A Comprehensive Review Uncovering the Challenges and Advancements in the In Vitro Propagation of Eucalyptus Plantations
Abstract
1. Introduction
1.1. Challenges with Conventional Breeding
1.2. A Crop with Potential Biomass Production
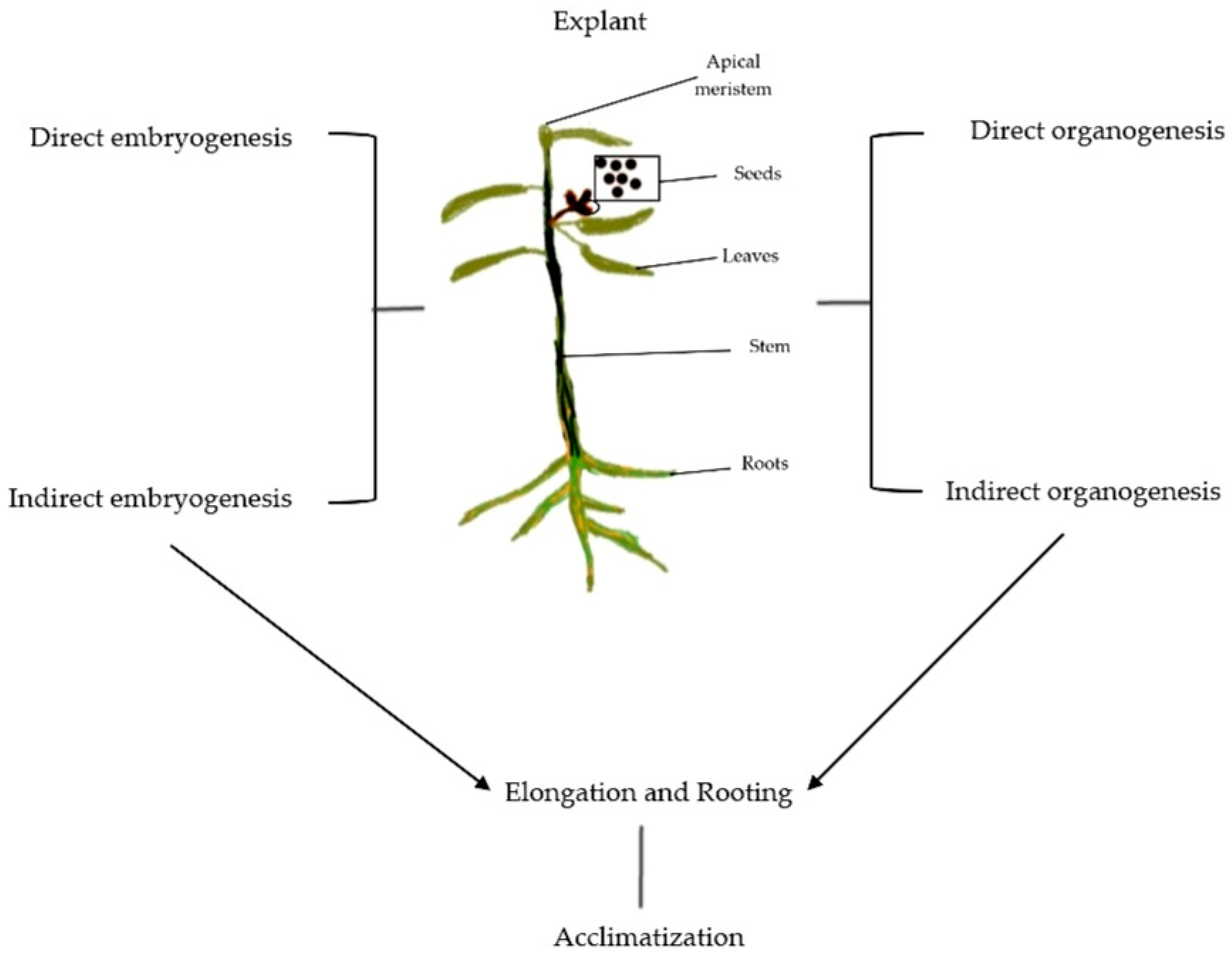
2. Micropropagation and Its Applications
2.1. Establishing Axenic Culture
2.2. In Vitro Proliferation of Shoot
2.2.1. Organogenesis
2.2.2. Somatic Embryogenesis
2.3. Adventitious Root Formation and Root Hardening
3. Factors Affecting the Efficiency of Micropropagation
3.1. Role of Plant Growth Regulators
3.2. Effect of Culture Media
3.3. Importance of Organic and Inorganic Elements
3.4. The Role of Carbohydrates
3.5. Effects of Radiation and Light Exposure
4. In Vitro Germplasm Preservation
5. Limitations, Challenges, and Future Directions
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Watt, M.P.; Blakeway, F.C.; Mokotedi, M.E.O.; Jain, S.M. Micropropagation of Eucalyptus. In Micropropagation of Woody Trees and Fruits; Jain, S.M., Ishii, K., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2003; Volume 75, pp. 217–244. [Google Scholar] [CrossRef]
- Trueman, S.J.; Hung, C.D.; Wendling, I. Tissue Culture of Corymbia and Eucalyptus. Forests 2018, 9, 84. [Google Scholar] [CrossRef]
- Nogueira, M.C.D.J.A.; Araujo, V.A.D.; Vasconcelos, J.S.; Christoforo, A.L.; Lahr, F.A.R. Sixteen Properties of Eucalyptus tereticornis Wood for Structural Uses. Biosci. J. 2020, 36, 449–457. [Google Scholar] [CrossRef]
- Silva, J.; Abebe, W.; Sousa, S.M.; Duarte, V.G.; Machado, M.I.L.; Matos, F.J.A. Analgesic and Anti-Inflammatory Effects of Essential Oils of Eucalyptus. J. Ethnopharmacol. 2003, 89, 277–283. [Google Scholar] [CrossRef]
- Kamal, B.; Arya, I.; Sharma, V.; Jadon, V.S. In Vitro Enhanced Multiplication and Molecular Validation of Eucalyptus F1 Hybrids. Plant Cell Biotechnol. Mol. Biol. 2016, 17, 167–175. [Google Scholar]
- Arya, I.D.; Chauhan, S.S.S.; Arya, S. Micropropagation of Superior Eucalyptus Hybrids FRI-5 (Eucalyptus camaldulensis Dehn × E. tereticornis Sm) and FRI-14(Eucalyptus Torelliana F.V. Muell × E. citriodora Hook): A Commercial Multiplication and Field Evaluation. Afr. J. Biotechnol. 2009, 8, 5718–5726. [Google Scholar] [CrossRef]
- Kendurkar, S.V.; Rangaswamy, M. Genetic Transformation in Eucalyptus. In Biotechnologies of Crop Improvement, Volume 2; Gosal, S.S., Wani, S.H., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 335–366. [Google Scholar] [CrossRef]
- El-Esawi, M.A. Micropropagation Technology and Its Applications for Crop Improvement. In Plant Tissue Culture: Propagation, Conservation and Crop Improvement; Anis, M., Ahmad, N., Eds.; Springer: Singapore, 2016; pp. 523–545. [Google Scholar] [CrossRef]
- Trueman, S.J. Clonal Propagation and Storage of Subtropical Pines in Queensland, Australia. S. Afr. For. J. 2006, 208, 49–52. [Google Scholar] [CrossRef]
- Booth, T.H. Eucalypt Plantations and Climate Change. Ecol. Manag. 2013, 301, 28–34. [Google Scholar] [CrossRef]
- Vilasboa, J.; Da Costa, C.T.; Fett-Neto, A.G. Environmental Modulation of Mini-Clonal Gardens for Cutting Production and Propagation of Hard- and Easy-to-Root Eucalyptus spp. Plants 2022, 11, 3281. [Google Scholar] [CrossRef]
- Shanthi, K.; Bachpai, V.K.W.; Anisha, S.; Ganesan, M.; Anithaa, R.G.; Subashini, V.; Chakravarthi, M.; Sivakumar, V.; Yasodha, R. Micropropagation of Eucalyptus camaldulensis for the Production of Rejuvenated Stock Plants for Microcuttings Propagation and Genetic Fidelity Assessment. New For. 2015, 46, 357–371. [Google Scholar] [CrossRef]
- Kataria, V.; Masih, A.; Chauhan, S.; Sharma, S.K.; Kant, A.; Arya, I.D. Clonal Propagation, a Tested Technique for Increasing Productivity: A Review of Bamboos, Eucalyptus and Chirpine. In Agricultural Biotechnology: Latest Research and Trends; Kumar Srivastava, D., Kumar Thakur, A., Kumar, P., Eds.; Springer Nature: Singapore, 2021; pp. 37–51. [Google Scholar] [CrossRef]
- Butcher, P.A.; Skinner, A.K.; Gardiner, C.A. Increased Inbreeding and Inter-Species Gene Flow in Remnant Populations of the Rare Eucalyptus benthamii. Conserv. Genet. 2005, 6, 213–226. [Google Scholar] [CrossRef]
- Potts, B.M.; Dungey, H.S. Interspecific Hybridization of Eucalyptus: Key Issues for Breeders and Geneticists. New For. 2004, 27, 115–138. [Google Scholar] [CrossRef]
