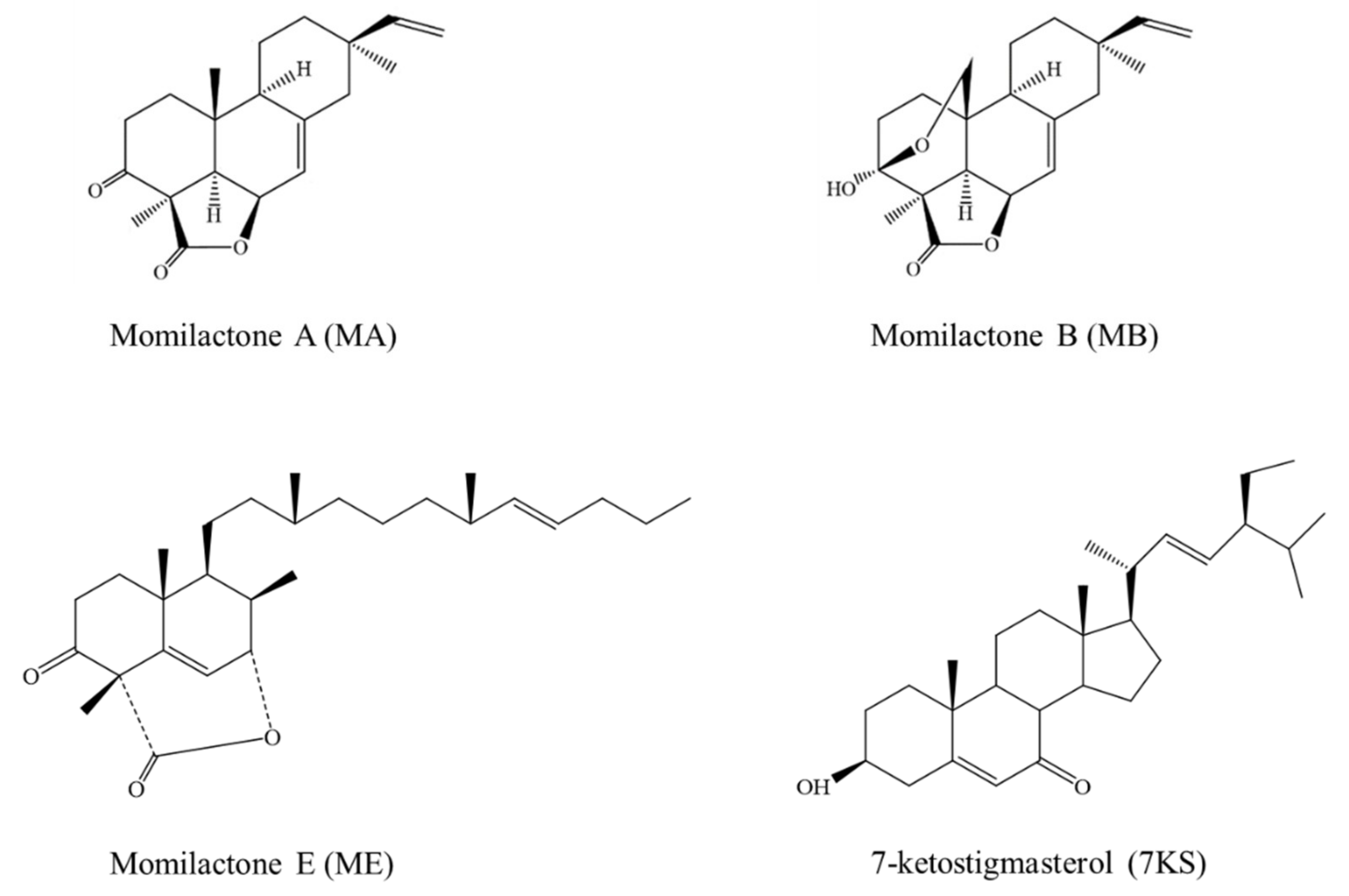
Inhibitory Activities of Momilactones A, B, E, and 7-Ketostigmasterol Isolated from Rice Husk on Paddy and Invasive Weeds
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Materials
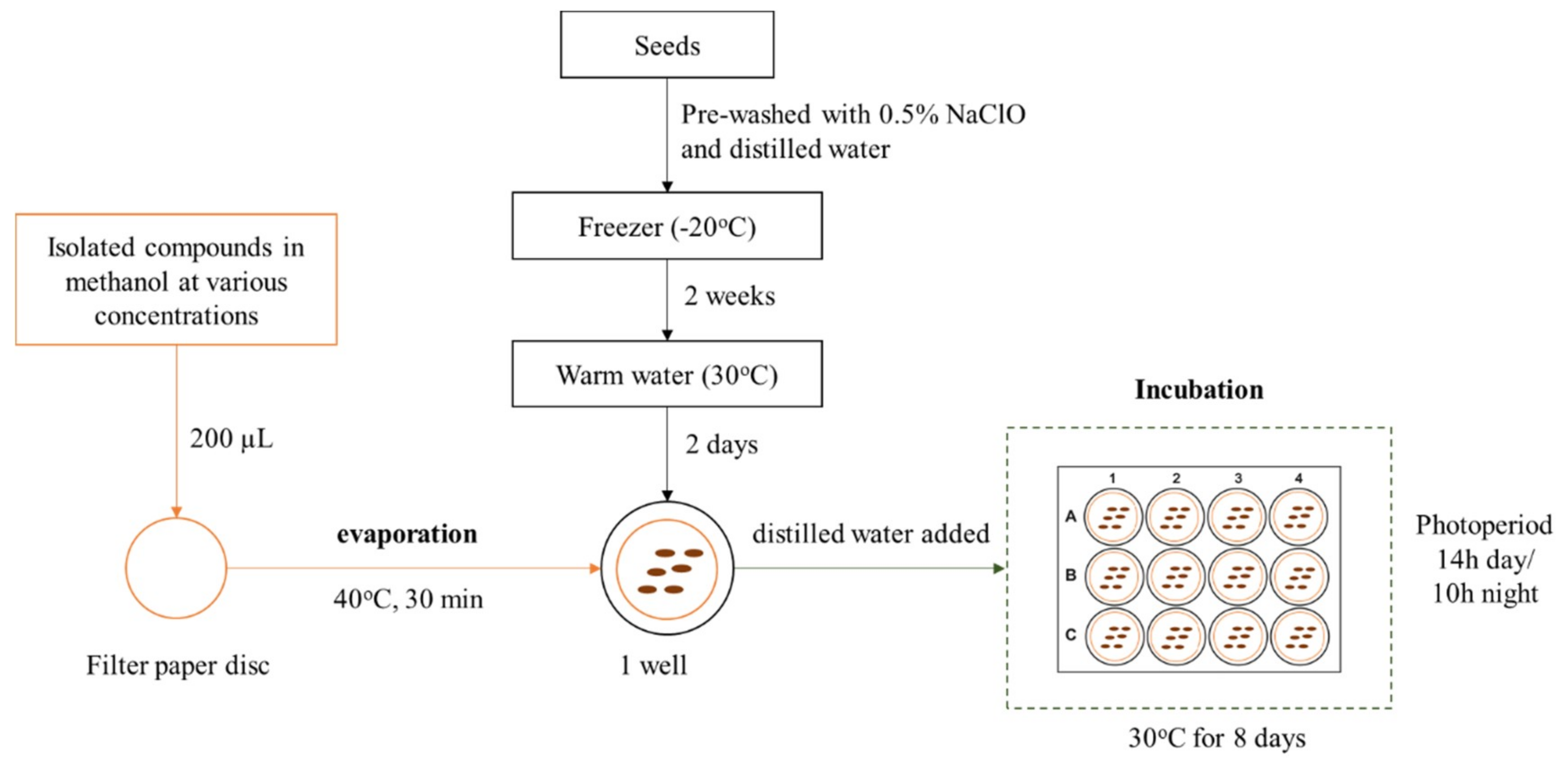
4.2. Extraction and Isolation of Active Compounds from Rice Husk
4.3. Allelopathic Bioassays
4.4. Quantification of Momilactones A and B by HPLC
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rice, E.L. Allelopathy—An Overview. In Chemically Mediated Interactions between Plants and Other Organisms. Recent Advances in Phytochemistry; Cooper-Driver, G.A., Swain, T., Conn, E.E., Eds.; Springer: Boston, MA, USA, 1985; Volume 19, pp. 81–99. [Google Scholar] [CrossRef]
- Xuan, T.D.; Elzaawely, A.A.; Deba, F.; Fukuta, M.; Tawata, S. Mimosine in Leucaena as a potent bio-herbicide. Agron. Sustain. Dev. 2006, 26, 89–97. [Google Scholar] [CrossRef]
- Khanh, T.D.; Xuan, T.D.; Chung, I.M. Rice allelopathy and the possibility for weed management. Ann. Appl. Biol. 2007, 151, 325–339. [Google Scholar] [CrossRef]
- Xuan, T.D.; Tawata, S.; Khanh, T.D. Herbicidal activity of mimosine and its derivatives. In Herbicides—Advances in Research; Price, A., Kelton, J., Eds.; IntechOpen: Rijeka, Croatia, 2013; pp. 299–312. [Google Scholar] [CrossRef]
- Xuan, T.D.; Tsuzuki, E.; Tawata, S.; Khanh, T.D. Method to determine allelopathic potential of crop plants for weed control. Allelopathy J. 2004, 13, 149–164. [Google Scholar]
- Chung, I.M.; Ali, M.; Hahn, S.J.; Siddiqui, N.A.; Lim, Y.H.; Ahmad, A. Chemical constituents from the hulls of Oryza sativa with cytotoxic activity. Chem. Nat. Compd. 2005, 41, 182–189. [Google Scholar] [CrossRef]
- Esa, N.M.; Ling, T.B.; Peng, L.S. By-products of rice processing: An overview of health benefits and applications. J. Rice Res. 2013, 1, 107. [Google Scholar] [CrossRef]
