Investigating the In Vitro Regeneration Potential of Commercial Cultivars of Brassica
Abstract
1. Introduction
2. Results
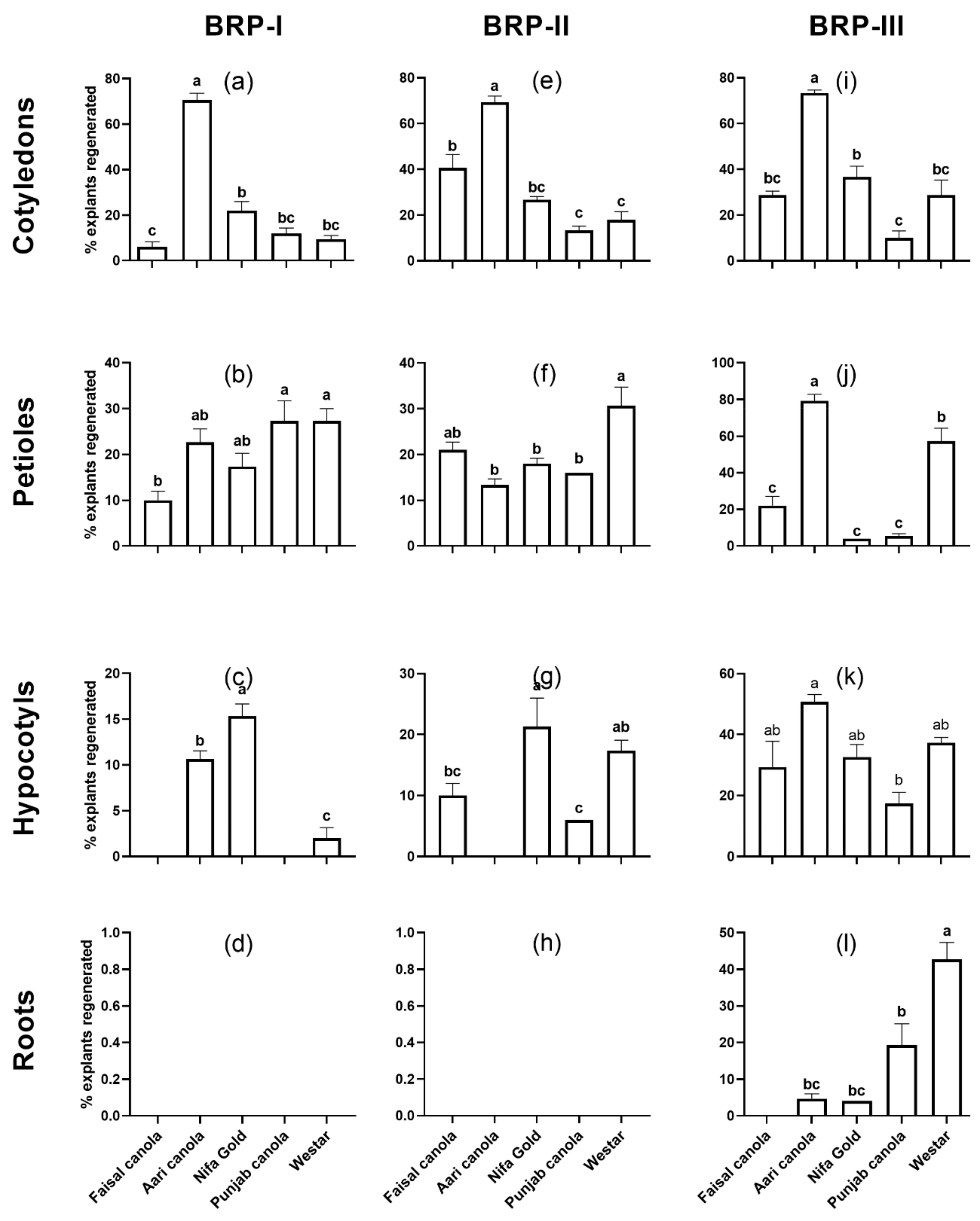
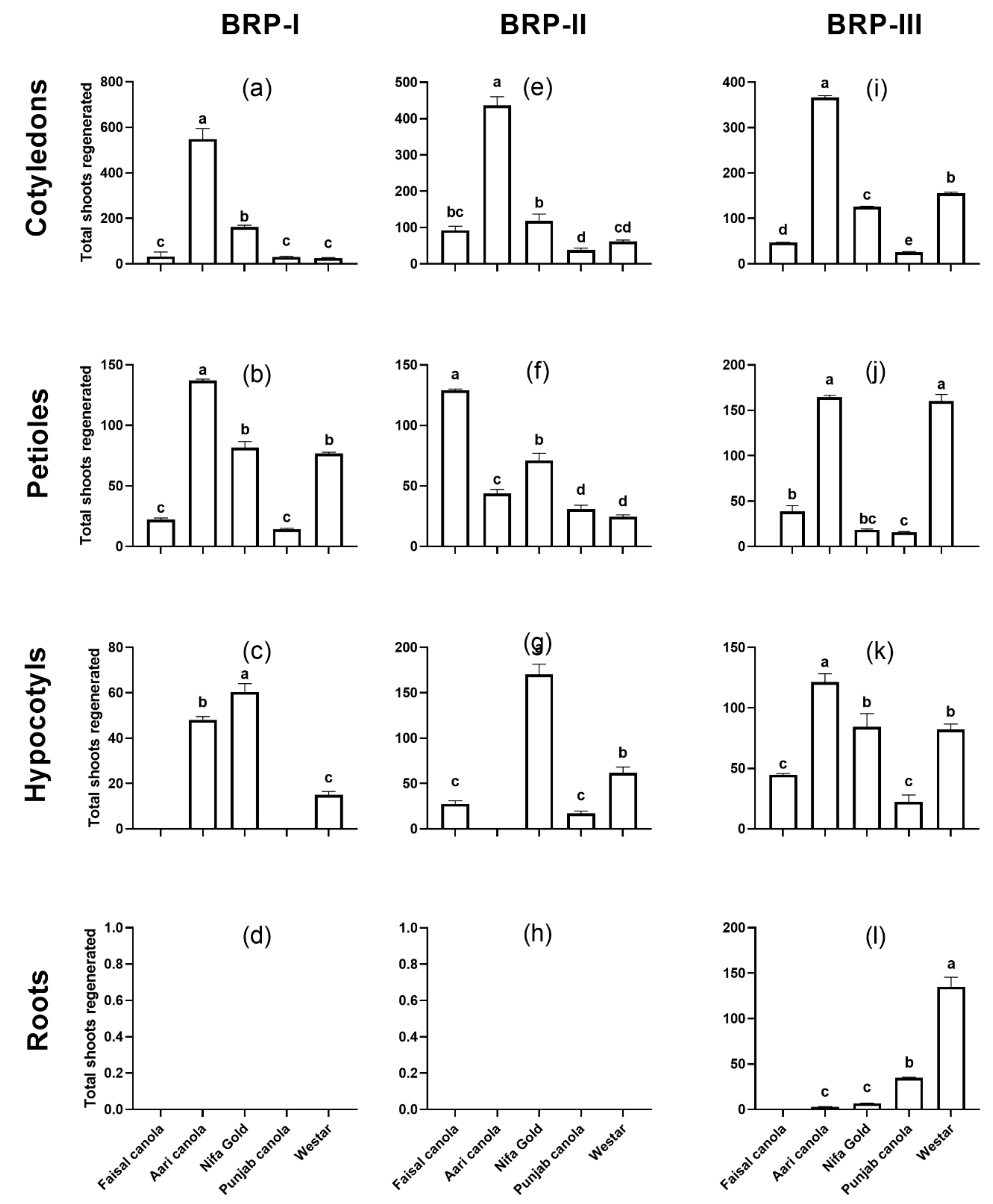
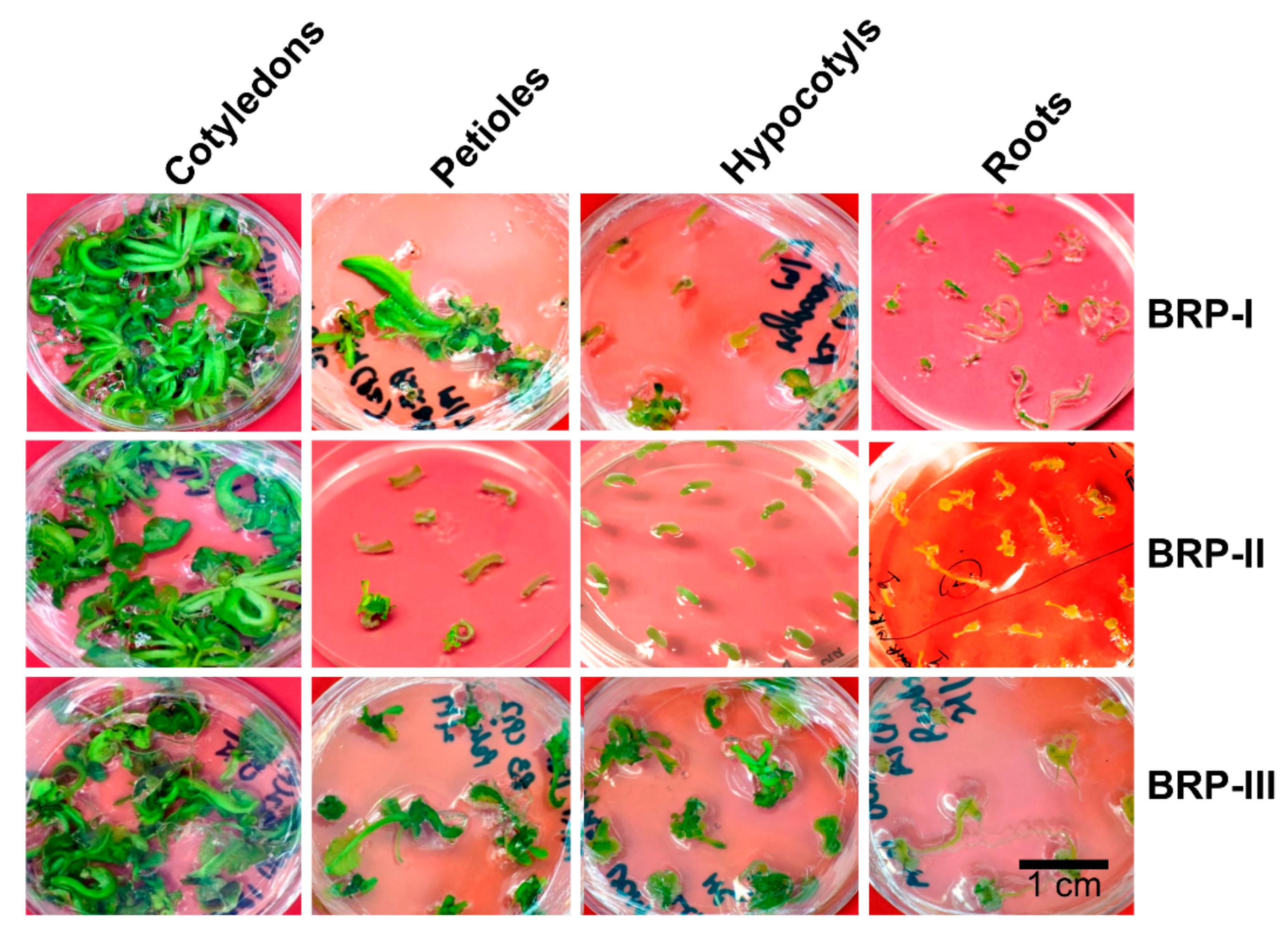
2.1. Shoot Regeneration from Cotyledons
2.2. Shoot Regeneration from Detached Petioles
2.3. Shoot Regeneration from Hypocotyls
2.4. Shoot Regeneration from Roots
2.5. Effect of Explant, Regeneration Conditions, and Their Interaction on In Vitro Regeneration
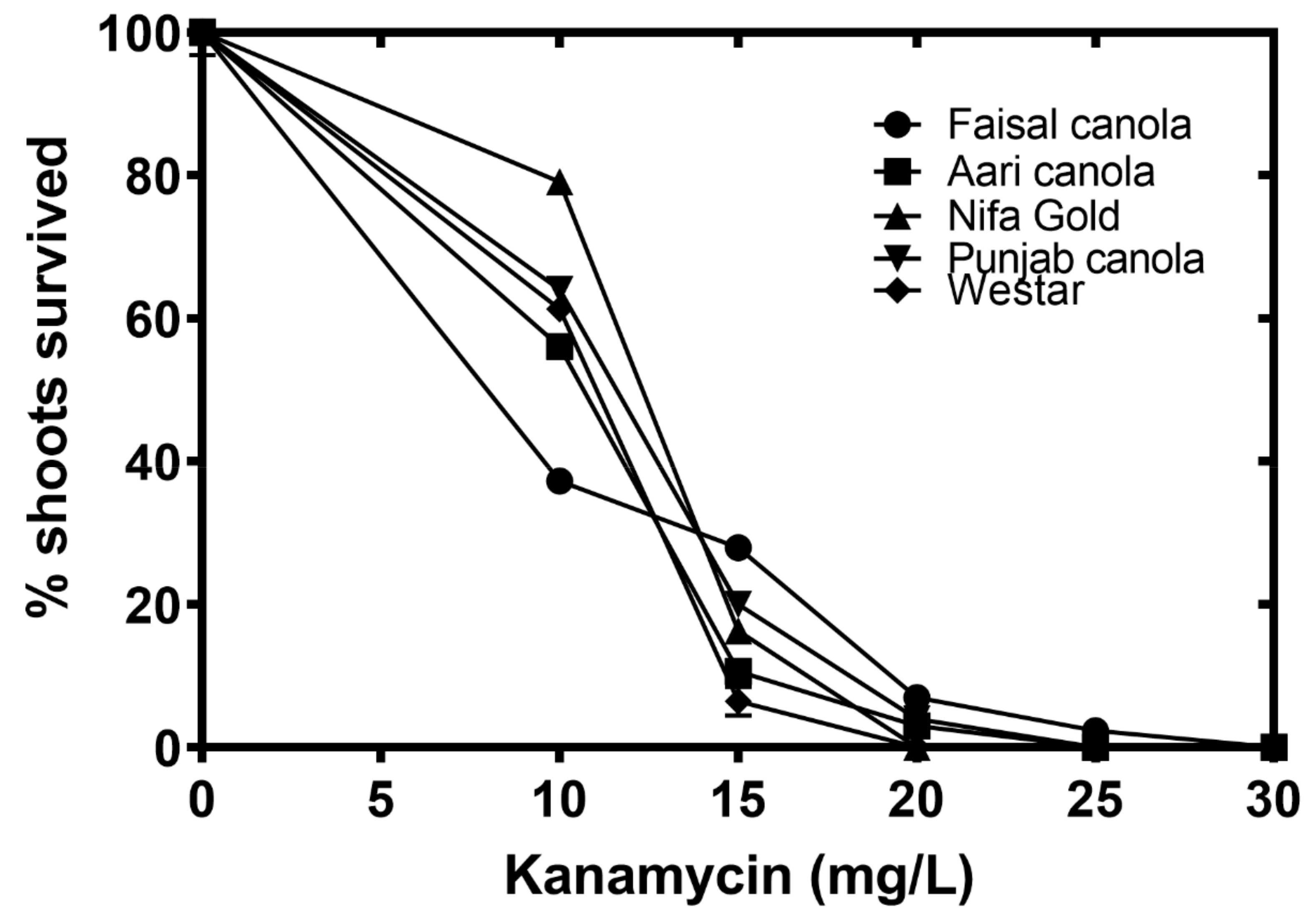
2.6. Determining Kanamycin Sensitivity Levels
