Glyphosate: Its Environmental Persistence and Impact on Crop Health and Nutrition
Abstract
1. Introduction
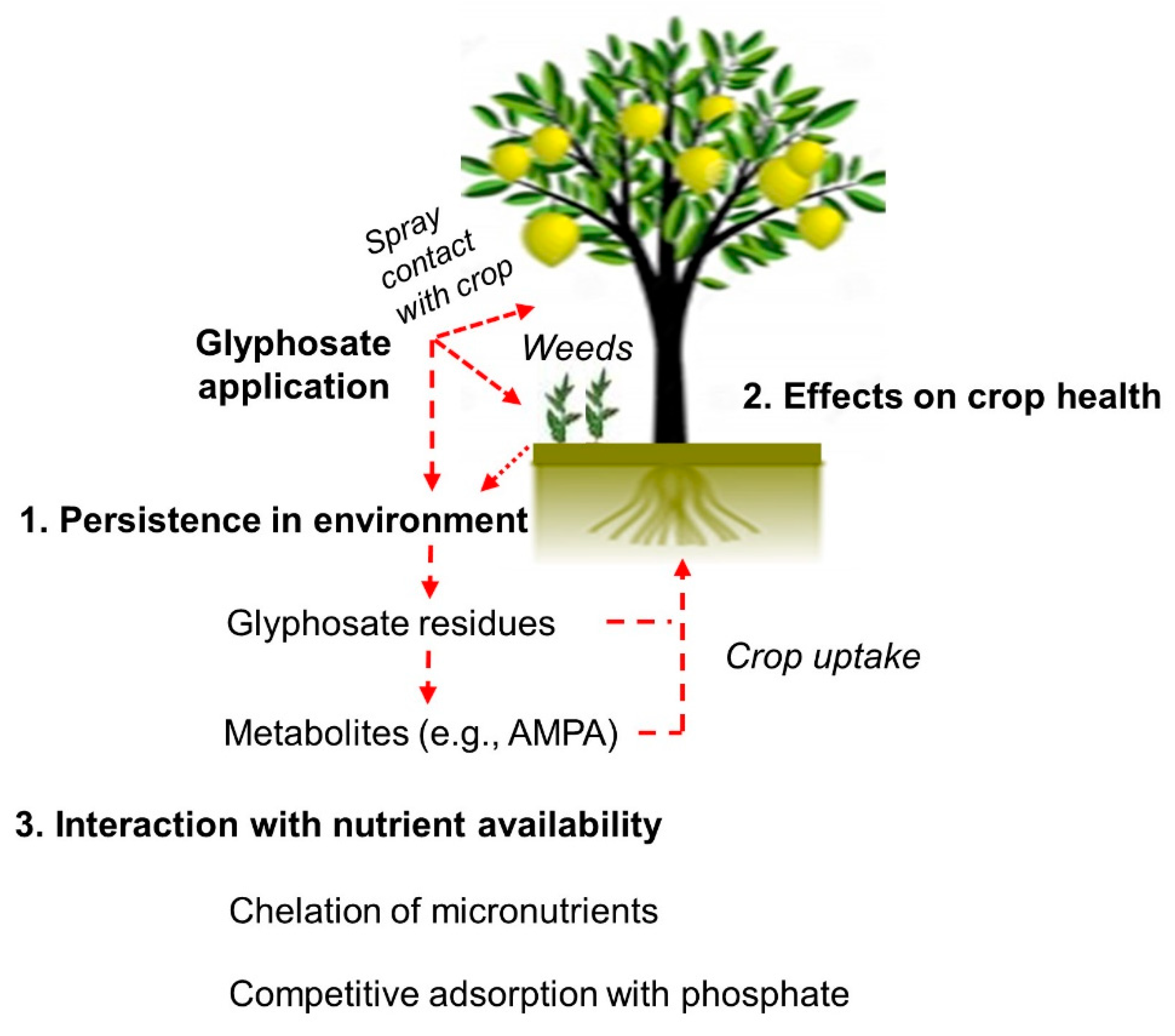
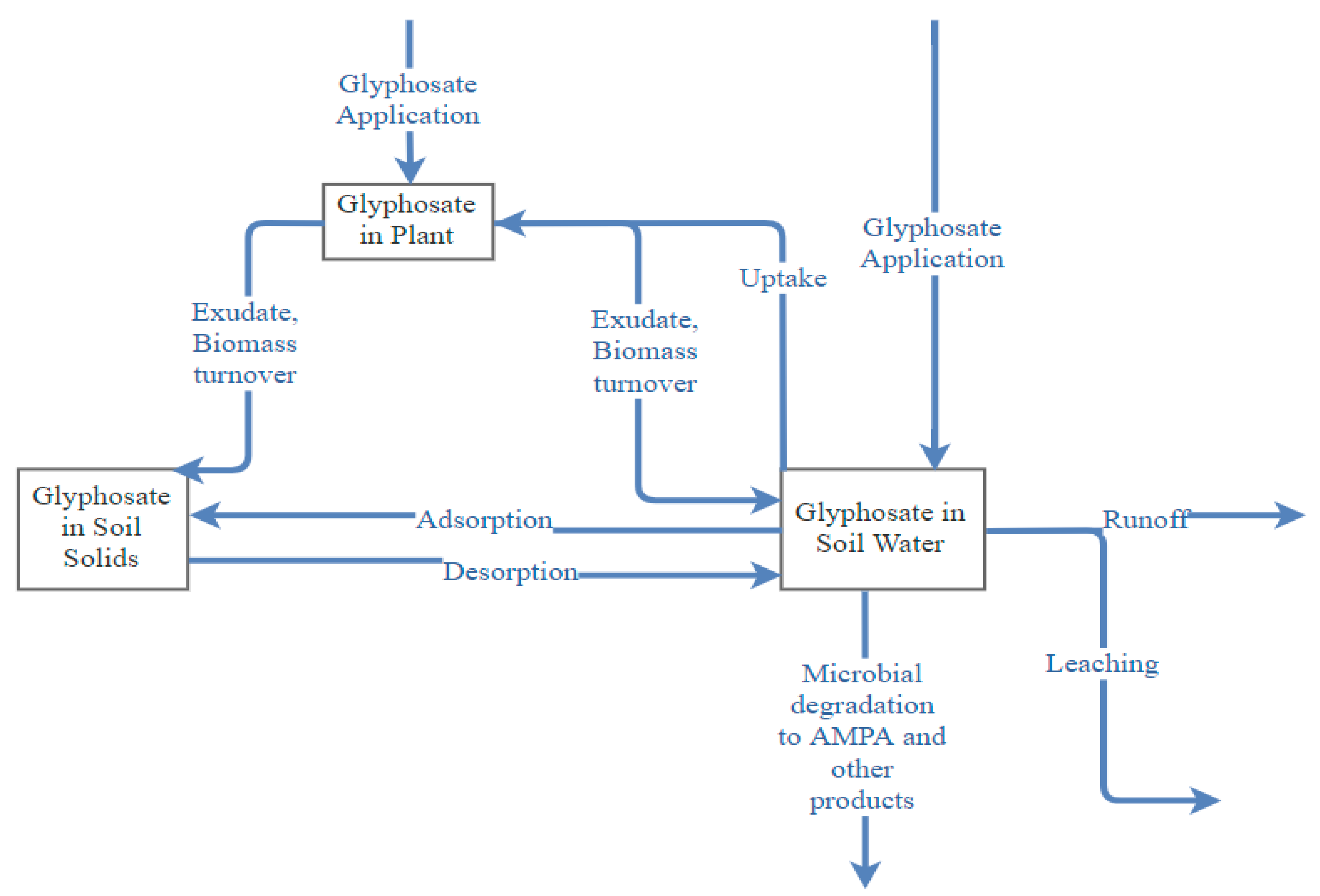
2. Glyphosate Persistence in the Environment
3. Glyphosate’s Effects on Crop Health
4. Glyphosate’s Interaction with Crop Nutrition
5. Conclusions and Future Direction
Author Contributions
Funding
Conflicts of Interest
References
- Rodrigues, B.N.; Almeida, F.S. Guide of Herbicides, 5th ed.; IAPAR: Londrina, Brazil, 2005. [Google Scholar]
- Sharma, S.D.; Singh, M. Environmental factors affecting absorption and bio-efficacy of glyphosate in Florida beggarweed (Desmodium tortuosum). Crop Prot. 2001, 20, 511–516. [Google Scholar] [CrossRef]
- Kataoka, H.; Ryu, S.; Sakiyama, N.; Makita, M. Simple and rapid determination of the herbicides glyphosate and glufosinate in river water, soil and carrot samples by gas chromatography with flame photometric detection. J. Chromatogr. A 1996, 726, 253–258. [Google Scholar] [CrossRef]
- Forlani, G.; Mangiagalli, A.; Nielsen, E.; Suardi, C.M. Degradation of the phosphonate herbicide glyphosate in soil: Evidence for a possible involvement of unculturable microorganisms. Soil Biol. Biochem. 1999, 31, 991–997. [Google Scholar] [CrossRef]
- Gravena, R.; Filho, R.V.; Alves, P.L.C.A.; Mazzafera, P.; Gravena, A.R. Glyphosate has low toxicity to citrus plants growing in the field. Can. J. Plant Sci. 2012, 92, 119–127. [Google Scholar] [CrossRef]
- Chang, S.Y.; Liao, C. Analysis of glyphosate, glufosinate and aminomethylphosphonic acid by capillary electrophoresis with indirect fluorescence detection. J. Chromatogr. A 2002, 959, 309–315. [Google Scholar] [CrossRef]
- Nandula, V.K. Glyphosate Resistance in Crops and Weeds: History, Development, and Management; John Wiley & Sons: Hoboken, NJ, USA, 2010; pp. 1–33. [Google Scholar]
- Motharasan, M.; Shukor, M.Y.; Yasid, N.A.; Wan Johari, W.L.; Ahmad, S.A. Environmental fate and degradation of glyphosate in soil. Pertanika J. Sch. Res. Rev. 2018, 4, 102–116. [Google Scholar]
- Quinn, J.P.; Peden, J.M.M.; Dick, R.E. Glyphosate tolerance and utilization by the microflora of soils treated with the herbicide. Appl. Microbiol. Biotechnol. 1988, 29, 511–516. [Google Scholar] [CrossRef]
- Battaglin, W.A.; Kolpin, D.W.; Scribner, E.A.; Kuivila, K.M.; Sandstrom, M.W. Glyphosate other herbicides and transformation products in midwestern streams, 2002. J. Am. Water Resour. Assoc. 2005, 41, 323–332. [Google Scholar] [CrossRef]
