The Effect of Granular Commercial Fertilizers Containing Elemental Sulfur on Wheat Yield under Mediterranean Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Commercial Field Trials
2.2. Experimental Field Trials
2.3. The Nature of the FBES Granules
2.4. Soil Analysis
2.5. Head Measurements
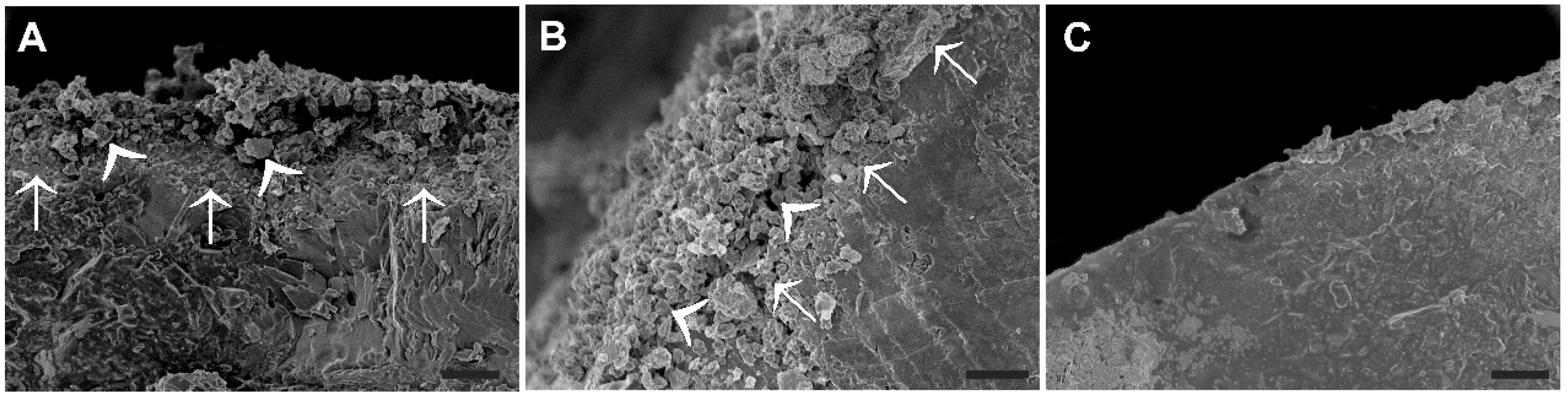
2.6. Scanning Electron Microscopy
2.7. Statistical Analysis
3. Results
3.1. Characteristics of ES Enriched Fertilizer Granules
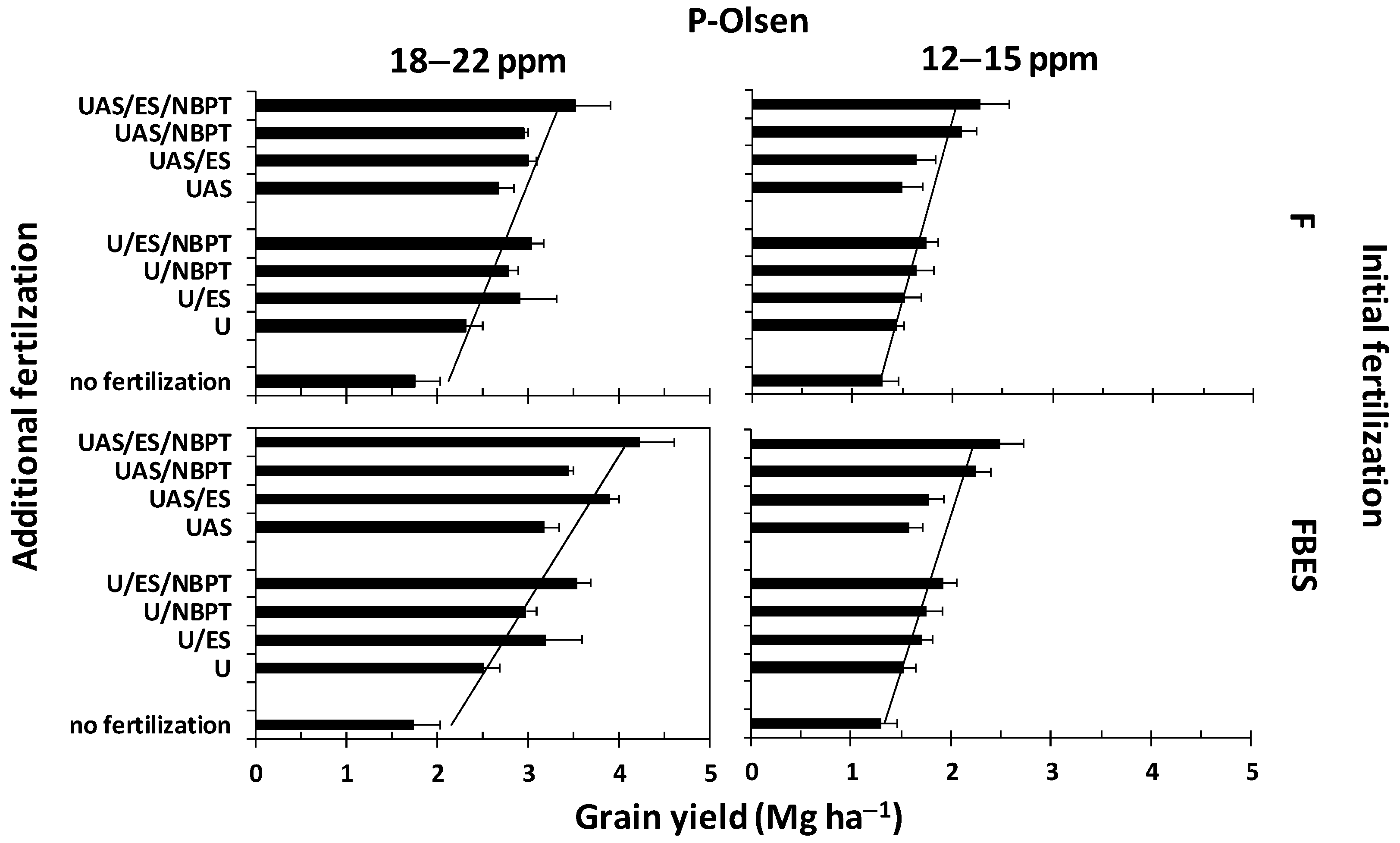
3.2. The Effect of the Fertilization Schemes on Yield
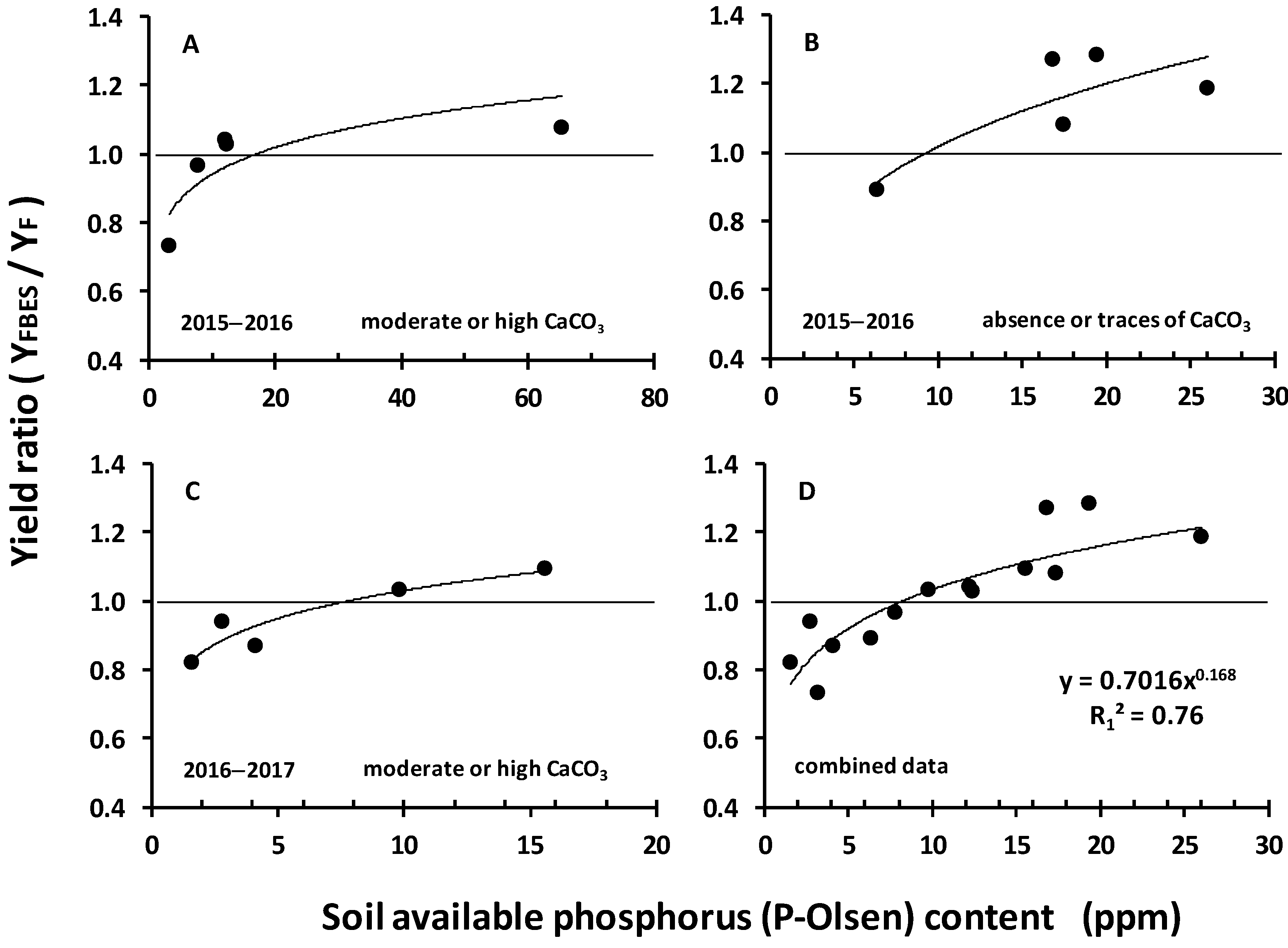
3.3. P-Olsen as a Yield-Limiting Factor
3.4. The effect of ES and NBPT Containing Fertilizers on Yield
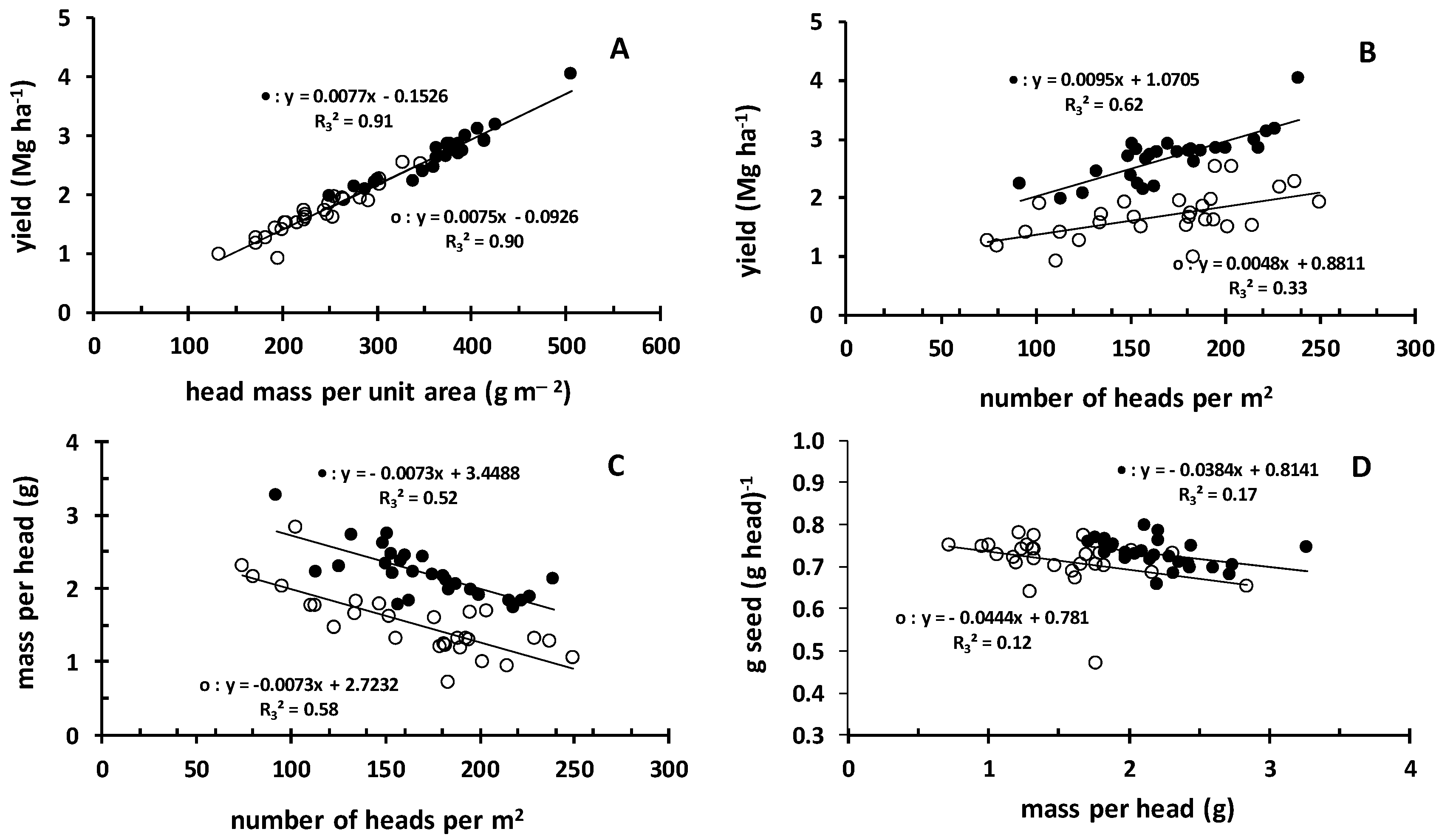
3.5. Yield and Head Characteristics
4. Discussion
4.1. The Role of Soil Available P on the Relative Yield
4.2. The Efficacy of ES Fertilization Schemes
4.3. The Efficacy of ES and NBPT Fertilization Schemes
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| RS | rhizosoil |
| OM | organic matter |
| HS | humic substances |
| F | granular conventional fertilizer |
| ES | elemental sulfur |
| B | binder |
| U | urea |
| UAS | urea ammonium sulfate |
| NBPT | N-(n-butyl) thiophosphoric triamide |
References
- Zhao, F.J.; Hawkesford, M.J.; McGrath, S.P. Sulphur assimilation and effects on yield and quality of wheat. J. Cer. Sci. 1999, 30, 1–17. [Google Scholar] [CrossRef]
- Eriksen, J. Soil sulfur cycling in temperate agricultural systems. Adv. Agron. 2009, 102, 55–89. [Google Scholar]