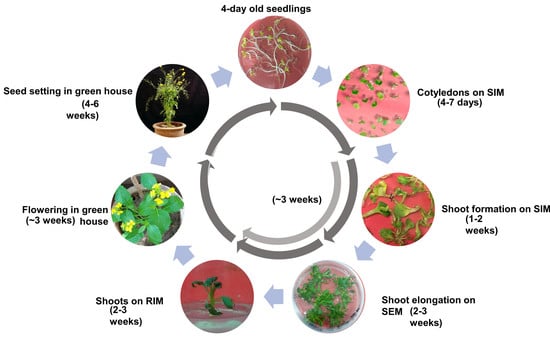
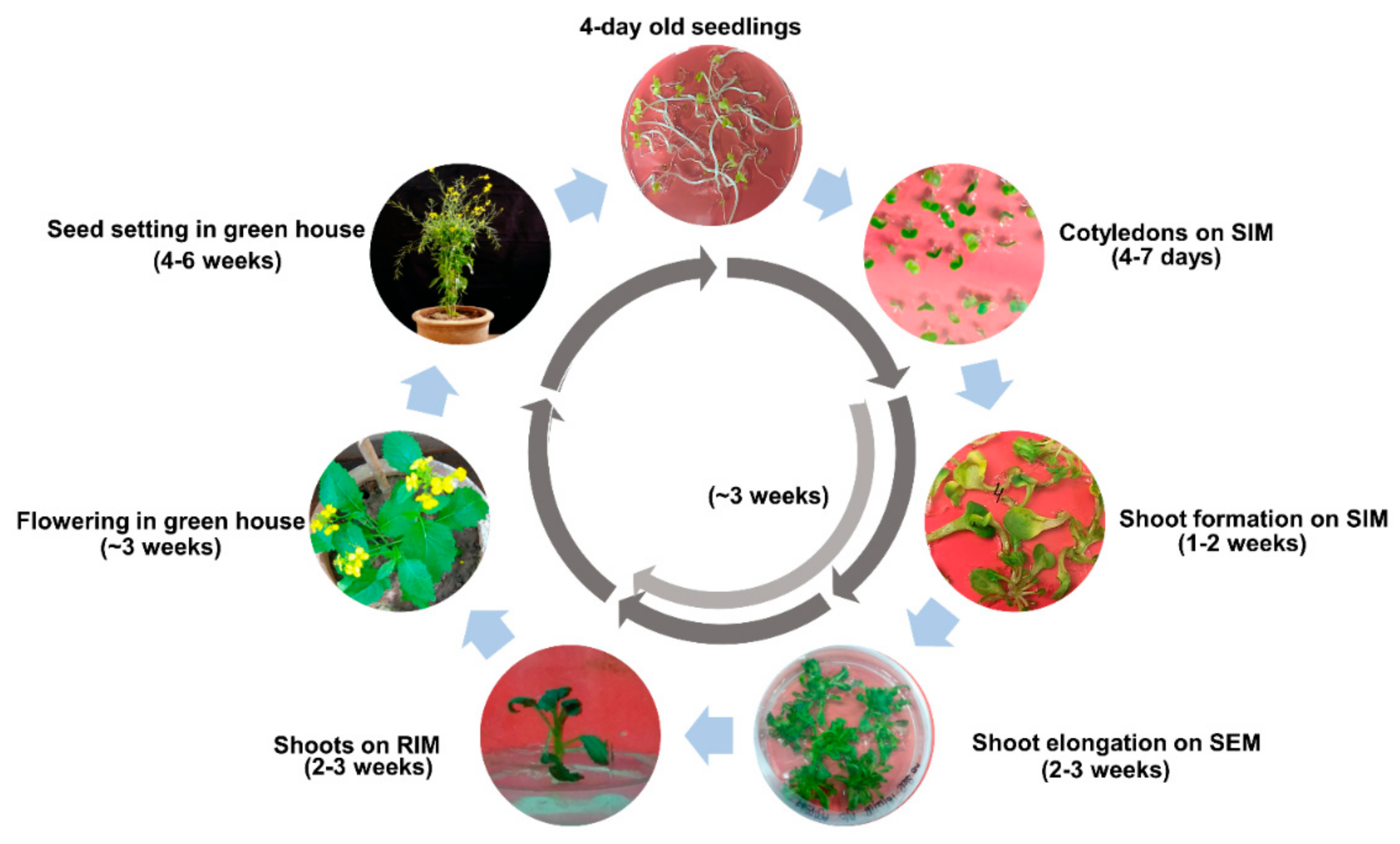
2.7. Completion of Regeneration Cycle for Aari canola
2.8. Rooting Efficiency
3. Discussion
4. Materials and Methods
4.1. Plant Material and Growth Conditions
4.2. Sterilization and Sowing
4.3. Explant Isolation and Shoot Induction
4.4. Kanamycin Selection Levels
4.5. Growing In Vitro Regenerated Shoots to Maturity
4.6. Rooting Efficiency
4.7. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Naz, S.; Siddiquiland, M.F.; Raza, S. Effect of different growth regulators