- Vereecken, H. Mobility and leaching of glyphosate: A review. Pest Manag. Sci. 2005, 61, 1139–1151. [Google Scholar] [CrossRef]
- Giesy, J.P.; Dobson, S.; Solomon, K.R. Ecotoxicological risk assessment for Roundup herbicide. In Reviews of Environmental Contamination and Toxicology; Springer: New York, NY, USA, 2000; Volume 167, pp. 35–120. [Google Scholar]
- Hanke, I.; Wittmer, I.; Bischofberger, S.; Stamm, C.; Singer, H. Relevance of urban glyphosate use for surface water quality. Chemosphere 2010, 81, 422–429. [Google Scholar] [CrossRef]
- Perez, G.L.; Vera, M.S.; Miranda, L.A. Effects of herbicide glyphosate and glyphosate-based formula-tions on aquatic ecosystems. In Herbicides and Environment; Kortekamp, A., Ed.; InTech: Rijeka, Croatia, 2011. [Google Scholar]
- Yamada, T.; Kremer, R.J.; Castro, P.R.C.; Wood, B.W. Glyphosate interactions with physiology, nutrition, and diseases of plants: Threat to agricultural sustainability? Eur. J. Agron. 2009, 31, 111–113. [Google Scholar] [CrossRef]
- Ellis, J.M.; Griffin, J.L. Soybean (Glycine max) and cotton (Gossypium hirsutum) response to simulated drift of glyphosate and glufosinate. Weed Technol. 2002, 16, 580–586. [Google Scholar] [CrossRef]
- Neumann, G.; Kohls, S.; Landesberg, E.; Stoch-Oliveira Souza, K.; Yamda, T.; Römheld, V. Relevance of glyphosate transfer to non-target plants via the rhizosphere. J. Plant Dis. Prot. 2006, 20, 963–969. [Google Scholar]
- Mamy, L.; Barriuso, E.; Gabrielle, B. Glyphosate fate in soils when arriving in plant residues. Chemosphere 2016, 154, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Lupi, L.; Miglioranza, K.S.; Aparicio, V.C.; Marino, D.; Bedmar, F.; Wunderlin, D.A. Occurrence of glyphosate and AMPA in an agricultural watershed from the southeastern region of Argentina. Sci. Total Environ. 2015, 536, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Newton, M.; Horner, L.M.; Cowell, J.E.; White, D.E.; Cole, E.C. Dissipation of glyphosate and aminomethylphosphonic acid in north American forests. J. Agric. Food Chem. 1994, 42, 1795–1802. [Google Scholar] [CrossRef]
- Shushkova, T.; Ermakova, I.; Leontievsky, A. Glyphosate bioavailability in soil. Biodegradation 2010, 21, 403–410. [Google Scholar] [CrossRef]
