The Essential Oil Composition and Antimicrobial Activity of Liquidambar formosana Oleoresin
Abstract
1. Introduction
2. Results and Discussion
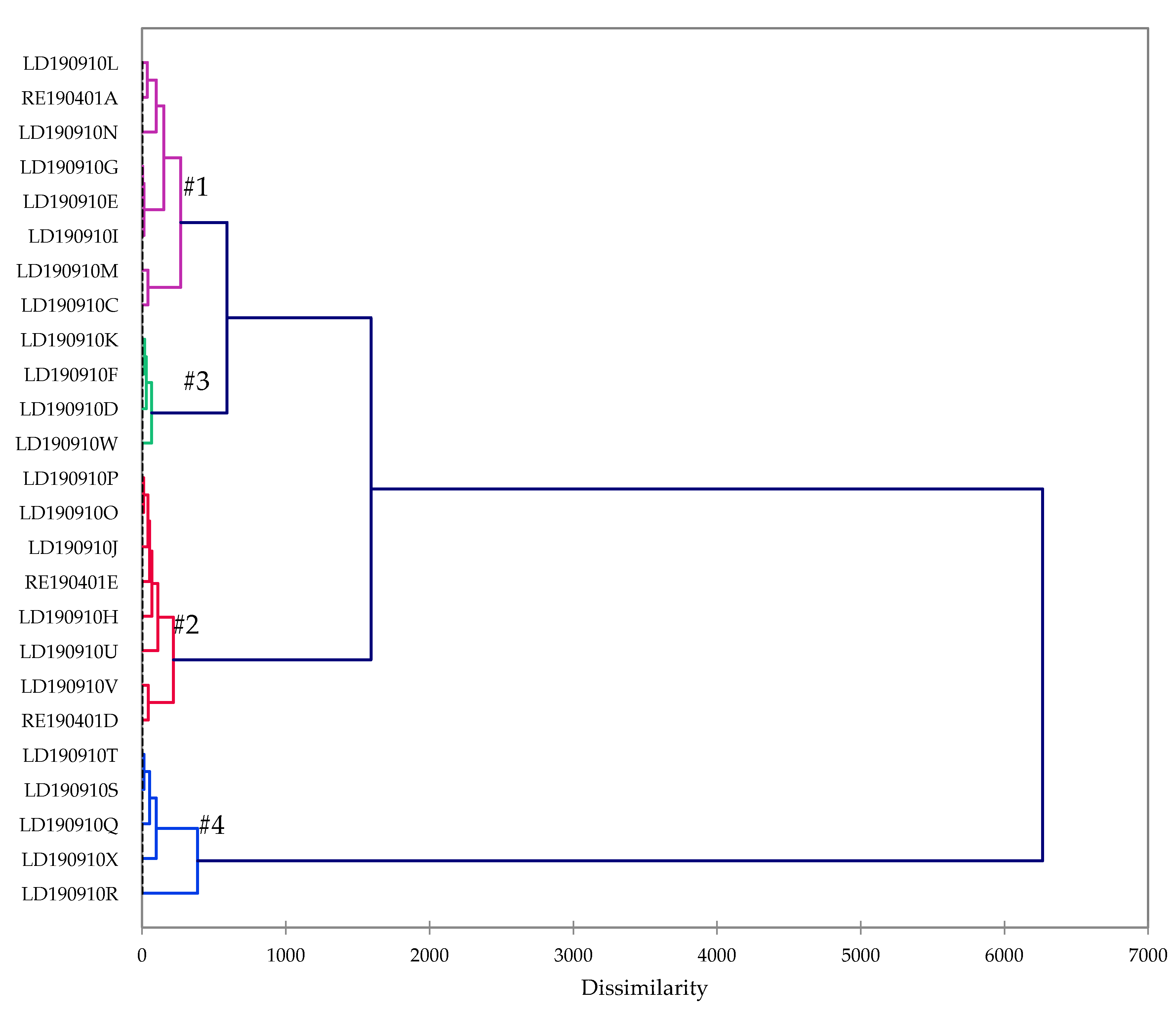
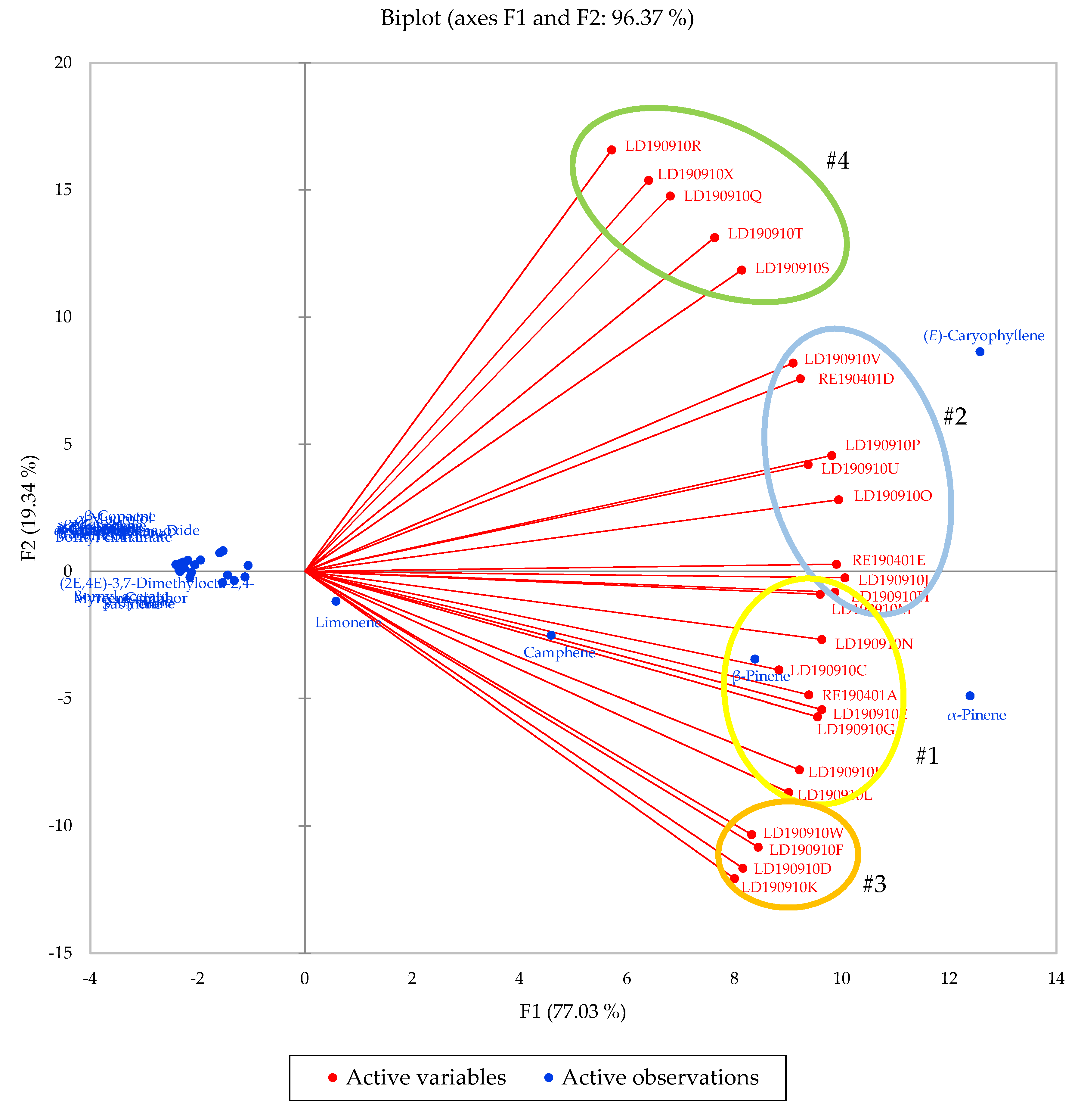
2.1. Essential Oil Chemical Composition
2.2. Antibacterial and Antifungal Activity
3. Materials and Methods
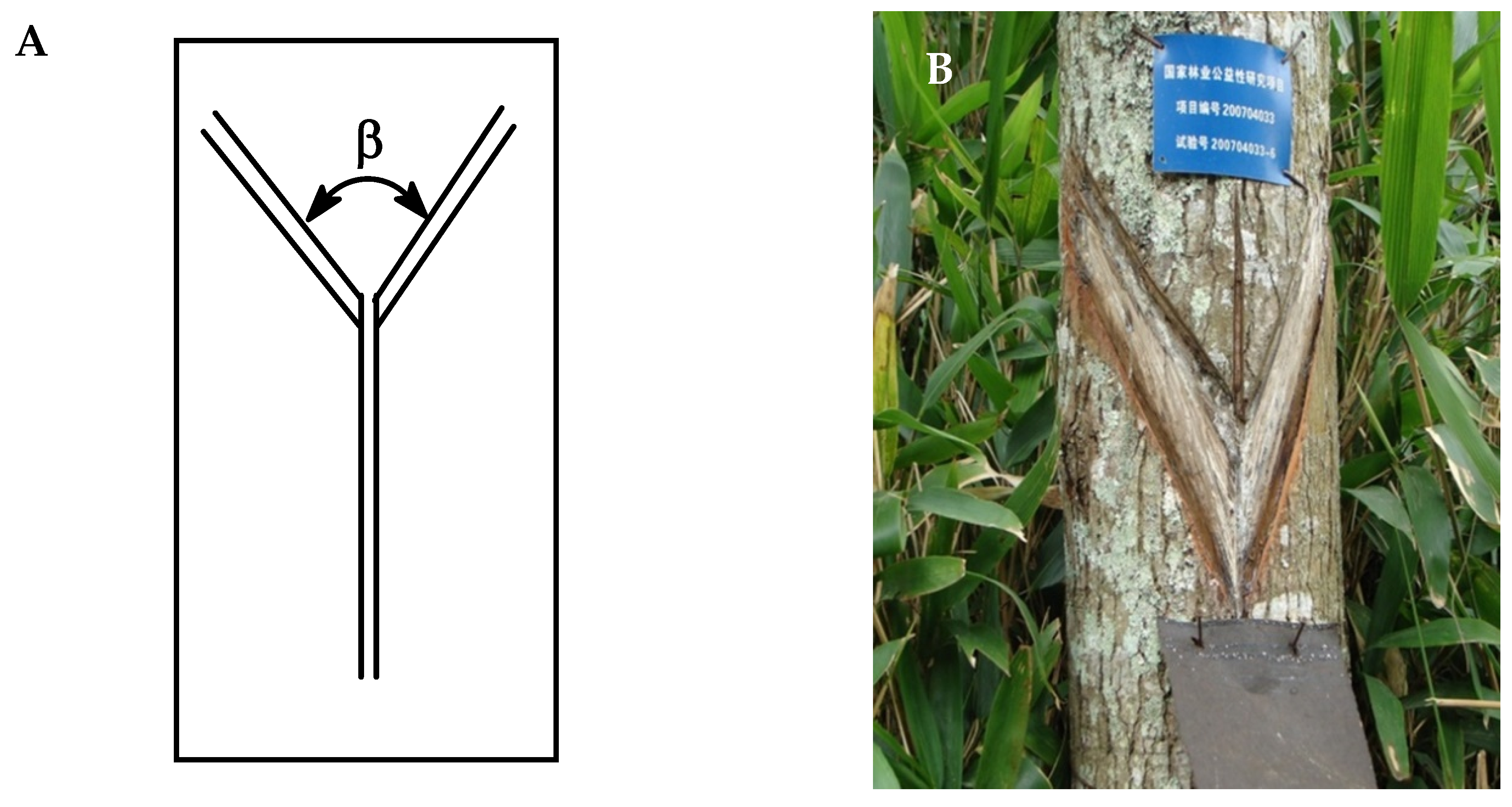
3.1. Oleoresin Collection
3.2. Oleoresin Hydrodistillation
3.3. Gas Chromatographic–Mass Spectral Analysis
3.4. Gas Chromatographic–Flame Ionization Detection
3.5. Chiral Gas Chromatography–Mass Spectrometry
