Silicon Promotes Agronomic Performance in Brassica napus Cultivated under Field Conditions with Two Nitrogen Fertilizer Inputs
Abstract
1. Introduction
2. Materials and Methods
2.1. Site, Climatic Conditions, and Lysimeter Description
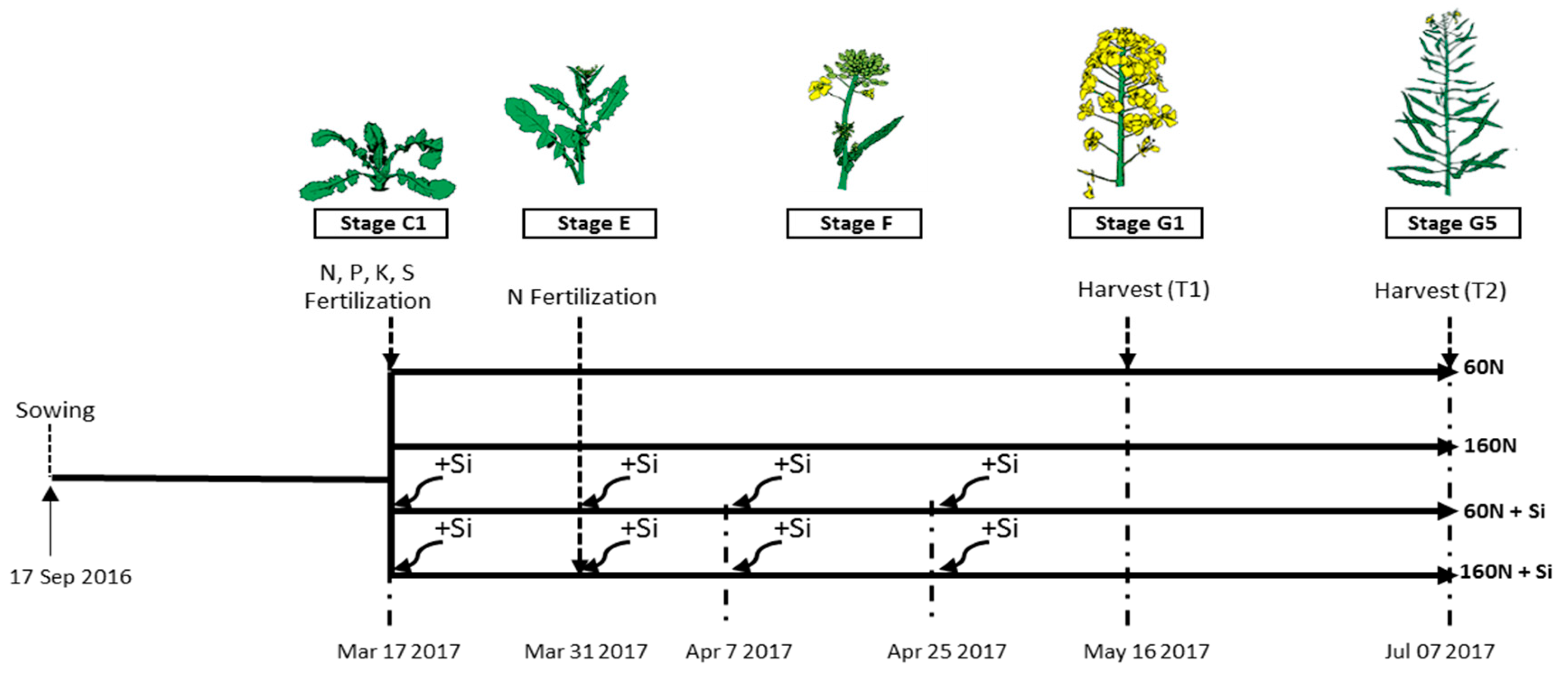
2.2. Experimental Design
2.3. Determination of the Percentages of Green and Senescent Leaves
2.4. Elemental Analysis
2.5. Determination of Oil, Protein, and Glucosinolate Concentrations in Mature Seeds with Near Infrared Spectroscopy (NIRS)
2.6. Determination of Nitrogen Use Efficiency Indexes
2.7. Statistical Analysis
3. Results
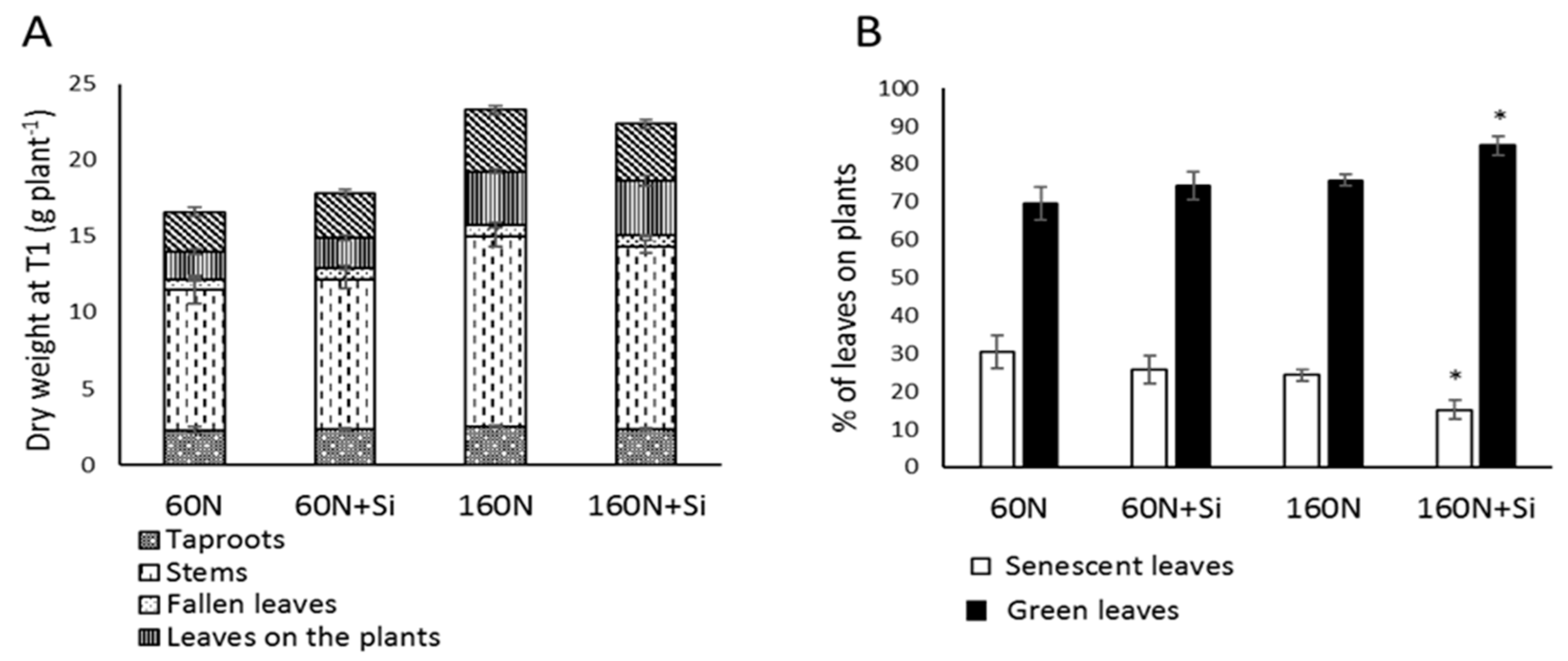
3.1. Effect of Silicon Supply on Plant Growth and Leaf Development
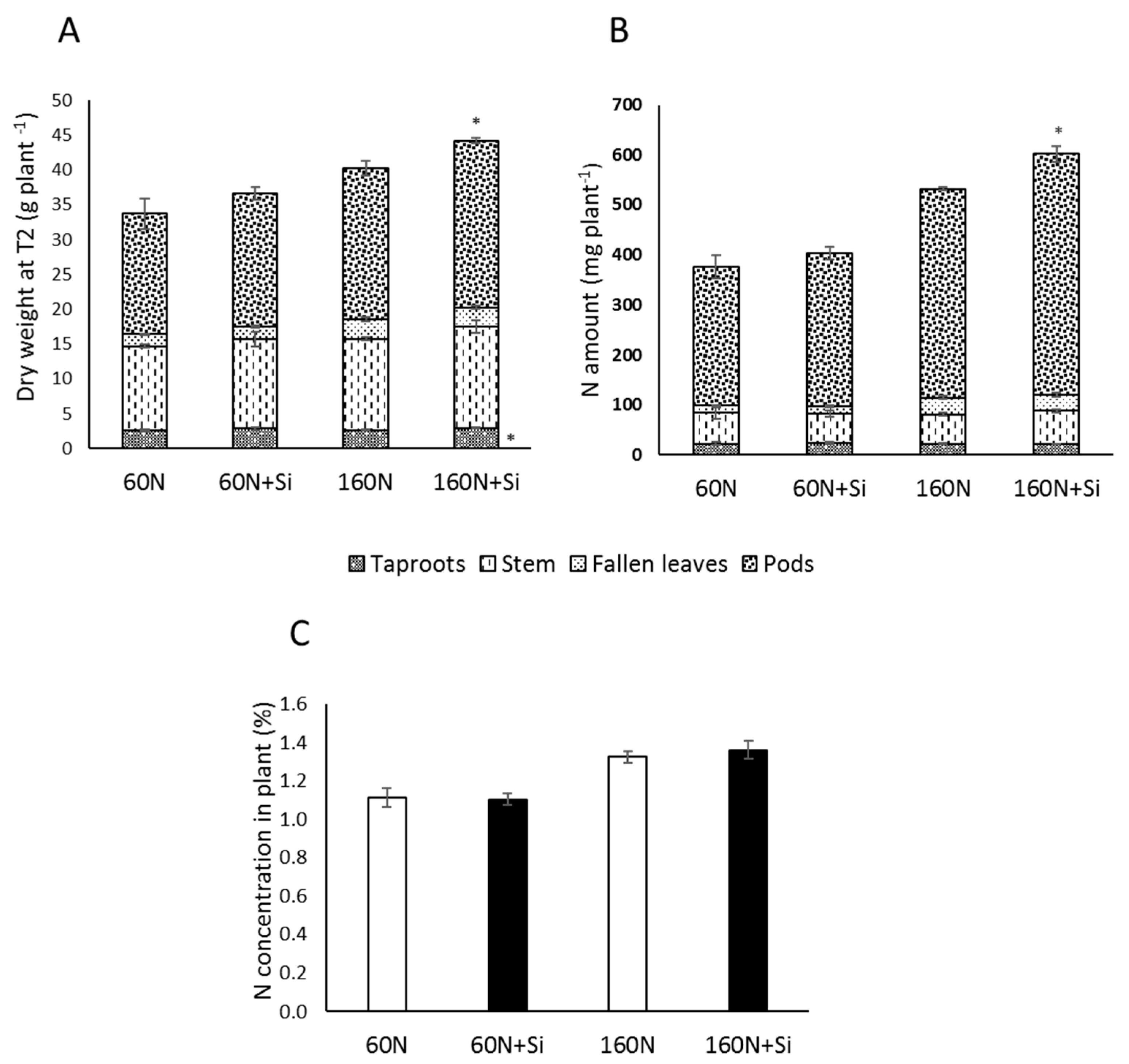
3.2. Effect of Silicon Supply on Yield and Seed Quality Components
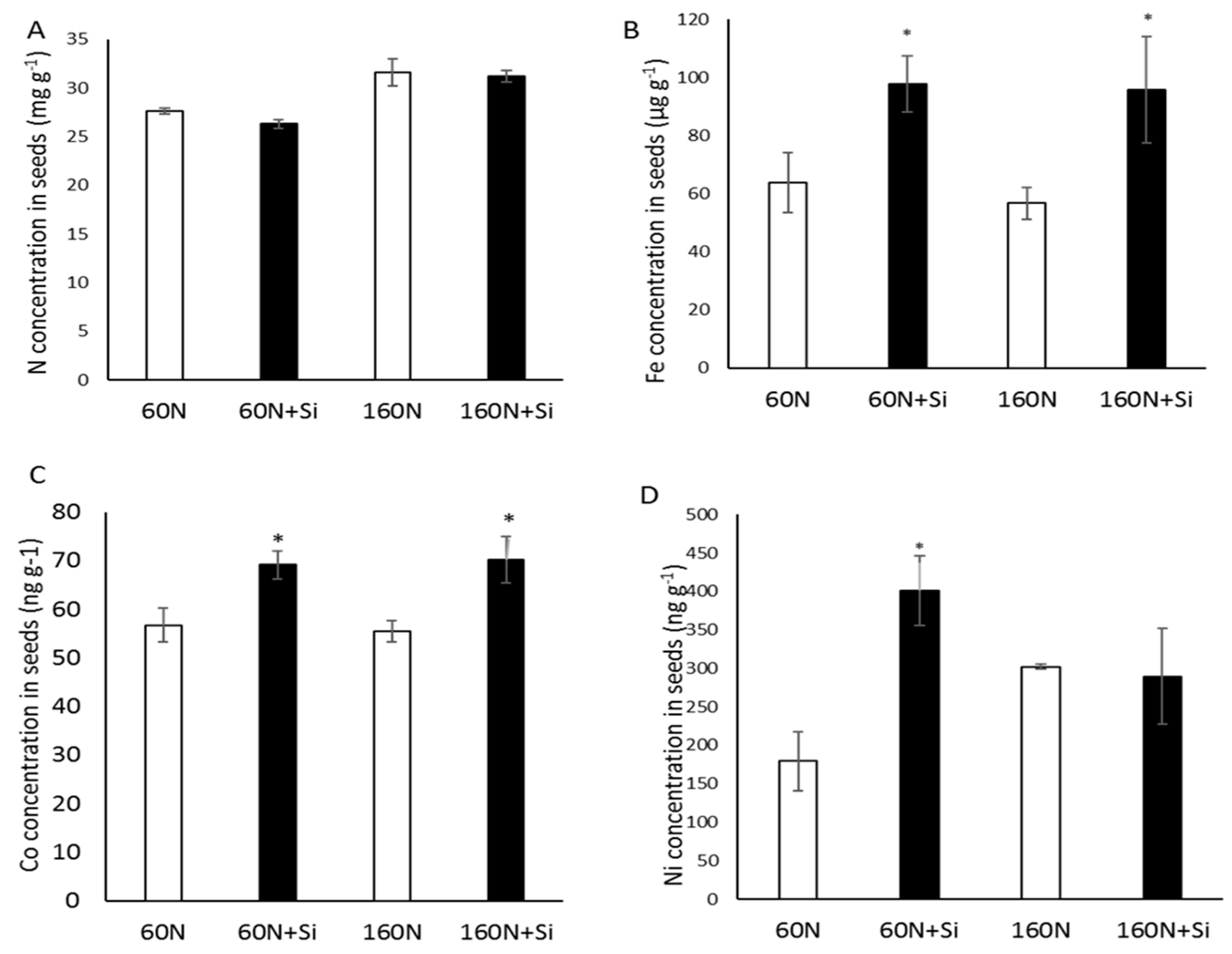
3.3. Effect of Silicon Supply on Elemental Seed Composition
