Breeding Buckwheat for Increased Levels of Rutin, Quercetin and Other Bioactive Compounds with Potential Antiviral Effects
Abstract
1. Introduction
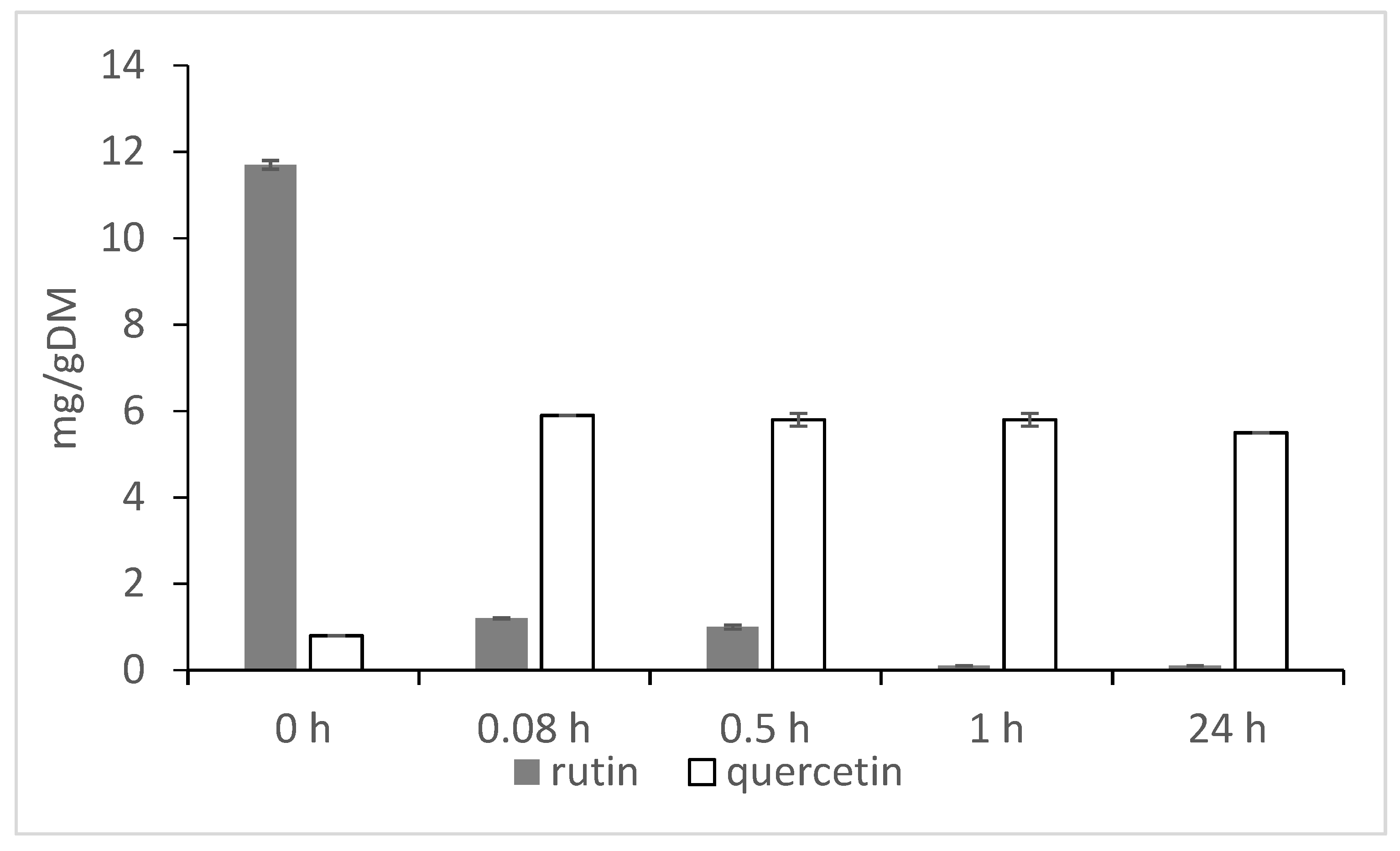
2. Rutin, Rutinosidase and Quercetin
| Species | Sample | Rutin | Quercetin | Literature Source |
|---|---|---|---|---|
| Tartary buckwheat | Flour | 1.46 | 0.19 | [52] |
| Tartary buckwheat | Bread | 0.51 | [52] | |
| Tartary buckwheat | Herb | 1.2–3.1 | [56] | |
| Tartary buckwheat | Grain | 1.3–1.6 | [56] | |
| Common buckwheat | Grain | 0.01 | [56] | |
| Common buckwheat | Flour | 0.0003 | [23] | |
| Tartary buckwheat | Wholemeal flour | 0.22 | 0.19 | [57] |
| Tartary buckwheat | Malt from wholemeal flour | 0.37 | 0.41 | [57] |
| Tartary buckwheat | Flour | 1.17 | 0.06 | [25] |
| Tartary buckwheat | Flour | 1.584–1.637 | [42] | |
| Common buckwheat | Grain | 0.017-0.070 | [58,59] |
3. Fagopyrin and Emodin
4. Buckwheat Breeding: Challenges and Prospects for the Future
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wei, Y.M. Buckwheat remains from the late Neolithic site of Donghuishan, Gansu Province, China. Cereal Chem. 2019, 96, 332–337. [Google Scholar] [CrossRef]
- Ohsawa, R. Current status and prospects of common buckwheat breeding in Japan. Breed. Sci. 2020, 70, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, Y.L.B.; Scott, I.M.; McNeil, J.N. The buckwheat effect: A biopesticide for Wireworm? J. Econ. Entomol. 2019, 112, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Morishita, T.; Hara, T.; Hara, T. Important agronomic characteristics of yielding ability in common buckwheat; ecotype and ecological differentiation, preharvest sprouting resistance, shattering resistance, and lodging resistance. Breed. Sci. 2020, 70, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Malhotra, N.; Sharma, K. Buckwheat (Fagopyrum sp.) genetic resources: What can they contribute towards nutritional security of changing world? Genet. Resour. Crop Evol. 2020, 67, 1639–1658. [Google Scholar] [CrossRef]
- Kreft, M. Buckwheat phenolic metabolites in health and disease. Nutr. Res. Rev. 2016, 29, 30–39. [Google Scholar] [CrossRef]
- Pongrac, P.; Kelemen, M.; Vavpetič, P.; Vogel-Mikuš, K.; Regvar, M.; Pelicon, P. Application of micro-PIXE (particle induced X-ray emission) to study buckwheat grain Structure and Composition. Fagopyrum 2020, 37, 5–10. [Google Scholar] [CrossRef]
- Sytar, O.; Svediene, J.; Loziene, K.; Paskevicius, A.; Kosyan, A.; Taran, N. Antifungal properties of hypericin, hypericin tetrasulphonic acid and fagopyrin on pathogenic fungi and Spoilage yeasts. Pharm. Biol. 2016, 54, 3121–3125. [Google Scholar] [CrossRef]
- Skrabanja, V.; Kreft, I. Resistant starch formation following autoclaving of buckwheat (Fagopyrum esculentum Moench) groats. An in vitro study. J. Agric. Food Chem. 1998, 46, 2020–2023. [Google Scholar] [CrossRef]
- Huda, M.N.; Lu, S.; Jahan, T.; Ding, M.; Jha, R.; Zhang, K.; Zhang, W.; Georgiev, M.I.; Park, S.U.; Zhou, M. Treasure from garden: Bioactive compounds of buckwheat. Food Chem. 2020, 335, 127653. [Google Scholar] [CrossRef]
- Kreft, I.; Zhou, M.L.; Golob, A.; Germ, M.; Likar, M.; Dziedzic, K.; Luthar, Z. Breeding Buckwheat for Nutritional Quality. Breed. Sci. 2020, 70, 67–73. [Google Scholar] [CrossRef]
