Phytotoxic and Antimicrobial Activities of Teucrium polium and Thymus decussatus Essential Oils Extracted Using Hydrodistillation and Microwave-Assisted Techniques
Abstract
1. Introduction
2. Results and Discussion
2.1. Chemical Profiles of EOs
2.2. Principal Component Analysis (PCA) of EO Chemical Compounds
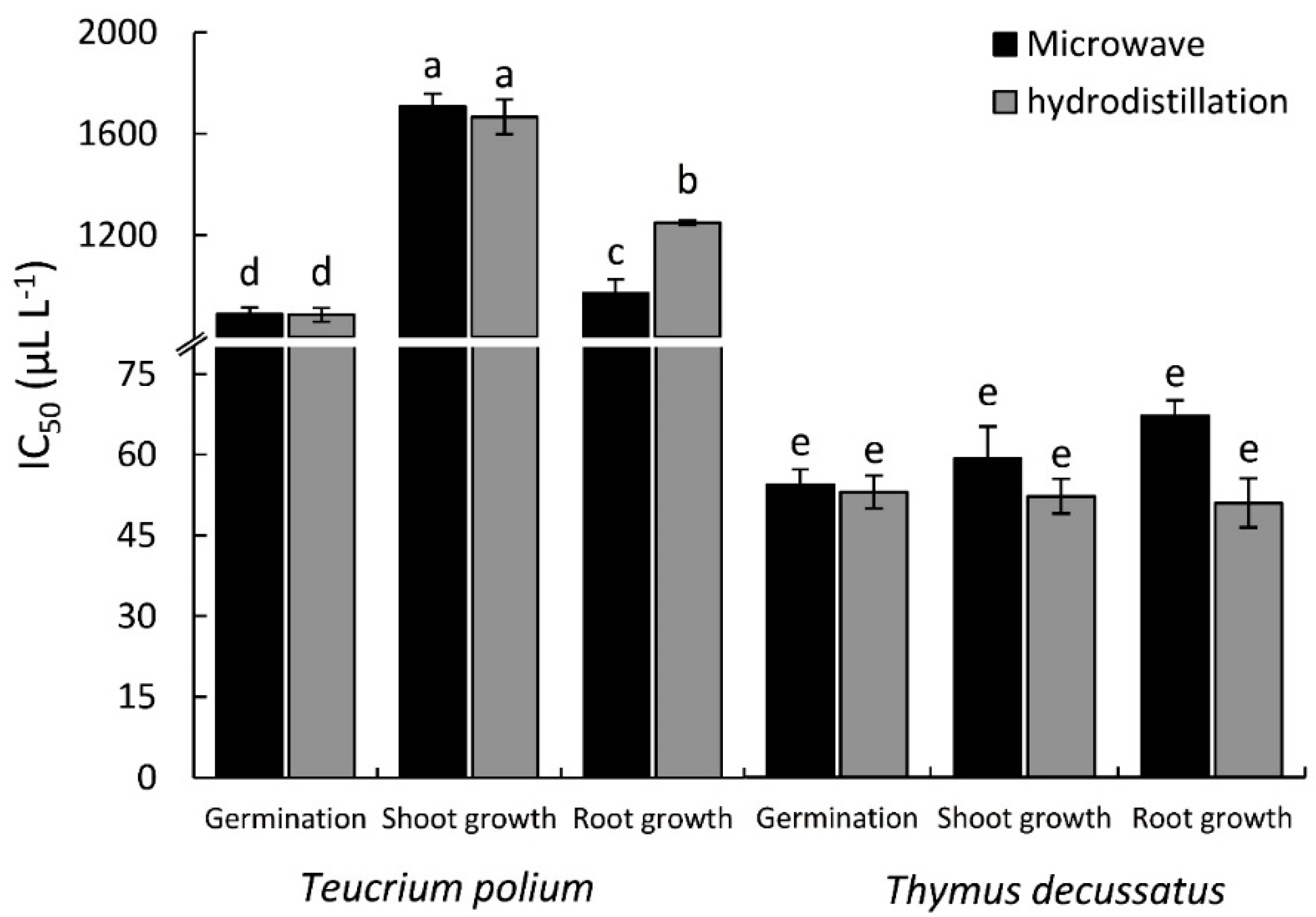
2.3. Phytotoxicity Activity
2.4. Antimicrobial Activities
3. Materials and Methods
3.1. Plant Materials
3.2. EO Extraction, Analysis, and Constituent Identification
3.3. Phytotoxic Activity of the Extracted EOs
3.4. Antimicrobial Properties of EOs
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zahran, M.A.; Willis, A.J. The Vegetation of Egypt; Springer Science & Business Media: Berlin, Germany, 2008; Volume 2. [Google Scholar]
- Eissa, T.; Palomino, O.; Carretero, M.; Gómez-Serranillos, M. Ethnopharmacological study of medicinal plants used in the treatment of CNS disorders in Sinai Peninsula, Egypt. J. Ethnopharmacol. 2014, 151, 317–332. [Google Scholar] [CrossRef] [PubMed]
- Boulos, L. Flora of Egypt Checklist; Al-Hadara Publishing: Cairo, Egypt, 2009; pp. 198–201. [Google Scholar]
- Batanouny, K.H.; Aboutabl, E.; Shabana, M.F.S. Wild Medicinal Plants in Egypt. An Inventory to Support Conservation and Sustainable Use; The Palm Press: Cairo, Egypt, 1999. [Google Scholar]
- Elshamy, A.I.; Abd El-Gawad, A.M.; El Gendy, A.E.-N.G.; Assaeed, A.M. Chemical characterization of Euphorbia heterophylla L. essential oils and their antioxidant activity and allelopathic potential on Cenchrus echinatus L. Chem. Biodivers. 2019, 16, e1900051. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Gawad, A.M.; Elshamy, A.I.; El Gendy, A.E.-N.; Gaara, A.; Assaeed, A.M. Volatiles profiling, allelopathic activity, and antioxidant potentiality of Xanthium strumarium leaves essential oil from Egypt: Evidence from chemometrics analysis. Molecules 2019, 24, 584. [Google Scholar] [CrossRef] [PubMed]
- Assaeed, A.; Elshamy, A.; El Gendy, A.E.-N.; Dar, B.; Al-Rowaily, S.; Abd-ElGawad, A. Sesquiterpenes-rich essential oil from above ground parts of Pulicaria somalensis exhibited antioxidant activity and allelopathic effect on weeds. Agronomy 2020, 10, 399. [Google Scholar] [CrossRef]
- Abd-ElGawad, A.M.; Elshamy, A.; El Gendy, A.E.-N.; Al-Rowaily, S.L.; Assaeed, A.M. Preponderance of oxygenated sesquiterpenes and diterpenes in the volatile oil constituents of Lactuca serriola L. revealed antioxidant and allelopathic activity. Chem. Biodivers. 2019, 16, e1900278. [Google Scholar] [CrossRef]
- Elshamy, A.; Abd-ElGawad, A.M.; El-Amier, Y.A.; El Gendy, A.; Al-Rowaily, S. Interspecific variation, antioxidant and allelopathic activity of the essential oil from three Launaea species growing naturally in heterogeneous habitats in Egypt. Flavour Fragr. J. 2019, 34, 316–328. [Google Scholar] [CrossRef]
- Abd-ElGawad, A.M.; Elshamy, A.; Al-Rowaily, S.; El-Amier, Y.A. Habitat affects the chemical profile, allelopathy, and antioxidant properties of essential oils and phenolic enriched extracts of the invasive plant Heliotropium Curassavicum. Plants 2019, 8, 482. [Google Scholar] [CrossRef]
- Deng, W.; Liu, K.; Cao, S.; Sun, J.; Zhong, B.; Chun, J. Chemical composition, antimicrobial, antioxidant, and antiproliferative properties of grapefruit essential oil prepared by molecular distillation. Molecules 2020, 25, 217. [Google Scholar] [CrossRef]
