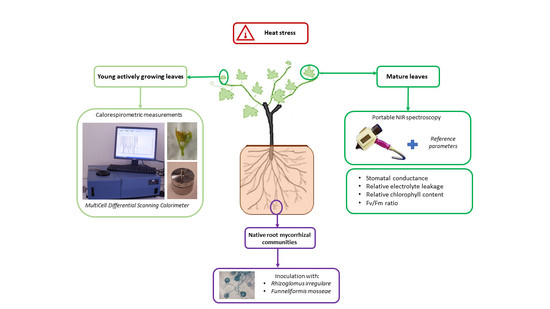
Response of Mycorrhizal ’Touriga Nacional‘ Variety Grapevines to High Temperatures Measured by Calorespirometry and Near-Infrared Spectroscopy
Abstract
1. Introduction
2. Results
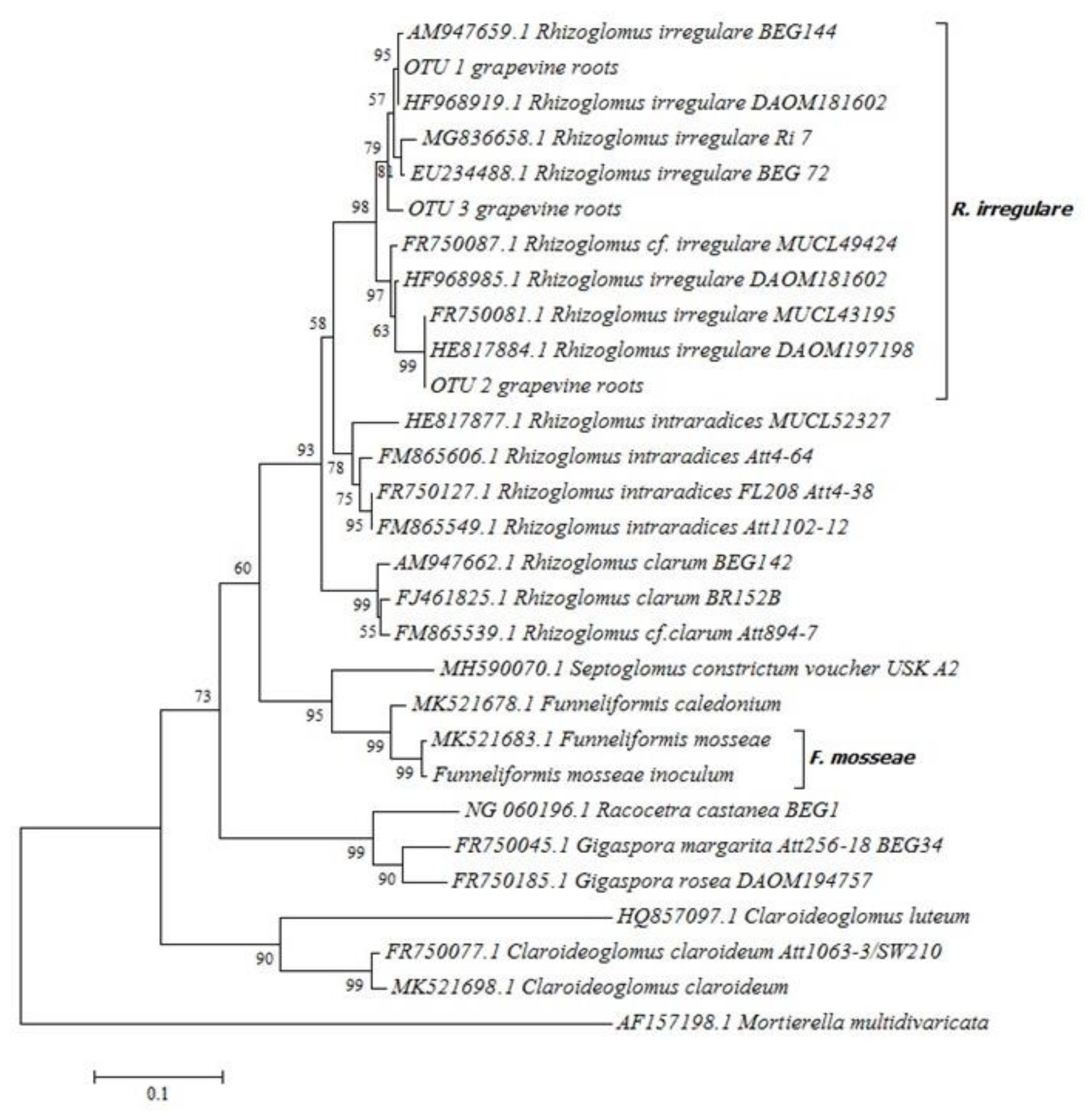
2.1. ‘Touriga Nacional’ Variety Vines Grafted onto 1103 Paulsen Rootstock were Naturally Colonized by Mycorrhizal Fungi
2.2. Grapevine Vegetative Growth Parameters were Influenced by Mycorrhizal Inoculation in the First Growing Season
2.3. ‘Touriga Nacional’ Variety Grapevines Adapted Their Physiology to Elevated Temperatures
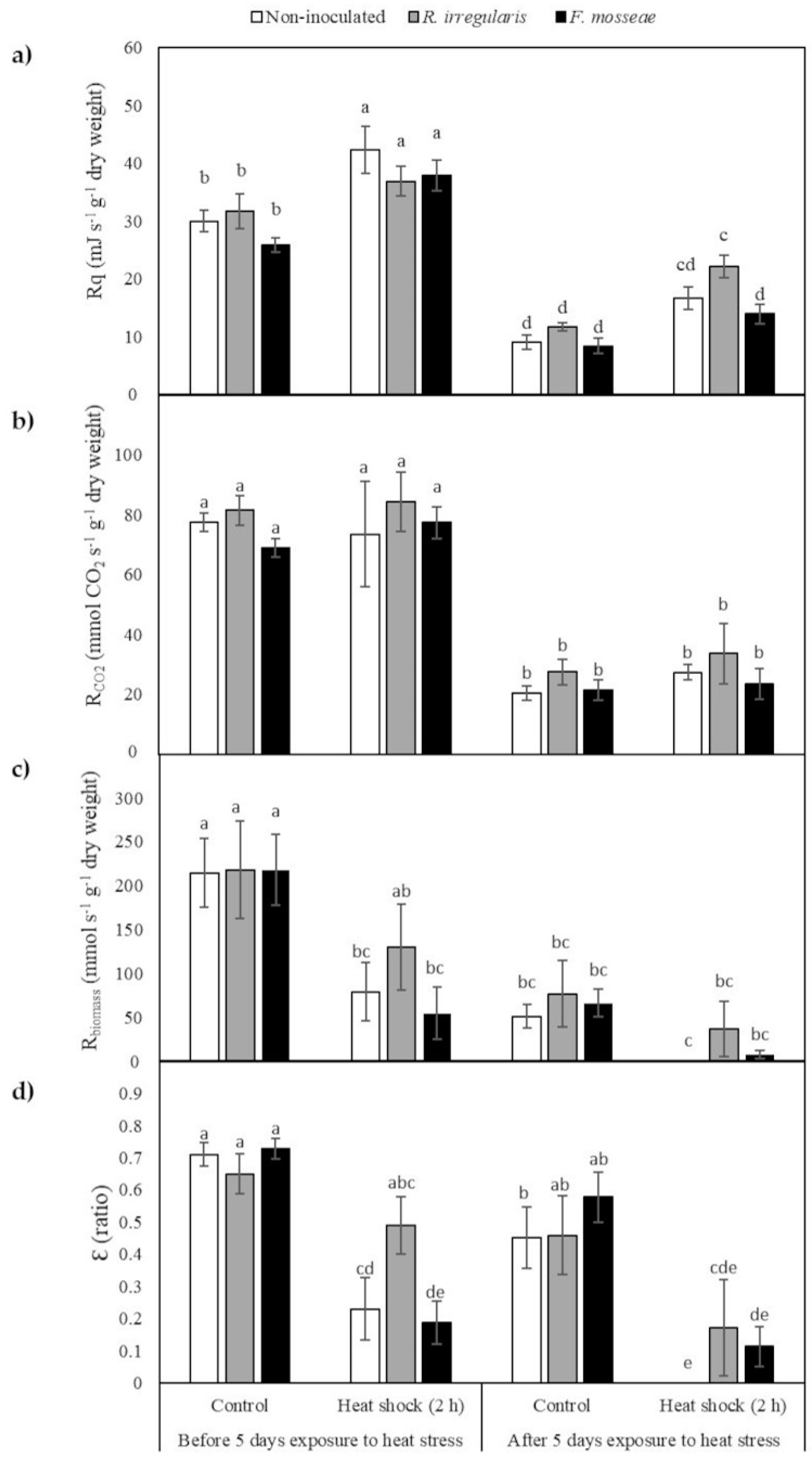
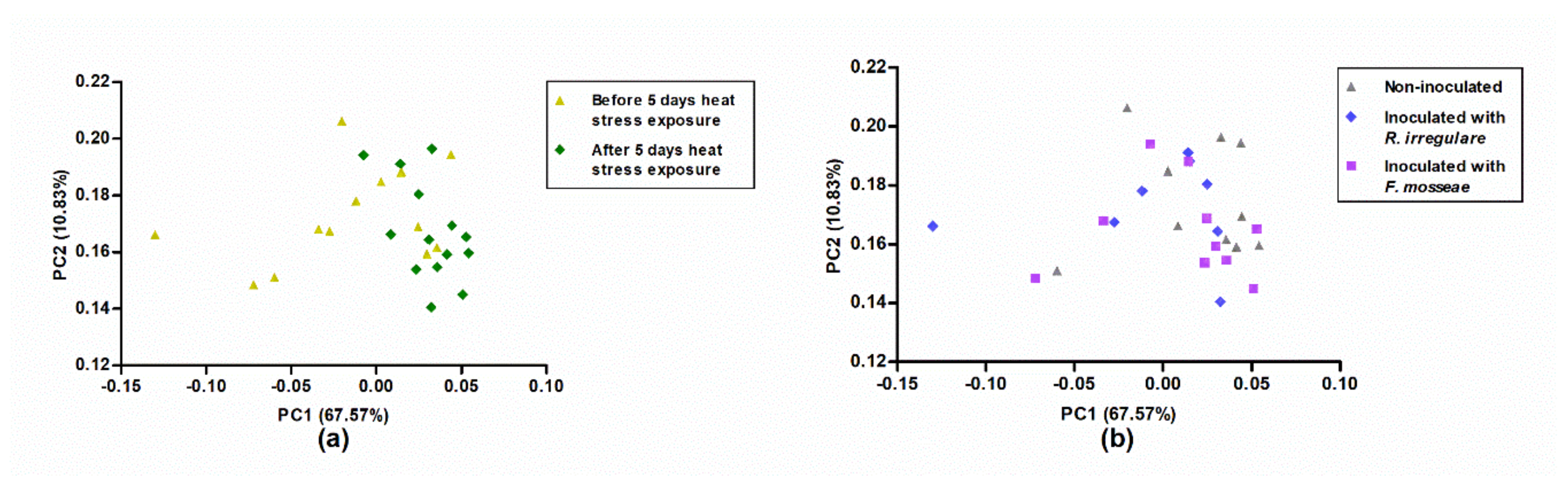
2.4. Calorespirometric Measurements Detected Short- and Long-Term Heat Stress Responses
