Bioactivity of Common Pesticidal Plants on Fall Armyworm Larvae (Spodoptera frugiperda)
Abstract
1. Introduction
2. Results and Discussion
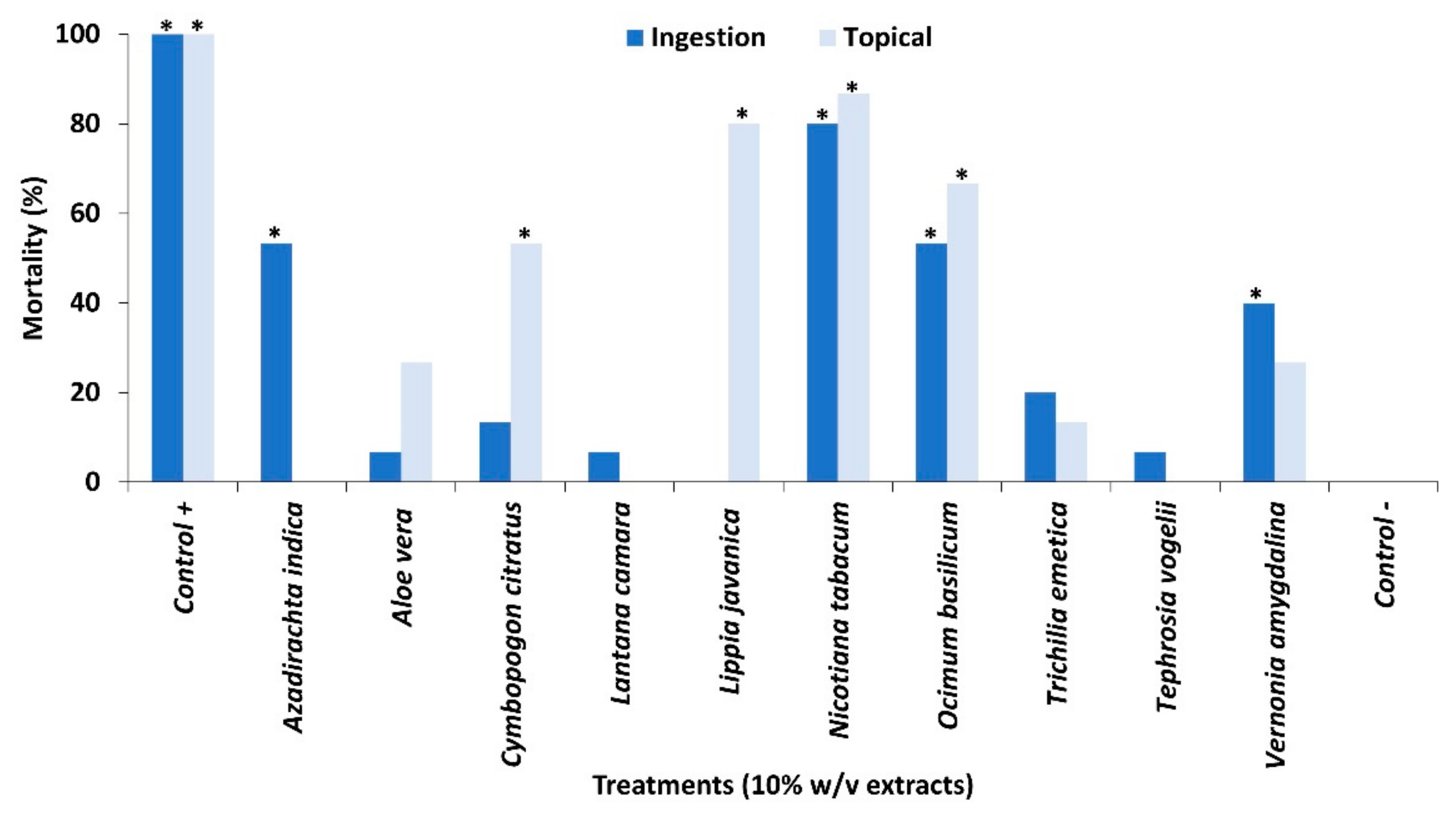
2.1. Contact Toxicity and Feeding Bioassays with Ten Pesticidal Plant Species
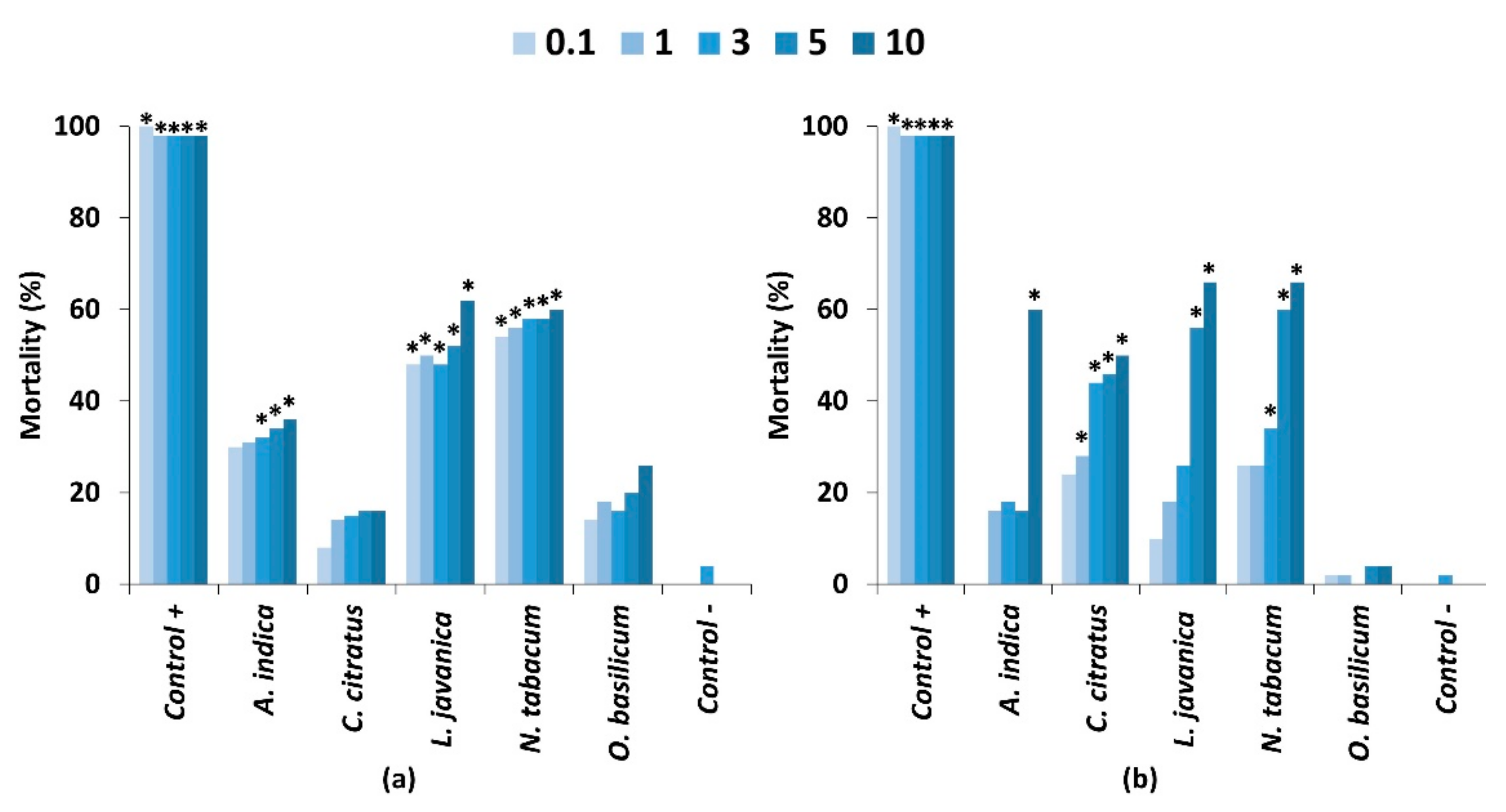
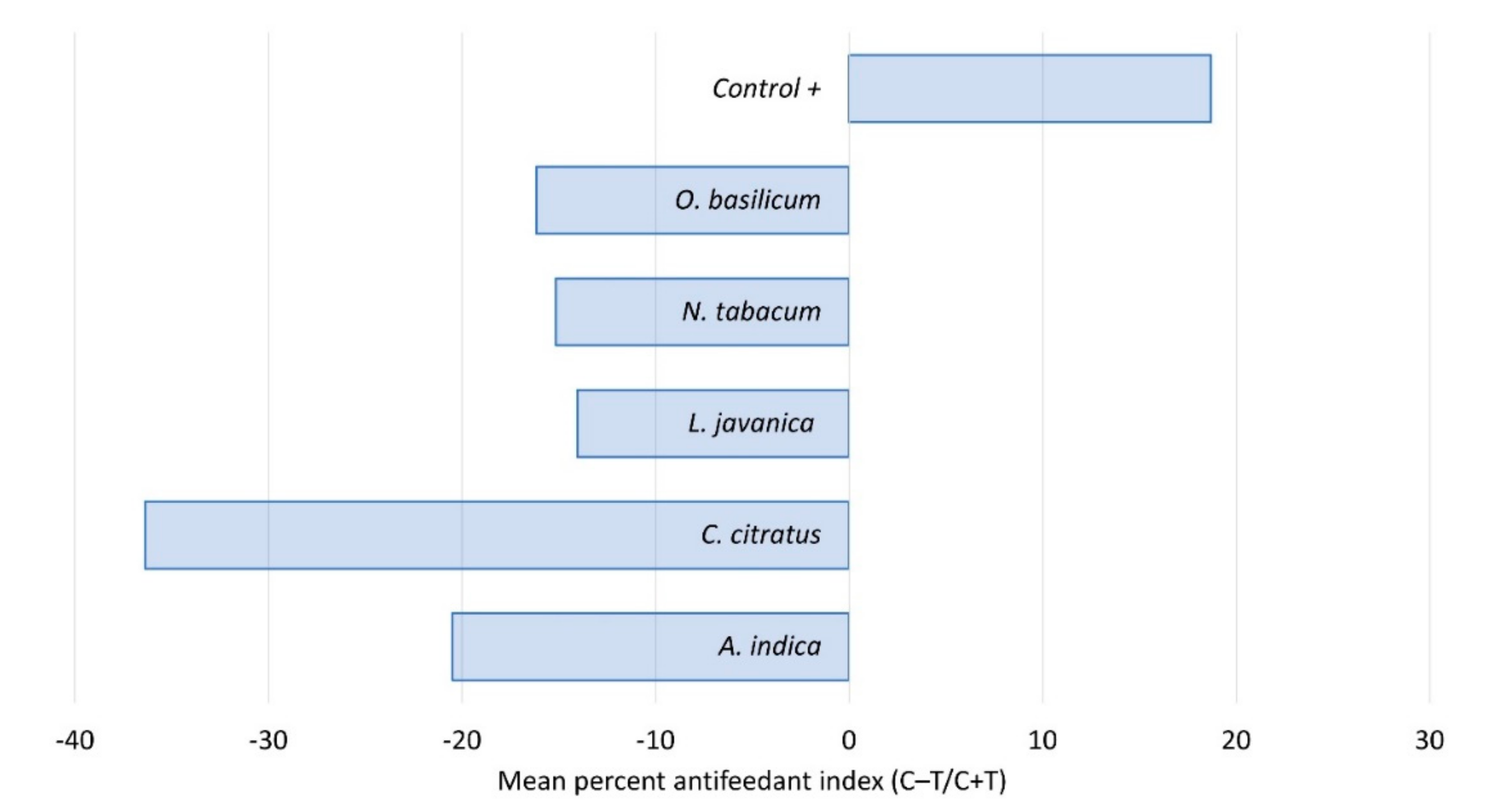
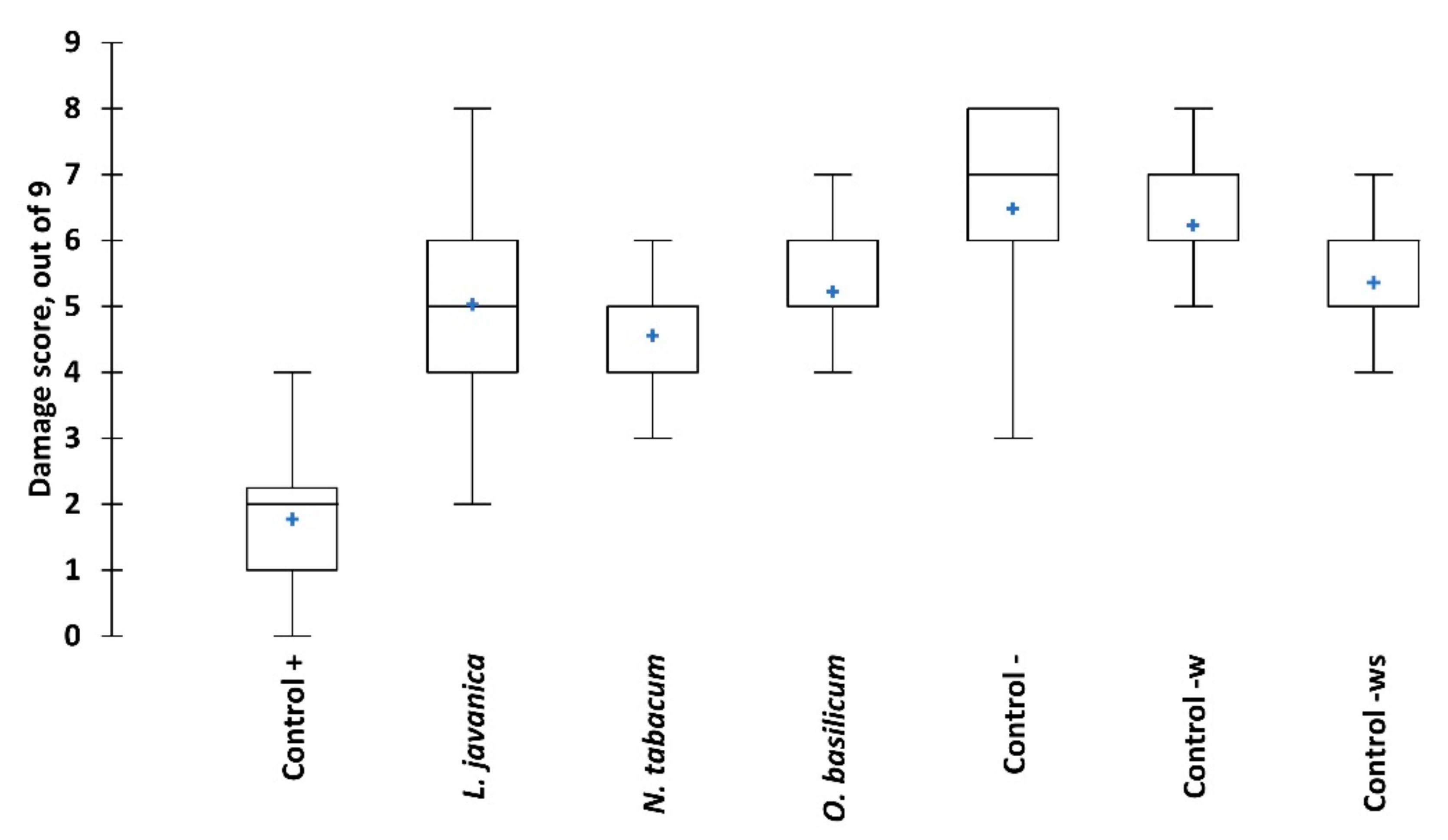
2.2. Contact Toxicity and Feeding Bioassays with Five Pesticidal Plant Species
3. Materials and Methods
3.1. Fall Armyworm Rearing
3.2. Plant Material Collection and Extract Preparation
3.3. Bioassay Methods
3.4. Data Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sisay, B.; Tefera, T.; Wakgari, M.; Ayalew, G.; Mendesil, E. The Efficacy of Selected Synthetic Insecticides and Botanicals against Fall Armyworm, Spodoptera frugiperda, in Maize. Insects 2019, 10, 45. [Google Scholar] [CrossRef]
- Goergen, G.; Kumar, P.L.; Sankung, S.B.; Togola, A.; Tamò, M. First Report of Outbreaks of the Fall Armyworm Spodoptera frugiperda (J E Smith) (Lepidoptera, Noctuidae), a New Alien Invasive Pest in West and Central Africa. PLoS ONE 2016, 11, e0165632. [Google Scholar] [CrossRef] [PubMed]
- Rwomushana, I.; Bateman, M.; Beale, T.; Beseh, P.; Cameron, K.; Chiluba, M.; Clottey, V.; Davis, T.; Day, R.; Early, R.; et al. Fall Armyworm: Impacts and Implications for Africa; Evidence Note Update, October 2018; Centre for Agriculture and Bioscience International: Wallingford, UK, 2018. [Google Scholar]
