Underlying Mechanisms of Increased Precipitation and Arbuscular Mycorrhizal (AM) Fungi on Plant Community by Mediating Soil Microbes in Desert Ecosystems
Abstract
1. Introduction
2. Results
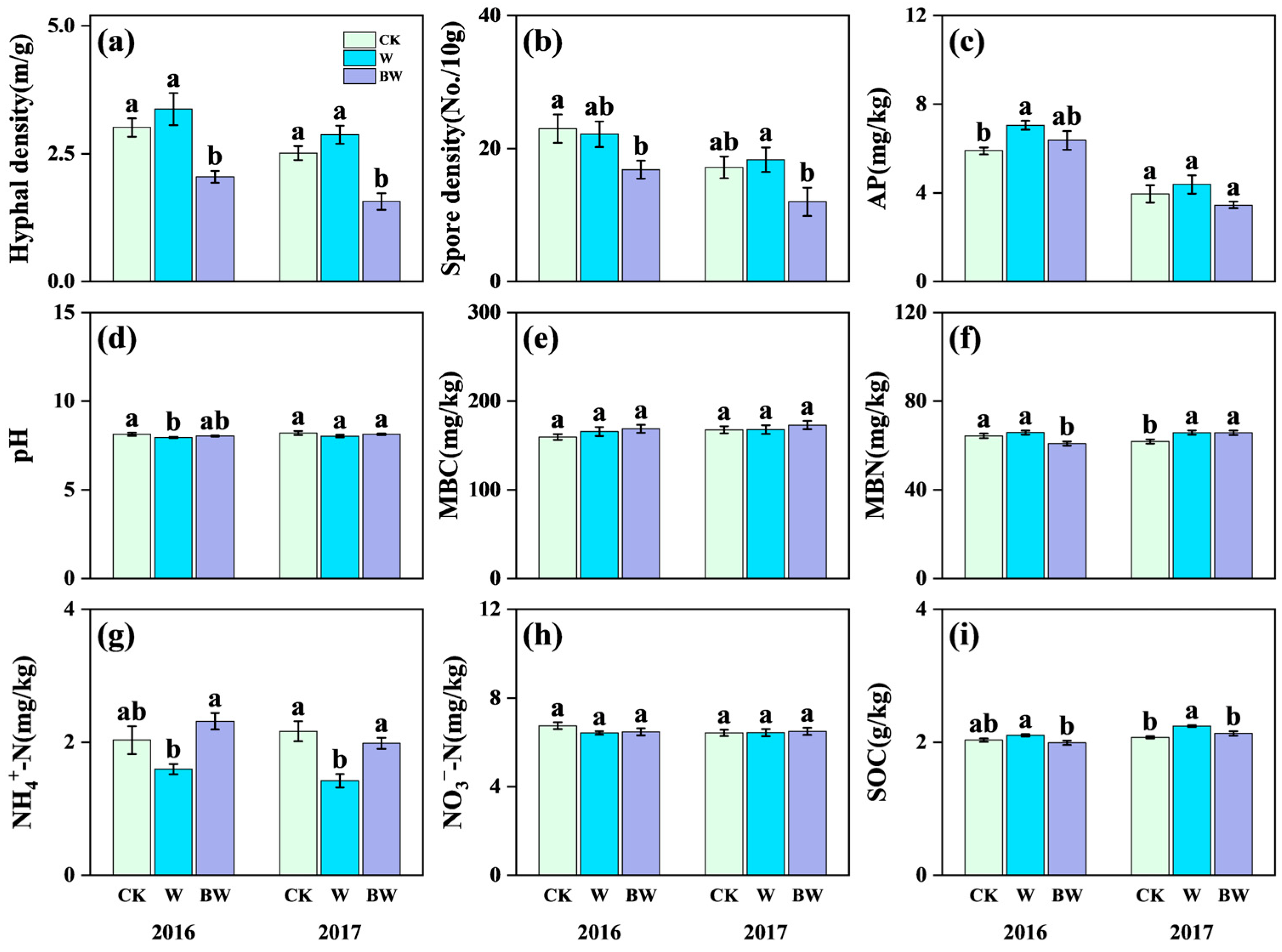
2.1. Responses of Soil Physicochemical Properties and Plant Community to Increased Precipitation and the Suppression of AMF
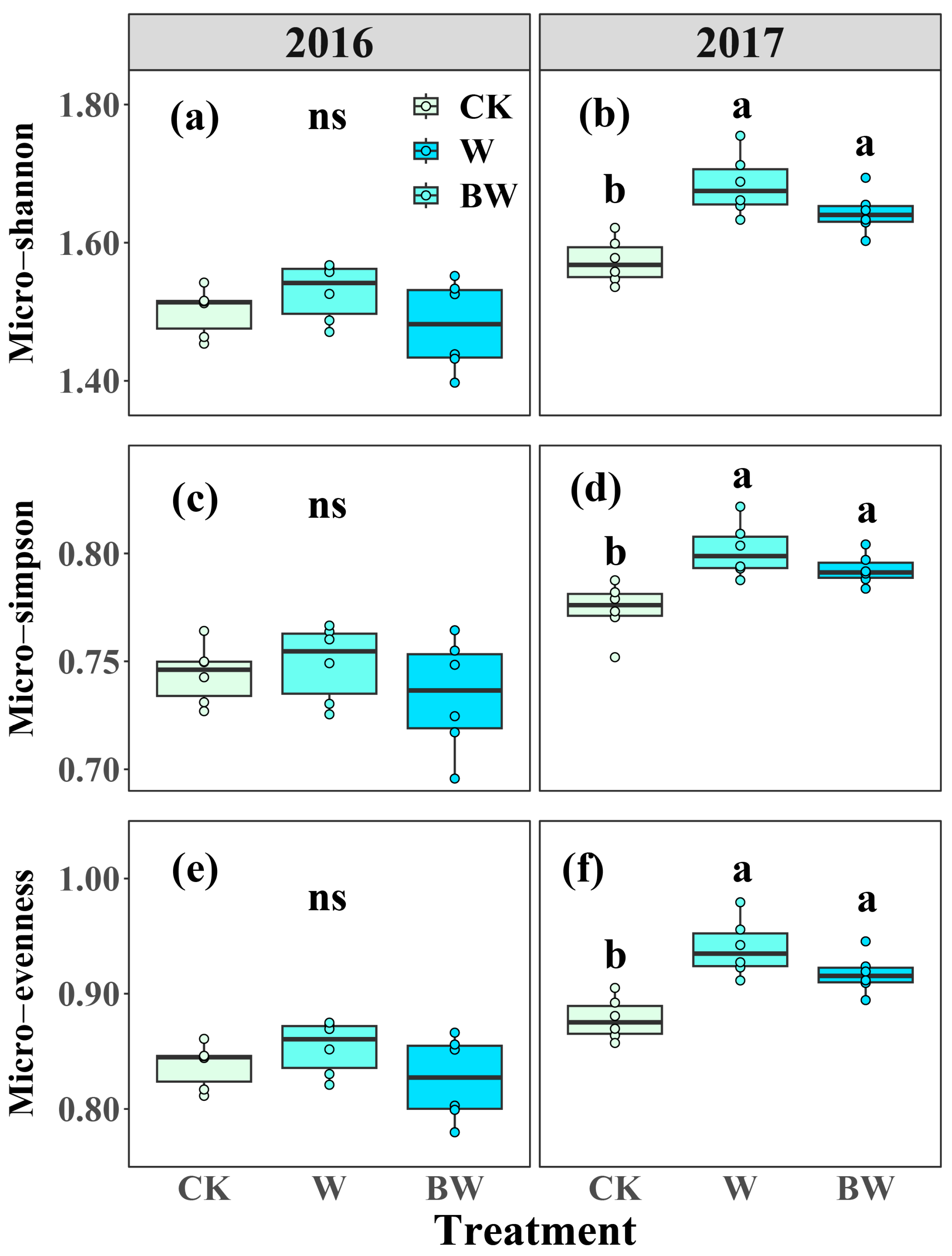
2.2. Responses of Soil Microbial Community to Increased Precipitation and the Suppression of AMF
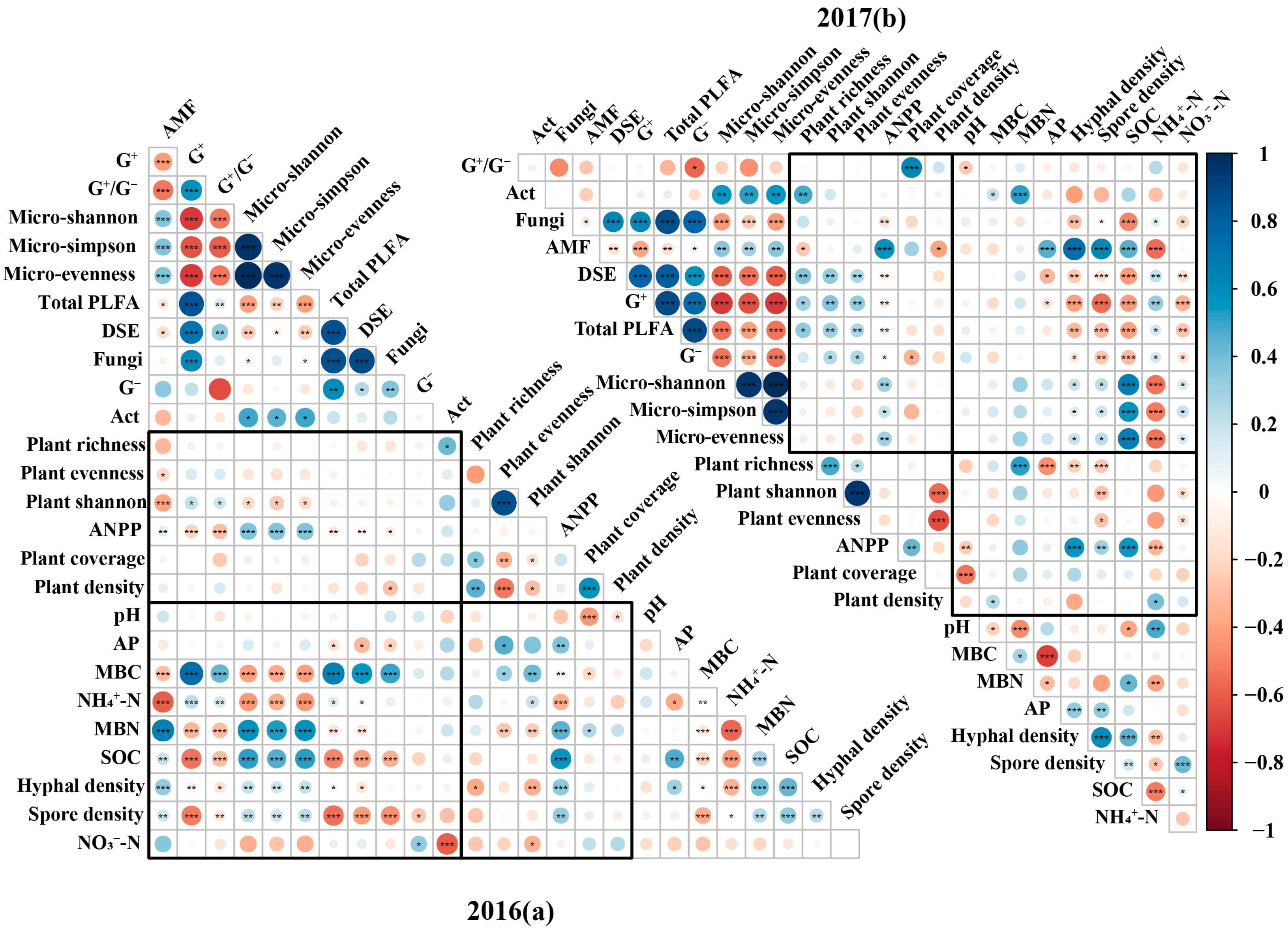
2.3. Underlying Mechanisms of Soil Microbial-Community-Mediated Effects of Increased Precipitation and AMF on Plant Community
3. Discussion
3.1. The Influence Mechanisms of Increased Precipitation on Soil Microbial Community
3.2. The Influence Mechanisms of Arbuscular Mycorrhizal Fungi on Soil Microbial Community
3.3. The Influence Mechanisms of Increased Precipitation and Arbuscular Mycorrhizal Fungi on Plant Community Through Altering Soil AMF and G+
4. Materials and Methods
4.1. Study Site
4.2. Experimental Design
4.3. PLFA Determination and Plant Community and Soil Parameters Measurements
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Xie, Z.J.; Fu, Y.Y.; He, H.S.; Wang, S.Q.; Wang, L.C.; Liu, C. Increases in extreme precipitation expected in Northeast China under continued global warming. Clim. Dyn. 2024, 62, 4943–4965. [Google Scholar] [CrossRef]
- Stott, P. How climate change affects extreme weather events Research can increasingly determine the contribution of climate change to extreme events such as droughts. Science 2016, 352, 1517–1518. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2021: The Physical Science Basis; AR6; IPCC: Geneva, Switzerland, 2021. [Google Scholar]
- Al-Rowaily, S.L.; Al-Dosari, D.H.; Assaeed, A.M.; Abd-ElGawad, A.M.; El-Sheikh, M.A.; El-Bana, M.I.; Al-Taisan, W.A. Native Perennial Plants Colonizing Abandoned Arable Fields in a Desert Area: Population Structure and Community Assembly. Agriculture 2020, 10, 550. [Google Scholar] [CrossRef]
- Huxman, T.E.; Smith, M.D.; Fay, P.A.; Knapp, A.K.; Shaw, M.R.; Loik, M.E.; Smith, S.D.; Tissue, D.T.; Zak, J.C.; Weltzin, J.F.; et al. Convergence across biomes to a common rain-use efficiency. Nature 2004, 429, 651–654. [Google Scholar] [CrossRef]
- Feng, W.; Lindner, H.; Robbins, N.E.; Dinneny, J.R. Growing Out of Stress: The Role of Cell- and Organ-Scale Growth Control in Plant Water-Stress Responses. Plant Cell 2016, 28, 1769–1782. [Google Scholar] [CrossRef]
- Kushwaha, A.; Bhatnagar, R.; Kumar, P.; Zucca, C.; Srivastava, S.; Ajai. Long Term Monitoring of Ecological Status of Major Deserts of the World. J. Indian Soc. Remote Sens. 2024, 52, 1819–1839. [Google Scholar] [CrossRef]
- Na, X.F.; Yu, H.L.; Wang, P.; Zhu, W.W.; Niu, Y.B.; Huang, J.Y. Vegetation biomass and soil moisture coregulate bacterial community succession under altered precipitation regimes in a desert steppe in northwestern China. Soil Biol. Biochem. 2019, 136, 107520. [Google Scholar] [CrossRef]
- Dong, Q.Q.; Ji, Z.Q.; Wang, H.; Duan, W.; Cao, W.L.; Li, W.S.; Jia, Y.Y. Interactive Effects of Precipitation and Nitrogen on Soil Microbial Communities in a Desert Ecosystem. Microorganisms 2025, 13, 1393. [Google Scholar] [CrossRef] [PubMed]