on in vitro propagation of Brassica napus L. Pak. J. Bot. 2018, 50, 1871–1876. [Google Scholar]
- Guoliang, L.; Lixin, Y.; Fei, L.; Zhang, S.; Zhang, H.; Wei, Q.; Zhiyuan, F.; Jian, W.; Xiaowu, W.; Zhang, S. Research progress on Agrobacterium tumefaciens-based transgenic technology in Brassica rapa. Hortic. Plant J. 2018, 4, 126–132. [Google Scholar]
- Gerszberg, A. Tissue culture and genetic transformation of cabbage (Brassica oleracea var. capitata): An overview. Planta 2018, 248, 1037–1048. [Google Scholar] [CrossRef]
- Rani, T.; Yadav, R.C.; Yadav, N.R.; Rani, A.; Singh, D. Genetic transformation in oilseed Brassicas–A review. Indian J. Agric. Sci. 2013, 83, 367–373. [Google Scholar]
- O’Brien, D. Canola: Good protein source for dairy cattle. Agric. Res. 2016, 64, 1–3. [Google Scholar]
- Wickramasuriya, S.S.; Yi, Y.-J.; Yoo, J.; Kang, N.K.; Heo, J.M. A review of canola meal as an alternative feed ingredient for ducks. J. Anim. Sci. Technol. 2015, 57, 29. [Google Scholar] [CrossRef]
- Government of Pakistan, E.A.W. Pakistan Economic Survey 2016–17–Agriculture; Division, F., Ed.; Ministry of Finance: Islamabad, Pakistan, 2017.