- Zhao, F.J.; McGrath, S.P.; Blake-Kalff, M.M.A.; Link, A.; Tucker, M. Crop responses to sulphur fertilisation in Europe. In Proceedings No.504, International Fertiliser Society; International Fertiliser Society: York, UK, 2002. [Google Scholar]
- Grant, C.; Hawkesford, M.J. Sulfur. In Handbook of Plant Nutrition, 2nd ed.; Barker, A.V., Pilbeam, D.J., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 261–302. [Google Scholar]
- Matamwa, W.; Blair, G.; Guppy, C.; Yunusa, I. Plant Availability of Sulfur Added to Finished Fertilizers. Commun. Soil Sci. Plant Anal. 2018, 49, 433–443. [Google Scholar] [CrossRef]
- Somani, L.L.; Totawat, K.L. Mined and industrial waste products capable of generating gypsum in soil. In Handbook of Soil Conditioners; Wallace, A., Terry, R.E., Eds.; Marcel Dekker, Inc.: New York, NY, USA; Basel, Switzerland; Hong Kong, China, 1998; pp. 257–291. [Google Scholar]
- Boswell, C.C.; Friesen, D.K. Elemental sulfur fertilizers and their use on crops and pastures. Fert. Res. 1993, 35, 127–149. [Google Scholar] [CrossRef]
- Chien, S.H.; Prochnow, L.I.; Cantarella, H. Recent developments of fertilizer production and use to increase nutrient efficiency and minimize environmental impacts. Adv. Agron. 2009, 102, 261–316. [Google Scholar]
- Benardos, D. Method for Coating Fertilizer Beads with Elemental Sulfur. U.S. Patent WO2017077350A, 5 November 2017. [Google Scholar]
- Chien, S.H.; Teixeira, L.A.; Cantarella, H.; Rehm, G.W.; Grant, C.A.; Gearhart, M.M. Agronomic Effectiveness of Granular Nitrogen/Phosphorus Fertilizers Containing Elemental Sulfur with and without Ammonium Sulfate: A Review. Agron. J. 2016, 108, 1203–1213. [Google Scholar] [CrossRef]