- Elshamy, A.I.; Ammar, N.M.; Hassan, H.A.; Al-Rowaily, S.L.; Raga, T.R.; El Gendy, A.; Abd-ElGawad, A.M. Essential oil and its nanoemulsion of Araucaria heterophylla resin: Chemical characterization, anti-inflammatory, and antipyretic activities. Ind. Crop. Prod. 2020, 148, 112272. [Google Scholar] [CrossRef]
- Arunachalam, K.; Balogun, S.O.; Pavan, E.; de Almeida, G.V.B.; de Oliveira, R.G.; Wagner, T.; Cechinel Filho, V.; de Oliveira Martins, D.T. Chemical characterization, toxicology and mechanism of gastric antiulcer action of essential oil from Gallesia integrifolia (Spreng.) Harms in the in vitro and in vivo experimental models. Biomed. Pharmacother. 2017, 94, 292–306. [Google Scholar] [CrossRef]
- Damtie, D.; Braunberger, C.; Conrad, J.; Mekonnen, Y.; Beifuss, U. Composition and hepatoprotective activity of essential oils from Ethiopian thyme species (Thymus serrulatus and Thymus schimperi). J. Essent. Oil Res. 2019, 31, 120–128. [Google Scholar] [CrossRef]
- Heywood, V.H.; Brummitt, R.; Culham, A.; Seberg, O. Flowering Plant Families of the World; Firefly Books: Ontario, ON, Canada, 2007; Volume 88. [Google Scholar]
- Harley, R.M.; Atkins, S.; Budantsev, A.L.; Cantino, P.D.; Conn, B.J.; Grayer, R.J.; Harley, M.M.; de Kok, R.P.; Morales, R.; Paton, A.J.; et al. Labiatae. In The Families and Genera of Vascular Plants; Kubitzki, K., Ed.; Springer: Berlin, Germany, 2004; Volume 7, pp. 2275–2283. [Google Scholar]
- Rafieian-Kopaei, M.; Nasri, H.; Baradaran, A. Teucrium polium: Liver and kidney effects. J. Res. Med Sci. 2014, 19, 478–479. [Google Scholar]
- Forouzandeh, H.; Azemi, M.E.; Rashidi, I.; Goudarzi, M.; Kalantari, H. Study of the protective effect of Teucrium polium L. extract on acetaminophen-induced hepatotoxicity in mice. Iran. J. Pharm. Res. 2013, 12, 123–129. [Google Scholar]
- Khazaei, M.; Nematollahi-Mahani, S.N.; Mokhtari, T.; Sheikhbahaei, F. Review on Teucrium polium biological activities and medical characteristics against different pathologic situations. J. Contemp. Med Sci. 2018, 4, 1–6. [Google Scholar]
- Venditti, A.; Frezza, C.; Trancanella, E.; Zadeh, S.M.M.; Foddai, S.; Sciubba, F.; Delfini, M.; Serafini, M.; Bianco, A. A new natural neo-clerodane from Teucrium polium L. collected in Northern Iran. Ind. Crop. Prod. 2017, 97, 632–638. [Google Scholar] [CrossRef]
- Elmasri, W.A.; Yang, T.; Tran, P.; Hegazy, M.-E.F.; Hamood, A.N.; Mechref, Y.; Paré, P.W. Teucrium polium phenylethanol and iridoid glycoside characterization and flavonoid inhibition of biofilm-forming Staphylococcus aureus. J. Nat. Prod. 2015, 78, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Purnavab, S.; Ketabchi, S.; Rowshan, V. Chemical composition and antibacterial activity of methanolic extract and essential oil of Iranian Teucrium polium against some of phytobacteria. Nat. Prod. Res. 2015, 29, 1376–1379. [Google Scholar] [CrossRef]
- Ben Othman, M.; Salah-Fatnassi, K.B.H.; Ncibi, S.; Elaissi, A.; Zourgui, L. Antimicrobial activity of essential oil and aqueous and ethanol extracts of Teucrium polium L. subsp. gabesianum (LH) from Tunisia. Physiol. Mol. Biol. Plants 2017, 23, 723–729. [Google Scholar] [CrossRef]
- Ibrahim, S.R.; Abdallah, H.M.; Mohamed, G.A.; Farag, M.A.; Alshali, K.Z.; Alsherif, E.A.; Ross, S.A. Volatile oil profile of some lamiaceous plants growing in Saudi Arabia and their biological activities. Z. Für Nat. C 2017, 72, 35–41. [Google Scholar] [CrossRef]