- Christou, P. Plant genetic engineering and agricultural biotechnology 1983–2013. Trends Biotechnol. 2013, 31, 125–127. [Google Scholar] [CrossRef]
- Ahmad, N.; Mukhtar, Z. Genetic manipulations in crops: Challenges and opportunities. Genomics 2017, 109, 494–505. [Google Scholar] [CrossRef]
- Baltes, N.J.; Gil-Humanes, J.; Voytas, D.F. Genome engineering and agriculture: Opportunities and challenges. In Progress in Molecular Biology and Translational Science; Weeks, D.P., Yang, B., Eds.; Academic Press, Elsevier: Cambridge, Ma, USA, 2017; Volume 149, pp. 1–26. [Google Scholar]
- Paul, M.J.; Nuccio, M.L.; Basu, S.S. Are GM crops for yield and resilience possible? Trends Plant Sci. 2017, 23, 10–16. [Google Scholar] [CrossRef]
- Tyczewska, A.; Woźniak, E.; Gracz, J.; Kuczyński, J.; Twardowski, T. Towards food security: Current state and future prospects of agrobiotechnology. Trends Biotechnol. 2018, 36, 1219–1229. [Google Scholar] [CrossRef]
- Augustine, R.; Bisht, N.C. Targeted silencing of genes in polyploids: Lessons learned from Brassica juncea-glucosinolate system. Plant Cell Rep. 2018. [Google Scholar] [CrossRef]
- Nour-Eldin, H.H.; Madsen, S.R.; Engelen, S.; Jørgensen, M.E.; Olsen, C.E.; Andersen, J.S.; Seynnaeve, D.; Verhoye, T.; Fulawka, R.; Denolf, P. Reduction of antinutritional glucosinolates in Brassica oilseeds by mutation of genes encoding transporters. Nat. Biotechnol. 2017, 35, 377–382. [Google Scholar] [CrossRef]
- Liang, Y.; Xiong, Z.; Zheng, J.; Xu, D.; Zhu, Z.; Xiang, J.; Gan, J.; Raboanatahiry, N.; Yin, Y.; Li, M. Genome-wide identification, structural analysis and new insights into late embryogenesis abundant (LEA) gene family formation pattern in Brassica napus. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Maheshwari, P.; Selvaraj, G.; Kovalchuk, I. Optimization of Brassica napus (canola) explant regeneration for genetic transformation. Nat. Biotechnol. 2011, 29, 144–155. [Google Scholar] [CrossRef]
- Cheng, L.; Li, H.-P.; Qu, B.; Huang, T.; Tu, J.-X.; Fu, T.-D.; Liao, Y.-C. Chloroplast transformation of rapeseed (Brassica napus) by particle bombardment of cotyledons. Plant Cell Rep. 2010, 29, 371–381. [Google Scholar] [CrossRef]
- Liu, C.W.; Lin, C.C.; Yiu, J.C.; Chen, J.J.; Tseng, M.J. Expression of a Bacillus thuringiensis toxin (Cry1Ab) gene in cabbage (Brassica oleracea L. var. capitata L.) chloroplasts confers high insecticidal efficacy against Plutella xylostella. Theor. Appl. Genet. 2008, 117, 75–88. [Google Scholar] [CrossRef]
- Bhalla, P.L.; Singh, M.B. Agrobacterium-mediated transformation of Brassica napus and Brassica oleracea. Nat. Protoc. 2008, 3, 181–189. [Google Scholar] [CrossRef]
- Liu, C.W.; Lin, C.C.; Chen, J.J.; Tseng, M.J. Stable chloroplast transformation in cabbage (Brassica oleracea L. var. capitata L.) by particle bombardment. Plant Cell Rep. 2007, 26, 1733–1744. [Google Scholar] [CrossRef]
- Skarjinskaia, M.; Svab, Z.; Maliga, P. Plastid transformation in Lesquerella fendleri, an oilseed Brassicacea. Transgenic Res. 2003, 12, 115–122. [Google Scholar] [CrossRef]
- Sparrow, P.; Dale, P.; Irwin, J. The use of phenotypic markers to identify Brassica oleracea genotypes for routine high-throughput Agrobacterium-mediated transformation. Plant Cell Rep. 2004, 23, 64–70. [Google Scholar] [CrossRef]
- Hundleby, P.A.; Irwin, J.A. Brassica oleracea and B. napus. Methods Mol. Biol. 2015, 1223, 287–297. [Google Scholar] [CrossRef]
- Sparrow, P.; Snape, J.; Dale, P.; Irwin, J. The rapid identification of B. napus genotypes, for high-throughput transformation, using phenotypic tissue culture markers. Acta Hortic. 2006, 706, 239–246. [Google Scholar] [CrossRef]
- Sparrow, P.; Townsend, T.; Arthur, A.; Dale, P.; Irwin, J. Genetic analysis of Agrobacterium tumefaciens susceptibility in Brassica oleracea. Theor. Appl. Genet. 2004, 108, 644–650. [Google Scholar] [CrossRef]
