Zeaxanthin and Other Carotenoids: Roles in Abiotic Stress Defense with Implications for Biotic Defense
Abstract
1. Introduction
2. Biosynthetic Pathways of Xanthophylls and Carotenoid-Derived Plant Stress Hormones
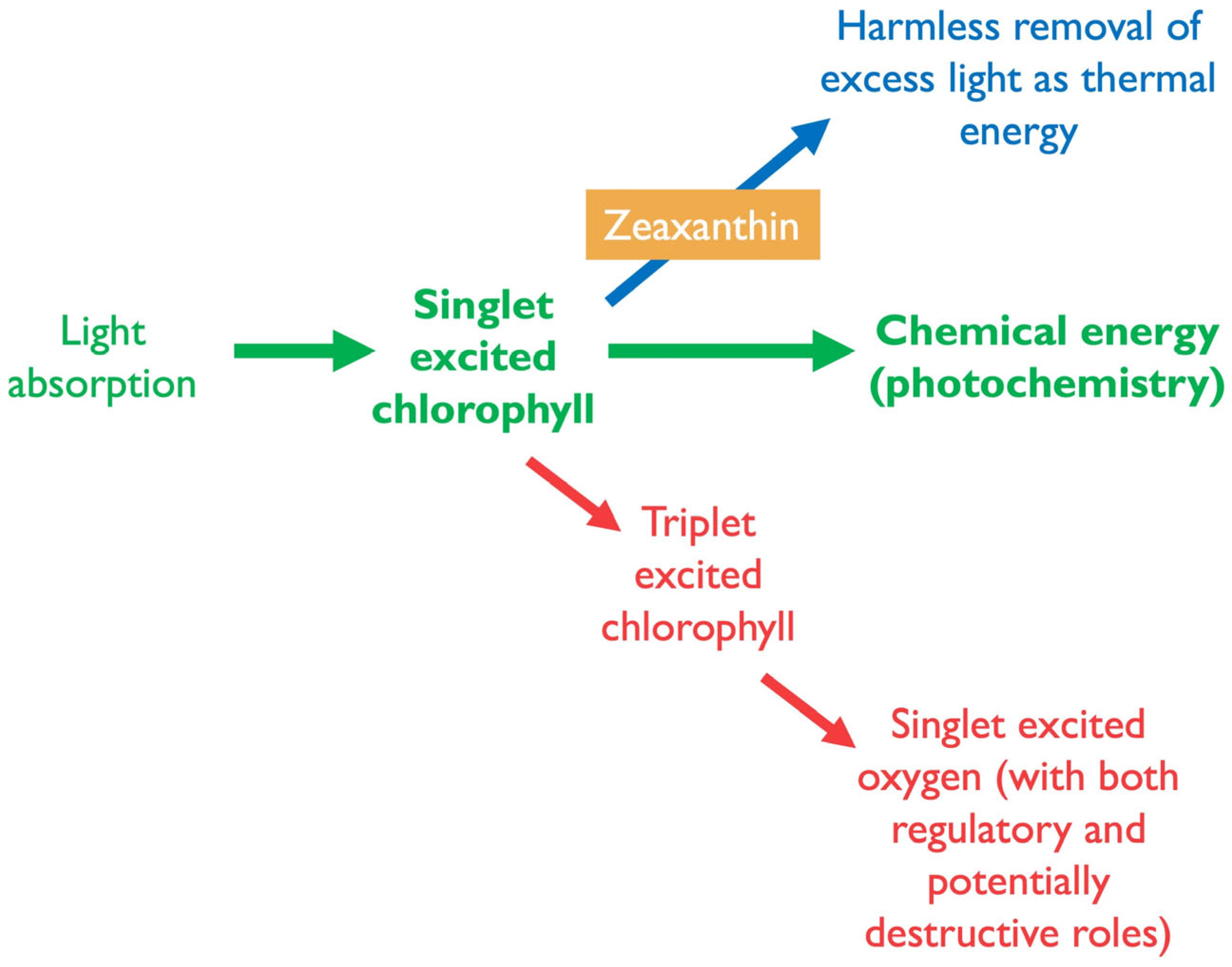
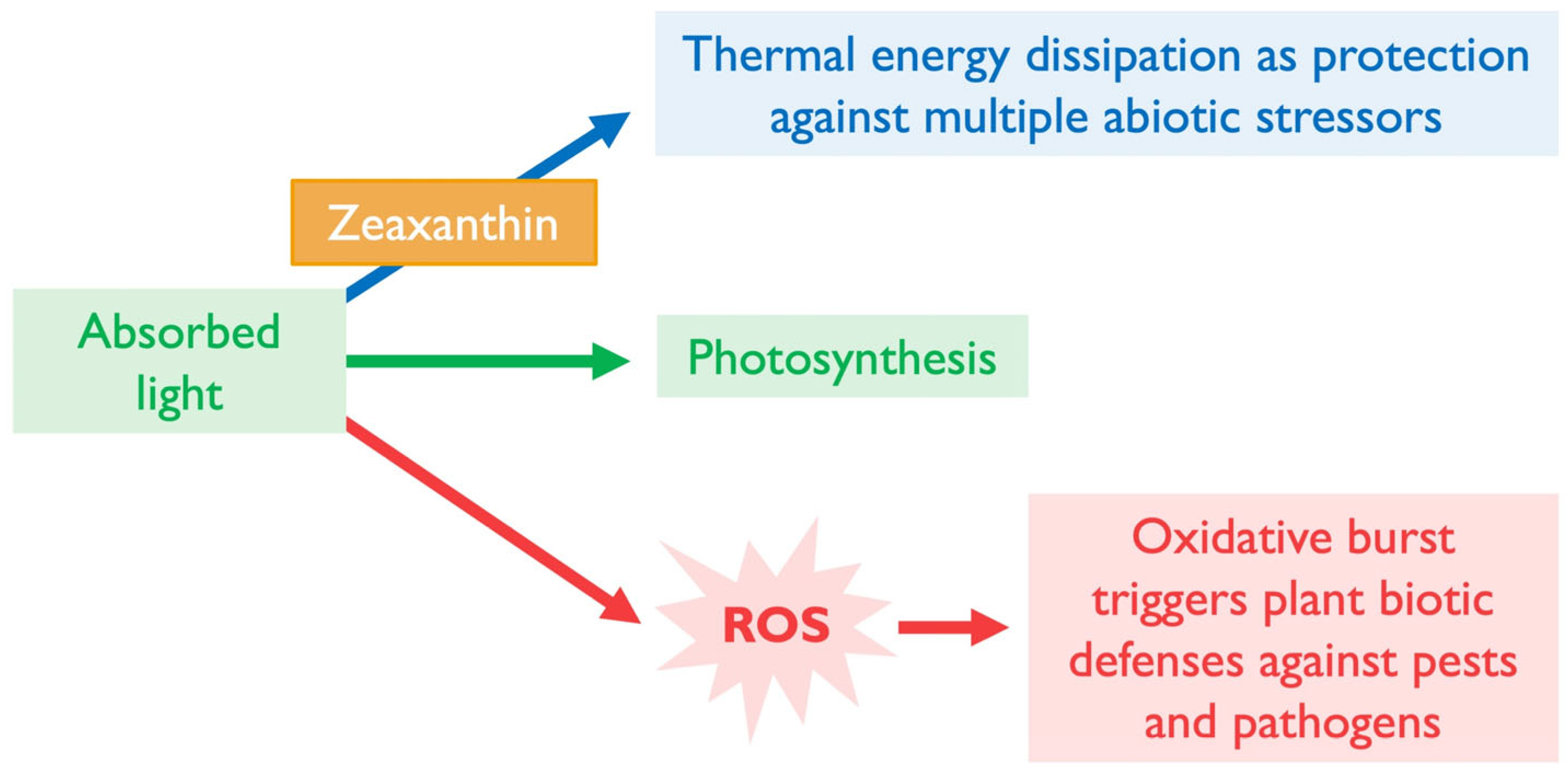
3. Photoprotection of Photosynthesis by Xanthophylls
4. Carotenoids as Essential Human Micronutrients with Roles in Stress Protection and Gene Regulation
5. Roles of Zeaxanthin in Protecting Membrane Integrity and Function
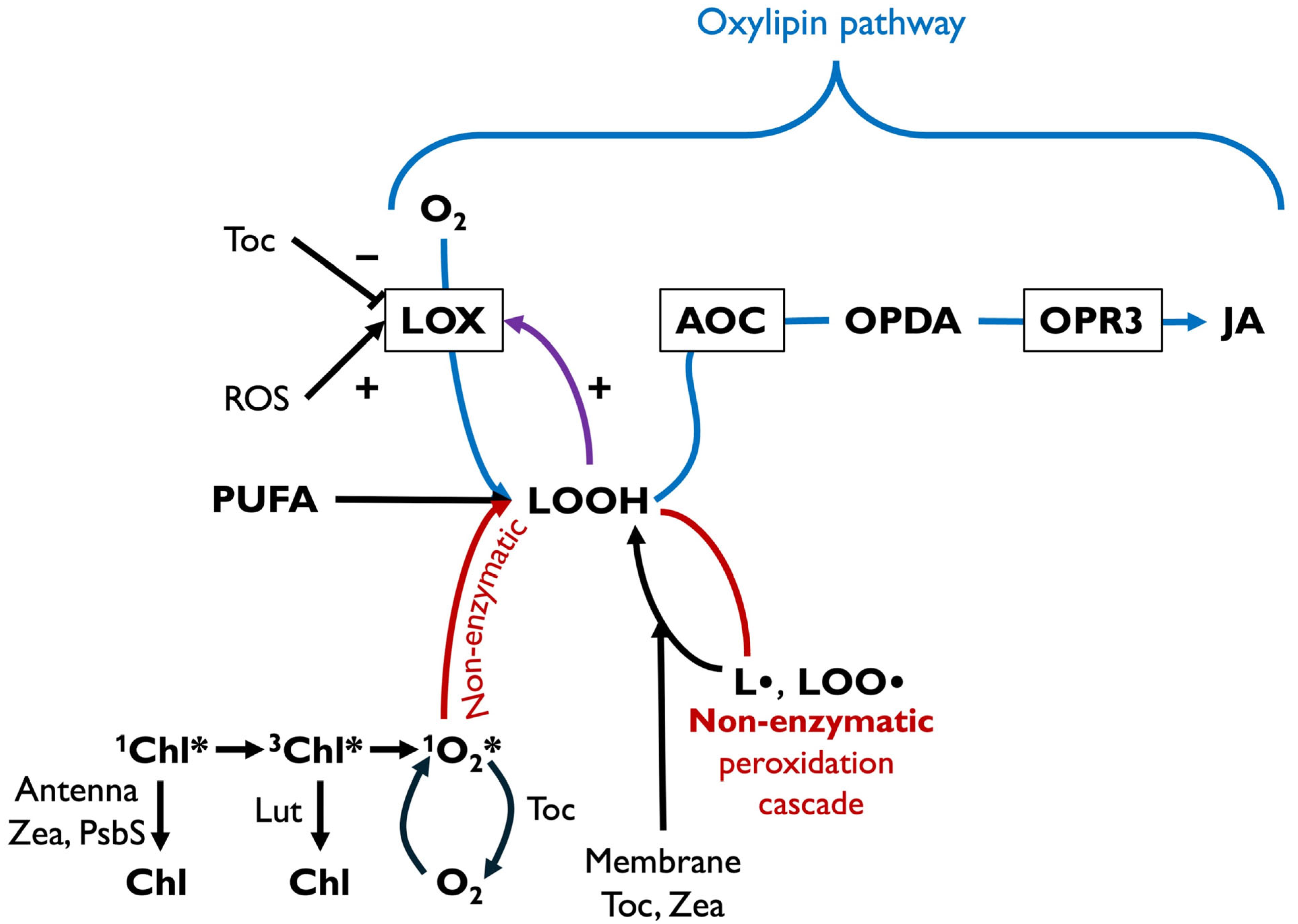
6. Specific Functions of Xanthophylls in Modulating Plant Stress Hormone Synthesis
6.1. Zeaxanthin, Thermal Dissipation, and Jasmonic Acid
6.2. Violaxanthin and ABA
6.3. Xanthophylls and Salicylic Acid
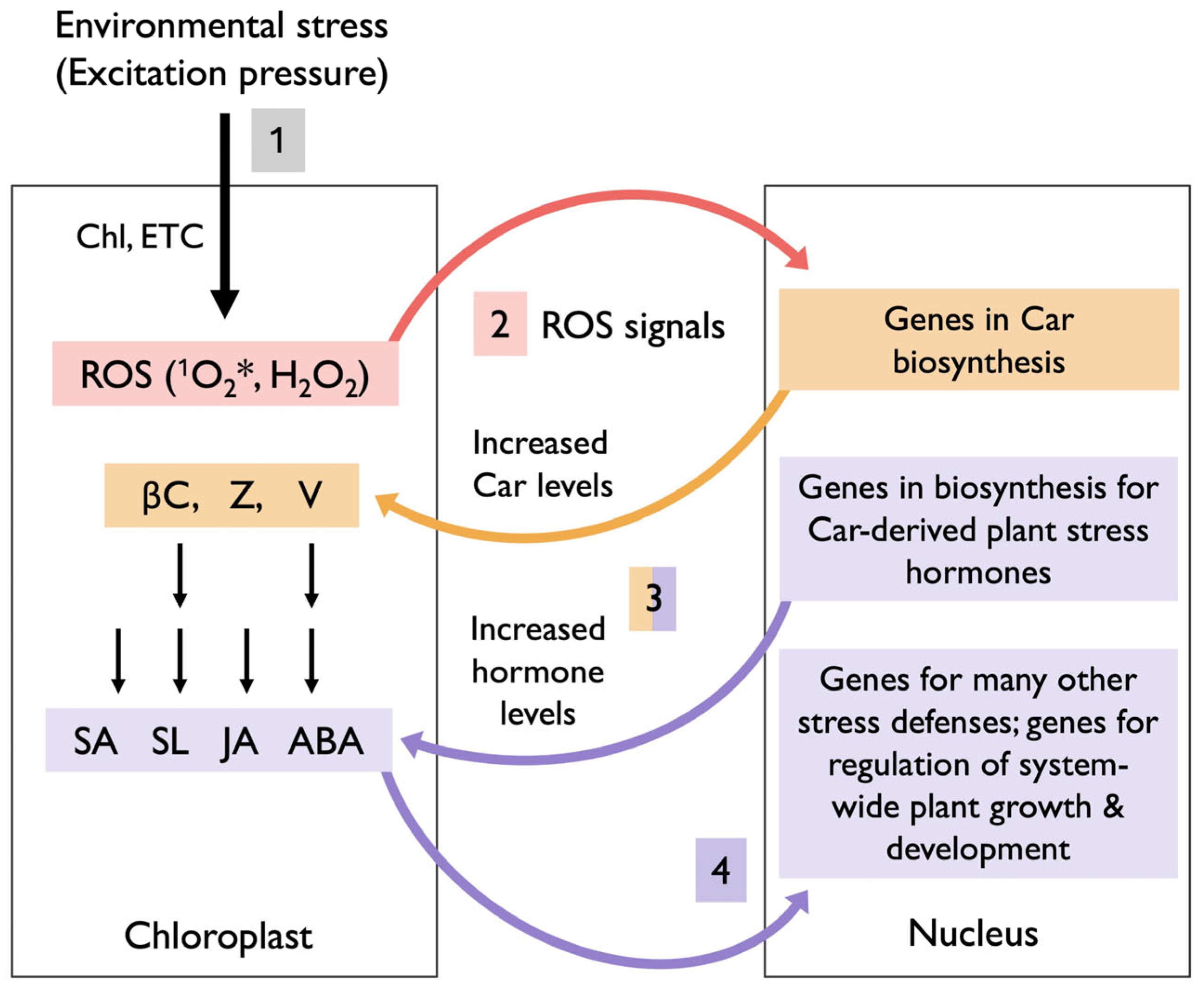
7. Summary of Multi-Loop Signaling Cascades Between Chloroplast and Nucleus
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Demmig-Adams, B.; Polutchko, S.K.; Adams, W.W., III. Structure-Function-Environment Relationship of the Isomers Zeaxanthin and Lutein. Photochem 2022, 2, 308–325. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Stewart, J.J.; López-Pozo, M.; Polutchko, S.K.; Adams, W.W., III. Zeaxanthin, a Molecule for Photoprotection in Many Different Environments. Molecules 2020, 25, 5825. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Kunert, K. The Ascorbate–Glutathione Cycle Coming of Age. J. Exp. Bot. 2024, 75, 2682–2699. [Google Scholar] [CrossRef]
- Niyogi, K.K. Photoprotection Revisited: Genetic and Molecular Approaches. Annu. Rev. Plant Biol. 1999, 50, 333–359. [Google Scholar] [CrossRef]
- Uarrota, V.G.; Stefen, D.L.V.; Leolato, L.S.; Gindri, D.M.; Nerling, D. Revisiting Carotenoids and Their Role in Plant Stress Responses: From Biosynthesis to Plant Signaling Mechanisms during Stress. In Antioxidants and Antioxidant Enzymes in Higher Plants; Gupta, D., Palma, J., Corpas, F., Eds.; Springer: Cham, Switzerland, 2018; pp. 207–232. [Google Scholar] [CrossRef]
- Zhao, B.; Liu, Q.; Wang, B.; Yuan, F. Roles of Phytohormones and Their Signaling Pathways in Leaf Development and Stress Responses. J. Agric. Food Chem. 2021, 69, 3566–3584. [Google Scholar] [CrossRef]
