Weather Conditions and Maturity Group Impacts on the Infestation of First Generation European Corn Borers in Maize Hybrids in Croatia
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Experimental Fields and Trial Design
4.2. Meteorological Data
4.3. Trial Assessments
4.4. Data Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Martel, C.; Réjasse, A.; Rousset, F.; Bethenod, M.T.; Bourguet, D. Host-plant-associated genetic differentiation in Northern French populations of the European corn borer. Heredity 2003, 90, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, W.D.; Burkness, E.C.; Mitchell, P.D.; Moon, R.D.; Leslie, T.W.; Fleischer, S.J.; Abrahamson, M.; Hamilton, K.L.; Steffey, K.L.; Gray, M.E.; et al. Areawide suppression of European corn borer with Bt maize reaps savings to non-Bt maize growers. Science 2010, 330, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Szőke, C.; Zsubori, Z.; Pók, I.; Rácz, F.; Illés, O.; Szegedi, I. Significance of the European corn borer (Ostrinia nubilalis Hübn.) in maize production. Acta Agron. Hung. 2002, 50, 447–461. [Google Scholar] [CrossRef]
- Ponsard, S.; Bethenod, M.T.; Bontemps, A.; Pélozuelo, L.; Souqual, M.C.; Bourguet, D. Carbon stable isotopes: A tool for studying the mating, oviposition, and spatial distribution of races of European corn borer, Ostrinia nubilalis, among host plants in the field. Can. J. Zool. 2004, 82, 1177–1185. [Google Scholar] [CrossRef]
- Wright, M.G.; Kuhar, T.P.; Hoffmann, M.P.; Chenus, S.A. Effect of inoculative releases of Trichogramma ostriniae on populations of Ostrinia nubilalis and damage to sweet corn and field corn. Biol. Control 2002, 23, 149–155. [Google Scholar] [CrossRef]
- Bohn, M.; Schulz, B.; Kreps, R.; Klein, D.; Melchinger, A.E. QTL mapping for resistance against the European corn borer (Ostrinia nubilalis H.) in early maturing European dent germplasm. Theor. Appl. Genet. 2000, 101, 907–917. [Google Scholar] [CrossRef]
- Raemisch, D.R.; Walgenbach, D.D. Evaluation of insecticides for control of first-brood European corn borer (Lepidoptera: Pyralidae) and effect on silage yield in South Dakota. J. Econ. Entomol. 1983, 76, 654–656. [Google Scholar] [CrossRef]
- Lew, H.; Adler, A.; Edinger, W. Moniliformin and the European corn borer (Ostrinia nubilalis). Mycotoxin Res. 1991, 7, 71–76. [Google Scholar] [CrossRef]
- Magg, T.; Melchinger, A.E.; Klein, D.; Bohn, M. Relationship between European corn borer resistance and concentration of mycotoxins produced by Fusarium spp. in grains of transgenic Bt maize hybrids, their isogenic counterparts, and commercial varieties. Plant Breed. 2002, 121, 146–154. [Google Scholar] [CrossRef]
- Munkvold, G.P.; Hellmich, R.L.; Showers, W.B. Reduced Fusarium ear rot and symptomless infection in kernels of maize genetically engineered for European corn borer resistance. Phytopathology 1997, 87, 1071–1077. [Google Scholar] [CrossRef]
- Hudon, M.; McLeod, D.G.R.; Foott, W.H. Control of the European Corn Borer. Agric. Can. 1982. Available online: https://atrium.lib.uoguelph.ca/xmlui/bitstream/handle/10214/15098/FDMR_control_european_corn_borer82.pdf?sequence=1&isAllowed=y (accessed on 1 September 2020).