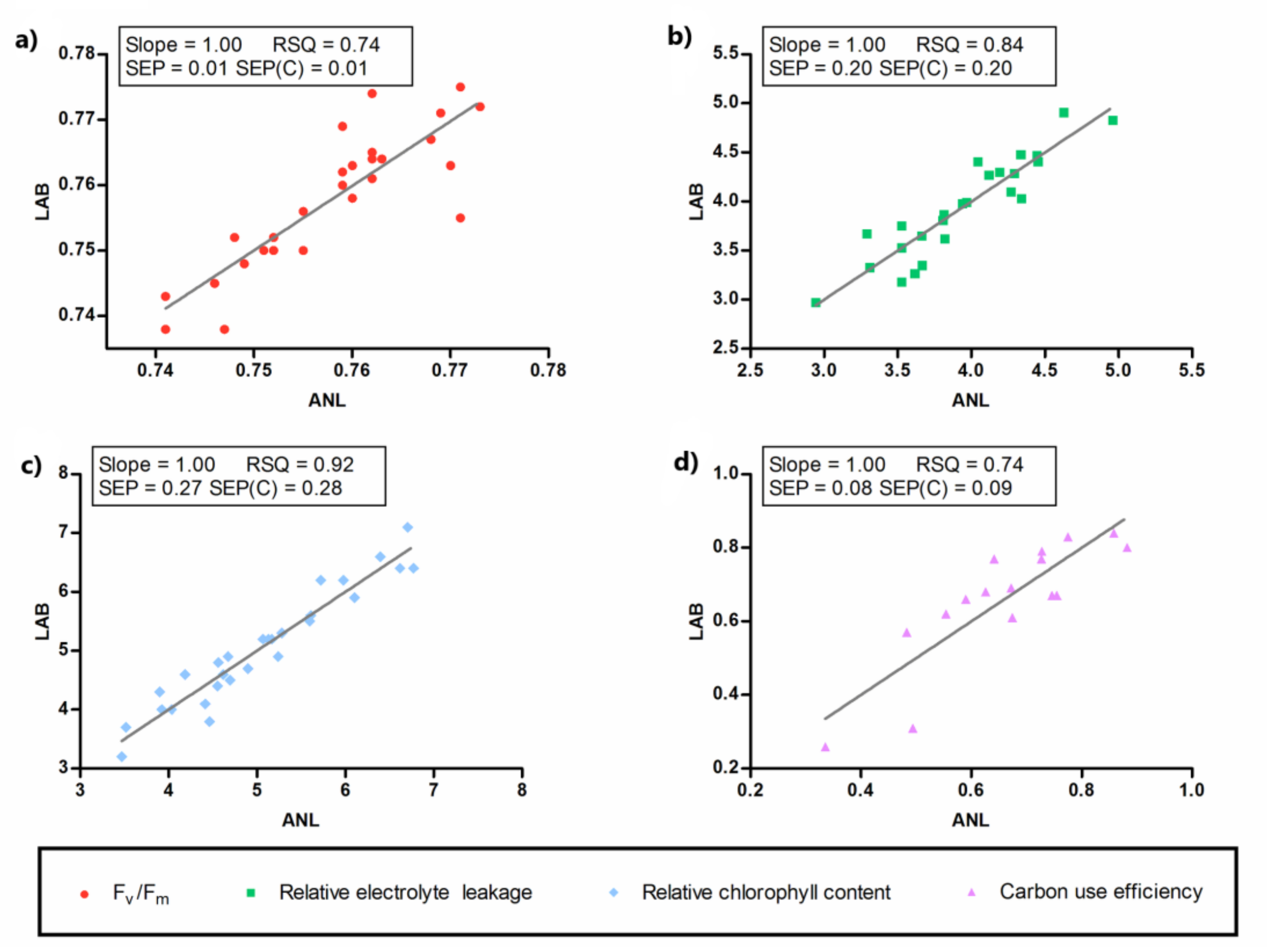
2.5. Near-Infrared Spectroscopy
3. Discussion
4. Material and Methods
4.1. Biological Material
4.2. Molecular Identification of Arbuscular Mycorrhizal Fungi
4.3. Plant Growth Conditions and High Temperature Treatment
4.4. Determination of Mycorrhizal Colonization Rate
4.5. Plant Growth and Physiology Measurements
4.6. Calorespirometric Measurements
4.7. Near-Infrared Spectroscopy
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- OIV—Organisation Internationale de la Vigne et du Vin Databases and Statistics. 2019. Available online: http://www.oiv.int/en/databases-and-statistics/statistics (accessed on 14 December 2019).