- Brewer, P.B.; Koltai, H.; Beveridge, C.A. Diverse Roles of Strigolactones in Plant Development. Mol. Plant 2013, 6, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Bhoi, A.; Yadu, B.; Chandra, J.; Keshavkant, S. Contribution of Strigolactone in Plant Physiology, Hormonal Interaction and Abiotic Stresses. Planta 2021, 254, 28. [Google Scholar] [CrossRef] [PubMed]
- Özbilen, A.; Sezer, F.; Taşkin, K.M. Identification and Expression of Strigolactone Biosynthesis and Signaling Genes and the in Vitro Effects of Strigolactones in Olive (Olea europaea L.). Plant Direct 2024, 8, e568. [Google Scholar] [CrossRef]
- Dall’Osto, L.; Lico, C.; Alric, J.; Giuliano, G.; Havaux, M.; Bassi, R. Lutein Is Needed for Efficient Chlorophyll Triplet Quenching in the Major LHCII Antenna Complex of Higher Plants and Effective Photoprotection in Vivo under Strong Light. BMC Plant Biol. 2006, 6, 32. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Cohu, C.M.; Amiard, V.; Van Zadelhoff, G.; Veldink, G.A.; Muller, O.; Adams, W.W., III. Emerging Trade—offs—Impact of Photoprotectants (PsbS, Xanthophylls, and Vitamin E) on Oxylipins as Regulators of Development and Defense. New Phytol. 2013, 197, 720–729. [Google Scholar] [CrossRef]
- North, H.M.; Almeida, A.D.; Boutin, J.; Frey, A.; To, A.; Botran, L.; Sotta, B.; Marion-Poll, A. The Arabidopsis ABA-deficient Mutant aba4 Demonstrates That the Major Route for Stress-induced ABA Accumulation Is via Neoxanthin Isomers. Plant J. 2007, 50, 810–824. [Google Scholar] [CrossRef]
- Huang, Y.; Guo, Y.; Liu, Y.; Zhang, F.; Wang, Z.; Wang, H.; Wang, F.; Li, D.; Mao, D.; Luan, S. 9-Cis-Epoxycarotenoid Dioxygenase 3 Regulates Plant Growth and Enhances Multi-Abiotic Stress Tolerance in Rice. Front. Plant Sci. 2018, 9, 162. [Google Scholar] [CrossRef]
- Slovik, S.; Daeter, W.; Hartung, W. Compartmental Redistribution and Long-Distance Transport of Abscisic Acid (ABA) in Plants as Influenced by Environmental Changes in the Rhizosphere—A Biomathematical Model. J. Exp. Bot. 1995, 46, 881–894. [Google Scholar] [CrossRef]
- Lee, T.-Y.; Lam, L.; Patel-Tupper, D.; Roy, P.P.; Ma, S.A.; Lam, H.E.; Lucas-DeMott, A.; Karavolias, N.G.; Iwai, M.; Niyogi, K.K. Chlorophyll to Zeaxanthin Energy Transfer in Nonphotochemical Quenching: An Exciton Annihilation-Free Transient Absorption Study. Proc. Natl. Acad. Sci. USA 2024, 121, e2411620121. [Google Scholar] [CrossRef] [PubMed]
- Krieger-Liszkay, A. Singlet Oxygen Production in Photosynthesis. J. Exp. Bot. 2005, 56, 337–346. [Google Scholar] [CrossRef]