- Lynch, R.E.; Robinson, J.F.; Berry, E.C. European corn borer: Yield losses and damage resulting from a simulated natural infestation. J. Econ. Entomol. 1980, 73, 141–144. [Google Scholar] [CrossRef]
- Ivezic, M. The corn-borer (Ostrinia nubilalis Hbn.) and the weed flora as influential factors in the corn production. Zb. Rad. 1976, 2, 153–163. [Google Scholar]
- Maceljski, M. Poljoprivredna Entomologija, 2nd ed.; Zrinski d.d: Čakovec, Croatia, 2002; pp. 323–337. [Google Scholar]
- Ivezić, M.; Raspudić, E. Tolerantnost pojedinih hibrida kukuruza na kukuruznog moljca (Ostrinia nubilalis Hbn.) na području Slavonije. In Zbornik Sažetaka, Proceedings of the 34. Znanstveni Skup Hrvatskih Agronoma s Međunarodnim Sudjelovanjem, Opatija, Croatia, 25–28 February 1998; Hitrec, I., Ed.; Agronomski Fakultet: Zagreb, Croatia, 1998; p. 223. [Google Scholar]
- Raspudić, E.; Ivezić, M.; Brmež, M.; Majić, I.; Sarajlić, A. Intenzitet napada kukuruznog moljca (Ostrinia nubilalis Hübner) u plodoredu i monokulturi kukuruza. In Zbornik Sažetaka, Proceedings of the 45. Hrvatski i 5. Međunarodni Simpozij Agronoma, Opatija, Croatia, 15–19 February 2010; Marić, S., Lončarić, Z., Eds.; Poljoprivredni Fakultet Osijek: Osijek, Croatia, 2010; pp. 901–905. [Google Scholar]
- Derozari, M.B.; Showers, W.B.; Shaw, R.H. Environment and the sexual activity of the European corn borer. Environ. Entomol. 1977, 6, 657–665. [Google Scholar] [CrossRef]
- Trnka, M.; Muška, F.; Semerádová, D.; Dubrovský, M.; Kocmánková, E.; Žalud, Z. European corn borer life stage model: Regional estimates of pest development and spatial distribution under present and future climate. Ecol. Model. 2007, 207, 61–84. [Google Scholar] [CrossRef]
- Raspudić, E.; Sarajlić, A.; Ivezić, M.; Majić, I.; Brmež, M.; Gumze, A. Efficiency of the chemical treatment against the European corn borer in seed maize production. Poljoprivreda 2013, 19, 11–15. [Google Scholar]
- Sarajlić, A.; Raspudić, E.; Lončarić, Z.; Josipović, M.; Brmež, M.; Ravlić, M.; Zebec, V.; Majić, I. Significance of irrigation treatments and weather conditions on European corn borer appearance. Maydica 2018, 62, 8. [Google Scholar]