- Kliewer, W.M. Effect of high temperatures during the bloom-set period on fruit-set, ovule fertility, and berry growth of several grape cultivars. Am. J. Enol. Vitic. 1977, 28, 215–222. [Google Scholar]
- Berry, J.; Bjorkman, O. Photosynthetic response and adaptation to temperature in higher plants. Annu. Rev. Plant. Physiol. 1980, 31, 491–543. [Google Scholar] [CrossRef]
- Mullins, M.G.; Bouquet, A.; Williams, L.E. Biology of the grapevine; Cambridge University Press: Cambridge, UK, 1992; ISBN 0521305071. [Google Scholar]
- Malheiro, A.; Santos, J.; Fraga, H.; Pinto, J. Climate change scenarios applied to viticultural zoning in Europe. Clim. Res. 2010, 43, 163–177. [Google Scholar] [CrossRef]
- Kenny, G.J.; Harrison, P.A. The effects of climate variability and change on grape suitability in Europe. J. Wine Res. 1992, 3, 163–183. [Google Scholar] [CrossRef]
- Jones, G.V.; White, M.A.; Cooper, O.R.; Storchmann, K. Climate change and global wine quality. Clim. Chang. 2005, 73, 319–343. [Google Scholar] [CrossRef]
- Fraga, H.; Santos, J.A.; Malheiro, A.C.; Oliveira, A.A.; Moutinho-Pereira, J.; Jones, G.V. Climatic suitability of Portuguese grapevine varieties and climate change adaptation. Int. J. Climatol. 2016, 36, 1–12. [Google Scholar] [CrossRef]
- Wahid, A.; Gelani, S.; Ashraf, M.; Foolad, M.R. Heat tolerance in plants: An overview. Environ. Exp. Bot. 2007, 61, 199–223. [Google Scholar] [CrossRef]
- Howarth, C.J. Genetic improvements of tolerance to high temperature. In Abiotic Stresses. Plant Resistance through Breeding and Molecular Approaches; Harris, P., Ashraf, M., Eds.; CRC Press: Boca Raton, FL, USA, 2005; pp. 299–322. [Google Scholar]
- Scafaro, A.P.; Haynes, P.A.; Atwell, B.J. Physiological and molecular changes in Oryza meridionalis Ng., a heat-tolerant species of wild rice. J. Exp. Bot. 2010, 61, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, S.; Jagadish, S.V.K.; Prasad, A.S.H.; Thomar, A.K.; Anand, A.; Pal, M.; Agarwal, P.K. Local climate affects growth, yield and grain quality of aromatic and non-aromatic rice in northwestern India. Agric. Ecosyst. Environ. 2010, 138, 274–281. [Google Scholar] [CrossRef]
- Singh, R.P.; Prasad, P.V.V.; Sunita, K.; Giri, S.N.; Reddy, K.R. Influence of high temperature and breeding for heat tolerance in cotton: A review. Adv. Agron. 2007, 93, 313–385. [Google Scholar] [CrossRef]
- Bita, C.E.; Gerats, T. Plant tolerance to high temperature in a changing environment: Scientific fundamentals and production of heat stress-tolerant crops. Front. Plant. Sci. 2013, 4, 273. [Google Scholar] [CrossRef] [PubMed]
- Driedonks, N.; Rieu, I.; Vriezen, W.H. Breeding for plant heat tolerance at vegetative and reproductive stages. Plant. Reprod. 2016, 29, 67–79. [Google Scholar] [CrossRef]
- Grover, M.; Ali, S.Z.; Sandhya, V.; Rasul, A.; Venkateswarlu, B. Role of microorganisms in adaptation of agriculture crops to abiotic stresses. World J. Microbiol. Biotechnol. 2011, 27, 1231–1240. [Google Scholar] [CrossRef]
- Magalhães, N. Tratado de Viticultura. A Videira a Vinha e o Terroir, 2nd ed.; Esfera Poética Ed.; Publicações Chaves Ferreira: Lisboa, Portugal, 2015; ISBN 9789899820739. [Google Scholar]
- Costa, J.M.; Ortuño, M.F.; Lopes, C.M.; Chaves, M.M. Grapevine varieties exhibiting differences in stomatal response to water deficit. Funct. Plant. Biol. 2012, 39, 179–189. [Google Scholar] [CrossRef]
- Carvalho, L.C.; Coito, J.L.; Colaço, S.; Sangioco, M.; Amâncio, S. Heat stress in grapevine: The pros and cons of acclimation. Plant. Cell Environ. 2015, 38, 777–789. [Google Scholar] [CrossRef]
- Schüßler, A.; Schwarzott, D.; Walker, C. A new fungal phylum, the Glomeromycota: Phylogeny and evolution. Mycol. Res. 2001, 105, 1413–1421. [Google Scholar] [CrossRef]
- Trouvelot, S.; Bonneau, L.; Redecker, D.; Tuinen, V.; Adrian, M.; Wipf, D.; van Tuinen, D. Arbuscular mycorrhiza symbiosis in viticulture: A review. Agron. Sustain. Dev. 2015, 35, 1449–1467. [Google Scholar] [CrossRef]
- Zhu, X.; Song, F.; Xu, H. Influence of arbuscular mycorrhiza on lipid peroxidation and antioxidant enzyme activity of maize plants under temperature stress. Mycorrhiza 2010, 20, 325–332. [Google Scholar] [CrossRef]
- Zhu, X.-C.; Song, F.-B.; Liu, S.-Q.; Liu, T.-D. Effects of arbuscular mycorrhizal fungus on photosynthesis and water status of maize under high temperature stress. Plant. Soil 2011, 346, 189–199. [Google Scholar] [CrossRef]
- Maya, M.A.; Matsubara, Y. Influence of arbuscular mycorrhiza on the growth and antioxidative activity in cyclamen under heat stress. Mycorrhiza 2013, 23, 381–390. [Google Scholar] [CrossRef]
- Torres, N.; Antolín, M.C.; Goicoechea, N. Arbuscular mycorrhizal symbiosis as a promising resource for improving berry quality in grapevines under changing environments. Front. Plant. Sci. 2018, 9, 897. [Google Scholar] [CrossRef]
- Badger, M.R.; Bjorkman, O.; Armond, P.A. An analysis of photosynthetic response and adaptation to temperature in higher plants: Temperature acclimation in the desert evergreen Nerium oleander L. Plant. Cell Environ. 1982, 5, 85–99. [Google Scholar] [CrossRef]
