Myrica esculenta Buch.-Ham. ex D. Don: A Natural Source for Health Promotion and Disease Prevention
Abstract
:1. Introduction
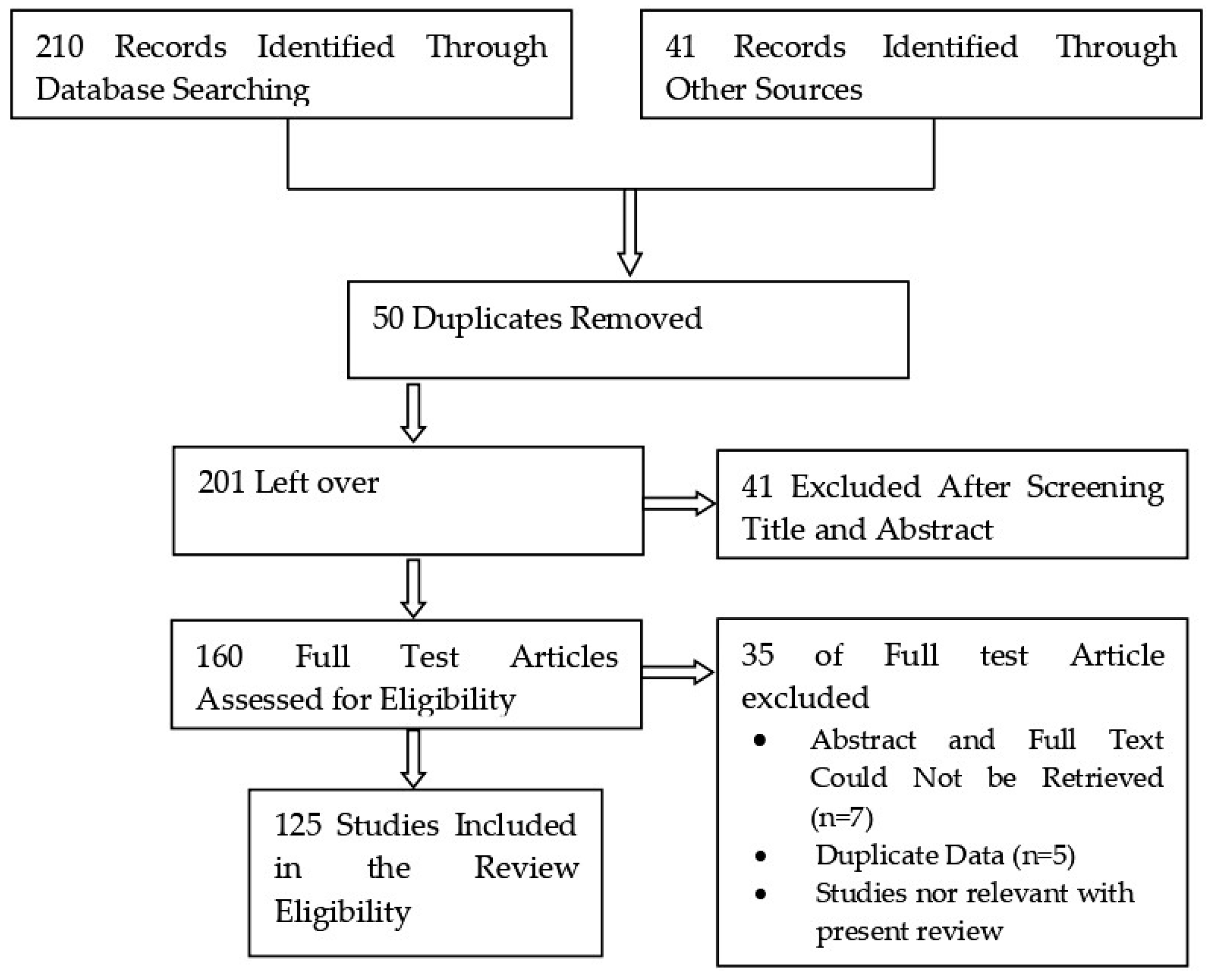
2. Research Methodology
3. Botanical Description
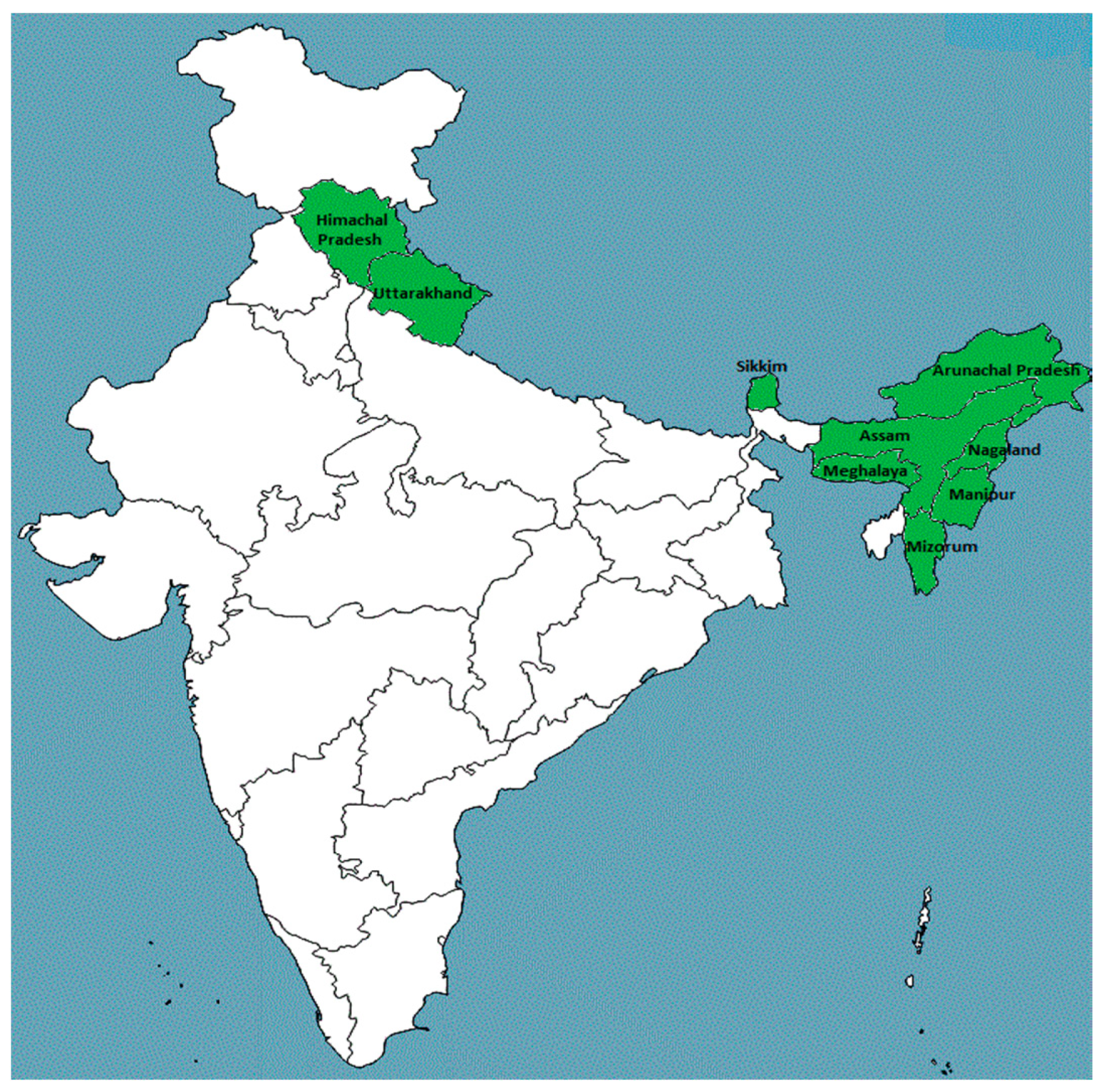
3.1. Habitat
3.2. Morphologicaland Microscopical Characteristics
4. Ethnomedicinal Uses
5. Physiochemical and Nutritional Analysis
6. Phytochemistry
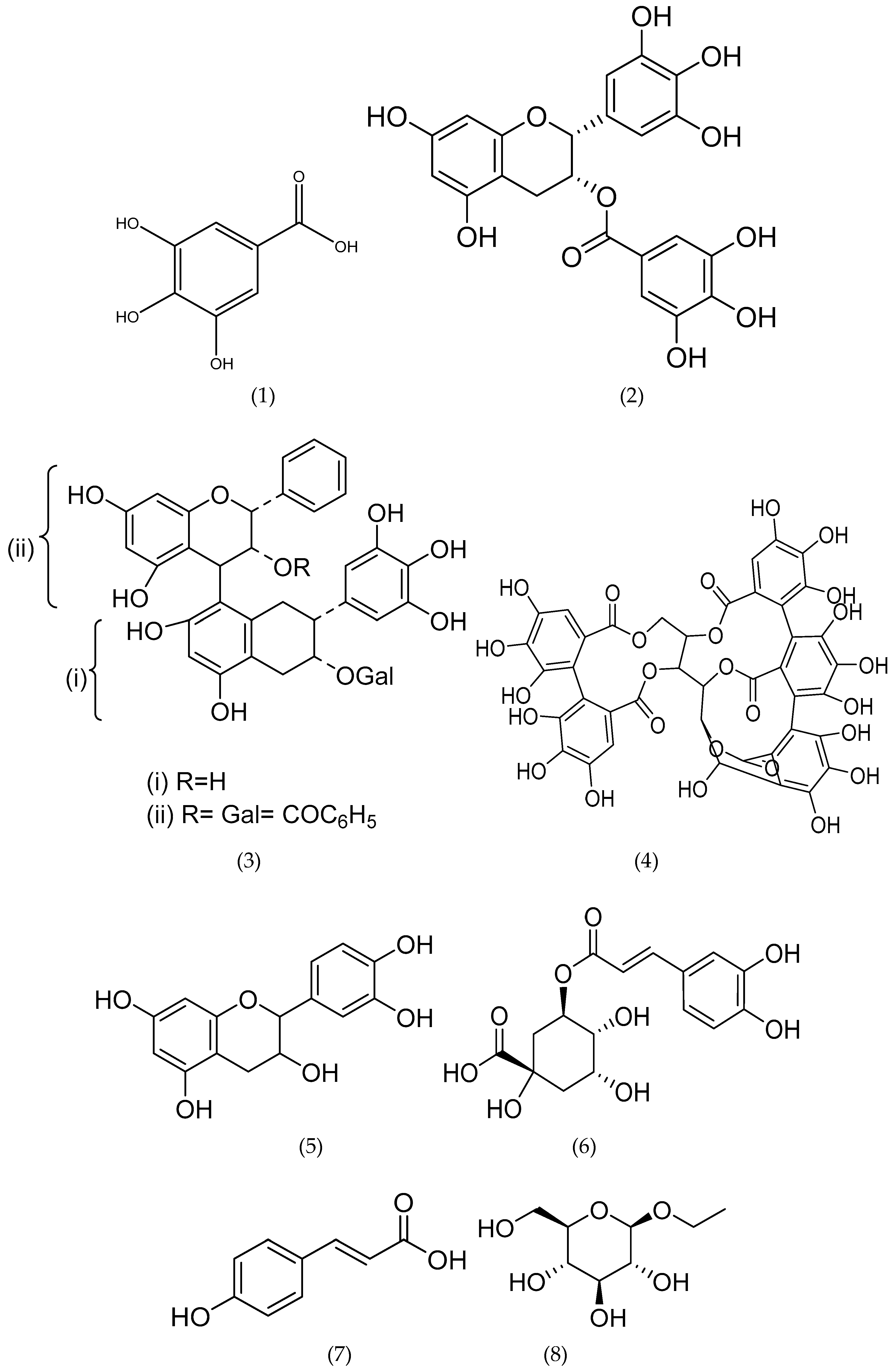
6.1. Tannins and Phenolic Acids
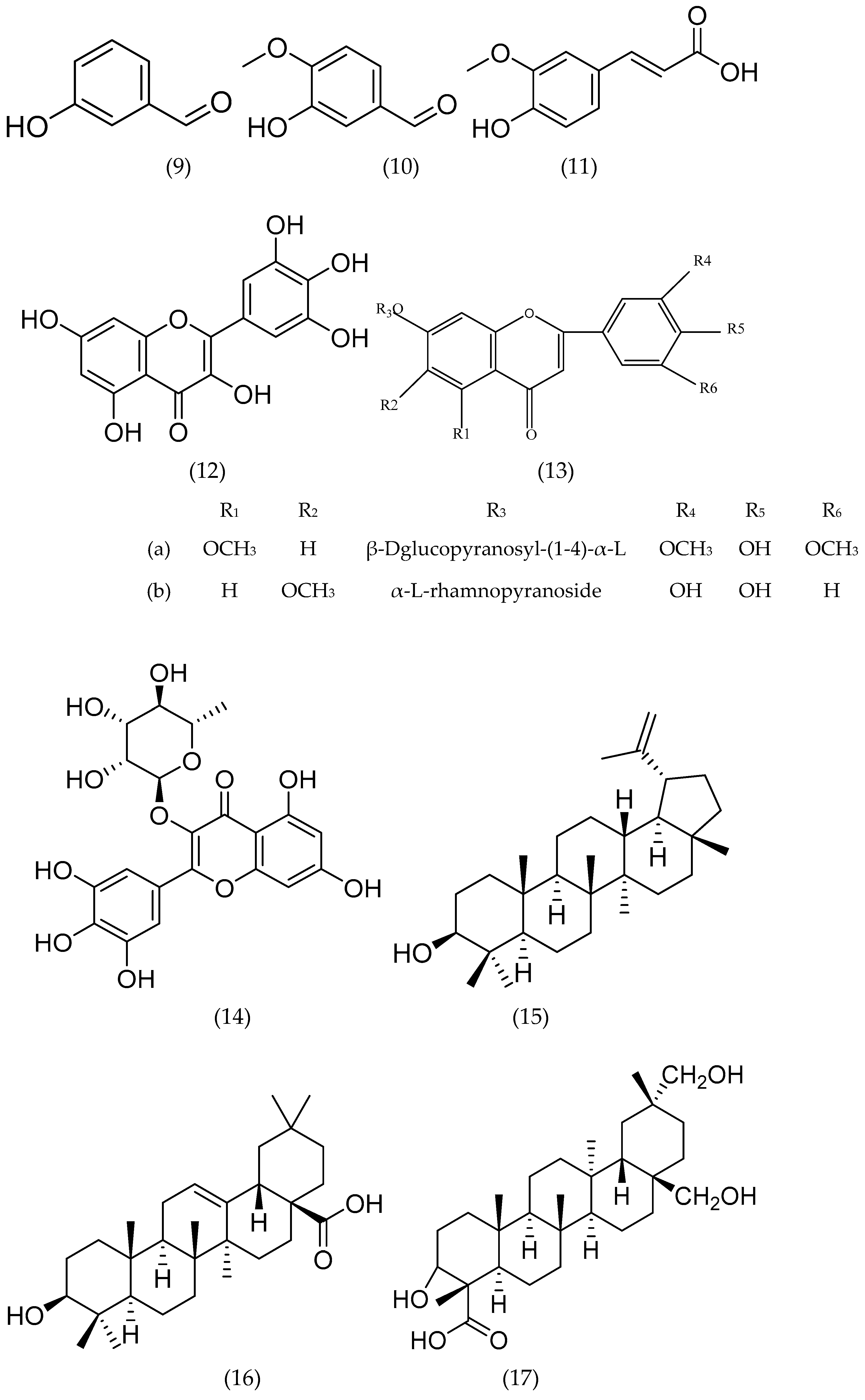
6.2. Flavonoids
6.3. Terpenes
Monoterpenoid
6.4. Triterpenoids
6.5. Volatile Compounds
6.6. Proanthocyanidins
6.7. Diarylheptanoids
6.8. Steroids
7. Pharmacological Profile
8. Conservation
9. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| HPTLC | High performance thin layer chromatography |
| LD50 | Lethal dose 50 |
| ME-EtAC | Ethyl acetate fraction-Myrica esculenta |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| ABTS | 2,2-azinobis (3-ethyl-benzothiazoline-6-sulfonic acid) |
| FRAP | Ferric reducing antioxidant power |
| MTT | Methyl thiazolyl tetrazolium |
| BP | Bacillus pumilus |
| SA | Staphylococcus aureus |
| SE | Staphylococcus epidermidis |
| EC | Escherichia coli |
| BS | Bacillus subtilis |
| PM | Proteus mirabilis |
| PA | Pseudomonas aeruginosa |
| CA | Candida albicans |
| AN | Aspergillus niger |
| SC | Saccharomyces cerevisiae |
| CCl4 | Carbon tetrachloride |
| H2O2 | Hydrogen peroxide |
| ACE | Angiotensin Converting Enzyme |
| MeOH | methanol |
| nm | Nanometer |
| EAE | Ethyl acetate extract |
| ME | Methanolic extract |
| AE | Aqueous extract |
| PE | Polar extract |
| NPE | Non polar extract |
| STZ | Streptozotocin |
| p.o. | Per oral |
| TBARS | Thio barbituric acid reactive substances |
| CAT | Catalase |
| SOD | Superoxide dismutase |
| GSH | Glutathione |
| MPO | Myeloperoxidase |
| GV | Gastric volume |
| FA | Free acidity |
| DLC | Differential Leukocyte Count |
| TLC | Total Leukocyte Count |