- Capinera, J.L. Encyclopedia of Entomology, 1st ed.; Springer Science & Business Media: Berlin, Germany, 2008; pp. 1374–1376. [Google Scholar]
- Rosca, I.; Rada, I. Entomology (Agriculture, Horticulture, Forest), 1st ed.; Alpha Mdn. Publishing House: London, UK, 2009; pp. 119–123. [Google Scholar]
- Barbulescu, A.; Popov, S.I.; Sabau, I. The behavior of a Monsanto maize hybrid-Dekalb 512 bt to the attack by the European corn borer in Romania. Rom. Agric. Res. 2001, 15, 65–73. [Google Scholar]
- Georgescu, E.; Toader, M.; Cană, L.; Rîşnoveanu, L. Researches Concerning European Corn Borer (Ostrinia nubilalis Hbn.) Control, In South-East of the Romania. Agron. Ser. Sci. Res. 2019, 62, 301–308. [Google Scholar]
- Lemić, D.; Mandić, J.; Čačija, M.; Drmić, Z.; Mrganić, M.; Čavlovićak, S.; Bažok, R.; Virić Gašparić, H. European corn borer and its parasites overwintering abundance and damages on different corn FAO maturity groups. J. Cent. Eur. Agric. 2019, 20, 447–460. [Google Scholar] [CrossRef]
- Condon, G.C. Ecological Engineering for Pest Management of Ostrinia nubilalis Hübner (Lepidoptera: Crambidae) in Peppers. Ph.D. Thesis, Rutgers University-School of Graduate Studies, New Brunswick, NJ, USA, 2020. [Google Scholar]
- Guthrie, W.D.; Barry, B.D. Methodologies used for screening and determining resistance in maize to the European corn borer. In Toward Insect Resistant Maize for the Third World, Proceedings of the International Symposium on Methodologies for Developing Host Plant Resistance to Maize Insects, Mexico City, Mexico, 9–14 March 1987; CIMMYT: Mexico, Mexico, 1989. [Google Scholar]
- Barry, D.; Darrah, L.L. Effect of research on commercial hybrid maize resistance to European corn borer (Lepidoptera: Pyralidae). J. Econ. Entomol. 1991, 84, 1053–1059. [Google Scholar] [CrossRef]
- Barry, B.D.; Darrah, L.L. Impact of mechanisms of resistance on European corn borer resistance in selected maize hybrids. Insect Resist. Maize 1994, 21, 21–28. [Google Scholar]
