Chemical Constituents and Antifungal Activity of Ficus hirta Vahl. Fruits
Abstract
:1. Introduction
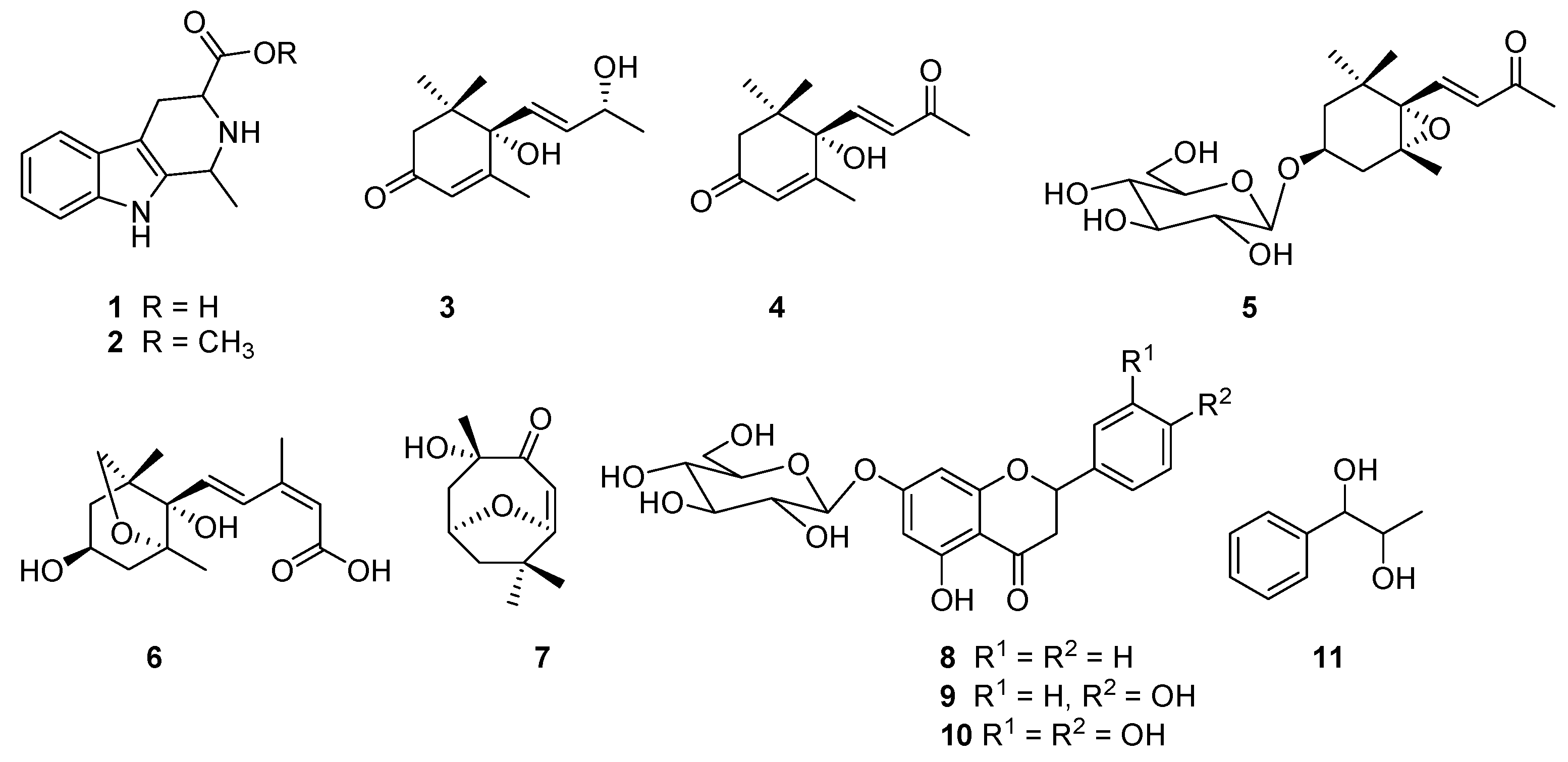
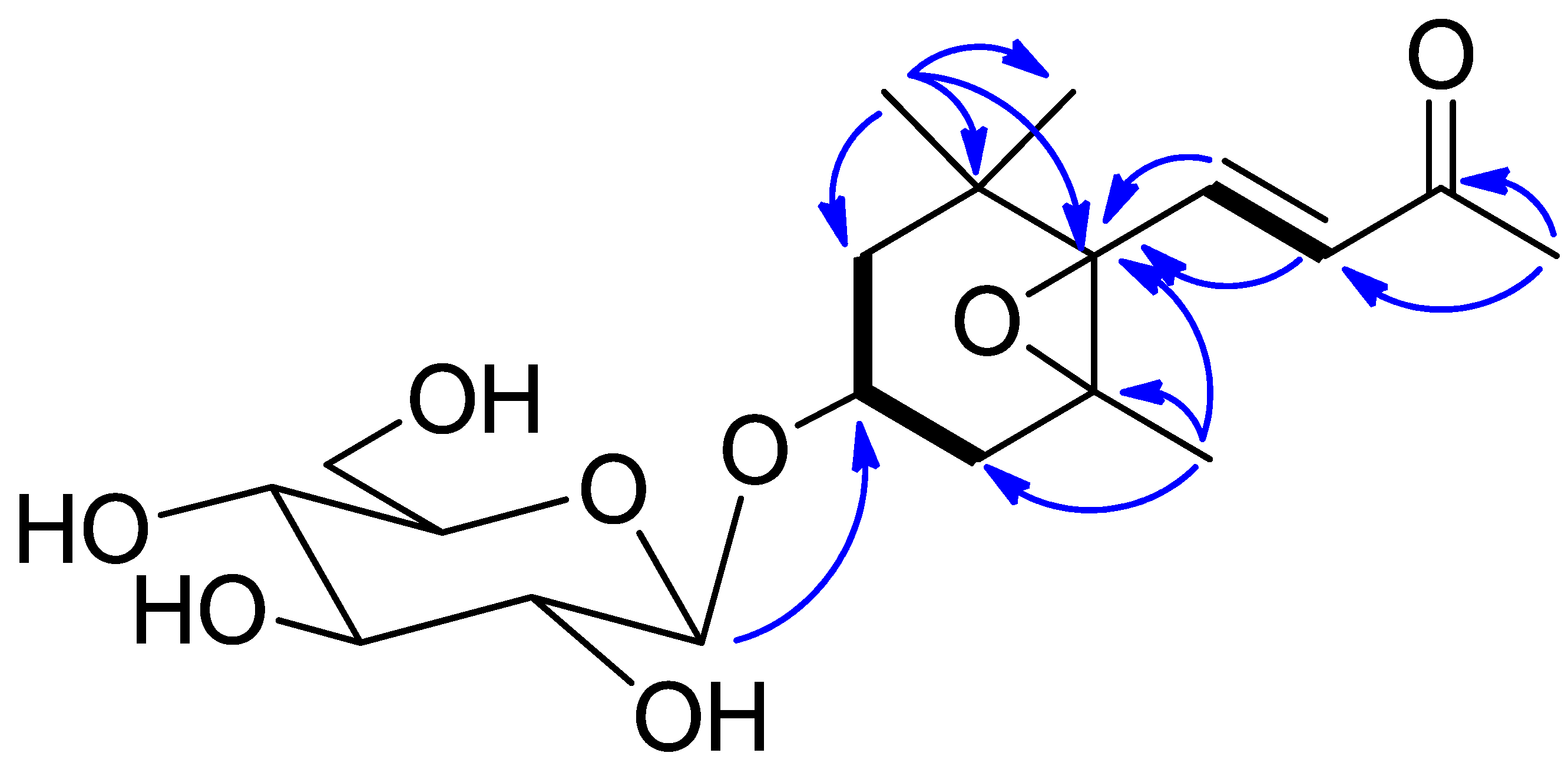
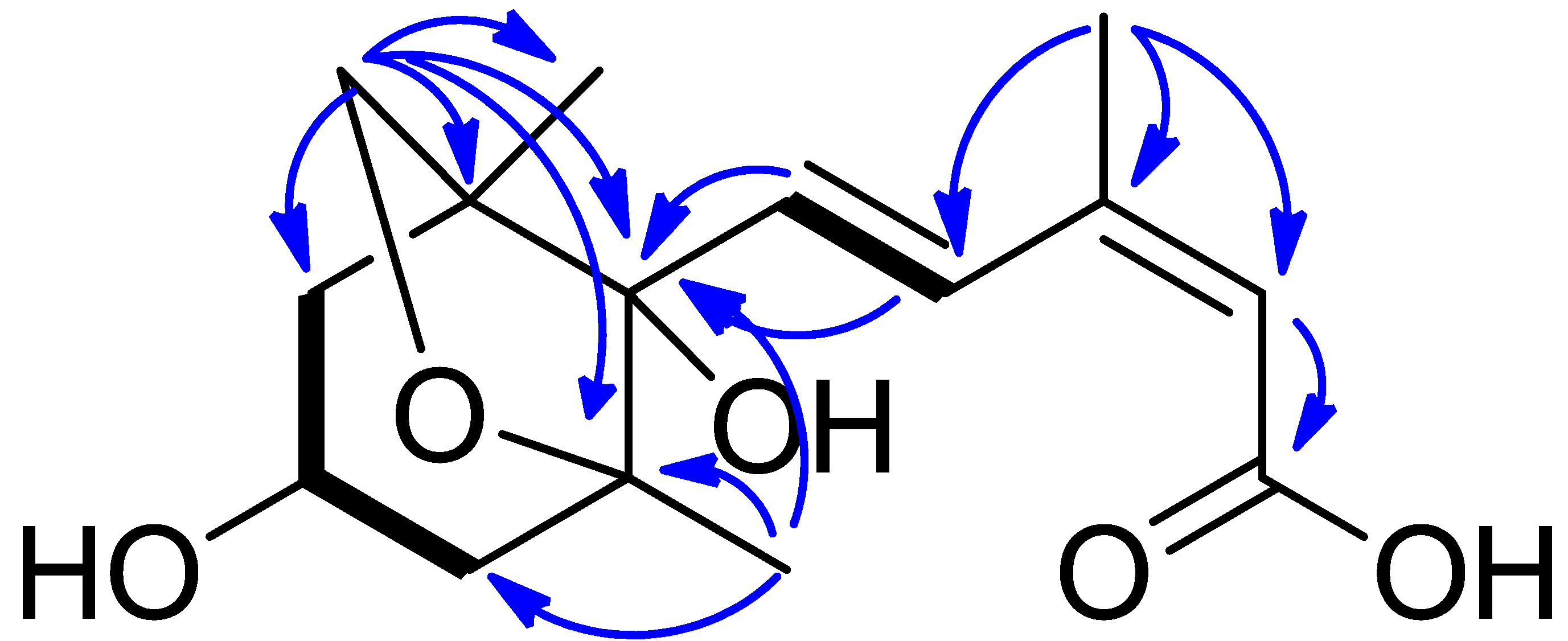
2. Results
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Equipment and Reagents
4.3. Extraction and Chromatography
4.4. Antifungal Activity Test
4.5. NMR and MS Data of Compounds 1–11
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Flora Compilation Committee of Chinese Academy of Science. Flora of China; Science Press: Beijing, China, 1998; Volume 23, pp. 67–160. [Google Scholar]
- Li, C.; Bu, P.B.; Qiu, D.K.; Sun, Y.F. Chemical constituents from roots of Ficus hirta. China J. Chin. Mater. Med. 2006, 31, 131–133. [Google Scholar]
- Ya, J.; Zhang, X.Q.; Wang, Y.; Li, Y.; Ye, W. Studies on flavonoids and coumarins in the roots of Ficus hirta Vahl. Chem. Ind. For. Prod. 2008, 28, 49–52. [Google Scholar]
- Ya, J.; Zhang, X.Q.; Wang, G.C. Flavonoids from the roots of Ficus hirta Vahl. Asia Chem. Lett. 2009, 13, 21–26. [Google Scholar]
- Ya, J.; Zhang, X.Q.; Wang, Y.; Zhang, Q.W.; Chen, J.X.; Ye, W.C. Two new phenolic compounds from the roots of Ficus hirta. Nat. Prod. Res. 2010, 24, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.P.; Di, B.; Feng, F. Chemical constituents from the roots of Ficus hirta. Pharm. Clin. Res. 2008, 16, 5–7. [Google Scholar]
- Zheng, R.R.; Ya, J.; Wang, W.J.; Yang, H.B.; Zhang, Q.W.; Zhang, X.Q.; Ye, W.C. Chemical studies on roots of Ficus hirta. China J. Chin. Mater. Med. 2013, 38, 3696–3701. [Google Scholar]
- Jiang, B.; Liu, Z.Q.; Zeng, Y.E.; Xu, H. Chemical constituents roots of Ficus hirta. Chin. Tradit. Herb. Drugs 2005, 36, 1141–1142. [Google Scholar]