| mg/g | Milligram per gram |
| mg/kg | Milligram per kilogram |
| % w/w | Percentage weight by weight |
| GAE/g QE/g | Gallic Acid Equivalent per gram Quercetin equivalent per gram |
References
- Yanthan, M.; Misra, A.K. Molecular approach to the classification of medicinally important actinorhizal genus Myrica. Indian J. Biotechnol. 2013, 12, 133–136. [Google Scholar]
- Silva, B.J.C.; Seca, A.M.L.; Barreto, C.M.D.; Pinto, D.C.G.A. Recent breakthroughs in the antioxidant and anti-inflammatory effects of Morella and Myrica species. Int. J. Mol. Sci. 2015, 16, 17160–17180. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Rana, A.C. Pharmacognostic and pharmacological profile of traditional medicinal plant: Myrica nagi. Int. Res. J. Pharm. 2013, 3, 32–37. [Google Scholar]
- Sun, C.; Huang, H.; Xu, C.; Li, X.; Chen, K. Biological activities of extracts from Chinese bayberry (Myrica rubra Sieb. et Zucc.): A review. Plant Foods Hum. Nutr. 2013, 68, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Sood, P.; Shri, R. A review on ethnomedicinal, phytochemical and pharmacological aspects of Myrica esculenta. Indian J. Pharm. Sci. 2018, 80, 2–13. [Google Scholar]
- Sun, D.; Zhao, Z.; Wong, H.; Foo, L.Y. Tannins and other phenolics from Myrica esculenta bark. Phytochemistry 1988, 27, 579–583. [Google Scholar]
- Annonymous. The Wealth of India; Council of Scientific and Industrial Research: New Delhi, India, 1962; p. 472. [Google Scholar]
- Srivastava, B.; Sharma, V.C.; Pant, P.; Pandey, N.K.; Jadhav, A.D. Evaluation for substitution of stem bark with small branches of Myrica esculenta for medicinal use-A comparative phytochemical study. J. Ayurveda Integr. Med. 2016, 7, 1–6. [Google Scholar] [CrossRef]
- Nadkarni, K.M. Indian Materia Medica, 3rd ed.; Popular Book Depot: Mumbai, India, 2002; p. 871. [Google Scholar]
- Huguet, V.; Gouy, M.; Normand, P.; Zimpfer, J.M.; Fernandez, M.P. Molecular phylogeny of Myricaceae: A reexamination of host symbiont specificity. Mol. Phylogenet. Evol. 2005, 34, 557–568. [Google Scholar] [CrossRef]
