Prospects of Arbuscular Mycorrhizal Fungi Utilization in Production of Allium Plants
Abstract
1. Introduction
- the chance to increase selenium accumulation in Allium plants and to produce products with high concentration of natural anti-carcinogens [19].
2. AMF Effect on Yield and Growth of Allium Species
3. Effects of AMF on Physiological, Quality, and Antioxidant Indicators in Allium Species
4. Effects of AMF on Elemental Composition
5. Selenium–AMF Interaction
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chen, M.; Arato, M.; Borghi, L.; Nouri, E.; Reinhardt, D. Beneficial Services of Arbuscular Mycorrhizal Fungi—From Ecology to Application. Front. Plant Sci. 2018, 9, 1270. [Google Scholar] [CrossRef]
- Khan, A.G. Mycorrhizoremediation—An enhanced form of phytoremediation. J. Zhejiang Univ. Sci. B 2006, 7, 503–514. [Google Scholar] [CrossRef]
- Bahadur, A.; Batool, A.; Nasir, F.; Jiang, S.; Mingsen, Q.; Zhang, Q.; Pan, J.; Liu, Y.; Feng, H. Mechanistic Insights into Arbuscular Mycorrhizal Fungi-Mediated Drought Stress Tolerance in Plants. Int. J. Mol. Sci. 2019, 20, 4199. [Google Scholar] [CrossRef]
- Zhu, X.; Song, F.; Liu, F. Arbuscular Mycorrhizal Fungi and Tolerance of Temperature Stress in Plants. In Arbuscular Mycorrhizas and Stress Tolerance of Plants; Wu, Q.-S., Ed.; Springer Science and Business Media LLC: Singapore, 2017; pp. 163–194. [Google Scholar]
- Begum, N.; Qin, C.; Ahanger, M.A.; Raza, S.; Khan, M.I.; Ashraf, M.; Ahmed, N.; Zhang, L. Role of Arbuscular Mycorrhizal Fungi in Plant Growth Regulation: Implications in Abiotic Stress Tolerance. Front. Plant Sci. 2019, 10, 1068. [Google Scholar] [CrossRef]
- Júnior, C.V.T.; Araujo, K.E.C.; De Souza, S.R.; Schultz, N.; Júnior, O.J.S.; Sperandio, M.V.L.; Zilli, J. Édson Plant-mycorrhizal fungi interaction and response to inoculation with different growth-promoting fungi. Pesqui. Agropecuária Bras. 2019, 54. [Google Scholar]
- Berruti, A.; Lumini, E.; Balestrini, R.M.; Bianciotto, V. AMF as natural biofertilizers: Lets benefits from past successes. Front. Microbiol. 2015, 6, 1559. [Google Scholar] [CrossRef]
- White, J.; Kingsley, K.L.; Zhang, Q.; Verma, R.; Obi, N.; Dvinskikh, S.; Elmore, M.T.; Verma, S.K.; Gond, S.K.; Kowalski, K.P. Review: Endophytic microbes and their potential applications in crop management. Pest Manag. Sci. 2019, 75, 2558–2565. [Google Scholar] [CrossRef]
- Caruso, G.; Golubkina, N.; Seredin, T.; Sellitto, B. Utilization of AMF in production of Allium species. Veg. Crop. Russ. 2018, 3, 85–90. [Google Scholar]
- Golubkina, N.A.; Caruso, G.; Vidican, R.; Sellitto, V.M.; Pietrantonio, L.D.; Martorana, M.E.; Lumini, E.F.; Balestrini, R. Effetto dei funghi micorrizici su fisiologia, produttività e qualità di cipolla, aglio e porro. Agrisicilia 2018, 4, 41–44. [Google Scholar]
- Golubkina, N.; Zamana, S.; Seredin, T.; Poluboyarinov, P.A.; Sokolov, S.; Baranova, H.; Krivenkov, L.; Pietrantonio, L.; Caruso, G. Effect of Selenium Biofortification and Beneficial Microorganism Inoculation on Yield, Quality and Antioxidant Properties of Shallot Bulbs. Plants 2019, 8, 102. [Google Scholar] [CrossRef]
- Deressa, T.G.; Schenk, M.K. Contribution of roots and hyphae to phosphorus uptake of mycorrhizal onion (Allium cepaL.)-A mechanistic modeling approach. J. Plant Nutr. Soil Sci. 2008, 171, 810–820. [Google Scholar] [CrossRef]
- Bowling, D.J.F.; Mengel, K.; Kirkby, E.A. Principles of Plant Nutrition. J. Ecol. 1980, 68, 324. [Google Scholar] [CrossRef]
- Greenwood, D.J.; Gerwitz, A.; Stone, D.A.; Barnes, A. Root development of vegetable crops. Plant Soil 1982, 68, 75–96. [Google Scholar] [CrossRef]
- Sanders, I.R.; Fitter, A. Evidence for differential responses between host-fungus combinations of vesicular-arbuscular mycorrhizas from a grassland. Mycol. Res. 1992, 96, 415–419. [Google Scholar] [CrossRef]
- Bever, J.D.; Morton, J.B.; Antonovics, J.; Schultz, P.A. Host-Dependent Sporulation and Species Diversity of Arbuscular Mycorrhizal Fungi in a Mown Grassland. J. Ecol. 1996, 84, 71. [Google Scholar] [CrossRef]
- FAOSTAT 2015. Available online: http://faostat3.fao.org/browse/Q/QC/E (accessed on 8 August 2015).