- Jovanovic, T.; Arnold, R.; Booth, T. Determining the Climatic Suitability of Eucalyptus dunnii for Plantations in Australia, China and Central and South America. New For. 2000, 19, 215–226. [Google Scholar] [CrossRef]
- Singh, D.; Kaur, S.; Kumar, A. In Vitro Drought Tolerance in Selected Elite Clones of Eucalyptus tereticornis Sm. Acta Physiol. Plant 2020, 42, 17. [Google Scholar] [CrossRef]
- Venkatesh, C.S.; Sharma, V.K. Hybrid Vigour in Controlled Interspecific Crosses of Eucalyptus tereticornis × E. camaldulensis. Silvae Genet. 1977, 26, 121–124. [Google Scholar]
- Paramathma, M.; Surendran, C.; Rai, R.S.V. Studies on heterosis in six Eucalyptus species. J. Trop. For. Sci. 1997, 9, 283–293. [Google Scholar]
- De Almeida, M.R.; De Bastiani, D.; Gaeta, M.L.; De Araujo Mariath, J.E.; De Costa, F.; Retallick, J.; Nolan, L.; Tai, H.H.; Strömvik, M.V.; Fett-Neto, A.G. Comparative Transcriptional Analysis Provides New Insights into the Molecular Basis of Adventitious Rooting Recalcitrance in Eucalyptus. Plant Sci. 2015, 239, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, G.R.; Wallace, H.M.; Lee, D.J. Reciprocal and Advanced Generation Hybrids between Corymbia citriodora and C. torelliana: Forestry Breeding and the Risk of Gene Flow. Ann. For. Sci. 2013, 70, 1–10. [Google Scholar] [CrossRef]
- Wendling, I.; Warburton, P.; Trueman, S. Maturation in Corymbia torelliana × C. citriodora Stock Plants: Effects of Pruning Height on Shoot Production, Adventitious Rooting Capacity, Stem Anatomy, and Auxin and Abscisic Acid Concentrations. Forests 2015, 6, 3763–3778. [Google Scholar] [CrossRef]
- Debergh, P.C.; Read, P.E. Micropropagation. In Micropropagation: Technology and Application; Debergh, P.C., Zimmerman, R.H., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 1991; pp. 1–13. [Google Scholar] [CrossRef]
- Cassells, A.C. Pathogen and Biological Contamination Management in Plant Tissue Culture: Phytopathogens, Vitro Pathogens, and Vitro Pests. In Plant Cell Culture Protocols; Loyola-Vargas, V.M., Ochoa-Alejo, N., Eds.; Humana Press: Totowa, NJ, USA, 2012; Volume 877, pp. 57–80. [Google Scholar] [CrossRef]
- Wendling, I.; Brooks, P.R.; Trueman, S.J. Topophysis in Corymbia torelliana × C. citriodora Seedlings: Adventitious Rooting Capacity, Stem Anatomy, and Auxin and Abscisic Acid Concentrations. New For. 2015, 46, 107–120. [Google Scholar] [CrossRef]
- Bag, N.; Chandra, S.; Palni, L.M.S.; Nandi, S.K. Micropropagation of Dev-Ringal [Thamnocalamus spathiflorus (Trin.) Munro]—A Temperate Bamboo, and Comparison between in Vitro Propagated Plants and Seedlings. Plant Sci. 2000, 156, 125–135. [Google Scholar] [CrossRef]
- Giri, C.C.; Shyamkumar, B.; Anjaneyulu, C. Progress in Tissue Culture, Genetic Transformation and Applications of Biotechnology to Trees: An Overview. Trees 2004, 18, 115–135. [Google Scholar] [CrossRef]
- Ansar, S.; Iqbal, M. Effect of Dietary Antioxidant on Mercuric Chloride Induced Lung Toxicity and Oxidative Stress. Toxin Rev. 2015, 34, 168–172. [Google Scholar] [CrossRef]
- Keret, R.; Nakhooda, M.; Jones, N.B.; Hills, P.N. Optimisation of Micropropagation Protocols for Temperate Eucalypt Hybrids in South Africa, with a Focus on Auxin Transport Proteins. South. For. J. For. Sci. 2021, 83, 254–263. [Google Scholar] [CrossRef]
- Trueman, S.J.; Richardson, D.M. In Vitro Propagation of Corymbia torelliana × C. citriodora (Myrtaceae) via Cytokinin-Free Node Culture. Aust. J. Bot. 2007, 55, 471. [Google Scholar] [CrossRef]
- Kuppusamy, S.; Ramanathan, S.; Sengodagounder, S.; Senniappan, C.; Shanmuganathan, R.; Brindhadevi, K.; Kaliannan, T. Optimizing the Sterilization Methods for Initiation of the Five Different Clones of the Eucalyptus Hybrid Species. Biocatalaysis Agric. Biotechnol. 2019, 22, 101361. [Google Scholar] [CrossRef]
- Kamal, B.; Arya, I.D.; Gupta, S. In-Vitro Regeneration of Interspecific F1 Hybrid (Eucalyptus citriodora and Eucalyptus torelliana) of Eucalyptus. J. Mt. Res. 2022, 17, 125–130. [Google Scholar] [CrossRef]
- Aggarwal, D.; Kumar, A.; Reddy, M.S. Shoot Organogenesis in Elite Clones of Eucalyptus tereticornis. Plant Cell Tiss. Organ. Cult 2010, 102, 45–52. [Google Scholar] [CrossRef]