3.6. Antimicrobial Screening
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hatano, T.; Kira, R.; Yoshizaki, M.; Okuda, T. Seasonal changes in the tannins of Liquidambar formosana reflecting their biogenesis. Phytochemistry 1986, 25, 2787–2789. [Google Scholar] [CrossRef]
- Yoshizaki, M.; Shingu, T.; Okuda, T.; Hatano, T.; Kaneda, T. Liquidambin, an ellagitannin from Liquidambar formosana. Phytochemistry 1987, 26, 2053–2055. [Google Scholar] [CrossRef]
- Feng, Y.; Liu, S.; Liu, G.; Yao, J. Facile and fast removal of oil through porous carbon spheres derived from the fruit of Liquidambar formosana. Chemosphere 2017, 170, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, X.; Yi, S.; Lu, H.; Wu, S.; Zhao, H. Liquidambar formosana Hance: A mini-review of chemical constituents and pharmacology. Eur. J. Med. Plants 2016, 17, 1–11. [Google Scholar] [CrossRef]
- Ma, H.; Wang, F.; Jiang, J.; Cheng, L.; Zhang, H.; Zhang, G. In vivo anti-inflammatory activity of Liquidambar formosana Hance infructescence extract. Trop. J. Pharm. Res. 2017, 16, 2403–2410. [Google Scholar]
- Dictionary of Natural Products on DVD, version 28.2; CRC Press: Boca Raton, FL, USA, 2019.