- Kjaer, J.; Ullum, M.; Olsen, P.; Helweg, A.; Mogensen, B.B.; Plauborg, F.; Grant, R.; Fomsgaard, I.S.; Brüsch, W. Geological Survey of Denmark and Greenland. In The Danish Pesticide Leaching Assessment Programme: Monitoring Results May 1999–June 2002; Third Report; Danish Institute of Agricultural Sciences, National Environmental Research Institute: Roskilde, Denmark, 2003. [Google Scholar]
- USEPA. Problem Formulation for the Ecological Risk and Drinking Water Exposure Assessments in Support of the Registration Review of Glyphosate and Its Salts; EPA-HQ-OPP-2009-0361-0007; Office of Prevention, Pesticides and Toxic Substances, Environmental Protection Agency: Washington, DC, USA, 2009.
- French Institute for the Environment. Les pesticides dans les eaux, bilan annuel. In Etudes et Travaux; French Institute for the Environment: Orléans, France, 2002; Volume 36. (In French) [Google Scholar]
- French Institute for the Environment. Les pesticides dans les eaux, cinqiuème bilan annuel. In Etudes et Travaux; Données 2001; French Institute for the Environment: Orléans, France, 2003; Volume 37. (In French) [Google Scholar]
- French Institute for the Environment. Les pesticides dans les eaux, sixième bilan annuel. In Etudes et Travaux; Données 2002; French Institute for the Environment: Orléans, France, 2004; Volume 42. (In French) [Google Scholar]
- Wang, S.; Seiwert, B.; Kastner, M.; Miltner, A.; Schaffer, A.; Reemtsma, T.; Yang, Q.; Nowak, K.M. (Bio)degradation of glyphosate in water sediment microcosms—A stable isotope co-labeling approach. Water Res. 2016, 99, 91–100. [Google Scholar] [CrossRef]
- Franz, J.E.; Mao, M.K.; Sikorski, J.A. Glyphosate: A Unique Global Herbicide; ACS Monograph 189; American Chemical Society: Washington, DC, USA, 1997; Volume 653. [Google Scholar]
- Sprankle, P.; Meggitt, W.F.; Penner, D. Adsorption, mobility and microbial degradation of glyphosate in the soil. Weed Sci. 1975, 23, 229–234. [Google Scholar] [CrossRef]
- Gimsing, A.L.; Borggaard, O.K.; Jacobsen, O.S.; Aamand, J.; Sorensen, J. Chemical and microbiological soil characteristics controlling glyphosate mineralization in Danish surface soils. Appl. Soil Ecol. 2004, 27, 233–242. [Google Scholar] [CrossRef]
- Torstensson, L. Behaviour of glyphosate in soils and its degradation. In The Herbicide Glyphosate; Grossbard, E., Atkinson, D., Eds.; Butterworths: London, UK, 1985; pp. 450–490. [Google Scholar]
- Haney, R.L.; Senseman, S.A.; Hons, F.M.; Zuberer, D.A. Effect of glyphosate on soil microbial activity and biomass. Weed Sci. 2000, 48, 89–93. [Google Scholar] [CrossRef]