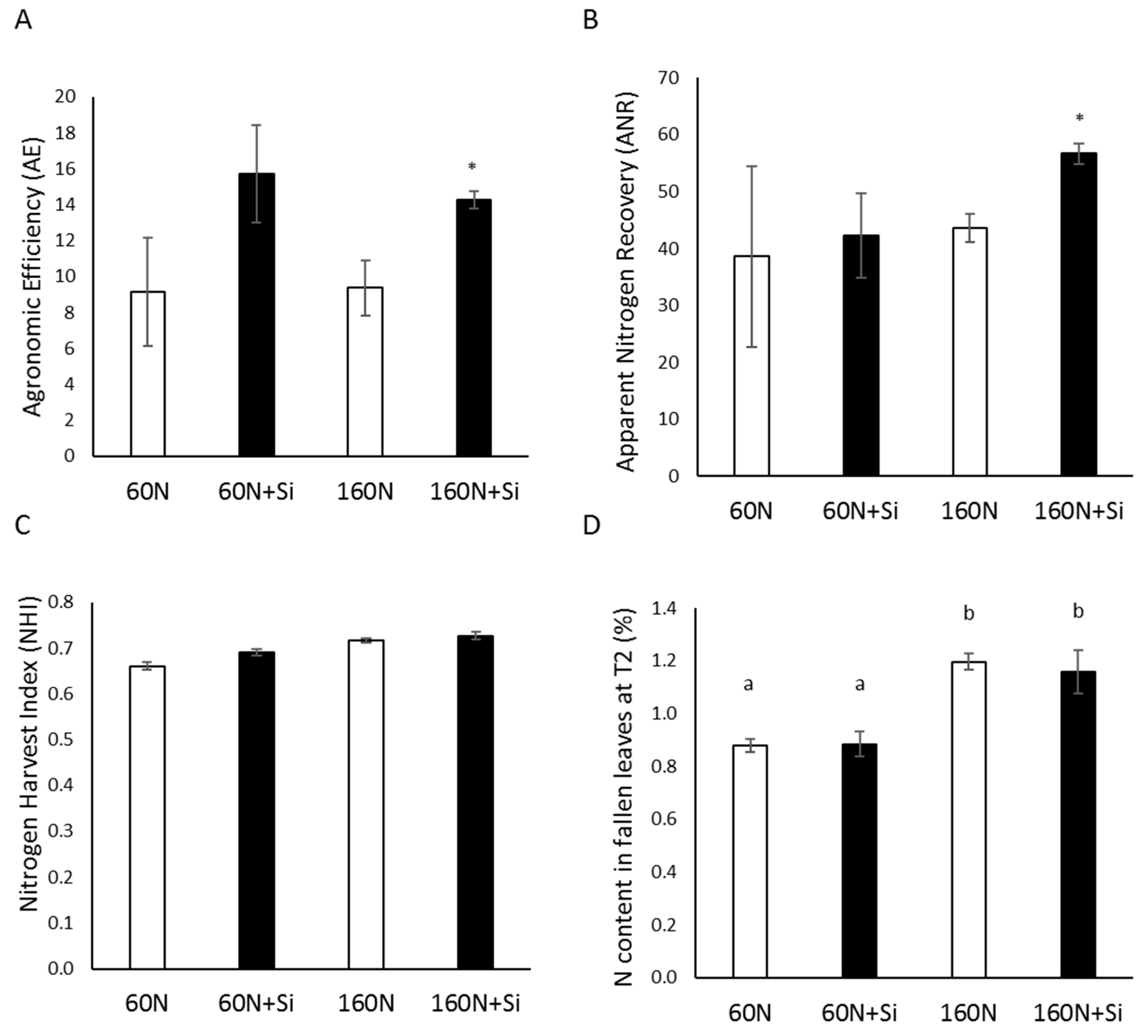
3.4. Effect of Silicon on the Nitrogen Use Efficiency Component of Plants
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ma, J.F.; Yamaji, N. Silicon uptake and accumulation in higher plants. Trends Plant Sci. 2006, 11, 392–397. [Google Scholar] [CrossRef]
- Sommer, M.; Kaczorek, D.; Kuzyakov, Y.; Breuer, T. Silicon pools and fluxes in soils and landscapes: A review. J. Plant Nutr. Soil Sci. 2006, 169, 310–329. [Google Scholar] [CrossRef]
- Epstein, E.; Bloom, A.J. Inorganic components of plants. In Inorganic Components of Plants: Mineral Nutrition of Plants: Principles and Perspectives, 2nd ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2005; pp. 44–45. [Google Scholar]
- Epstein, E. The anomaly of silicon in plant biology. Proc. Natl. Acad. Sci. USA 1994, 91, 11–17. [Google Scholar] [CrossRef]
- Ma, J.F.; Tamai, K.; Yamaji, N.; Mitani, N.; Konishi, S.; Katsuhara, M.; Yano, M. A silicon transporter in rice. Nature 2006, 440, 688–691. [Google Scholar] [CrossRef] [PubMed]