- Su, Y.C.; Ho, C.L. Composition, in vitro cytotoxicity, anti-mildew and anti-wood-decay fungal activities of the fruit essential oil of Liquidambar formosana from Taiwan. Nat. Prod. Commun. 2017, 12, 287–290. [Google Scholar] [CrossRef]
- Hatano, T.; Kira, R.; Yasuhara, T.; Okuda, T. Tannins of hamamelidaceous plants. III. Isorugosins A, B and D, new ellagitannins from Liquidambar formosana. Chem. Pharm. Bull. 1988, 36, 3920–3927. [Google Scholar] [CrossRef]
- Wang, K.; Pan, Y.; Wang, H.; Zhang, Y.; Lei, Q.; Zhu, Z.; Li, H.; Liang, M. Antioxidant activities of Liquidambar formosana Hance leaf extracts. Med. Chem. Res. 2010, 19, 166–176. [Google Scholar] [CrossRef]
- Zhang, L.; Zhu, M.F.; Tu, Z.C.; Zhao, Y.; Wang, H.; Li, G.J.; Wang, H.; Sha, X.M. α-Glucosidase inhibition, anti-glycation and antioxidant activities of Liquidambar formosana Hance leaf, and identification of phytochemical profile. South Afr. J. Bot. 2017, 113, 239–247. [Google Scholar] [CrossRef]
- Chen, C.-J.; Chu, F.-H.; Chien, S.-C.; Tsao, N.-W.; Wang, S.-Y. Comparative analysis of phytoncides released frm Liqudambar formosana Hance trees and seedlings. J. Agric. For. 2013, 62, 137–144. [Google Scholar]
- Hua, K.-F.; Yang, T.-J.; Chiu, H.-W.; Ho, C.-L. Essential oil from leaves of Liquidambar formosana ameliorates inflammatory response in lipopolysaccharide-activated mouse macrophages. Nat. Prod. Commun. 2014, 9, 869–872. [Google Scholar] [CrossRef] [PubMed]
- Shang, H.-J.; Li, D.-Y.; Wang, W.-J.; Li, Z.-L.; Hua, H.-M. Three new diterpenoids from the resin of Liquidambar formosana. Nat. Prod. Res. 2014, 28, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.-Y.; Chen, J.-H.; Zhou, G.-S.; Tang, Y.-P.; Duan, J.-A.; Tian, L.-J.; Liu, X.-H. Pentacyclic triterpenes from the resin of Liquidambar formosana. Fitoterapia 2011, 82, 927–931. [Google Scholar] [CrossRef] [PubMed]
- Chien, S.-C.; Xiao, J.-H.; Tseng, Y.-H.; Kuo, Y.-H.; Wang, S.-Y. Composition and antifungal activity of balsam from Liquidambar formosana Hance. Holzforschung 2013, 67, 345–351. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, H.; Zhu, P.; Wu, X.; Yao, H.; Ye, W.; Jiang, J.; Xu, J. Synthesis and biological evaluation of ambradiolic acid as an inhibitor of glycogen phosphorylase. Fitoterapia 2015, 100, 50–55. [Google Scholar] [CrossRef]
- Dat, N.T.; Lee, I.S.; Cai, X.F.; Shen, G.; Kim, Y.H. Oleanane triterpenoids with inhibitory activity against NFAT transcription factor from Liquidambar formosana. Biol. Pharm. Bull. 2004, 27, 426–428. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, W.; Zeng, T.; Shen, X. Toxicity and antibacterial activity of Chinese sweetgum resin. Nat. Prod. Res. Dev. 2015, 27, 1651–1656. [Google Scholar]
- Song, X.; Zeng, T. Analysis on chemical components of essential oil of Chinese sweetgum resin. Chem. Ind. For. Prod. 2010, 30, 40–44. [Google Scholar]
- Yi, L.P.; Wen, S.Z.; Wang, Z.Z.; Yang, L.L. Biomass and productivity of Liquidambar formosana Hance. J. Cent. South Univ. For. Technol. 2018, 2, 50–53. [Google Scholar]
- Hunter, I.; von Hahn, C.-G.; Zhu, Z.; Zhou, Y. Stabilising forest margins by growing non-timber forest products: A novel example from Hainan Island, China. Land Use Policy 2003, 20, 225–230. [Google Scholar] [CrossRef]
- Zeng, T.; Song, X.; Zeng, C.; Luo, J.; Yang, X. Method for Collecting Resin of Sweetgum 2012. China patent CN 102090300A, 4 January 2012. [Google Scholar]
- Liu, H.; Chen, M.Y. Studies on the chemical constituents of Liquidambar formosana resin. Chem. Ind. For. Prod. 1995, 15, 61–66. [Google Scholar]
- Sartoratto, A.; Machado, A.L.M.; Delarmelina, C.; Figueira, G.M.; Duarte, M.C.T.; Rehder, V.L.G. Composition and antimicrobial activity of essential oils from aromatic plants used in Brazil. Braz. J. Microbiol. 2004, 35, 275–280. [Google Scholar] [CrossRef]