- Showers, W.B.; Witkowski, J.F.; Mason, C.E.; Poston, F.L.; Welch, S.M. Management of the European corn borer. North Central Reg. Publ. Iowa State Univ. Ames Iowa 1983, 22, 1–57. [Google Scholar]
- Rice, M.E.; Ostlie, K. European corn borer management in field corn: A survey of perceptions and practices in Iowa and Minnesota. J. Prod. Agric. 1997, 10, 628–634. [Google Scholar] [CrossRef]
- Raspudić, E.; Ivezić, M.; Mlinarević, M. Utjecaj transgenih Bt hibrida kukuruza na kukuruznog moljca (Ostrinia nubilalis Hübner). In Glasnik Zaštite Bilja, Proceedings of the Sažeci Priopćenja, 43. Seminara iz Zaštite Bilja, Opatija, Croatia, 9–11 February 1999; Maceljski, M., Ed.; Zadružna štampa: Zagreb, Croatia, 1999; pp. 6–7. [Google Scholar]
- Augustinović, Z.; Raspudić, E.; Ivezić, M.; Brmež, M.; Andreata-Koren, M.; Ivanek-Martinčić, M.; Samobor, V. Utjecaj kukuruznog moljca (Ostrinia nubilalis Hübner) na hibride kukuruza u sjeverozapadnoj i istočnoj Hrvatskoj. Poljoprivreda 2005, 11, 24–29. [Google Scholar]
- Kumar, H.; Mihm, J.A. Resistance in maize hybrids and inbreds to first-generation southwestern corn borer, Diatraea grandiosella (Dyar) and sugarcane borer, Diatraea saccharalis Fabricius. Crop Prot. 1996, 15, 311–317. [Google Scholar] [CrossRef]
- Patch, L.H. Some factors determining corn borer damage. J. Econ. Entomol. 1929, 22, 174–183. [Google Scholar] [CrossRef]
- Leppik, E.; Frérot, B. Maize field odorscape during the oviposition flight of the European corn borer. Chemoecology 2014, 24, 221–228. [Google Scholar] [CrossRef]
- Ivezić, M.; Raspudić, E.; Brmež, M.; Majić, I.; Brkić, I.; Tollefson, J.J.; Bohn, M.; Hibbard, B.E.; Šimić, D. A review of resistance breeding options targeting western corn rootworm (Diabrotica virgifera virgifera LeConte). Agric. For. Entomol. 2009, 11, 307–311. [Google Scholar] [CrossRef]
