In Vitro Decoated Seed Germination and Seedling Development for Propagation of Wild Mandrake (Mandragora autumnalis Bertol.)
Abstract
1. Introduction
2. Results and Discussion
2.1. Lifespan and Phenology of Wild Mandrake (Mandragora autumnalis Bertol.) Plants
2.2. Seed Viability vs. Periodic Storage Conditions
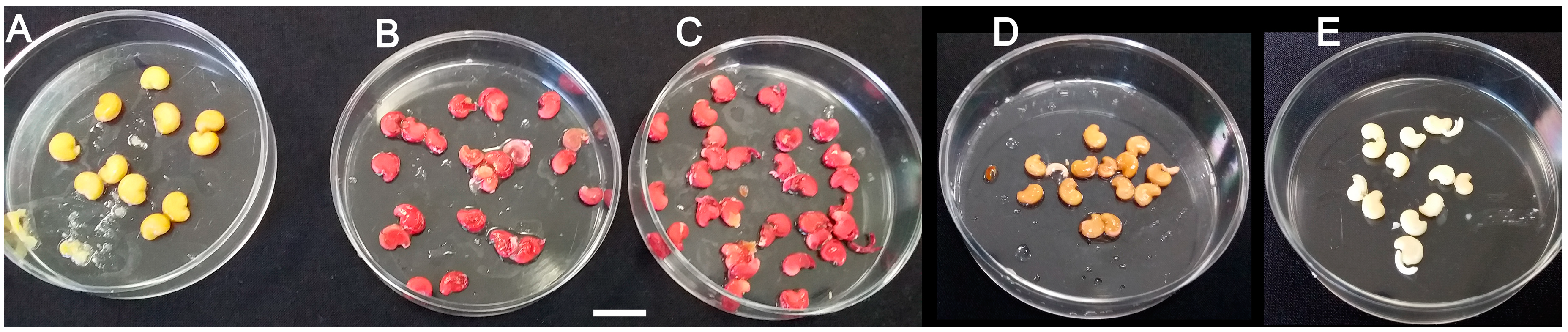
2.3. Water Permeability of M. autumnalis Seed Coats
2.4. Intact Mature Seed Germination and Seedling Emergence: Preliminary Tests
2.5. Intact Seed Storage Conditions vs. Decoated Seed Germinability
2.6. Removal of Seed Coats and Exogenous Application of Gibberellic acid Promote In Vitro Seed Germination and Subsequent Seedling Emergence
2.7. Growth Features of the In Vitro Established M. autumnalis Plantlets Grown on SGM Cultures
2.8. Growth Features of the In Vitro Established M. autumnalis Plants Transplanted into Potting Soil and Grown under Glasshouse Conditions
3. Materials and Methods
3.1. Collection of Mature Seeds
3.2. Surface Sterilization and Decoating of the Mature Seeds
3.3. Seed Viability Testing
3.4. Water Permeability of Seed Coats
3.5. In Vitro Seed Germination Conditions
3.6. Controlled Maintenance Conditions of Cultured Plant Materials
3.7. Plantlets Transplantation and Acclimatization
3.8. Statistical Analysis
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Brown, D. Encyclopedia of Herbs and Their Uses; The Herb Society of America: Willoughby, OH, USA; Dorling Kindersley: London, UK, 1995; pp. 156–308. [Google Scholar]
- Jackson, B.P.; Berry, M.I. Mandragora taxonomy and chemistry of the European species. In The Biology and Taxonomy of the Solanaceae. Linnean Society Symposium Series No. 7; Hawkes, J.G., Lester, R.N., Skelding, A.D., Eds.; Academic Press: London, UK, 1979; pp. 505–512. [Google Scholar]
- Fakir, H.; Özçelik, H. Mandragora officinarum L. (Solanaceae): A new record for the flora of Turkey. Afr. J. Biotechnol. 2009, 8, 3560–3564. [Google Scholar]
- Piccillo, G.A.; Mondati, E.G.; Moro, P.A. Six clinical cases of Mandragora autumnalis poisoning: Diagnosis and treatment. Eur. J. Emerg. Med. 2002, 9, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Carter, A.J. Myths and mandrakes. J. R. Soc. Med. 2003, 96, 144–147. [Google Scholar] [CrossRef]
- Chidiac, E.J.; Kaddoum, R.N.; Fuleihan, S.F. Mandragora: Anesthetic of the ancients. Anesth. Analg. 2012, 115, 1437–1441. [Google Scholar] [CrossRef]
- Aburjai, T.; Hudaib, M.; Tayyema, R.; Yousef, M.; Qishawi, M. Ethnopharmacological survey of medicinal herbs in Jordan, the Ajloun Heights region. J. Ethnopharmacol. 2007, 110, 294–304. [Google Scholar] [CrossRef]
- Al-Quran, S. Ethnopharmacological survey of wild medicinal plants in Showbak, Jordan. J. Ethnopharmacol. 2009, 123, 45–50. [Google Scholar] [CrossRef]
- Hanuš, L.O.; Řezanka, T.; Spížek, J.; Dembitsky, V.M. Substances isolated from Mandragora species. Phytochemistry 2005, 66, 2408–2417. [Google Scholar] [CrossRef]
- Bekkouche, K.; Daali, Y.; Cherkaoui, S.; Veuthey, J.L.; Christen, P. Calystegine distribution in some solanaceous species. Phytochemistry 2001, 58, 455–462. [Google Scholar] [CrossRef]