- Wieslander, G.; Fabjan, N.; Vogrinčič, M.; Kreft, I.; Janson, C.; Spetz-Nystrom, U.; Vombergar, B.; Tagesson, C.; Leanderson, P.; Norbäck, D. Eating buckwheat cookies is associated with the reduction in serum levels of myeloperoxidase and cholesterol: A double blind crossover study in day-care centre staffs. Tohoku J. Exp. Med. 2011, 225, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Wieslander, G.; Fabjan, N.; Vogrinčič, M.; Kreft, I.; Vombergar, B.; Norbäck, D. Effects of common and Tartary buckwheat consumption on mucosal symptoms, headache and tiredness: A double-blind crossover intervention study. J. Food Agric. Environ. 2012, 10, 107–110. [Google Scholar]
- Ikeda, K.; Ishida, Y.; Ikeda, S.; Asami, Y.; Lin, R. Tartary, but not common, buckwheat inhibits α-glucosidase activity: Its nutritional implications. Fagopyrum 2017, 34, 13–18. [Google Scholar] [CrossRef]
- Zhang, C.N.; Zhang, R.; Li, Y.M.; Liang, N.; Zhao, Y.M.; Zhu, H.Y.; He, Z.Y.; Liu, J.H.; Hao, W.J.; Jiao, R.; et al. Cholesterol-lowering activity of Tartary buckwheat protein. J. Agric. Food Chem. 2017, 65, 1900–1906. [Google Scholar] [CrossRef] [PubMed]
- Gullon, B.; Lu-Chau, T.A.; Moreira, M.T.; Lema, J.M.; Eibes, G. Rutin: A review on extraction, Identification and Purification Methods, Biological Activities and Approaches to enhance its bioavailability. Trends Food Sci. Technol. 2017, 67, 220–235. [Google Scholar] [CrossRef]
- Kawabata, K.; Mukai, R.; Ishisaka, A. Quercetin and related polyphenols: New insights and implications for their bioactivity and bioavailability. Food Funct. 2015, 6, 1399–1417. [Google Scholar] [CrossRef]
- Kreft, S.; Štrukelj, B.; Gaberščik, A.; Kreft, I. Rutin in buckwheat herbs grown at different UV-B radiation levels: Comparison of two UV spectrophotometric and an HPLC method. J. Exp. Bot. 2002, 53, 1801–1804. [Google Scholar] [CrossRef]
- Suzuki, T.; Honda, Y.; Mukasa, Y. Effects of UV-B radiation, cold and desiccation stress on rutin concentration and rutin glucosidase activity in Tartary buckwheat (Fagopyrum tataricum) leaves. Plant Sci. 2005, 168, 1303–1307. [Google Scholar] [CrossRef]
- Kalinova, J.; Vrchotova, N.; Triska, J. Exudation of allelopathic substances in buckwheat (Fagopyrum esculentum Moench). J. Agric. Food Chem. 2007, 55, 6453–6459. [Google Scholar] [CrossRef]
- Gfeller, A.; Glauser, G.; Etter, C.; Signarbieux, C.; Wirth, J. Fagopyrum esculentum alters its root exudation after Amaranthus retroflexus recognition and suppresses weed growth. Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, T.; Nakagawa, H. Purification and characterization of the rutin-degrading enzymes in Tartary buckwheat seeds. Phytochemistry 1994, 37, 133–136. [Google Scholar] [CrossRef]
- Suzuki, T.; Honda, Y.; Mukasa, Y.; Kim, S. Effects of lipase, lipoxygenase, peroxidase, and rutin on quality deteriorations in buckwheat flour. J. Agric. Food Chem. 2005, 53, 8400–8405. [Google Scholar] [CrossRef]
- Fujita, K.; Yoshihashi, T. Heat-treatment of Tartary buckwheat (Fagopyrum tataricum Gaertn.) provides dehulled and gelatinized product with denatured rutinosidase. Food Sci. Technol. Res. 2019, 25, 613–618. [Google Scholar] [CrossRef]
- Vombergar, B. Rutin and quercetin in common buckwheat and Tartary buckwheat flour. Folia Biol. Geol. 2020, 61, 257–280. [Google Scholar] [CrossRef]