- Cantarella, H.; Otto, R.; Rodrigues Soares, J.; Gomes de Brito Silva, A. Agronomic efficiency of NBPT as a urease inhibitor: A review. J. Adv. Res. 2018, 13, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Whitehurst, G.B.; Whitehurst, B.M. Aminoalcohol Solutions of N-(N-butyl)thio-Phosphoric Triamine (NBPT) and Urea Fertilizers Using such Solutions as Urease Inhibitors. U.S. Patent 2010/0206031A1, 19 August 2010. [Google Scholar]
- Chorianopoulou, S.N.; Saridis, G.I.; Sigalas, P.; Margetis, M.; Benardos, D.; Mavrogiannis, H.; Bouranis, D.L. The application of S0-coated fertilizer to durum wheat crop. In Plant Sulfur Proceedings; Sulfur Metabolism in Higher Plants—Fundamental, Environmental and Agricultural Aspects; De Kok, L.J., Haneklaus, S.H., Hawkesford, M.J., Schnug, E., Eds.; Springer: Berlin, Germany, 2017; Volume 3, pp. 115–122. [Google Scholar]
- Bouranis, D.L.; Chorianopoulou, S.N.; Margetis, M.; Saridis, G.I.; Sigalas, P.P. Effect of Elemental Sulfur as Fertilizer Ingredient on the Mobilization of Iron from the Iron Pools of a Calcareous Soil Cultivated with Durum Wheat and the Crop’s Iron and Sulfur Nutrition. Agriculture 2018, 8, 20. [Google Scholar] [CrossRef]
- Gee, G.W.; Bauder, J.W. Particle-size analysis. In Methods of Soil Analysis, Part 1. Physical and Mineralogical Methods; Klute, A., Ed.; ASA, SSSA: Madison, WI, USA, 1986; pp. 383–411. [Google Scholar]
- Bilias, F.; Barbayiannis, N. Evaluation of sodium tetraphenylboron (NaBPh4) as a soil test of potassium availability. Arch. Agron. Soil Sci. 2017, 63, 468–476. [Google Scholar]