- Sparrow, P.; Townsend, T.; Morgan, C.; Dale, P.; Arthur, A.; Irwin, J. Genetic analysis of in vitro shoot regeneration from cotyledonary petioles of Brassica oleracea. Theor. Appl. Genet. 2004, 108, 1249–1255. [Google Scholar] [CrossRef]
- Khehra, G.; Mathias, R. The interaction of genotype, explant and media on the regeneration of shoots from complex explants of Brassica napus L. J. Exp. Bot. 1992, 43, 1413–1418. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Arumugam, N.; Nandakumar, P.B.A.; Pradhan, A.K.; Gupta, V.; Pental, D. Agrobacterium-mediated genetic transformation of oilseed Brassica campestris: Transformation frequency is strongly influenced by the mode of shoot regeneration. Plant Cell Rep. 1992, 11, 506–513. [Google Scholar] [CrossRef]
- Zhang, Y.; Singh, M.B.; Swoboda, I.; Bhalla, P.L. Agrobacterium-mediated transformation and generation of male sterile lines of Australian canola. Aust. J. Agric. Res. 2005, 56, 353–361. [Google Scholar] [CrossRef]
- Das, B.; Goswami, L.; Ray, S.; Ghosh, S.; Bhattacharyya, S.; Das, S.; Majumder, A.L. Agrobacterium-mediated transformation of Brassica juncea with a cyanobacterial (Synechocystis PCC6803) delta-6 desaturase gene leads to production of gamma-linolenic acid. Plant Cell Tissue Organ Cult. 2006, 86, 219–231. [Google Scholar] [CrossRef]
- Dutta, I.; Saha, P.; Das, S. Efficient Agrobacterium-mediated genetic transformation of oilseed mustard [Brassica juncea (L.) Czern.] using leaf piece explants. In Vitro Cell. Dev. Biol.-Plant 2008, 44, 401–411. [Google Scholar] [CrossRef]
- Baskar, V.; Gangadhar, B.H.; Park, S.W.; Nile, S.H. A simple and efficient Agrobacterium tumefaciens-mediated plant transformation of Brassica rapa ssp. pekinensis. 3 Biotech 2016, 6, 88. [Google Scholar] [CrossRef]
- Daud, N.; Hasbullah, N.; Azis, N.; Rasad, F.; Amin, M.; Lassim, M. In vitro regeneration of Brassica oleracea var. capitata trough stems, roots, leaves and petioles cultures. In Proceedings of the International Conference on Agricultural, Ecological and Medical Sciences (AEMS-2015), Phuket, Thailand, 7–8 April 2015; pp. 7–8. [Google Scholar]
- Zhang, Y.; Bhalla, P.L. In vitro shoot regeneration from commercial cultivars of Australian canola (Brassica napus L.). Aust. J. Agric. Res. 2004, 55, 753–756. [Google Scholar] [CrossRef]
- Darçın, E.S.; Kolsarıcı, Ö.; Yıldız, M. Establishment of efficient regeneration protocol for three rapeseed cultivars. Biotechnol. Biotechnol. Equip. 2014, 28, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Srivastava, D. High frequency organogenesis in hypocotyl, cotyledon, leaf and petiole explants of broccoli (Brassica oleracea L. var. italica), an important vegetable crop. Physiol. Mol. Biol. Plants 2015, 21, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Gambhir, G.; Kumar, P.; Srivastava, D. High frequency regeneration of plants from cotyledon and hypocotyl cultures in Brassica oleracea cv. Pride of India. Biotechnol. Rep. 2017, 15, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, B.; Shariatpanahi, M.E. Proline and chitosan enhanced efficiency of microspore embryogenesis induction and plantlet regeneration in Brassica napus L. Plant Cell Tissue Organ Cult. 2015, 123, 57–65. [Google Scholar] [CrossRef]
- Chikkala, V.R.; Nugent, G.D.; Dix, P.J.; Stevenson, T.W. Regeneration from leaf explants and protoplasts of Brassica oleracea var. botrytis (cauliflower). Sci. Hortic. 2009, 119, 330–334. [Google Scholar] [CrossRef]
- Khan, M.; Robin, A.A.H.K.; Nazim-Ud-Dowla, M.; Talukder, S.; Hassan, L. In vitro regeneration potentiality of Brassica genotypes in differential growth regulators. Bangladesh J. Agric. Res. 2010, 35, 189–199. [Google Scholar] [CrossRef]
- Cao, J.; Earle, E. Transgene expression in broccoli (Brassicaoleracea var. italica) clones propagated in vitro via leaf explants. Plant Cell Rep. 2003, 21, 789–796. [Google Scholar]