- Sisay, B.; Simiyu, J.; Mendesil, E.; Likhayo, P.; Ayalew, G.; Mohamed, S.; Subramanian, S.; Tefera, T. Fall Armyworm, Spodoptera frugiperda Infestations in East Africa: Assessment of Damage and Parasitism. Insects 2019, 10, 195. [Google Scholar] [CrossRef] [PubMed]
- Day, R.; Abrahams, P.; Bateman, M.; Beale, T.; Clottey, V.; Cock, M.; Colmenarez, Y.; Corniani, N.; Early, R.; Godwin, J.; et al. Fall armyworm: Impacts and implications for Africa. Outlooks Pest Manag. 2017, 28, 196–201. [Google Scholar] [CrossRef]
- Yu, S.J. Insecticide resistance in the fall armyworm, Spodoptera frugiperda (Smith, J.E.). Pestic. Biochem. Physiol. 1991, 39, 84–91. [Google Scholar] [CrossRef]
- Belmain, S.R.; Stevenson, P.C. Ethnobotanicals in Ghana: Revising and modernising age-old farmer practice. Pestic. Outlook 2001, 6, 233–238. [Google Scholar]
- Nyirenda, S.P.; Sileshi, G.W.; Belmain, S.R.; Kamanula, J.F.; Mvumi, M.; Sola, P.; Nyirenda, G.K.C.; Stevenson, P.C. Farmers’ ethno-ecological knowledge of vegetable pests and pesticidal plant use in northern Malawi and eastern Zambia. Afr. J. Agric. Res. 2011, 6, 1525–1537. [Google Scholar]
- Kamanula, J.; Sileshi, G.W.; Belmain, S.R.; Sola, P.; Mvumi, B.M.; Nyirenda, G.K.C.; Nyirenda, S.P.; Stevenson, P.C. Farmers’ insect pest management practices and pesticidal plant use in the protection of stored maize and beans in Southern Africa. Int. J. Pest Manag. 2011, 57, 41–49. [Google Scholar] [CrossRef]
- Isman, M.B. Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu. Rev. Entomol. 2006, 51, 45–66. [Google Scholar] [CrossRef]