- Khang, D.T.; Anh, L.H.; Ha, P.T.T.; Tuyen, P.T.; Quan, N.V.; Minh, L.T.; Quan, N.T.; Minh, T.N.; Xuan, T.D.; Khanh, T.D.; Trung, K.H. Allelopathic activity of dehulled rice and its allelochemicals on weed germination. Int. Lett. Nat. Sci. 2016, 58, 1–10. [Google Scholar] [CrossRef]
- Kato, T.; Kabuto, C.; Sasaki, N.; Tsunagawa, M.; Aizawa, H.; Fujita, K.; Kato, Y.; Kitahara, Y.; Takahashi, N. Momilactones, growth inhibitors from rice, Oryza sativa L. Tetrahedron Lett. 1973, 14, 3861–3864. [Google Scholar] [CrossRef]
- Berendji, S.; Asghari, J.B.; Matin, A.A. Allelopathic potential of rice (Oryza sativa) varieties on seedling growth of barnyardgrass (Echinochloa crus-galli). J. Plant Interact. 2008, 3, 175–180. [Google Scholar] [CrossRef]
- Minh, T.N.; Xuan, T.D.; Ahmad, A.; Elzaawely, A.A.; Teschke, R.; Van, T.M. Efficacy from different extractions for chemical profile and biological activities of rice husk. Sustainability 2018, 10, 1356. [Google Scholar] [CrossRef]
- Minh, T.N.; Xuan, T.D.; Ahmad, A.; Elzaawely, A.A.; Teschke, R.; Van, T.M. Momilactones A and B: Optimization of yields from isolation and purification. Separations 2018, 5, 28. [Google Scholar] [CrossRef]
- Quan, N.V.; Hoang-Dung, T.; Xuan, T.D.; Ahmad, A.; Dat, T.D.; Khanh, T.D.; Teschke, R. Momilactones A and B are α-amylase and α-glucosidase inhibitors. Molecules 2019, 24, 482. [Google Scholar] [CrossRef] [PubMed]
- Quan, N.V.; Xuan, T.D.; Hoang-Dung, T.; Ahmad, A.; Khanh, T.D.; Dat, T.D. Contribution of momilactones A and B to diabetes inhibitory potential of rice bran: Evidence from in vitro assays. Saudi Pharm. J. 2019, in press. [Google Scholar] [CrossRef]
- Tsunakawa, M.; Ohba, A.; Sasaki, N.; Kabuto, C. Momilactone C, a minor constituent of growth inhibitors in rice husk. Chem. Lett. 1976, 1157–1158. [Google Scholar] [CrossRef]