- Chauhan, Y.S.; Senboku, T. Thermostabilities of cell-membrane and photosynthesis in cabbage cultivars differing in heat tolerance. J. Plant Physiol. 1996, 149, 729–734. [Google Scholar] [CrossRef]
- Stafne, E.T.; Clark, J.R.; Rom, C.R. Leaf gas exchange characteristics of red raspberry germplasm in a hot environment. HortScience 2000, 35, 278–280. [Google Scholar] [CrossRef]
- Stafne, E.T.; Clark, J.R.; Rom, C.R. Leaf gas exchange response of “Arapaho” blackberry and six red raspberry cultivars to moderate and high temperatures. HortScience 2001, 36, 880–883. [Google Scholar] [CrossRef]
- Herzog, H.; Chai-Arree, W. Gas exchange of five warm-season grain legumes and their susceptibility to heat stress. J. Agron. Crop. Sci. 2012, 198, 466–474. [Google Scholar] [CrossRef]
- Nagarajan, S.; Bansal, K.C. Measurement of cellular membrane thermostability to evaluate foliage heat tolerance of potato. Potato Res. 1986, 29, 163–167. [Google Scholar] [CrossRef]
- Srinivasan, A.; Takeda, H.; Senboku, T. Heat tolerance in food legumes as evaluated by cell membrane thermostability and chlorophyll fluorescence techniques. Euphytica 1996, 88, 35–45. [Google Scholar] [CrossRef]
- Yeh, D.M.; Lin, H.F. Thermostability of cell membranes as a measure of heat tolerance and relationship to flowering delay in Chrysanthemum. J. Am. Soc. Hort. Sci. 2003, 128, 656–660. [Google Scholar] [CrossRef]
- Rahman, U.H.; Malik, S.A.; Saleem, M. Heat tolerance of upland cotton during the fruiting stage evaluated using cellular membrane thermostability. F. Crop. Res. 2004, 85, 149–158. [Google Scholar] [CrossRef]
- Xu, W.; Subudhi, P.K.; Crasta, O.R.; Rosenow, D.T.; Mullet, J.E.; Nguyen, H.T. Molecular mapping of QTLs conferring stay-green in grain sorghum (Sorghum bicolor L. Moench). Genome 2000, 43, 461–469. [Google Scholar] [CrossRef]
- Rosyara, U.R.; Subedi, S.; Duveiller, E.; Sharma, R.C. The effect of spot blotch and heat stress on variation of canopy temperature depression, chlorophyll fluorescence and chlorophyll content of hexaploid wheat genotypes. Euphytica 2010, 174, 377–390. [Google Scholar] [CrossRef]
- Rosyara, U.R.; Subedi, S.; Duveiller, E.; Sharma, R.C. Photochemical efficiency and SPAD value as indirect selection criteria for combined selection of spot blotch and terminal heat stress in wheat. J. Phytopathol. 2010, 158, 813–821. [Google Scholar] [CrossRef]
- Xu, H.; Liu, G.; Liu, G.; Yan, B.; Duan, W.; Wang, L.; Li, S. Comparison of investigation methods of heat injury in grapevine (Vitis) and assessment to heat tolerance in different cultivars and species. BMC Plant Biol. 2014, 14, 156. [Google Scholar] [CrossRef]
- Agarie, S.; Hanaoka, N.; Kubota, F.; Agata, W.; Kaufman, P.B. Measurement of cell membrane stability evaluated by electrolyte leakage as a drought and heat tolerance test in rice (Oryza sativa L.). J. Fat. Agr. Kyushu Univ. 1995, 40, 233–240. [Google Scholar]
- Chenu, K.; Van Oosterom, E.J.; Mclean, G.; Deifel, K.S.; Fletcher, A.; Geetika, G.; Tirfessa, A.; Mace, E.S.; Jordan, D.R.; Sulman, R.; et al. Integrating modelling and phenotyping approaches to identify and screen complex traits—Illustration for transpiration efficiency in cereals. bioRxiv 2018. [Google Scholar] [CrossRef]
- Nogales, A.; Muñoz-Sanhueza, L.; Hansen, L.D.; Arnholdt-Schmitt, B. Calorespirometry as a tool for studying temperature response in carrot (Daucus carota L.). Eng. Life Sci. 2013, 13. [Google Scholar] [CrossRef]
- Nogales, A.; Muñoz-Sanhueza, L.; Hansen, L.D.; Arnholdt-Schmitt, B. Phenotyping carrot (Daucus carota L.) for yield-determining temperature response by calorespirometry. Planta 2015, 241. [Google Scholar] [CrossRef]
- Hansen, L.D.; Criddle, R.S.; Smith, B.N. Calorespirometry in plant biology. In Plant Respiration; Springer-Verlag: Berlin/Heidelberg, Germany, 2005; pp. 17–30. [Google Scholar]
- Criddle, R.S.; Smith, B.N.; Hansen, L.D. A respiration based description of plant growth rate responses to temperature. Planta 1997, 201, 441–445. [Google Scholar] [CrossRef]
- Criddle, R.S.; Hansen, L.D.; Smith, B.N.; Macfarlane, C.; Church, J.N.; Thygerson, T.; Jovanovic, T.; Booth, T. Thermodynamic law for adaptation of plants to environmental temperatures. Pure Appl. Chem 2005, 77, 1425–1444. [Google Scholar] [CrossRef]
- Wadsö, L.; Hansen, L.D. Calorespirometry of terrestrial organisms and ecosystems. Methods 2015, 76, 11–19. [Google Scholar] [CrossRef]
- Momen, B. Calorespirometry: A promising method for plant ecophysiology Research. Usda For. Serv. Proc. 2018, 6, 276–279. [Google Scholar]
- Klepsatel, P.; Wildridge, D.; Gáliková, M. Temperature induces changes in Drosophila energy stores. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Nelson, C.J. Genetic associations between photosynthetic characteristics and yield: review of the evidence. Plant Physiol. Biochem. 1988, 26, 543–554. [Google Scholar]
- McCarlie, V.W.; Hansen, L.D.; Smith, B.N.; Monsen, S.B.; Ellingson, D.J. Anabolic rates measured by calorespirometry for eleven subpopulation of Bromus tectorum match temperature profiles of local microclimates. Russ. J. Plant. Physiol. 2003, 50, 183–191. [Google Scholar] [CrossRef]