- Bussey, M.D.; Ratcliff, A.W.; Shestak, C.J.; Powers, R.F. Glyphosate toxicity and the effects of long-term vegetation control on soil microbial communities. Soil Biol. Biochem. 2001, 33, 1777–1789. [Google Scholar] [CrossRef]
- Araujo, A.S.F.; Monteiro, R.T.R.; Abarkeli, R.B. Effect of glyphosate on the microbial activity of two Brazilian soils. Chemosphere 2003, 52, 799–804. [Google Scholar] [CrossRef]
- Rueppel, M.L.; Brightwell, B.B.; Schaefer, J.; Marvel, J. Metabolism and degradation of glyphosate in soil and water. J. Agric. Food Chem. 1977, 25, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Stenrod, M.; Eklo, O.M.; Charnay, M.-P.; Benoit, B. Effect of freezing and thawing on microbial activity and glyphosate degradation in two Norwegian soils. Pest Manag. Sci. 2005, 61, 887–898. [Google Scholar] [CrossRef]
- Lancaster, S.H.; Hollister, E.B.; Senseman, S.A.; Gentry, T.J. Effects of repeated glyphosate applications on soil microbial community composition and the mineralization of glyphosate. Pest Manag. Sci. 2009, 66, 59–64. [Google Scholar] [CrossRef]
- Accinelli, C.; Koskinen, W.C.; Seebinger, J.D.; Vicari, A.; Sadowsky, M.J. Effects of incorporated corn residues on glyphosate mineralization and sorption in soil. J. Agric. Food Chem. 2005, 53, 4110–4117. [Google Scholar] [CrossRef]
- Dick, R.E.; Quinn, J.P. Glyphosate degrading isolates from environmental samples: Occurrence and pathways of degradation. Appl. Microbiol. Biotechnol. 1995, 43, 545–550. [Google Scholar] [CrossRef]
- Haney, R.L.; Senseman, S.A.; Hons, F.M. Effect of Roundup Ultra on microbial activity and biomass from selected soils. J. Environ. Qual. 2002, 31, 730–735. [Google Scholar] [CrossRef]
- Sorensen, S.R.; Schultz, A.; Jacobsen, O.S.; Aamand, J. Sorption, desorption and mineralisation of the herbicides glyphosate and MCPA in samples from two Danish soil and subsurface profiles. Environ. Pollut. 2006, 141, 184–194. [Google Scholar] [CrossRef]
- Zaranyika, M.F.; Nyandoro, M.G. Degradation of glyphosate in the aquatic environment: An enzymatic kinetic model that takes into account microbial degradation of both free and colloidal (or sediment) particle adsorbed glyphosate. J. Agric. Food Chem. 1993, 41, 838–842. [Google Scholar] [CrossRef]
- Glass, R.L. Adsorption of glyphosate by soils and clay minerals. J. Agric. Food Chem. 1987, 35, 497–500. [Google Scholar] [CrossRef]