- Bhatt, I.D.; Rawal, R.S.; Dhar, U. Improvement in seed germination of Myrica esculenta Buch. Ham. Ex D. Don- A high value tree species of Kumanun Himalaya, India. Seed Sci. Technol. 2000, 28, 597–605. [Google Scholar]
- Catalogue of Life: 2019 Annual Checklist. Available online: http://www.catalogueoflife.org/col/search/all/key/myrica+esculenta+/fossil/1/match/1 (accessed on 15 May 2019).
- Pandey, G.; Sharma, B.D.; Hore, D.K.; Rao, N.K. Indigenous minor fruits genetic resources and their marketing status in north-eastern hills of India. J. Hill Res. 1993, 6, 1–4. [Google Scholar]
- Makdoh, K.; Lynser, M.B.; Pala, K.H.M. Marketing of Indigenous Fruits: A Source of Income among Khasi Women of Meghalaya, North East India. J. Agric. Sci. 2014, 5, 1–9. [Google Scholar] [CrossRef]
- MacDnald, A.D. The morphology and relationships of the Myricaceae. Evol. Syst. Foss. Hist. Hamamelidae 1989, 2, 147–165. [Google Scholar]
- Haridasan, K.; Rao, R.R. Forest Flora of Meghalaya; Caprifoliaceae to Salicaceae; Bishan Singh Mahendra Pal Singh: Dehradun, India, 1987; Volume 2, pp. 851–852. [Google Scholar]
- Paranjpe, P. Indian Medicinal Plants, 3rd ed.; Chaukhamba Sanskrit Pratishthan: New Delhi, India, 2012; p. 128. [Google Scholar]
- Bhatt, I.D.; Dhar, U. Factors controlling micropropagation of Myrica esculenta Buch. -Ham. ex D. Don: A high value wild edible of Kumaun Himalaya. Afr. J. Biotechnol. 2004, 3, 534–540. [Google Scholar]
- Bao, J.; Cai, Y.; Sun, M.; Wang, G.; Corke, H. Anthocyanins, flavonols, and free radical scavenging activity of Chinese Bayberry (Myrica rubra) extracts and their color properties and stability. J. Agric. Food Chem. 2005, 53, 2327–2332. [Google Scholar] [CrossRef] [PubMed]
- The Ayurvedic Pharmacopoeia of India, part I, 1st ed.; Government of India, Ministry of Health and Family Welfare, Department of Indian System of Medicine and Homeopathy: New Delhi, India, 1999; Volume III, pp. 92–93.