- Girijashankar, V. In Vitro Regeneration of Eucalyptus camaldulensis. Physiol. Mol. Biol. Plants 2012, 18, 79–87. [Google Scholar] [CrossRef][Green Version]
- Fernando, S.C.; Goodger, J.Q.D.; Gutierrez, S.S.; Johnson, A.A.T.; Woodrow, I.E. Plant Regeneration through Indirect Organogenesis and Genetic Transformation of Eucalyptus polybractea R.T. Baker. Ind. Crops Prod. 2016, 86, 73–78. [Google Scholar] [CrossRef]
- Sluis, A.; Hake, S. Organogenesis in Plants: Initiation and Elaboration of Leaves. Trends Genet. 2015, 31, 300–306. [Google Scholar] [CrossRef]
- Oberschelp, G.P.J.; Gonçalves, A.N. Assessing the Effects of Basal Media on the in Vitro Propagation and Nutritional Status of Eucalyptus dunnii Maiden. In Vitro Cell. Dev. Biol.—Plant 2016, 52, 28–37. [Google Scholar] [CrossRef]
- Méndez-Hernández, H.A.; Ledezma-Rodríguez, M.; Avilez-Montalvo, R.N.; Juárez-Gómez, Y.L.; Skeete, A.; Avilez-Montalvo, J.; De-La-Peña, C.; Loyola-Vargas, V.M. Signaling Overview of Plant Somatic Embryogenesis. Front. Plant Sci. 2019, 10, 77. [Google Scholar] [CrossRef]
- Martínez, M.T.; del San-José, M.C.; Arrillaga, I.; Cano, V.; Morcillo, M.; Cernadas, M.J.; Corredoira, E. Holm Oak Somatic Embryogenesis: Current Status and Future Perspectives. Front Plant Sci 2019, 10, 239. [Google Scholar] [CrossRef]
- Pinto, G.; Silva, S.; Park, Y.-S.; Neves, L.; Araújo, C.; Santos, C. Factors Influencing Somatic Embryogenesis Induction in Eucalyptus globulus Labill.: Basal Medium and Anti-Browning Agents. Plant Cell Tiss. Organ Cult. 2008, 95, 79–88. [Google Scholar] [CrossRef]
- Pinto, G.; Park, Y.-S.; Neves, L.; Araújo, C.; Santos, C. Genetic Control of Somatic Embryogenesis Induction in Eucalyptus globulus Labill. Plant Cell Rep. 2008, 27, 1093–1101. [Google Scholar] [CrossRef]
- Pinto, G.; Park, Y.-S.; Silva, S.; Neves, L.; Araújo, C.; Santos, C. Factors Affecting Maintenance, Proliferation, and Germination of Secondary Somatic Embryos of Eucalyptus globulus Labill. Plant Cell Tiss. Organ. Cult. 2008, 95, 69–78. [Google Scholar] [CrossRef]
- Pinto, G.; Silva, S.; Neves, L.; Araújo, C.; Santos, C. Histocytological Changes and Reserve Accumulation during Somatic Embryogenesis in Eucalyptus globulus. Trees 2010, 24, 763–769. [Google Scholar] [CrossRef]
- Glocke, P.; Delaporte, K.; Collins, G.; Sedgley, M. Micropropagation of Juvenile Tissue of Eucalyptus erythronema × Eucalyptus stricklandii Cv. ‘Urrbrae Gem’. In Vitro Cell. Dev. Biol.—Plant 2006, 42, 139–143. [Google Scholar] [CrossRef]
- Tanaka, M.; Giang, D.T.T.; Murakami, A. Application of a Novel Disposable Film Culture System to Photoautotrophic Micropropagation of Eucalyptus uro-grandis (Urophylia × grandis). In Vitro Cell. Dev. Biol.—Plant 2005, 41, 173–180. [Google Scholar] [CrossRef]
- Trueman, S.J.; McMahon, T.V.; Bristow, M. Production of Eucalyptus cloeziana Cuttings in Response to Stock Plant Temperature. J. Trop. For. Sci. 2013, 25, 60–69. [Google Scholar]
- De Oliveira, L.S.; Brondani, G.E.; Molinari, L.V.; Dias, R.Z.; Teixeira, G.L.; Gonçalves, A.N.; De Almeida, M. Optimal Cytokinin/Auxin Balance for Indirect Shoot Organogenesis of Eucalyptus cloeziana and Production of Ex Vitro Rooted Micro-Cuttings. J. For. Res. 2022, 33, 1573–1584. [Google Scholar] [CrossRef]
- Prakash, M.G.; Gurumurthi, K. Effects of Type of Explant and Age, Plant Growth Regulators and Medium Strength on Somatic Embryogenesis and Plant Regeneration in Eucalyptus camaldulensis. Plant Cell Tiss. Organ Cult. 2010, 100, 13–20. [Google Scholar] [CrossRef]
- Brondani, G.E.; Dutra, L.F.; Wendling, I.; Grossi, F.; Hansel, F.A.; Araujo, M.A. Micropropagation of an Eucalyptus hybrid (Eucalyptus benthamii × Eucalyptus dunnii). Acta Sci. Agron. 2011, 33, 8317. [Google Scholar] [CrossRef]
- Brondani, G.E.; de Wit Ondas, H.W.; Baccarin, F.J.B.; Gonçalves, A.N.; de Almeida, M. Micropropagation of Eucalyptus benthamii to Form a Clonal Micro-Garden. In Vitro Cell. Dev. Biol.—Plant 2012, 48, 478–487. [Google Scholar] [CrossRef]
- Gomes, F.; Canhoto, J.M. Micropropagation of Eucalyptus nitens Maiden (Shining Gum). In Vitro Cell. Dev. Biol.—Plant 2003, 39, 316–321. [Google Scholar] [CrossRef]
- Mankessi, F.; Saya, A.; Baptiste, C.; Nourissier, S.; Monteuuis, O. In Vitro Rooting of Genetically Related Eucalyptus urophylla × Eucalyptus grandis Clones in Relation to the Time Spent in Culture. Trees 2009, 23, 931–940. [Google Scholar] [CrossRef]
