Plant Growth Inhibitory Activities and Volatile Active Compounds of 53 Spices and Herbs
Abstract
1. Introduction
2. Results and Discussion
2.1. Screening of Allelopathic Activity
2.2. The GC-MS Analysis of Volatiles Constituents
2.3. Evaluation of Allelopathic Activities of the Detected Volatiles
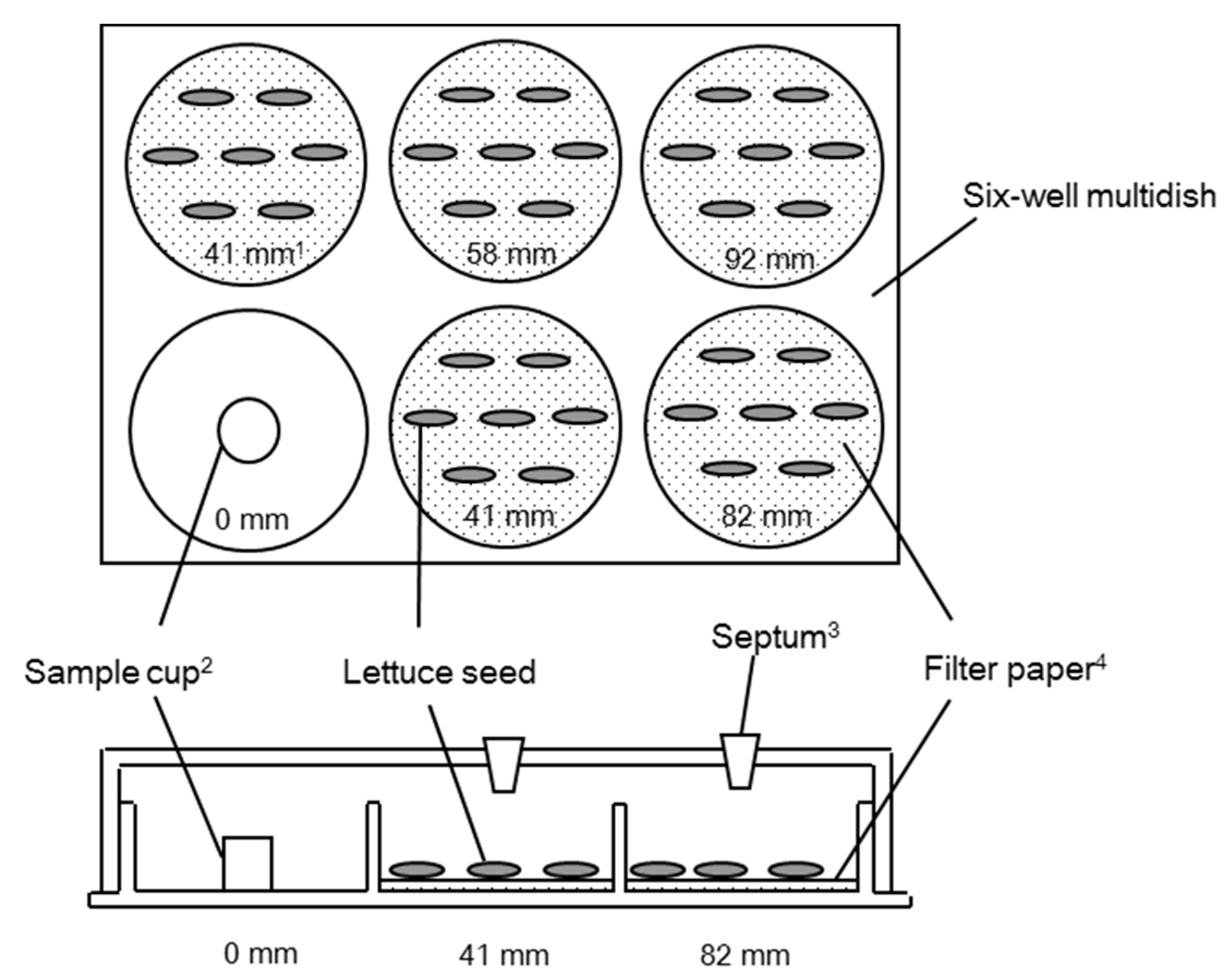
3. Materials and Methods
3.1. Screening of Spices and Herbs
3.2. Sandwich Method
3.3. Dish Pack Method
3.4. Identification and Evaluation of Plant Growth Inhibitory Volatiles
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Benner, J.P. Pesticides from nature. Part I: Crop protection agents from higher plants—An overview. In Crop Protection Agents from Nature; Copping, L.G., Ed.; The Royal Society of Chemistry: Cambridge, UK, 1996; pp. 217–229. [Google Scholar]
- Abraham, D.; Braguini, W.L.; Kelmer–Bracht, A.M.; Ishii-Iwamoto, E.L. Effects of four monoterpenes of germination, primary root growth, and mitochondrial respiration of maize. J. Chem. Ecol. 2000, 26, 611–624. [Google Scholar] [CrossRef]
- Barney, J.; Hays, A.; Weston, L.A. Isolation and characterization of allelopathic volatiles from mugwort (Artemisia vulgaris). J. Chem. Ecol. 2005, 31, 247–265. [Google Scholar] [CrossRef] [PubMed]
- Del Amo, S.; Anaya, A.L. Effect of some sesquiterpene lactones on the growth of certain secondary tropical species. J. Chem. Ecol. 1978, 4, 305–313. [Google Scholar] [CrossRef]
- Dudai, N.; Mayer, A.M.; Poljakoff-Mayber, A.; Putievsky, E.; Lerner, H.R. Essential oils as allelochemicals and their potential use as bioherbicides. J. Chem. Ecol. 1999, 25, 1079–1089. [Google Scholar] [CrossRef]
- Al-Khatib, K.; Libbey, C.; Boydston, R. Weed suppression with Brassica green manure crops in green pea. Weed Sci. 1997, 45, 439–445. [Google Scholar] [CrossRef]
- Mayton, H.S.; Olivier, C.; Vaughn, S.F.; Loria, R. Correlation of fungicidal activity of Brassica species with allyl isothiocyanate production in macerated leaf tissue. Phytopathology 1996, 86, 267–271. [Google Scholar] [CrossRef]
- Olivier, C.; Vaughn, S.F.; Mizubuti, E.S.G.; Loria, R. Variation in allyl isothiocyanate production within Brassica species and correlation with fungicidal activity. J. Chem. Ecol. 1998, 25, 2687–2701. [Google Scholar] [CrossRef]
- Vaughn, S.F.; Boydston, R.A. Volatile allelochemicals released by crucifer green manures. J. Chem. Ecol. 1997, 23, 2107–2116. [Google Scholar] [CrossRef]
- Vaughn, S.F.; Isbell, T.A.; Weisleder, D.; Berhow, M.A. Biofumigant compounds released by field pennycress (Thlaspi arvense) seed meal. J. Chem. Ecol. 2005, 31, 167–177. [Google Scholar] [CrossRef]