- Gaberščik, A.; Vončina, M.; Trošt, T.; Germ, M.; Björn, L.O. Growth and production of buckwheat (Fagopyrum esculentum) treated with reduced, ambient, and enhanced UV-B radiation. J. Photochem. Photobiol. B 2002, 66, 30–36. [Google Scholar] [CrossRef]
- Holasova, M.; Fiedlerova, V.; Smrcinova, H.; Orsak, M.; Lachman, J.; Vavreinova, S. Buckwheat—The source of antioxidant activity in functional foods. Food Res. Int. 2002, 35, 207–211. [Google Scholar] [CrossRef]
- Matsui, K.; Oshima, Y.; Mitsuda, N.; Sakamoto, S.; Nishiba, Y.; Walker, A.R.; Ohme-Takagi, M.; Robinson, S.P.; Yasui, Y.; Mori, M.; et al. Buckwheat R2R3 MYB transcription factor FeMYBF1 regulates flavonol biosynthesis. Plant Sci. 2018, 274, 466–475. [Google Scholar] [CrossRef]
- Kalinova, J.P.; Vrchotova, N.; Triska, J. Phenolics levels in different parts of common buckwheat (Fagopyrwn esculentum) achenes. J. Cereal Sci. 2019, 85, 243–248. [Google Scholar] [CrossRef]
- Zhang, K.X.; Logacheva, M.D.; Meng, Y.; Hu, J.P.; Wan, D.P.; Li, L.; Janovska, D.; Wang, Z.Y.; Georgiev, M.I.; Yu, Z.; et al. Jasmonate-responsive MYB factors spatially repress rutin biosynthesis in Fagopyrum tataricum. J. Exp. Bot. 2018, 69, 1955–1966. [Google Scholar] [CrossRef]
- Sikder, K.; Kesh, S.B.; Das, N.; Manna, K.; Dey, S. The high antioxidative power of quercetin (aglycone flavonoid) and its glycone (rutin) avert high cholesterol diet induced hepatotoxicity and inflammation in swiss albino mice. Food Funct. 2014, 5, 1294–1303. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Li, Y.M.; Zhang, K.S.; Jiao, R.; Ma, K.Y.; Zhang, R.; Ren, G.X.; Chen, Z.Y. Hypocholesterolemic activity of buckwheat flour is mediated by increasing sterol excretion and down-regulation of intestinal NPC1L1 and ACAT2. J. Funct. Foods 2014, 6, 311–318. [Google Scholar] [CrossRef]
- Vogrinčič, M.; Timoracka, M.; Melichacova, S.; Vollmannova, A.; Kreft, I. Degradation of rutin and polyphenols during the preparation of Tartary buckwheat bread. J. Agric. Food Chem. 2010, 58, 4883–4887. [Google Scholar] [CrossRef] [PubMed]
- Vogrinčič, M.; Kreft, I.; Filipič, M.; Žegura, B. Antigenotoxic Effect of Tartary (Fagopyrum tataricum) and common (Fagopyrum esculentum) buckwheat flour. J. Med. Food 2013, 16, 944–952. [Google Scholar] [CrossRef] [PubMed]
- Chitarrini, G.; Nobili, C.; Pinzari, F.; Antonini, A.; De Rossi, P.; Del Fiore, A.; Procacci, S.; Tolaini, V.; Scala, V.; Scarpari, M.; et al. Buckwheat achenes antioxidant profile modulates Aspergillus flavus growth and aflatoxin production. Int. J. Food Microbiol. 2014, 189, 1–10. [Google Scholar] [CrossRef]
- Suzuki, T.; Honda, Y.; Funatsuki, W.; Nakatsuka, K. Purification and characterization of flavonol 3-glucosidase, and its activity during ripening in Tartary buckwheat seeds. Plant Sci. 2002, 163, 417–423. [Google Scholar] [CrossRef]
- Suzuki, T.; Morishita, T.; Mukasa, Y.; Takigawa, S.; Yokota, S.; Ishiguro, K.; Noda, T. Breeding of ‘Manten-Kirari’, a non-bitter and trace-rutinosidase variety of Tartary Buckwheat (Fagopyrum tataricum Gaertn.). Breeding Sci. 2014, 64, 344–350. [Google Scholar] [CrossRef]
- Suzuki, T.; Morishita, T.; Kim, S.J.; Park, S.U.; Woo, S.H.; Noda, T.; Takigawa, S. Physiological roles of rutin in the buckwheat plant. Jarq-Jpn. Agric. Res. Q. 2015, 49, 37–43. [Google Scholar] [CrossRef]