- Li, X.; He, T.; Wang, X.; Shen, M.; Yan, X.; Fan, S.; Wang, L.; Wang, X.; Xu, X.; Sui, H. Traditional uses, chemical constituents and biological activities of plants from the genus Thymus. Chem. Biodivers. 2019, 16, e1900254. [Google Scholar] [CrossRef]
- El-Hela, A. Chemical composition and biological studies of the essential oil of Thymus decussatus Benth growing in Egypt. Egypt. J. Biomed. Sci. 2007, 23, 146–153. [Google Scholar] [CrossRef]
- Abd El-Mohsen, M.M.; Abd El-Mohsen, M.M.; Ehsan, N.A.; Hussein, A.A.; Hammouda, F.M.; Ismail, S.I.; Hiffnawy, M.S. Hepatoprotective and antioxidative activities of the essential oils of Origanum syriacum (L.), Thymus decussatus (Benth) and Salvia multicaulis (Vahl Enum.). J. Arab Soc. Med Res. 2009, 4, 19–24. [Google Scholar]
- Amin, S.M.; Hassan, H.M.; El Gendy, A.E.N.G.; El-Beih, A.A.; Mohamed, T.A.; Elshamy, A.I.; Bader, A.; Shams, K.A.; Mohammed, R.; Hegazy, M.E.F. Comparative chemical study and antimicrobial activity of essential oils of three Artemisia species from Egypt and Saudi Arabia. Flavour Fragr. J. 2019, 34, 450–459. [Google Scholar] [CrossRef]
- Elmasri, W.A.; Hegazy, M.E.F.; Aziz, M.; Koksal, E.; Amor, W.; Mechref, Y.; Hamood, A.N.; Cordes, D.B.; Paré, P.W. Biofilm blocking sesquiterpenes from Teucrium polium. Phytochemistry 2014, 103, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Gamal-Eldeen, A.M.; Hegazy, M.-E. A crystal lapiferin derived from Ferula vesceritensis induces apoptosis pathway in MCF-7 breast cancer cells. Nat. Prod. Res. 2010, 24, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Gawad, A.M.; El-Amier, Y.A.; Bonanomi, G. Essential oil composition, antioxidant and allelopathic activities of Cleome droserifolia (Forssk.) Delile. Chem. Biodivers. 2018, 15, e1800392. [Google Scholar] [CrossRef]
- Reda, E.; Saleh, I.; El Gendy, A.N.; Talaat, Z.; Hegazy, M.-E.; Haggag, E. Chemical constituents of Euphorbia sanctae-catharinae Fayed essential oil: A comparative study of hydro-distillation and microwave-assisted extraction. J. Adv. Pharm. Res. 2017, 1, 155–159. [Google Scholar] [CrossRef][Green Version]
- Aburjai, T.; Hudaib, M.; Cavrini, V. Composition of the essential oil from Jordanian germander (Teucrium polium L.). J. Essent. Oil Res. 2006, 18, 97–99. [Google Scholar] [CrossRef]
- Cozzani, S.; Muselli, A.; Desjobert, J.M.; Bernardini, A.F.; Tomi, F.; Casanova, J. Chemical composition of essential oil of Teucrium polium subsp. capitatum (L.) from Corsica. Flavour Fragr. J. 2005, 20, 436–441. [Google Scholar] [CrossRef]
- Kabouche, A.; Kabouche, Z.; Ghannadi, A.; Sajjadi, S. Analysis of the essential oil of Teucrium polium ssp. aurasiacum from Algeria. J. Essent. Oil Res. 2007, 19, 44–46. [Google Scholar] [CrossRef]
- Lograda, T.; Ramdani, M.; Chalard, P.; Figueredo, G.; Deghar, A. Chemical analysis and antimicrobial activity of Teucrium polium L. essential oil from eastern Algeria. Am. J. Adv. Drug Deliv. 2014, 2, 697–710. [Google Scholar]
- Kerbouche, L.; Hazzit, M.; Ferhat, M.-A.; Baaliouamer, A.; Miguel, M.G. Biological activities of essential oils and ethanol extracts of Teucrium polium subsp. capitatum (L.) Briq. and Origanum floribundum Munby. J. Essent. Oil Bear. Plants 2015, 18, 1197–1208. [Google Scholar] [CrossRef]
- Kovacevic, N.N.; Lakusic, B.S.; Ristic, M.S. Composition of the essential oils of seven Teucrium species from Serbia and Montenegro. J. Essent. Oil Res. 2001, 13, 163–165. [Google Scholar] [CrossRef]