- Zeng, Y.; Li, Y.; Yang, J.; Pu, X.; Du, J.; Yang, X.; Yang, T.; Yang, S. Therapeutic Role of Functional Components in Alliums for Preventive Chronic Disease in Human Being. Evid. Based Complement. Altern. Med. 2017, 2017, 1–13. [Google Scholar] [CrossRef]
- Sugihara, S.; Kondo, M.; Chihara, Y.; Yuji, M.; Hattori, H.; Yoshida, M. Preparation of selenium-enriched sprouts and identification of their selenium species by high-performance liquid chromatography-inductively coupled plasma mass spectrometry. Biosci. Biotechnol. Biochem. 2004, 68, 193–199. [Google Scholar] [CrossRef]
- Brewster, J.L. Onions and Other Vegetable Alliums; CAB International: Wallingford, UK, 2008. [Google Scholar]
- Sekara, A.; Pokluda, R.; Del Vacchio, L.; Somma, S.; Caruso, G.; Agnieszka, S.; Robert, P.; Del, V.L.; Silvano, S.; Gianluca, C. Interactions among genotype, environment and agronomic practices on production and quality of storage onion (Allium cepa L.)—A review. Hortic. Sci. 2017, 44, 21–42. [Google Scholar] [CrossRef]
- Etana, M.B.; Aga, M.C.; Fufa, B.O. Major onion (Allium cepa L.) production challenges in Ethiopia: A review. J. Biol. Agric. Healthc. 2019, 9, 42–47. [Google Scholar]
- Suhardi, H.A. Effect of planting date and fungicide applications on the intensity of anthracnose on shallot. Indones. J. Hortic. 1996, 6, 172–180. [Google Scholar]
- Mohammadi, K.; Khalesro, S.; Sohrabi, Y.; Heidari, G.A. Review: Beneficial effects of the mycorrhizal fungi for plant growth. J. Appl. Environ. Biol. Sci. 2011, 1, 310–319. [Google Scholar]
- Plenchette, C.; Clermont-Dauphin, C.; Meynard, J.M.; Fortin, J.A. Managing arbuscular mycorrhizal fungi in cropping systems. Can. J. Plant Sci. 2005, 85, 31–40. [Google Scholar] [CrossRef]
- Serra, A.D.B. Agronomy of onions. In Allium Crop Science: Recent Advances; Rabinowitch, H.D., Currah, L., Eds.; CABI Publishing: New York, NY, USA, 2002; pp. 187–232. [Google Scholar]
- Scullion, J.; Eason, W.; Scott, E. The effectivity of arbuscular mycorrhizal fungi from high input conventional and organic grassland and grass-arable rotations. Plant Soil 1998, 204, 243–254. [Google Scholar] [CrossRef]
- Patharajan, S.; Raaman, N. Influence of arbuscular mycorrhizal fungi on growth and selenium uptake by garlic plants. Arch. Phytopathol. Plant Prot. 2012, 45, 138–151. [Google Scholar] [CrossRef]
- Larsen, E.H.; Łobiński, R.; Burger-Meÿer, K.; Hansen, M.; Ruzik, L.; Mazurowska, L.; Rasmussen, P.H.; Sloth, J.J.; Scholten, O.; Kik, C. Uptake and speciation of selenium in garlic cultivated in soil amended with symbiotic fungi (mycorrhiza) and selenate. Anal. Bioanal. Chem. 2006, 385, 1098–1108. [Google Scholar] [CrossRef]
- Mollavali, M.; Bolandnazar, S.; Nazemieh, H.; Aliasgharzad, N. The effect of mycorrhizal fungi on antioxidant activity of various cultivars of onion (Allium cepa L.). Int. J. Biosci. 2015, 6, 66–79. [Google Scholar]
- Kostin, M.; Podkovyrov, I. Using mycorrhiza in onion growing: Russian experience. GISAP Biol. Veter Med. Agric. Sci. 2017, 7–9. [Google Scholar] [CrossRef]
- Bolandnazar, S. The effect of mycorrhizal fungi on onion (Allium cepa L.) growth and yield under three irrigation intervals at field condition. J. Food Agric. Environ. 2009, 7, 360–362. [Google Scholar]