- Suzuki, T.; Morishita, T.; Noda, T.; Ishiguro, K. Acute and subacute toxicity studies on rutin-rich Tartary buckwheat dough in experimental animals. J. Nutr. Sci. Vitaminol. 2015, 61, 175–181. [Google Scholar] [CrossRef][Green Version]
- Suzuki, T.; Morishita, T.; Takigawa, S.; Noda, T.; Ishiguro, K. Characterization of rutin-rich bread made with ‘Manten-Kirari’, a trace-rutinosidase variety of Tartary buckwheat (Fagopyrum tataricum Gaertn.). Food Sci. Technol. Res. 2015, 21, 733–738. [Google Scholar] [CrossRef][Green Version]
- Jin, H.R.; Yu, J.; Choi, S.J. Hydrothermal treatment enhances antioxidant activity and intestinal absorption of rutin in Tartary buckwheat flour extracts. Foods 2020, 9, 8. [Google Scholar] [CrossRef] [PubMed]
- Dziedzic, K.; Górecka, D.; Szwengiel, A.; Olejnik, A.; Rychlik, J.; Kreft, I.; Drożdżyńska, A.; Walkowiak, J. The cytotoxic effect of artificially digested buckwheat products on HT-29 colon cancer cells. J. Cereal Sci. 2018, 83, 68–73. [Google Scholar] [CrossRef]
- Sun, Y.L.; Zhou, W.M.; Huang, Y.G. Encapsulation of Tartary buckwheat flavonoids and application to yoghurt. J. Microencapsul. 2020, 37, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Levit, A.; Nowak, S.; Peters, M.; Wiener, A.; Meyerhof, W.; Behrens, M.; Niv, M.Y. The bitter pill: Clinical drugs that activate the human bitter taste receptor TAS2R14. FASEB J. 2013, 28, 1181–1197. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.C.; Santos, J.; Costa, A.R.; Ferreira, C.L.; Tomas, J.; Quintela, T.; Ishikawa, H.; Schwerk, C.; Schroten, H.; Ferrer, I.; et al. Bitter taste receptors profiling in the human blood-cerebrospinal fluid-barrier. Biochem. Pharmacol. 2020, 177, 113954. [Google Scholar] [CrossRef]
- Shawky, E.; Nada, A.A.; Ibrahim, R.S. Potential role of medicinal plants and their constituents in the mitigation of Sars-Cov-2: Identifying related therapeutic targets using network pharmacology and molecular docking analyses. RSC Adv. 2020, 10, 27961–27983. [Google Scholar] [CrossRef]
- Das, P.; Majumder, R.; Mandal, M.; Basak, P. In-silico approach for identification of effective and stable inhibitors for Covid-19 main protease (M-pro) from flavonoid based phytochemical constituents of Calendula officinalis. J. Biomol. Struct. Dyn. 2020. [Google Scholar] [CrossRef]
- Al-Zahrani, A.A. Rutin as a promising inhibitor of main protease and other protein targets of Covid-19: In silico study. Nat. Prod. Commun. 2020, 15. [Google Scholar] [CrossRef]
- Bhowmik, D.; Nandi, R.; Jagadeesan, R.; Kumar, N.; Prakash, A.; Kumar, D. Identification of potential inhibitors against Sars-Cov-2 by targeting proteins responsible for envelope formation and virion assembly using docking based virtual screening, and pharmacokinetics approaches. Infect. Genet. Evol. 2020, 84, 1–14. [Google Scholar] [CrossRef]
- Shivanika, C.; Kumar, S.D.; Ragunathan, V.; Tiwari, P.; Sumitha, A.; Devi, P.B. Molecular docking, validation, dynamics simulations, and pharmacokinetic prediction of natural compounds against the Sars-Cov-2 main-protease. J. Biomol. Struct. Dyn. 2020, 83, 1–28. [Google Scholar] [CrossRef]
- Germ, M.; Arvay, J.; Vollmannova, A.; Toth, T.; Golob, A.; Luthar, Z.; Kreft, I. The temperature threshold for the transformation of rutin to quercetin in Tartary buckwheat dough. Food Chem. 2019, 283, 28–31. [Google Scholar] [CrossRef]