- Ji, Z.Q.; Dong, Q.Q.; Yang, R.; Qin, W.H.; Peng, Y.; Jia, Y.Y. From Ordinary to Extraordinary: The Crucial Role of Common Species in Desert Plant Community Stability with Arbuscular Mycorrhizal (AM) Fungi Under Increased Precipitation. Plants 2025, 14, 1099. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Huang, Z.Z.; Chen, W.W.; Xing, A.; Zhao, S.X.; Wan, W.F.; Hu, H.N.; Li, H.G. Mild to moderate drought stress reinforces the role of functional microbiome in promoting growth of a dominant forage species (Neopallasia pectinata) in desert steppe. Front. Microbiol. 2024, 15, 1371208. [Google Scholar] [CrossRef]
- Li, Z.L.; Peng, Q.; Dong, Y.S.; Guo, Y. The influence of increased precipitation and nitrogen deposition on the litter decomposition and soil microbial community structure in a semiarid grassland. Sci. Total Environ. 2022, 844, 157115. [Google Scholar] [CrossRef]
- Zhou, Z.H.; Wang, C.K.; Luo, Y.Q. Meta-analysis of the impacts of global change factors on soil microbial diversity and functionality. Nat. Commun. 2020, 11, 3072. [Google Scholar] [CrossRef] [PubMed]
- Quiroga-González, C.; Prada-Salcedo, L.D.; Buscot, F.; Tarkka, M.; Herrmann, S.; Bouffaud, M.L.; Goldmann, K. Severe drought impacts tree traits and associated soil microbial communities of clonal oaks. Environ. Microbiome 2025, 20, 63. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.H.; Wang, Z.R.; Li, T.P.; Wang, C.; Dang, N.; Wang, R.Z.; Jiang, Y.; Wang, H.Y.; Li, H. N and P fertilization enhanced carbon decomposition function by shifting microbes towards an r-selected community in meadow grassland soils. Ecol. Indic. 2021, 132, 108306. [Google Scholar] [CrossRef]
- Wu, H.W.; Cui, H.L.; Fu, C.X.; Li, R.; Qi, F.Y.; Liu, Z.L.; Yang, G.; Xiao, K.Q.; Qiao, M. Unveiling the crucial role of soil microorganisms in carbon cycling: A review. Sci. Total Environ. 2024, 909, 168627. [Google Scholar] [CrossRef]
- Zheng, T.T.; Miltner, A.; Liang, C.; Nowak, K.M.; Kästner, M. Turnover of gram-negative bacterial biomass-derived carbon through the microbial food web of an agricultural soil. Soil Biol. Biochem. 2021, 152, 108070. [Google Scholar] [CrossRef]
- Derakhshan-Nejad, Z.; Lee, W.; Han, S.; Choi, J.; Yun, S.-T.; Lee, G. Effects of soil moisture content on CO2 triggered soil physicochemical properties in a near-surface environment. J. Soils Sediments 2020, 20, 2107–2120. [Google Scholar] [CrossRef]
- DerakhshanNejad, Z.; Yun, H.; Choi, J.; Yun, S.T.; Lee, G. Effects of soil moisture content on soil physicochemical properties exposed to CO2. Geol. Soc. Korea 2017, 10, 213. [Google Scholar]
- Chytry, M.; Danihelka, J.; Ermakov, N.; Hájek, M.; Hájková, P.; Koci, M.; Kubesová, S.; Lustyk, P.; Otypková, Z.; Popov, D.; et al. Plant species richness in continental southern Siberia: Effects of pH and climate in the context of the species pool hypothesis. Glob. Ecol. Biogeogr. 2007, 16, 668–678. [Google Scholar] [CrossRef]
- Rousk, J.; Bååth, E.; Brookes, P.C.; Lauber, C.L.; Lozupone, C.; Caporaso, J.G.; Knight, R.; Fierer, N. Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J. 2010, 4, 1340–1351. [Google Scholar] [CrossRef]
