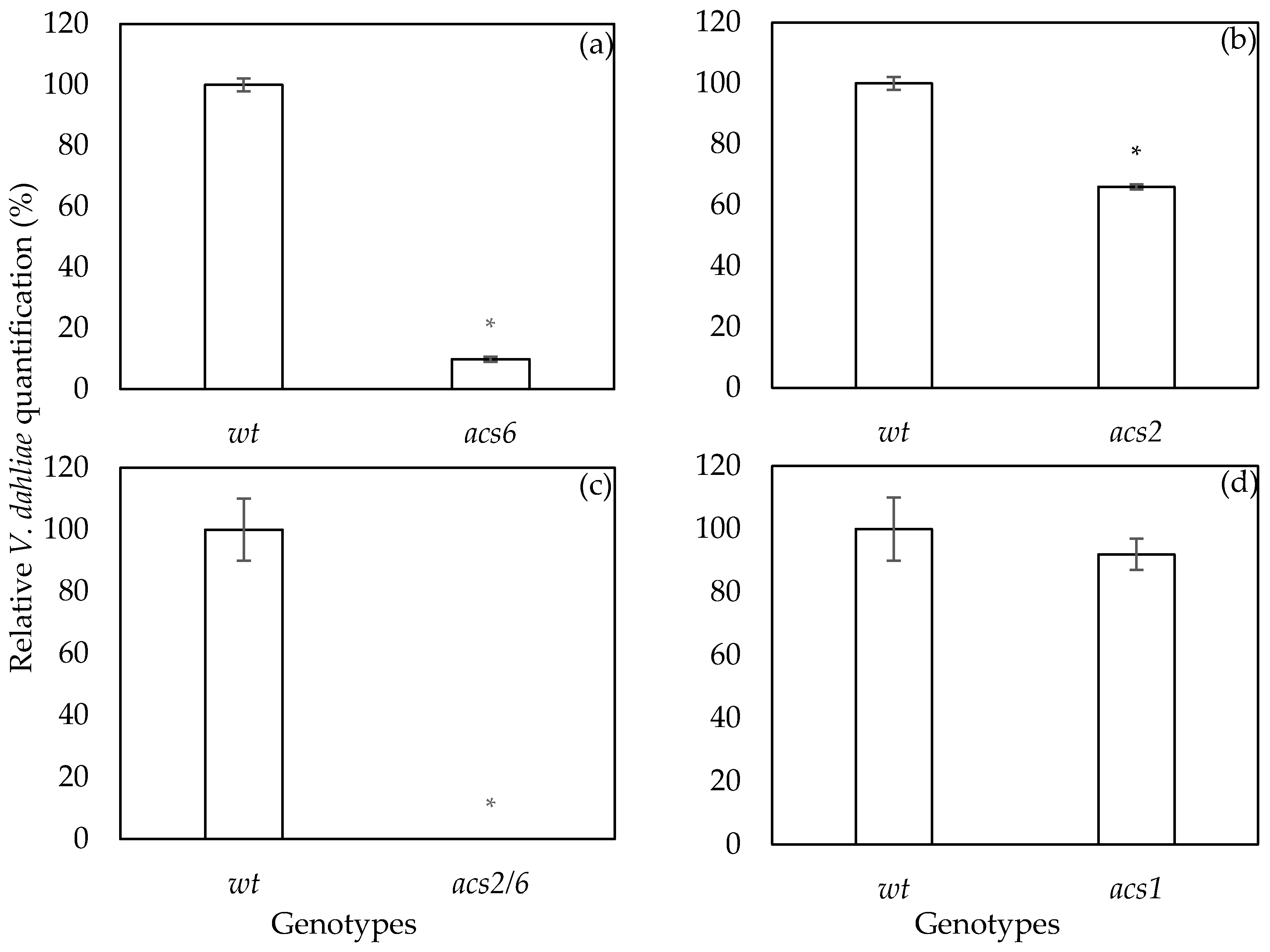
2.2. Verticillium dahliae DNA qPCR Quantification
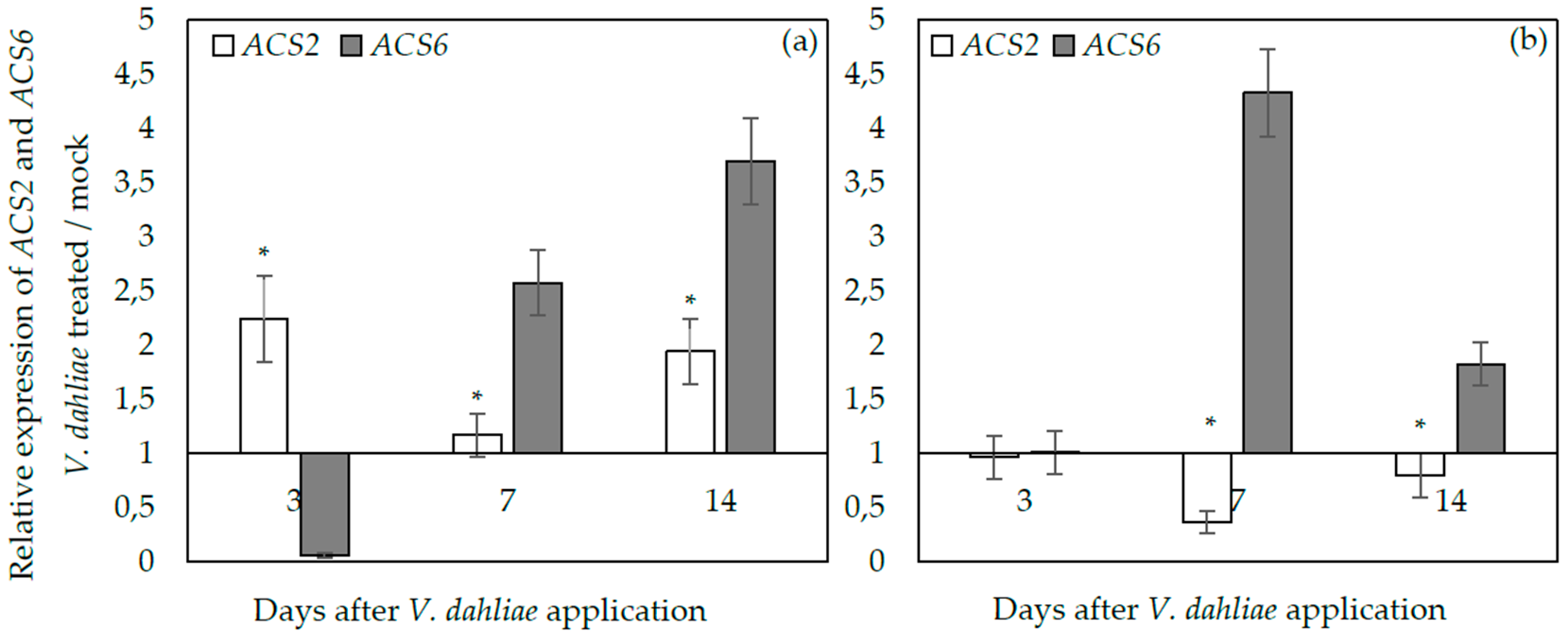
2.3. ACS2 and ACS6 Expression upon Verticillium dahliae Plant Infection
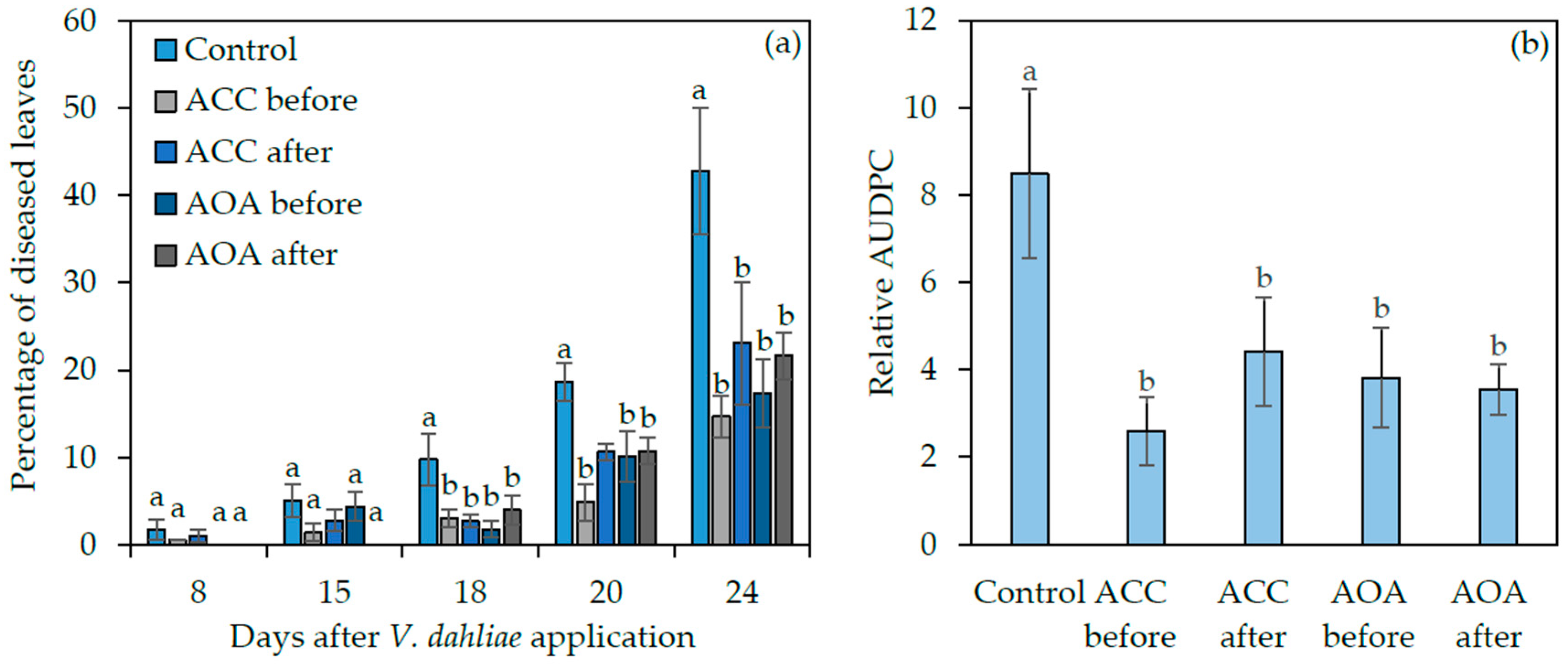
2.4. Exogenous ACC and AOA Application to wt Arabidopsis thaliana before or after Verticillium dahliae Inoculation
3. Discussion
4. Materials and Methods
4.1. Fungal Culture
4.2. Seeds Origin and Plant Growth Conditions
4.3. Pathogenicity Experiments
4.4. DNA Extraction and qPCR Fungal Quantification
4.5. Determination of Transcript Levels Using RT-PCR Assay
4.6. Statistics
Author Contributions
Funding
Conflicts of Interest
References
- Pegg, G.F.; Brady, B.L. Verticillium Wilts; CABI Publishing: New York, NY, USA, 2002. [Google Scholar]
- Schnathorst, W.C. Life cycle and epidemiology of Verticillium. In Fungal Wilt Diseases of Plants; Mace, M.A., Bell, A.A., Beckman, C.H., Eds.; Academic Press: New York, NY, USA, 1981; pp. 81–111. [Google Scholar]
- Abeles, F.B.; Morgan, P.W.; Saltveit, M.E., Jr. Ethylene in Plant Biology, 2nd ed.; Academic Press: San Diego, CA, USA, 1992. [Google Scholar]
- Johnson, P.R.; Ecker, J.R. The ethylene gas signal transduction pathway: A molecular perspective. Annu. Rev. Genet. 1998, 32, 227–254. [Google Scholar] [CrossRef]
- Pantelides, I.S.; Tjamos, S.E.; Paplomatas, E.J. Ethylene perception via ETR1 is required in Arabidopsis infection by Verticillium dahliae. Mol. Plant Pathol. 2010, 11, 191–202. [Google Scholar] [CrossRef]
- Johansson, A.; Staal, J.; Dixelius, C. Early responses in the Arabidopsis—Verticillium longisporum pathosystem are dependent on NDR1, JA- and ET-associated signals via cytosolic NPR-1 and RFO1. Mol. Plant Microbe-Interact. 2006, 19, 958–969. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Ecker, J.R. The ethylene signaling pathway: New insights. Curr. Opin. Plant Biol. 2004, 7, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Kende, H. Ethylene biosynthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1993, 44, 283–307. [Google Scholar] [CrossRef]