- Fett-Neto, A.G.; Fett, J.P.; Goulart, L.W.V.; Pasquali, G.; Termignoni, R.R.; Ferreira, A.G. Distinct Effects of Auxin and Light on Adventitious Root Development in Eucalyptus saligna and Eucalyptus globulus. Tree Physiol. 2001, 21, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Watt, M.P.; Thokoane, N.L.; Mycock, D.; Blakeway, F. In Vitro Storage of Eucalyptus grandis Germplasm under Minimal Growth Conditions. Plant Cell Tissue Organ Cult. 2000, 61, 161–164. [Google Scholar] [CrossRef]
- Blakesley, D.; Kiernan, R. Cryopreservation of Axillary Buds of a Eucalyptus grandis × Eucalyptus camaldulensis Hybrid. Cryo Lett. 2001, 22, 13–18. [Google Scholar]
- Souza, D.M.S.C.; Fernandes, S.B.; Avelar, M.L.M.; Frade, S.R.D.P.; Molinari, L.V.; Gonçalves, D.S.; Pinto, J.E.B.P.; Brondani, G.E. Light Quality in Micropropagation of Eucalyptus grandis × Eucalyptus urophylla. Sci. For. 2020, 48, e3329. [Google Scholar] [CrossRef]
- Thomas, T.D. The Role of Activated Charcoal in Plant Tissue Culture. Biotechnol. Adv. 2008, 26, 618–631. [Google Scholar] [CrossRef]
- Jones, N.B.; van Staden, J. Micropropagation and Establishment of Eucalyptus grandis Hybrids. S. Afr. J. Bot. 1994, 60, 122–126. [Google Scholar] [CrossRef]
- Sapaeing, A.; Sutthinon, P.; Hilae, A.; Wattanapan, N. Effects of BA, NAA, and Activated Charcoal on Micropropagation of Nepenthes Mirabilis (Lour.) Druce. Acta Hortic. 2020, 1298, 281–286. [Google Scholar] [CrossRef]
- Xu, Y.; Liang, Y.; Yang, M. Effects of Composite LED Light on Root Growth and Antioxidant Capacity of Cunninghamia lanceolata Tissue Culture Seedlings. Sci. Rep. 2019, 9, 9766. [Google Scholar] [CrossRef] [PubMed]
- Murthy, H.N.; Joseph, K.S.; Paek, K.Y.; Park, S.Y. Bioreactor Systems for Micropropagation of Plants: Present Scenario and Future Prospects. Front. Plant Sci. 2023, 14, 1159588. [Google Scholar] [CrossRef]
- Zobayed, S. Mass Propagation of Eucalyptus camaldulensis in a Scaled-up Vessel Under In Vitro Photoautotrophic Condition. Ann. Bot. 2000, 85, 587–592. [Google Scholar] [CrossRef]
- Kozai, T.; Afreen, F.; Zobayed, S.M.A. (Eds.) . Photoautotrophic (Sugar-Free Medium) Micropropagation as a New Micropropagation and Transplant Production System; Springer: Berlin/Heidelberg, Germany, 2005. [Google Scholar] [CrossRef]
- Mendonça, E.G.; Batista, T.R.; Stein, V.C.; Balieiro, F.P.; de Abreu, J.R.; Pires, M.F.; de Souza, P.A.; Paiva, L.V. In Vitro Serial Subculture to Improve Rooting of Eucalyptus urophylla. New For. 2020, 51, 801–816. [Google Scholar] [CrossRef]
- Leva, A. (Ed.) Recent Advances in Plant In Vitro Culture; InTech: London, UK, 2012. [Google Scholar] [CrossRef]
- Zhao, Y. Auxin Biosynthesis. Arab. Book 2014, 12, e0173. [Google Scholar] [CrossRef]
- Frick, E.M.; Strader, L.C. Roles for IBA-Derived Auxin in Plant Development. J. Exp. Bot. 2018, 69, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Strader, L.C.; Bartel, B. Transport and Metabolism of the Endogenous Auxin Precursor Indole-3-Butyric Acid. Mol. Plant. 2011, 4, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Nazirah, A.; Nor-Hasnida, H.; Mohd-Saifuldullah, A.W.; Muhammad-Fuad, Y.; Ahmad-Zuhaidi, Y.; Rozidah, K. Development of an Efficient Micropropagation Protocol for Eucalyptus Hybrid (E. urophylla × E. grandis) through axillary shoot proliferation. J. Trop. For. Sci. 2021, 33, 391–397. [Google Scholar] [CrossRef]
- Shwe, S.S.; Leung, D.W.M. Plant Regeneration from Eucalyptus bosistoana Callus Culture. In Vitro Cell. Dev.Biol.—Plant 2020, 56, 718–725. [Google Scholar] [CrossRef]
- Faria, J.C.T.; Ribeiro-Kumara, C.; Costa, R.S.D.R.; Nieri, E.M.; De Carvalho, D.; Pinto, J.E.B.P.; Neto, A.R.D.S.; Brondani, G.E. Use of Biodegradable Polyester-Based Microvessels for Micropropagation of Mature Eucalyptus microcorys. N. Z. J. For. Sci. 2022, 52, 1–13. [Google Scholar] [CrossRef]
- Skůpa, P.; Opatrný, Z.; Petrášek, J. Auxin Biology: Applications and the Mechanisms Behind. In Applied Plant Cell Biology; Nick, P., Opatrny, Z., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; Volume 22, pp. 69–102. [Google Scholar] [CrossRef]