- Gerritse, R.G.; Beltran, J.; Hernandez, F. Adsorption of atrazine, simazine, and glyphosate in soils of the Gnangara Mound, Western Australia. Aust. J. Soil. Res. 1996, 34, 599–607. [Google Scholar] [CrossRef]
- Scow, K.M.; Hutson, J. Effect of diffusion and sorption on the kinetics of biodegradation: Theoretical considerations. Soil Sci. Soc. Am. J. 1992, 56, 119–127. [Google Scholar] [CrossRef]
- Eberbach, P. Applying non-steady-state compartmental analysis to investigate the simultaneous degradation of soluble and sorbed glyphosate (N-(phosphonomethyl)glycine) in four soils. Pestic. Sci. 1998, 52, 229–240. [Google Scholar] [CrossRef]
- Schnurer, Y.; Persson, P.; Nilsson, M.; Nordgren, A.; Giesler, R. Effects of surface sorption on microbial degradation of glyphosate. Environ. Sci. Technol. 2006, 40, 4145–4150. [Google Scholar] [CrossRef]
- Kononova, S.V.; Nesmeyanova, M.A. Phosphonates and their degradation by microorganisms. Biochemistry (Moscow) 2002, 67, 184–195. [Google Scholar] [CrossRef]
- Singh, B.K.; Walker, A. Microbial degradation of organophosphorous compounds. FEMS Microbiol. Rev. 2006, 30, 428–471. [Google Scholar] [CrossRef]
- Quinn, J.P.; Peden, J.M.M.; Dick, R.E. Carbon–phosphorus bond cleavage by Gram positive and Gram-negative bacteria. Appl. Microbiol. Biotechnol. 1989, 31, 283–287. [Google Scholar] [CrossRef]
- Liu, C.M.; McLean, P.A.; Sookdeo, C.C.; Cannon, F.C. Degradation of the herbicide glyphosate by members of the family Rhizobiaceae. Appl. Environ. Microbiol. 1991, 57, 1799–1804. [Google Scholar]
- Barry, G.F.; Kishore, G.M. Glyphosate Tolerant Plant. U.S. Patent 5,776,760, 7 July 1998. [Google Scholar]
- Al-Khatib, K.; Peterson, D. Soybean (Glycine max) response to simulated drift from selected sulfonylurea herbicides, dicamba, glyphosate, and glufosinate. Weed Technol. 1999, 13, 264–270. [Google Scholar] [CrossRef]
- Kanissery, R. Herbicide Exposure to Crops—“Making a Foe out of a Friend”. Acta Sci. Agric. 2019, 3, 161–162. [Google Scholar]
- Reddy, K.N.; Rimando, A.M.; Duke, S.O. Aminomethylphosphonic acid, a metabolite of glyphosate, causes injury in glyphosate-treated, glyphosate resistant soybean. J. Agric. Food Chem. 2004, 52, 5139–5143. [Google Scholar] [CrossRef] [PubMed]
- Coupland, D.; Casely, J.C. Presence of 14C activity in root exudates and gutation fluid from Agropyron repens treated with 14C-labelled glyphosate. New Phytol. 1979, 83, 17–22. [Google Scholar] [CrossRef]