- Sonah, H.; Deshmukh, R.K.; Labbé, C.; Bénlanger, R.R. Analysis of aquaporins in Brassicaceae species reveals high-level of conservation and dynamic role against biotic and abiotic stress in canola. Sci. Rep. 2017, 7, 2771. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.H.P.; Handreck, K.A. Silica in soils, plants, and animals. Adv. Agron. 1967, 19, 107–149. [Google Scholar]
- Epstein, E. Silicon. Ann. Rev. Plant Biol. 1999, 50, 641–664. [Google Scholar] [CrossRef] [PubMed]
- Broadley, M.; Brown, P.; Cakmak, I.; Ma, J.F.; Rengel, Z.; Zhao, F. Beneficial elements. In Mineral Nutrition of Higher Plants, 3rd ed.; Marschner, P., Ed.; Elsevier Science: Amsterdam, The Netherlands, 2011; pp. 257–261. [Google Scholar]
- Haddad, C.; Arkoun, M.; Jamois, F.; Schwarzenberg, A.; Yvin, J.C.; Etienne, P.; Laîné, P. Silicon promotes growth of Brassica napus L. and delays leaf senescence induced by nitrogen starvation. Front. Plant Sci. 2018, 9, 516. [Google Scholar] [CrossRef] [PubMed]
- Zaid, A.; Gul, F.; Ahanger, M.A.; Ahmad, P. Silicon-mediated alleviation of stresses in plants. In Plant Metabolites and Regulation under Environmental Stress, 1st ed.; Ahmad, P., Ahanger, M.A., Singh, V.T., Tripathi, D.K., Alam, P., Alyemeni, M.N., Eds.; Elsevier Academic Press: Amsterdam, The Netherlands, 2018; pp. 377–387. [Google Scholar]
- Ma, J.F.; Miyake, Y.; Takahashi, E. Silicon as a beneficial element for crop plants. Stud. Plant Sci. 2001, 8, 17–39. [Google Scholar]
- Datnoff, L.E.; Deren, C.W.; Snyder, G.H. Silicon fertilization for disease management of rice in Florida. Crop Prot. 1997, 16, 525–531. [Google Scholar] [CrossRef]
- Rodrigues, F.Á.; Benhamou, N.; Datnoff, L.E.; Jones, J.B.; Bélanger, R.R. Ultrastructural and cytochemical aspects of silicon-mediated rice blast resistance. Phytopathology 2003, 93, 535–546. [Google Scholar] [CrossRef]