- Teale, W.D.; Paponov, I.A.; Palme, K. Auxin in Action: Signalling, Transport and the Control of Plant Growth and Development. Nat. Rev. Mol. Cell. Biol. 2006, 7, 847–859. [Google Scholar] [CrossRef]
- Bunn, E. Development of in Vitro Methods for Ex Situ Conservation of Eucalyptus impensa, an Endangered Mallee from Southwest Western Australia. Plant Cell. Tiss. Organ. Cult. 2005, 83, 97–102. [Google Scholar] [CrossRef]
- Kaur, S. In Vitro Regeneration of Shoots From Nodal Explants of Dendrobium chrysotoxum Lindl. J. Hortic. Res. 2017, 25, 27–34. [Google Scholar] [CrossRef][Green Version]
- Woodward, A.W.; Bartel, B. Auxin: Regulation, Action, and Interaction. Ann. Bot. 2005, 95, 707–735. [Google Scholar] [CrossRef]
- Chen, J.; Ziv, M. The Effect of Ancymidol on Hyperhydricity, Regeneration, Starch and Antioxidant Enzymatic Activities in Liquid-Cultured Narcissus. Plant Cell Rep. 2001, 20, 22–27. [Google Scholar] [CrossRef]
- Ngomuo, M.; Mneney, E.; Ndakidemi, P.A. The In Vitro Propagation Techniques for Producing Banana Using Shoot Tip Cultures. Am. J. Plant Sci. 2014, 05, 1614–1622. [Google Scholar] [CrossRef]
- Al-Aizari, A.A.; Al-Obeed, R.S.; Mohamed, M.A.H. Improving Micropropagation of Some Grape Cultivars via Boron, Calcium and Phosphate. Electron. J. Biotechnol. 2020, 48, 95–100. [Google Scholar] [CrossRef]
- George, E.F.; Hall, M.A.; Klerk, G.-J.D. (Eds.) The Components of Plant Tissue Culture Media I: Macro- and Micro-Nutrients. In Plant Propagation by Tissue Culture; Springer Netherlands: Dordrecht, The Netherlands, 2007; pp. 65–113. [Google Scholar] [CrossRef]
- Pérez-Tornero, O.; Burgos, L. Different Media Requirements for Micropropagation of Apricot Cultivars. Plant Cell Tissue Organ Cult. 2000, 63, 133–141. [Google Scholar] [CrossRef]
- White, P.J.; Broadley, M.R. Calcium in Plants. Ann. Bot. 2003, 92, 487–511. [Google Scholar] [CrossRef] [PubMed]
- Al-Mayahi, A.M.W. Effect of Calcium and Boron on Growth and Development of Callus and Shoot Regeneration of Date Palm ‘Barhee’. Can. J. Plant Sci. 2020, 100, 357–364. [Google Scholar] [CrossRef]
- Brunoni, F.; Rolli, E.; Dramis, L.; Incerti, M.; Abarca, D.; Pizarro, A.; Diaz-Sala, C.; Ricci, A. Adventitious Rooting Adjuvant Activity of 1,3-Di(Benzo[d]Oxazol-5-Yl)Urea and 1,3-Di(Benzo[d]Oxazol-6-Yl)Urea: New Insights and Perspectives. Plant Cell Tiss. Organ Cult. 2014, 118, 111–124. [Google Scholar] [CrossRef]
- Lopes da Silva, A.L.; Gollo, A.; Brondani, G.; Horbach, M.; Oliveira, L.; Machado, M.; Lima, K.; Costa, J. Micropropagation of Eucalyptus saligna Sm. from Cotyledonary Nodes. Pak. J. Bot. 2015, 47, 311–318. [Google Scholar]
- Sharma, S.; Ramamurthy, V. Micropropagation of 4-Year-Old Elite Eucalyptus tereticornis Trees. Plant Cell Rep. 2000, 19, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Gago, D.; Vilavert, S.; Bernal, M.Á.; Sánchez, C.; Aldrey, A.; Vidal, N. The Effect of Sucrose Supplementation on the Micropropagation of Salix viminalis L. Shoots in Semi-solid Medium and Temporary Immersion Bioreactors. Forests 2021, 12, 1408. [Google Scholar] [CrossRef]
- Joshi, I.; Bisht, P.; Sharma, V.K.; Uniyal, D.P. In Vitro Clonal Propagation of Mature Eucalyptus F1 Hybrid (Eucalyptus tereticornis Sm. x E. grandis Hill Ex. Maiden). Silvae Genet. 2003, 52, 110–113. [Google Scholar]
- Gago, D.; Bernal, M.Á.; Sánchez, C.; Aldrey, A.; Cuenca, B.; Christie, C.B.; Vidal, N. Effect of Sucrose on Growth and Stress Status of Castanea sativa × C. crenata Shoots Cultured in Liquid Medium. Plants 2022, 11, 965. [Google Scholar] [CrossRef]
- Sandhu, M.; Wani, S.H.; Jiménez, V.M. In Vitro Propagation of Bamboo Species through Axillary Shoot Proliferation: A Review. Plant Cell Tiss Organ Cult. 2018, 132, 27–53. [Google Scholar] [CrossRef]
- Nadgauda, R.S.; Parasharami, V.A.; Mascarenhas, A.F. Precocious Flowering and Seeding Behaviour in Tissue-Cultured Bamboos. Nature 1990, 344, 335–336. [Google Scholar] [CrossRef]
- Shen, G.; Tan, S.; Sun, X.; Chen, Y.; Li, B. Experimental Evidence for the Importance of Light on Understory Grass Communities in a Subtropical Forest. Front Plant Sci. 2020, 11, 1051. [Google Scholar] [CrossRef] [PubMed]
- Fogaça, C.M.; Fett-Neto, A.G. Role of Auxin and Its Modulators in the Adventitious Rooting of Eucalyptus Species Differing in Recalcitrance. Plant Growth Regul. 2005, 45, 1–10. [Google Scholar] [CrossRef]