- Ellingson, D.; Olson, A.; Matheson, S.; Criddle, R.S.; Smith, B.N.; Hansen, L.D. Determination of the enthalpy change for anabolism by four methods. Thermochim. Acta 2003, 400, 79–85. [Google Scholar] [CrossRef]
- Gutiérrez, S.; Tardaguila, J.; Fernández-Novales, J.; Diago, M.P. Data mining and NIR spectroscopy in viticulture: Applications for plant phenotyping under field conditions. Sensors 2016, 16, 236. [Google Scholar] [CrossRef]
- Roberts, C.A.; Workman, J.; James, B.; Reeves, I. Near-Infrared Spectroscopy in Agriculture; Robers, C.A., WorkMan, J., Jr., Reeves, J.B., III, Eds.; American Society of Agronomy, Crop Science Society of America, Soil Science Society of America: Madison, WI, USA, 2004; ISBN 978-0-89118-236-8. [Google Scholar]
- Cozzolino, D.; Esler, M.B.; Dambergs, R.G.; Cynkar, W.U.; Boehm, D.R.; Francis, I.L.; Gishen, M. Prediction of colour and pH in grapes using a diode array spectrophotometer (400–1100 nm). J. Near Infrared Spectrosc. 2004, 12, 105–111. [Google Scholar] [CrossRef]
- Nogales-Bueno, J.; Hernández-Hierro, J.M.; Rodríguez-Pulido, F.J.; Heredia, F.J. Determination of technological maturity of grapes and total phenolic compounds of grape skins in red and white cultivars during ripening by near infrared hyperspectral image: A preliminary approach. Food Chem. 2014, 152, 586–591. [Google Scholar] [CrossRef]
- Nogales-Bueno, J.; Rodríguez-Pulido, F.J.; Heredia, F.J.; Hernández-Hierro, J.M. Comparative study on the use of anthocyanin profile, color image analysis and near-infrared hyperspectral imaging as tools to discriminate between four autochthonous red grape cultivars from La Rioja (Spain). Talanta 2015, 131, 412–416. [Google Scholar] [CrossRef]
- Nogales-Bueno, J.; Baca-Bocanegra, B.; Rodríguez-Pulido, F.J.; Heredia, F.J.; Hernández-Hierro, J.M. Use of near infrared hyperspectral tools for the screening of extractable polyphenols in red grape skins. Food Chem. 2015, 172, 559–564. [Google Scholar] [CrossRef]
- Smith, J.P.; Schmidtke, L.M.; Müller, M.C.; Holzapfel, B.P. Measurement of the concentration of nutrients in grapevine petioles by attenuated total reflectance Fourier transform infrared spectroscopy and chemometrics. Aust. J. Grape Wine Res. 2014, 20, 299–309. [Google Scholar] [CrossRef]
- Rustioni, L.; Rocchi, L.; Guffanti, E.; Cola, G.; Failla, O. Characterization of grape (Vitis vinifera L.) berry sunburn symptoms by reflectance. J. Agric. Food Chem. 2014, 62, 3043–3046. [Google Scholar] [CrossRef]
- Santos, A.O.; Kaye, O. Grapevine leaf water potential based upon near infrared spectroscopy. Sci. Agric. 2009, 66, 287–292. [Google Scholar] [CrossRef]
- Tardaguila, J.; Fernández-Novales, J.; Gutiérrez, S.; Diago, M.P. Non-destructive assessment of grapevine water status in the field using a portable NIR spectrophotometer. J. Sci. Food Agric. 2017, 97, 3772–3780. [Google Scholar] [CrossRef]
- Nogales-Bueno, J.; Ayala, F.; Hernández-Hierro, J.M.; Rodríguez-Pulido, F.J.; Echávarri, J.F.; Heredia, F.J. Simplified method for the screening of technological maturity of red grape and total phenolic compounds of red grape skin: Application of the characteristic vector method to near-infrared spectra. J. Agric. Food Chem. 2015, 63, 4284–4290. [Google Scholar] [CrossRef]
- van Tuinen, D.; Jacquot, E.; Zhao, B.; Gollotte, A.; Gianinazzi-Pearson, V. Characterization of root colonization profiles by a microcosm community of arbuscular mycorrhizal fungi using 25S rDNA-targeted nested PCR. Mol. Ecol. 1998, 7, 879–887. [Google Scholar] [CrossRef]
- Gollotte, A.; Van Tuinen, D.; Atkinson, D. Diversity of arbuscular mycorrhizal fungi colonising roots of the grass species Agrostis capillaris and Lolium perenne in a field experiment. Mycorrhiza 2004, 14, 111–117. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Tamura, K.; Nei, M. Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol. Biol. Evol. 1993, 10, 512–526. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Linderman, R.G.; Davis, E.A. Comparative response of selected grapevine rootstocks and cultivars to inoculation with different mycorrhizal fungi. Am. J. Enol. Vitic. 2001, 52, 8–11. [Google Scholar]
- Sembiring, H.; Raun, W.R.; Johnson, G.V.; Stone, M.L.; Solie, J.B.; Phillips, S.B. Detection of nitrogen and phosphorus nutrient status in winter wheat using spectral radiance. J. Plant. Nutr. 1998, 21, 1207–1233. [Google Scholar] [CrossRef]
- Edwards, D.; Berry, J.J. The efficiency of simulation-based multiple comparisons. Biometrics 1987, 43, 913–928. [Google Scholar] [CrossRef]
- Shenk, J.; Westerhaus, M. Routine Operation, Calibration, Development and Network System Management Manual; Shenk, J., Westerhaus, M., Eds.; NIRSystems Inc.: Silver Spring, MD, USA, 1995. [Google Scholar]
- Hepper, C.M.; Azcon-Aguilar, C.; Rosendahl, S.; Sen, R. Competition between three species of Glomus used as spatially separated introduced and indigenous mycorrhizal inocula for leek (Allium porrum L.). New Phytol. 1988, 110, 207–215. [Google Scholar] [CrossRef]