- Wang, K.L.; Li, H.; Ecker, J.R. Ethylene biosynthesis and signaling networks. Plant Cell 2002, 14 (Suppl. 1), S131–S151. [Google Scholar] [CrossRef]
- Tsuchisaka, A.; Yu, G.; Jin, H.; Alonso, J.M.; Ecker, J.R.; Zhang, X.; Gao, S.; Theologis, A. A combinatorial interplay among the 1-aminocyclopropane-1-carboxylate isoforms regulates ethylene biosynthesis in Arabidopsis thaliana. Genetics 2009, 183, 979–1003. [Google Scholar] [CrossRef]
- Bleecker, A.B.; Kende, H. Ethylene: A Gaseous Signal Molecule in Plants. Annu. Rev. Cell Dev. Biol. 2000, 16, 1–18. [Google Scholar] [CrossRef]
- Guan, R.; Su, J.; Meng, X.; Li, S.; Liu, Y.; Xu, J.; Zhang, S. Multilayered regulation of ethylene induction plays a positive role in Arabidopsis resistance against Pseudomonas syringae. Plant Physiol. 2015, 169, 299–312. [Google Scholar] [CrossRef]
- Han, L.; Li, G.J.; Yang, K.Y.; Mao, G.; Wang, R.; Liu, Y.; Zhang, S. Mitogen-activated protein kinase 3 and 6 regulate Botrytis cinerea-induced ethylene production in Arabidopsis. Plant J. 2010, 64, 114–127. [Google Scholar] [CrossRef]
- Li, G.; Meng, X.; Wang, R.; Mao, G.; Han, L.; Liu, Y.; Zhang, S. Dual-level regulation of ACC synthase activity by MPK3/MPK6 cascade and its downstream WRKY transcription factor during ethylene induction in Arabidopsis. PLoS Genet. 2012, 8, e1002767. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.B.; Adams, D.O.; Yang, S.F. 1-Aminocyclopropanecarboxylate synthase, a key enzyme in ethylene biosynthesis. Arch. Biochem. Biophys. 1979, 198, 280–286. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, S. Phosphorylation of 1-aminocyclopropane-1-carboxylic acid synthase by MPK6, a stress-responsive mitogen-activated protein kinase, induces ethylene biosynthesis in Arabidopsis. Plant Cell 2004, 16, 3386–3399. [Google Scholar] [CrossRef]