- Ono, Y.; Takahata, Y.; Kaizuma, N. Effect of genotype on shoot regeneration from cotyledonary explants of rapeseed (Brassica napus L.). Plant Cell Rep. 1994, 14, 13–17. [Google Scholar] [CrossRef]
- Jonoubi, P.; Mousavi, A.; Majd, A.; Salmanian, A.; Javaran, M.J.; Daneshian, J. Efficient regeneration of Brassica napus L. hypocotyls and genetic transformation by Agrobacterium tumefaciens. Biol. Plant. 2005, 49, 175–180. [Google Scholar] [CrossRef]
- Tang, G.; Zhou, W.; Li, H.; Mao, B.; He, Z.; Yoneyama, K. Medium, explant and genotype factors influencing shoot regeneration in oilseed Brassica spp. J. Agron. Crop Sci. 2003, 189, 351–358. [Google Scholar] [CrossRef]
- Ċosiċ, T.; Motyka, V.; Raspor, M.; Savić, J.; Cingel, A.; Vinterhalter, B.; Vinterhalter, D.; Trávníčková, A.; Dobrev, P.I.; Bohanec, B. In vitro shoot organogenesis and comparative analysis of endogenous phytohormones in kohlrabi (Brassica oleracea var. gongylodes): Effects of genotype, explant type and applied cytokinins. Plant Cell Tissue Organ Cult. 2015, 121, 741–760. [Google Scholar] [CrossRef]
- Gerszberg, A.; Hnatuszko-Konka, K.; Kowalczyk, T. In vitro regeneration of eight cultivars of Brassica oleracea var. capitata. In Vitro Cell. Dev. Biol.-Plant 2015, 51, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Uliaie, E.D.; Farsi, M.; Ghreyazie, B.; Imani, J. Effects of genotype and AgNO3 on shoot regeneration in winter cultivars of rapeseed (Brassica napus). Pak. J. Biol. Sci. 2008, 11, 2040–2043. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hachey, J.E.; Sharma, K.K.; Moloney, M.M. Efficient shoot regeneration of Brassica campestris using cotyledon explants cultured in vitro. Plant Cell Rep. 1991, 9, 549–554. [Google Scholar] [CrossRef]
- Bano, R.; Khan, M.H.; Khan, R.S.; Rashid, H.; Swati, Z.A. Development of an efficient regeneration protocol for three genotypes of Brassica juncea. Pak. J. Bot. 2010, 42, 963–969. [Google Scholar]
- Zhao, S.; Lei, J.; Chen, G.; Cao, B. Application of kanamycin in transgenic mustard (Brassica juncea Coss.). Yi Chuan Hered. 2008, 30, 501–507. [Google Scholar] [CrossRef]
- Mahmood, T.; Hussain, M.; Mustafa, H.; Hasan, E.; Aftab, M. Aari canola: Pakistan’s first ever canola quality and short duration mustard (Brassica juncea L.) cultivar resilient to climate change. Int. J. Biol. Pharm. Al. Sci 2017, 6, 777–787. [Google Scholar]
- Ribaut, J.-M.; Hoisington, D. Marker-assisted selection: New tools and strategies. Trends Plant Sci. 1998, 3, 236–239. [Google Scholar] [CrossRef]
- Lu, X.-M.; Yin, W.-B.; Hu, Z.-M. Chloroplast transformation. In Plant Cell Culture Protocols; Springer: Berlin, Germany, 2006; pp. 285–303. [Google Scholar]
- Díaz, A.H. Regeneration and plastid transformation approaches in Arabidopsis thaliana and Rapid-Cycling Brassica rapa; Ludwig Maximilian University of Munich: Munich, Germany, 2011. [Google Scholar]
- Cogbill, S.; Faulcon, T.; Jones, G.; McDaniel, M.; Harmon, G.; Blackmon, R.; Young, M. Adventitious shoot regeneration from cotyledonary explants of rapid-cycling fast plants of Brassica rapa L. Plant Cell Tiss. Org. Cult. 2010, 101, 127–133. [Google Scholar] [CrossRef]
- Khan, M.R.; Rashid, H.; Ansar, M.; Chaudry, Z. High frequency shoot regeneration and Agrobacterium-mediated DNA transfer in Canola (Brassica napus). Plant Cell Tiss. Org. Cult. 2003, 75, 223–231. [Google Scholar] [CrossRef]
- Khan, M.R.; Rashid, H.; Quraishi, A. High frequency shoot regeneration from hypocotyl of canola (Brassica napus L.) cv. Dunkled. Plant Tissue Cult 2002, 12, 131–138. [Google Scholar]
- Zhang, Y.; Bhalla, P. Shoot regeneration potential from seedling explants of Australian cultivars of oil seed rape (Brassica napus L.). In Proceedings of the New Horizons for an Old Crop, 10th International Rapeseed Congress, Canberra, Australia, 26–29 September 1999. [Google Scholar]
- Damgaard, O.; Rasmussen, O. Direct regeneration of transformed shoots in Brassica napus from hypocotyl infections with Agrobacterium rhizogenes. Plant Mol. Biol. 1991, 17, 1–8. [Google Scholar] [CrossRef]