- Kabra, A.; Sharma, R.; Singla, S.; Kabra, R.; Baghel, U.S. Pharmacognostic characterization of Myrica esculenta leaves. J. Ayurveda Integr. Med. 2017, 10, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Dollo, M.; Samal, P.K.; Sundriyal, R.C.; Kumar, K. Environmentally sustainable traditional natural resource management and conservation in Ziro valley, Arunachal Himalaya, India. J. Am. Sci. 2009, 5, 41–52. [Google Scholar]
- Kumar, J.K.; Sinha, A.K. Resurgence of natural colourants: A holistic view. Nat. Prod. Res. 2004, 18, 59–84. [Google Scholar] [CrossRef]
- Jeeva, S.; Lyndem, F.B.; Sawian, J.T.; Laloo, R.C.; Mishra, B.P. Myrica esculenta Buch.-Ham. ex D. Don.—A potential ethnomedicinal species in a subtropical forest of Meghalaya, northeast India. Asian Pac. J. Trop. Biomed. 2011, 1, S174–S177. [Google Scholar] [CrossRef]
- Bhatt, B.P.; Tomar, J.M.S.; Bujarbaruah, K.M. Characteristics of Some Firewood trees and shrubs of the North Eastern Himalayan Region, India. Renew. Energy 2004, 29, 1401–1405. [Google Scholar] [CrossRef]
- Kala, C.P. Prioritization of cultivated and wild edibles by local people in the Uttaranchal hills of Indian Himalaya. Indian J. Tradit. Knowl. 2007, 6, 239–243. [Google Scholar]
- Gusain, Y.S.; Khanduri, V.P. Myrica esculenta wild edible fruit of Indian Himalaya: Need a sustainable approach for indigenous utilization. Ecol. Environ. Conserv. 2016, 22, S267–S270. [Google Scholar]
- Gupta, R.K. The Living Himalaya; Today and Tomorrow Printers and Publishers: Delhi, India, 1989; Volume 2. [Google Scholar]
- Fang, Z.; Zhang, M.; Wang, L. HPLC-DAD-ESIMS analysis of phenolic compounds in bayberries (Myrica rubra Sieb. et Zucc.). Food Chem. 2007, 100, 845–852. [Google Scholar] [CrossRef]
- Zhang, W.S.; Li, X.; Zheng, J.T.; Wang, G.Y.; Sun, C.D.; Ferguson, I.B. Bioactive components and antioxidant capacity of Chinese bayberry (Myrica rubra Sieb. and Zucc.) fruit in relation to fruit maturity and post harvesting storage. Eur. Food Res. Technol. 2008, 227, 1091–1097. [Google Scholar] [CrossRef]
- Fang, Z.; Zhang, Y.; Lu, Y.; Ma, G.; Chen, J.; Liu, D.; Ye, X. Phenolic compounds and antioxidant capacities of bayberry juices. Food Chem. 2009, 113, 884–888. [Google Scholar] [CrossRef]
- Zhou, S.H.; Fang, Z.X.; Lu, Y.; Chen, J.C.; Liu, D.H.; Ye, X.Q. Phenolics and antioxidant properties of bayberry (Myrica rubra Sieb. et Zucc.) pomace. Food Chem. 2009, 112, 394–399. [Google Scholar] [CrossRef]
- Lau-Cam, C.A.; Chan, H.H. Flavonoids from Comptonia peregrine. Phytochemistry 1973, 12, 1829. [Google Scholar] [CrossRef]
- Jones, J.R.; Lebar, M.D.; Jinwal, U.K.; Abisambra, J.F.; Koren, J.; Blair, L.; O’Leary, J.C.; Davey, Z.; Trotter, J.; Johnson, A.G. The diarylheptanoid (+)-aR,11S-myricanol and two flavones from bayberry (Myrica cerifera) destabilize the microtubule-associated protein tau. J. Nat. Prod. 2011, 74, 38–44. [Google Scholar] [CrossRef]
- Semwal, D.K.; Semwal, R.B.; Combrinck, S.; Viljoen, A. Myricetin: A Dietary Molecule with Diverse Biological Activities. Nutrients 2016, 8, 90. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, K.; Wang, Y.; Ma, R. Biological effects study of Myricitrin and relevant molecular mechanisms. Curr. Stem Cell Res. Ther. 2019, 14. [Google Scholar] [CrossRef]
- Perkin, A.G.; Hummel, J.J. LXXVI-The colouring principle contained in the bark of Myrica nagi Part I. J. Chem. Soc. Trans. 1896, 69, 1287–1294. [Google Scholar] [CrossRef]
- Chauhan, N.S. Medicinal and Aromatic Plants of Himachal Pradesh; Indus Publishing Company: New Delhi, India, 1999; p. 226. [Google Scholar]
- Anonymous. Ayurvedic Pharmacopoeia of India, Part 1, Vol III.; Ministry of Health and Family Welfare, Department of Indian System of Medicine and Homeopathy: New Delhi, India, 2007; pp. 90–96.
- Mozhui, R.; Rongsensashi, L.; Changkija, S. Wild edible fruits used by the tribals of Dimapur district of Nagaland, India, East Himalayan Society for Spermatophyte Taxonomy. Pleione 2011, 5, 56–64. [Google Scholar]
- Kirtikar, K.R.; Basu, B.D. Indian Medicinal Plants. Vol. III, 2nd ed.; International book distributors: New Delhi, India, 1999; p. 1699. [Google Scholar]
- Osmaston, A.E. A Forest Flora of Kumaon; Bishen Singh Mahinder Pal Singh: Dehradun, India, 1994. [Google Scholar]
- Singh, N.P.; Gajurel, P.R.; Rethy, P. Ethnomedicinal value of traditional food plants used by the Zeliang tribe of Nagaland. Indian J. Tradit. Knowl. 2015, 14, 298–305. [Google Scholar]
- Gyawali, R.; Hengaju, A.; Thapa, P.M.; Khadka, P.; Sah, R.; Bhandari, S.; Adhikari, S.; Subedi, S.; Shrestha, A.K.; Shrestha, T.M. Antioxidant and Wound Healing Property of Polyherbal Ointment of Nepalese Medicinal Plants. Int. J. Allied Med. Sci. Clin. Res. 2016, 4, 275–283. [Google Scholar]
- Shrestha, P.M.; Dhillion, S.S. Medicinal plant diversity and use in the highlands of Dolakha district, Nepal. J. Ethnopharmacol. 2003, 86, 81–96. [Google Scholar] [CrossRef]
- Nguyen, X.N.; Phan, V.K.; Chau, V.M.; Bui, H.T.; Nguyen, X.C.; Vu, K.T.; Hoang le, T.A.; Jo, S.H.; Jang, H.D.; Kwon, Y.I.; et al. A new monoterpenoid glycoside from Myrica esculenta and the inhibition of Angiotensin I-Converting Enzyme. Chem. Pharm. Bull. 2010, 58, 1408–1410. [Google Scholar] [PubMed]
- Kankanamalage, T.N.M.; Dharmadasa, R.M.; Abeysinghe, D.C.; Wijesekara, R.G.S. A survey on medicinal materials used in traditional systems of medicine in Sri Lanka. J. Ethnopharmacol. 2014, 155, 679–691. [Google Scholar] [CrossRef] [PubMed]
- Kuang, K.Z.; Lu, A.M. Myricaceae, Flora Reipublicae Popularis Sinicae; Science Press: Beijing, China, 1979; pp. 1–6. [Google Scholar]
- Parmar, C.; Kaushal, M.K. Wild Fruits of the Sub-Himalayan Region. In Myrica nagi; Parmar, C., Kaushal, M.K., Eds.; Kalyani Publishers: New Delhi, India, 1982; pp. 49–53. [Google Scholar]
- Dhani, A. Major wild edible fruits used by locals of Garhwal Himalaya. Int. J. Adv. Life Sci. 2013, 6, 145–149. [Google Scholar]
- Pundir, S.; Tomar, S.; Upadhyay, N.; Sharma, V. Antioxidant, anti-inflammatory and analgesic activity of bioactive fraction of leaves of Myrica esculenta Buch.-Ham along with its pharmacognostic and chromatographic evaluation. Int. J. Biol. Pharm. Allied Sci. 2015, 4, 6509–6524. [Google Scholar]
- Singh, J.; Lan, V.K.; Trivedi, V.P. Pharmacognostic evaluation of Katphala (The bark of Myrica esculenta Buch–Ham). Anc. Sci. Life 1986, 6, 85–87. [Google Scholar]
- Sahu, S.; Sahu, C.R.; Yadav, A.; Rathod, P.; Chaturvedi, S.; Tripathi, R. Review on Myrica esculenta a popular plant of Himalayan region. J. Chem. Pharm. Sci. 2013, 6, 93–97. [Google Scholar]