- McLean, E.O. Soil pH and lime requirement. In Methods of Soil Analysis, Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA, SSSA: Madison, WI, USA, 1982; pp. 199–224. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon and organic matter. In Methods of Soil Analysis, Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA, SSSA: Madison, WI, USA, 1982; pp. 539–547. [Google Scholar]
- Olsen, S.R.; Sommers, L.E. Phosphorus. In Methods of Soil Analysis, Part 2, 2nd ed.; Page, A.L., Ed.; Agron Monogr. ASA and ASSA: Madison, WI, USA, 1982; pp. 403–430. [Google Scholar]
- Thomas, G.W. Exchangeable cations. In Methods of Soil Analysis, Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA, SSSA: Madison, WI, USA, 1982; pp. 159–166. [Google Scholar]
- Lindsay, W.L.; Norvell, W.A. Development of DTPA soil test for zinc, iron, manganese and copper. Soil Sci. Soc. Am. J. 1978, 42, 421–428. [Google Scholar]
- Roussos, P.A.; Gasparatos, D.; Kechrologou, K.; Katsenos, P.; Bouchagier, P. Impact of organic fertilization on soil properties, physiology and yield in two newly planted Greek olive (Olea europaea L.) cultivars under Mediterranean conditions. Sci. Hortic. 2017, 220, 11–19. [Google Scholar] [CrossRef]
- Chatzistathis, T.; Papaioannou, A.; Gasparatos, D.; Molassiotis, A. From which soil metal fractions Fe, Mn, Zn and Cu are taken up by olive trees (Olea europaea L., ‘Chondrolia Chalkidikis’) in organic groves? J. Environ. Manag. 2017, 203, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.B., Jr. Soil Analysis Handbook of Reference Methods; CRC Press: Boca Raton, FL, USA; London, UK; New York, NY, USA; Washington, DC, USA, 1999. [Google Scholar]
- Stroup, W.W. Generalized Linear Mixed Models; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 2 December 2018).