- Sabzeghabaie, A.; Asgarpanah, J. Essential oil composition of Teucrium polium L. fruits. J. Essent. Oil Res. 2016, 28, 77–80. [Google Scholar] [CrossRef]
- Kabouche, Z.; Boutaghane, N.; Laggoune, S.; Kabouche, A.; Ait-Kaki, Z.; Benlabed, K. Comparative antibacterial activity of five Lamiaceae essential oils from Algeria. Int. J. Aromather. 2005, 15, 129–133. [Google Scholar] [CrossRef]
- Sadeghi, H.; Jamalpoor, S.; Shirzadi, M.H. Variability in essential oil of Teucrium polium L. of different latitudinal populations. Ind. Crop. Prod. 2014, 54, 130–134. [Google Scholar] [CrossRef]
- De Martino, L.; Formisano, C.; Mancini, E.; Feo, V.D.; Piozzi, F.; Rigano, D.; Senatore, F. Chemical composition and phytotoxic effects of essential oils from four Teucrium species. Nat. Prod. Commun. 2010, 5, 1969–1976. [Google Scholar] [CrossRef]
- Menichini, F.; Conforti, F.; Rigano, D.; Formisano, C.; Piozzi, F.; Senatore, F. Phytochemical composition, anti-inflammatory and antitumour activities of four Teucrium essential oils from Greece. Food Chem. 2009, 115, 679–686. [Google Scholar] [CrossRef]
- Mitić, V.; Jovanović, O.; Stankov-Jovanović, V.; Zlatkovic, B.; Stojanovic, G. Analysis of the Essential oil of Teucrium polium ssp. capitatum from the Balkan Peninsula. Nat. Prod. Commun. 2012, 7, 83–86. [Google Scholar]
- Boulos, L. Flora of Egypt, Vol. III (Verbenaceae—Compositae); Al-Hadara Publishing: Cairo, Egypt, 2002; Volume 3. [Google Scholar]
- Guan, X.; Ge, D.; Li, S.; Huang, K.; Liu, J.; Li, F. Chemical composition and antimicrobial activities of Artemisia argyi Lévl. et Vant essential oils extracted by simultaneous distillation-extraction, subcritical extraction and hydrodistillation. Molecules 2019, 24, 483. [Google Scholar] [CrossRef]
- Mohammed, M.S.A.; Tawfik, M.S.H.; Ibrahim, A.E.-G. Influence of two extraction methods on essential oils of some Apiaceae family plants. Egypt. Pharm. J. 2019, 18, 160–166. [Google Scholar]
- De Martino, L.; Bruno, M.; Formisano, C.; De Feo, V.; Napolitano, F.; Rosselli, S.; Senatore, F. Chemical composition and antimicrobial activity of the essential oils from two species of Thymus growing wild in southern Italy. Molecules 2009, 14, 4614–4624. [Google Scholar] [CrossRef] [PubMed]
- Kordali, S.; Cakir, A.; Ozer, H.; Cakmakci, R.; Kesdek, M.; Mete, E. Antifungal, phytotoxic and insecticidal properties of essential oil isolated from Turkish Origanum acutidens and its three components, carvacrol, thymol and p-cymene. Bioresour. Technol. 2008, 99, 8788–8795. [Google Scholar] [CrossRef] [PubMed]
- Saharkhiz, M.J.; Smaeili, S.; Merikhi, M. Essential oil analysis and phytotoxic activity of two ecotypes of Zataria multiflora Boiss. growing in Iran. Nat. Prod. Res. 2010, 24, 1598–1609. [Google Scholar] [CrossRef] [PubMed]
- Aragão, F.; Palmieri, M.; Ferreira, A.; Costa, A.; Queiroz, V.; Pinheiro, P.; Andrade-Vieira, L. Phytotoxic and cytotoxic effects of Eucalyptus essential oil on lettuce (Lactuca sativa L.). Allelopath. J. 2015, 35, 259–272. [Google Scholar]
- Ulukanli, Z.; Bozok, F.; Cenet, M.; Ince, H.; Demirci, S.C.; Sezer, G. Secondary metabolites and bioactivities of Thymbra spicata var. spicata in Amanos mountains. J. Essent. Oil Bear. Plants 2016, 19, 1754–1761. [Google Scholar] [CrossRef]