- Gkizi, D.; Lehmann, S.; L’Haridon, F.; Serrano, M.; Paplomatas, E.J.; Métraux, J.P.; Tjamos, S.E. The innate immune signaling system as a regulator of disease resistance and induced systemic resistance activity against Verticillium dahliae. Mol. Plant Microbe Interact. 2016, 29, 313–323. [Google Scholar] [CrossRef]
- Liang, X.; Oono, Y.; Shen, N.F.; Köhler, C.; Li, K.; Scolnik, P.A.; Theologis, A. Characterization of two members (ACS1 and ACS3) of the 1-aminocyclopropane-1-carboxylate synthase gene family of Arabidopsis thaliana. Gene 1995, 167, 17–24. [Google Scholar] [CrossRef]
- Veronese, P.; Narasimhan, M.L.; Stevenson, R.A.; Zhu, J.K.; Weller, S.C.; Subbarao, K.V.; Bressan, R.A. Identification of a locus controlling Verticillium disease symptom response in Arabidopsis thaliana. Plant J. 2003, 35, 574–587. [Google Scholar] [CrossRef]
- Paula, X.; Kover, P.X.; Schaal, B.A. Genetic variation for disease resistance and tolerance among Arabidopsis thaliana accessions. Proc. Natl. Acad. Sci. USA 2002, 20, 11270–11274. [Google Scholar]
- Denby, K.J.; Kumar, P.; Kliebenstein, D.J. Identification of Botrytis cinerea susceptibility loci in Arabidopsis thaliana. Plant J. 2004, 38, 473–486. [Google Scholar] [CrossRef]
- Poland, J.A.; Balint-Kurti, P.J.; Wisser, R.J.; Pratt, R.C.; Nelson, R.J. Shades of gray: The world of quantitative disease resistance. Trends Plant Sci. 2009, 14, 21–29. [Google Scholar] [CrossRef]
- Van Loon, L.C.; Geraats, B.P.J.; Linthorst, H.J.M. Ethylene as a modulator of disease resistance in plants. Trends Plant Sci. 2006, 11, 184–191. [Google Scholar] [CrossRef]
- Robison, M.M.; Griffith, M.; Pauls, K.P.; Glick, B.R. Dual role for ethylene in susceptibility of tomato to Verticillium wilt. J. Phytopathol. 2001, 149, 385–388. [Google Scholar] [CrossRef]