- Lukšič, L.; Bonafaccia, G.; Timoracka, M.; Vollmannova, A.; Trček, J.; Nyambe, T.K.; Melini, V.; Acquistucci, R.; Germ, M.; Kreft, I. Rutin and quercetin transformation during preparation of buckwheat sourdough bread. J. Cereal Sci. 2016, 69, 71–76. [Google Scholar] [CrossRef]
- Brajčič, F.; Luštek, P.; Šuštarič, T.; Pust, J. Extraction of rutin and quercetin from Tartary buckwheat grains, hydrothermally treated at different temperatures. Fagopyrum 2020, 37, 37–40. [Google Scholar] [CrossRef]
- Braune, A.; Blaut, M. Bacterial Species Involved in the conversion of dietary flavonoids in the human gut. Gut Microbes 2016, 7, 216–234. [Google Scholar] [CrossRef] [PubMed]
- Shin, N.R.; Moon, J.S.; Shin, S.Y.; Li, L.; Lee, Y.B.; Kim, T.J.; Han, N.S. Isolation and characterization of human intestinal enterococcus avium EFEL009 converting rutin to quercetin. Lett. Appl. Microbiol. 2016, 62, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Fabjan, N.; Rode, J.; Košir, I.J.; Wang, Z.H.; Zhang, Z.; Kreft, I. Tartary buckwheat (Fagopyrum tataricum Gaertn.) as a source of dietary rutin and quercitrin. J. Agric. Food Chem. 2003, 51, 6452–6455. [Google Scholar] [CrossRef]
- Molinari, R.; Costantini, L.; Timperio, A.M.; Lelli, V.; Bonafaccia, F.; Bonafaccia, G.; Merendino, N. Tartary buckwheat malt as igredient of gluten-free cookies. J. Cereal Sci. 2018, 80, 37–43. [Google Scholar] [CrossRef]
- Suzuki, T.; Hara, T.; Hara, T.; Katsu, K. Effect of storage temperature on occurrence of secondary dormancy in buckwheat Seeds. Seed Sci. Technol. 2020, 48, 257–267. [Google Scholar] [CrossRef]
- Suzuki, T.; Noda, T.; Morishita, T.; Ishiguro, K.; Otsuka, S.; Brunori, A. Present status and future perspectives of breeding for buckwheat quality. Breed. Sci. 2020, 70, 48–66. [Google Scholar] [CrossRef]
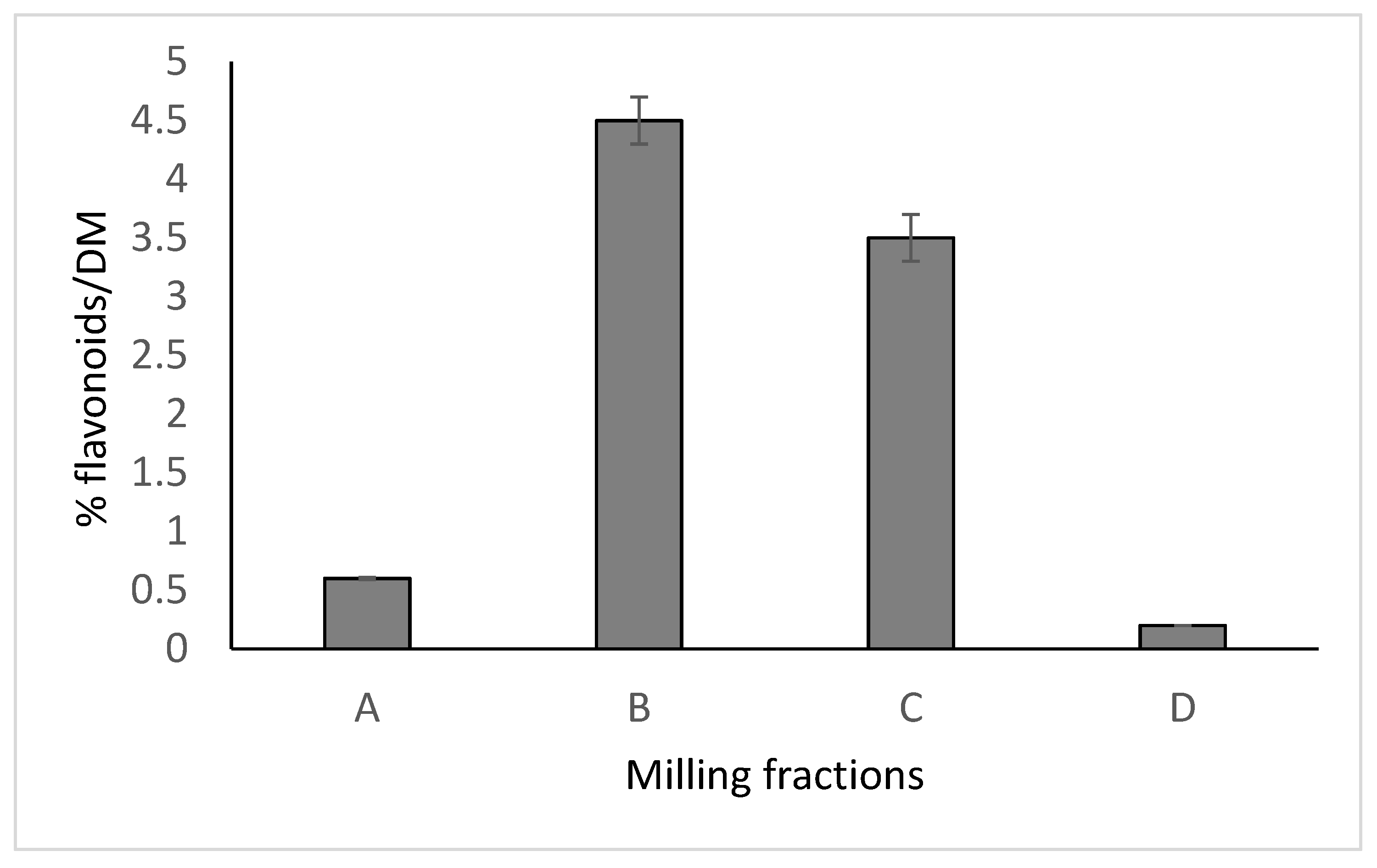
- Vombergar, B.; Škrabanja, V.; Germ, M. Flavonoid concentration in milling fractions of Tartary and common buckwheat. Fagopyrum 2020, 37, 11–21. [Google Scholar] [CrossRef]
- Ren, Q.; Liu, W.; Zhao, M.; Sai, C.M.; Wang, J.A. Changes in α-glucosidase inhibition, antioxidant, and phytochemical profiles during the growth of Tartary buckwheat (Fagopyrum tataricum Gaertn). Int. J. Food Prop. 2018, 21, 2689–2699. [Google Scholar] [CrossRef]
- Li, Y.Q.; Zhou, F.C.; Gao, F.; Bian, J.S.; Shan, F. Comparative evaluation of quercetin, isoquercetin and rutin as inhibitors of α-glucosidase. J. Agric. Food Chem. 2009, 57, 11463–11468. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Ghimeray, A.K.; Gurung, A.; Jin, C.W.; Rho, H.S.; Cho, D.H. Phenolic contents, antioxidant and α-glucosidase inhibition properties of nepalese strain buckwheat vegetables. Afr. J. Biotechnol. 2012, 11, 184–190. [Google Scholar] [CrossRef]
- Kočevar Glavač, N.; Stojilkovski, K.; Kreft, S.; Park, C.H.; Kreft, I. Determination of fagopyrins, rutin, and quercetin in Tartary buckwheat products. LWT Food Sci. Technol. 2017, 79, 423–427. [Google Scholar] [CrossRef]
- Kreft, S.; Janež, D.; Kreft, I. The content of fagopyrin and polyphenols in common and Tartary buckwheat sprouts. Acta Pharm. 2013, 63, 553–560. [Google Scholar] [CrossRef]
- Stojilkovski, K.; Kočevar Glavač, N.; Kreft, S.; Kreft, I. Fagopyrin and flavonoid contents in common, Tartary, and cymosum buckwheat. J. Food Compos. Anal. 2013, 32, 126–130. [Google Scholar] [CrossRef]