- Kato-Noguchi, H.; Kimura, F.; Ohno, O.; Suenaga, K. Involvement of allelopathy in inhibition of understory growth in red pine forests. J. Plant Physiol. 2017, 218, 66–73. [Google Scholar] [CrossRef]
- Kobayashi, K. Factors affecting phytotoxic activity of allelochemicals in soil. Weed Biol. Manag. 2004, 4, 1–7. [Google Scholar] [CrossRef]
- Tukey, H.B., Jr. Implications of allelopathy in agricultural plant science. Bot. Rev. 1969, 35, 1–16. [Google Scholar] [CrossRef]
- Alsaadawi, I.S.; Alrubeaa, A.J. Allelopathic effects of Citrus aurantium L. I. Vegetational patterning. J. Chem. Ecol. 1985, 11, 1527–1534. [Google Scholar] [CrossRef] [PubMed]
- Bradow, J.M.; Connick, W.J., Jr. Allelochemicals from Palmer amaranth, Amaranthus palmeri S. Wats. J. Chem. Ecol. 1987, 13, 185–202. [Google Scholar] [CrossRef]
- Connick, W.J., Jr.; Bradow, J.M.; Legendre, M.G.; Vail, S.L.; Menges, R.M. Identification of volatile allelochemicals from Amaranthus palmeri S. Wats. J. Chem. Ecol. 1987, 13, 463–472. [Google Scholar] [CrossRef]
- Fujii, Y. Screening of allelopathic candidates by new specific discrimination, and assessment methods for allelopathy, and the identification of L-DOPA as the allelopathic substance from the most promising velvet beans (Mucuna pruriens). Bull. Natl. Inst. Agro-Environ. Sci. 1994, 10, 115–218, (In Japanese with English summary). [Google Scholar]
- Haig, T. Application of hyphenated chromatography-mass spectrometry techniques to plant allelopathy research. J. Chem. Ecol. 2001, 27, 2363–2396. [Google Scholar] [CrossRef]
- Nishida, N.; Tamotsu, S.; Nagata, N.; Saito, C.; Sakai, A. Allelopathic effects of volatile monoterpenoids produced by Salvia leucophylla: Inhibition of cell proliferation and DNA synthesis in the root apical meristem of Brassica campestris seedlings. J. Chem. Ecol. 2005, 31, 1187–1203. [Google Scholar] [CrossRef]
- Fujii, Y.; Parvez, S.S.; Parves, M.M.; Ohmae, Y.; Iida, O. Screening of 239 medicinal plant species for allelopathic activity using the sandwich method. Weed Biol. Manag. 2003, 3, 233–241. [Google Scholar] [CrossRef]
- Morikawa, C.I.O.; Miyaura, R.; Tapia, Y.; Figueroa, M.D.L.; Rengifo Salgado, E.L.; Fujii, Y. Screening of 170 Peruvian plant species for allelopathic activity by using the Sandwich Method. Weed Biol. Manag. 2012, 12, 1–11. [Google Scholar] [CrossRef]
- Shiraishi, S.; Watanabe, I.; Kuno, K.; Fujii, Y. Allelopathic activity of leaching from dry leaves and exudates from roots of groundcover plants assayed on agar. Weed Biol. Manag. 2002, 2, 133–142. [Google Scholar] [CrossRef]