- Kozuleva, M.A.; Ivanov, B.N. Superoxide Anion Radical Generation in Photosynthetic Electron Transport Chain. Biochem. Mosc. 2023, 88, 1045–1060. [Google Scholar] [CrossRef] [PubMed]
- Pospíšil, P. Production of Reactive Oxygen Species by Photosystem II. Biochim. Biophys. Acta BBA-Bioenerg. 2009, 1787, 1151–1160. [Google Scholar] [CrossRef]
- Murchie, E.H.; Ruban, A.V. Dynamic Non-photochemical Quenching in Plants: From Molecular Mechanism to Productivity. Plant J. 2020, 101, 885–896. [Google Scholar] [CrossRef]
- Baroli, I.; Niyogi, K.K. Molecular Genetics of Xanthophyll–Dependent Photoprotection in Green Algae and Plants. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2000, 355, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Adams, W.W., III. Carotenoid Composition in Sun and Shade Leaves of Plants with Different Life Forms. Plant Cell Environ. 1992, 15, 411–419. [Google Scholar] [CrossRef]
- Logan, B.A.; Demmig-Adams, B.; Adams, W.W., III; Grace, S.C. Antioxidants and Xanthophyll Cycle-Dependent Energy Dissipation in Cucurbita pepo L. and Vinca major L. Acclimated to Four Growth PPFDs in the Field. J. Exp. Bot. 1998, 49, 1869–1879. [Google Scholar] [CrossRef]
- Li, X.-P.; Gilmore, A.M.; Caffarri, S.; Bassi, R.; Golan, T.; Kramer, D.; Niyogi, K.K. Regulation of Photosynthetic Light Harvesting Involves Intrathylakoid Lumen pH Sensing by the PsbS Protein. J. Biol. Chem. 2004, 279, 22866–22874. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Adams, W.W., III. Photoprotection in an Ecological Context: The Remarkable Complexity of Thermal Energy Dissipation. New Phytol. 2006, 172, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Hüner, N.P.; Bode, R.; Dahal, K.; Hollis, L.; Rosso, D.; Krol, M.; Ivanov, A.G. Chloroplast Redox Imbalance Governs Phenotypic Plasticity: The “Grand Design of Photosynthesis” Revisited. Front. Plant Sci. 2012, 3, 255. [Google Scholar] [CrossRef]
- Havaux, M. Carotenoids as Membrane Stabilizers in Chloroplasts. Trends Plant Sci. 1998, 3, 147–151. [Google Scholar] [CrossRef]
- Johansson Jänkänpää, H.; Frenkel, M.; Zulfugarov, I.; Reichelt, M.; Krieger-Liszkay, A.; Mishra, Y.; Gershenzon, J.; Moen, J.; Lee, C.-H.; Jansson, S. Non-Photochemical Quenching Capacity in Arabidopsis thaliana Affects Herbivore Behaviour. PLoS ONE 2013, 8, e53232. [Google Scholar] [CrossRef]