- von Wiren-Lehr, S.; Komoßa, D.; Glaesgen, W.E.; Sandermann, H., Jr.; Scheunert, I. Mineralization of [14C] Glyphosate and its plant-associated residues in arable soils originating from different farming systems. Pestic. Sci. 1997, 54, 436–442. [Google Scholar] [CrossRef]
- Laitinen, P.; Sari Rämö, S.; Siimes, K. Glyphosate translocation from plants to soil—Does this constitute a significant proportion of residues in soil? Plant Soil. 2007, 300, 51–60. [Google Scholar] [CrossRef]
- Pline, W.A.; Wilcut, J.W.; Edmisten, K.L.; Wells, R. Physiological and morphological response of glyphosate resistant and non-glyphosate-resistant cotton seedlings to root-absorbed glyphosate. Pestic. Biochem. Phys. 2002, 73, 48–58. [Google Scholar] [CrossRef]
- Wagner, R.; Kogan, M.; Parada, A.M. Phytotoxic activity of root absorbed glyphosate in corn seedlings (Zea mays L.). Weed Biol. Manag. 2003, 3, 228–232. [Google Scholar] [CrossRef]
- Petersen, I.L.; Hansen, H.C.; Ravn, H.W.; Sørensen, J.C.; Sørensen, H. Metabolic effects in rapeseed (Brassica napus L.) seedlings after root exposure to glyphosate. Pestic. Biochem. Phys. 2007, 89, 220–229. [Google Scholar] [CrossRef]
- Johal, G.S.; Rahe, J.E. Glyphosate, hypersensitivity and phytoalexin accumulation in the incompatible bean anthracnose host-parasite interaction. Physiol. Mol. Plant Pathol. 1988, 32, 267–281. [Google Scholar] [CrossRef]
- Lévesque, C.A.; Rahe, J.E.; Eaves, D.M. Effects of glyphosate on Fusarium spp.: Its influence on root colonization of weeds, propagule density in the soil, and crop emergence. Can. J. Microbiol. 1987, 33, 354–360. [Google Scholar] [CrossRef]
- Fernandez, M.R.; Zentner, R.P.; Basnyat, P.; Gehl, D.; Selles, F.; Huber, D. Glyphosate associations with cereal diseases caused by Fusarium spp. in the Canadian Prairies. Eur. J. Agron. 2009, 31, 133–143. [Google Scholar] [CrossRef]
- Fernandez, M.R.; Zentner, R.P.; DePauw, R.M.; Gehl, D.; Stevenson, F.C. Impacts of crop production factors on common root rot of barley in Eastern Saskatchewan. Crop Sci. 2007, 47, 1585–1595. [Google Scholar] [CrossRef]
- Rahe, J.E.; Lévesque, C.A.; Johal, G.S. Synergistic role of soil fungi in the herbicidal efficacy of glyphosate. ACS Symp. Ser. 1990, 439, 260–275. [Google Scholar]
- Cornish, P.S.; Burgin, S. Residual effects of glyphosate herbicide in ecological restoration. Restor. Ecol. 2005, 13, 695–702. [Google Scholar] [CrossRef]