- Joshi, D.C.; Zhang, K.; Wang, C.; Chandora, R.; Khurshid, M.; Li, J.; He, M.; Georgiev, M.I.; Zhou, M. Strategic enhancement of genetic gain for nutraceutical development in buckwheat: A genomics-driven perspective. Biotechnol. Adv. 2020, 39, 107479. [Google Scholar] [CrossRef]
- Kim, J.; Hwang, K.T. Fagopyrins in different parts of common buckwheat (Fagopyrum esculentum) and Tartary buckwheat (F. tataricum) during growth. J. Food Compos. Anal. 2020, 86. [Google Scholar] [CrossRef]
- Zambounis, A.; Sytar, O.; Valasiadis, D.; Hilioti, Z. Effect of photosensitisers on growth and morphology of Phytophthora citrophthora coupled with leaf bioassays in pear seedlings. Plant Prot. Sci. 2020, 56, 74–82. [Google Scholar] [CrossRef]
- Rolta, R.; Yadav, R.; Salaria, D.; Sourirajan, A.; Dev, K. In silico screening of hundred phytocompounds of ten medicinal plants as potential inhibitors of nucleocapsid phosphoprotein of Covid-19: An approach to prevent virus assembly. J. Biomol. Struct. Dyn. 2020. [Google Scholar] [CrossRef]
- Peng, L.X.; Wang, J.B.; Hu, L.X.; Zhao, J.L.; Xiang, D.B.; Zou, L.; Zhao, G. Rapid and simple method for the determination of emodin in Tartary buckwheat (Fagopyrum tataricum) by high-performance liquid chromatography coupled to a diode array detector. J. Agric. Food Chem. 2013, 61, 854–857. [Google Scholar] [CrossRef] [PubMed]
- Robson, B. Computers and viral diseases. Preliminary bioinformatics studies on the design of a synthetic vaccine and a preventative peptidomimetic antagonist against the Sars-Cov-2 (2019-Ncov, Covid-19) coronavirus. Comput. Biol. Med. 2020, 119. [Google Scholar] [CrossRef] [PubMed]
- Subbaiyan, A.; Ravichandran, K.; Singh, S.V.; Sankar, M.; Thomas, P.; Dhama, K.; Malik, Y.S.; Singh, R.K.; Chaudhuri, P. In silico molecular docking analysis targeting Sars-Cov-2 spike protein and selected herbal constituents. J. Pure Appl. Microbiol. 2020, 14, 989–998. [Google Scholar] [CrossRef]
- Yin, Q.G.; Han, X.Y.; Han, Z.X.; Chen, Q.F.; Shi, Y.H.; Gao, H.; Zhang, T.Y.; Dong, G.Q.; Xiong, C.; Song, C.; et al. Genome-wide analyses reveals a glucosyltransferase involved in rutin and emodin glucoside biosynthesis in Tartary buckwheat. Food Chem. 2020, 318. [Google Scholar] [CrossRef]
- Qu, X.B.; Su, Z.M.; Hu, D.H.; Bao, Y.L.; Meng, X.Y.; Wu, Y.; Li, Y.X. Studies on molecular structure of hypericin and its interactions with HIV-1 protease by molecular modeling. Chem. J. Chin. Univ. 2009, 30, 1402–1405. [Google Scholar]
- Luthar, Z. Buckwheat genetic resources in Central Europe. In Buckwheat Germplasm in the World; Zhou, M., Kreft, I., Suvorova, G., Tang, Y., Woo, S.H., Eds.; Academic Press, An Imprint of Elsevier: London, UK, 2018; pp. 127–143. [Google Scholar] [CrossRef]
- Škrabanja, V.; Kreft, I.; Germ, M. Screening of common buckwheat genetic resources for recessive genes. In Buckwheat Germplasm in the World; Zhou, M., Kreft, I., Suvorova, G., Tang, Y., Woo, S.H., Eds.; Academic Press, An Imprint of Elsevier: London, UK, 2018; pp. 127–143. [Google Scholar] [CrossRef]
- Mizuno, N.; Yasui, Y. Gene flow signature in the S-allele region of cultivated buckwheat. BMC Plant Biol. 2019, 19, 1–9. [Google Scholar] [CrossRef]