- Göhre, V.; Jones, A.M.E.; Sklenář, J.; Robatzek, S.; Weber, A.P.M. Molecular Crosstalk Between PAMP-Triggered Immunity and Photosynthesis. Mol. Plant-Microbe Interact. 2012, 25, 1083–1092. [Google Scholar] [CrossRef]
- Semba, R.D. Vitamin A, Immunity, and Infection. Clin. Infect. Dis. 1994, 19, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Banik, S.P.; Ohia, S.E.; Moriyama, H.; Chakraborty, S.; Wang, C.-K.; Song, Y.S.; Goel, A.; Bagchi, M.; Bagchi, D. Current Insights on the Photoprotective Mechanism of the Macular Carotenoids, Lutein and Zeaxanthin: Safety, Efficacy and Bio-Delivery. J. Am. Nutr. Assoc. 2024, 43, 505–518. [Google Scholar] [CrossRef]
- Mrowicka, M.; Mrowicki, J.; Kucharska, E.; Majsterek, I. Lutein and Zeaxanthin and Their Roles in Age-Related Macular Degeneration—Neurodegenerative Disease. Nutrients 2022, 14, 827. [Google Scholar] [CrossRef]
- Perry, A.; Rasmussen, H.; Johnson, E.J. Xanthophyll (Lutein, Zeaxanthin) Content in Fruits, Vegetables and Corn and Egg Products. J. Food Compos. Anal. 2009, 22, 9–15. [Google Scholar] [CrossRef]
- Kanterman, J.; Sade-Feldman, M.; Baniyash, M. New Insights into Chronic Inflammation-Induced Immunosuppression. Semin. Cancer Biol. 2012, 22, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, A.; Harrison, E.H. Carotenoid Metabolism in Mammals, Including Man: Formation, Occurrence, and Function of Apocarotenoids. J. Lipid Res. 2013, 54, 1719–1730. [Google Scholar] [CrossRef]
- McGrane, M.M. Vitamin A Regulation of Gene Expression: Molecular Mechanism of a Prototype Gene. J. Nutr. Biochem. 2007, 18, 497–508. [Google Scholar] [CrossRef]
- Polutchko, S.K.; Glime, G.N.E.; Demmig-Adams, B. Synergistic Action of Membrane-Bound and Water-Soluble Antioxidants in Neuroprotection. Molecules 2021, 26, 5385. [Google Scholar] [CrossRef]
- Kumar, A.; Prasad, A.; Sedlářová, M.; Ksas, B.; Havaux, M.; Pospíšil, P. Interplay between Antioxidants in Response to Photooxidative Stress in Arabidopsis. Free Radic. Biol. Med. 2020, 160, 894–907. [Google Scholar] [CrossRef]
- Havaux, M.; García-Plazaola, J.I. Beyond Non-Photochemical Fluorescence Quenching: The Overlapping Antioxidant Functions of Zeaxanthin and Tocopherols. In Non-Photochemical Quenching and Energy Dissipation in Plants, Algae and Cyanobacteria; Demmig-Adams, B., Garab, G., Adams, W.W., III, Govindjee, Eds.; Springer: Dordrecht, The Netherlands, 2014; Volume 40, pp. 583–603. [Google Scholar] [CrossRef]