- Tong, M.; Gao, W.; Jiao, W.; Zhou, J.; Li, Y.; He, L.; Hou, R. Uptake, translocation, metabolism, and distribution of glyphosate in nontarget tea plant (Camellia sinensis L.). J. Agric. Food. Chem. 2017, 65, 7638–7646. [Google Scholar] [CrossRef]
- Bai, S.H.; Ogbourne, S.M. Glyphosate environmental contamination, toxicity and potential risks to human health via food contamination. Environ. Sci. Pollut. Res. 2016, 23, 18988–19001. [Google Scholar] [CrossRef]
- Bott, S.; Tesfamariam, T.; Candan, H.; Cakmak, I.; Römheld, V.; Neumann, G. Glyphosate induced impairment of plant growth and micronutrient status in glyphosate-resistant soybean (Glycine max L.). Plant Soil 2008, 312, 185. [Google Scholar] [CrossRef]
- Feng, P.C.; Pratley, J.E.; Bohn, J.A. Resistance to glyphosate in Lolium rigidum. II. Uptake, translocation, and metabolism. Weed Sci. 1999, 47, 412–415. [Google Scholar] [CrossRef]
- Hest, D. Citrus managers strive for top weed control, low fruit drop. In Citrus Industry; AgNet Media, Inc.: Gainesville, FL, USA, 1996; Volume 77, pp. 54–57. [Google Scholar]
- Kanissery, R.G.; Fernando, A.; Batuman, O. How to handle glyphosate-related fruit drop. In Citrus Industry; AgNet Media, Inc.: Gainesville, FL, USA, 2018. [Google Scholar]
- Kanissery, R.G.; Welsh, A.; Sims, G.K. Effect of Soil Aeration and Phosphate Addition on the Microbial Bioavailability of Carbon-14-Glyphosate. J. Environ. Qual. 2015, 44, 137–144. [Google Scholar] [CrossRef]
- Eker, S.; Ozturk, L.; Yazici, A.; Erenoglu, B.; Romheld, V.; Cakmak, I. Foliar-applied glyphosate substantially reduced uptake and transport of iron and manganese in sunflower (Helianthus annuus L.) plants. J. Agric. Food. Chem. 2006, 54, 10019–10025. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.S.; Ozturk, L.; Cakmak, I.; Budak, H. Turfgrass species response exposed to increasing rates of glyphosate application. Eur. J. Agron. 2009, 31, 120–125. [Google Scholar]
- Cakmak, I.; Yazici, A.; Tutus, Y.; Ozturk, L. Glyphosate reduced seed and leaf concentrations of calcium, manganese, magnesium, and iron in non-glyphosate resistant soybean. Eur. J. Agron. 2009, 31, 114–119. [Google Scholar] [CrossRef]
- Tesfamariam, T.; Bott, S.; Cakmak, I.; Römheld, V.; Neumann, G. Glyphosate in the rhizosphere—Role of waiting times and different glyphosate binding forms in soils for phytotoxicity to non-target plants. Eur. J. Agron. 2009, 31, 126–132. [Google Scholar] [CrossRef]