- Germida, J.J.; Jansen, H.H. Factors affecting the oxidation of elemental sulfur in soils. Fertil. Res. 1993, 35, 101–114. [Google Scholar] [CrossRef]
- Zhao, C.; Gupta, V.V.S.R.; Degryse, F.; McLaughlin, M.J. Abundance and diversity of sulphur-oxidising bacteria and their role in oxidizing elemental sulphur in cropping soils. Biol. Fertil. Soils 2017, 53, 157–169. [Google Scholar] [CrossRef]
- Zhao, C.; Degryse, F.; Gupta, V.; McLaughlin, M.J. Elemental Sulfur oxidation in Australian cropping soils. Soil Sci. Soc. Am. J. 2015, 75, 89–96. [Google Scholar] [CrossRef]
- Chorianopoulou, S.N.; Venieraki, A.; Maniou, F.; Mavrogiannis, H.; Benardos, D.; Katinakis, P.; Bouranis, D.L. Sulfogrow®: A new type of plant biostimulant? In Proceedings of the 13th International Conference on Protection and Restoration of the Environment, Mykonos, Greece, 3–8 July 2016; pp. 733–738. [Google Scholar]
- Kertesz, M.A.; Mirleau, P. The role of soil microbes in plant sulphur nutrition. J. Exp. Bot. 2004, 55, 1939–1945. [Google Scholar] [CrossRef] [PubMed]
- Gahan, J.; Schmalenberger, A. The role of bacteria and mycorrhiza in plant sulfur supply. Front. Plant Sci. 2014, 5, 723. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.E.; Barea, J.-M.; McNeill, A.M.; Prigent-Combaret, C. Acquisition of phosphorus and nitrogen in the rhizosphere and plant growth promotion by microorganisms. Plant Soil 2009, 321, 305–339. [Google Scholar] [CrossRef]
- Shen, J.; Yuan, L.; Zhang, J.; Li, H.; Bai, Z.; Chen, X.; Zhang, W.; Zhang, F. Phosphorus Dynamics: From Soil to Plant. Plant Physiol. 2011, 156, 997–1005. [Google Scholar] [CrossRef]
- Ziadi, N.; Whalen, J.K.; Messiga, A.J.; Morel, C. Assessment and modeling of soil available phosphorus in sustainable cropping systems. Adv. Agron. 2013, 122, 85–126. [Google Scholar]
- Rodriguez, D.; Andrade, F.H.; Goudriaan, J. Effects of phosphorus nutrition on tiller emergence in wheat. Plant Soil 1999, 209, 283–295. [Google Scholar] [CrossRef]
- Otto, W.M.; Kilian, W.H. Response of Soil Phosphorus Content, Growth and Yield of Wheat to Long-Term Phosphorus Fertilization in A Conventional Cropping System. Nutr. Cycl. Agroecos. 2001, 61, 283–292. [Google Scholar] [CrossRef]
- Alam, S.M.; Azam, S.; Ali, S.; Iqbal, M. Wheat yield and P fertilizer efficiency as influenced by rate and integrated use of chemical and organic fertilizers. Pak. J. Soil Sci. 2003, 22, 72–76. [Google Scholar]
- Gill, H.S.; Singh, H.B.A.; Sethia, S.K.; Behla, R.K. Phosphorus uptake and efficiency in different varieties of bread wheat (Triticum aestivum L.). Arch. Agron. Soil Sci. 2004, 50, 563–572. [Google Scholar] [CrossRef]
- Kosmas, C.; Vassiliou, P.; Dolopoulou, C.; Gouvela, T. Spatial Analysis of Soil Characteristics of Aliartos Field; Agricultural University of Athens: Athens, Greece, 2015. (In Greek) [Google Scholar]
- Haneklaus, S.; Schnug, E. An agronomic, ecological and economic assessment of site-specific fertilisation. Landbauforsch. Völkenrode 2002, 52, 123–133. [Google Scholar]
- Friesen, D.K. Influence of co-granulated nutrients and granule size on plant response to elemental sulfur in compound fertilizers. Nutr. Cycl. Agroecosyst. 1996, 46, 81–90. [Google Scholar] [CrossRef]
- Degryse, F.; da Silva, R.C.; Baird, R.; Beyrer, T.; Below, F.; McLaughlina, M.J. Uptake of elemental or sulfate-S from fall- or spring-applied co-granulated fertilizer by corn—A stable isotope and modeling study. Field Crops Res. 2018, 221, 322–332. [Google Scholar] [CrossRef]