- Yoon, J.H.; Adhikari, M.; Jeong, S.S.; Lee, S.P.; Kim, H.S.; Lee, G.S.; Park, D.H.; Kim, H.; Yang, J.E. Microbial diversity of soils under different land use and chemical conditions. Appl. Biol. Chem. 2024, 67, 111. [Google Scholar] [CrossRef]
- Feng, K.; Deng, Y. Intertwined Relationship Between Soil pH and Microbes in Biogeography. Glob. Change Biol. 2025, 31, e70208. [Google Scholar] [CrossRef]
- Wang, M.M.; Sun, X.; Cao, B.C.; Chiariello, N.R.; Docherty, K.M.; Field, C.B.; Gao, Q.; Gutknecht, J.L.M.; Guo, X.; He, G.H.; et al. Long-term elevated precipitation induces grassland soil carbon loss via microbe-plant-soil interplay. Glob. Change Biol. 2023, 29, 5429–5444. [Google Scholar] [CrossRef]
- Yu, H.Y.; Li, L.; Ma, Q.H.; Liu, X.D.; Li, Y.B.; Wang, Y.H.; Zhou, G.S.; Xu, Z.Z. Soil microbial responses to large changes in precipitation with nitrogen deposition in an arid ecosystem. Ecology 2023, 104, e4020. [Google Scholar] [CrossRef]
- Hartmann, M.; Six, J. Soil structure and microbiome functions in agroecosystems. Nat. Rev. Earth Environ. 2023, 4, 4–18. [Google Scholar] [CrossRef]
- Wang, X.J.; Li, Z.; Li, Q.; Hu, Z.Q. Alleviation of Plant Abiotic Stress: Mechanistic Insights into Emerging Applications of Phosphate-Solubilizing Microorganisms in Agriculture. Plants 2025, 14, 1558. [Google Scholar] [CrossRef] [PubMed]
- Kakouridis, A.; Yuan, M.T.; Nuccio, E.E.; Hagen, J.A.; Fossum, C.A.; Moore, M.L.; Estera-Molina, K.Y.; Nico, P.S.; Weber, P.K.; Pett-Ridge, J.; et al. Arbuscular mycorrhiza convey significant plant carbon to a diverse hyphosphere microbial food web and mineral-associated organic matter. New Phytol. 2024, 242, 1661–1675. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Liao, S. Advances in the Research on the Effects of Arbuscular Mycorrhizal Fungi on Plant Stress Resistance. World For. Res. 2005, 18, 26–29. [Google Scholar]
- Shi, Z.Y.; Feng, G.; Christie, P.; Li, X.L. Arbuscular mycorrhizal status of spring ephemerals in the desert ecosystem of Junggar Basin, China. Mycorrhiza 2006, 16, 269–275. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kang, J.; Yang, W.L.; Liu, S.W.; Xuan, N.; Shao, Y.H.; Geng, Y.; Afzal, M.; Zhang, Y.X.; Yue, S.S.; Mushtaq, R.; et al. Arbuscular mycorrhizal fungi increased peanut (Arachis hypogaea L.) yield by changing the rhizosphere microbial community structure in saline-alkali soil. Front. Microbiol. 2023, 14, 1303979. [Google Scholar] [CrossRef]
- Fan, L.; Zhang, P.; Cao, F.Z.; Liu, X.P.; Ji, M.J.; Xie, M. Effects of AMF on Maize Yield and Soil Microbial Community in Sandy and Saline Soils. Plants 2024, 13, 2056. [Google Scholar] [CrossRef]
- Leigh, J.; Fitter, A.H.; Hodge, A. Growth and symbiotic effectiveness of an arbuscular mycorrhizal fungus in organic matter in competition with soil bacteria. Fems Microbiol. Ecol. 2011, 76, 428–438. [Google Scholar] [CrossRef]
- Herman, D.J.; Firestone, M.K.; Nuccio, E.; Hodge, A. Interactions between an arbuscular mycorrhizal fungus and a soil microbial community mediating litter decomposition. Fems Microbiol. Ecol. 2012, 80, 236–247. [Google Scholar] [CrossRef] [PubMed]
- Duan, S.; Huo, Y.; Tian, Y.; Yan, W.; George Timothy, S.; Huang, C.; Feng, G.; Zhang, L. The interplay of direct and mycorrhizal pathways for plants to efficiently acquire phosphorus from soil. Front. Agric. Sci. Eng. 2025, 12, 47–56. [Google Scholar]