- Morgan, K.T.; Rouse, R.E.; Ebel, R.C. Foliar Applications of Essential Nutrients on Growth and Yield of ‘Valencia’Sweet Orange Infected with Huanglongbing. HortScience 2016, 51, 1482–1493. [Google Scholar] [CrossRef]
- Spann, T.M.; Schumann, A.W. The role of plant nutrients in disease development with emphasis on citrus and huanglongbing. Proc. Fla. State Hort. Soc. 2009, 122, 169–171. [Google Scholar]
- Gimsing, A.L.; Borggaard, O.K. Competitive adsorption and desorption of glyphosate and phosphate on clay silicates and oxides. Clay Miner. 2002, 37, 509–515. [Google Scholar] [CrossRef]
- Hance, R.J. Adsorption of glyphosate by soils. Pest Manag. Sci. 1976, 7, 363–366. [Google Scholar] [CrossRef]
- Wang, Y.J.; Zhou, D.M.; Sun, R.J. Effects of phosphate on the adsorption of glyphosate on three different types of Chinese soils. J. Environ. Sci. China 2004, 17, 711–715. [Google Scholar]
| Chemical structure |  |
| CAS number | 1071-83-6 |
| Chemical name | N-(phosphonomethyl) glycine |
| Empirical formula | C3H8NO5P |
| Molecular weight (g mol−1) | 169.08 |
| Water solubility (mg L−1 at 25 °C) | 10,000 to 15,700 [10] |
| Octanol–water coeff. (Kow) | −4.6 to −1.6 [10] |
| Vapor pressure (mm Hg at 25 °C) | 4.3 × 10−10 [10] |
| Freundlich adsorption coeff. (Kads) (L Kg−1) | 0.6 to 303 [11] |
| Degradation half-life in soil (T1/2) (days) | 7–60 [12] |
| Photolysis half-life (days) | Not substantial |
| EPA maximum contamination level (μg L−1) | 700 [10] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kanissery, R.; Gairhe, B.; Kadyampakeni, D.; Batuman, O.; Alferez, F. Glyphosate: Its Environmental Persistence and Impact on Crop Health and Nutrition. Plants 2019, 8, 499. https://doi.org/10.3390/plants8110499
Kanissery R, Gairhe B, Kadyampakeni D, Batuman O, Alferez F. Glyphosate: Its Environmental Persistence and Impact on Crop Health and Nutrition. Plants. 2019; 8(11):499. https://doi.org/10.3390/plants8110499
Chicago/Turabian StyleKanissery, Ramdas, Biwek Gairhe, Davie Kadyampakeni, Ozgur Batuman, and Fernando Alferez. 2019. "Glyphosate: Its Environmental Persistence and Impact on Crop Health and Nutrition" Plants 8, no. 11: 499. https://doi.org/10.3390/plants8110499
APA StyleKanissery, R., Gairhe, B., Kadyampakeni, D., Batuman, O., & Alferez, F. (2019). Glyphosate: Its Environmental Persistence and Impact on Crop Health and Nutrition. Plants, 8(11), 499. https://doi.org/10.3390/plants8110499