- Shinde, S.K.; Shinde, B.P. Consequence of arbuscular mycorrhiza on enhancement, growth and yield of onion (Allium cepa L.). Int. J. Life. Sci. Sci. Res. 2016, 2, 206–211. [Google Scholar]
- Bolandnazar, S.; Aliasgarzad, N.; Neishabury, M.; Chaparzadeh, N. Mycorrhizal colonization improves onion (Allium cepa L.) yield and water use efficiency under water deficit condition. Sci. Hortic. 2007, 114, 11–15. [Google Scholar] [CrossRef]
- Azcón, R.; Tobar, R.M. Activity of nitrate reductase and glutamine synthetase in shoot and root of mycorrhizal Allium cepa. Plant Sci. 1998, 133, 1–8. [Google Scholar] [CrossRef]
- Mohamed, A.A.; Eweda, W.E.; Heggo, A.; Hassan, E.A. Effect of dual inoculation with arbuscular mycorrhizal fungi and sulphur-oxidising bacteria on onion (Allium cepa L.) and maize (Zea mays L.) grown in sandy soil under green house conditions. Ann. Agric. Sci. 2014, 59, 109–118. [Google Scholar] [CrossRef]
- Bettoni, M.M.; Mogor, Á.F.; Pauletti, V.; Goicoechea, N. Growth and metabolism of onion seedlings as affected by the application of humic substances, mycorrhizal inoculation and elevated CO2. Sci. Hortic. 2014, 180, 227–235. [Google Scholar] [CrossRef]
- Albrechtová, L.S.J.; Latr, A.; Nedorost, L.; Pokluda, R.; Posta, K.; Vosátka, M. Dual Inoculation with Mycorrhizal and Saprotrophic Fungi Applicable in Sustainable Cultivation Improves the Yield and Nutritive Value of Onion. Sci. World J. 2012, 2012, 1–8. [Google Scholar] [CrossRef]
- Bago, B.; Azcon, C. Changes in the rhizospheric pH induced by arbuscular mycorrhiza formation in onion (Allium cepa L.). J. Plant Nutr. Soil Sci. 1997, 160, 333–339. [Google Scholar] [CrossRef]
- Galván, G.A.; Kuyper, T.W.; Burger, K.; Keizer, L.C.P.; Hoekstra, R.F.; Kik, C.; Scholten, O.E. Genetic analysis of the interaction between Allium species and arbuscular mycorrhizal fungi. Theor. Appl. Genet. 2011, 122, 947–960. [Google Scholar] [CrossRef]
- Scholten, O.E.; Galvan-Vivero, G.; Burger-Meijer, B.J.; Kik, D.C. Effect of arbuscular mycorrhiza fungi on growth and development of onion and wild relatives. In Proceedings of the Poster at: Joint Organic Congress, Odense, Denmark, 30–31 May 2006. [Google Scholar]
- Priyadharsini, P.; Pandey, R.; Muthukumar, T. Arbuscular mycorrhizal and dark septate fungal associations in shallot (Allium cepa L. var. aggregatum) under conventional agriculture. Acta Bot. Croat. 2012, 71, 159–175. [Google Scholar] [CrossRef]
- Kučová, L.; Kopta, T.; Sekara, A.; Pokluda, R. Controlling Nitrate and Heavy Metals Content in Leeks (Allium porrum L.) Using Arbuscular Mycorrhizal Fungi Inoculation. Pol. J. Environ. Stud. 2018, 27, 137–143. [Google Scholar] [CrossRef]
- Kučová, L.; Záhora, J.; Pokluda, R. Effect of mycorrhizal inoculation of leek Allium porrum L. on mineral nitrogen leaching. Hortic. Sci. 2016, 43, 195–202. [Google Scholar] [CrossRef]
- Han, Y.; Douds, J.D.D.; Boateng, A.A.; Douds, D.D. Effect of biochar soil-amendments on Allium porrum growth and arbuscular mycorrhizal fungus colonization. J. Plant Nutr. 2015, 39, 1654–1662. [Google Scholar] [CrossRef]
- Perner, H.; Schwar, D.; George, E. Effect of mycorrhyzal inoculation and compost supply on growth and nutrient uptake of young leek plants grown on peat-based substrates. Hortic. Sci. 2006, 4, 628–632. [Google Scholar]