- Amini, S.; Azizi, M.; Joharchi, M.R.; Shafei, M.N.; Moradinezhad, F.; Fujii, Y. Determination of allelopathic potential in some medicinal and wild plant species of Iran by dish pack method. Theor. Exp. Plant Physiol. 2014, 26, 189–199. [Google Scholar] [CrossRef]
- Fujii, Y.; Matsuyama, M.; Hiradate, S.; Shimozawa, H. Dish Pack Method: A New Bioassay for volatile allelopathy. In Proceedings of the Fourth World Congress on Allelopathy, New South Wales, Australia, 21–26 August 2005; pp. 493–497. [Google Scholar]
- Sunohara, Y.; Baba, Y.; Matsuyama, S.; Fujimura, K.; Matsumoto, H. Screening and identification of phytotoxic volatile compounds in medicinal plants and characterizations of a selected compound, eucarvone. Protoplasma 2015, 252, 1047–1059. [Google Scholar] [CrossRef] [PubMed]
- Sekine, T.; Sugano, M.; Azizi, M.; Fujii, Y. Antifungal effects of volatile compounds from black zira (Bunium percisum) and other spices and herbs. J. Chem. Ecol. 2007, 33, 2123–2132. [Google Scholar] [CrossRef] [PubMed]
- Itani, T.; Hirai, K.; Fujii, Y.; Kohda, H.; Tamaki, M. Screening for allelopathic activity among weeds and medical plants using the “sandwich method”. J. Weed Sci. Tech. 1998, 43, 258–266, (In Japanese with English summary). [Google Scholar] [CrossRef]
- Hirai, N.; Sakashita, S.; Sano, T.; Inoue, T.; Ohigashi, H.; Premasthira, C.; Asakawa, Y.; Harada, J.; Fujii, Y. Allelochemicals of the tropical weed Sphenoclea zeylanica. Phytochemistry 2000, 55, 131–140. [Google Scholar] [CrossRef]
- Takemura, T.; Sakuno, E.; Kamo, T.; Hiradate, S.; Fujii, Y. Screening of the growth-inhibitory effects of 168 plant species against lettuce seedlings. Am. J. Plant. Sci. 2013, 4, 1095–1104. [Google Scholar] [CrossRef]
- Vokou, D.; Douvli, P.; Blionis, G.T.; Halley, J.M. Effects of monoterpenoids, acting alone or in pairs, on seed germination and subsequent seedling growth. J. Chem. Ecol. 2003, 29, 2281–2301. [Google Scholar] [CrossRef]
- Weidenhamer, J.D.; Macias, F.A.; Fischer, N.H.; Williamson, G.B. Just how insoluble are monoterpenes? J. Chem. Ecol. 1993, 19, 1799–1807. [Google Scholar] [CrossRef]
- Itani, T.; Fujita, T.; Tamaki, M.; Kuroyanagi, M.; Fujii, Y. Allelopathic activity and oxalate content in oxalate-rich plants. J. Weed Sci. Technol. 1999, 44, 316–323, (In Japanese with English summary). [Google Scholar] [CrossRef][Green Version]
- Harada, J. Plant growth-inhibiting substance contained in parsley plants. Jpn. J. Crop. Sci. 1984, 53, 128–129. (In Japanese) [Google Scholar]
- Harada, J. Plant growth-inhibiting substances contained in parsley seed oil. Jpn. J. Crop. Sci. 1984, 53, 130–131. (In Japanese) [Google Scholar]
- Muller, C.H.; Muller, W.H.; Haines, B.L. Volatile growth inhibitors produced by aromatic shrubs. Science 1964, 143, 471–473. [Google Scholar] [CrossRef] [PubMed]