- Ulukanli, Z.; Cenet, M.; Ince, H.; Yilmaztekin, M. Antimicrobial and herbicidal activities of the essential oil from the Mediterranean Thymus eigii. J. Essent. Oil Bear. Plants 2018, 21, 214–222. [Google Scholar] [CrossRef]
- Kashkooli, A.B.; Saharkhiz, M.J. Essential oil compositions and natural herbicide activity of four Denaei Thyme (Thymus daenensis Celak.) ecotypes. J. Essent. Oil Bear. Plants 2014, 17, 859–874. [Google Scholar] [CrossRef]
- Pinheiro, P.F.; Costa, A.V.; Alves, T.d.A.; Galter, I.N.; Pinheiro, C.A.; Pereira, A.F.; Oliveira, C.M.R.; Fontes, M.M.P. Phytotoxicity and cytotoxicity of essential oil from leaves of Plectranthus amboinicus, carvacrol, and thymol in plant bioassays. J. Agric. Food Chem. 2015, 63, 8981–8990. [Google Scholar] [CrossRef]
- De Almeida, L.F.R.; Frei, F.; Mancini, E.; De Martino, L.; De Feo, V. Phytotoxic activities of Mediterranean essential oils. Molecules 2010, 15, 4309–4323. [Google Scholar] [CrossRef]
- Verdeguer, M.; Blázquez, M.A.; Boira, H. Phytotoxic effects of Lantana camara, Eucalyptus camaldulensis and Eriocephalus africanus essential oils in weeds of Mediterranean summer crops. Biochem. Syst. Ecol. 2009, 37, 362–369. [Google Scholar] [CrossRef]
- Taban, A.; Saharkhiz, M.J.; Hadian, J. Allelopathic potential of essential oils from four Satureja spp. Biol. Agric. Hortic. 2013, 29, 244–257. [Google Scholar] [CrossRef]
- Ben Arfa, A.; Combes, S.; Preziosi-Belloy, L.; Gontard, N.; Chalier, P. Antimicrobial activity of carvacrol related to its chemical structure. Lett. Appl. Microbiol. 2006, 43, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Roller, S.; Seedhar, P. Carvacrol and cinnamic acid inhibit microbial growth in fresh-cut melon and kiwifruit at 4 and 8 °C. Lett. Appl. Microbiol. 2002, 35, 390–394. [Google Scholar] [CrossRef]
- Belmekki, N.; Bendimerad, N.; Bekhechi, C.; Fernandez, X. Chemical analysis and antimicrobial activity of Teucrium polium L. essential oil from Western Algeria. J. Med. Plants Res. 2013, 7, 897–902. [Google Scholar]
- Magi, G.; Marini, E.; Facinelli, B. Antimicrobial activity of essential oils and carvacrol, and synergy of carvacrol and erythromycin, against clinical, erythromycin-resistant Group A Streptococci. Front. Microbiol. 2015, 6, 165. [Google Scholar] [CrossRef]
- Eusepi, P.; Marinelli, L.; García-Villén, F.; Borrego-Sánchez, A.; Cacciatore, I.; Di Stefano, A.; Viseras, C. Carvacrol prodrugs with antimicrobial activity loaded on clay nanocomposites. Materials 2020, 13, 1793. [Google Scholar] [CrossRef]
- Aznar, A.; Fernández, P.S.; Periago, P.M.; Palop, A. Antimicrobial activity of nisin, thymol, carvacrol and cymene against growth of Candida lusitaniae. Food Sci. Technol. Int. 2015, 21, 72–79. [Google Scholar] [CrossRef]
- Abd El-Gawad, A.M. Chemical constituents, antioxidant and potential allelopathic effect of the essential oil from the aerial parts of Cullen plicata. Ind. Crop. Prod. 2016, 80, 36–41. [Google Scholar] [CrossRef]
- Prabuseenivasan, S.; Jayakumar, M.; Ignacimuthu, S. In vitro antibacterial activity of some plant essential oils. BMC Complement. Altern. Med. 2006, 6, 39. [Google Scholar] [CrossRef]
- Rota, C.; Carraminana, J.; Burillo, J.; Herrera, A. In vitro antimicrobial activity of essential oils from aromatic plants against selected foodborne pathogens. J. Food Prot. 2004, 67, 1252–1256. [Google Scholar] [CrossRef] [PubMed]
- Legendre, P.; Legendre, L.F. Numerical Ecology; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