- Dougoud, J.; Toepfer, S.; Bateman, M.; Jenner, W.H. Efficacy of homemade botanical insecticides based on traditional knowledge. A review. Agron. Sustain. Dev. 2019, 39, 37. [Google Scholar] [CrossRef]
- Isman, M.B. Botanical insecticides: For richer, for poorer. Pest Manag. Sci. 2008, 64, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Mkindi, A.; Mpumi, N.; Tembo, Y.; Stevenson, P.C.; Ndakidemi, P.A.; Mtei, K.; Machunda, R.; Belmain, S.R. Invasive weeds with pesticidal properties as potential new crops. Ind. Crops Prod. 2017, 110, 113–122. [Google Scholar] [CrossRef]
- Tembo, Y.; Mkindi, A.G.; Mkenda, P.A.; Mpumi, N.; Mwanauta, R.; Stevenson, P.C.; Ndakidemi, P.A.; Belmain, S.R. Pesticidal Plant Extracts Improve Yield and Reduce Insect Pests on Legume Crops without Harming Beneficial Arthropods. Front. Plant Sci. 2018, 9, 1425. [Google Scholar] [CrossRef] [PubMed]
- Mkenda, P.; Mwanauta, R.; Stevenson, P.C.; Ndakidemi, P.; Mtei, K.; Belmain, S.R. Extracts from field margin weeds provide economically viable and environmentally benign pest control compared to synthetic pesticides. PLoS ONE 2015, 10, e0143530. [Google Scholar] [CrossRef]
- Belmain, S.R.; Amoah, B.A.; Nyirenda, S.P.; Kamanula, J.F.; Stevenson, P.C. Highly Variable Insect Control Efficacy of Tephrosia vogelii Chemotypes. J. Agric. Food Chem. 2012, 60, 10055–10063. [Google Scholar] [CrossRef]
- Stevenson, P.C.; Kite, G.C.; Lewis, G.P.; Forest, F.; Nyirenda, S.P.; Belmain, S.R.; Sileshi, G.W.; Veitch, N.C. Distinct chemotypes of Tephrosia vogelii and implications for their use in pest control and soil enrichment. Phytochemistry 2012, 78, 135–146. [Google Scholar] [CrossRef]