- Yun, K.W.; Kil, B.-S. Assessment of allelopathic potential in Artemisia princeps var. Orientalis Residues. J. Chem. Ecol. 1992, 18, 1933–1940. [Google Scholar] [CrossRef]
- Morinaka, Y.; Fukuda, N.; Takayanagi, K. Evaluation of perilla (Perilla frutescens) aroma–Analysis of volatile aromatic components in fresh perilla leaves by adsorptive column method. J. Jpn. Soc. Hort. Sci. 2002, 71, 411–418, (In Japanese with English summary). [Google Scholar] [CrossRef][Green Version]
- Asplund, R.O. Monoterpenes: Relationship between structure and inhibition of germination. Phytochemistry 1968, 7, 1995–1997. [Google Scholar] [CrossRef]
- Fischer, N.H.; Williamson, G.B.; Weidenhamer, J.D.; Richardson, D.R. In search of allelopathy in the Florida scrub: The role of terpenoids. J. Chem. Ecol. 1994, 20, 1355–1380. [Google Scholar] [CrossRef]
- Halligan, J.P. Toxic terpenes from Artemisia californica. Ecology 1975, 56, 999–1003. [Google Scholar] [CrossRef]
- Romagni, J.G.; Allen, S.N.; Dayan, F.E. Allelopathic effects of volatile cineoles on two weedy plants species. J. Chem. Ecol. 2000, 26, 303–313. [Google Scholar] [CrossRef]
- Kang, G.; Mishyna, M.; Appiah, K.S.; Yamada, M.; Takano, A.; Prokhorov, V.; Fujii, Y. Screening for plant volatile emissions with allelopathic activity and the identification of L-Fenchone and 1,8-cineole from anise (Illicium verum) leaves. Plants 2019, 8, 457. [Google Scholar] [CrossRef]
- Fujii, Y.; Hiradate, S. A critical survey of allelochemicals in action—The importance of total activity and the weed suppression equation. In Proceedings of the Fourth World Congress on Allelopathy, New South Wales, Australia, 21–26 August 2005; pp. 73–76. [Google Scholar]
- Hiradate, S. Isolation strategies for finding bioactive compounds: Specific activity vs. total activity. In Natural Products for Pest Management in ACS Symposium Series 927; Rimando, A.M., Duke, S.O., Eds.; ACS Publications: Washington, DC, USA, 2006; pp. 113–126. [Google Scholar]
- Hiradate, S.; Morita, S.; Sugie, H.; Fujii, Y.; Harada, J. Phytotoxic cis-cinnamoyl glucosides from Spiraea thunbergii. Phytochemistry 2004, 65, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Finney, D.J. Probit Analysis; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
| Donor Spice and Herb | Inhibition (%) 3 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sandwich Method 4 | Dish Pack Method 5 | |||||||||||
| Common Names (Part Used) 1 | Scientific Name | Family | R | H | Radicle | Hypocotyl | ||||||
| 41 | 58 | 82 | 92 | 41 | 58 | 82 | 92 | |||||
| Onion 2,* | Allium cepa | Liliaceae | 38 | 9 | 17 | 5 | 12 | −1 | 14 | 21 | 16 | 13 |