- Chien, S.H.; Prochnow, L.I.; Tu, S.; Synder, C.S. The agronomic and environmental aspects of phosphate fertilizers varying in source and solubility: An update review. Nutr. Cycl. Agroecosyst. 2011, 89, 229–255. [Google Scholar] [CrossRef]
- Janzen, H.H.; Bettany, J.R. Measurement of sulfur oxidation in soils. Soil Sci. 1987, 143, 444–452. [Google Scholar] [CrossRef]
- Cantarella, H.; Mattos Jr, D.; Quaggio, J.A.; Rigolin, A.T. Fruit yield of Valencia sweet orange fertilized with different N sources and the loss of applied N. Nutr. Cycl. Agroecosyst. 2003, 67, 215–223. [Google Scholar] [CrossRef]
- Shivay, Y.S.; Pooniya, V.; Prasad, R.; Pal, M.; Bansal, R. Sulphur-coated urea as a source of sulphur and an enhanced efficiency of N fertilizer for spring wheat. Cereal Res. Commun. 2016, 44, 513–523. [Google Scholar] [CrossRef]
- Ding, W.X.; Chen, Z.M.; Yu, H.Y.; Luo, J.F.; Yoo, G.Y.; Xiang, J.; Zhang, H.J.; Yuan, J.J. Nitrous oxide emission and nitrogen use efficiency in response to nitrophosphate, N-(n-butyl) thiophosphoric triamide and dicyandiamide of a wheat cultivated soil under sub-humid monsoon conditions. Biogeosciences 2015, 12, 803–815. [Google Scholar] [CrossRef]
- Zanin, L.; Venuti, S.; Tomasi, N.; Zamboni, A.; De Brito Francisco, R.M.; Varanini, Z.; Pinton, R. Short-term treatment with the uease inhibitor N-(n-Butyl) Thiophosphoric Triamide (NBPT) alters urea assimilation and modulates transcriptional profiles of genes involved in primary and secondary metabolism in maize seedlings. Front. Plant Sci. 2016, 8, 845. [Google Scholar]
- Zaman, M.; Nguyen, M.L.; Blennerhassett, J.D.; Quin, B.F. Reducing NH3, N2O and NO3−–N losses from a pasture soil with urease or nitrification inhibitors and elemental S-amended nitrogenous fertilizers. Biol. Fertil. Soils 2008, 44, 693–705. [Google Scholar] [CrossRef]
| Group | Field Trial | Area ha | Seed kg ha−1 | Cultivar | F1 kg ha−1 | Fertilizer Type | F2a kg ha−1 | Fertilizer Type | F2b kg ha−1 | Fertilizer Type | Δx/x (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 2015–2016 | |||||||||||
| A1 | FT_13C | 1.4 | 280 | Meridiano | 250 | 16-20-0 | 120 | 46-0-0 | |||
| FT_13S | 1.4 | 280 | Meridiano | 250 | 16-20-0/ES | 120 | 46-0-0 | 28.5 | |||
| FT_08C | 1.4 | 250 | Quadrato | 300 | 20-10-0 | 200 | 40-0-0 | ||||
| FT_08S | 1.4 | 250 | Quadrato | 300 | 20-10-0/ES | 200 | 40-0-0 | 26.9 | |||
| FT_03C | 1.5 | 250 | Quadrato | 300 | 16-20-0 | 250 | 40-0-0 | ||||
| FT_03S | 1.5 | 250 | Quadrato | 300 | 16-20-0/ES | 250 | 40-0-0/ES | 18.5 | |||
| FT_12C | 1.35 | 250 | Simeto | 250 | 10-20-0 | 100 | 46-0-0 | 100 | 34.5-0-0 | ||
| FT_12S | 1.35 | 250 | Simeto | 250 | 10-20-0/ES | 100 | 46-0-0 | 100 | 34.5-0-0 | 8.1 | |
| FT_07C | 1.4 | 220 | Normano | 280 | 20-10-0 | 250 | 45-0-0 | ||||
| FT_07S | 1.4 | 220 | Normano | 280 | 20-10-0/ES | 250 | 45-0-0 | −10.9 | |||
| A2 | FT_02C | 1.0 | 250 | Quadrato | 300 | 16-20-0 | 100 | 40-0-0 | 150 | 34.5-0-0 | |
| FT_02S | 1.0 | 250 | Quadrato | 300 | 16-20-0/ES | 100 | 40-0-0 | 150 | 34.5-0-0 | 7.8 | |
| FT_10C | 1.1 | 280 | Meridiano | 300 | 16-20-0 | 220 | 40-0-0 | ||||
| FT_10S | 1.1 | 280 | Meridiano | 300 | 16-20-0/ES | 220 | 40-0-0 | 4.2 | |||
| FT_11C | 1.5 | 280 | Normano | 300 | 16-20-0 | 300 | 40-0-0 | ||||
| FT_11S | 1.5 | 280 | Normano | 300 | 16-20-0/ES | 300 | 40-0-0 | 2.7 | |||
| FT_01C | 0.9 | 250 | Simeto | 275 | 16-20-0 | 180 | 40-0-0 | 180 | 40-0-0 | ||