- Fisher, K.; Phillips, C. Potential antimicrobial uses of essential oils in food: Is citrus the answer? Trends Food Sci. Technol. 2008, 19, 156–164. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Smith-Palmer, A.; Stewart, J.; Fyfe, L. Antimicrobial properties of plant essential oils and essences against five important food-borne pathogens. Lett. Appl. Microbiol. 1998, 26, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Izadi, Z.; Esna-Ashari, M.; Piri, K.; Davoodi, P. Chemical composition and antimicrobial activity of feverfew (Tanacetum parthenium) essential oil. Int. J. Agric. Biol. 2010, 12, 759–763. [Google Scholar]
- Djihane, B.; Wafa, N.; Elkhamssa, S.; Pedro, D.H.J.; Maria, A.E.; Mohamed Mihoub, Z. Chemical constituents of Helichrysum italicum (Roth) G. Don essential oil and their antimicrobial activity against Gram-positive and Gram-negative bacteria, filamentous fungi and Candida albicans. Saudi Pharm. J. 2017, 25, 780–787. [Google Scholar] [CrossRef]
- Inouye, S.; Takizawa, T.; Yamaguchi, H. Antibacterial activity of essential oils and their major constituents against respiratory tract pathogens by gaseous contact. J. Antimicrob. Chemother. 2001, 47, 565–573. [Google Scholar] [CrossRef]
- Herman, A.; Tambor, K.; Herman, A. Linalool affects the antimicrobial efficacy of essential oils. Curr. Microbiol. 2016, 72, 165–172. [Google Scholar] [CrossRef]
- Requena, R.; Vargas, M.; Chiralt, A. Study of the potential synergistic antibacterial activity of essential oil components using the thiazolyl blue tetrazolium bromide (MTT) assay. LWT Food Sci. Technol. 2019, 101, 183–190. [Google Scholar] [CrossRef]
- Crevelin, E.J.; Caixeta, S.C.; Dias, H.J.; Groppo, M.; Cunha, W.R.; Martins, C.H.G.; Crotti, A.E.M. Antimicrobial activity of the essential oil of Plectranthus neochilus against cariogenic bacteria. Evid. Based Complement. Altern. Med. 2015, 2015, 102317. [Google Scholar] [CrossRef] [PubMed]
- DeCarlo, A.; Johnson, S.; Okeke-Agulu, K.I.; Dosoky, N.S.; Wax, S.J.; Owolabi, M.S.; Setzer, W.N. Compositional analysis of the essential oil of Boswellia dalzielii frankincense from West Africa reveals two major chemotypes. Phytochemistry 2019, 164, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007. [Google Scholar]
- Mondello, L. FFNSC 3; Shimadzu Scientific Instruments: Columbia, MD, USA, 2016. [Google Scholar]
- National Institute of Standards and Technology. NIST17; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2017.
- Satyal, P. Development of GC-MS Database of Essential Oil Components by the Analysis of Natural Essential Oils and Synthetic Compounds and Discovery of Biologically Active Novel Chemotypes in Essential Oils. Ph.D. Dissertation, University of Alabama in Huntsville, Huntsville, AL, USA, 2015. [Google Scholar]
- Sahm, D.H.; Washington, J.A. Antibacterial susceptibility tests: Dilution methods. In Manual of Clinical Microbiology; Balows, A., Hausler, W.J., Herrmann, K.L., Isenberg, H.D., Shamody, H.J., Eds.; American Society for Microbiology: Washington, DC, USA, 1991. [Google Scholar]
- EUCAST. Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution. Clin. Microbiol. Infect. 2003, 9, ix–xv. [Google Scholar] [CrossRef]
- Setzer, M.C.; Setzer, W.N.; Jackes, B.R.; Gentry, G.A.; Moriarity, D.M. The medicinal value of tropical rainforest plants from Paluma, North Queensland, Australia. Pharm. Biol. 2001, 39, 67–78. [Google Scholar] [CrossRef]
- Satyal, P.; Paudel, P.; Poudel, A.; Dosoky, N.S.; Pokharel, K.K.; Setzer, W.N. Bioactivities and compositional analyses of Cinnamomum essential oils from Nepal: C. camphora, C. tamala, and C. glaucescens. Nat. Prod. Commun. 2013, 8, 1777–1784. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, T.G.; Nedrud, J.G. Laboratory maintenance of Helicobacter species. Curr. Protoc. Microbiol. 2006, 1, 8B-1. [Google Scholar] [CrossRef]