| Scallion (root) | Allium fistuiosum | Liliaceae | 22 | 0 | 45 | 12 | −10 | −5 | 11 | −3 | −7 | 7 |
| Scallion (leaf & stem) | Allium fistuiosum | Liliaceae | 58 | 24 | −2 | 9 | −10 | 16 | 11 | −3 | −7 | −5 |
| Chinese pepper | Zanthoxylum bungeanum | Rutaceae | 68 | 62 | 89 | 100 | 44 | 69 | 94 | 100 | 72 | 81 |
| Chinpi * | Citrus unshiu | Rutaceae | 38 | 13 | 8 | 4 | −24 | −3 | 3 | 6 | −11 | −23 |
| Clove * | Eugenia aromatica | Myrtaceae | 49 | 72 | 69 | 8 | 31 | 39 | 47 | 6 | 19 | 16 |
| Allspice * | Pimenta officinalis | Myrtaceae | 35 | 21 | 38 | −27 | 38 | 12 | 49 | 36 | 46 | 18 |
| Juniper berry * | Juniperus communis | Cupressaceae | 11 | −9 | 75 | 37 | 20 | 33 | 71 | 26 | 18 | 29 |
| Mace * | Myristica fragrans | Myristicaceae | 44 | 46 | 95 | 90 | 54 | −4 | 97 | 92 | 71 | 51 |
| Nutmeg * | Myristica fragrans | Myristicaceae | 21 | 31 | 24 | −2 | −11 | −9 | 58 | 39 | 18 | 26 |
| Red pepper * | Capsicum annuum | Solanaceae | 49 | 19 | 48 | 53 | 43 | 25 | 15 | 30 | 25 | 10 |
| Houttuynia (root) | Houttuynia cordata | Saururaceae | 18 | −6 | 47 | 44 | −1 | 23 | 44 | 34 | −1 | 7 |
| Houttuynia (leaf & stem | Houttuynia cordata | Saururaceae | 38 | −8 | 13 | 3 | 26 | 22 | 17 | −5 | 40 | 29 |
| Caraway * | Carum carvi | Umbelliferae | 60 | 44 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Dill (seed) * | Anethum graveolens | Umbelliferae | 58 | 54 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Dill (leaf & stem) | Anethum graveolens | Umbelliferae | 62 | 37 | 8 | 22 | 34 | −2 | 3 | 5 | 36 | −9 |
| Black zira | Bunium persicum | Umbelliferae | 68 | 52 | 93 | 85 | 47 | 21 | 94 | 90 | 71 | 51 |
| Coriander * | Coriandrum sativum | Umbelliferae | 68 | 31 | 20 | 52 | 30 | 26 | 11 | 23 | 21 | 21 |
| Funnel (seed) * | Foeniculum vulgare | Umbelliferae | 41 | 18 | 48 | 10 | 7 | 3 | 72 | 53 | 34 | 36 |
| Parsley * | Petroselium sativum | Umbelliferae | 77 | 62 | 20 | 32 | 24 | 11 | 19 | 25 | 25 | 7 |
| Celery (seed) * | Apium graveolens | Umbelliferae | 46 | 52 | 21 | 22 | 22 | 12 | 18 | 16 | 13 | 14 |
| Seri (root) | Oenanthe javanica | Umbelliferae | 33 | −23 | 20 | 26 | 9 | 19 | 10 | 11 | 1 | −3 |
| Seri (leaf and stem) | Oenanthe javanica | Umbelliferae | 42 | 5 | 9 | 6 | 17 | 20 | 0 | 38 | 13 | 11 |
| Cumin * | Cuminum cyminum | Umbelliferae | 60 | 31 | 50 | 20 | −37 | 1 | 79 | 46 | 8 | 32 |
| Anise * | Pinpinella anisum | Umbelliferae | 48 | 7 | 17 | 35 | −1 | −6 | 48 | 50 | 36 | 28 |
| Ajowan * | Trachyspermum ammi | Umbelliferae | 56 | 52 | −60 | 1 | −53 | −12 | 16 | 29 | −4 | 20 |
| Cardamom * | Elettaria cardamomum | Zingiberaceae | 29 | −3 | 100 | 100 | 85 | 75 | 100 | 100 | 89 | 78 |