| FT_01S | 0.9 | 250 | Simeto | 275 | 16-20-0/ES | 180 | 40-0-0/ES | 180 | 40-0-0/ES | −3.4 | |
| FT_09 | 1.5 | 250 | Meridiano | 300 | 16-20-0 | 200 | 40-0-0 | ||||
| FT_09 | 1.5 | 250 | Meridiano | 300 | 16-20-0/ES | 200 | 40-0-0/ES | −26.8 | |||
| 2016–2017 | |||||||||||
| A3 | FT_6C | 1.3 | 280 | Meridiano | 300 | 16-20-0 | 200 | 40-0-0 | |||
| FT_6S | 1.3 | 280 | Meridiano | 300 | 16-20-0/ES | 200 | 40-0-0/ES | 9.5 | |||
| FT_7C | 1.5 | 280 | Meridiano | 300 | 16-20-0 | 200 | 40-0-0 | ||||
| FT_7S | 1.5 | 280 | Meridiano | 300 | 16-20-0/ES | 200 | 40-0-0/ES | 3.2 | |||
| FT_3C | 1.5 | 280 | Meridiano | 300 | 16-20-0 | 200 | 40-0-0 | ||||
| FT_3S | 1.5 | 280 | Meridiano | 300 | 16-20-0/ES | 200 | 40-0-0/ES | −13.0 | |||
| FT_4C | 0.7 | 250 | Simeto | 300 | 20-10-0 | 230 | 45-0-0 | ||||
| FT_4S | 0.7 | 250 | Simeto | 300 | 20-10-0/ES | 230 | 45-0-0 | −5.9 | |||
| FT_5C | 0.9 | 250 | Simeto | 300 | 20-10-0 | 220 | 40-0-0 | ||||
| FT_5S | 0.9 | 250 | Simeto | 300 | 20-10-0/ES | 220 | 40-0-0 | −18.0 | |||
| Group | Field Trial | Sand (%) | Silt (%) | Clay (%) | Type | pH | ECe μS/cm | NO3−N mg/kg | YF | YFBS | Δx/x (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mg ha−1 | |||||||||||
| 2015–2016 | |||||||||||
| A1 | FT_13 | 34 | 30 | 36 | CL | 6.20 | 479 | 20.2 | 5.19 | 6.67 | 28.5 |
| FT_08 | 24 | 30 | 46 | C | 7.53 | 505 | 16.0 | 3.60 | 4.57 | 26.9 | |
| FT_03 | 26 | 24 | 50 | C | 6.95 | 352 | 11.5 | 4.65 | 5.51 | 18.5 | |
| FT_12 | 22 | 44 | 34 | CL | 7.17 | 603 | 25.7 | 4.30 | 4.65 | 8.1 | |
| FT_07 | 22 | 44 | 34 | CL | 6.65 | 386 | 17.1 | 4.60 | 4.10 | −10.9 | |
| A2 | FT_02 | 28 | 40 | 32 | CL | 8.02 | 572 | 15.6 | 6.16 | 6.64 | 7.8 |
| FT_10 | 28 | 34 | 38 | CL | 8.10 | 453 | 9.1 | 4.30 | 4.48 | 4.2 | |
| FT_11 | 34 | 38 | 28 | CL | 7.96 | 675 | 16.6 | 4.40 | 4.52 | 2.7 | |
| FT_01 | 28 | 30 | 42 | C | 8.01 | 613 | 24.6 | 5.06 | 4.89 | −3.4 | |
| FT_09 | 10 | 42 | 48 | SiC | 8.00 | 592 | 23.2 | 3.32 | 2.43 | −26.8 | |
| 2016–2017 | |||||||||||
| A3 | FT_6 | 30 | 34 | 36 | CL | 7.81 | 667 | 23.1 | 1.68 | 1.84 | 9.5 |
| FT_7 | 14 | 40 | 46 | SiC/C | 7.88 | 610 | 25.5 | 4.73 | 4.88 | 3.2 | |
| FT_3 | 16 | 38 | 46 | C | 7.74 | 491 | 10.5 | 2.62 | 2.28 | −13.0 | |
| FT_4 | 26 | 28 | 46 | C | 7.80 | 684 | 19.6 | 1.53 | 1.44 | −5.9 | |
| FT_5 | 30 | 28 | 42 | C | 7.74 | 571 | 19.9 | 1.78 | 1.46 | −18.0 | |
| B1 | 23 | 25 | 52 | C | 8.57 | 543 | 19.2 | ||||
| B2 | 23 | 24 | 53 | C | 8.13 | 527 | 17.1 | ||||
| Field | CaCO3 | P-Olsen | Kexch | Fe-DTPA | Mn-DTPA | Cu-DTPA | Zn-DTPA | SOM | HS | HS/SOM | |
| Trial | (%) | mg/kg | mg/kg | mg/kg | mg/kg | mg/kg | mg/kg | (%) | (%) | (%) | |
| 2015–2016 | |||||||||||
| A1 | FT_13 | 0 | 19.4 | 230 | 22.7 | 13.4 | 1.5 | 0.8 | 1.5 | 0.2 | 11.7 |
| FT_08 | 0 | 16.8 | 300 | 17.7 | 12.1 | 1.5 | 0.7 | 2.3 | 0.5 | 21.1 | |
| FT_03 | 0 | 26.0 | 230 | 26.3 | 13.4 | 4.7 | 1.8 | 1.8 | 0.2 | 12.7 | |
| FT_12 | 0 | 17.4 | 270 | 25.8 | 12.2 | 2.2 | 1.8 | 1.9 | 0.2 | 8.1 | |
| FT_07 | 0 | 6.4 | 180 | 27.3 | 13.4 | 2.2 | 0.7 | 2.3 | 0.3 | 14.3 | |
| A2 | FT_02 | 5.2 | 65.4 | 240 | 13.0 | 8.4 | 1.5 | 1.1 | 2.1 | 0.2 | 9.9 |
| FT_10 | 49.1 | 12.2 | 390 | 15.9 | 9.3 | 1.8 | 0.7 | 1.8 | 0.1 | 2.8 | |
| FT_11 | 6.6 | 12.4 | 270 | 11.2 | 7.3 | 1.1 | 0.7 | 1.8 | 0.1 | 5.0 | |
| FT_01 | 24.5 | 7.8 | 270 | 5.8 | 6.3 | 1.4 | 0.8 | 1.8 | 0.1 | 6.7 | |
| FT_09 | 26.6 | 3.2 | 290 | 8.0 | 6.2 | 1.3 | 0.5 | 2.9 | 0.1 | 2.4 | |
| 2016–2017 | |||||||||||
| A3 | FT_6 | 41.0 | 15.6 | 450 | 5.8 | 9.6 | 1.7 | 0.8 | 2.4 | 0.1 | 4.2 |