- Xiao, Y.; Niu, G.; Kozai, T. Development and Application of Photoautotrophic Micropropagation Plant System. Plant Cell Tiss. Organ Cult. 2011, 105, 149–158. [Google Scholar] [CrossRef]
- Miranda, N.A.; Xavier, A.; Otoni, W.C.; Gallo, R.; Gatti, K.C.; de Moura, L.C.; Souza, D.M.S.C.; Maggioni, J.H.; de Santos, S.S.O. Quality and Intensity of Light in the In Vitro Development of Microstumps of Eucalyptus urophylla in a Photoautotrophic System. For. Sci. 2020, 66, 754–760. [Google Scholar] [CrossRef]
- Rangel Do Prado Frade, S.; Santana Costa Souza, D.M.; Fernandes, S.B.; Lopes Martins Avelar, M.; Vaz Molinari, L.; Santos Gonçalves, D.; Alves Magalhães, T.; Brondani, G.E. Spectral Quality Influence on in Vitro Morphophysiological Responses of Eucalyptus dunnii Maiden and Eucalyptus grandis W.Hill Ex Maiden × E. urophylla ST Blake. N. Z. J. For. Sci. 2023, 53, 1–16. [Google Scholar] [CrossRef]
- Souza, D.M.S.C.; Avelar, M.L.M.; Fernandes, S.B.; Silva, E.O.; Duarte, V.P.; Molinari, L.V.; Brondani, G.E. Spectral Quality and Temporary Immersion Bioreactor for in Vitro Multiplication of Eucalytpus grandis × Eucalyptus urophylla. 3 Biotech 2020, 10, 457. [Google Scholar] [CrossRef]
- Matheus, D.; Souza, S.C.; Martins, A.R.; Fernandes, S.B.; Lopes Martins Avelar, M.; Vaz Molinari, L.; Santos Gonçalves, D.; Brondani, G.E. In Vitro Multiplication of Eucalyptus pilularis and Eucalyptus grandis × E. urophylla (Urograndis Eucalypt): Effect of Light Quality in Temporary Immersion Bioreactor. Mindanao J. Sci. Technol. 2022, 20, 72–86. [Google Scholar]
- Roque-Borda, C.A.; Kulus, D.; Vacaro De Souza, A.; Kaviani, B.; Vicente, E.F. Cryopreservation of Agronomic Plant Germplasm Using Vitrification-Based Methods: An Overview of Selected Case Studies. Int. J. Mol. Sci. 2021, 22, 6157. [Google Scholar] [CrossRef]
- Padayachee, K.; Watt, M.; Edwards, N.; Mycock, D. Cryopreservation as a Tool for the Conservation of Eucalyptus Genetic Variability: Concepts and Challenges. South. For. 2009, 71, 165–170. [Google Scholar] [CrossRef]
- Hung, C.D.; Trueman, S.J. Preservation of Encapsulated Shoot Tips and Nodes of the Tropical Hardwoods Corymbia torelliana × C. citriodora and Khaya Senegalensis. Plant Cell Tiss. Organ Cult. 2012, 109, 341–352. [Google Scholar] [CrossRef]


| Explant-Source/Type | Culture Media/PGRs/Additives | Experimental Outcomes/Remarks/Productivity/Root Hardening, etc. | References |
|---|---|---|---|
| Nodal segments | MS media, WPM, 0.5 mg L−1 IBA, 1.0 mg L−1 BAP, B5, NAA, Kinetin | Full-strength MS media supplemented with 1.0 mg L−1 BAP showed the best shoot elongation; ½ MS media supplemented with 0.5 mg L−1 IBA showed the best rooting; the resulting hybrid produced yielded 3–5 times more wood than parental species. | [5] |
| Nodal segments (30–32-year trees) | MS media, WPM, B5 medium, SH medium, IBA, BAP, NAA, | Best in vitro studies were obtained from the explants collected during the period of January to February and August to September; ½ MS media + BAP resulted in best rooting (92%); up to 98% of plants survived acclimatization. | [6] |
| Stem cuttings and axillary buds | MS Media, 2.22 µm BA, 1.16 µm Kinetin, 0.029 µm gibberellic acid, 400 mg L−1 PVP, 30 g L−1 sucrose | Micropropagation and microcuttings showed higher adventitious rooting (24.8–100% and 43–95%, respectively) than stem cuttings (9.3–75.5%). | [12] |
| Maintained Elite clones (KE8, CE2, T1, and Y8) | Basal MS media, 2.5 µM BA, 0.5 µM NAA, D-mannitol (0, 250, 500, 750, and 1000 mM) | The culture growth index of all clones was reduced significantly because of drought stress. | [17] |
| Cuttings from plants grown in vitro | Full and ½ strength MS media | Concentrations of IAA, IBA, and stem anatomy had no effect on the rooting potential of shoots. | [22] |
| Nodal cuttings | IAA, IBA (0, 1, 3, 8 g Kg−1) | The position from which the explant is harvested can affect the rooting potential and seed vigor. Explants obtained from 7/8 and 9/10 apical positions showed enhanced rooting and shooting. | [25] |
| Cuttings from 6-month-old parental plants | MS media, IAA, meta-topolin, kinetin, BAP, vit. B5, biotin, sucrose | 0.5 mg L−1 meta-topolin and 1 mg L−1 IAA enhanced shoot elongation as well as bud proliferation, while 0.5 mg L−1 IAA resulted in the most consistent rooting percentages. Moreover, equal expression of AUX1 and PIN1 transporter genes increased responsiveness toward PGRs. | [29] |