- Yi, T.; Chen, Q.; He, X.; So, S.; Lo, Y.; Fan, L.; Chen, H. Chemical quantification and antioxidant assay of four active components in Ficus hirta root using UPLC-PAD-MS fingerprinting combined with cluster analysis. Chem. Cent. J. 2013, 7, 115. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Liu, X.; Lv, Z.; Peng, Y.H. Effects of Ficus hirta Vahl.(Wuzhimaotao) extracts on growth inhibition of HeLa cells. Exp. Toxicol. Pathol. 2012, 64, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.N.; Wang, Y.; Tang, L.H.; Liu, D.D.; Hou, S.Z.; Deng, X.C.; Ye, M.R. Study of Radix Fici hirtae on anti-inflammatory analgesic and effect of acute liver injury of mice. Pharm. Today 2008, 18, 55–58. [Google Scholar]
- Zhou, T.N.; Tang, L.H.; Huang, S.C.; Lu, D.D.; Wang, Y.; Liu, L.F.; Ye, M.R. Study on the antitussive and antiasthmatic effects of radix Fici hirtae. J. Chin. Med. Mater. 2009, 32, 571–574. [Google Scholar]
- Jia, F.L.; Ruan, M. Radix Fici hirtae protective effect of aqueous extract on acute hepatic injury in mice induced by two methyl farmamide. J. Chin. Med. Mater. 2008, 31, 1364–1368. [Google Scholar]
- Wang, X.P.; Duan, L.J.; Huang, X.; Cen, Y.W.; Li, G.F. Protective Role Of Aqueous Extract From Fici hirtae radix for DNA damage of bone marrow cells by~(60) Coγ-ray in mice. Chin. J. Mod. Appl. Pharm. 2011, 28, 284–287. [Google Scholar]
- Zhou, T.N.; Wang, Y.; Liu, D.D.; Tang, L.H.; Xiao, X.J.; Liu, L.F.; Ye, M.R. Experimental study on the tonic effect of different extracts from Radix Fici hirtae. J. Chin. Med. Mater. 2009, 32, 753–757. [Google Scholar]
- Chen, Q.; Ye, S.X.; Yu, J. Antibacterial activity of Radix Fici hirtae by chromotest microassay. Med. Plant 2012, 3, 13–16. [Google Scholar]