- Green, P.W.C.; Belmain, S.R.; Ndakidemi, P.A.; Farrell, I.W.; Stevenson, P.C. Insecticidal activity of Tithonia diversifolia and Vernonia amygdalina. Ind. Crops Prod. 2017, 110, 15–21. [Google Scholar] [CrossRef]
- Mkenda, P.P.A.; Stevenson, P.C.P.; Ndakidemi, P.; Farman, D.I.; Belmain, S.R. Contact and fumigant toxicity of five pesticidal plants against Callosobruchus maculatus (Coleoptera: Chrysomelidae) in stored cowpea (Vigna unguiculata). Int. J. Trop. Insect Sci. 2015, 35, 1–13. [Google Scholar] [CrossRef]
- Munyemana, F.; Alberto, A.L. Evaluation of larvicidal activity of selected plant extracts against Plutella xylostella (Lepidoptera: Plutellidae) larvae on cabbage. Adv. Med. Plant Res. 2017, 5, 11–20. [Google Scholar] [CrossRef]
- Pavela, R.; Benelli, G. Ethnobotanical knowledge on botanical repellents employed in the African region against mosquito vectors—A review. Exp. Parasitol. 2016, 167, 103–108. [Google Scholar] [CrossRef]
- Trdan, S.; Cirar, A.; Bergant, K.; Andjus, L.; Kač, M.; Vidrih, M.; Rozman, L. Effect of temperature on efficacy of three natural substances to Colorado potato beetle, Leptinotarsa decemlineata (Coleoptera: Chrysomelidae). Acta Agric. Scand. Sect. B Soil Plant Sci. 2007, 57, 293–296. [Google Scholar] [CrossRef]
- Stevenson, P.C.; Belmain, S.R. Pesticidal plants in African agriculture: Local uses and global perspectives. Outlooks Pest Manag. 2016, 27, 226–230. [Google Scholar] [CrossRef]