| Nodal segments | MS media, 58 mM sucrose, 0.5 μM NAA, 2.5 μM BA, | Media supplemented with 1.0 μM 2,4-D, 5.0 μM BA and 500 mg L−1 cefotaxime showed maximum (44.6%) shoot bud organogenesis. | [33] |
| Nodal segments | MS media, 2 mg L−1 BAP, 0.1 mg L−1 NAA | Media supplemented with 0.5 mg L−1 showed the best shoot elongation, ½ MS + 1 mg L−1 IAA showed the best root induction and elongation, and direct regeneration was observed in MS + 20:1 BAP: NAA. | [34] |
| Young shoot segments | WPM, MS media, 2iP, NAA, BAP, sucrose | Media supplemented with BA resulted in 99% shoot proliferation, media supplemented with 2iP resulted in 93% shoot regeneration, and IBA promoted rooting in 60% of the clones. | [35] |
| Nodal segments | EDM basal media (a novel basal media for E. dunnii) supplemented with 20 g L−1 sucrose and without PGRs | Higher Fe, Cu, Zn, and Mn concentrations in EDMm media increased rooting. Moreover, high S and K concentrations in EDMm increased growth rate and multiplication. Also, no Fe chlorosis/oxidation was observed in shoots cultured on EDMm. | [37] |
| Zygotic embryo | One of the following media and growth regulators: ½ or full-strength MS media/WPM/B5/DKW/JADS media/3 mg L−1 NAA/10 mL−1 silver nitrate/0.5 mg L−1 DTT/100 mg L−1 ascorbic acid/0.5 mg L−1 DTE/1% m/v PVP/1% m/v PVPP/0.01% w/v activated charcoal | The best media for somatic embryogenesis were B5 and MS. Moreover, Silver nitrate, activated charcoal, and DTE reduced the browning of explants. | [41] |
| Somatic embryos | MS media supplemented with 3 mg L−1 NAA | MS medium without PGRs is highly efficient for promoting cotyledonary embryo proliferation and germination. | [42] |
| Zygotic embryo cotyledons | Hormone-free MS media | The reserve accumulation of mature zygotic embryos was analyzed. Cotyledonary somatic embryos possess a low density of starch and no lipids/proteins. | [43] |
| Axillary shoots | ½ MS, 4.4 μM, 1 μM NAA, 1 g L−1 sucrose | WPM and QL media supplemented with Gibberellic acids showed enhanced shoot proliferation, ½ WPM supplemented with 20 μM IBA showed enhanced rooting, and 67% Plantlet hardening was achieved. | [44] |
| Shoot segments | MS media, 0.02 mg L−1 IBA | Vitron vessel placed in Low Photon Flux density at 3000 ppm CO2 for 24 h/day yielded the best growth and quality of plantlets. | [45] |
| Seedlings grown from seeds | Variable potting mixture | Low temperatures of 18 °C/13 °C to 23 °C/18 °C (day/night) reduced the number of harvested cuttings; however, they did not affect the percentage of roots proliferated from cuttings. By contrast, increasing the temperature to 33 °C/28 °C resulted in an increased number of cutting per stock plant. | [46] |
| Hypocotyl segments and cotyledonary leaves | MS media supplemented with different concentrations of NAA and TDZ, 0.8 g L−1 PVP, 0.1 g L−1 biotin, 0.1 g L−1 calcium pantothenate, 30 g L−1 sucrose | 0.44 µM BAP increased the regeneration of adventitious buds. | [47] |
| Zygotic embryos and cotyledons | MS media supplemented with 3 g L−1 sucrose and different concentrations of NAA, 2,4-D, BA, ABA | 1 mg L−1 NAA resulted in maximum callus induction, the frequency of callus proliferation depends on the age of the explant, with 10-year-old explants showing maximum proliferation, the highest frequency of somatic embryogenesis was observed in callus from mature zygotic embryos, low ABA concentrations increased number of somatic embryos. | [48] |
| Nodal segments | 1/2 MS supplemented with different concentrations of BAP, NAA, and GA3 | 0.050 mg L−1 BAP achieved optimal bud proliferation + 0.50 mg L−1 NAA, while ½ MS media supplemented with 0.2−1 and 0.10 mg L−1 GA3 + 0.10 mg L−1 BAP showed highest shoot elongation. | [49] |
| Nodal segments | MS media without PGRs | Media free from GA3 + BAP resulted in best shoot elongation, and WPM + 0.05 mg L−1 NAA + 0.5 mg L−1 BAP resulted in maximum axillary bud proliferation. | [50] |
| Nodal segments | ½ MS media, De-Fossard Medium, 0.9 µg L−1 BA, 0.5 µM NAA | The best multiplication rate (2.25) was achieved, and 93% of the plants survived acclimatization. | [51] |