- Fernández-Marín, B.; Roach, T.; Verhoeven, A.; García-Plazaola, J.I. Shedding Light on the Dark Side of Xanthophyll Cycles. New Phytol. 2021, 230, 1336–1344. [Google Scholar] [CrossRef] [PubMed]
- Triantaphylidès, C.; Havaux, M. Singlet Oxygen in Plants: Production, Detoxification and Signaling. Trends Plant Sci. 2009, 14, 219–228. [Google Scholar] [CrossRef]
- Schaller, F.; Biesgen, C.; Müssig, C.; Altmann, T.; Weiler, E.W. 12-Oxophytodienoate Reductase 3 (OPR3) Is the Isoenzyme Involved in Jasmonate Biosynthesis. Planta 2000, 210, 979–984. [Google Scholar] [CrossRef]
- Jones, G.D.; Russell, L.; Darley-Usmar, V.M.; Stone, D.; Wilson, M.T. Role of Lipid Hydroperoxides in the Activation of 15-Lipoxygenase. Biochemistry 1996, 35, 7197–7203. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Brash, A.R. On the Role of Molecular Oxygen in Lipoxygenase Activation. J. Biol. Chem. 2010, 285, 39876–39887. [Google Scholar] [CrossRef]
- Maccarrone, M.; Lorenzon, T.; Guerrieri, P.; Agrò, A.F. Resveratrol Prevents Apoptosis in K562 Cells by Inhibiting Lipoxygenase and Cyclooxygenase Activity. Eur. J. Biochem. 1999, 265, 27–34. [Google Scholar] [CrossRef]
- Park, N.-Y.; Im, S.; Jiang, Q. Different Forms of Vitamin E and Metabolite 13′-Carboxychromanols Inhibit Cyclooxygenase-1 and Its Catalyzed Thromboxane in Platelets, and Tocotrienols and 13′-Carboxychromanols Are Competitive Inhibitors of 5-Lipoxygenase. J. Nutr. Biochem. 2022, 100, 108884. [Google Scholar] [CrossRef]
- Maccarrone, M.; Van Zadelhoff, G.; Veldink, G.A.; Vliegenthart, J.F.G.; Finazzi-Agrò, A. Early Activation of Lipoxygenase in Lentil (Lens culinaris) Root Protoplasts by Oxidative Stress Induces Programmed Cell Death. Eur. J. Biochem. 2000, 267, 5078–5084. [Google Scholar] [CrossRef]
- Agrawal, G. Cloning of Novel Rice Allene Oxide Cyclase (OsAOC): mRNA Expression and Comparative Analysis with Allene Oxide Synthase (OsAOS) Gene Provides Insight into the Transcriptional Regulation of Octadecanoid Pathway Biosynthetic Genes in Rice. Plant Sci. 2003, 164, 979–992. [Google Scholar] [CrossRef]
- Breeze, E.; Mullineaux, P.M. The Passage of H2O2 from Chloroplasts to Their Associated Nucleus during Retrograde Signalling: Reflections on the Role of the Nuclear Envelope. Plants 2022, 11, 552. [Google Scholar] [CrossRef]