- Anonymous. Data base on Medicinal Plants used in Ayurveda and Siddha, Volume VIII.; CCRAS: New Delhi, India, 2007; p. 207. [Google Scholar]
- Mallya, S.V.; Nesari, T.; Kumar, K.N. Pharmacognostic standards of Katphala (Myrica nagi Hook. f. non-Thumb); A potent bark drug used in Indian systems of medicine. J. Sci. Innov. Res. 2016, 5, 135–137. [Google Scholar]
- Panthari, P.; Kharkwal, H.; Kharwal, H.; Joshi, D.D. Myrica nagi: A review on active constituents, biological and therapeutic effects. Int. J. Pharm. Pharm. Sci. 2012, 4, 38–42. [Google Scholar]
- Boloor, V.A.; Hosadurga, R.; Rao, A.; Jenifer, H.; Pratap, S. Unconventional Dentistry in India – An Insight into the Traditional Methods. J. Trad. Complement. Med. 2014, 4, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Manandhar, N.P. A survey of medicinal plants of Jajarkot district, Nepal. J. Ethnopharmacol. 1995, 48, 1–6. [Google Scholar] [CrossRef]
- Nainwal, P.; Kalra, K. Study on the wound activity potential on the aqueous extract of the bark of Myrica esculenta Buch. &Ham. Int. J. Pharm. Clin. Res. 2009, 1, 85–87. [Google Scholar]
- Gaire, B.P.; Subedi, L. Medicinal Plant Diversity and their Pharmacological Aspects of Nepal Himalayas. Pharmacogn. J. 2011, 25, 6–17. [Google Scholar] [CrossRef]
- Khan, M.Y.; Sagrawat, H.; Upmanyu, N.; Siddique, S. Anxiolytic Properties of Myrica nagi Bark Extract. Pharm. Biol. 2008, 46, 757–761. [Google Scholar] [CrossRef]
- Bich, D.H.; Chung, D.Q.; Chuong, B.X.; Dong, N.T.; Dam, D.T.; Hien, P.V.; Lo, V.N.; Mai, P.D.; Man, P.K.; Nhu, D.T.; et al. The Medicinal Plants and Animals in Vietnam; Hanoi Science and Technology Publishing House: Hanoi, Vietnam, 2004; Volume 1, pp. 612–613. [Google Scholar]
- Joshi, A.R.; Edington, J.M. The use of medicinal plants by two village communities in the Central Development Region of Nepal. Econ. Bot. 1990, 44, 71–83. [Google Scholar] [CrossRef]
- Sharma, H.K.; Chhangte, L.; Dolui, A.K. Traditional medicinal plants in Mizoram, India. Fitoterapia 2001, 72, 146–161. [Google Scholar] [CrossRef]
- Shrestha, N.; Prasai, D.; Shrestha, K.K.; Shrestha, S.; Zhang, X.C. Ethnomedicinal practices in the highlands of central Nepal: A case study of Syaphru and Langtang village in Rasuwa district. J. Ethnopharmacol. 2014, 155, 1204–1221. [Google Scholar] [CrossRef]
- Kumari, P.; Joshi, G.C.; Tewari, L.M. Diversity and status of ethnomedicinal trees of Almora district in Uttarakhand, India. Int. J. Biodivers. Conserv. 2011, 3, 298–326. [Google Scholar]
- Kichu, M.; Malewska, T.; Akter, K.; Imchen, I.; Harrington, D.; Kohen, J.; Vemulpad, S.R.; Jamie, J.F. An ethnobotanical study of medicinal plants of Chungtia village, Nagaland, India. J. Ethnopharmacol. 2015, 166, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Semwal, D.P.; Saradhi, P.P.; Kala, C.P.; Sajwan, B.S. Medicinal plants used by local Vaidyas in Ukhimath block, Uttarakhand. Indian J. Tradit. Knowl. 2010, 9, 480–485. [Google Scholar]
- Bhatt, I.D.; Rawat, S.; Badhani, A.; Rawal, R.S. Nutraceutical potential of selected wild edible fruits of the Indian Himalayan region. Food Chem. 2017, 215, 84–91. [Google Scholar] [CrossRef]
- Dhyani, P.P.; Dhar, U. Myrica esculenta, Box Myrtle (Kaiphal); Himavikas Occasional Publication; GB Pant Institute of Himalayan Environment and Development: Almora, India, 1994.
- Pala, N.A.; Negi, A.K.; Todaria, N.P. Traditional uses of medicinal plants of Pauri Garhwal, Uttrakhand. Nat. Sci. 2010, 8, 57–61. [Google Scholar]
- Seal, T. Nutritional composition of wild edible fruits in Meghalaya state of India and their ethnobotanical Importance. Res. J. Bot. 2011, 6, 58–67. [Google Scholar]
- Chandra, S.; Saklani, S.; Mishra, A.P.; Badoni, P.P. Nutritional evaluation, antimicrobial activity and phytochemical screening of wild edible fruit of Myrica nagi pulp. Int. J. Pharm. Pharm. Sci. 2012, 4, 407–411. [Google Scholar]
- Patel, V.G.; Patel, K.G.; Patel, K.V.; Gandhi, T.R. Development of Standardisation parameters and Isolation of Phytomarker Myricetin from stem bark of Myrica esculenta Buch. Ham. Ex d. Don. J. Pharmacogn. Phytochem. 2017, 6, 29–34. [Google Scholar]
- Singh, N.; Khatoon, S.; Srivastava, N.; Rawat, A.; Mehrotra, S. Qualitative and quantitative standardization of Myrica esculenta Buch.-Ham. Stem bark by use of HPTLC. J. Planar Chromatogr. 2009, 22, 287–291. [Google Scholar] [CrossRef]
- Rawat, S.; Jugran, A.; Giri, L.; Bhatt, I.D.; Rawal, R.S. Assessment of antioxidant properties in fruits of Myrica esculenta: A popular wild edible species in Indian Himalayan Region. Evid. Based Complet. Altern. Med. 2011, 2011, 1–8. [Google Scholar] [CrossRef]
- Wei, Y.; Chang-ming, T.; Xian, L.; Ya, Z.; Li, W.; Liang, L. Study on the chemical constituents of Myrica esculenta. J. Yunnan Univ. (Nat. Sci.) 2011, 33, 453–457. [Google Scholar]
- Mann, S.; Satpathy, G.; Gupta, R.K. In-vitro evaluation of bioprotective properties of underutilized Myrica esculenta Buch.-Ham. ex D. Don fruit of Meghalaya. Indian J. Nat. Prod. Resour. 2015, 6, 183–188. [Google Scholar]
- Dawang, S.; Zuchun, Z.; Foo, L.Y.; Wong, H. Flavonols from Myrica esculenta bark. Chem. Indus. Forest Prod. 1991, 4, 251–257. [Google Scholar]
- Bamola, A.; Semwal, D.K.; Semwal, S.; Rawat, U. Flavonoid glycosides from Myrica esculenta leaves. J. Indian Chem. Soc. 2009, 86, 535–536. [Google Scholar]
- Available online: http://www.niscair.res.in/activitiesandservices/products/wealth-ofIndiaFolder2010.pdf (accessed on 15 May 2019).
- Agnihotri, S.; Wakode, S.; Ali, M. Triterpenoids from the stem bark of Myrica esculenta Buch Ham. World J. Pharm. Pharm. Sci. 2016, 5, 1319–1327. [Google Scholar]
- Hui-fen, M.A.; Zheng-liang, Y.; Sang-zi, Z.E.; Yong-jie, L.; De-lu, N.; Zhen, Y.U. GC/MS analysis of volatile components from leaf of Myrica esculenta Buch.-Ham. Guangdong Agric. Sci. 2011, 16, 18. [Google Scholar]
- Krishnamoorthy, V.; Seshadri, T.R. A new Proanthocyanidin from the stem bark of Myrica nagi thumb. Tetrahedron 2001, 22, 2367–2371. [Google Scholar] [CrossRef]
- Mei, W.D.; Hong, C.J.; Mei, W.Y.; Man, X.; Song, W.Z. Study on ultrasound-assisted extraction of proanthocyanidins from Myrica esculenta Bark. Chem. Ind. Forest Prod. 2009, 29, 105–109. [Google Scholar]
- Begley, M.J.; Campbell, R.V.M.; Crombie, L.; Tuck, B.; Whiting, D.A. Constitution and absolute configuration of meta, metabridged, stained biphenyls from Myrica nagi: X-ray analysis of 16-bromomyricanol. J. Chem. Soc. C Org. 1971, 1970, 3634–3642. [Google Scholar]
- Malterud, K.E.; Anthonsen, T. 13-oxomyricanol, a new [7.0]-metacyclophane from Myrica nagi. Phytochemistry 1980, 19, 705–707. [Google Scholar] [CrossRef]