- Hanuš, L.O.; Dembitsky, V.M.; Moussaieff, A. Comparative study of volatile compounds in the fresh fruits of Mandragora autumnalis. Acta Chromatogr. 2006, 17, 151–160. [Google Scholar]
- Suleiman, R.K.; Zarga, M.A.; Sabri, S.S. New withanolides from Mandragora officinarum: First report of withanolides from the Genus Mandragora. Fitoterapia 2010, 81, 864–868. [Google Scholar] [CrossRef]
- Obeidat, M. Antimicrobial activity of some medicinal plants against multidrug resistant skin pathogens. J. Med. Plant Res. 2011, 5, 3856–3860. [Google Scholar]
- Kuete, V. Medicinal Plant Research in Africa: Pharmacology and Chemistry; Elsevier: London, UK, 2013. [Google Scholar]
- Rankou, H.; Ouhammou, A.; Taleb, M.; Martin, G. Mandragora autumnalis. The IUCN Red List of Threatened Species 2015. Available online: https://dx.doi.org/10.2305/IUCN.UK.2015-4.RLTS.T53785790A53798742.en (accessed on 31 August 2020).
- Al-Quran, S. Conservation of medicinal plants in Ajlun woodland/Jordan. J. Med. Plants Res. 2011, 5, 5857–5862. [Google Scholar]
- Paudel, M.; Pradhan, S.; Pant, B. In vitro seed germination and seedling development of Esmeralda clarkei Rchb.f. (Orchidaceae). Plant Tissue Cult. Biotechnol. 2012, 22, 107–111. [Google Scholar] [CrossRef]
- Utami, E.S.W.; Hariyanto, S. In vitro seed germination and seedling development of a rare Indonesian native orchid Phalaenopsis amboinensis JJ Sm. Scientifica 2019, 2019, 8105138. [Google Scholar] [CrossRef] [PubMed]
- Coelho, N.; Gonçalves, S.; Romano, A. Endemic plant species conservation: Biotechnological approaches. Plants 2020, 9, 345. [Google Scholar] [CrossRef]
- Verma, P.; Majee, M. Seed germination and viability test in tetrazolium (TZ) assay. Bio-Protocol 2013, 3, e884. [Google Scholar] [CrossRef]
- Pribil, M.; Sandoval-Ibáñez, O.; Xu, W.; Sharma, A.; Labs, M.; Liu, Q.; Galgenmüller, C.; Schneider, T.; Wessels, M.; Matsubara, S.; et al. Fine-tuning of photosynthesis requires CURVATURE THYLAKOID1- mediated thylakoid plasticity. Plant Physiol. 2018, 176, 2351–2364. [Google Scholar] [CrossRef]
- Tezuka, T.; Yokoyama, H.; Tanaka, H.; Shiozaki, S.; Oda, M. Seed and embryo germination in Ardisia crenata. J. Bot. 2012, 2012, 679765. [Google Scholar]
- Weitbrecht, K.; Muller, K.; Leubner-Metzger, G. First off the mark: Early seed germination. J. Exp. Bot. 2011, 62, 3289–3309. [Google Scholar] [CrossRef]
- Nonogaki, H. Seed dormancy and germination-emerging mechanisms and new hypotheses. Front. Plant. Sci. 2014, 5, 233. [Google Scholar] [CrossRef]
- Kucera, B.; Cohn, M.A.; Leubner-Metzger, G. Plant hormone interactions during seed dormancy release and germination. Seed Sci. Res. 2005, 15, 281–307. [Google Scholar] [CrossRef]
- Leubner-Metzger, G. Brassinosteroids and gibberellins promote tobacco seed germination by distinct pathways. Planta 2001, 213, 758–763. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.-N.; Ryu, J.-Y.; Jeong, Y.-M.; Park, J.; Song, J.-J.; Amasino, R.M.; Noh, B.; Noh, Y.-S. Control of seed germination by light-induced histone arginine demethylation activity. Dev. Cell. 2012, 22, 736–748. [Google Scholar] [CrossRef] [PubMed]