- Matsui, K.; Mizuno, N.; Ueno, M.; Takeshima, R.; Yasui, Y. Development of co-dominant markers linked to a hemizygous region that is related to the self-compatibility locus (S) in buckwheat (Fagopyrum esculentum). Breed. Sci. 2020, 70, 112–117. [Google Scholar] [CrossRef]
- Matsui, K.; Yasui, Y. Genetic and genomic research for the development of an efficient breeding system in heterostylous self-incompatible common buckwheat (Fagopyrum esculentum). Theor. Appl. Genet. 2020, 133, 1641–1653. [Google Scholar] [CrossRef]
- Cawoy, V.; Ledent, J.F.; Kinet, J.M.; Jacquemart, A.L. Floral biology of common buckwheat (Fagopyrum esculentum Moench). Eur. J. Plant Sci. Biotechnol. 2009, 3, 1–9. [Google Scholar]
- Yabe, S.; Hara, T.; Ueno, M.; Enoki, H.; Kimura, T.; Nishimura, S.; Yasui, Y.; Ohsawa, R.; Iwata, H. Potential of genomic selection in mass selection breeding of an allogamous crop: An empirical study to increase yield of common buckwheat. Front. Plant Sci. 2018, 9, 1–12. [Google Scholar] [CrossRef]
- Yabe, S.; Iwata, H. Genomics-assisted breeding in minor and pseudo-cereals. Breed. Sci. 2020, 70, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.M.; Zhang, Y.L.; Zhang, Y.K.; Huang, K.H.; Yang, W.J.; Li, X.Y.; Zhang, Z.Y.; Wu, K.H.; Xu, X.; Ruan, R.W.; et al. De novo transcriptome assembly and identification of genes related to seed size in common buckwheat (Fagopyrum esculentum M.). Breed. Sci. 2019, 69, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Hara, T.; Shima, T.; Nagai, H.; Ohsawa, R. Genetic analysis of photoperiod sensitivity associated with difference in ecotype in common buckwheat. Breed. Sci. 2020, 70, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.J.; Ma, M.C.; Liu, L.L. Identification of genetic locus underlying easy dehulling in rice-Tartary for easy postharvest processing of Tartary buckwheat. Genes 2020, 11, 459. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.F.; Huang, X.Y.; Li, H.Y.; Yang, L.J.; Cui, Y.S. Recent progress in perennial buckwheat development. Sustainability 2018, 10, 536. [Google Scholar] [CrossRef]
- Morishita, T.; Shimizu, A.; Yamaguchi, H.; Degi, K. Development of common buckwheat cultivars with high antioxidative activity—‘Gamma no irodori’, ‘Cobalt no chikara’ and ‘Ruchiking’. Breed. Sci. 2019, 69, 514–520. [Google Scholar] [CrossRef]
- Gao, J.; Wang, T.T.; Liu, M.X.; Liu, J.; Zhang, Z.W. Transcriptome analysis of filling stage seeds among three buckwheat species with emphasis on rutin accumulation. PLoS ONE 2017, 12, e0189672. [Google Scholar] [CrossRef]
- Wang, L.J.; Sheng, M.Y.; Wen, P.C.; Du, J.Y. Morphological, physiological, cytological and phytochemical studies in diploid and colchicine-induced tetraploid plants of Fagopyrum tataricum (L.) Gaertn. Bot. Stud. 2017, 58, 1–12. [Google Scholar] [CrossRef][Green Version]
- Li, C.H.; Xie, Z.M.; Wang, Y.Q.; Lu, W.J.; Yin, G.F.; Sun, D.W.; Ren, C.Z.; Wang, L.H. Correlation and genetic analysis of seed shell thickness and yield factors in Tartary buckwheat (Fagopyrum tataricum (L.) Gaertn.). Breed. Sci. 2019, 69, 464–470. [Google Scholar] [CrossRef]
- Srejović, V.; Nesković, M. Regeneration of plants from cotyledon fragments of buckwheat (Fagopyrum esculentum Moench). Z. Pflanzenphysiol. 1981, 104, 37–42. [Google Scholar] [CrossRef]