- Borde, M.; Dudhane, M.; Jite, P.K. Role of bioinoculant (AM Fungi) increasing in growth, flavor content and yield in Allium sativum L. under field condition. Not. Bot. Hortic. Agrobot. 2009, 37, 124–128. [Google Scholar]
- Al-Karaki, G.N. Field response of garlic inoculated with arbuscular mycorrhizal fungi to phosphorus fertilization. J. Plant Nutr. 2002, 25, 747–756. [Google Scholar] [CrossRef]
- Smith, S.E.; Smith, F.A. Fresh perspectives on the roles of arbuscular mycorrhizal fungi in plant nutrition and growth. Mycologia 2012, 104, 1–13. [Google Scholar] [CrossRef]
- Johnson, N.C.; Graham, J.H.; Smith, F.A. Functioning of mycorrhizal associations along the mutualism-parasitism continuum. New Phytol. 1997, 135, 575–585. [Google Scholar] [CrossRef]
- Helgason, T.; Daniell, T.; Husband, R.; Fitter, A.H.; Young, J.P.W. Ploughing up the wood-wide web? Nature 1998, 394, 431. [Google Scholar] [CrossRef]
- Daniell, T.; Husband, R.; Fitter, A.H.; Young, J.P.W. Molecular diversity of arbuscular mycorrhizal fungi colonizing arable crops. FEMS Microbiol. Ecol. 2001, 36, 203–209. [Google Scholar] [CrossRef]
- Jansa, J.; Mozafar, A.; Anken, T.; Ruh, R.; Sanders, I.; Frossard, E. Diversity and structure of AMF communities as affected by tillage in a temperate soil. Mycorrhiza 2002, 12, 225–234. [Google Scholar]
- Galván, G.A.; Parádi, I.; Burger, K.; Baar, J.; Kuyper, T.W.; Scholten, O.E.; Kik, C. Molecular diversity of arbuscular mycorrhizal fungi in onion roots from organic and conventional farming systems in the Netherlands. Mycorrhiza 2009, 19, 317–328. [Google Scholar] [CrossRef]
- Mayr, R.; Godoy, R. Seasonal patterns in vesicular-arbuscular mycorrhiza in Melic-Beech Forest. Agric. Ecosyst. Environ. 1990, 29, 281–288. [Google Scholar] [CrossRef]
- Jansa, J.; Smith, F.A.; Smith, S.E. Are there benefits of simultaneous root colonization by different arbuscular mycorrhizal fungi? New Phytol. 2008, 177, 779–789. [Google Scholar] [CrossRef]
- Golubkina, N.A.; Amagova, Z.; Matsadze, V.; Zamana, S.; Tallarita, A.; Caruso, G. Effects of Arbuscular Mycorrhizal Fungi on Yield, Biochemical Characteristics, and Elemental Composition of Garlic and Onion under Selenium Supply. Plants 2020, 9, 84. [Google Scholar] [CrossRef]
- Tawaraya, K.; Tokairin, K.; Wagatsuma, T. Dependence of Allium fistulosum cultivars on the arbuscular mycorrhizal fungus, Glomus fasciculatum. Appl. Soil Ecol. 2001, 17, 119–124. [Google Scholar] [CrossRef]
- Azcon, R.; Ocampo, J.A. Factors affecting the vesicular-arbuscular infection and mycorrhizal dependency of thirteen wheat cultivars. New Phytol. 1981, 87, 677–685. [Google Scholar] [CrossRef]
- Baon, J.B.; Smith, S.E.; Alston, A.M. Mycorrhizal responses of barley cultivars differing in P efficiency. Plant Soil 1993, 157, 97–105. [Google Scholar] [CrossRef]
- Bryla, D.; Koide, R.T. Role of mycorrhizal infection in the growth and reproduction of wild vs. cultivated plants. Oecologia 1990, 84, 82–92. [Google Scholar] [CrossRef]
- Bååth, E.; Spokes, J. The effect of added nitrogen and phosphorus on mycorrhizal growth response and infection in Allium schoenoprasum. Can. J. Bot. 1989, 67, 3227–3232. [Google Scholar] [CrossRef]