- Muralidhara, B.M.; Reddy, Y.T.N.; Srilatha, V.; Akshitha, H.J. Effect of seed coat removal treatments on seed germination and seedling attributes in mango varieties. Int. J. Fruit Sci. 2016, 16, 1–9. [Google Scholar] [CrossRef]
- Kim, D.H. Practical methods for rapid seed germination from seed coat-imposed dormancy of Prunus yedoensis. Sci. Hortic. 2019, 243, 451–456. [Google Scholar] [CrossRef]
- Finch-Savage, W.E.; Leubner-Metzger, G. Seed dormancy and the control of germination. New Phytol. 2006, 171, 501–523. [Google Scholar] [CrossRef]
- Ravindran, P.; Kumar, P.P. Regulation of seed germination: The involvement of multiple forces exerted via gibberellic acid signaling. Mol. Plant. 2019, 12, 24–26. [Google Scholar] [CrossRef]
- Yan, A.; Chen, Z. The control of seed dormancy and germination by temperature, light and nitrate. Bot. Rev. 2020, 86, 39–75. [Google Scholar] [CrossRef]
- Rawat, B.S.; Khanduri, V.P.; Sharma, C.M. Beneficial effects of cold-moist stratification on seed germination behaviors of Abies pindrow and Picea smithiana. J. For. Res. 2008, 19, 125. [Google Scholar] [CrossRef]
- Horimoto, T.; Koshioka, M.; Kubota, S.; Mander, L.N.; Hirai, N.; Ishida, N.; Suh, J.K.; Lee, A.K.; Roh, M.S. Effect of warm and cold stratification on H-NMR profiles, endogenous gibberellins and abscisic acid in Styrax japonicus seeds. Hortic. Environ. Biotechnol. 2011, 52, 233. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, A.K.; Suh, J.K. Effect of warm and cold stratification and ethanol treatment on germination of Corylopsis seeds. Hort. Sci. 2016, 43, 84–91. [Google Scholar]
- Footitt, S.; Finch-Savage, W.E. Dormancy and control of seed germination. In Plant Physiology and Function, the Plant Sciences Vol. 6; Clemens, S., Ed.; Springer: New York, NY, USA, 2017; pp. 1–30. [Google Scholar]
- Baskin, J.M.; Baskin, C.C. A classification system for seed dormancy. Seed Sci. Res. 2004, 14, 1–16. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. Seeds: Ecology, Biogeography, and Evolution of Dormancy and Germination; Academic Press: San Diego, CA, USA, 2014. [Google Scholar]
- Baskin, J.M.; Carol, C.; Baskin, C.C.; Xiaojie, L.I. Taxonomy, anatomy and evolution of physical dormancy in seeds. Plant Spec. Biol. 2000, 15, 139–152. [Google Scholar] [CrossRef]
- Carrera-Castaño, G.; Calleja-Cabrera, J.; Pernas, M.; Gómez, L.; Oñate-Sánchez, L. An updated overview on the regulation of seed germination. Plants 2020, 9, 703. [Google Scholar] [CrossRef] [PubMed]
- Baskin, C.C. Breaking physical dormancy in seeds—Focussing on the lens. New. Phytol. 2003, 158, 227–238. [Google Scholar] [CrossRef]
- Kildisheva, O.A.; Dumroese, R.K.; Davis, A.S. Overcoming dormancy and enhancing germination of Sphaeralcea munroana seeds. HortScience 2011, 46, 1672–1676. [Google Scholar] [CrossRef]
- Orozco-Segovia, A.; Márquez-Guzmán, J.; Sánchez-Coronado, M.E.; de Buen, A.G.; Baskin, J.M.; Baskin, C.C. Seed anatomy and water uptake in relation to seed dormancy in Opuntiato mentosa (Cactaceae, Opuntioideae). Ann. Bot. 2007, 99, 581–592. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissues. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]