- Kadličko, S.R.; Tollefson, J.J.; Prasifka, J.; Bača, F.; Stanković, G.J.; Delić, N. Evaluation of Serbian commercial maize hybrid tolerance to feeding by larval Western corn rootworm (Diabrotica virgifera virgifera LeConte) using the novel ‘difference approach’. Maydica 2010, 55, 179–185. [Google Scholar]
- Fontes-Puelba, A.A.; Bernal, J.S. Resistance and Tolerance to Root Herbivory in Maize Were Mediated by Domestication, Spread, and Breeding. Front. Plant Sci. 2020, 11, 223. [Google Scholar] [CrossRef]
- Szulc, P.; Idziak, R.; Woznica, Z.; Sobiech, L. Assessment of susceptibility of “stay-green” type maize cultivars (Zea mays L.) to maize smut (Ustilago maydis (DC.) Corda) and European corn borer (Ostrinia nubilalis Hbn.). Prog. Plant Prot. 2014, 54, 368–374. [Google Scholar]
- Demirel, N.; Konuskan, O. A study on percentages of damage ratios of the European corn borer (ECB), Ostrinia nubilalis (Hübner) (Lepidoptera: Pyralidae) on sweet corn cultivars. Entomol. Appl. Sci. Lett. 2017, 4, 1–4. [Google Scholar] [CrossRef]
- Everett, T.R.; Chiang, H.C.; Hibbs, E.T. Some Factors Influencing Populations of European Corn Borer (Pyrausta nubilalis [Hbn.]) in the North Central States; University of Minnesota, Agricultural Experiment Station: Columbia, MN, USA, 1958; Technical Bulletin 229. [Google Scholar]
- Chiang, H.C.; Jarvis, J.L.; Burkhardt, C.C.; Fairchild, M.L.; Weekman, G.T.; Triplehorn, C.A. Populations of European Corn Borer Ostrinia nubilalis (Hbn.) in Field Corn, Zea mays (L.); University of Missouri, College of Agriculture, Agricultural Experiment Station: Columbia, MO, USA, 1961; Research Bulletin 776. [Google Scholar]
- Sparks, A.N.; Triplehorn, C.A.; Chiang, H.C.; Guthrie, W.D.; Brindley, T.A. Some factors influencing populations of European corn borer, Ostrinia nubilalis (Hubner) in the north central states. Resistance of corn, time of planting, and weather conditions. Part II, 1958–1962. Iowa Agric. Home Econ. Exp. Stn. Res. Bull. 1967, 559, 66–103. [Google Scholar]
- Bažok, R.; Igrc Barčić, J.; Kos, T.; Gotlin Čuljak, T.; Šilović, M.; Jelovčan, S.; Kozina, A. Monitoring and efficacy of selected insecticides for European corn borer (Ostrinia nubilalis Hubn., Lepidoptera: Crambidae) control. J. Pest Sci. 2009, 82, 311–319. [Google Scholar] [CrossRef]