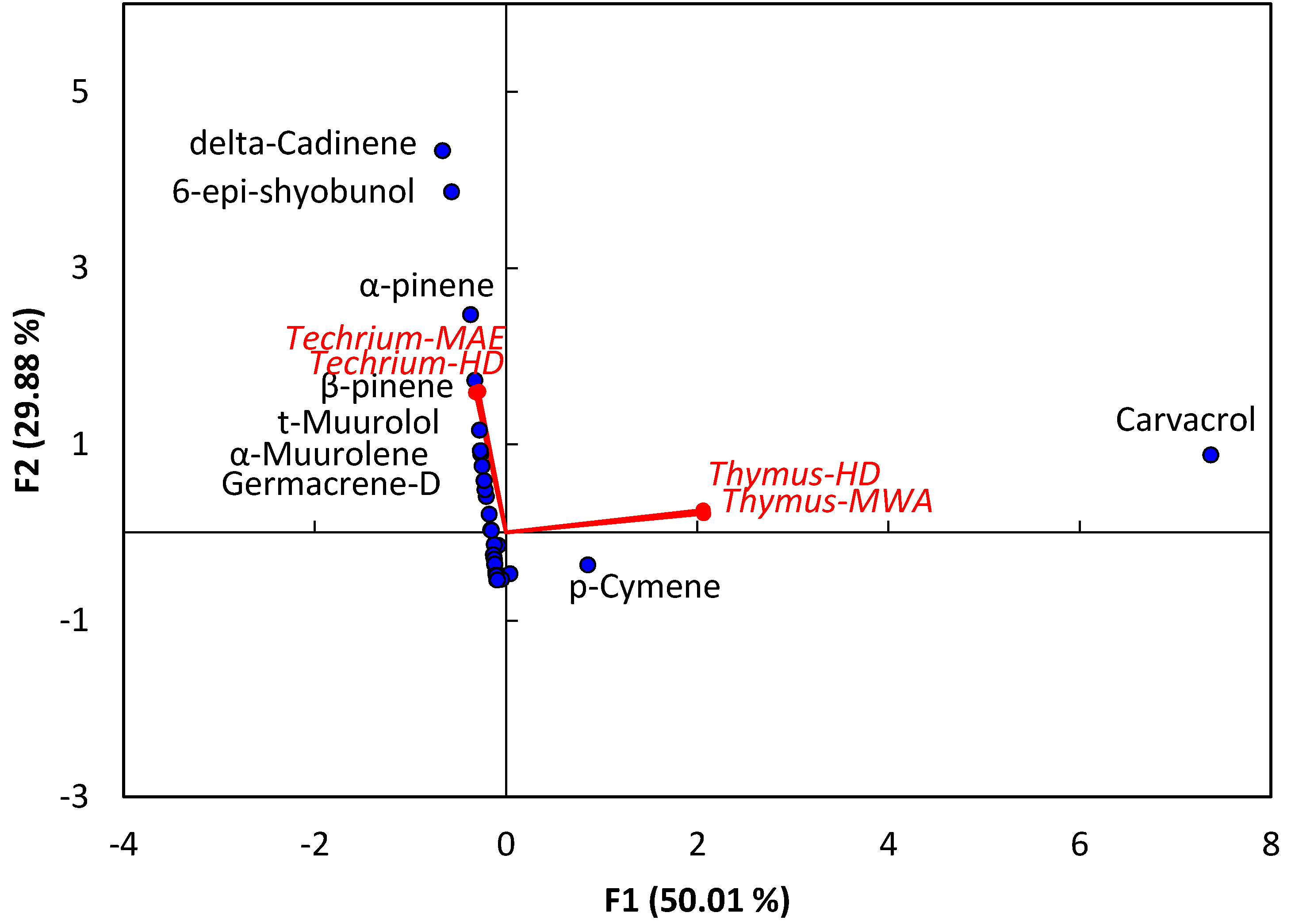
| No. | Compound | Rt | KIExp | KILit | T. polium | T. decussatus | Identification b | ||
|---|---|---|---|---|---|---|---|---|---|
| HD | MAE | HD | MAE | ||||||
| Monoterpene hydrocarbons | |||||||||
| 1 | α-Thujene | 4.57 | 925 | 925 | 0.52 ± 0.01 a | 1.26 ± 0.03 | 0.04 ± 0.01 | ---- | MS, KI |
| 2 | α-Pinene | 4.77 | 935 | 936 | 8.94 ± 0.06 | 11.58 ± 0.09 | ---- | ---- | MS, KI |
| 3 | Camphene | 5.80 | 954 | 954 | ---- | 0.44 ± 0.02 | ---- | 0.30 ± 0.02 | MS, KI |
| 4 | 2,4(10)-Thujadiene | 5.96 | 961 | 960 | ---- | 0.32 ± 0.01 | ---- | ---- | MS, KI |
| 5 | Sabinene | 7.44 | 975 | 975 | 2.94 ± 0.04 | 1.14 ± 0.03 | ---- | ---- | MS, KI |
| 6 | β-Pinene | 7.54 | 978 | 979 | 7.38 ± 0.05 | 8.20 ± 0.09 | 0.09 ± 0.01 | 0.54 ± 0.03 | MS, KI |
| 7 | α-Myrcene | 10.19 | 986 | 986 | 2.90 ± 0.04 | ---- | 0.16 ± 0.01 | 1.04 ± 0.03 | MS, KI |
| 8 | p-Cymene | 10.20 | 1024 | 1026 | ---- | ---- | 3.61 ± 0.04 | 16.98 ± 0.10 | MS, KI |
| 9 | Limonene | 10.94 | 1034 | 1033 | 1.10 ± 0.02 | 0.58 ± 0.03 | ---- | ---- | MS, KI |
| 10 | α-Terpinene | 12.18 | 1018 | 1018 | ---- | ---- | ---- | 1.01 ± 0.03 | MS, KI |
| 11 | β-Terpinene | 12.23 | 1031 | 1032 | ---- | ---- | 0.23 ± 0.01 | ---- | MS, KI |
| 12 | β-Phellandrene | 12.64 | 1045 | 1044 | ---- | ---- | ---- | 0.21 ± 0.01 | MS, KI |
| 13 | γ-Terpinene | 14.43 | 1061 | 1061 | 0.34 ± 0.02 | ---- | 0.49 ± 0.01 | 2.51 ± 0.06 | MS, KI |
| 14 | β-Terpinolene | 15.15 | 1084 | 1086 | ---- | ---- | 0.12 ± 0.01 | 0.34 ± 0.01 | MS, KI |
| Oxygenated monoterpenes | |||||||||
| 15 | α-Fenchyl alcohol | 12.70 | 1106 | 1107 | 0.39 ± 0.02 | ---- | ---- | ---- | MS, KI |
| 16 | cis-Verbenol | 13.65 | 1142 | 1144 | 0.34 ± 0.03 | ---- | ---- | ---- | MS, KI |
| 17 | Menthol | 14.17 | 1181 | 1182 | ---- | ---- | ---- | 0.54 ± 0.01 | MS, KI |
| 18 | α-Terpineol | 20.28 | 1189 | 1189 | 0.44 ± 0.03 | ---- | ---- | ---- | MS, KI |
| 19 | Thymol methyl ether | 20.41 | 1236 | 1237 | ---- | ---- | ---- | 0.03 ± 0.01 | MS, KI |
| 20 | Dihydrocarvone | 21.31 | 1243 | 1242 | ---- | ---- | ---- | 0.06 ± 0.01 | MS, KI |
| 21 | Carvacrol | 22.79 | 1304 | 1304 | ---- | ---- | 94.40 ± 0.52 | 75.91 ± 0.61 | MS, KI |
| 22 | α-Terpinenyl acetate | 25.21 | 1349 | 1350 | 0.51 ± 0.02 | ---- | ---- | ---- | MS, KI |
| 23 | Thymyl acetate | 28.55 | 1357 | 1357 | ---- | ---- | 0.05 ± 0.01 | 0.06 ± 0.01 | MS, KI |
| Sesquiterpene hydrocarbons | |||||||||
| 24 | α-Cubebene | 21.37 | 1351 | 1351 | ---- | 1.10 ± 0.03 | ---- | ---- | MS, KI |
| 25 | α-Bourbonene | 25.43 | 1374 | 1374 | ---- | 0.48 ± 0.04 | ---- | ---- | MS, KI |
| 26 | α-Copaene | 25.77 | 1377 | 1376 | 0.47 ± 0.02 | 1.34 ± 0.04 | ---- | ---- | MS, KI |
| 27 | β-Elemene | 26.52 | 1385 | 1384 | ---- | 0.42 ± 0.03 | ---- | ---- | MS, KI |
| 28 | Longifolene | 27.61 | 1401 | 1402 | ---- | ---- | 0.07 ± 0.01 | ---- | MS, KI |
| 29 | cis-Caryophyllene | 27.89 | 1410 | 1409 | 0.97 ± 0.03 | 1.72 ± 0.05 | 0.33 ± 0.02 | 0.17 ± 0.01 | MS, KI |
| 30 | Clovene | 29.37 | 1426 | 1425 | ---- | ---- | 0.13 ± 0.01 | ---- | MS, KI |
| 31 | Aromadendrene | 29.55 | 1440 | 1439 | 3.86 ± 0.04 | 0.33 ± 0.03 | 0.10 ± 0.01 | 0.04 ± 0.01 | MS, KI |
| 32 | β-Neoclovene | 30.56 | 1455 | 1454 | ---- | ---- | 0.04 ± 0.01 | ---- | MS, KI |
| 33 | α-Gurjunene | 30.74 | 1475 | 1475 | 0.68 ± 0.03 | ---- | ---- | ---- | MS, KI |
| 34 | Germacrene-D | 31.99 | 1480 | 1480 | 2.21 ± 0.06 | 7.25 ± 0.46 | ---- | ---- | MS, KI |