| Nodal segments | MS media supplemented with 0.05 μM NAA, 0.4 μM BA, 1 mg L−1 nicotinic acid, 1 mg L−1 pyridoxine-HCl, 1 mg L−1 thiamine, 2 mg L−1 glycine, 50 mg L−1 myo-inositol, and 30 g L−1 sucrose | Endogenous rhythms cause time-related fluctuations, resulting in rooting variations among closely related genotypes. | [52] |
| Epicotyl segments | ½ MS supplemented with 1/6× CaCl2, 2% (w/v) sucrose | Auxin reduced mean rooting time, and light conditions did not affect the rooting efficiency; with increased age, decreased rooting capability was observed. | [53] |
| Axillary buds | ½ MS supplemented with 1 g L−1 ABA | Encapsulation by calcium alginate and storing under low light intensities resulted in the preservation of cultures for up to 3 months without affecting their viability. | [54] |
| Apical shoots | MS media supplemented with 0.04 mg L−1 BA, 1% sucrose, with/without charcoal | 38–85% survival was observed with plants exposed to PSV2 for 30 min in liquid nitrogen. | [55] |
| Nodal segments | MS media supplemented with 30 g L−1 sucrose | The best in vitro establishment, multiplication, shooting, and rooting were achieved using red–blue LEDs and fluorescent lamps. | [56] |
| Species | Explant | Sterilant | Media; PGR (If Any) | Area Studied and Scope of Work | References |
|---|---|---|---|---|---|
| E. camaldulensis × E. tereticornis and E. torelliana × E. citriodora | Nodal segments from mature trees (30–32 yrs) | 0.15% HgCl2 | MS media, WPM, SH medium, B5 medium; BAP, NAA | Hybridization of Eucalyptus species. The study reported that two hybrids developed that showed superior performance than parental genotypes. | [17] |
| E. grandis × E. nitens | Axial buds | 10 g L−1 CaOCl | MS media; BAP, IAA, metatopolin, kinetin | Individual evaluation of each stage of micropropagation. The study reported that Auxins are principal components of media, and expression of different auxin transporters might be used as markers to identify Eucalyptus spp. amenable for micropropagation. | [29] |
| E. erythronema × E. stricklandii | Seedlings germinated in vitro | 3% NaOCl | MS media supplemented with sucrose 30 g L−1; IBA, NAA, Gibberellic acids | First micropropagation report of ornamental Eucalyptus spp. The study reported that successful micropropagation from juvenile seedlings was achieved. | [44] |
| E. benthamii × E. dunni | Nodal segments from 1-year-old plants | NaOCl | ½ strength MS media; PVP40, NAA, BAP | Optimization of chlorine concentration for explant sterilization and optimum ratio of PGRs for shoot elongation. The study reported that 0.5% NaOCl is suggested for nodal segments; 0.50 mg L−1 BAP + 0.05 mg L−1 NAA provides the highest number of bud proliferation. | [49] |
| E. erythronema × E. stricklandii | Nodal segments | 1% NaOCl | MS media supplemented with sucrose 30 g L−1; 0.05 μM NAA and 2.22 μM BAP | Effect of different light intensities on micropropagation efficiency. The study reported that red–blue LEDs and fluorescent light result in higher vigor, high photosynthesis, and increased shoot and root proliferation. | [56] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, V.; Ankita; Karnwal, A.; Sharma, S.; Kamal, B.; Jadon, V.S.; Gupta, S.; Sivanasen, I. A Comprehensive Review Uncovering the Challenges and Advancements in the In Vitro Propagation of Eucalyptus Plantations. Plants 2023, 12, 3018. https://doi.org/10.3390/plants12173018
Sharma V, Ankita, Karnwal A, Sharma S, Kamal B, Jadon VS, Gupta S, Sivanasen I. A Comprehensive Review Uncovering the Challenges and Advancements in the In Vitro Propagation of Eucalyptus Plantations. Plants. 2023; 12(17):3018. https://doi.org/10.3390/plants12173018
Chicago/Turabian StyleSharma, Vikas, Ankita, Arun Karnwal, Shivika Sharma, Barkha Kamal, Vikash S. Jadon, Sanjay Gupta, and Iyyakkannu Sivanasen. 2023. "A Comprehensive Review Uncovering the Challenges and Advancements in the In Vitro Propagation of Eucalyptus Plantations" Plants 12, no. 17: 3018. https://doi.org/10.3390/plants12173018
APA StyleSharma, V., Ankita, Karnwal, A., Sharma, S., Kamal, B., Jadon, V. S., Gupta, S., & Sivanasen, I. (2023). A Comprehensive Review Uncovering the Challenges and Advancements in the In Vitro Propagation of Eucalyptus Plantations. Plants, 12(17), 3018. https://doi.org/10.3390/plants12173018