- Miljuš Djukić, J.; Nesković, M.; Ninković, S.; Crkvenjakov, R. Agrobacterium-mediated transformation and plant-regeneration of buckwheat (Fagopyrum esculentum Moench). Plant Cell Tissue Organ Cult. 1992, 29, 101–108. [Google Scholar] [CrossRef]
- Luthar, Z.; Marchetti, S. Plant regeneration from mature cotyledons in a buckwheat (Fagopyrum esculentum Moench.) germplasm collection. Fagopyrum 1994, 14, 65–69. [Google Scholar]
- Woo, S.H.; Nair, A.; Adachi, T.; Campbell, C.G. Plant regeneration from cotyledon tissues of common buckwheat (Fagopyrum esculentum Moench). In Vitro Cell. Dev. Biol. Plant 2000, 36, 358–361. [Google Scholar] [CrossRef]
- Yamane, Y. Induced differentiation of buckwheat plants from subcultured calluses in Vitro. Jpn. J. Genet. 1974, 49, 139–146. [Google Scholar] [CrossRef]
- Lachmann, S.; Adachi, T. Callus regeneration from hypocotyl protoplasts of Tartary buckwheat (Fagopyrum tataricum Gartn.). Fagopyrum 1990, 10, 62–64. [Google Scholar]
- Suvorova, G. Buckwheat tissue cultures and genetic transformation. Mol. Breed. Nutr. Asp. Buckwheat 2016, 365–375. [Google Scholar] [CrossRef]
- Acquaah, G. Principles of Plant Genetics and Breeding, 2nd ed.; Wiley-Blackwell: Oxford, UK, 2012; p. 758. [Google Scholar]
- Brown, J.; Caligari, P.; Campos, H. Plant Breeding. In Introduction to Plant Breeding—Revised and Updated, 2nd ed.; Wiley-Blackwell: Oxford, UK, 2014; p. 287. [Google Scholar]
- Niroula, R.K.; Sah, B.P. Interspecific hybrids of buckwheat (Fagpyrum spp.) regenerated through embryo rescue. Sci. World 2006, 4, 74–77. [Google Scholar]
- Woo, S.H.; Roy, S.K.; Kwon, J.S.; Cho, S.W.; Kim, H.H. Interspecific crosses between Fagopyrum cymosum and other species through embryo culture techniques. In Buckwheat Germplasm in the World; Zhou, M., Kreft, I., Suvorova, G., Tang, Y., Woo, S.H., Eds.; Academic Press, An Imprint of Elsevier: London, UK, 2018; pp. 249–258. [Google Scholar] [CrossRef]
- Fesenko, N.N.; Fesenko, I.N. Silencing of dominant genes in heterozygous genotypes of interspecific hybrids Fagopyrum esculentum Moench X C2026 F. homotropicum Ohnishi. Russ. J. Genet. 2016, 52, 383–390. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luthar, Z.; Germ, M.; Likar, M.; Golob, A.; Vogel-Mikuš, K.; Pongrac, P.; Kušar, A.; Pravst, I.; Kreft, I. Breeding Buckwheat for Increased Levels of Rutin, Quercetin and Other Bioactive Compounds with Potential Antiviral Effects. Plants 2020, 9, 1638. https://doi.org/10.3390/plants9121638
Luthar Z, Germ M, Likar M, Golob A, Vogel-Mikuš K, Pongrac P, Kušar A, Pravst I, Kreft I. Breeding Buckwheat for Increased Levels of Rutin, Quercetin and Other Bioactive Compounds with Potential Antiviral Effects. Plants. 2020; 9(12):1638. https://doi.org/10.3390/plants9121638
Chicago/Turabian StyleLuthar, Zlata, Mateja Germ, Matevž Likar, Aleksandra Golob, Katarina Vogel-Mikuš, Paula Pongrac, Anita Kušar, Igor Pravst, and Ivan Kreft. 2020. "Breeding Buckwheat for Increased Levels of Rutin, Quercetin and Other Bioactive Compounds with Potential Antiviral Effects" Plants 9, no. 12: 1638. https://doi.org/10.3390/plants9121638
APA StyleLuthar, Z., Germ, M., Likar, M., Golob, A., Vogel-Mikuš, K., Pongrac, P., Kušar, A., Pravst, I., & Kreft, I. (2020). Breeding Buckwheat for Increased Levels of Rutin, Quercetin and Other Bioactive Compounds with Potential Antiviral Effects. Plants, 9(12), 1638. https://doi.org/10.3390/plants9121638