- Fredotović, Ž.; Puizina, J. Edible Allium species: Chemical composition, biological activity and health effects. Ital. J. Food Sci. 2019, 31, 19–39. [Google Scholar]
- Hanen, N.; Fattouch, S.; Ammar, E.; Neffati, M. Allium Species, Ancient Health Food for the Future; Chapter 17 in Scientific, Health and Social Aspects of the Food Industry; Valdez, B., Ed.; InTechOpen: London, UK, 2012; pp. 343–354. [Google Scholar]
- Kertesz, M. The role of soil microbes in plant sulphur nutrition. J. Exp. Bot. 2004, 55, 1939–1945. [Google Scholar] [CrossRef]
- Guo, T.; Zhang, J.; Christie, P.; Li, X. Effects of Arbuscular Mycorrhizal Fungi and Ammonium: Nitrate Ratios on Growth and Pungency of Onion Seedlings. J. Plant Nutr. 2006, 29, 1047–1059. [Google Scholar] [CrossRef][Green Version]
- Guo, T.; Zhang, J.; Christie, P.; Li, X. Influence of Nitrogen and Sulfur Fertilizers and Inoculation with Arbuscular Mycorrhizal Fungi on Yield and Pungency of Spring Onion. J. Plant Nutr. 2006, 29, 1767–1778. [Google Scholar] [CrossRef]
- Salvioli, A.; Bonfante, P. Systems biology and “omics” tools: A cooperation for next-generation mycorrhizal studies. Plant Sci. 2013, 203, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Shuab, R.; Lone, R.; Naidu, J.; Sharma, V.; Imtiyaz, S.; Koul, K.K. Benefits of inoculation of arbuscular mycorrhizal fungi on growth and development of onion (Allium cepa) plant. American-Eurasian. J. Agric. Environ. Sci. 2014, 14, 527–535. [Google Scholar] [CrossRef]
- Rolland, F.; Baena-González, E.; Sheen, J. Sugar sensing and signaling in plants: Conserved and Novel Mechanisms. Annu. Rev. Plant Boil. 2006, 57, 675–709. [Google Scholar] [CrossRef]
- Lone, R.; Shuab, R.; Wani, K.; Ganaie, M.A.; Tiwari, A.; Koul, K. Mycorrhizal influence on metabolites, indigestible oligosaccharides, mineral nutrition and phytochemical constituents in onion (Allium cepa L.) plant. Sci. Hortic. 2015, 193, 55–61. [Google Scholar] [CrossRef]
- Regvar, M.; Vogel-Mikuš, K.; Ševerkar, T. Effect of AMF inoculum from field isolates on the yield of green pepper, parsley, carrot, and tomato. Folia Geobot. Phytotaxon. 2003, 38, 223–234. [Google Scholar] [CrossRef]
- Igamberdiev, A.U.; Eprintsev, A.T. Organic Acids: The Pools of Fixed Carbon Involved in Redox Regulation and Energy Balance in Higher Plants. Front. Plant Sci. 2016, 7, 513. [Google Scholar] [CrossRef]
- Rozpądek, P.; Rapala-Kozik, M.; Wężowicz, K.; Grandin, A.; Karlsson, S.; Ważny, R.; Anielska, T.; Turnau, K. Arbuscular mycorrhiza improves yield and nutritional properties of onion (Allium cepa). Plant Physiol. Biochem. 2016, 107, 264–272. [Google Scholar] [CrossRef]
- Charron, G.; Furlan, V.; Bernier-Cardou, M.; Doyon, G. Response of onion plants to arbuscular mycorrhizae. 1. Effects of inoculation method and phosphorus fertilization on biomass and bulb firmness. Mycorrhiza 2001, 11, 187–197. [Google Scholar] [CrossRef]
- Charron, G.; Furlan, V.; Bernier-Cardou, M.; Doyon, G. Response of onion plants to arbuscular mycorrhizae. 2. Effects of nitrogen fertilization on biomass and bulb firmness. Mycorrhiza 2001, 11, 145–150. [Google Scholar] [CrossRef]