- Foyer, C.H.; Lopez-Delgado, H.; Dat, J.F.; Scott, I.M. Hydrogen Peroxide- and Glutathione-associated Mechanisms of Acclimatory Stress Tolerance and Signalling. Physiol. Plant. 1997, 100, 241–254. [Google Scholar] [CrossRef]
- Creelman, R.A.; Mullet, J.E. Jasmonic Acid Distribution and Action in Plants: Regulation during Development and Response to Biotic and Abiotic Stress. Proc. Natl. Acad. Sci. USA 1995, 92, 4114–4119. [Google Scholar] [CrossRef] [PubMed]
- Santino, A.; Taurino, M.; De Domenico, S.; Bonsegna, S.; Poltronieri, P.; Pastor, V.; Flors, V. Jasmonate Signaling in Plant Development and Defense Response to Multiple (a)Biotic Stresses. Plant Cell Rep. 2013, 32, 1085–1098. [Google Scholar] [CrossRef]
- Roach, T.; Krieger-Liszkay, A. The Role of the PsbS Protein in the Protection of Photosystems I and II against High Light in Arabidopsis thaliana. Biochim. Biophys. Acta BBA—Bioenerg. 2012, 1817, 2158–2165. [Google Scholar] [CrossRef] [PubMed]
- Külheim, C.; Jansson, S. What Leads to Reduced Fitness in Non-photochemical Quenching Mutants? Physiol. Plant. 2005, 125, 202–211. [Google Scholar] [CrossRef]
- Frenkel, M.; Külheim, C.; Jänkänpää, H.J.; Skogström, O.; Dall’Osto, L.; Ågren, J.; Bassi, R.; Moritz, T.; Moen, J.; Jansson, S. Improper Excess Light Energy Dissipation in Arabidopsis Results in a Metabolic Reprogramming. BMC Plant Biol. 2009, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Glazebrook, J. Contrasting Mechanisms of Defense Against Biotrophic and Necrotrophic Pathogens. Annu. Rev. Phytopathol. 2005, 43, 205–227. [Google Scholar] [CrossRef]
- Pokotylo, I.; Hodges, M.; Kravets, V.; Ruelland, E. A Ménage à Trois: Salicylic Acid, Growth Inhibition, and Immunity. Trends Plant Sci. 2022, 27, 460–471. [Google Scholar] [CrossRef]
- Toçilla, S. In the Spotlight: The Plant Growth-Defence Dilemma: A Hormonal Balancing Act. Physiol. Plant. 2024, 176, e14557. [Google Scholar] [CrossRef]
- Yang, L. You Can Have Your Cake and Eat It Too: Ectopic Expression of Cold-Regulated Genes Reshapes the Salicylic Acid–Mediated Growth-Defense Tradeoff. Plant Cell 2024, 36, 4270–4271. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Zhi, L.; Li, P.; Hancock, J.T.; Hu, X. The Role of Salicylic Acid Signal in Plant Growth, Development and Abiotic Stress. Phyton 2022, 91, 2591–2605. [Google Scholar] [CrossRef]