- Cho, J.; Cha, B.; Min Lee, S.; Shrestha, S.; Jeong, R.; Sung Lee, D.; Kim, Y.; Lee, D.; Kang, H.; Kim, J.; Baek, N. Diterpenes from the roots of Oryza sativa L. and their inhibition activity on NO production in LPS-stimulated RAW264.7 macrophages. Chem. Biodivers. 2015, 12, 1356–1364. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Xuan, T.D.; Minh, T.N.; Siddiqui, N.A.; Quan, N.V. Comparative extraction and simple isolation improvement techniques of active constituents’ momilactone A and B from rice husks of Oryza sativa by HPLC analysis and column chromatography. Saudi Pharm. J. 2019, 27, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Alemany, L.; Laparra, J.M.; Barberá, R.; Alegría, A. Evaluation of the cytotoxic effect of 7keto-stigmasterol and 7keto-cholesterol in human intestinal (Caco-2) cells. Food Chem. Toxicol. 2012, 50, 3106–3113. [Google Scholar] [CrossRef] [PubMed]
- Laparra, J.M.; Alfonso-García, A.; Alegría, A.; Barberá, R.; Cilla, A. 7keto-stigmasterol and 7keto-cholesterol induce differential proteome changes to intestinal epitelial (Caco-2) cells. Food Chem. Toxicol. 2015, 84, 29–36. [Google Scholar] [CrossRef]
- Marinho, R.d.S.S.; Ramos, C.J.B.; Leite, J.P.G.; Teixeira, V.L.; Paixão, I.C.N.d.P.; Belo, C.A.D.; Pereira, A.B.; Pinto, A.M.V. Antiviral activity of 7-keto-stigmasterol obtained from green Antarctic algae Prasiola crispa against equine herpesvirus 1. J. Appl. Phycol. 2017, 29, 555–562. [Google Scholar] [CrossRef]
- Khanh, T.D.; Cong, L.C.; Chung, I.M.; Xuan, T.D.; Tawata, S. Variation of weed-suppressing potential of Vietnamese rice cultivars against barnyardgrass (Echinochloa crus-galli) in laboratory, greenhouse and field screenings. J. Plant Interact. 2009, 4, 209–218. [Google Scholar] [CrossRef]
- Fujii, Y. Toxic chemicals from invasive alien plants. In Plant Toxins. Toxinology; Gopalakrishnakone, P., Carlini, C., Ligabue-Braun, R., Eds.; Springer: Dordrecht, The Netherlands, 2017; pp. 25–36. [Google Scholar] [CrossRef]