- Tsolakidou, M.D.; Pantelides, L.S.; Tzima, A.K.; Kang, S.; Paplomatas, E.J.; Tsaltas, D. Disruption and overexpression of the gene encoding acc (1-aminocyclopropane-1-carboxylic acid) deaminase in soil-borne fungal pathogen Verticillium dahliae revealed the role of ACC as a potential regulator of virulence and plant defense. Mol. Plant Microbe Interact. 2019, 32, 639–653. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Gomez, L.; Boller, T. FLS2: An LRR receptor-like kinase involved in the perception of the bacterial elicitor flagellin in Arabidopsis. Mol. Cell 2000, 5, 1003–1011. [Google Scholar] [CrossRef]
- Zipfel, C.; Kunze, G.; Chinchilla, D.; Caniard, A.; Jones, J.D.G.; Boller, T.; Felix, G. Perception of the bacterial PAMP EF-Tu by the receptor EFR restricts Agrobacterium-mediated transformation. Cell 2006, 125, 749–760. [Google Scholar] [CrossRef] [PubMed]
- Tintor, N.; Ross, A.; Kanehara, K.; Yamada, K.; Fan, L.; Kemmerling, B.; Nürnberger, T.; Tsuda, K.; Saijo, Y. Layered pattern receptor signaling via ethylene and endogenous elicitor peptides during Arabidopsis immunity to bacterial infection. Proc. Natl. Acad. Sci. USA 2013, 110, 6211–6216. [Google Scholar] [CrossRef] [PubMed]
- Robb, J.; Lee, B.; Nazar, R.N. Gene suppression in a tolerant tomato–vascular pathogen interaction. Planta 2007, 226, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Tjamos, S.E.; Flemetakis, E.; Paplomatas, E.J.; Katinakis, P. Induction of resistance to Verticillium dahliae in Arabidopsis thaliana by the biocontrol agent K165 and pathogenesis-related proteins gene expression. Mol. Plant Microbe Interact. 2005, 18, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Maniatis, T.; Fritsch, E.F.; Sambrook, J. Molecular Cloning, a Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1982. [Google Scholar]
- Sinha, A.K.; Wood, R.K.S. Studies on the nature of resistance in tomato plants to Verticillium albo-atrum. Ann. Appl. Biol. 1968, 62, 319–327. [Google Scholar] [CrossRef]
- Campbell, C.L.; Madden, L.V. Introduction to Plant Disease Epidemiology; Wiley: New York, NY, USA, 1990. [Google Scholar]