- Duan, S.L.; Feng, G.; Limpens, E.; Bonfante, P.; Xie, X.N.; Zhang, L. Cross-kingdom nutrient exchange in the plant-arbuscular mycorrhizal fungus-bacterium continuum. Nat. Rev. Microbiol. 2024, 22, 773–790. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Hou, Y. Application of phospholipid fatty acid method in soil microbial analysis. Microbiology 2004, 31, 114–117. [Google Scholar]
- Zelles, L.; Bai, Q.Y.; Beck, T.; Beese, F. Signature fatty acids in phospholipids and lipopolysaccharides as indicators of microbial biomass and community structure in agricultural soils. Soil Biol. Biochem. 1992, 24, 317–323. [Google Scholar] [CrossRef]
- Frostegård, A.; Bååth, E. The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol. Fertil. Soils 1996, 22, 59–65. [Google Scholar] [CrossRef]
- Maestre, F.T.; Biancari, L.; Chen, N.; Corrochano-Monsalve, M.; Jenerette, G.D.; Nelson, C.; Shilula, K.N.; Shpilkina, Y. Research needs on the biodiversity-ecosystem functioning relationship in drylands. npj Biodivers. 2024, 3, 12. [Google Scholar] [CrossRef]
- Zhai, C.; Han, L.; Xiong, C.; Ge, A.; Yue, X.; Li, Y.; Zhou, Z.; Feng, J.; Ru, J.; Song, J.; et al. Soil microbial diversity and network complexity drive the ecosystem multifunctionality of temperate grasslands under changing precipitation. Sci. Total Environ. 2024, 906, 167217. [Google Scholar] [CrossRef]
- Ren, C.J.; Zhao, F.Z.; Shi, Z.; Chen, J.; Han, X.H.; Yang, G.H.; Feng, Y.Z.; Ren, G.X. Differential responses of soil microbial biomass and carbon-degrading enzyme activities to altered precipitation. Soil Biol. Biochem. 2017, 115, 1–10. [Google Scholar] [CrossRef]
- Zheng, J.Q.; Cui, M.M.; Wang, C.; Wang, J.; Wang, S.L.; Sun, Z.J.; Ren, F.R.; Wan, S.Q.; Han, S.J. Elevated CO2 warming, N addition, and increased precipitation affect different aspects of the arbuscular mycorrhizal fungal community. Sci. Total Environ. 2022, 806, 150522. [Google Scholar] [CrossRef] [PubMed]
- Tu, N.; Hong, M.; Ye, H.; Zhang, Y.; He, S.; Zhang, Y.; Zhao, Y.; Hu, J. Effects of Precipitation Change and Nitrogen Deposition on Soil Arbuscular Mycorrhizal Fungi (AMF) Community Structure in Desert Steppe. Soils 2023, 55, 1251–1260. [Google Scholar]
- Guo, H.; He, X. Effects of AM fungi on the growth and drought resistance of Astragalus adsurgens Pall. under water stress. Acta Ecol. Sin. 2010, 30, 5933–5940. [Google Scholar]
- Fu, W.; Chen, B.D.; Rillig, M.C.; Jansa, J.; Ma, W.; Xu, C.; Luo, W.T.; Wu, H.H.; Hao, Z.P.; Wu, H.; et al. Community response of arbuscular mycorrhizal fungi to extreme drought in a cold-temperate grassland. New Phytol. 2022, 234, 2003–2017. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.Q.; Fan, C.K.; Zhao, S.M.; Xiao, R.; Miao, R.H.; Yang, Z.L.; Wan, S.Q. Effect of Litter Changes on Soil Microbial Community and Respiration in a Coniferous-Broadleaf Mixed Forest. Ecosystems 2025, 28, 26. [Google Scholar] [CrossRef]
- Sun, X.N.; Gong, L.; Ma, X.Y.; Yin, K.J.; Li, X.C. The driving role of microorganisms in the soil carbon cycle of mountain forests in arid areas under different carbon inputs. Plant Soil 2025, 507, 1–17. [Google Scholar] [CrossRef]