- Showers, W.B.; De Rozari, M.B.; Reed, G.L.; Shaw, R.H. Temperature-Related Climatic Effects on Survivorship of the European Corn Borer. Environ. Entomol. 1978, 7, 717–723. [Google Scholar] [CrossRef]
- Gylling Data Management Inc. ARM 9® GDM Software, Revision 9.2014.7 January 28, 2015 (B = 20741); Gylling Data Management Inc.: Brookings, SD, USA, 2019. [Google Scholar]
| Weather Indicator | Average Value ± SD for Year 1 | HSDp = 0.05 | |
|---|---|---|---|
| 2017 | 2018 | ||
| Average monthly temperature in May (°C) | 17.78 ± 0.83 b 2 | 19.45 ± 0.83 a | 1.5 |
| Average monthly temperature in June (°C) | 22.90 ± 1.02 a | 21.55 ± 1.49 b | 0.752 |
| Total monthly amount of rainfall in May (mm) | 58.48 ± 12.95 | 103.60 ± 40.21 | ns 3 |
| Total monthly amount of rainfall in June (mm) | 45.45 ± 24.99 | 118.20 ± 52.16 | ns |
| Average air humidity in May (%) | 68.00 ± 4.08 | 71.93 ± 5.91 | ns |
| Average air humidity in June (%) | 65.00 ± 5.66 | 70.23 ± 11.53 | ns |
| Weather Indicator | Average Value ± SD for Location 1 | HSDp = 0.05 | |||
|---|---|---|---|---|---|
| Šašinovec | Tovarnik | Gola | Vrana | ||
| Average monthly temperature in May (°C) | 18.10 ± 0.71 | 19.10 ± 1.98 | 17.95 ± 1.48 | 19.30 ± 0.57 | ns 2 |
| Average monthly temperature in June (°C) | 21.45 ± 1.20 b 3 | 22.35 ± 0.92 ab | 21.15 ± 1.20 b | 23.95 ± 0.49 a | 1.613 |
| Total monthly amount of rainfall in May (mm) | 98.60 ± 80.75 | 80.15 ± 30.05 | 65.30 ± 10.89 | 80.10 ± 27.72 | ns |
| Total monthly amount of rainfall in June (mm) | 108.25 ± 69.65 | 99.65 ± 91.57 | 83.90 ± 16.83 | 35.50 ± 27.72 | ns |
| Average air humidity in May (%) | 75.00 ± 2.83 | 65.75 ± 3.18 | 71.35 ± 4.74 | 67.75 ± 6.72 | ns |
| Average air humidity in June (%) | 75.20 ± 3.11 | 65.85 ± 6.86 | 72.00 ± 9.90 | 57.40 ± 5.09 | ns |
| FAO Maturity Group | 2017 | 2018 |
|---|---|---|
| FAO 300 | 14.13 ± 14.28 b 1 | 20.57 ± 20.89 ab |
| FAO 400 | 15.57 ± 14.65 b | 18.95 ± 18.03 b |