- Cano, C.; Bago, A. Competition and substrate colonization strategies of three polyxenically grown arbuscular mycorrhizal fungi. Mycologia 2005, 97, 1201–1214. [Google Scholar] [CrossRef]
- Verbruggen, E.; van der Heijden, M.G.A.; Rillig, M.C.; Kiers, E.T. Mycorrhizal fungal establishment in agricultural soils: Factors determining inoculation success. New Phytol. 2013, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Thonar, C.; Frossard, E.; Šmilauer, P.; Jansa, J. Competition and facilitation in synthetic communities of arbuscular mycorrhizal fungi. Mol. Ecol. 2014, 23, 733–746. [Google Scholar] [CrossRef]
- Lu, N.; Zhou, X.; Cui, M.; Yu, M.; Zhou, J.; Qin, Y.; Li, Y. Colonization with arbuscular mycorrhizal fungi promotes the growth of Morus alba L. seedlings under greenhouse conditions. Forests 2015, 6, 734–747. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D. Mycorrhizal Symbiosis; Academic press: Cambridge, MA, USA, 2008; ISBN 9780123705266. [Google Scholar]
- Munkvold, L.; Kjøller, R.; Vestberg, M.; Rosendahl, S.; Jakobsen, I. High functional diversity within species of arbuscular mycorrhizal fungi. New Phytol. 2004, 164, 357–364. [Google Scholar] [CrossRef]
- Camprubí, A.; Estaún, V.; Nogales, A.; García-Figueres, F.; Pitet, M.; Calvet, C. Response of the grapevine rootstock Richter 110 to inoculation with native and selected arbuscular mycorrhizal fungi and growth performance in a replant vineyard. Mycorrhiza 2008, 18. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.-H.; Eo, J.-K.; Ka, K.-H.; Eom, A.-H. Diversity of arbuscular mycorrhizal fungi and their roles in ecosystems. Mycobiology 2013, 41, 121–125. [Google Scholar] [CrossRef]
- Duc, N.H.; Csintalan, Z.; Posta, K. Arbuscular mycorrhizal fungi mitigate negative effects of combined drought and heat stress on tomato plants. Plant. Physiol. Biochem. 2018, 132, 297–307. [Google Scholar] [CrossRef]
- Ashraf, M.; Saeed, M.M.; Qureshi, M.J. Tolerance to high temperature in cotton (Gossypium hirsutum L.) at initial growth stages. Environ. Exp. Bot. 1994, 34, 275–283. [Google Scholar] [CrossRef]
- Marcum, K.B. Cell membrane thermostability and whole-plant heat tolerance of Kentucky Bluegrass. Crop. Sci. 1998, 38, 1214. [Google Scholar] [CrossRef]
- Blum, A.; Klueva, N.; Nguyen, H.T. Wheat cellular thermotolerance is related to yield under heat stress. Euphytica 2001, 117, 117–123. [Google Scholar] [CrossRef]
- Liu, D.-F.; Zhang, D.; Liu, G.-Q.; Hussain, S.; Teng, Y.-W. Influence of heat stress on leaf ultrastructure, photosynthetic performance, and ascorbate peroxidase gene expression of two pear cultivars (Pyrus pyrifolia). J. Zhejiang Univ-Sci. B (Biomed. Biotechnol.) 2013, 14, 1070–1083. [Google Scholar] [CrossRef]
- Feng, B.; Liu, P.; Li, G.; Dong, S.T.; Wang, F.H.; Kong, L.A.; Zhang, J.W. Effect of heat stress on the photosynthetic characteristics in flag leaves at the grain-filling stage of different heat-resistant winter wheat varieties. J. Agron. Crop. Sci. 2014, 200, 143–155. [Google Scholar] [CrossRef]
- Todorov, D.T.; Karanov, E.N.; Smith, A.R.; Hall, M.A. Chlorophyllase activity and chlorophyll content in wild and mutant plants of Arabidopsis thaliana. Biol. Plant 2003, 46, 125–127. [Google Scholar] [CrossRef]
- Sharkey, T.D.; Zhang, R. High temperature effects on electron and proton circuits of photosynthesis. J. Integr. Plant Biol. 2010, 52, 712–722. [Google Scholar] [CrossRef]
- Kadir, S.; Weihe, M. Von; Al-Khatib, K. Photochemical efficiency and recovery of photosystem II in grapes after exposure to sudden and gradual heat stress. J. Am. Soc. Hortic. Sci. 2007, 132, 764–769. [Google Scholar] [CrossRef]
- Marias, D.E.; Meinzer, F.C.; Still, C. Impacts of leaf age and heat stress duration on photosynthetic gas exchange and foliar nonstructural carbohydrates in Coffea arabica. Ecol. Evol. 2017, 7, 1297–1310. [Google Scholar] [CrossRef]
- Monteiro de Paula, F.; Pham Thi, A.; Zuily-Fodil, Y.; Ferrari-Iliou, R.; Vieira da Silva, J.; Mazliak, P. Effects of water stress on the biosynthesis and degradation of poly-unsaturated lipid molecular species in leaves of Vigna unguiculata. Plant. Physiol. Biochem. 1993, 31, 707–715. [Google Scholar]
- Costa Oliveira, M. Estudo da Composição Lipídica e Efeito do Estresse Hídrico em Membranas Foliares de Cajueiro-Anão-Precoce (Anacardium Occidentale L.); Universidade Federal do Ceará: Fortaleza, Brasil, 2001. [Google Scholar]
- Murakami, Y.; Tsuyama, M.; Kobayashi, Y.; Kodama, H.; Iba, K. Trienoic fatty acids and plant tolerance of high temperature. Science 2000, 287, 476–479. [Google Scholar] [CrossRef]
- Zheng, G.; Tian, B.; Zhang, F.; Tao, F.; Li, W. Plant adaptation to frequent alterations between high and low temperatures: Remodelling of membrane lipids and maintenance of unsaturation levels. Plant Cell Environ. 2011, 34, 1431–1442. [Google Scholar] [CrossRef]
- Su, K.; Bremer, D.J.; Jeannotte, R.; Welti, R.; Yang, C. Membrane lipid composition and heat tolerance in cool-season turfgrasses, including a hybrid bluegrass. J. Am. Soc. Hortic. Sci. 2009, 134, 511–520. [Google Scholar] [CrossRef]