| Brown cardamom * | Elettaria cardamomum | Zingiberaceae | 14 | 4 | 58 | 17 | 17 | 10 | 63 | 38 | 3 | 14 |
| Ginger * | Zingiber officinale | Zingiberaceae | 36 | 26 | 85 | 52 | 25 | 46 | 84 | 53 | 16 | 48 |
| Turmeric * | Curcuma domestica | Zingiberaceae | 19 | 39 | −22 | −23 | −2 | −29 | 16 | 22 | 10 | −11 |
| Rosemary * | Rosemarinus officinalis | Labiatae | 59 | 61 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Sage * | Salvia officinalis | Labiatae | 14 | 20 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Thyme * | Thymus vulgaris | Labiatae | 20 | 33 | 100 | 100 | 85 | 64 | 100 | 100 | 85 | 63 |
| Lavender (leaf&stem) | Lavandula augustifolia | Labiatae | 43 | 62 | 100 | 100 | 67 | 58 | 100 | 100 | 56 | 58 |
| Lavender (root) | Lavandula augustifolia | Labiatae | 15 | 3 | 77 | 52 | 27 | 39 | 61 | 13 | −3 | 7 |
| Savory * | Satureja hortensis | Labiatae | 21 | 21 | 29 | 13 | 24 | 11 | 27 | 18 | 25 | 7 |
| Basil * | Ocimum basilicum | Labiatae | 68 | 21 | 51 | −12 | −60 | −10 | 65 | 27 | 3 | 27 |
| Origan * | Origanum vulgare | Labiatae | 11 | 22 | −7 | −37 | 30 | 14 | 36 | 19 | 31 | 4 |
| Shiso (green) | Perilla frutescens | Labiatae | 37 | 29 | −64 | −96 | −103 | −114 | 30 | 20 | 11 | 13 |
| Shiso (red) | Perilla frutescens | Labiatae | 49 | −24 | −122 | −107 | −104 | −116 | −2 | −14 | −20 | −12 |
| White pepper * | Piper nigricum | Piperaceae | 67 | 66 | 85 | 16 | 56 | 40 | 82 | 16 | 63 | 29 |
| Black pepper * | Piper nigricum | Piperaceae | 60 | 61 | 15 | −13 | −10 | −3 | 42 | 53 | 34 | 36 |
| Laurel * | Laurus nobilis | Lauraceae | 24 | 26 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Cassis * | Cinnmomum cassia | Lauraceae | 42 | 29 | 100 | 48 | 20 | 32 | 100 | 65 | 38 | 21 |
| Cinnamon * | Cinnamomum verum | Lauraceae | 42 | 21 | 65 | 20 | 10 | −7 | 68 | 48 | 20 | 10 |
| Tarragon * | Artemisia dracunculus | Compositae | 72 | 40 | 84 | 33 | 15 | 0 | 71 | 65 | 28 | 16 |
| Camomile (leaf & stem) | Anthemis nobilis | Compositae | 59 | 16 | 40 | 20 | 24 | 12 | 17 | 3 | 11 | −1 |
| Chamomile (root) | Anthemis nobilis | Compositae | 17 | −8 | 50 | 19 | −1 | −9 | 13 | 11 | 17 | −3 |
| Chamomile (flower) | Anthemis nobilis | Compositae | 61 | 39 | 8 | 25 | −4 | −1 | 5 | 40 | −5 | 5 |
| Pepper tree * | Schinus molle | Anacardiaceae | 13 | 0 | 100 | 77 | 21 | −2 | 100 | 91 | 32 | 20 |
| Oriental mustard * | Brassica juncea | Brassicaceae | 64 | 69 | 99 | 97 | 54 | 43 | 97 | 96 | 45 | 32 |
| Yellow mustard * | Brassica alba | Brassicaceae | 46 | 42 | 14 | 32 | 17 | 19 | 24 | 44 | 33 | 34 |
| Brown mustard * | Brassica nigra | Brassicaceae | 32 | 32 | 44 | 45 | 10 | 10 | 33 | 43 | 32 | 18 |