- Kato, T.; Tsunakawa, M.; Sasaki, N.; Aizawa, H.; Fujita, K.; Kitahara, Y.; Takahashi, N. Growth and germination inhibitors in rice husks. Phytochemistry 1977, 16, 45–48. [Google Scholar] [CrossRef]
- Cartwright, D.W.; Langcake, P.; Pryce, R.J.; Leworthy, D.P.; Ride, J.P. Isolation and characterization of two phytoalexins from rice as momilactones A and B. Phytochemistry 1981, 20, 535–537. [Google Scholar] [CrossRef]
- Fukuta, M.; Xuan, T.D.; Deba, F.; Tawata, S.; Khanh, T.D.; Chung, I.M. Comparative efficacies in vitro of antibacterial, fungicidal, antioxidant, and herbicidal activities of momilactones A and B. J. Plant Interact. 2007, 2, 245–251. [Google Scholar] [CrossRef]
- Xuan, T.D.; Shinkichi, T.; Khanh, T.D.; Chung, I.M. Biological control of weeds and plant pathogens in paddy rice by exploiting plant allelopathy: An overview. Crop Prot. 2005, 24, 197–206. [Google Scholar] [CrossRef]
- War, A.R.; Paulraj, M.G.; Ahmad, T.; Buhroo, A.A.; Hussain, B.; Ignacimuthu, S.; Sharma, H.C. Mechanisms of plant defense against insect herbivores. Plant Signal Behav. 2012, 7, 1306–1320. [Google Scholar] [CrossRef] [PubMed]
- Khanh, T.D.; Xuan, T.D.; Chung, I.M.; Tawata, S. Allelochemicals of barnyardgrass-infested soil and their activities on crops and weeds. Weed Biol. Manag. 2008, 8, 267–275. [Google Scholar] [CrossRef]
- Fischer, N.H.; Quijano, L. Allelopathic agents from common weeds. In The Chemistry of Allelopathy; Thompson, A.C., Ed.; ACS Symposium Series 268–American Chemical Society: Washington, DC, USA, 1985; pp. 133–147. [Google Scholar] [CrossRef]
- Walck, J.L.; Baskin, J.M.; Baskin, C.C. A comparative study of the seed germination biology of a narrow endemic and two geographically-widespread species of Solidago (Asteraceae). 1. Germination phenology and effect of cold stratification on germination. Seed Sci. Res. 1997, 7, 47–58. [Google Scholar] [CrossRef]