- Chen, Q.; Niu, B.; Hu, Y.; Luo, T.; Zhang, G. Warming and increased precipitation indirectly affect the composition and turnover of labile-fraction soil organic matter by directly affecting vegetation and microorganisms. Sci. Total Environ. 2020, 714, 136787. [Google Scholar] [CrossRef]
- Yang, X.C.; Zhu, K.; Loik, M.E.; Sun, W. Differential responses of soil bacteria and fungi to altered precipitation in a meadow steppe. Geoderma 2021, 384, 114812. [Google Scholar] [CrossRef]
- Boukhatem, Z.F.; Merabet, C.; Tsaki, H. Plant Growth Promoting Actinobacteria, the Most Promising Candidates as Bioinoculants? Front. Agron. 2022, 4, 849911. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, J.; Wang, Y.; Zhang, Y.; Zhao, Y.; Luo, X. Effects of short-term precipitation changes on soil microbial communities in desert grasslands. Acta Ecol. Sin. 2025, 45, 64–79. [Google Scholar]
- Wu, K.; Xu, W.X.; Yang, W.K. Effects of precipitation changes on soil bacterial community composition and diversity in the Junggar desert of Xinjiang, China. Peerj 2020, 8, e8433. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.J.; Yan, Y.; Li, X.Q.; Liu, Y.L.; Lu, X.Y. Rhizosphere soil microbial community and its response to different utilization patterns in the semi-arid alpine grassland of northern Tibet. Front. Microbiol. 2022, 13, 931795. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Yue, P.; Cui, X.; Li, K.; Liu, X. Response of Soil Microbial Biomass Carbon and Nitrogen Deposition to Precipitation and Temperature in the Gurbantunggut Desert. Arid Zone Res. 2018, 35, 515–523. [Google Scholar]
- Huang, G.; Li, Y.; Su, Y.G. Effects of increasing precipitation on soil microbial community composition and soil respiration in a temperate desert, Northwestern China. Soil Biol. Biochem. 2015, 83, 52–56. [Google Scholar] [CrossRef]
- Kang, F.; Yang, B.; Wujisiguleng, Y.X.; Wang, L.; Guo, J.; Sun, W.; Zhang, Q.; Zhang, T. Arbuscular mycorrhizal fungi alleviate the negative effect of nitrogen deposition on ecosystem functions in meadow grassland. Land Degrad. Dev. 2020, 31, 748–759. [Google Scholar] [CrossRef]
- Tang, B.; Man, J.; Xiang, G.; Wang, Y.; Bai, Y. Heavy grazing disrupts positive effects of arbuscular mycorrhizae symbiosis on community productivity and stability under low and high phosphorus conditions. Plant Soil 2020, 457, 375–387. [Google Scholar] [CrossRef]
- O’Connor, P.J.; Smith, S.E.; Smith, F.A. Arbuscular mycorrhizas influence plant diversity and community structure in a semiarid herbland. New Phytol. 2002, 154, 209–218. [Google Scholar] [CrossRef]
- Pech, P.; Heneberg, P. Benomyl treatment decreases fecundity of ant queens. J. Invertebr. Pathol. 2015, 130, 61–63. [Google Scholar] [CrossRef]
- Smith, M.D.; Hartnett, D.C.; Rice, C.W. Effects of long-term fungicide applications on microbial properties in tallgrass prairie soil. Soil Biol. Biochem. 2000, 32, 935–946. [Google Scholar] [CrossRef]
- Yang, G.; Zhang, Y.; Yang, X.; Liu, N.; Rillig, M.C. Mycorrhizal suppression and phosphorus addition influence the stability of plant community composition and function in a temperate steppe. Oikos 2021, 130, 354–365. [Google Scholar] [CrossRef]