- Hernández-Lambraño, R.; Caballero-Gallardo, K.; Olivero-Verbel, J. Toxicity and antifeedant activity of essential oils from three aromatic plants grown in Colombia against Euprosterna elaeasa and Acharia fusca (Lepidoptera: Limacodidae). Asian Pac. J. Trop. Biomed. 2014, 4, 695–700. [Google Scholar] [CrossRef]
- Martinez, S.S.; van Emden, H.F. Sublethal concentrations of azadirachtin affect food intake, conversion efficiency and feeding behaviour of Spodoptera littoralis (Lepidoptera: Noctuidae). Bull. Entomol. Res. 1999, 89, 65–71. [Google Scholar] [CrossRef]
- Liang, G.-M.; Chen, W.; Liu, T.-X. Effects of three neem-based insecticides on diamondback moth (Lepidoptera: Plutellidae). Crop Prot. 2003, 22, 333–340. [Google Scholar] [CrossRef]
- Roel, A.R.; Dourado, D.M.; Matias, R.; Porto, K.R.A.; Bednaski, A.V.; Costa, R.B. da The effect of sub-lethal doses of Azadirachta indica (Meliaceae) oil on the midgut of Spodoptera frugiperda (Lepidoptera, Noctuidae). Rev. Bras. Entomol. 2010, 54, 505–510. [Google Scholar] [CrossRef]
- Amoabeng, B.W.; Gurr, G.M.; Gitau, C.W.; Nicol, H.I.; Munyakazi, L.; Stevenson, P.C. Tri-trophic insecticidal effects of African plants against cabbage pests. PLoS ONE 2013, 8, e78651. [Google Scholar] [CrossRef]
- Amoabeng, B.W.; Gurr, G.M.; Gitau, C.W.; Stevenson, P.C. Cost:benefit analysis of botanical insecticide use in cabbage: Implications for smallholder farmers in developing countries. Crop Prot. 2014, 57, 71–76. [Google Scholar] [CrossRef]