- Smith, S.E. Mycorrhizal Fungi Can Dominate Phosphate Supply to Plants Irrespective of Growth Responses. Plant Physiol. 2003, 133, 16–20. [Google Scholar] [CrossRef]
- Mosse, B.; Stribley, D.P.; LeTacon, F. Ecology of Mycorrhizae and Mycorrhizal Fungi. Adv. Microb. Ecol. 1981, 5, 137–210. [Google Scholar]
- Fitter, A.H. Functioning of vesicular-arbuscular mycorrhizas under field conditions. New Phytol. 1985, 99, 257–265. [Google Scholar] [CrossRef]
- Linderman, R.; Davis, E. Vesicular-Arbuscular Mycorrhiza and Plant Growth Response to Soil Amendment with Composted Grape Pomace or Its Water Extract. HortTechnology 2001, 11, 446–450. [Google Scholar] [CrossRef]
- Sato, T.; Ezawa, T.; Cheng, W.; Tawaraya, K. Release of acid phosphatase from extraradical hyphae of arbuscular mycorrhizal fungus Rhizophagus clarus. Soil Sci. Plant Nutr. 2015, 61, 269–274. [Google Scholar] [CrossRef]
- Golubkina, N.A.; Papazyan, T.T. Selenium in Food, Plants, Animals and Human Beings; Pechatny Gorod: Moscow, Russia, 2006. [Google Scholar]
- Pilon-Smits, E.A.H. On the Ecology of Selenium Accumulation in Plants. Plants 2019, 8, 197. [Google Scholar] [CrossRef]
- Bhattacharya, A. Methylselenocysteine: A promising antiangiogenic agent for overcoming drug delivery barriers in solid malignancies for therapeutic synergy with anticancer drugs. Expert Opin. Drug Deliv. 2011, 8, 749–763. [Google Scholar] [CrossRef]
- Ip, C.; Lisk, D.J. Enrichment of selenium in allium vegetables for cancer prevention. Carcinogenesis 1994, 15, 1881–1885. [Google Scholar] [CrossRef]
- El-Bayoumy, K.; Sinha, R.; Pinto, J.T.; Rivlin, R.S. Cancer Chemoprevention by Garlic and Garlic-Containing Sulfur and Selenium Compounds. J. Nutr. 2006, 136, 864S–869S. [Google Scholar] [CrossRef]
- Fairweather-Tait, S.J.; Bao, Y.; Broadley, M.R.; Collings, R.; Ford, D.; Hesketh, J.E.; Hurst, R. Selenium in Human Health and Disease. Antioxid. Redox Signal. 2011, 14, 1337–1383. [Google Scholar] [CrossRef]
- Dwivedi, S.; Al-Khedhairy, A.; Ahamed, M.; Musarrat, J. Biomimetic Synthesis of Selenium Nanospheres by Bacterial Strain JS-11 and Its Role as a Biosensor for Nanotoxicity Assessment: A Novel Se-Bioassay. PLoS ONE 2013, 8, e57404. [Google Scholar] [CrossRef] [PubMed]
- Duran, P.; Acuña, J.; Jorquera, M.; Azcón, R.; Borie, F.; Cornejo, P.; Mora, M.D.L.L. Enhanced selenium content in wheat grain by co-inoculation of selenobacteria and arbuscular mycorrhizal fungi: A preliminary study as a potential Se biofortification strategy. J. Cereal Sci. 2013, 57, 275–280. [Google Scholar] [CrossRef]
- Sanmartín, C.; Garmendia, I.; Romano, B.; Díaz, M.; Palop, J.A.; Goicoechea, N. Mycorrhizal inoculation affected growth, mineral composition, proteins and sugars in lettuces biofortified with organic or inorganic selenocompounds. Sci. Hortic. 2014, 180, 40–51. [Google Scholar] [CrossRef]
- Yü, Y.; Zhang, S.; Wen, B.; Huang, H.; Luo, L. Accumulation and Speciation of Selenium in Plants as Affected by Arbuscular Mycorrhizal Fungus Glomus mosseae. Boil. Trace Elem. Res. 2011, 143, 1789–1798. [Google Scholar] [CrossRef]
- González-Morales, S.; Pérez-Labrada, F.; García-Enciso, E.L.; Leija-Martínez, P.; Medrano-Macías, J.; Dávila-Rangel, I.E.; Juárez-Maldonado, A.; Rivas-Martínez, E.N.; Benavides-Mendoza, A. Selenium and sulfur to produce Allium functional crops. Molecules 2017, 22, 558. [Google Scholar] [CrossRef]