- Chen, C.; Wan, C.; Peng, X.; Chen, Y.; Chen, M.; Chen, J. Optimization of antifungal extracts from Ficus hirta fruits using response surface methodology and antifungal activity tests. Molecules 2015, 20, 19647–19649. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Han, J.; Chen, C.; Yao, L.; Chen, J.; Yuan, T. Monosubstituted benzene derivatives from fruits of Ficus hirta and their antifungal activity against phytopathogen Penicillium italicum. J. Agric. Food Chem. 2016, 64, 5621–5624. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Peng, X.; Zeng, R.; Chen, M.; Wan, C.; Chen, J. Ficus hirta fruits extract incorporated into an alginate-based edible coating for Nanfeng mandarin preservation. Sci. Hortic. 2016, 202, 41–48. [Google Scholar] [CrossRef]
- Wei, W.; Fan, C.L.; Wang, G.Y.; Tang, H.J.; Wang, Y.; Ye, W.C. Chemical constituents from Ficus pumila. Chin. Tradit. Herb. Drugs 2014, 45, 615–621. [Google Scholar]
- Zeng, Y.; Zhang, Y.; Weng, Q.; Hu, M.; Zhong, G. Cytotoxic and insecticidal activities of derivatives of harmine, a natural insecticidal component isolated from Peganum harmala. Molecules 2010, 15, 7775–7791. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.Y.; Duan, J.A.; Li, P.; Qian, S.H. Chemical constituents of Glechoma longituba. Acta Pharm. Sin. 2006, 41, 431–434. [Google Scholar]
- Woo, K.W.; Lee, K.R. Phytochemical constituents of Allium victorialis var. platyphyllum. Nat. Prod. Sci. 2013, 19, 221–226. [Google Scholar]
- Cai, L.; Liu, C.S.; Fu, X.W.; Shen, X.J.; Yin, T.P.; Yang, Y.B.; Ding, Z.T. Two new glucosides from the pellicle of the walnut (Juglans regia). Nat. Prod. Bioprospect. 2012, 2, 150–153. [Google Scholar] [CrossRef]
- He, J.B.; Niu, Y.F.; Li, J.X.; Wang, L.B.; Zi, T.P.; Yu, S.; Tao, J. Studies on terpenoids from Zygophyllum fabago. China J. Chin. Mater. Med. 2015, 40, 4634–4638. [Google Scholar]
- Zhao, Q.; Liu, F.; Li, Q.J.; Chen, W.P. Chemical constituents from flowers of Rosa chinensis. Chin. Tradit. Herb. Drugs 2012, 43, 1484–1488. [Google Scholar]