| 35 | β-Selinene | 33.44 | 1485 | 1485 | ---- | ---- | 0.04 ± 0.01 | ---- | MS, KI |
| 36 | Guaia-1(10),11-diene | 33.72 | 1488 | 1488 | ---- | 1.69 ± 0.07 | ---- | ---- | MS, KI |
| 37 | β-Muurolene | 34.08 | 1493 | 1493 | 0.57 ± 0.04 | 5.55 ± 0.06 | ---- | 0.05 ± 0.01 | MS, KI |
| 38 | α-Muurolene | 34.54 | 1498 | 1499 | 0.72 ± 0.01 | 7.64 ± 0.58 | ---- | ---- | MS, KI |
| 39 | δ-Cadinene | 35.03 | 1525 | 1524 | 7.04 ± 0.31 | 25.13 ± 0.43 | 0.06 ± 0.01 | 0.08 ± 0.01 | MS, KI |
| 40 | Cadina-1(10),4-diene | 35.89 | 1530 | 1530 | ---- | 1.83 ± 0.05 | ---- | ---- | MS, KI |
| Oxygenated sesquiterpenes | |||||||||
| 41 | Spathulenol | 28.61 | 1515 | 1516 | ---- | 0.51 ± 0.02 | ---- | ---- | MS, KI |
| 42 | 1,5-Epoxy-salvial-4(14)-ene | 29.64 | 1548 | 1548 | 0.86 ± 0.02 | 0.78 ± 0.03 | ---- | ---- | MS, KI |
| 43 | Ledol | 29.67 | 1564 | 1565 | 0.69 ± 0.04 | 0.58 ± 0.03 | ---- | ---- | MS, KI |
| 44 | 4-epi-Cubebol | 29.29 | 1572 | 1574 | ---- | 0.30 ± 0.01 | ---- | ---- | MS, KI |
| 45 | Germacrene D-4-ol | 30.71 | 1578 | 1577 | 5.65 ± 0.31 | ---- | ---- | ---- | MS, KI |
| 46 | Τ-Cadinol | 32.23 | 1638 | 1640 | 2.06 ± 0.05 | 7.61 ± 0.12 | ---- | ---- | MS, KI |
| 47 | Cubenol | 32.31 | 1641 | 1642 | ---- | 0.35 ± 0.01 | ---- | ---- | MS, KI |
| 48 | Τ-Muurolol | 32.74 | 1646 | 1646 | 12.88 ± 0.15 | ---- | ---- | ---- | MS, KI |
| 49 | α-Cadinol | 34.16 | 1654 | 1653 | 2.01 ± 0.03 | 5.49 ± 0.10 | ---- | ---- | MS, KI |
| 50 | Alloaromadendrene oxide | 39.89 | 1672 | 1672 | 0.53 ± 0.01 | 6.09 ± 0.23 | ---- | ---- | MS, KI |
| 51 | 6-Epi-shyobunol | 41.92 | 1682 | 1680 | 33.00 ± 0.72 | 0.29 ± 0.01 | ---- | ---- | MS, KI |
| Non-oxygenated hydrocarbons | |||||||||
| 52 | Nonadecane | 45.04 | 1900 | 1900 | ---- | ---- | 0.03 ± 0.01 | 0.03 ± 0.01 | MS, KI |
| 53 | Heptacosane | 51.54 | 2700 | 2700 | ---- | ---- | 0.01 ± 0.00 | 0.10 ± 0.02 | MS, KI |
| Monoterpene hydrocarbons | 24.12 | 23.52 | 4.74 | 22.93 | |||||
| Oxygenated monoterpenes | 1.68 | 0 | 94.45 | 76.6 | |||||
| Sesquiterpene hydrocarbons | 16.52 | 54.48 | 0.73 | 0.34 | |||||
| Oxygenated sesquiterpenes | 57.68 | 22 | 0 | 0 | |||||
| Non-oxygenated hydrocarbons | 0 | 0 | 0.04 | 0.13 | |||||
| Total | 100% | 100% | 100% | 100% | |||||
| Plant Species | Extraction Method | Conc. (µL L−1) | Inhibition (%) * | ||
|---|---|---|---|---|---|
| Germination | Shoot Growth | Root Growth | |||
| Teucrium polium | Hydrodistillation | 250 | 19.6 ± 0.95 | 8.2 ± 0.74 | 14.9 ± 2.76 |
| 500 | 33.0 ± 1.50 | 10.8 ± 0.35 | 23.2 ± 2.03 | ||
| 750 | 43.0 ± 1.98 | 21.5 ± 1.47 | 35.8 ± 1.52 | ||
| 1000 | 51.9 ± 2.09 | 31.4 ± 1.63 | 54.4 ± 1.90 | ||
| Microwave-assisted extraction | 250 | 24.1 ± 1.01 | 0.4 ± 0.35 | 15.3 ± 3.41 | |
| 500 | 37.5 ± 1.13 | 6.0 ± 2.03 | 18.7 ± 2.45 | ||
| 750 | 40.0 ± 2.01 | 17.7 ± 1.44 | 26.0 ± 2.94 | ||
| 1000 | 50.9 ± 2.25 | 39.8 ± 1.30 | 42.3 ± 2.75 | ||
| p-value (0.05) Extraction method (E) | 0.438 ns | 0.0150 * | <0.001 *** | ||
| p-value (0.05) Concentration (C) | <0.001 *** | <0.001 *** | <0.001 *** | ||
| p-value (0.05) Interaction (E × C) | 0.878 ns | <0.001 *** | 0.031 * | ||
| Thymus decussatus | Hydrodistillation | 25 | 50.9 ± 1.59 | 33.0 ± 2.37 | 11.6 ± 0.98 |
| 50 | 55.4 ± 1.55 | 45.7 ± 1.42 | 25.2 ± 3.06 | ||
| 75 | 68.8 ± 2.11 | 52.1 ± 1.48 | 49.8 ± 1.22 | ||
| 100 | 77.7 ± 2.36 | 85.8 ± 1.18 | 84.6 ± 1.02 | ||
| Microwave-assisted extraction | 25 | 42.0 ± 1.11 | 45.6 ± 1.18 | 42.1 ± 2.54 | |