- Rivas-San Vicente, M.; Plasencia, J. Salicylic Acid beyond Defence: Its Role in Plant Growth and Development. J. Exp. Bot. 2011, 62, 3321–3338. [Google Scholar] [CrossRef]
- Jang, G.; Yoon, Y.; Choi, Y.D. Crosstalk with Jasmonic Acid Integrates Multiple Responses in Plant Development. Int. J. Mol. Sci. 2020, 21, 305. [Google Scholar] [CrossRef]
- Liu, H.; Timko, M.P. Jasmonic Acid Signaling and Molecular Crosstalk with Other Phytohormones. Int. J. Mol. Sci. 2021, 22, 2914. [Google Scholar] [CrossRef] [PubMed]
- Santisree, P.; Jalli, L.C.L.; Bhatnagar-Mathur, P.; Sharma, K.K. Emerging Roles of Salicylic Acid and Jasmonates in Plant Abiotic Stress Responses. In Protective Chemical Agents in the Amelioration of Plant Abiotic Stress; Aryadeep Roychoudhury, A., Kumar Tripathi, D., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2020; pp. 342–373. [Google Scholar] [CrossRef]
- Lu, Y.; Yao, J. Chloroplasts at the Crossroad of Photosynthesis, Pathogen Infection and Plant Defense. Int. J. Mol. Sci. 2018, 19, 3900. [Google Scholar] [CrossRef]
- Reymond, P.; Farmer, E.E. Jasmonate and Salicylate as Global Signals for Defense Gene Expression. Curr. Opin. Plant Biol. 1998, 1, 404–411. [Google Scholar] [CrossRef]
- Li, N.; Han, X.; Feng, D.; Yuan, D.; Huang, L.-J. Signaling Crosstalk between Salicylic Acid and Ethylene/Jasmonate in Plant Defense: Do We Understand What They Are Whispering? Int. J. Mol. Sci. 2019, 20, 671. [Google Scholar] [CrossRef] [PubMed]
- Klessig, D.F.; Choi, H.W.; Dempsey, D.A. Systemic Acquired Resistance and Salicylic Acid: Past, Present, and Future. Mol. Plant-Microbe Interact. 2018, 31, 871–888. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Stewart, J.J.; Adams, W.W., III. Less Photoprotection Can Be Good in Some Genetic and Environmental Contexts. Biochem. J. 2019, 476, 2017–2029. [Google Scholar] [CrossRef]
- Zhou, J.; Zeng, L.; Liu, J.; Xing, D. Manipulation of the Xanthophyll Cycle Increases Plant Susceptibility to Sclerotinia sclerotiorum. PLoS Pathog. 2015, 11, e1004878. [Google Scholar] [CrossRef]
- Kerchev, P.I.; Van Breusegem, F. Improving Oxidative Stress Resilience in Plants. Plant J. 2022, 109, 359–372. [Google Scholar] [CrossRef]
- Fichman, Y.; Rowland, L.; Oliver, M.J.; Mittler, R. ROS Are Evolutionary Conserved Cell-to-Cell Stress Signals. Proc. Natl. Acad. Sci. USA 2023, 120, e2305496120. [Google Scholar] [CrossRef]