- Ward, J.H. Hierarchical grouping to optimize an objective function. J. Am. Stat. Assoc. 1963, 58, 236–244. [Google Scholar] [CrossRef]
| Sample Code | Collection Site | dbh, cm | GPS Coordinates | Collection Date | Oleoresin Mass (g) | Essential Oil Yield (%) |
|---|---|---|---|---|---|---|
| RE190401A | Longsheng (tree A) | 170 | 25°41′51.70”N, 110° 9′8.80”E, elev 1204 m | 3/16/2019 | 23.10 | 11.43 |
| RE190401D | Wangmo (tree C) | 105 | 25°22′35.22”N, 106° 2′6.54”E, elev 573 m | 3/17/2019 | 173.60 | 13.61 |
| RE190401E | Wangmo (tree F) | 87 | 25°24′32.88”N, 105°58′23.16”E, elev 780 m | 3/17/2019 | 92.00 | 18.00 |
| LD190910C | 4 Leye | 200 | 24°37′36.23”N, 106°33′25.27”E, elev 921 m | 8/27/2019 | 55.10 | 23.96 |
| LD190910D | 5 Leye | 303 | 24°37′36.23”N, 106°33′25.26”E, elev 924 m | 8/27/2019 | 41.20 | 14.20 |
| LD190910E | 6 Leye | 150 | 24°52′56.10”N, 106°26′20.36”E, elev 1111 m | 8/27/2019 | 49.40 | 19.98 |
| LD190910F | 7 Leye | 190 | 24°52′56.10”N, 106°26′20.36”E, elev 1111 m | 8/27/2019 | 40.54 | 30.17 |
| LD190910G | 8 Leye | 158 | 24°52′58.85”N, 106°25′52.28”E, elev 1213 m | 8/27/2019 | 52.80 | 17.92 |
| LD190910H | 9 Leye | 144 | 24°52′58.85”N, 106°25′52.28”E, elev 1213 m | 8/27/2019 | 40.00 | 17.53 |
| LD190910I | 10 Leye | 187 | 24°52′59.58”N, 106°26′19.95”E, elev 1127 m | 8/27/2019 | 38.80 | 23.30 |
| LD190910J | 11 Leye | 94 | 24°47′20.39”N, 106°34′28.89”E, elev 1118 m | 8/27/2019 | 55.34 | 17.96 |
| LD190910K | 12 Leye | 244 | 24°47′20.47”N, 106°34′30.28”E, elev 1130 m | 8/27/2019 | 41.60 | 25.84 |
| LD190910L | 13 Leye | 116 | 24°47′3.38”N, 106°34′35.56”E, elev 1136 m | 8/27/2019 | 25.93 | 7.71 |
| LD190910M | 14 Leye | 92 | 24°47′2.77”N, 106°34′35.08”E, elev 1142 m | 8/27/2019 | 49.36 | 15.80 |
| LD190910N | 15 Leye | 265 | 24°38′56.00”N, 106°39′0.70”E, elev 883 m | 8/27/2019 | 45.87 | 20.58 |
| LD190910O | 16 Wangmo | 120 | 25°14′59.65”N, 106°28′7.61”E, elev 717 m | 8/31/2019 | 60.50 | 13.59 |
| LD190910P | 17 Wangmo | 106 | 25°14′56.69”N, 106°28′8.62”E, elev 731 m | 8/31/2019 | 38.10 | 20.29 |
| LD190910Q | 18 Wangmo | 110 | 25°14′56.11”N, 106°28′9.10”E, elev 739 m | 8/31/2019 | 52.40 | 14.56 |
| LD190910R | 19 Wangmo | 70 | 25°14′40.42”N, 106°28′17.92”E, elev 761 m | 8/31/2019 | 44.40 | 9.28 |
| LD190910S | 20 Wangmo | 72 | 25°14′40.50”N, 106°28′18.04”E, elev 764 m | 8/31/2019 | 49.60 | 15.73 |
| LD190910T | 21 Wangmo | 85 | 25°14′40.90”N, 106°28′17.84”E, elev 765 m | 8/31/2019 | 43.25 | 21.32 |
| LD190910U | 22 Wangmo | 107 | 25°14′40.89”N, 106°28′17.08”E, elev 757 m | 8/31/2019 | 47.60 | 21.32 |
| LD190910V | 23 Wangmo | 145 | 25°13′48.24”N, 106° 7′44.64”E, elev 991 m | 8/31/2019 | 61.40 | 17.51 |
| LD190910W | 24 Wangmo | 80 | 25°13′47.85”N, 106° 7′44.85”E, elev 993 m | 8/31/2019 | 36.65 | 14.05 |
| LD190910X | 25 Wangmo | 105 | 25°13′50.79”N, 106° 7′44.79”E, elev 992 m | 8/31/2019 | 3.96 | 15.04 |
| Compound | Group #1 | Group #2 | Group #3 | Group #4 | Overall |
|---|---|---|---|---|---|
| Average (Range) | Average (Range) | Average (Range) | Average (Range) | Average (Range) | |
| (E)-Caryophyllene | 14.2 (8.9–18.5) | 26.0 (19.9–34.8) | 5.0 (3.3–6.5) | 49.0 (42.0–64.4) | 23.5 (3.3–64.4) |
| α-Pinene | 23.4 (18.7–27.8) | 19.3 (15.3–25.3) | 31.7 (29.1–34.5) | 6.3 (0.6–11.8) | 20.0 (0.6–34.5) |
| β-Pinene | 16.9 (12.0–20.7) | 14.2 (10.9–18.7) | 23.3 (20.6–26.0) | 5.4 (0.6–9.2) | 14.8 (0.6–26.0) |
| Camphene | 12.3 (9.2–15.3) | 8.6 (6.0–10.9) | 14.5 (11.0–17.3) | 2.7 (0.3–5.2) | 9.6 (0.3–17.3) |