| 50 | 50.9 ± 1.99 | 58.5 ± 1.78 | 56.1 ± 1.47 | ||
| 75 | 68.8 ± 2.25 | 65.7 ± 1.18 | 70.6 ± 1.22 | ||
| 100 | 86.6 ± 2.98 | 87.4 ± 3.55 | 89.9 ± 2.04 | ||
| p-value (0.05) Extraction method (E) | 0.715 ns | <0.001 *** | <0.001 *** | ||
| p-value (0.05) Concentration (C) | <0.001 *** | <0.001 *** | <0.001 *** | ||
| p-value (0.05) Interaction (E × C) | 0.250 ns | 0.007 ** | <0.001 *** | ||
| Plant | Extraction Method | Bacteria | Fungi | ||||
|---|---|---|---|---|---|---|---|
| Gram-Positive | Gram-Negative | ||||||
| B. subtilis ATCC6633 | S. aureus ATCC29213 | E. coli ATCC25922 | C. albicans ATCC10321 | A. niger NRC53 | F. solani NRC15 | ||
| Teucrium polium | HD | 10.00 ± 0.79 * | 10.00 ± 2.12 | 10.00 ± 0.71 | NA | NA | NA |
| MAE | 11.00 ± 2.12 | 11.00 ± 0.71 | 10.00 ± 1.41 | NA | NA | NA | |
| Thymus decussatus | HD | 39.00 ± 0.71 | 34.00 ± 1.40 | 35.00 ± 2.12 | 34.00 ± 2.12 | 47.00 ± 2.12 | 40.00 ± 2.83 |
| MAE | 38.00 ± 2.90 | 35.00 ± 0.70 | 36.00 ± 1.34 | 35.00 ± 0.00 | 48.00 ± 3.14 | 40.00 ± 1.78 | |
| Thiophenicol (100 μg/disc) | 30.0 ± 0.71 | 30.00 ± 0.71 | 27.00 ± 0.77 | 25.00 ± 1.55 | ---- | ---- | |
| Treflucan (100 μg/disc) | ---- | ---- | ---- | 29.00 ± 0.70 | 13.00 ± 0.14 | 11.00 ± 0.71 | |
| Plant | Extraction Method | Bacteria | Fungi | ||||
|---|---|---|---|---|---|---|---|
| Gram-Positive | Gram-Negative | ||||||
| B. subtilis ATCC6633 | S. aureus ATCC29213 | E. coli ATCC25922 | C. albicans ATCC10321 | A. niger NRC53 | F. solani NRC15 | ||
| Teucrium polium | HD | 62.50 | 125.00 | 250.00 | ---- | ---- | ---- |
| MAE | 62.50 | 125.00 | 250.00 | ---- | ---- | ---- | |
| Thymus decussatus | HD | 3.91 | 1.95 | 0.98 | 3.91 | 0.49 | 7.81 |
| MAE | 3.91 | 1.95 | 0.98 | 3.91 | 0.49 | 7.81 | |
| Thiophenicol | 3.13 | 3.13 | 6.25 | ---- | ---- | ---- | |
| Treflucan | ---- | ---- | ---- | 12.5 | 50 | 50 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saleh, I.; Abd-ElGawad, A.; El Gendy, A.E.-N.; Abd El Aty, A.; Mohamed, T.; Kassem, H.; Aldosri, F.; Elshamy, A.; Hegazy, M.-E.F. Phytotoxic and Antimicrobial Activities of Teucrium polium and Thymus decussatus Essential Oils Extracted Using Hydrodistillation and Microwave-Assisted Techniques. Plants 2020, 9, 716. https://doi.org/10.3390/plants9060716
Saleh I, Abd-ElGawad A, El Gendy AE-N, Abd El Aty A, Mohamed T, Kassem H, Aldosri F, Elshamy A, Hegazy M-EF. Phytotoxic and Antimicrobial Activities of Teucrium polium and Thymus decussatus Essential Oils Extracted Using Hydrodistillation and Microwave-Assisted Techniques. Plants. 2020; 9(6):716. https://doi.org/10.3390/plants9060716
Chicago/Turabian StyleSaleh, Ibrahim, Ahmed Abd-ElGawad, Abd El-Nasser El Gendy, Abeer Abd El Aty, Tarik Mohamed, Hazem Kassem, Fahd Aldosri, Abdelsamed Elshamy, and Mohamed-Elamir F. Hegazy. 2020. "Phytotoxic and Antimicrobial Activities of Teucrium polium and Thymus decussatus Essential Oils Extracted Using Hydrodistillation and Microwave-Assisted Techniques" Plants 9, no. 6: 716. https://doi.org/10.3390/plants9060716
APA StyleSaleh, I., Abd-ElGawad, A., El Gendy, A. E.-N., Abd El Aty, A., Mohamed, T., Kassem, H., Aldosri, F., Elshamy, A., & Hegazy, M.-E. F. (2020). Phytotoxic and Antimicrobial Activities of Teucrium polium and Thymus decussatus Essential Oils Extracted Using Hydrodistillation and Microwave-Assisted Techniques. Plants, 9(6), 716. https://doi.org/10.3390/plants9060716