- Wang, Q.-L.; Chen, J.-H.; He, N.-Y.; Guo, F.-Q. Metabolic reprogramming in chloroplasts under heat stress in plants. Int. J. Mol. Sci. 2018, 19, 849. [Google Scholar] [CrossRef] [PubMed]
- Sepúlveda, G.; Kliewer, W.M. Stomatal response of three grapevine cultivars (Vitis vinifera L.) to high temperature. Am. J. Enol. Vitic. 1986, 37, 44–52. [Google Scholar]
- Urban, J.; Ingwers, M.; McGuire, M.A.; Teskey, R.O. Stomatal conductance increases with rising temperature. Plant. Signal. Behav. 2017, 12, 3–6. [Google Scholar] [CrossRef]
- Smertenko, A.; Draber, P.; Viklicky, V.; Opatrny, Z. Heat stress affects the organization of microtubules and cell division in Nicotiana tabacum cells. Plant Cell Environ. 1997, 20, 1534–1542. [Google Scholar] [CrossRef]
- Potters, G.; Pasternak, T.P.; Guisez, Y.; Palme, K.J.; Jansen, M.A.K. Stress-induced morphogenic responses: Growing out of trouble? Trends Plant. Sci. 2007, 12, 98–105. [Google Scholar] [CrossRef]
- Stoutemyer, M.R.; Burger, D.W. Calorespirometric studies of in vitro-grown carnation (Dianthus caryophyllus L. var. ’Improved White Sim’) shoot tips. Plant Cell. Tissue Organ. Cult. 1998, 53, 189–196. [Google Scholar] [CrossRef]
- Yu, W.; Hansen, L.D.; Fan, W.; Zhao, W.; McArthur, E.D. Adaptation of growth and respiration of three varieties of Caragana to environmental temperature. Asian J. Plant. Sci. 2008, 7, 67–72. [Google Scholar] [CrossRef]
- Rocheta, M.; Coito, J.L.; Ramos, M.J.N.; Carvalho, L.; Becker, J.D.; Carbonell-Bejerano, P.; Amâncio, S. Transcriptomic comparison between two Vitis vinifera L. varieties (Trincadeira and Touriga Nacional) in abiotic stress conditions. BMC Plant Biol. 2016, 16, 1–19. [Google Scholar] [CrossRef]
- Al-Karaki, G.N. Salt stress response of salt-sensitive and tolerant durum wheat cultivars inoculated with mycorrhizal fungi. Acta Agron. Hung. 2001, 49, 25–34. [Google Scholar] [CrossRef]
- Al-Karaki, G.N.; Hammad, R. Mycorrhizal influence on fruit yield and mineral content of tomato grown under salt stress. J. Plant Nutr. 2001, 24, 1311–1323. [Google Scholar] [CrossRef]
- Al-Karaki, G.N.; Hammad, R.; Rusan, M. Response of two tomato cultivars differing in salt tolerance to inoculation with mycorrhizal fungi under salt stress. Mycorrhiza 2001, 11, 43–47. [Google Scholar] [CrossRef]
- Quiroga, G.; Erice, G.; Aroca, R.; Chaumont, F.; Ruiz-Lozano, J.M. Enhanced drought stress tolerance by the arbuscular mycorrhizal symbiosis in a drought-sensitive maize cultivar is related to a broader and differential regulation of host plant aquaporins than in a drought-tolerant cultivar. Front. Plant Sci. 2017, 8, 1056. [Google Scholar] [CrossRef]
- Nogales, A.; Santos, E.S.; Abreu, M.M.; Arán, D.; Victorino, G.; Pereira, H.S.; Lopes, C.M.; Viegas, W. Mycorrhizal inoculation differentially affects grapevine’s performance in copper contaminated and non-contaminated soils. Front. Plant Sci. 2019, 9, 1906. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Calvet, C.; Camprubi, A.; Pérez-Hernández, A.; Lovato, P.E. Plant growth stimulation and root colonization potential of in vivo versus in vitro arbuscular mycorrhizal inocula. HortScience 2013, 48, 897–901. [Google Scholar] [CrossRef]
- Saitou, N.; Nei, M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef]
- Phillips, J.M.; Hayman, D.S. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc. 1970, 55, 158–161. [Google Scholar] [CrossRef]
- Koske, R.E.; Gemma, J.N. A modified procedure for staining roots to detect VA mycorrhizas. Mycol. Res. 1989, 92, 486–488. [Google Scholar] [CrossRef]
- Giovannetti, M.; Mosse, B. An evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol. 1980, 84, 489–500. [Google Scholar] [CrossRef]
- Lopes, C.M.; Pinto, P.A. Easy and accurate estimation of grapevine leaf area with simple mathematical models. Vitis 2005, 44, 55–61. [Google Scholar]
- Liu, W.; Yu, K.; He, T.; Li, F.; Zhang, D.; Liu, J. The low temperature induced physiological responses of Avena nuda L., a cold-tolerant plant species. Scientificworldjournal, 2013; 93. [Google Scholar] [CrossRef]
- Hansen, L.D.; Church, J.N.; Matheson, S.; McCarlie, V.W.; Thygerson, T.; Criddle, R.S.; Smith, B.N. Kinetics of plant growth and metabolism. Thermochim. Acta 2002, 388, 415–425. [Google Scholar] [CrossRef]
- Macfarlane, C.; Adams, M.A.; Hansen, L.D. Application of an enthalpy balance model of the relation between growth and respiration to temperature acclimation of Eucalyptus globulus seedlings. Proc. R. Soc. London Ser. B Biol. Sci. 2002. [Google Scholar] [CrossRef]
- Brereton, R.G. Calibration. In Chemometrics; Brereton, R.B., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2003; pp. 271–338. [Google Scholar]
- Geladi, P.; MacDougall, D.; Martens, H. Linearization and scatter-correction for near-infrared reflectance spectra of meat. Appl. Spectrosc. 1985, 39, 491–500. [Google Scholar] [CrossRef]