- Okrikata, E.; Bukar, M.; Ali, B. Economic Viability of Chilli Pepper and Neem Seed Kernel Powdered Formulations Vis-à-vis Sevin Dust (85%) in the Management of Lepidopterous Stemborers on Sorghum in North Eastern Nigeria. J. Biol. Agric. Healthc. 2016, 6, 99–103. [Google Scholar]
- Prasanna, B.M.; Huesing, J.E.; Eddy, R.; Peschke, V.M. Fall Armyworm in Africa: A guide for Integrated Pest Management; International Maize and Wheat Improvement Center: Mexico City, Mexico, 2018. [Google Scholar]
- Miranda, M.A.F.M.; Varela, R.M.; Torres, A.; Molinillo, J.M.G.; Gualtieri, S.C.J.; Macías, F.A. Phytotoxins from Tithonia diversifolia. J. Nat. Prod. 2015, 78, 1083–1092. [Google Scholar] [CrossRef]
- Rabe, T.; Mullholland, D.; van Staden, J. Isolation and identification of antibacterial compounds from Vernonia colorata leaves. J. Ethnopharmacol. 2002, 80, 91–94. [Google Scholar] [CrossRef]
- Koul, O.; Wahab, S. (Eds.) Neem: Today and in the New Millennium; Springer: Dordrecht, The Netherlands, 2004; ISBN 978-1-4020-1229-7. [Google Scholar]
- Singh, P.; Jayaramaiah, R.H.; Sarate, P.; Thulasiram, H.V.; Kulkarni, M.J.; Giri, A.P. Insecticidal potential of defense metabolites from Ocimum kilimandscharicum against Helicoverpa armigera. PLoS ONE 2014, 9, e104377. [Google Scholar] [CrossRef] [PubMed]
- Adeniyi, S.A.; Orjiekwe, C.L.; Ehiagbonare, J.E.; Arimah, B.D. Preliminary phytochemical analysis and insecticidal activity of ethanolic extracts of four tropical plants (Vernonia amygdalina, Sida acuta, Ocimum gratissimum and Telfaria occidentalis) against beans weevil (Acanthscelides obtectus). Int. J. Phys. Sci. 2010, 5, 753–762. [Google Scholar]