- Xue, Y.; Pierre, M.; Jixun, G.; Yann, H.; Tao, Z. Suppression of arbuscular mycorrhizal fungi decreases the temporal stability of community productivity under elevated temperature and nitrogen addition in a temperate meadow. Sci. Total Environ. 2020, 762, 143137. [Google Scholar] [CrossRef]
- Li, X.; Zuo, Y.; Xue, Z.; Zhang, L.; Zhao, L.; He, X. Characteristics of microbial community structure in rhizosphere soil of different desert plants. Acta Ecol. Sin. 2018, 38, 2855–2863. [Google Scholar] [CrossRef]
- Bortolot, M.; Buffoni, B.; Mazzarino, S.; Hoff, G.; Martino, E.; Fiorilli, V.; Fossalunga, A.S.D. The Importance of Mycorrhizal Fungi and Their Associated Bacteria in Promoting Crops’ Performance: An Applicative Perspective. Horticulturae 2024, 10, 1326. [Google Scholar] [CrossRef]
- Wang, L.T.; Zhang, L.; George, T.S.; Feng, G. A core microbiome in the hyphosphere of arbuscular mycorrhizal fungi has functional significance in organic phosphorus mineralization. New Phytol. 2023, 238, 859–873. [Google Scholar] [CrossRef]
- Duan, H.; Luo, C.; Shi, Q.; Kang, S.; Zhao, L.; Xiong, Y. Research progress in the effects of arbuscular mycorrhizal fungi on plant-soil systems. Acta Ecol. Sin. 2025, 45, 475–491. [Google Scholar]
- Welc, M.; Ravnskov, S.; Kieliszewska-Rokicka, B.; Larsen, J. Suppression of other soil microorganisms by mycelium of arbuscular mycorrhizal fungi in root-free soil. Soil Biol. Biochem. 2010, 42, 1534–1540. [Google Scholar] [CrossRef]
- Leake, J.; Johnson, D.; Donnelly, D.; Muckle, G.; Boddy, L.; Read, D. Networks of power and influence: The role of mycorrhizal mycelium in controlling plant communities and agroecosystem functioning. Can. J. Bot. 2004, 82, 1016–1045. [Google Scholar] [CrossRef]
- Ruan, C.J.; Ramoneda, J.; Gogia, G.; Wang, G.; Johnson, D.R. Fungal hyphae regulate bacterial diversity and plasmid-mediated functional novelty during range expansion. Curr. Biol. 2022, 32, 5285–5294.e4. [Google Scholar] [CrossRef]
- Yuan, H.; Ye, Y.; Si, H.; Ji, Q.; Zhang, Y. Effects of Arbuscular Mycorrhizal Fungi on Plant Stress Resistance and Secondary Metabolite Synthesis. Biotechnol. Bull. 2024, 40, 45–56. [Google Scholar]
- Groten, K.; Yon, F.; Baldwin, I.T. Arbuscular mycorrhizal fungi influence the intraspecific competitive ability of plants under field and glasshouse conditions. Planta 2023, 258, 60. [Google Scholar] [CrossRef]
- Xu, X.Y.; Qiu, Y.P.; Zhang, K.C.; Yang, F.; Chen, M.F.; Luo, X.; Yan, X.B.; Wang, P.; Zhang, Y.; Chen, H.H.; et al. Climate warming promotes deterministic assembly of arbuscular mycorrhizal fungal communities. Glob. Change Biol. 2022, 28, 1147–1161. [Google Scholar] [CrossRef]
- Kaiser, C.; Kilburn, M.R.; Clode, P.L.; Fuchslueger, L.; Koranda, M.; Cliff, J.B.; Solaiman, Z.M.; Murphy, D.V. Exploring the transfer of recent plant photosynthates to soil microbes: Mycorrhizal pathway vs direct root exudation. New Phytol. 2015, 205, 1537–1551. [Google Scholar] [CrossRef]
- Javed, Z.; Tripathi, G.D.; Mishra, M.; Dashora, K. Actinomycetes—The microbial machinery for the organic-cycling, plant growth, and sustainable soil health. Biocatal. Agric. Biotechnol. 2021, 31, 101893. [Google Scholar] [CrossRef]