- Mittler, R. ROS Are Good. Trends Plant Sci. 2017, 22, 11–19. [Google Scholar] [CrossRef]
- Jeong, M.-J.; Choi, B.S.; Bae, D.W.; Shin, S.C.; Park, S.U.; Lim, H.-S.; Kim, J.; Kim, J.B.; Cho, B.-K.; Bae, H. Differential Expression of Kenaf Phenylalanine Ammonia-Lyase (PAL) Ortholog during Developmental Stages and in Response to Abiotic Stresses. Plant Omics 2012, 5, 392–399. [Google Scholar]
- Chaouch, S.; Queval, G.; Vanderauwera, S.; Mhamdi, A.; Vandorpe, M.; Langlois-Meurinne, M.; Van Breusegem, F.; Saindrenan, P.; Noctor, G. Peroxisomal Hydrogen Peroxide Is Coupled to Biotic Defense Responses by ISOCHORISMATE SYNTHASE1 in a Daylength-Related Manner. Plant Physiol. 2010, 153, 1692–1705. [Google Scholar] [CrossRef] [PubMed]
- Lefevere, H.; Bauters, L.; Gheysen, G. Salicylic Acid Biosynthesis in Plants. Front. Plant Sci. 2020, 11, 338. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Noctor, G. Redox Signaling in Plants. Antioxid. Redox Signal. 2013, 18, 2087–2090. [Google Scholar] [CrossRef]
- Mittler, R.; Jones, D.P. The Redox Code of Plants. Plant Cell Environ. 2024, 47, 2821–2829. [Google Scholar] [CrossRef]
- Noctor, G.; Reichheld, J.-P.; Foyer, C.H. ROS-Related Redox Regulation and Signaling in Plants. Semin. Cell Dev. Biol. 2018, 80, 3–12. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demmig-Adams, B.; Hodges, A.K.; Polutchko, S.K.; Adams, W.W., III. Zeaxanthin and Other Carotenoids: Roles in Abiotic Stress Defense with Implications for Biotic Defense. Plants 2025, 14, 2703. https://doi.org/10.3390/plants14172703
Demmig-Adams B, Hodges AK, Polutchko SK, Adams WW III. Zeaxanthin and Other Carotenoids: Roles in Abiotic Stress Defense with Implications for Biotic Defense. Plants. 2025; 14(17):2703. https://doi.org/10.3390/plants14172703
Chicago/Turabian StyleDemmig-Adams, Barbara, Amy K. Hodges, Stephanie K. Polutchko, and William W. Adams, III. 2025. "Zeaxanthin and Other Carotenoids: Roles in Abiotic Stress Defense with Implications for Biotic Defense" Plants 14, no. 17: 2703. https://doi.org/10.3390/plants14172703
APA StyleDemmig-Adams, B., Hodges, A. K., Polutchko, S. K., & Adams, W. W., III. (2025). Zeaxanthin and Other Carotenoids: Roles in Abiotic Stress Defense with Implications for Biotic Defense. Plants, 14(17), 2703. https://doi.org/10.3390/plants14172703