| FAO 500 | 18.46 ± 16.77 ab | 23.54 ± 22.00 a |
| FAO 600 | 20.27 ± 20.31 a | 23.71 ± 22.94 a |
| HSDp = 0.05 2 | 4.525 | 4.487 |
| Locality | FAO Maturity Group | HSDp = 0.05 3 | |||
|---|---|---|---|---|---|
| 300 1 | 400 1 | 500 1 | 600 1 | ||
| Šašinovec | 1.54 ± 0.89 d 2 B 3 | 1.6 ± 0.97 d AB | 3.76 ± 1.00 b A | 1.99 ± 1.08 c AB | 2.21 |
| Gola | 6.29 ± 1.73 c | 8.14 ± 1.74 c | 7.93 ± 1.52 b | 9.87 ± 1.99 b | ns 4 |
| Tovarnik | 15.83 ± 1.55 b | 17.43 ± 1.74 b | 23.50 ± 1.47 a | 23.58 ± 1.79 a | ns |
| Vrana | 24.61 ± 1.50 a | 27.02 ± 1.16 a | 30.52 ± 1.71 a | 34.47 ± 1.80 a | ns |
| HSDp = 0.05 2 | 3.44 | 3.69 | 4.54 | 4.99 | |
| Locality | FAO Group | HSDp = 0.05 3 | |||
|---|---|---|---|---|---|
| 300 1 | 400 1 | 500 1 | 600 1 | ||
| Šašinovec | 6.33 ± 1.17 c | 8.56 ± 1.04 c | 8.39 ± 0.95 c | 8.78 ± 1.15 c | ns 4 |
| Gola | 1.35 ± 1.00 d | 1.68 ± 1.09 d | 2.21 ± 1.09 d | 1.36 ± 1.20 d | ns |
| Tovarnik | 27.03 ± 1.95 b | 25.03 ± 1.69 b | 31.21 ± 1.84 b | 30.22 ± 1.66 b | ns |
| Vrana | 39.97 ± 1.29 a AB | 33.97 ± 1.27 a B | 45.59 ± 1.22 a A | 47.14 ± 1.43 a A | 9.85 |
| HSDp = 0.05 2 | 3.36 | 3.05 | 3.50 | 3.26 | |
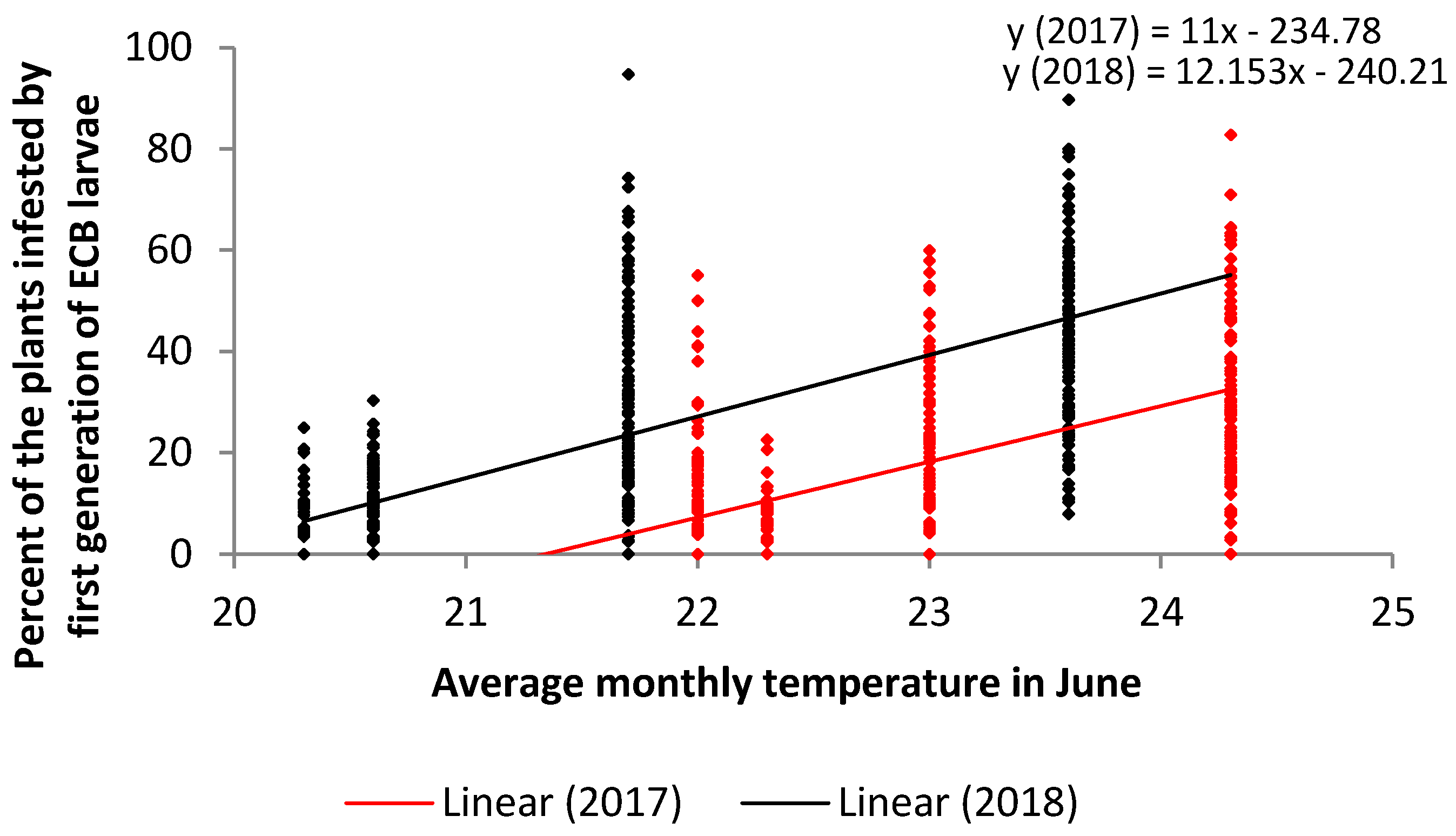
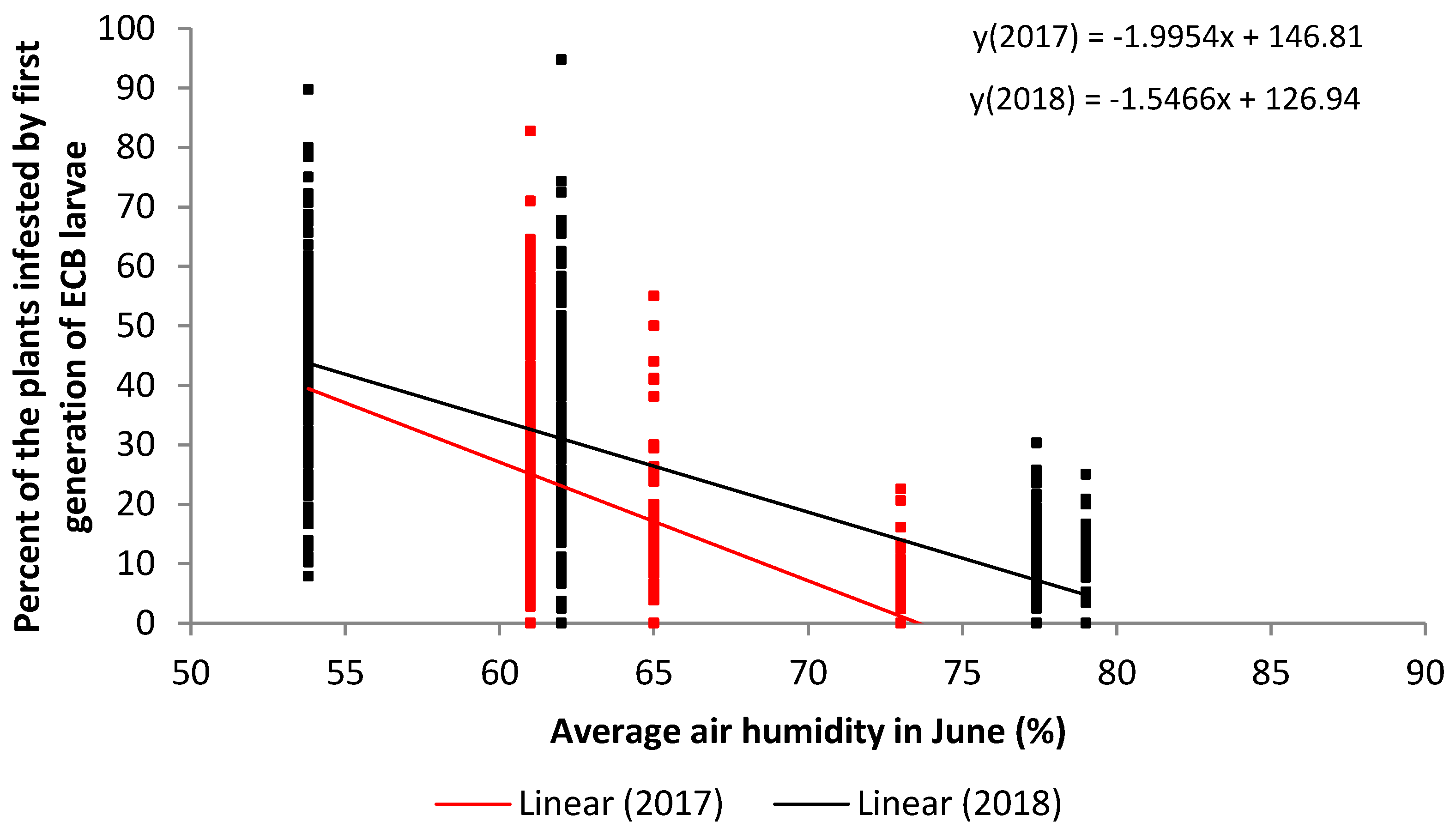
| Independent Variable | Month | Year | n | Correlation Coefficient r | Coefficient of Determination r2 | p | Type of Correlation |
|---|---|---|---|---|---|---|---|
| Mean air temperature | May | 2017 | 512 | 0.48 | 0.2375 | 0.0001 | medium |
| May | 2018 | 512 | 0.57 | 0.3274 | 0.0001 | strong | |
| June | 2017 | 512 | 0.59 | 0.3563 | 0.0001 | strong | |
| June | 2018 | 512 | 0.74 | 0.5570 | 0.0001 | strong | |
| Total amount of rainfall | May | 2017 | 512 | 0.23 | 0.0574 | 0.0001 | very weak |
| May | 2018 | 512 | 0.03 | 0.0014 | 0.0405 | not existing | |
| June | 2017 | 512 | −0.57 | 0.3298 | 0.0001 | strong | |
| June | 2018 | 512 | −0.25 | 0.0667 | 0.0001 | weak | |
| Average air humidity | May | 2017 | 512 | −0.59 | 0.3567 | 0.0001 | strong |
| May | 2018 | 512 | 0.14 | 0.0225 | 0.0007 | very weak | |