- Ouyang, J.; Wei, L.; Yu, C. Preliminary study on factors driving arbuscular mycorrhizal fungi diversity with elevation in the South-Tibet River Basin. Plant Sci. J. 2024, 42, 34–42. [Google Scholar] [CrossRef]
- Chen, Y.F.; Zhang, L.W.; Shi, X.; Liu, H.L.; Zhang, D.Y. Life history responses of two ephemeral plant species to increased precipitation and nitrogen in the Gurbantunggut Desert. Peerj 2019, 7, e6158. [Google Scholar] [CrossRef]
- Duan, C.; Wu, L.; Wang, S.; He, L. Analysis of spatio-temporal patterns of ephemeral plants in the Gurbantunggut Desert over the last 30 years. Acta Ecol. Sin. 2017, 37, 2642–2652. [Google Scholar] [CrossRef][Green Version]
- Gao, J.; Wang, Y.; Sayit, H.; Mamtimin, A.; Liu, Y.; Zhao, X.; Yang, X.; Huo, W.; Yang, F.; Zhou, C. Characteristics of surface radiation budget in Gurbantunggut Desert. J. Desert Res. 2021, 41, 47–58. [Google Scholar]
- Bronick, C.J.; Lal, R. Soil structure and management: A review. Geoderma 2005, 124, 3–22. [Google Scholar] [CrossRef]
- Tian, C.Y.; Shi, Z.; Chen, Z.; Feng, G. Study on arbuscular mycorrhizal symbionts in the Gurbantunggut Desert. Chin. Sci. Bull. 2006, 51, 140–146. [Google Scholar] [CrossRef]
- Zeng, Y.; Liu, T.; Zhou, X.B.; Sun, Q.M.; Han, Z.Q.; Liu, K. Effects of climate change on plant composition and diversity in the Gurbantünggüt Desert of northwestern China. Ecol. Res. 2016, 31, 427–439. [Google Scholar] [CrossRef]
- Zhao, Z.; Ding, Y.; Xu, Y.; Zhang, J. Detection and Prediction of Climate Change for the 20th and 21st Century Due to Human Activity in Northwest China. Clim. Environ. Res. 2003, 8, 26–34. [Google Scholar]
- Takayabu, I.; Kato, H.; Nishizawa, K.; Takayabu, Y.N.; Sato, Y.; Sasaki, H.; Kurihara, K.; Kitoh, A. Future projections in precipitation over Asia simulated by two RCMs nested into MRI-CGCM2.2. J. Meteorol. Soc. Jpn. 2007, 85, 511–519. [Google Scholar] [CrossRef][Green Version]
- Hartnett, D.C.; Wilson, G.W.T. Mycorrhizae influence plant community structure and diversity in tallgrass prairie. Ecology 1999, 80, 1187–1195. [Google Scholar] [CrossRef]
- Yang, G.W.; Liu, N.; Lu, W.J.; Wang, S.; Kan, H.M.; Zhang, Y.J.; Xu, L.; Chen, Y.L. The interaction between arbuscular mycorrhizal fungi and soil phosphorus availability influences plant community productivity and ecosystem stability. J. Ecol. 2014, 102, 1072–1082. [Google Scholar] [CrossRef]
- Woiwod, I.P. Ecological Diversity and Its Measurement. Biometrics 1990, 46, 547. [Google Scholar] [CrossRef]
- Qiu-Fang, Z.; Bo, L.I.U.; Ying-Zhi, L.I.N.; Huai, S.H.I.; Shu-Sheng, Y.; Xian-Zhi, Z. The diversity of phospholipid fatty acid (PLFA) biomarker for the microbial community in soil. Acta Ecol. Sin. 2009, 29, 4127–4137. [Google Scholar]
- Sun, H.; Liu, X. Microbes studies of tea rhizosphere. Acta Ecol. Sin. 2004, 24, 1353–1357. [Google Scholar]
- Jiang, X.Y.; Cao, L.X.; Zhang, R.D. Changes of labile and recalcitrant carbon pools under nitrogen addition in a city lawn soil. J. Soils Sediments 2014, 14, 515–524. [Google Scholar] [CrossRef]
- Zhang, M.; Bai, X.X.; Wang, Y.; Li, Y.; Cui, Y.X.; Hu, S.L.; Jonathan, M.A.; Dong, L.G.; Yu, X. Soil microbial trait-based strategies drive the storage and stability of the soil carbon pool in Robinia pseudoacacia plantations. Catena 2023, 222, 106894. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Y.; Liu, Z.; Han, F.; Chen, S.G.; Zhou, W.Z. Integrated application of phosphorus-accumulating bacteria and phosphorus-solubilizing bacteria to achieve sustainable phosphorus management in saline soils. Sci. Total Environ. 2023, 885, 163971. [Google Scholar] [CrossRef] [PubMed]