- Minard, H.R.G. Effect of clove size, spacing, fertilisers, and lime on yield and nutrient content of garlic (Allium sativum). N. Z. J. Exp. Agric. 1978, 6, 139–143. [Google Scholar] [CrossRef]
- Randle, W.M.; Kopsell, D.E.; Kopsell, D.A.; Snyder, R.L. Total sulfur and sulfate accumulation in onion is affected by sulfur fertility. J. Plant Nutr. 1999, 22, 45–51. [Google Scholar] [CrossRef]
- Bloem, E.; Haneklaus, S.; Schnug, E. Influence of Nitrogen and Sulfur Fertilization on the Alliin Content of Onions and Garlic. J. Plant Nutr. 2004, 27, 1827–1839. [Google Scholar] [CrossRef]
- Shannon, M.; Grieve, C. Tolerance of vegetable crops to salinity. Sci. Hortic. 1998, 78, 5–38. [Google Scholar] [CrossRef]
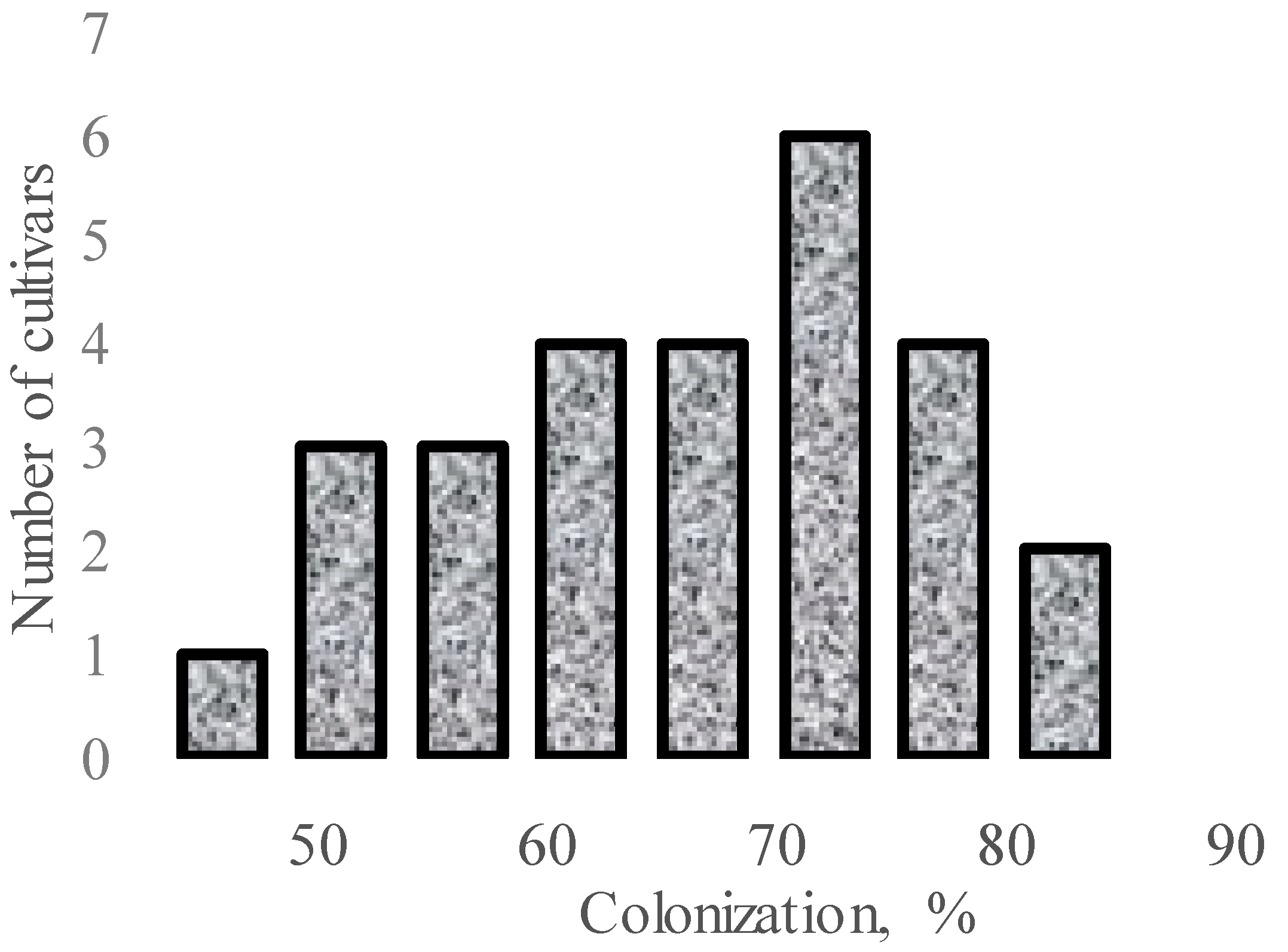
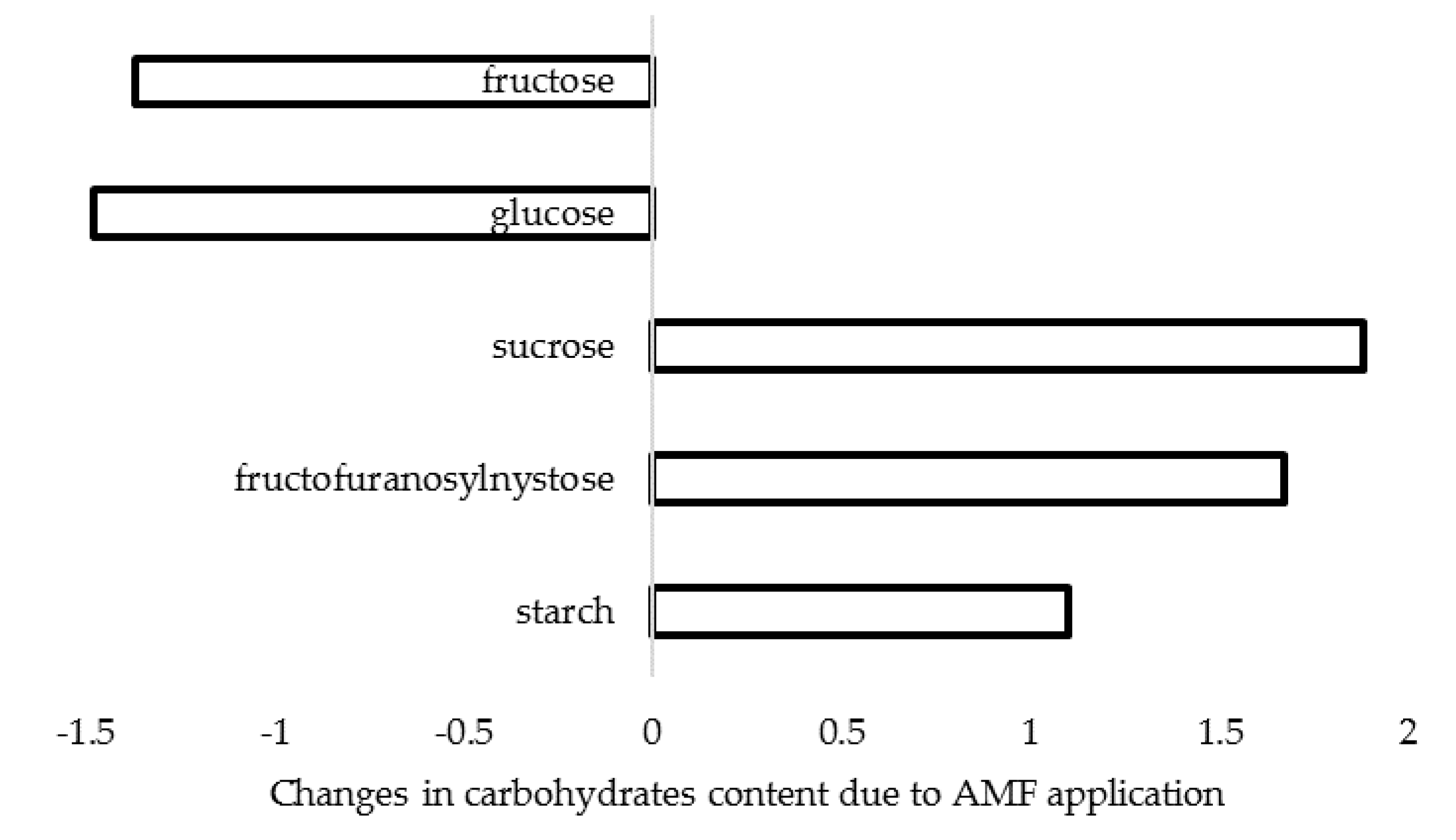

| AMF | Effect | References |
|---|---|---|
| Allium cepa | ||
| Glomus versiforme | Increase in antioxidant activity (AOA). Intervarietal differences in red, pink, yellow, and white A. cepa varieties. | [30] |
| G. mosseae, G. soronatum, G. caledonium, G. geosporum | The highest crop yield under G. mosseae and G. caledonium application. | [31] |
| G. versiforme, G. intraradices, G. etunicatum | Enhancement of seedling growth, mineral element, water use efficiency, and 3 times yield increase (>35 t/ha). Highest increase of leaves’ surface promoted by G. versiforme. | [32] |
| G. mosseae | Field and greenhouse results: significant yield and growth increase both in winter and autumn. | [33] |
| G. versiforme, G. intraradices, G. etunicatum | G. etonicatum gave the highest increase in bulb caliber, biomass, leaf surface, dry matter, chlorophyll content. | [34] |
| G. fasciculatum | Stimulation of nitrate reductase and glutathione synthetase activity in shoots and roots under water stress. | [35] |
| AMF + Thiobacillus sp. | High concentrations of N, P, K, and S in soil rhizosphere at 60 and 90 days from planting. | [36] |
| Rhizophagus intraradices + humic substances + CO2 | Enhancement of growth and quality of onion seedlings, chlorophyll, soluble solids, proteins, and proline content in leaves. | [37] |
| AMF mixture of Glomus etunicatum, G. microaggregatum, G. intraradices, G. claroideum, G. mosseae, and G. geosporum; single-fungus inoculum of G. intraradices BEG140; bark chips pre-inoculated with saprotrophic fungi mixture: Gymnopilus sp., Agrocybe praecox + Marasmius androsaceus | Higher beneficial effect of joint AMF application compared to a single-fungus; a synergism between AMF and saprotrophic fungi. | [38] |
| AMF | AMF reduces soil pH more than non-inoculated plants in the presence of NH4. | [39] |
| Allium cepa, Allium roylei, Allium fistulosum, A. fistulosum×A. roylei hybrid | ||
| Glomus intraradices | Interspecies differences in response to AMF inoculation. | [40] |
| Allium fistulosum, Allium roylei, Allium galanthum | ||
| G. mosseae, G. intraradices | Interspecies differences to AMF response. No differences between the effects of the two AMF species. | [41] |
| Allium cepa L. var. aggregatum | ||
| AMF and dark septate fungal endophyte (DSE) associations | Significant correlations between soil P and microsclerotia and between soil N, K and AMF spore number. | [42] |