| FT_7 | 1.4 | 9.8 | 350 | 14.8 | 10.1 | 1.5 | 0.7 | 3.7 | 0.4 | 10.8 | |
| FT_3 | 13.9 | 4.1 | 420 | 12.6 | 7.5 | 1.2 | 0.5 | 3.0 | 0.2 | 7.8 | |
| FT_4 | 11.1 | 2.8 | 240 | 10.1 | 7.3 | 0.9 | 0.9 | 1.9 | 0.1 | 7.2 | |
| FT_5 | 8.9 | 1.6 | 240 | 8.0 | 6.4 | 0.7 | 0.9 | 2.1 | 0.2 | 9.4 | |
| B1 | 18.1 | 20.2 | 230 | 24.3 | 12.1 | 1.8 | 0.9 | 2.5 | 0.2 | 9.6 | |
| B2 | 21.7 | 13.1 | 224 | 21.3 | 11.7 | 2.0 | 0.9 | 2.3 | 0.2 | 9.9 | |
| Treatment | Group B1 | Group B2 | Group B1 | Group B2 | ||||||
| P-Olsen (ppm) | P-Olsen (ppm) | |||||||||
| 18–22 | 12–15 | 18–22 | 12–15 | |||||||
| Grain yield (Mg ha−1) | Yield relative to the combination F; U (%) | |||||||||
| no fertilization | 1.75 | 1.30 | −25 | −10 | ||||||
| F | FBES | RC (%) | F | FBES | RC (%) | F | FBES | F | FBS0 | |
| U | 2.32 | 2.51 | 8.2 | 1.44 | 1.52 | 5.6 | 0 | 8 | 0 | 6 |
| U/ES | 2.90 | 3.19 | 10.0 | 1.53 | 1.71 | 11.8 | 25 | 38 | 6 | 19 |
| U/NBPT | 2.78 | 2.98 | 7.2 | 1.64 | 1.76 | 7.3 | 20 | 28 | 14 | 22 |
| U/NBPT/ES | 3.04 | 3.54 | 16.4 | 1.74 | 1.93 | 10.9 | 31 | 53 | 21 | 34 |
| UAS | 2.68 | 3.18 | 18.7 | 1.50 | 1.58 | 5.3 | 16 | 37 | 4 | 10 |
| UAS/ES | 3.00 | 3.90 | 30.0 | 1.64 | 1.78 | 8.5 | 29 | 68 | 14 | 24 |
| UAS/NBPT | 2.95 | 3.45 | 16.9 | 2.09 | 2.25 | 7.7 | 27 | 49 | 45 | 56 |
| UAS/NBPT/ES | 3.52 | 4.22 | 19.9 | 2.27 | 2.49 | 9.7 | 52 | 82 | 58 | 73 |
| Variable | Sum Sq | Mean Sq | NumDF | DenDF | P-Value |
|---|---|---|---|---|---|
| P-Olsen | 11.6159 | 11.6159 | 1 | 87 | <0.001 |
| IF | 3.0111 | 1.5056 | 2 | 87 | <0.001 |
| AF | 10.5053 | 1.5008 | 7 | 87 | <0.001 |
| P-Olsen:IF | 1.7335 | 0.8667 | 2 | 87 | 0.0013 |
| Variable | Beta | 95% CI | P-Value | |
|---|---|---|---|---|
| Intercept | 2.38 | 2.14 | 2.61 | <0.001 |
| P-Olsen; Low | −1.17 | −1.35 | −0.98 | <0.001 |
| AF; UAS/ES/NBPT | 1.18 | 0.91 | 1.44 | <0.001 |
| AF; UAS/NBPT | 0.74 | 0.47 | 1.00 | <0.001 |
| P-Olsen; Low: IF; NF | 0.72 | 0.16 | 1.28 | 0.019 |
| AF; UAS/ES | 0.63 | 0.37 | 0.90 | <0.001 |
| IF; FBES | 0.47 | 0.29 | 0.66 | <0.001 |
| IF; NF | −0.63 | −1.06 | −0.19 | 0.009 |
| AF; U/ES/NBPT | 0.61 | 0.35 | 0.88 | <0.001 |
| AF; U/ES | 0.38 | 0.12 | 0.65 | 0.008 |
| AF; U/NBPT | 0.34 | 0.08 | 0.61 | 0.018 |
| AF; UAS | 0.29 | 0.02 | 0.55 | 0.046 |
| P-Olsen; Low: IF; FBES | −0.33 | −0.59 | −0.06 | 0.024 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouranis, D.L.; Gasparatos, D.; Zechmann, B.; Bouranis, L.D.; Chorianopoulou, S.N. The Effect of Granular Commercial Fertilizers Containing Elemental Sulfur on Wheat Yield under Mediterranean Conditions. Plants 2019, 8, 2. https://doi.org/10.3390/plants8010002
Bouranis DL, Gasparatos D, Zechmann B, Bouranis LD, Chorianopoulou SN. The Effect of Granular Commercial Fertilizers Containing Elemental Sulfur on Wheat Yield under Mediterranean Conditions. Plants. 2019; 8(1):2. https://doi.org/10.3390/plants8010002
Chicago/Turabian StyleBouranis, Dimitris L., Dionisios Gasparatos, Bernd Zechmann, Lampros D. Bouranis, and Styliani N. Chorianopoulou. 2019. "The Effect of Granular Commercial Fertilizers Containing Elemental Sulfur on Wheat Yield under Mediterranean Conditions" Plants 8, no. 1: 2. https://doi.org/10.3390/plants8010002
APA StyleBouranis, D. L., Gasparatos, D., Zechmann, B., Bouranis, L. D., & Chorianopoulou, S. N. (2019). The Effect of Granular Commercial Fertilizers Containing Elemental Sulfur on Wheat Yield under Mediterranean Conditions. Plants, 8(1), 2. https://doi.org/10.3390/plants8010002