- Brookes, P.C.; Powlson, D.S.; Jenkinson, D.S. Measurement of microbial biomass phosphorus in soil. Soil Biol. Biochem. 1982, 14, 319–329. [Google Scholar] [CrossRef]
- Redmile-Gordon, M.A.; Brookes, P.C.; Evershed, R.P.; Goulding, K.W.T.; Hirsch, P.R. Measuring the soil-microbial interface: Extraction of extracellular polymeric substances (EPS) from soil biofilms. Soil Biol. Biochem. 2014, 72, 163–171. [Google Scholar] [CrossRef]
- Jakobsen, I.; Abbott, L.; Robson, A.D. External hyphae of vesicular-arbuscular mycorrhizal fungi associated with Trifolium subterraneum L. 1. Spread of hyphae and phosphorus inflow into roots. New Phytol. 1992, 120, 371–380. [Google Scholar] [CrossRef]
- White, D.C.; Flemming, C.A.; Leung, K.T.; Macnaughton, S.J. In situ microbial ecology for quantitative appraisal, monitoring, and risk assessment of pollution remediation in soils, the subsurface, the rhizosphere and in biofilms. J. Microbiol. Methods 1998, 32, 93–105. [Google Scholar] [CrossRef]
- Yang, Y.L.; Xie, H.T.; Mao, Z.; Bao, X.L.; He, H.B.; Zhang, X.D.; Liang, C. Fungi determine increased soil organic carbon more than bacteria through their necromass inputs in conservation tillage croplands. Soil Biol. Biochem. 2022, 167, 108587. [Google Scholar] [CrossRef]
- Bossio, D.A. Impacts of Carbon and Flooding on Soil Microbial Communities: Phospholipid Fatty Acid Profiles and Substrate Utilization Patterns. Microb. Ecol. 1998, 35, 265–278. [Google Scholar] [CrossRef]
- Zhang, C.H.; Xi, N.X. Precipitation Changes Regulate Plant and Soil Microbial Biomass Via Plasticity in Plant Biomass Allocation in Grasslands: A Meta-Analysis. Front. Plant Sci. 2021, 12, 614968. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duan, W.; Wang, H.; Ji, Z.; Dong, Q.; Li, W.; Cao, W.; Zhang, F.; Jia, Y. Underlying Mechanisms of Increased Precipitation and Arbuscular Mycorrhizal (AM) Fungi on Plant Community by Mediating Soil Microbes in Desert Ecosystems. Plants 2025, 14, 3386. https://doi.org/10.3390/plants14213386
Duan W, Wang H, Ji Z, Dong Q, Li W, Cao W, Zhang F, Jia Y. Underlying Mechanisms of Increased Precipitation and Arbuscular Mycorrhizal (AM) Fungi on Plant Community by Mediating Soil Microbes in Desert Ecosystems. Plants. 2025; 14(21):3386. https://doi.org/10.3390/plants14213386
Chicago/Turabian StyleDuan, Wan, Hui Wang, Zhanquan Ji, Qianqian Dong, Wenshuo Li, Wenli Cao, Fangwei Zhang, and Yangyang Jia. 2025. "Underlying Mechanisms of Increased Precipitation and Arbuscular Mycorrhizal (AM) Fungi on Plant Community by Mediating Soil Microbes in Desert Ecosystems" Plants 14, no. 21: 3386. https://doi.org/10.3390/plants14213386
APA StyleDuan, W., Wang, H., Ji, Z., Dong, Q., Li, W., Cao, W., Zhang, F., & Jia, Y. (2025). Underlying Mechanisms of Increased Precipitation and Arbuscular Mycorrhizal (AM) Fungi on Plant Community by Mediating Soil Microbes in Desert Ecosystems. Plants, 14(21), 3386. https://doi.org/10.3390/plants14213386