| Limonene | 5.8 (3.9–7.9) | 3.6 (2.5–5.6) | 6.7 (6.4–7.3) | 0.9 (0.2–1.9) | 4.3 (0.2–7.9) |
| Germacrene D | 2.2 (0.0–6.5) | 2.9 (0.0–9.9) | 0.9 (0.2–1.8) | 2.3 (0.2–5.5) | 2.2 (0.0–9.9) |
| Camphor | 3.3 (0.3–7.9) | 1.6 (0.2–2.8) | 1.2 (0.0–3.6) | 1.0 (0.0–2.6) | 1.9 (0.0–7.9) |
| β-Copaene | 0.9 (0.3–1.5) | 2.1 (0.4–5.3) | 0.3 (0.1–0.6) | 3.7 (2.7–4.9) | 1.8 (0.1–5.3) |
| Bornyl acetate | 1.7 (0.0–8.7) | 1.3 (0.2–6.1) | 3.1 (1.0–6.3) | 1.3 (0.2–3.0) | 1.7 (0.0–8.7) |
| α-Muurolol | 1.1 (0.3–2.0) | 1.8 (0.4–2.9) | 0.3 (0.1–0.6) | 3.7 (2.5–5.7) | 1.7 (0.1–5.7) |
| p-Cymene | 3.7 (0.1–13.6) | 0.7 (0.1–1.9) | 1.1 (0.2–3.8) | 0.4 (0.0–0.7) | 1.7 (0.0–13.6) |
| Sabinene | 1.8 (0.4–3.1) | 1.3 (0.4–1.9) | 3.0 (2.4–3.6) | 0.4 (0.0–1.1) | 1.5 (0.0–3.6) |
| β-Cubebene | 0.7 (0.2–1.2) | 1.1 (0.4–1.8) | 0.3 (0.1–0.6) | 2.4 (0.1–3.4) | 1.1 (0.1–3.4) |
| Caryophyllene oxide | 0.8 (0.3–2.5) | 1.0 (0.5–1.5) | 0.3 (0.2–0.5) | 1.6 (0.7–4.4) | 0.9 (0.2–4.4) |
| α-Muurolene | 0.4 (0.1–0.6) | 0.9 (0.2–2.1) | 0.1 (0.0–0.2) | 1.9 (1.4–2.4) | 0.8 (0.0–2.4) |
| Myrcene | 0.7 (0.0–1.6) | 0.4 (0.1–1.3) | 2.2 (0.1–5.1) | 0.4 (0.1–1.3) | 0.8 (0.0–5.1) |
| (2E,4E)-3,7-Dimethylocta-2,4-diene | 1.3 (0.3–3.9) | 0.5 (0.1–1.0) | 0.5 (0.1–0.9) | 0.3 (0.0–0.6) | 0.7 (0.0–3.9) |
| δ-Cadinene | 0.3 (0.1–0.5) | 0.7 (0.3–1.9) | 0.1 (0.1–0.1) | 1.5 (0.7–2.2) | 0.7 (0.1–2.2) |
| γ-Muurolene | 0.3 (0.1–0.5) | 0.7 (0.2–1.7) | 0.1 (0.0–0.2) | 1.5 (1.0–1.9) | 0.6 (0.0–1.9) |
| α-Humulene | 0.3 (0.2–0.4) | 0.7 (0.5–0.9) | 0.1 (0.1–0.1) | 1.4 (1.1–2.0) | 0.6 (0.1–2.0) |
| Cubebol | 0.3 (0.1–0.7) | 0.6 (0.1–1.3) | 0.1 (0.0–0.2) | 1.2 (0.7–2.1) | 0.5 (0.0–2.1) |
| trans-β-Elemene | 0.1 (0.0–0.1) | 1.3 (0.0–5.3) | 0.2 (0.0–0.9) | 0.2 (0.0–0.3) | 0.5 (0.0–5.3) |
| Bornyl cinnamate | 0.5 (0.1–1.2) | 0.5 (0.1–0.8) | 0.5 (0.3–0.8) | 0.4 (0.2–1.0) | 0.5 (0.1–1.2) |
| Borneol | 0.4 (0.1–0.8) | 0.6 (0.3–1.0) | 0.5 (0.2–0.7) | 0.4 (0.0–0.8) | 0.5 (0.0–1.0) |
| α-Cubebene | 0.1 (0.0–0.3) | 0.6 (0.1–2.5) | 0.0 (0.0–0.1) | 0.8 (0.4–1.4) | 0.4 (0.0–2.5) |
| Essential Oil Sample | Gram-Positive Bacteria | Gram-Negative Bacteria | |||||
|---|---|---|---|---|---|---|---|
| Bacillus Cereus | Cutibacterium Acnesb | Staphylococcus Aureus | Staphylococcus Epidermidis | Streptococcus Pyogenes | Pseudomonas Aeruginosa | Serratia Marcescens | |
| LD190910C | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910D | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910E | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910F | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910G | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910H | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910I | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910J | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910K | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910L | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910M | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910N | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910O | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910P | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910Q | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910R | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910S | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910T | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910U | 156 | 78 | 156 | 78 | 156 | 313 | 625 |
| LD190910V | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910W | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| LD190910X | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| Re190401A | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| Re190401D | 78 | 156 | 156 | 78 | 156 | 313 | 625 |
| Re190401E | 156 | 156 | 156 | 78 | 156 | 313 | 625 |