- Dhanoa, M.S.; Lister, S.J.; Barnes, R.J. On the scales associated with near-infrared reflectance difference spectra. Appl. Spectrosc. 1995, 49, 765–772. [Google Scholar] [CrossRef]
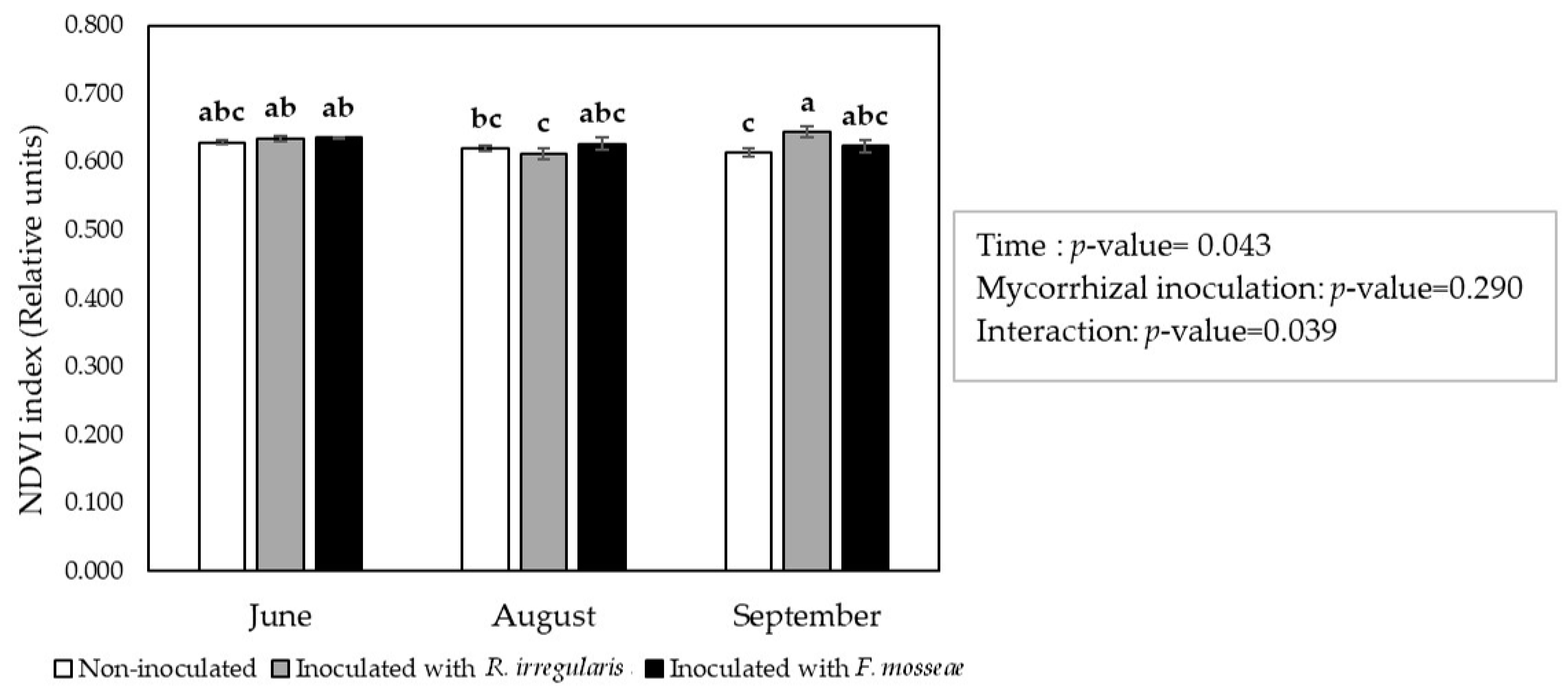
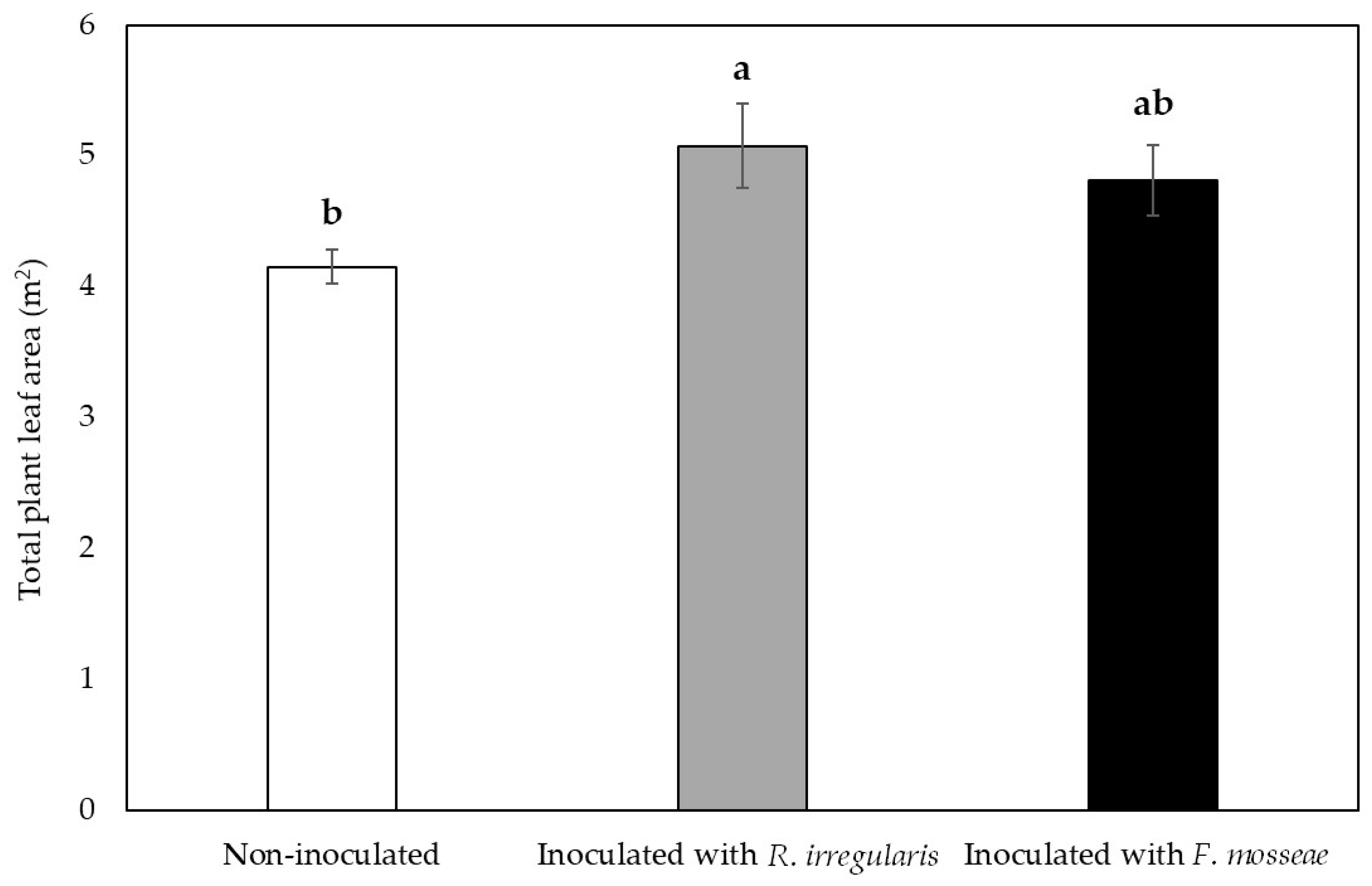

| Inoculation Treatment | 3 Months after Inoculation | 10 Months after Inoculation |
|---|---|---|
| Non-inoculated | 0.78 ± 0.007 a | 0.81 ± 0.296 a |
| Inoculated with R. irregulare | 0.63 ± 0.008 b | 0.75 ± 0.164 b |
| Inoculated with F. mosseae | 0.70 ± 0.039 ab | 0.76 ± 0.379 ab |
| Effects | p-Value | |||
|---|---|---|---|---|
| Relative Electrolite Leakage | Stomatal Conductance | Ratio of Variable to Maximum Chlorophyll Fluorescence | Relative Chlorophyll Content | |
| Long-term heat stress exposure | 0.004 | <0.001 | 0.325 | <0.001 |
| Mycorrhizal inoculation | 0.638 | 0.006 | 0.959 | 0.675 |
| Long-term heat stress exposure x Mycorrhizal inoculation | 0.276 | 0.061 | 0.907 | 0.586 |
| Mycorrhizal Treaments | Relative Electrolite Leakage (%) | Stomatal Conductance (mmol. m2.s−1) | Ratio of Variable to Maximum Chlorophyll Fluorescence (Ratio) | Relative Chlorophyll Content (SPAD Units) | ||||
|---|---|---|---|---|---|---|---|---|
| Before Stress | After 5-Day Exposure to Heat Stress | Before Stress | After 5-Day Exposure to Heat stress | Before Stress | After 5-Day Exposure to Heat Stress | Before Stress | After 5-Day Exposure to Heat Stress | |
| Non-inoculated | 4.51 ± 0.438 | 3.63 ± 0.121 | 67 ± 8.5 b | 119 ± 19.6 ab | 0.7587 ± 0.0046 | 0.7579 ± 0.0047 | 4.24 ± 0.327 | 5.47 ± 0.282 |
| Inoculated with R. irregulare | 5.11 ± 0.583 | 3.81 ± 0.255 | 58 ± 1.8 b | 228 ± 29.0 a | 0.7625 ± 0.0077 | 0.7574 ± 0.0031 | 4.32 ± 0.186 | 5.91 ± 0.303 |
| Inoculated with F. mosseae | 4.21 ± 0.219 | 3.90 ± 0.160 | 65 ± 7.8 b | 190 ± 8.8 a | 0.7606 ± 0.0046 | 0.7552 ± 0.0046 | 4.69 ± 0.259 | 5.67 ± 0.478 |
| Effects | p-Value | |||
|---|---|---|---|---|
| Metabolic Heat Rate (Rq) | Respiratory Rate (Rco2) | Structural Biomass Formation Rate (Rbiomass) | Carbon use Efficiency (Ɛ) | |
| Long-term heat stress exposure | <0.0001 | <0.0001 | <0.0001 | 0.0003 |
| Short-term heat stress exposure | <0.0001 | 0.3799 | <0.0001 | <0.0001 |
| Mycorrhizal inoculation | 0.1069 | 0.1602 | 0.6960 | 0.4647 |
| Long-term heat stress exposure x mycorrhizal inoculation | 0.5035 | 0.9855 | 0.8484 | 0.3530 |
| Short-term heat stress exposure x mycorrhizal inoculation | 0.8677 | 0.9479 | 0.6674 | 0.1634 |
| Long-term heat stress exposure x short-term heat stress exposure | 0.5035 | 0.7915 | 0.0556 | 0.9512 |
| Long-term heat stress exposure x short-term stress x mycorrhizal inoculation | 0.2361 | 0.7279 | 0.8596 | 0.7685 |
| Spectral Pre-Treatments a | Reference Variables | N b | PLS Factors | Mean | SD c | SEC d | RSQ e | SECV f | SECV (%) | SEP g | SEP (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| SNV + detrend 2,5,5,1 | Fv/Fm1 | 27 | 2 | 0.76 | 0.01 | 0.01 | 0.72 | 0.01 | 1.81 | 0.01 | 0.66 |
| Detrend 0,0,1,1 | EL2 | 25 | 4 | 3.94 | 0.51 | 0.22 | 0.81 | 0.33 | 8.43 | 0.20 | 5.05 |
| SNV 2,10,10,1 | ChlC3 | 28 | 4 | 5.05 | 0.98 | 0.30 | 0.91 | 0.67 | 13.37 | 0.27 | 5.35 |
| MSC 0,0,1,1 | Ɛ4 | 16 | 3 | 0.66 | 0.17 | 0.10 | 0.67 | 0.14 | 20.78 | 0.08 | 12.60 |
| SNV + detrend 0,0,1,1 | Rq5 | 25 | 2 | 5343.0 | 1530.57 | 1117.66 | 0.47 | 1229.66 | 23.01 | 1048.46 | 19.62 |
| SNV 2,5,5,1 | Rco26 | 28 | 3 | 12.02 | 6.19 | 2.37 | 0.85 | 4.76 | 31.14 | 2.20 | 14.38 |
| MSC 2,10,10,1 | gs7 | 27 | 1 | 112.37 | 62.92 | 49.14 | 0.39 | 56.83 | 37.74 | 47.29 | 31.41 |
| MSC 0,0,1,1 | Rbiomass8 | 16 | 3 | 45.69 | 26.65 | 16.21 | 0.63 | 24.39 | 38.82 | 14.04 | 22.34 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nogales, A.; Ribeiro, H.; Nogales-Bueno, J.; Hansen, L.D.; Gonçalves, E.F.; Coito, J.L.; Rato, A.E.; Peixe, A.; Viegas, W.; Cardoso, H. Response of Mycorrhizal ’Touriga Nacional‘ Variety Grapevines to High Temperatures Measured by Calorespirometry and Near-Infrared Spectroscopy. Plants 2020, 9, 1499. https://doi.org/10.3390/plants9111499
Nogales A, Ribeiro H, Nogales-Bueno J, Hansen LD, Gonçalves EF, Coito JL, Rato AE, Peixe A, Viegas W, Cardoso H. Response of Mycorrhizal ’Touriga Nacional‘ Variety Grapevines to High Temperatures Measured by Calorespirometry and Near-Infrared Spectroscopy. Plants. 2020; 9(11):1499. https://doi.org/10.3390/plants9111499
Chicago/Turabian StyleNogales, Amaia, Hugo Ribeiro, Julio Nogales-Bueno, Lee D. Hansen, Elsa F. Gonçalves, João Lucas Coito, Ana Elisa Rato, Augusto Peixe, Wanda Viegas, and Hélia Cardoso. 2020. "Response of Mycorrhizal ’Touriga Nacional‘ Variety Grapevines to High Temperatures Measured by Calorespirometry and Near-Infrared Spectroscopy" Plants 9, no. 11: 1499. https://doi.org/10.3390/plants9111499
APA StyleNogales, A., Ribeiro, H., Nogales-Bueno, J., Hansen, L. D., Gonçalves, E. F., Coito, J. L., Rato, A. E., Peixe, A., Viegas, W., & Cardoso, H. (2020). Response of Mycorrhizal ’Touriga Nacional‘ Variety Grapevines to High Temperatures Measured by Calorespirometry and Near-Infrared Spectroscopy. Plants, 9(11), 1499. https://doi.org/10.3390/plants9111499