- Ding, Y.X.; Guo, Y.J.; Ren, Y.L.; Dou, D.; Li, Q. Isolation of flavonoids from male flowers of Eucommia ulmoides and their anti-oxidantive activities. Chin. Tradit. Herb. Drugs 2014, 45, 323–327. [Google Scholar]
- Deng, R.X.; Zhang, C.F.; Liu, P.; Duan, W.L.; Yin, W.P. Separation and identification of flavonoids from Chinese Fringetree Flowers (Chionanthus retusa Lindl et Paxt). Food Sci. 2014, 35, 74–78. [Google Scholar]
- Mayorga, H.; Knapp, H.; Winterhalter, P.; Duque, C. Glycosidically bound flavor compounds of cape gooseberry (Physalis peruviana L.). J. Agric. Food Chem. 2001, 49, 1904–1908. [Google Scholar]
- Fan, M.S.; Ye, G.; Huang, C.G. The advances of chemistry and pharmacological study of Ficus genus. Nat. Prod. Res. Dev. 2005, 17, 497–504. [Google Scholar]
- El-Hela, A.; Mohammed, A.E.; Ragab, E.; Afifi, W. Chemical constituents and biological activity of Ficus platypoda (Miq.) leaves. J. Biomed. Pharm. Res. 2014, 3, 21–37. [Google Scholar]
- Kitajima, J.; Kimizuka, K.; Tanak, Y. Three new sesquiterpenoid glucosides of Ficus pumila fruit. Chem. Pharm. Bull. 2000, 48, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Somwong, P.; Suttisri, R.; Buakeaw, A. New sesquiterpenes and phenolic compound from Ficus foveolata. Fitoterapia 2013, 85, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Kuo, Y.H. A monoterpenoid and two simple phenols from heartwood of Ficus microcarpa. Phytochemistry 1998, 49, 2417–2419. [Google Scholar] [CrossRef]
- Kuo, Y.H.; Li, Y.C. Three new compounds, ficusone, ficuspirolide, and ficusolide from the heartwood of Ficus microcarpa. Chem. Pharm. Bull. 1999, 47, 299–301. [Google Scholar] [CrossRef]