- Nhan, P.P.; Hai, N.T. Amelioration of aluminum toxicity on OM4900 rice seedlings by sodium silicate. Afr. J. Plant Sci. 2013, 7, 208–212. [Google Scholar] [CrossRef]
- Zhang, S.R.; Li, S.; Ding, X.; Li, F.; Liu, C.; Liao, X.; Wang, R. Silicon mediated the detoxification of Cr on pakchoi (Brassica chinensis L.) in Cr-contaminated soil. Int. J. Food Agric. Environ. 2013, 11, 814–819. [Google Scholar]
- Farshidi, M.; Abdolzadeh, A.; Sadeghipour, H.R. Silicon nutrition alleviates physiological disorders imposed by salinity in hydroponically grown canola (Brassica napus L.) plants. Act. Physiol. Plant 2012, 34, 1779–1788. [Google Scholar] [CrossRef]
- Haddad, C.; Trouverie, J.; Arkoun, M.; Yvin, J.C.; Caïus, J.; Brunaud, V.; Laîné, P.; Etienne, P. Silicon supply affects the root transcriptome of Brassica napus L. Planta 2019, 249, 1645–1651. [Google Scholar] [CrossRef] [PubMed]
- Malagoli, P.; Laîné, P.; Rossato, L.; Ourry, A. Dynamics of nitrogen uptake and mobilization in field-grown winter oilseed rape (Brassica napus) from stem extension to harvest. II. An 15N-labelling based simulation model of N partitioning between vegetative and reproductive tissues. Ann. Bot. 2005, 95, 1187–1198. [Google Scholar] [CrossRef]
- Terre Inovia. Les stades repères du colza. Available online: https://www.terresinovia.fr/-/les-stades-reperes-du-colza (accessed on 25 April 2019).
- Génard, T.; Laîné, P.; Diquélou, S.; Yvin, J.C.; Etienne, P. Impact of modulation of sulfur applications on the agronomic performance of rapeseed-clover mixtures. J. Plant Nutr. Soil Sci. 2017, 180, 676–682. [Google Scholar] [CrossRef]
- Terre Inovia. Fertilisation phosphate et potassique. Available online: https://www.terresinovia.fr/-/colza-fertilisation-phosphatee-et-potassique-a-l-implantation (accessed on 26 April 2019).