| Rhizotech MB formulate: Glomus intraradices with low concentrations of Trichoderma harzianum and Bacillus subtilis | Increase in Se accumulation. | [11] |
| Allium porrum | ||
| Rhizophagus intraradices (RI), Claroideoglomus claroideum (CC), Funneliformis mosseae (FM) | Highest colonization with (RI + FM) and (RI + CC). | [43] |
| Funneliformis mosseae, Claroideoglomus claroideum, Rhizophagus intraradices | AMF decreases nitrate leaching. | [44] |
| AMF + biochar | Protection from heavy metals. Biochar decreases colonization degree of AMF. | [45] |
| AMF + compost + peat | Increase of leaf Zn and K, but no effect on dry matter, N, and P. | [46] |
| Allium sativum | ||
| G. mosseaeG. fasciculatum + SeO2 | The highest beneficial effect of G. fasciculatum application:decreases of AMF colonization degree due to SeO2 application | [28] |
| G. fasciculatum | Increase in alliin content and alliinase activity. | [47] |
| G. fasciculatum + P | Highest colonization and yield at 20 kg·ha−1 P. | [48] |
| Glomus intraradices | Increase of selenate accumulation. | [29] |
| Parameter | AMF | AMF + Se | Se | |||
|---|---|---|---|---|---|---|
| A. sativum | A. cepa | A. sativum | A. cepa | A. sativum | A. cepa | |
| Yield | 149 | 145 | 156 | 150 | 100 | 100 |
| Dry matter | 108 | 119 | 104 | 119 | 109 | 100 |
| monosaccharides | 100 | 168 | 175 | 200 | 129 | 141 |
| Total sugar | 119 | 100 | 121 | 100 | 119 | 100 |
| AOA | 100 | 115 | 108 | 137 | 100 | 100 |
| Flavonoids | 100 | 100 | 118 | 169 | 100 | 100 |
| Ascorbic acid | 100 | 100 | 110 | 124 | 100 | 112 |
| TA | 88 | 145 | 118 | 111 | 100 | 131 |
| Minerals | ||||||
| Se | 488 | 1000 | 28,200 | 43,800 | 21,300 | 33,200 |
| P | 110 | 130 | 122 | 130 | 110 | 100 |
| K | 100 | 123 | 123 | 140 | 112 | 100 |
| Ca | 145 | 155 | 100 | 100 | 100 | 100 |
| Mg | 191 | 100 | 186 | 123 | 191 | 100 |
| Na | 206 | 100 | 141 | 100 | 168 | 129 |
| B | 100 | 108 | 100 | 126 | 100 | 126 |
| Fe | 100 | 131 | 116 | 236 | 89 | 100 |
| Cu | 100 | 143 | 100 | 135 | 100 | 100 |
| Mo | 140 | 60 | 284 | 144 | 216 | 100 |
| Zn | 100 | 100 | 142 | 154 | 125 | 100 |
| Si | 68 | 108 | 100 | 152 | 100 | 132 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golubkina, N.; Krivenkov, L.; Sekara, A.; Vasileva, V.; Tallarita, A.; Caruso, G. Prospects of Arbuscular Mycorrhizal Fungi Utilization in Production of Allium Plants. Plants 2020, 9, 279. https://doi.org/10.3390/plants9020279
Golubkina N, Krivenkov L, Sekara A, Vasileva V, Tallarita A, Caruso G. Prospects of Arbuscular Mycorrhizal Fungi Utilization in Production of Allium Plants. Plants. 2020; 9(2):279. https://doi.org/10.3390/plants9020279
Chicago/Turabian StyleGolubkina, Nadezhda, Leonid Krivenkov, Agnieszka Sekara, Viliana Vasileva, Alessio Tallarita, and Gianluca Caruso. 2020. "Prospects of Arbuscular Mycorrhizal Fungi Utilization in Production of Allium Plants" Plants 9, no. 2: 279. https://doi.org/10.3390/plants9020279
APA StyleGolubkina, N., Krivenkov, L., Sekara, A., Vasileva, V., Tallarita, A., & Caruso, G. (2020). Prospects of Arbuscular Mycorrhizal Fungi Utilization in Production of Allium Plants. Plants, 9(2), 279. https://doi.org/10.3390/plants9020279