- Kamanula, J.F.; Belmain, S.R.; Hall, D.R.; Farman, D.I.; Goyder, D.J.; Mvumi, B.M.; Masumbu, F.F.; Stevenson, P.C. Chemical variation and insecticidal activity of Lippia javanica (Burm. f.) Spreng essential oil against Sitophilus zeamais Motschulsky. Ind. Crops Prod. 2017, 110, 75–82. [Google Scholar] [CrossRef]
- Mkindi, A.G.; Tembo, Y.; Ndakidemi, P.A.; Belmain, S.R.; Stevenson, P.C. Phytochemical Analysis of Tephrosia vogelii across East Africa Reveals Three Chemotypes that Influence Its Use as a Pesticidal Plant. Plants 2019, 8, 597. [Google Scholar] [CrossRef]
- Dos Santos, A.C.V.; de Almeida, W.A.; Fernandes, C.C.; de Sousa, A.H. Extractos hidroalcohólicos de plantas propias de la Amazonía suroccidental como alternativa de control de la oruga militar tardía. Idesia 2016, 34, 63–67. [Google Scholar]
- Abbott, W.S. A Method of Computing the Effectiveness of an Insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Green, P.W.C.; Veitch, N.C.; Stevenson, P.C.; Simmonds, M.S.J. Cardenolides from Gomphocarpus sinaicus and Pergularia tomentosa (Apocynaceae: Asclepiadoideae) deter the feeding of Spodoptera littoralis. Arthropod. Plant. Interact. 2011, 5, 219–225. [Google Scholar] [CrossRef]
- Hummelbrunner, L.A.; Isman, M.B. Acute, sublethal, antifeedant, and synergistic effects of monoterpenoid essential oil compounds on the tobacco cutworm, Spodoptera litura (Lep., Noctuidae). J. Agric. Food Chem. 2001, 49, 715–720. [Google Scholar] [CrossRef]