| June | 2017 | 512 | −0.58 | 0.3392 | 0.0001 | strong | |
| June | 2018 | 512 | −0.77 | 0.6027 | 0.0001 | very strong |
| FAO 300 | FAO 400 | FAO 500 | FAO 600 | ||||
|---|---|---|---|---|---|---|---|
| Hybrid | Company | Hybrid | Company | Hybrid | Company | Hybrid | Company |
| Bc 344 | Bc 1 | Os 444 | Os | Os 552 | Os | Bc 682 | Bc |
| Bc 323 | Bc | Bc 406 | Bc | Bc 525 | Bc | Bc 616 | Bc |
| Bc 306 | Bc | Bc 424 | Bc | Bc 575 | Bc | Bc 626 | Bc |
| TRIO | Bc | Bc 482 | Bc | Klipan | Bc | Riđan | Bc |
| P9903 | DuPont 2 | DKC 4608 | DeKalb 4 | DKC 5830 | DeKalb | P1535 | DuPont |
| Os 378 | Os 3 | Kulak | Os | Velimir | Os | Rudolf 60 | Os |
| Os 398 | Os | Tomasov | Os | Os 5922 | Os | Os 6217 | Os |
| Os 3617 | Os | Drava 404 | Os | Os 515 | Os | Os 635 | Os |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bažok, R.; Pejić, I.; Čačija, M.; Virić Gašparić, H.; Lemić, D.; Drmić, Z.; Kadoić Balaško, M. Weather Conditions and Maturity Group Impacts on the Infestation of First Generation European Corn Borers in Maize Hybrids in Croatia. Plants 2020, 9, 1387. https://doi.org/10.3390/plants9101387
Bažok R, Pejić I, Čačija M, Virić Gašparić H, Lemić D, Drmić Z, Kadoić Balaško M. Weather Conditions and Maturity Group Impacts on the Infestation of First Generation European Corn Borers in Maize Hybrids in Croatia. Plants. 2020; 9(10):1387. https://doi.org/10.3390/plants9101387
Chicago/Turabian StyleBažok, Renata, Ivan Pejić, Maja Čačija, Helena Virić Gašparić, Darija Lemić, Zrinka Drmić, and Martina Kadoić Balaško. 2020. "Weather Conditions and Maturity Group Impacts on the Infestation of First Generation European Corn Borers in Maize Hybrids in Croatia" Plants 9, no. 10: 1387. https://doi.org/10.3390/plants9101387
APA StyleBažok, R., Pejić, I., Čačija, M., Virić Gašparić, H., Lemić, D., Drmić, Z., & Kadoić Balaško, M. (2020). Weather Conditions and Maturity Group Impacts on the Infestation of First Generation European Corn Borers in Maize Hybrids in Croatia. Plants, 9(10), 1387. https://doi.org/10.3390/plants9101387