- Barsby, T.L.; Yarrow, S.A.; Shepard, J.F. A rapid and efficient alternative procedure for the regeneration of plants from hypocotyl protoplasts of Brassica napus. Plant Cell Rep. 1986, 5, 101–103. [Google Scholar] [CrossRef] [PubMed]
| Source of Variation | DF | Faisal Canola | Aari Canola | Nifa Gold | Punjab Canola | Westar |
|---|---|---|---|---|---|---|
| Explant (E) | 3 | 6552 **** | 368,487 **** | 26,280 *** | 6644 **** | 10,035 **** |
| Regeneration Conditions (RC) | 2 | 8297 *** | 11,116 *** | 1026 NS | 8261 **** | 35,692 ** |
| Interaction (E x RC) | 6 | 2540 **** | 9938 **** | 1762 NS | 3202 **** | 2323 ** |
| Replicate | 8 | 115.9 NS | 795.6 NS | 891.1 NS | 318.7 *** | 302.9 NS |
| Residual Error | 16 | 236.1 | 596.7 | 769.1 | 180.1 | 432.5 |
| Source of Variation | Percent Contribution of a Treatment to the Total Experimental Variations | ||||
|---|---|---|---|---|---|
| Faisal canola | Aari canola | Nifa Gold | Punjab canola | Westar | |
| Explant Type (E) | 34.98 | 91.87 | 71.09 | 32.62 | 24.13 |
| Regeneration Conditions (RC) | 29.53 | 1.848 | 1.850 | 27.04 | 57.21 |
| Interaction (E x RC) | 27.12 | 4.956 | 9.533 | 31.44 | 11.17 |
| Replicate | 1.650 | 0.529 | 6.428 | 4.173 | 1.942 |
| Residual Error | 6.720 | 0.797 | 11.09 | 4.727 | 5.548 |
| Total | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Sr. No. | Cultivar | Species | Source |
|---|---|---|---|
| 1 | Aari canola | Brassica juncea | Oilseed Research Institute, Ayyub Agriculture Research Institute (AARI), Faisalabad, Pakistan |
| 2 | Faisal canola | Brassica napus | Oilseed Research Institute, Ayyub Agriculture Research Institute (AARI), Faisalabad, Pakistan |
| 3 | Punjab canola | Brassica napus | Oilseed Research Institute, Ayyub Agriculture Research Institute (AARI), Faisalabad, Pakistan |
| 4 | Nifa Gold | Brassica napus | Nuclear Institute for Food and Agriculture, Peshawar, Pakistan |
| 5 | Westar | Brassica napus | National Oilseed Development Program (NODP), National Agricultural Research Centre (NARC), Islamabad, Pakistan |
| Reagents | BRP-I | BRP-II | BRP-III | ||||||
|---|---|---|---|---|---|---|---|---|---|
| SIM | GRM | CIM | SIM | RIM | CIM | SIM | SOM | RIM | |
| MS salts (mg/L) | 4.43 | 4.43 | 4.43 | 4.43 | 4.43 | 4.43 | 4.43 | 2.215 | |
| Gamborg’s salts (mg/L) | 3.1 | ||||||||
| Sucrose (mg/L) | 30 | 10 | 30 | 30 | 30 | 20 | 20 | 20 | 10 |
| Vitamin Stock (ml/L) | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||
| Phytagel (g/L) | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 |
| BAP (mg/L) | 2 | 6 | 1 | 0.75 | 3 | 0.00125 | |||
| NAA (mg/L) | 0.1 | 0.2 | 0.2 | ||||||
| IBA (mg/L) | 1 | ||||||||
| AgNO3 (mg/L) | 5 | 5 | |||||||
| GA3 (mg/L) | 0.01 | 0.01 | |||||||
| CaCl2 (mg/L) | 435 | 435 | 435 | ||||||
| KI (mg/L) | 0.75 | 0.75 | 0.75 | 0.375 | |||||
| Adenine hemisulfate (mg/L) | 40 | ||||||||
| PVP 40,000 (mg/L) | 500 | ||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farooq, N.; Nawaz, M.A.; Mukhtar, Z.; Ali, I.; Hundleby, P.; Ahmad, N. Investigating the In Vitro Regeneration Potential of Commercial Cultivars of Brassica. Plants 2019, 8, 558. https://doi.org/10.3390/plants8120558
Farooq N, Nawaz MA, Mukhtar Z, Ali I, Hundleby P, Ahmad N. Investigating the In Vitro Regeneration Potential of Commercial Cultivars of Brassica. Plants. 2019; 8(12):558. https://doi.org/10.3390/plants8120558
Chicago/Turabian StyleFarooq, Nisma, Muhammad Asif Nawaz, Zahid Mukhtar, Iftikhar Ali, Penny Hundleby, and Niaz Ahmad. 2019. "Investigating the In Vitro Regeneration Potential of Commercial Cultivars of Brassica" Plants 8, no. 12: 558. https://doi.org/10.3390/plants8120558
APA StyleFarooq, N., Nawaz, M. A., Mukhtar, Z., Ali, I., Hundleby, P., & Ahmad, N. (2019). Investigating the In Vitro Regeneration Potential of Commercial Cultivars of Brassica. Plants, 8(12), 558. https://doi.org/10.3390/plants8120558