- Agarwal, K.P.; Roy, A.C.; Dhar, M.L. Triterpenes from the Bark of Myrica esculenta Buch.-Ham. Indian J. Chem. 1963, 1, 28–30. [Google Scholar]
- Husain, S.R.; Cillard, J.; Cillard, P. Hydroxyl radical scavenging activity of flavonoids. Phytochemistry 1987, 26, 2489–2491. [Google Scholar] [CrossRef]
- Afanaslev, I.B.; Dorozhko, A.I.; Bordskii, A.V. Chelating and free radical scavenging mechanisms of inhibitory action of rutin and quercetin in lipid peroxidation. Biochem. Pharmacol. 1989, 38, 1763–1769. [Google Scholar] [CrossRef]
- Torel, J.; Cillard, J.; Cillard, P. Antioxidant activity of flavonoids and reactivity with peroxy radical. Phytochemistry 1986, 25, 383–385. [Google Scholar] [CrossRef]
- Pant, G.; Prakash, O.; Chandra, M.; Sethi, S.; Punetha, H.; Dixit, S. Biochemical analysis, pharmacological activity, antifungal activity and mineral analysis in methanol extracts of Myrica esculenta and Syzygiumcumini: The Indian traditional fruits growing in Uttarakhand Himalaya. Indian J. Pharm. Biol. Res. 2014, 2, 26–34. [Google Scholar]
- Middha, S.K.; Kumar, G.A.; Talambedu, U.; Babu, D.; Misra, A.K.; Prakash, L. Evaluation of antioxidative, analgesic and antiinflammatory activities of methanolic extract of Myrica nagi leaves—An animal model approach. Symbiosis 2016, 13, 179–184. [Google Scholar] [CrossRef]
- Seal, T. Antioxidant Activity of Some Wild Edible Fruits of Meghalaya State in India. Adv. Biol. Res. 2011, 5, 155–160. [Google Scholar]
- Goyal, A.K.; Mishra, T.; Bhattacharya, M.; Kar, P.; Sen, A. Evaluation of phytochemical constituents and antioxidant activity of selected actinorhizal fruits growing in the forests of Northeast India. J. Biosci. 2013, 38, 797–803. [Google Scholar] [CrossRef]
- Chen, J.; Wang, Y.; Wu, D.; Wu, Z. Preliminary study on antioxidative and radical scavenging activities of extracts from Myrica esculenta Buch.-Ham. Bark. Chem. Ind. Forest Prod. 2007, S1, 1–7. [Google Scholar]
- Rana, R.K.; Patel, R.K. Antioxidant Activity of Bark of Myrica nagi. Int. J. Pharm. Sci. Rev. Res. 2014, 28, 99–101. [Google Scholar]
- Patel, K.G.; Bhalodia, P.N.; Patel, A.D.; Patel, K.V.; Gandhi, T.R. Evaluation of bronchodilator and antianphylactic activity of Myrica sapida. Iran. Biomed. J. 2008, 12, 191–196. [Google Scholar]
- Patel, K.G.; Rao, N.J.; Gajera, V.G.; Bhatt, P.A.; Patel, K.V.; Gandhi, T.R. Antiallergic activity of stem bark of Myrica esculenta Buch.-Ham. (Myricaceae). J. Young Pharm. 2010, 2, 74–78. [Google Scholar] [CrossRef]
- Patel, T.; Ladani, K.; Shah, S. Antiasthmatic activity of aqueous extract of Myrica nagi bark. Int. J. Phytopharm. Res. 2013, 4, 40–45. [Google Scholar]
- Rana, R.K.; Patel, R.K. Pharmacological Evaluation of Antiasthmatic Activity of Myrica nagi Bark Extracts. Antiinflamm. Antiallergy Agents Med. Chem. 2016, 15, 145–152. [Google Scholar] [CrossRef]
- Patel, T.; Rajshekar, C.; Parmar, R. Mast cell stabilizing activity of Myrica nagi bark. J. Pharmacogn. Phytother. 2011, 3, 114–117. [Google Scholar]
- Saini, R.; Garg, V.; Dangwal, K. Effect of extraction solvents on polyphenolic composition and antioxidant, antiproliferative activities of Himalayan bayberry (Myrica esculenta). Food Sci. Biotechnol. 2013, 22, 887–894. [Google Scholar] [CrossRef]
- Syed, S.; Ahmad, M.; Fatima, N.; Mahjabeen; Jahan,, N. Neuropharmacological studies of Myrica nagi bark. Int. J. Biol. Biotechnol. 2013, 10, 553–558. [Google Scholar]
- Rawat, S.; Kumar, N.; Kothiyal, P. Evaluate the antidiabetic activity of Myrica esculenta leaves in streptozotocin induced diabetes in rat. Int. J. Univ. Pharm. Bio. Sci. 2013, 2, 510–525. [Google Scholar]
- Nayak, B.K.; Deka, P.; Eloziia, N. Assessment of phytochemical & pharmacological activities of the ethanol leaves extracts of Myrica esculenta Buch. Ham. J. Pharm. Res. 2017, 11, 444–449. [Google Scholar]
- Jain, V.K.; Jain, B. Anthihelmintic Activity of ethanolic extract of bark of Myrica esculenta. Int. J. Pharm. Sci. Res. 2010, 1, 129–131. [Google Scholar]
- Patel, T.; Dudhpejiya, A.; Sheath, N. Antiinflammatory activity of Myrica nagi Linn. Bark. Anc. Sci. Life 2011, 30, 100–103. [Google Scholar]
- Suryawanshi, J.S.; Karande, K.M.; Udugade, B.V. Antibacterial activity of bark and fruits of Myrica nagi. Indian J. Nat. Prod. 2009, 25, 21–23. [Google Scholar]
- Shan, B.; Cai, Y.Z.; Brooks, J.D.; Corke, H. The in vitro antibacterial activity of dietary spice and medicinal herb extracts. Int. J. Food Microbiol. 2007, 117, 112–119. [Google Scholar] [CrossRef]
- Agnihotri, S.; Wakode, S.; Ali, M. Essential oil of Myrica esculenta Buch. Ham: Composition, antimicrobial and topical antiinflammatory activities. Nat. Prod. Res. 2012, 26, 2266–2269. [Google Scholar] [CrossRef]
- Swathi, D.; Prasad, K.V.S.R.G. Antioxidant and antiulcer potential of ethanolic extract of bark of Myrica esculenta in pyloric ligation ulcer model. Int. J. Pharm. Pharm. Sci. 2015, 7, 195–198. [Google Scholar]
- Alam, A.; Iabal, M.; Saleem, M.; Ahmed, S.U.; Sultana, S. Myrica nagi attenuates cumene hydroperoxide-induced cutaneous oxidative stress and toxicity in swiss albino mice. Pharmacol. Toxicol. 2000, 86, 209–214. [Google Scholar] [CrossRef]
- Samundeeswari, C.; Rajadurai, M.; Periasami, R.; Kanchana, G. Hepatoprotective effect of Herbitars, A polyherbal against CCl4 induced hepatotoxicity in rats. J. Pharm. Res. 2011, 4, 676–679. [Google Scholar]
- Patil, S.P.; Pardeshi, M.L.; Ghongane, B.B. Screening for Anti-allergic and Anti-histaminic Activity of Extract of Momordicadioica, Myrica esculenta and Euphorbiahirta in Animal Models. Res. J. Pharmaceu. Biol. Chem. Sci. 2016, 7, 21–28. [Google Scholar]
- Sharma, R.; Kabra, A.; Rao, M.M.; Prajapati, P.K. Herbal and Holistic solutions for Neurodegenerative and Depressive disorders Leads from Ayurveda. Curr. Pharm. Des. 2018, 24, 2597–2608. [Google Scholar] [CrossRef]
- Rawal, R.S.; Pandey, B.; Dhar, U. Himalayan forest database-Thinking beyond dominants. Curr. Sci. 2003, 84, 990–994. [Google Scholar]
- Lohani, N.; Tewari, L.M.; Joshi, G.C.; Kumar, R.; Kishor, K.; Upreti, M.R. Population assessment and threat categorization of endangered medicinal orchid Malaxis acuminata D. Don. From North-West Himalaya. Int. J. Conserv. Sci. 2013, 4, 483–940. [Google Scholar]
- Chapin, F.S.; Zavaleta, E.S.; Eviner, V.T.; Naylor, R.L.; Vitousek, P.M.; Reynolds, H.L.; Hooper, D.U.; Lavorel, S.; Sala, O.E.; Hobbie, S.E.; et al. Consequences of changing biodiversity. Nature 2000, 405, 234–242. [Google Scholar] [CrossRef]