| Plant Species | Concentration (μg/g Plant) 1 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| α-Pinene | Camphene | β-Pinene | β-Myrcene | 3-Carene | p-Cymene | Limonene | 1,8-Cineole | γ-Terpinene | Camphor | Borneol | Carvone | |
| Caraway | ND | ND | ND | ND | ND | ND | 4.85 | ND | ND | ND | ND | 0.289 |
| Dill (seed) | ND | ND | ND | ND | ND | ND | 57.2 | ND | 0.0947 | ND | ND | 6.47 |
| Cardamom | ND | ND | ND | 0.0417 | 0.424 | ND | 0.288 | 8.21 | ND | ND | ND | ND |
| Rosemary | 9.59 | 2.06 | ND | 0.437 | ND | 2.66 | 0.543 | 18.6 | ND | 1.51 | ND | ND |
| Sage | 7.64 | 1.08 | 1.28 | 1.26 | ND | 2.39 | 0.419 | 57.2 | 0.136 | 0.474 | ND | ND |
| Thyme | 2.83 | 2.80 | 0.308 | 0.110 | 0.853 | 4.57 | 0.149 | ND | ND | ND | 0.968 | ND |
| Laurel | 8.76 | 0.724 | 5.78 | ND | ND | 8.56 | ND | 155 | ND | ND | ND | ND |
| LOD | 0.008 | 0.005 | 0.007 | 0.006 | 0.012 | 0.056 | 0.004 | 0.010 | 0.003 | 0.007 | 0.007 | 0.006 |
| Compound 1 | Amount Added | Distance (mm) 2 | Inhibition (%) 3 | Vapour (ppm v/v) 4 | Compound 1 | Amount Added | Distance (mm) 2 | Inhibition (%) 3 | Vapour (ppm v/v) 4 | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| R | H | R | H | ||||||||
| α-pinene | γ-terpinene | ||||||||||
| 50 μL | 41 | 65.2 | 73.9 | 861 | 50 μL | 41 | 80.0 | 90.1 | 101 | ||
| 58 | 56.8 | 72.6 | 58 | 35.6 | 65.5 | ||||||
| 82 | 84.7 | 82.6 | 82 | 38.1 | 58.9 | ||||||
| 92 | 61.0 | 62.7 | 776 | 92 | 48.5 | 67.1 | 78.9 | ||||
| camphene | 5 μL | 41 | −14.9 | 32.2 | |||||||
| 50 mg | 41 | 34.9 | 40.0 | 950 | 58 | −17.3 | 32.2 | ||||
| 58 | 44.6 | 47.4 | 82 | −19.9 | 20.3 | ||||||
| 82 | −4.4 | −25.0 | 92 | −10.5 | 27.0 | ||||||
| 92 | −54.7 | −15.1 | 744 | camphor | |||||||
| β-pinene | 50 mg | 41 | 100 | 100 | |||||||
| 50 μL | 41 | 91.6 | 93.3 | 896 | 58 | 100 | 100 | ||||
| 58 | 86.5 | 90.5 | 82 | 100 | 100 | ||||||
| 82 | 77.5 | 81.0 | 92 | 100 | 100 | ||||||
| 92 | 77.5 | 88.6 | 875 | 5 mg | 41 | 100 | 100 | ||||
| 5 μL | 41 | 39.1 | 16.7 | 58 | 100 | 100 | |||||
| 58 | 32.7 | 8.8 | 82 | 100 | 100 | ||||||
| 82 | 36.1 | 14.0 | 92 | 100 | 100 | ||||||
| 92 | 33.6 | 7.0 | 0.5 mg | 41 | 100 | 100 | 1.76 | ||||
| β-myrcene | 58 | 100 | 100 | ||||||||
| 50 μL | 41 | 21.7 | 64.9 | 209 | 82 | 100 | 100 | ||||
| 58 | 47.8 | 67.6 | 92 | 90.6 | 94.6 | 0.482 | |||||
| 82 | 5.7 | 35.1 | 0.1 mg | 41 | 83.2 | 94.8 | 0.240 | ||||
| 92 | −1.0 | 43.2 | 154 | 58 | 67.5 | 86.6 | |||||
| 3-carene | 82 | 64.4 | 83.6 | ||||||||
| 50 μL | 41 | 91.0 | 89.3 | 580 | 92 | 49.7 | 70.2 | ||||
| 58 | 88.0 | 91.5 | 0.05 mg | 41 | 41.4 | 59.0 | 0.0472 | ||||
| 82 | 72.9 | 80.8 | 58 | 19.4 | 38.9 | ||||||