- Good, A.G.; Shrawat, A.K.; Muench, D.G. Can less yield more? Is reducing nutrient input into the environment compatible with maintaining crop production? Trends Plant Sci. 2004, 9, 597–605. [Google Scholar] [CrossRef]
- Markovich, O.; Steiner, E.; Kouřil, Š.; Tarkowski, P.; Aharoni, A.; Elbaum, R. Silicon promotes cytokinin biosynthesis and delays senescence in Arabidopsis and Sorghum. Plant Cell Environ. 2017, 40, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Gottardi, S.; Iacuzzo, F.; Tomasi, N.; Cortella, G.; Manzocco, L.; Pinton, R.; Römheld, V.; Mimmo, T.; Scampicchio, M.; Costa, L.D.; et al. Beneficial effects of silicon on hydroponically grown corn salad (Valerianella locusta (L.) Laterr) plants. Plant Physiol. Biochem. 2012, 56, 14–23. [Google Scholar] [CrossRef]
- Matichenkov, V.V.; Bocharnikova, E.A. The Relationship between silicon and soil physical and chemical properties. In Silicon in Agriculture. Studies in Plant Science; Datnoff, L.E., Snyder, G.H., Korndorfer, G.H., Eds.; Elsevier: Amsterdam, The Netherlands, 2001; Volume 8, pp. 209–219. [Google Scholar]
- Siam, H.S.; Abd El-Moez, M.R.; Holah, S.S.; Abou Zeid, S.T. Effect of silicon addition to different fertilizer on yield of rice (Oryza sativa L.) plants.I-Macro nutrients by different rice parts. Middle East J. Appl. Sci. 2018, 8, 177–190. [Google Scholar]
- Rangaraj, S.; Gopalu, K.; Rathinam, Y.; Periasamy, P.; Venkatachalam, R.; Narayanasamy, K. Effect of silica nanoparticles on microbial biomass and silica availability in maize rhizosphere. Biotechnol. Appl. Biochem. 2014, 61, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Greger, M.; Landberg, T.; Vaculík, M. Silicon Influences Soil Availability and Accumulation of Mineral Nutrients in Various Plant Species. Plants 2018, 7, 41. [Google Scholar] [CrossRef] [PubMed]
- Cuong, T.X.; Ullah, H.; Datta, A.; Hanh, T.C. Effects of silicon-based fertilizer on growth, yield and nutrient uptake of rice in tropical zone of Vietnam. Rice Sci. 2017, 24, 283–290. [Google Scholar] [CrossRef]
- Wallace, A. Relationships among nitrogen, silicon, and heavy metal uptake. Soil Sci. 1989, 147, 457–460. [Google Scholar] [CrossRef]
- Wallace, A. Participation of silicon in cation-anion balance as a possible mechanism foe aluminum and iron tolerance in some gramineae. J. Plant Nutr. 1993, 16, 547–553. [Google Scholar] [CrossRef]
- Wallace, A.; Romney, E.M.; Mueller, R.T. Nitrogen-silicon interaction in plants grown in desert soil with nitrogen deficiency. Agron. J. 1976, 68, 529–530. [Google Scholar] [CrossRef]
- Singh, A.K.; Singh, R.; Singh, K. Growth, yield, and economics of rice (Oryza sativa) as influenced by level and time of silicon application. Ind. J. Agron. 2005, 50, 190–193. [Google Scholar]
- Kuai, J.; Sun, Y.; Guo, C.; Zhao, L.; Zuo, Q.; Wu, J.; Zhou, G. Root-applied silicon in the early bud stage increases the rapeseed yield and optimizes the mechanical harvesting characteristics. Field Crops Res. 2017, 200, 88–97. [Google Scholar] [CrossRef]
- Tottey, S.; Block, M.A.; Allen, M.; Westergren, T.; Albrieux, C.; Scheller, H.V.; Merchant, S.; Jensen, P.E. Arabidopsis CHL27, located in both envelope and thylakoid membranes, is required for the synthesis of protochlorophyllide. Proc. Natl. Acad. Sci. USA 2003, 100, 16119–16124. [Google Scholar] [CrossRef]
- Abbaspour, N.; Hurrell, R.; Kelishadi, R. Review on iron and its importance for human health. J. Res. Med. Sci. 2014, 419, 164–174. [Google Scholar]
- Yip, R.; Dallman, P.R. Present knowledge in nutrition. In Iron, 7th ed.; Ziegler, E.E., Filer, L.J., Eds.; ILSI Press: Washington, DC, USA, 1996; Volume 14, pp. 278–292. [Google Scholar]
- McDowell, L.R. Minerals in Animal and Human Nutrition, 2nd ed.; McDowell, L.R., Ed.; Elsevier Science: Amsterdam, The Netherlands, 2003; p. 660. [Google Scholar]
- World Health Organization. The World Health Report: Reducing Risks, Promoting Healthy Life. Home Page. Available online: http://www.who.int/whr/2002/en/ (accessed on 25 April 2019).