- Koorbanally, C.; Crouch, N.R.; Mulholland, D.A. The phytochemistry and ethnobotany of the Southern African genus Eucomis (Hyacinthaceae: Hya- cinthoideae). In Phytochemistry: Advances in Research; Imperato, F., Ed.; Research Signpost: Kerala, India, 2006; pp. 69–85. [Google Scholar]
- Sharma, R.; Kuca, K.; Nepovimova, E.; Kabra, A.; Rao, M.M.; Prajapati, P.K. Traditional Ayurvedic and herbal remedies for Alzheimers disease from bench to bedside. Expert Rev. Neurother. 2019, 19, 359–374. [Google Scholar] [CrossRef]
- Sharma, R.; Martins, N.; Kuca, K.; Chaudhary, A.; Kabra, A.; Rao, M.M.; Prajapati, P.K. Chyawanprash A Traditional Indian Bioactive Health Supplement. Biomolecules 2019, 9, 161. [Google Scholar] [CrossRef]


| Formulation | Uses | Manufacturers | References |
|---|---|---|---|
| “Chwayanprash” | Improved digestion and strength and enhanced energy | Dabur, Patanjali, Nature & Nurture Healthcare | [19,20] |
| “Katphaladi Churna” | Treatment of fever, throat infection, respiratory disorders, and abdominal pain | VHCA Ayurveda | [19,20] |
| “Pushyanuga Churna” | Treatment for bleeding disorders and candidiasis | AVN Ayurveda,Baidyanat-h | [19,20] |
| “Katphala Taila” | Treatment of joint pain | VHCA Ayurveda | [19,20] |
| “Arimedadi Taila” | Helps to relieve tooth decay and breath problem | IMIS Pharmaceuticals | [19,20] |
| “Mahavisagarbha Taila” | Used for vata imbalance, neuromuscular conditions | VHCA Ayurveda | [19,20] |
| “Bala Taila” | Treatment of vata disorders, respiratory infections and weakness | Patanjali | [19,20] |
| “Khadiradi Gutika” | Treatment of dental, oral, throat and tonsillar infections | Zandu | [19,20] |
| “Maha Vatagajankusa Rasa” | Rheumatoid arthritis, Migraine, Paralysis, Cough, Cold, Asthma | Dabur, Baidyanath, Shree Dhootapapeshwar | [19,20] |
| “Brihat Phala Ghrta” | Treatment of infertility | SN Pandit Ayurvedic | [19,20] |
| Plant Part Used | Uses | Region/Tribe | References |
|---|---|---|---|
| Leaf, fruit, root, bark | Jaundice | Meghalaya, India | [23] |
| Leaf | Inflammation of vocal cord | Meghalaya, India | [24] |
| Bark | Antiseptic | Meghalaya, India Khasi tribe | [24] |
| Fruit, bark, leaf | Fever | Meghalaya, India Vietnam, South China | [24] |
| Bark | Anemia | Meghalaya, India Khasi tribe | [24] |
| Fruit | Refreshing drink “Um Soh-Phi” | Meghalaya, India Khasi tribe | [24] |
| Bark | Sore | Nagaland, India Zeliang tribe | [43] |
| Bark | Toothache | Meghalaya, India Khasi tribe Almora, Uttarakhand, India | [24,41,57] |
| Bark | Sprain | Far-flung village, Jajarkot, Nepal | [58] |
| Flower, bark, leaf | Inflammation, paralysis | Meghalaya, India Khasi tribe Vietnam, South China | [24,59] |
| Unripe fruit | Anthelmintic | Himachal Pradesh, India | [59] |
| Fruit | Bronchitis, dysentery | Nepalese community, Nepal | [60] |
| Bark | Mental illness | Orissa, India | [61] |
| Bark | Skin disorder | Vietnam, South China | [62] |
| Bark | Cholera | Mizoram, India | [63,64] |
| Bark | Cardiac debility, cardiac edema | Meghalaya, India | [64] |
| Bark | Carminative | Meghalaya, India Khasi tribe Mizoram, India | [22,64] |
| Bark, leaf | Asthma, chronic bronchitis, lung infection | Meghalaya, India Khasi tribe Vietnam, South China Chaubas and Syabru, Nepal | [23,63,65] |
| Flower | Earache | Meghalaya, India Khasi tribe Almor, Uttarakhand, India Himachal Pradesh, India | [24,41,49,66] |
| Bark, flower, leaf, fruit | Diarrhea, dysentery, stomach problem | Meghalaya, India Khasi tribe Almora, Uttarakhand, India Chungtia village, Nagaland, India | [24,66,67] |
| Leaf | Redness of mucosa | Chungtia village, Nagaland, India | [67] |
| Fruit | Body ache | Ukhimath block, Uttarakhand, India | [68] |
| Bark, fruit | Headache | Mizoram, India Ukhimath block, Uttarakhand. India | [64,68] |
| Fruit | Ulcer | Himalaya, India | [69] |
| Parameters | Results | References | |||
|---|---|---|---|---|---|
| Leaves | Bark | Stem Bark | Small Branches | ||
| Extractive value (%w/w) | [8,21,55] | ||||
| Methanolic extract | 28.32 | 38.52 | 23.57 | 5.03 | |
| Ethyl acetate extract | 25.46 | 21.20 | NR | NR | |
| Aqueous extract | 21.28 | 15.7 | 18.36 | 3.52 | |
| Ash Values (%w/w) | [8,21,55] | ||||
| Total ash | 2.83 | 3.3312 | 1.010 | 1.856 | |
| Acid insoluble ash | 0.52 | 1.2300 | 0.187 | 0.320 | |
| Foreign matter (% w/w) | <1% | NR | Nil | Nil | |
| Loss on drying (%w/w) | 5 | 6.47 | 6.81 | ||
| Total phenolics mg of GAE/g d.w. | NR | NR | 276.78 ± 5.36 | 31.24 ± 2.57 | [8] |
| Total flavonoids mg of QE/g d.w. | NR | NR | 121.68 ± 6.81 | 12.94 ± 1.12 | [8] |
| Minerals (mg/g) | Fruit | Stem Bark | Reference |
|---|---|---|---|
| Calcium | 4.63 ± 0.06 | 3.155 ± 0.18 | [72,73,74] |
| Potassium | 7.75 ± 0.11 | 2.939 ± 0.23 | [72,73,74] |
| Magnesium | 8.4 ± 0.20 | 1.061 ± 0.4 | [72,74] |
| Sodium | 0.81 ± 0.013 | 0.060 ± 0.03 | [72,74] |
| Phosphorous | 0.24 ± 0.25 | 0.030 ± 0.01 | [73,74] |
| Manganese | 0.032 ± 0.0001 | NR | [72] |
| Iron | 0.404 ± 0.0021 | 0.123 ± 0.16 | [72,73] |
| Zinc | 0.216 ± 0.0016 | 0.006 ± 0.001 | [72,73] |
| Copper | 0.004 ± 0.0002 | NR | [72] |
| Sulphur | NR | 0.277 ± 0.34 | [73] |
| Extract | Wavelength (nm) | Rf Value | References | ||
|---|---|---|---|---|---|
| Stem Bark | Small Branches | Leaves | |||
| n-hexane | 254 | 0.49, 0.69, 0.88 | 0.49, 0.78 | NR | [8] |
| 366 | 0.42, 0.51, 0.59, 0.74, 0.83,0.91 | 0.42, 0.51, 0.74,0.83,0.91 | |||
| Ethyl acetate | 254 | 0.07, 0.12, 0.36, 0.47, 0.61, 0.67, 0.84 | 0.47, 0.67 | 0.15, 0.6, 0.8 | [8,21] |
| 366 | 0.11, 0.15, 0.18, 0.33, 0.38, 0.55, 0.49, 0.65, 0.75, 0.85, 0.90 | 0.18, 0.30, 0.49, 0.65, 0.75, 0.85, 0.90 | 0.11, 0.22, 0.38, 0.53, 0.69, 0.82, 0.93 | ||
| Ethanol | 254 | 0.23, 0.54 | 0.23, 0.54 | NR | [8] |
| 366 | 0.54, 0.73, 0.84 | 0.25, 0.45, 0.54, 0.73, 0.84 | |||
| Methanol | 254 | NR | NR | 0.625, 0.875 | [21] |
| 366 | 0.46, 0.58, 0.81, 0.86, 0.93 | ||||
| Aqueous | 254 | NR | NR | 0.1, 0.63 | [21] |
| 366 | 0.093, 0.65, 0.81 | ||||
| Part Use | Extract/Fraction | Dose Tested/Route of Administration | Animals/Cell Lines | Experimental Models | Result | Reference |
|---|---|---|---|---|---|---|
| Anti-inflammatory | ||||||
| Leaves | Methanolic | 200 mg/kg, p.o. | Rat | Carrageenan-induced rat paw edema | Significant anti-inflammatory activity | [93] |
| Stem Bark | Essential oil | 10 mL per ear | Swiss albino mice | In vitro [ear] | Significant anti-inflammatory potential | [111] |
| Leaves | ME- EtAC | 100, 200 and 400 mg/kg, p.o. | Wistar rats | Carrageenan-induced rat paw edema | Significant anti-inflammatory activity | [50] |
| Bark | Ethyl acetate and aqueous | 100 and 200 mg/kg, p.o. | Wistar albino rats | Carrageenan and histamine induced rat paw edema | Significant anti-inflammatory potential (EAE> AE) | [102] |