- Meyer, A.H.; Schmid, B. Seed dynamics and seedling establishment invading perennial Solidago altissima under different experimental treatments. J. Ecol. 1999, 87, 28–41. [Google Scholar] [CrossRef]
- Sakata, Y.; Kaneko, S.; Hayano, A.; Inoue-Murayama, M.; Ohgushi, T.; Isagi, Y. Isolation and characterization of microsatellite loci in the invasive herb Solidago altissima (Asteraceae). Appl. Plant Sci. 2013, 1, 1200313. [Google Scholar] [CrossRef]
- Heath, J.J.; Kessler, A.; Woebbe, E.; Cipollini, D.; Stireman, J.O. Exploring plant defense theory in tall goldenrod, Solidago altissima. New Phytol. 2014, 202, 1357–1370. [Google Scholar] [CrossRef]
| Sample | L. sativa | E. crus-galli | S. altissima |
|---|---|---|---|
| MA | nd | 229.67 ± 1.20 b | 119.80 ± 11.50 b |
| MB | 178.46 ± 0.03 b | 31.88 ± 0.18 a | 20.36 ± 0.91 a |
| MAB | 327.20 ± 27.50 a | 28.26 ± 3.05 a | 23.97 ± 1.89 a |
| ME | nd | nd | 1162.50 ± 14.10 d |
| 7KS | nd | nd | nd |
| ME-7KS | nd | nd | 358.30 ± 8.39 c |
| pHA | nd | nd | 1074.20 ± 66.60 d |
| Sample | L. sativa | E. crus-galli | ||
|---|---|---|---|---|
| RL | SL | RL | SL | |
| MA | 348.00 ± 11.30 b | 388.20 ± 10.70 b | 147.60 ± 0.37 b | 123.87 ± 1.78 b |
| MB | 6.49 ± 0.04 a | 58.64 ± 0.99 a | 4.00 ± 0.03 a | 4.46 ± 0.05 a |
| MAB | 5.63 ± 0.05 a | 63.77 ± 0.64 a | 5.13 ± 0.05 a | 4.47 ± 0.05 a |
| pHA | 820.57 ± 9.8 c | 1453.30 ± 48.10 c | 936.66 ± 4.49 c | 1464.6 ± 40.30 c |
| Sample | Concentration (µg/mL) | L. sativa | E. crus-galli | S. altissima | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| GR | RL | SL | GR | RL | SL | GR | ||||
| Methanol | (8/8) a 0.0 | (1.95) b 0.0 | (0.76) c 0.0 | (8/8) 0.0 | (4.53) 0.0 | (3.03) 0.0 | (13/20) 0.0 | |||
| MA | 400 | 0.0 | 52.38 | 50.67 | 78.57 | 95.96 | 96.70 | 86.75 | ||
| MB | 100.00 | 94.71 | 84.59 | 100.00 | 100.00 | 100.00 | 100.00 | |||