- Billard, V.; Etienne, P.; Jannin, L.; Garnica, M.; Cruz, F.; Garcia-Mina, J.M.; Cruz, F.; Yvin, J.C.; Ourry, A. Two biostimulants derived from algae or humic acid induce similar responses in the mineral content and gene expression of winter oilseed rape (Brassica napus L.). J. Plant Growth Reg. 2013, 33, 305–316. [Google Scholar] [CrossRef]
| Depth (cm) | Particle Size Distribution (%) | pH (Water) | Total S (mg S g−1) | Organic C (mg C g−1) | Total N (mg N g−1) | Inorganic N (µg N g−1) | C:N Ratio | CEC (mol kg−1) | CaCO3 (%) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sand | Silt | Clay | |||||||||
| 0–20 | 32 | 38 | 30 | 7.9 | 0.1 | 25.4 | 3.2 | 7.4 | 7.9 | 16.5 | 10 |
| 20–40 | 34 | 38 | 28 | 8.1 | 0.1 | 15.0 | 1.9 | 7.5 | 8.0 | 11.5 | 12 |
| 40–65 | 41 | 35 | 24 | 8.3 | 0.1 | 7.6 | 0.9 | 4.5 | 8.2 | 7.2 | 28 |
| 65–100 | 49 | 34 | 17 | 8.4 | nd | 3.4 | 0.4 | 4.7 | 8.5 | 4.6 | 38 |
| Yield (kg ha−1) | Thousand-Seed Weight (g) | Glucosinolate Concentration (µg g−1 DW) | Oil Concentration (% DW) | ω-6:ω-3 Ratio | Protein Concentration (% DW) | |
|---|---|---|---|---|---|---|
| 60 N | 2696.96 ± 217.90 | 5.27 ± 0.33 | 13.10 ± 0.85 | 49.70 ± 0.96 | 2.37 ± 0.27 | 14.77 ± 0.13 |
| 60 N + Si | 3122.13 ± 74.37 | 4.92 ± 0.21 | 15.43 ± 0.55 | 50.45 ± 0.55 | 2.42 ± 0.13 | 14.58 ± 0.45 |
| 160 N | 3644.37 ± 182.48 | 4.81 ± 0.40 | 17.97 ± 0.66 | 47.4 ± 0.67 | 2.48 ± 0.06 | 16.50 ± 0.10 |
| 160 N + Si | 4247.20 ± 75.23 * | 4.54 ± 0.28 | 16.53 ± 1.54 | 47.93 ± 0.42 | 2.24 ± 0.10 | 17.33 ± 0.26 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laîné, P.; Haddad, C.; Arkoun, M.; Yvin, J.-C.; Etienne, P. Silicon Promotes Agronomic Performance in Brassica napus Cultivated under Field Conditions with Two Nitrogen Fertilizer Inputs. Plants 2019, 8, 137. https://doi.org/10.3390/plants8050137
Laîné P, Haddad C, Arkoun M, Yvin J-C, Etienne P. Silicon Promotes Agronomic Performance in Brassica napus Cultivated under Field Conditions with Two Nitrogen Fertilizer Inputs. Plants. 2019; 8(5):137. https://doi.org/10.3390/plants8050137
Chicago/Turabian StyleLaîné, Philippe, Cylia Haddad, Mustapha Arkoun, Jean-Claude Yvin, and Philippe Etienne. 2019. "Silicon Promotes Agronomic Performance in Brassica napus Cultivated under Field Conditions with Two Nitrogen Fertilizer Inputs" Plants 8, no. 5: 137. https://doi.org/10.3390/plants8050137
APA StyleLaîné, P., Haddad, C., Arkoun, M., Yvin, J.-C., & Etienne, P. (2019). Silicon Promotes Agronomic Performance in Brassica napus Cultivated under Field Conditions with Two Nitrogen Fertilizer Inputs. Plants, 8(5), 137. https://doi.org/10.3390/plants8050137