| Number of Leaves/Plant [Range] | Number of Flowers/Plant [Range] | Number of Immature Fruits/Plant [Range] | Number of Mature Seeds/Ripe Fruit [Range] |
|---|---|---|---|
| 14.4 ± 1.2 [3–35] (n = 40) | 13.5 ± 3.1 [0–46] (n = 19) | 2.3 ± 0.5 [0–19] (n = 58) | 44 ± 1.5 [18–61] (n = 46) |
| Intact Seed Length (mm) | Intact Seed Width (mm) | Intact Seed Weight (mg) | Decoated Seed Weight (mg) |
|---|---|---|---|
| 5.9 ± 0.1 (n = 21) | 4.4 ± 0.1 (n = 21) | 36.7 ± 0.9 (n = 50) | 19.1 ± 0.8 (n = 21) |
| Pre-Storage Temp. (°C) of Intact Seeds | Decoated Seeds on SGM + mg/L GA3 | Number of Cultured Seeds | Number of Developed Seedlings (%) |
|---|---|---|---|
| Cold (4–5) | MSO | 25 | 7 (28.0) |
| 30 | 49 | 27 (55.1) | |
| 60 | 63 | 42 (66.7) | |
| 100 | 67 | 43 (64.2) | |
| Soil controls of intact seeds | - | 22 | 10 (45.5) |
| Room temp. (22–25) | MSO | 15 | 3 (20.0) |
| 30 | 16 | 7 (43.8) | |
| 60 | 14 | 9 (64.3) | |
| 100 | 23 | 10 (43.5) | |
| Soil controls of intact seeds | - | 21 | 9 (42.9) |
| Origin of Plantlets | Number of True Leaves/5 Weeks-Old Plantlet | Number of True Leaves/10 Weeks-Old Plantlet | Taproot Length (cm)/10 Weeks-Old Plantlet | Total Fresh Weight/10 Weeks-Old Plantlet (g) |
|---|---|---|---|---|
| In vitro decoated seeds germinated in SGM cultures | 3.3 ± 0.2 a | 5.9 ± 0.7 a | 12.4 ± 0.5 a | 3.22 ± 0.23 a |
| In vivo intact seeds germinated in potting soil | 0.0 ± 0.0 b | 1.4 ± 0.2 b | 9.7 ± 1.2 b | 2.74 ± 0.20 a |
| Origin of Plants | Number of True Leaves/Plant | Taproot Length (cm)/Plant | Fresh Weight of Shoot/Plant (g) | Fresh Weight of Root/Plant (g) | Fresh Weight of The Whole Plant (g) |
|---|---|---|---|---|---|
| In vitro decoated seeds germinated in SGM cultures | 7.1 ± 0.7 a (n = 12) | 18.8 ± 1.3 a (n = 11) | 8.84 ± 1.57 a (n = 11) | 11.65 ± 1.98 a (n = 11) | 20.49 ± 3.50 a (n = 11) |
| In vivo intact seeds germinated in potting soil | 2.9 ± 0.4 b (n = 14) | 15.4 ± 1.8 a (n = 9) | 3.72 ± 0.79 b (n = 9) | 6.07 ± 1.11 b (n = 9) | 9.79 ± 1.82 b (n = 9) |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Ahmad, H. In Vitro Decoated Seed Germination and Seedling Development for Propagation of Wild Mandrake (Mandragora autumnalis Bertol.). Plants 2020, 9, 1339. https://doi.org/10.3390/plants9101339
Al-Ahmad H. In Vitro Decoated Seed Germination and Seedling Development for Propagation of Wild Mandrake (Mandragora autumnalis Bertol.). Plants. 2020; 9(10):1339. https://doi.org/10.3390/plants9101339
Chicago/Turabian StyleAl-Ahmad, Hani. 2020. "In Vitro Decoated Seed Germination and Seedling Development for Propagation of Wild Mandrake (Mandragora autumnalis Bertol.)" Plants 9, no. 10: 1339. https://doi.org/10.3390/plants9101339
APA StyleAl-Ahmad, H. (2020). In Vitro Decoated Seed Germination and Seedling Development for Propagation of Wild Mandrake (Mandragora autumnalis Bertol.). Plants, 9(10), 1339. https://doi.org/10.3390/plants9101339