| Pure Compounds | |||||||
| (E)-Caryophyllene | 313 | 625 | 313 | 313 | 313 | 313 | 313 |
| (+)-α-Pinene | 313 | 625 | 625 | 313 | 625 | 313 | 313 |
| (–)-α-Pinene | 313 | 625 | 313 | 313 | 313 | 313 | 313 |
| (–)-β-Pinene | 313 | 313 | 156 | 313 | 625 | 313 | 313 |
| Camphene | 313 | 625 | 313 | 313 | 313 | 313 | 313 |
| (+)-Limonene | 313 | 625 | 313 | 313 | 313 | 313 | 625 |
| (–)-Limonene | 313 | 39 | 313 | 78 | 625 | 313 | 313 |
| Gentamicin | <19.5 | <19.5 | <19.5 | <19.5 | <19.5 | <19.5 | <19.5 |
| Essential Oil Sample | Molds | Yeast | |||||
|---|---|---|---|---|---|---|---|
| Aspergillus Fumigatus | Aspergillus Niger | Microsporum Canis | Microsporum Gypseum | Trichophyton Mentagrophytes | Trichophyton Rubrum | Candida Albicans | |
| LD190910C | 156 | 313 | 313 | 313 | 156 | 313 | 313 |
| LD190910D | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| LD190910E | 156 | 156 | 156 | 313 | 156 | 313 | 313 |
| LD190910F | 156 | 313 | 313 | 313 | 156 | 313 | 313 |
| LD190910G | 156 | 313 | 313 | 313 | 156 | 313 | 313 |
| LD190910H | 156 | 78 | 313 | 313 | 156 | 313 | 313 |
| LD190910I | 156 | 313 | 313 | 313 | 156 | 313 | 313 |
| LD190910J | 156 | 156 | 313 | 313 | 156 | 313 | 313 |
| LD190910K | 156 | 156 | 313 | 313 | 156 | 313 | 313 |
| LD190910L | 156 | 313 | 313 | 313 | 156 | 313 | 313 |
| LD190910M | 156 | 156 | 313 | 313 | 156 | 313 | 313 |
| LD190910N | 156 | 78 | 156 | 313 | 156 | 313 | 313 |
| LD190910O | 156 | 78 | 156 | 313 | 156 | 313 | 313 |
| LD190910P | 156 | 156 | 156 | 313 | 156 | 313 | 313 |
| LD190910Q | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| LD190910R | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| LD190910S | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| LD190910T | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| LD190910U | 156 | 156 | 156 | 313 | 156 | 313 | 313 |
| LD190910V | 156 | 156 | 156 | 313 | 156 | 313 | 313 |
| LD190910W | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| LD190910X | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| Re190401A | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| Re190401D | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| Re190401E | 156 | 313 | 156 | 313 | 156 | 313 | 313 |
| Pure Compounds | |||||||
| (E)-Caryophyllene | 156 | 1250 | 313 | 313 | 625 | 313 | 156 |
| (+)-α-Pinene | 156 | 78 | 313 | 156 | 156 | 313 | 156 |
| (–)-α-Pinene | 313 | 156 | 313 | 313 | 313 | 313 | 156 |
| (–)-β-Pinene | 156 | 78 | 313 | 313 | 156 | 313 | 156 |
| Camphene | 313 | 156 | 313 | 313 | 625 | 313 | 156 |
| (+)-Limonene | 156 | 156 | 313 | 313 | 313 | 313 | 156 |
| (–)-Limonene | 156 | 156 | 313 | 156 | 156 | 313 | 156 |
| Amphotericin B | <19.5 | <19.5 | <19.5 | <19.5 | <19.5 | <19.5 | <19.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
DeCarlo, A.; Zeng, T.; Dosoky, N.S.; Satyal, P.; Setzer, W.N. The Essential Oil Composition and Antimicrobial Activity of Liquidambar formosana Oleoresin. Plants 2020, 9, 822. https://doi.org/10.3390/plants9070822
DeCarlo A, Zeng T, Dosoky NS, Satyal P, Setzer WN. The Essential Oil Composition and Antimicrobial Activity of Liquidambar formosana Oleoresin. Plants. 2020; 9(7):822. https://doi.org/10.3390/plants9070822
Chicago/Turabian StyleDeCarlo, Anjanette, Tao Zeng, Noura S. Dosoky, Prabodh Satyal, and William N. Setzer. 2020. "The Essential Oil Composition and Antimicrobial Activity of Liquidambar formosana Oleoresin" Plants 9, no. 7: 822. https://doi.org/10.3390/plants9070822
APA StyleDeCarlo, A., Zeng, T., Dosoky, N. S., Satyal, P., & Setzer, W. N. (2020). The Essential Oil Composition and Antimicrobial Activity of Liquidambar formosana Oleoresin. Plants, 9(7), 822. https://doi.org/10.3390/plants9070822