- Silva, M.S.; Broglio, S.M.F.; Trindade, R.C.P.; Ferrreira, E.S.; Gomes, I.B.; Micheletti, L.B. Toxicity and application of neem in fall armyworm. Comun. Sci. 2015, 6, 359–364. [Google Scholar] [CrossRef]
- Williams, W.P.; Buckley, P.M.; Daves, C.A. Identifying resistance in corn to southwestern corn borer (lepidoptera: Crambidae), fall armyworm (Lepidoptera: Noctuidae), and corn earworm (lepidoptera; Noctuidae). J. Agric. Urban Entomol. 2006, 23, 87–95. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Phambala, K.; Tembo, Y.; Kasambala, T.; Kabambe, V.H.; Stevenson, P.C.; Belmain, S.R. Bioactivity of Common Pesticidal Plants on Fall Armyworm Larvae (Spodoptera frugiperda). Plants 2020, 9, 112. https://doi.org/10.3390/plants9010112
Phambala K, Tembo Y, Kasambala T, Kabambe VH, Stevenson PC, Belmain SR. Bioactivity of Common Pesticidal Plants on Fall Armyworm Larvae (Spodoptera frugiperda). Plants. 2020; 9(1):112. https://doi.org/10.3390/plants9010112
Chicago/Turabian StylePhambala, Kelita, Yolice Tembo, Trust Kasambala, Vernon H. Kabambe, Philip C. Stevenson, and Steven R. Belmain. 2020. "Bioactivity of Common Pesticidal Plants on Fall Armyworm Larvae (Spodoptera frugiperda)" Plants 9, no. 1: 112. https://doi.org/10.3390/plants9010112
APA StylePhambala, K., Tembo, Y., Kasambala, T., Kabambe, V. H., Stevenson, P. C., & Belmain, S. R. (2020). Bioactivity of Common Pesticidal Plants on Fall Armyworm Larvae (Spodoptera frugiperda). Plants, 9(1), 112. https://doi.org/10.3390/plants9010112