- Ammar, S.; del Mar Contreras, M.; Belguith-Hadrich, O.; Bouaziz, M.; Segura-Carretero, A. New insights into the qualitative phenolic profile of Ficus carica L. fruits and leaves from Tunisia using ultra-high-performance liquid chromatography coupled to quadrupole-time-of-flight mass spectrometry and their antioxidant activity. RSC Adv. 2015, 26, 20035–20050. [Google Scholar] [CrossRef]
- Yang, S.; Liu, L.; Li, D.; Xia, H.; Su, X.; Peng, L.; Pan, S. Use of active extracts of poplar buds against Penicillium italicum and possible modes of action. Food Chem. 2016, 196, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Yang, S.; Cheng, Y.J.; Chen, F.; Pan, S.; Fan, G. Antifungal activity and action mode of pinocembrin from propolis against Penicillium italicum. Food Sci. Biotechnol. 2012, 6, 1533–1539. [Google Scholar] [CrossRef]
| Concentration (µg/mL) | Inhibition Rate |
|---|---|
| 25 | 13.70 ± 1.81 |
| 50 | 36.88 ± 1.08 |
| 100 | 56.10 ± 2.55 |
| 200 | 74.65 ± 1.61 |
| 400 | 92.05 ± 1.55 |
| 800 | 100 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wan, C.; Chen, C.; Li, M.; Yang, Y.; Chen, M.; Chen, J. Chemical Constituents and Antifungal Activity of Ficus hirta Vahl. Fruits. Plants 2017, 6, 44. https://doi.org/10.3390/plants6040044
Wan C, Chen C, Li M, Yang Y, Chen M, Chen J. Chemical Constituents and Antifungal Activity of Ficus hirta Vahl. Fruits. Plants. 2017; 6(4):44. https://doi.org/10.3390/plants6040044
Chicago/Turabian StyleWan, Chunpeng, Chuying Chen, Mingxi Li, Youxin Yang, Ming Chen, and Jinyin Chen. 2017. "Chemical Constituents and Antifungal Activity of Ficus hirta Vahl. Fruits" Plants 6, no. 4: 44. https://doi.org/10.3390/plants6040044
APA StyleWan, C., Chen, C., Li, M., Yang, Y., Chen, M., & Chen, J. (2017). Chemical Constituents and Antifungal Activity of Ficus hirta Vahl. Fruits. Plants, 6(4), 44. https://doi.org/10.3390/plants6040044