| Antimicrobial | ||||||
| Stem bark | Volatile oil | 10 mL | BP, SA, SE, EC, PA, CA, AN and SC | --- | Significant antimicrobial potential | [111] |
| Bark and fruit | Methanolic and chloroform | --- | --- | Agar Well diffusion method | Significant antimicrobial potential (Bark> Fruits) | [109] |
| Fruit pulp | Ethanolic | 10 and 50 mg/ml | In vitro | Disc diffusion assay | Dose dependent antimicrobial potential | [73] |
| Fruit | Methanolic | 50 μL | SA, SE, BS, PM, EC, SE | Agar Well diffusion method | Significant potential against Pathogens | |
| Antifungal | ||||||
| Fruit | Methanolic, ethanolic and aqueous | 10 and 50mg/ml | Candida albicans, Aspergillus flavus and Aspergillus parasiticus | Disc diffusion assay | Significant antifungal activity | [73] |
| Anthelmintic | ||||||
| Bark | 50% Aqueous Ethanolic | 12.5, 25 and 50 mg/ml | Earthworms (Pheretima posthuma) | --- | Paralysis and death at 12.5 mg.ml | [107] |
| Anticancer | ||||||
| Fruit | Methanol, acid methanol acetone and acidic acetone | 66.7, 166.5, 333, 500, 667 µg f.w./100 µL culture medium | C33A, SiHa and HeLa cell lines | --- | Acetone and acidic acetone extracts showed anticancer potential | [108] |
| Fruit | Methanolic | 5mg/ml | HepG2, Hela and MDA-MB-231 cells | MTT assay | Moderate anticancer activity | [78] |
| Chemopreventive | ||||||
| Bark | Ethanolic | 2.0 mg and 4.0 mg/kg | Swiss albino mice | Cumene hydroperoxide-mediated cutaneous oxidative stress and toxicity | ↑ antioxidant enzymes activity | [113] |
| Antioxidant | ||||||
| Fruit | Methanolic | --- | In vitro | DPPH, ABTS and FRAP assay | Significant antioxidant activity | [78] |
| Fruit pulp | Methanolic | 0.10 ml | In vitro | DPPH, ABTS and FRAP assay | Good scavenging potential | [76] |
| Fruit | Aqueous methanol and acetone | 100 µl | In vitro | DPPH assay | Acetone extract showed higher scavenging potential | [94] |
| Fruit | Methanol, acidic methanol, acetone, and acidic-acetone | --- | In vitro | DPPH, ABTS, FRAP and Superoxide anion radicals scavenging assay | MeAA showed higher antimicrobial potential and MeAM and MeA intermediate | [103] |
| Fruit | Fruit Juice | 0.2–2.0 mg/mL | In vitro | DPPH, H2O2 and NO scavenging activity | Moderate antioxidant activity | [95] |
| Antidiabetic | ||||||
| Leaves | Methanolic | 50,100 and 150 mg/kg, p.o. | Albino wistar rats | STZ induced diabetes | Significant anti-dyslipidemic effect at 150 mg/kg and maintain blood glucose level | [105] |
| Hepatoprotective | ||||||
| Polyherbal formulation (Herbitars) | --- | 50 and 100 mg/kg | Wistar rat | CCl4 induced hepatotoxicity | Extract ↓TBARS, ↑SOD, CAT, GSH | [114] |
| Antidepressant | ||||||
| Bark | Methanolic | 300, 500 mg/kg, p.o. | Albino mice | Open field test, cage-crossing test, head-dip test, rearing test, traction test, forced swimming test | Significant antidepressant activity | [104] |
| Anxiolytic | ||||||
| Bark | Ethanolic | 100, 200 and 400 mg/kg | Rats | Tail suspension test | Significant and dose dependent anxiolytic activity | [61] |
| Forced swimming test | ||||||
| Antihypertensive | ||||||
| Leaves | Methanolic | 100 mM | In vitro | ACE inhibitory activity | Potent ACE inhibition potential | [45] |
| Antiasthmatic | ||||||
| Bark | Ethanolic | 75 mg/kg, p.o. | Guinea pig | Acetylcholine induced bronchospasm | Protection against bronchospasm and anaphylaxis | [98] |
| Bark | Ethanolic | 75 mg/kg, p.o. | In vitro | Guinea pig tracheal strip | ↓pD2 value of histamine and acetylcholine | [98] |
| Stem bark | Ethanolic | 150 mg/kg, p.o. | Guinea pig | Histamine induced bronchospasm | ↓TLC and DLC | [115] |
| Stem Bark | Ethanolic | 75 and 150 mg/kg, p.o. | Mice | Acetic acid induced vascular permeability and allergic pleurisy | [99] | |
| Stem bark | Aqueous extract | 27 & 54mg/kg p.o. | Guinea pig | histamine induced bronchospasm | Significant antiasthamtic potential | [100] |
| In vitro | Guinea pig tracheal chain | |||||
| Bark | Polar, non-polar and methanolic | 200 mg/kg, p.o. | Rat and in vitro | Acetylcholine induced bronchospasm in conscious guinea pigs; acetylcholine induced contraction on isolated guinea pig tracheal chain preparation; compound 48/80 induced mast cell degranulation using rat; and trypsin and egg albumin induced bronchospasm | PE showed higher potential than NPE and ME | [101] |
| Antiulcer | ||||||
| Bark | Ethanolic | 100 and 200 mg/kg | Albino rat | Pyloric ligation induced ulcer | ↓level of GV, FA, LPO and GSH and ↑ CAT, nitrate and MPO↓level of GV, FA, LPO and GSH and ↑ CAT, nitrate and MPO | [112] |
| Antidiarrheal | ||||||
| Leaves | Ethanolic | 250 and 500 mg/kg, p.o. | Mice | Castor-oil induced diarrhea | Significant antidiarrheal activity | [106] |
| Antipruritic | ||||||
| Stem bark | Aqueous | 150 mg/kg | Male mice | Compound 48/80-induced | Significantly decrease in scratching effect | [116] |
| Analgesic | ||||||
| Fruit | Methanolic | 50, 100 mg/kg, p.o. | Mice | Eddy’s hot plate method | Significant analgesic activity | [92] |
| Leaves | ME-EtAC | 100, 200 mg/kg, p.o. | Mice | Acetic acid induced writhing and tail immersion assay | Significant response at 200 mg/kg | [50] |
| Leaves | Methanolic | 200 mg/kg, p.o. | Mice | Acetic acid induced writhing | 54.56% inhibition of writhing | [93] |
| Antipyretic | ||||||
| Fruit | Methanolic | 50 and 100 mg/kg | Mice | Yeast induced pyrexia in mice | Significant antipyretic effect at 100 mg/kg | [92] |
| Wound healing | ||||||
| Bark | Aqueous | Ointment (100 mg/500mm2) | Albino rats | Wound excision and incision | Significant wound healing potential | [59] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kabra, A.; Martins, N.; Sharma, R.; Kabra, R.; Baghel, U.S. Myrica esculenta Buch.-Ham. ex D. Don: A Natural Source for Health Promotion and Disease Prevention. Plants 2019, 8, 149. https://doi.org/10.3390/plants8060149
Kabra A, Martins N, Sharma R, Kabra R, Baghel US. Myrica esculenta Buch.-Ham. ex D. Don: A Natural Source for Health Promotion and Disease Prevention. Plants. 2019; 8(6):149. https://doi.org/10.3390/plants8060149
Chicago/Turabian StyleKabra, Atul, Natália Martins, Rohit Sharma, Ruchika Kabra, and Uttam Singh Baghel. 2019. "Myrica esculenta Buch.-Ham. ex D. Don: A Natural Source for Health Promotion and Disease Prevention" Plants 8, no. 6: 149. https://doi.org/10.3390/plants8060149
APA StyleKabra, A., Martins, N., Sharma, R., Kabra, R., & Baghel, U. S. (2019). Myrica esculenta Buch.-Ham. ex D. Don: A Natural Source for Health Promotion and Disease Prevention. Plants, 8(6), 149. https://doi.org/10.3390/plants8060149