| 92 | 89.0 | 93.6 | 625 | 82 | 16.2 | 34.4 | |||||
| 5 μL | 41 | 38.2 | 17.5 | 92 | 33.0 | 55.3 | |||||
| 58 | 49.7 | 14.0 | borneol | ||||||||
| 82 | 40.4 | 8.8 | 50 mg | 41 | 100 | 100 | |||||
| 92 | 28.4 | −8.8 | 58 | 100 | 100 | ||||||
| p-cymene | 82 | 100 | 100 | ||||||||
| 50 μL | 41 | 52.9 | 70.3 | 103 | 92 | 100 | 100 | ||||
| 58 | 19.2 | 51.4 | 5 mg | 41 | 100 | 100 | |||||
| 82 | 14.1 | 40.5 | 58 | 100 | 100 | ||||||
| 92 | 7.4 | 51.4 | 54.8 | 82 | 100 | 100 | |||||
| 92 | 100 | 100 | |||||||||
| limonene | (Borneol) | ||||||||||
| 50 μL | 41 | 3.2 | 32.8 | 183 | 0.5 mg | 41 | 100 | 100 | 5.53 | ||
| 58 | 2.5 | 32.8 | 58 | 100 | 100 | ||||||
| 82 | −0.3 | 40.3 | 82 | 93.4 | 94.6 | ||||||
| 92 | 29.0 | 47.8 | 167 | 92 | 96.2 | 94.6 | 0.435 | ||||
| 1,8-cineole | 0.1 mg | 41 | 81.7 | 78.9 | 0.0923 | ||||||
| 50 μL | 41 | 100 | 100 | 58 | 83.8 | 75.4 | |||||
| 58 | 100 | 100 | 82 | 56.6 | 31.6 | ||||||
| 82 | 100 | 100 | 92 | 52.3 | 21.1 | ||||||
| 92 | 100 | 100 | carvone | ||||||||
| 5 μL | 41 | 100 | 100 | 50 μL | 41 | 100 | 100 | 0.184 | |||
| 58 | 100 | 100 | 58 | 100 | 100 | ||||||
| 82 | 100 | 100 | 82 | 100 | 100 | ||||||
| 92 | 100 | 100 | 92 | 100 | 100 | ||||||
| 2 μL | 41 | 100 | 100 | 5 μL | 41 | 64.8 | 84.0 | 0.0529 | |||
| 58 | 100 | 100 | 58 | 95.9 | 97.3 | ||||||
| 82 | 100 | 100 | 82 | 82.0 | 89.4 | ||||||
| 92 | 100 | 100 | 34.6 | 92 | 97.2 | 100 | |||||
| 1 μL | 41 | 85.5 | 91.2 | 10.3 | 2 μL | 41 | 28.5 | 70.5 | |||
| 58 | 90.6 | 91.2 | 58 | 27.9 | 62.0 | ||||||
| 82 | 76.1 | 82.5 | 82 | 33.6 | 71.5 | ||||||
| 92 | 82.1 | 87.7 | 9.43 | 92 | 41.4 | 77.2 | |||||
| 0.5 μL | 41 | 9.0 | 42.9 | 4.72 | |||||||
| 58 | 15.1 | 35.7 | |||||||||
| 82 | 16.0 | 37.5 | |||||||||
| 92 | 15.1 | 46.4 | |||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sekine, T.; Appiah, K.S.; Azizi, M.; Fujii, Y. Plant Growth Inhibitory Activities and Volatile Active Compounds of 53 Spices and Herbs. Plants 2020, 9, 264. https://doi.org/10.3390/plants9020264
Sekine T, Appiah KS, Azizi M, Fujii Y. Plant Growth Inhibitory Activities and Volatile Active Compounds of 53 Spices and Herbs. Plants. 2020; 9(2):264. https://doi.org/10.3390/plants9020264
Chicago/Turabian StyleSekine, Takayuki, Kwame Sarpong Appiah, Majid Azizi, and Yoshiharu Fujii. 2020. "Plant Growth Inhibitory Activities and Volatile Active Compounds of 53 Spices and Herbs" Plants 9, no. 2: 264. https://doi.org/10.3390/plants9020264
APA StyleSekine, T., Appiah, K. S., Azizi, M., & Fujii, Y. (2020). Plant Growth Inhibitory Activities and Volatile Active Compounds of 53 Spices and Herbs. Plants, 9(2), 264. https://doi.org/10.3390/plants9020264