| MAB | 58.33 | 91.82 | 82.82 | 100.00 | 100.00 | 100.00 | 100.00 | |||
| ME | 1000 | 0.0 | +92.68 | 6.61 | 0.0 | +16.48 | +8.01 | 39.53 | ||
| 7KS | 0.0 | +60.93 | 13.65 | nd | nd | nd | nd | |||
| ME-7KS | 0.0 | +93.00 | 5.71 | 0.0 | 13.33 | 29.52 | 94.66 | |||
| pHA | 0.0 | 63.14 | 37.01 | 0.0 | 58.46 | 36.12 | 44.66 | |||
| Parameters | This Study | Previous Study [13] |
|---|---|---|
| Sep-Pak C18 cartridge | Used | Not use |
| HPLC column | 10 µm, 150 mm × 4.6 mm i.d. | 10 µm, 250 mm × 4.6 mm i.d. |
| Flow rate | 0.5 mL/min | 0.4 mL/min |
| LOD | 0.05 ng/mL (MA), 0.48 ng/mL (MB) | 0.43 ng/mL (MA), 0.18 ng/mL (MB) |
| LOQ | 0.14 ng/mL (MA), 1.46 ng/mL (MB) | 1.31 ng/mL (MA), 0.54 ng/mL (MB) |
| Quantity of MA | 51.96 µg/mL | 16.44 µg/mL |
| Quantity of MB | 42.33 µg/mL | 9.24 µg/mL |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quan, N.V.; Xuan, T.D.; Tran, H.-D.; Dieu Thuy, N.T. Inhibitory Activities of Momilactones A, B, E, and 7-Ketostigmasterol Isolated from Rice Husk on Paddy and Invasive Weeds. Plants 2019, 8, 159. https://doi.org/10.3390/plants8060159
Quan NV, Xuan TD, Tran H-D, Dieu Thuy NT. Inhibitory Activities of Momilactones A, B, E, and 7-Ketostigmasterol Isolated from Rice Husk on Paddy and Invasive Weeds. Plants. 2019; 8(6):159. https://doi.org/10.3390/plants8060159
Chicago/Turabian StyleQuan, Nguyen Van, Tran Dang Xuan, Hoang-Dung Tran, and Nguyen Thi Dieu Thuy. 2019. "Inhibitory Activities of Momilactones A, B, E, and 7-Ketostigmasterol Isolated from Rice Husk on Paddy and Invasive Weeds" Plants 8, no. 6: 159. https://doi.org/10.3390/plants8060159
APA StyleQuan, N. V., Xuan, T. D., Tran, H.-D., & Dieu Thuy, N. T. (2019). Inhibitory Activities of Momilactones A, B, E, and 7-Ketostigmasterol Isolated from Rice Husk on Paddy and Invasive Weeds. Plants, 8(6), 159. https://doi.org/10.3390/plants8060159