- Dellaporta, S.L.; Wood, J.; Hicks, J.B. A plant DNA minipreparation, version II. Plant Mol. Biol. Rep. 1983, 1, 19–21. [Google Scholar] [CrossRef]
- Czechowski, T.; Stitt, M.; Altmann, T.; Udvardi, M.K.; Scheible, W.R. Genome wide identification and testing of superior reference genes for transcript normalization in Arabidopsis. Plant Physiol. 2005, 139, 5–17. [Google Scholar] [CrossRef]
- Ramakers, C.; Ruijter, J.M.; Deprez, R.H.; Moorman, A.F. Assumption free analysis of quantitative real-time polymerase chain reaction (PCR) data. Neurosci. Lett. 2003, 339, 62–66. [Google Scholar] [CrossRef]

| ACS2 | ACS6 | |||
|---|---|---|---|---|
| Source | df | F | df | F |
| Tissue | 1 | 413.06 ** | 1 | 2.53 |
| Time | 2 | 90.09 ** | 2 | 118.14 ** |
| Tissue × Time | 2 | 7.29 * | 2 | 46.80 ** |
| Error | 48 | 48 | ||
| Total | 54 | 54 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poulaki, E.G.; Tsolakidou, M.-D.; Gkizi, D.; Pantelides, I.S.; Tjamos, S.E. The Ethylene Biosynthesis Genes ACS2 and ACS6 Modulate Disease Severity of Verticillium dahliae. Plants 2020, 9, 907. https://doi.org/10.3390/plants9070907
Poulaki EG, Tsolakidou M-D, Gkizi D, Pantelides IS, Tjamos SE. The Ethylene Biosynthesis Genes ACS2 and ACS6 Modulate Disease Severity of Verticillium dahliae. Plants. 2020; 9(7):907. https://doi.org/10.3390/plants9070907
Chicago/Turabian StylePoulaki, Eirini G., Maria-Dimitra Tsolakidou, Danai Gkizi, Iakovos S. Pantelides, and Sotirios E. Tjamos. 2020. "The Ethylene Biosynthesis Genes ACS2 and ACS6 Modulate Disease Severity of Verticillium dahliae" Plants 9, no. 7: 907. https://doi.org/10.3390/plants9070907
APA StylePoulaki, E. G., Tsolakidou, M.-D., Gkizi, D., Pantelides, I. S., & Tjamos, S. E. (2020). The Ethylene Biosynthesis Genes ACS2 and ACS6 Modulate Disease Severity of Verticillium dahliae. Plants, 9(7), 907. https://doi.org/10.3390/plants9070907








