Effects of Condensed Tannins on Bacterial and Fungal Communities during Aerobic Exposure of Sainfoin Silage
Abstract
:1. Introduction
2. Results
2.1. Characteristics of Fermentation during Aerobic Exposure in Sainfoin Silage
2.2. Bacterial Community during Aerobic Exposure
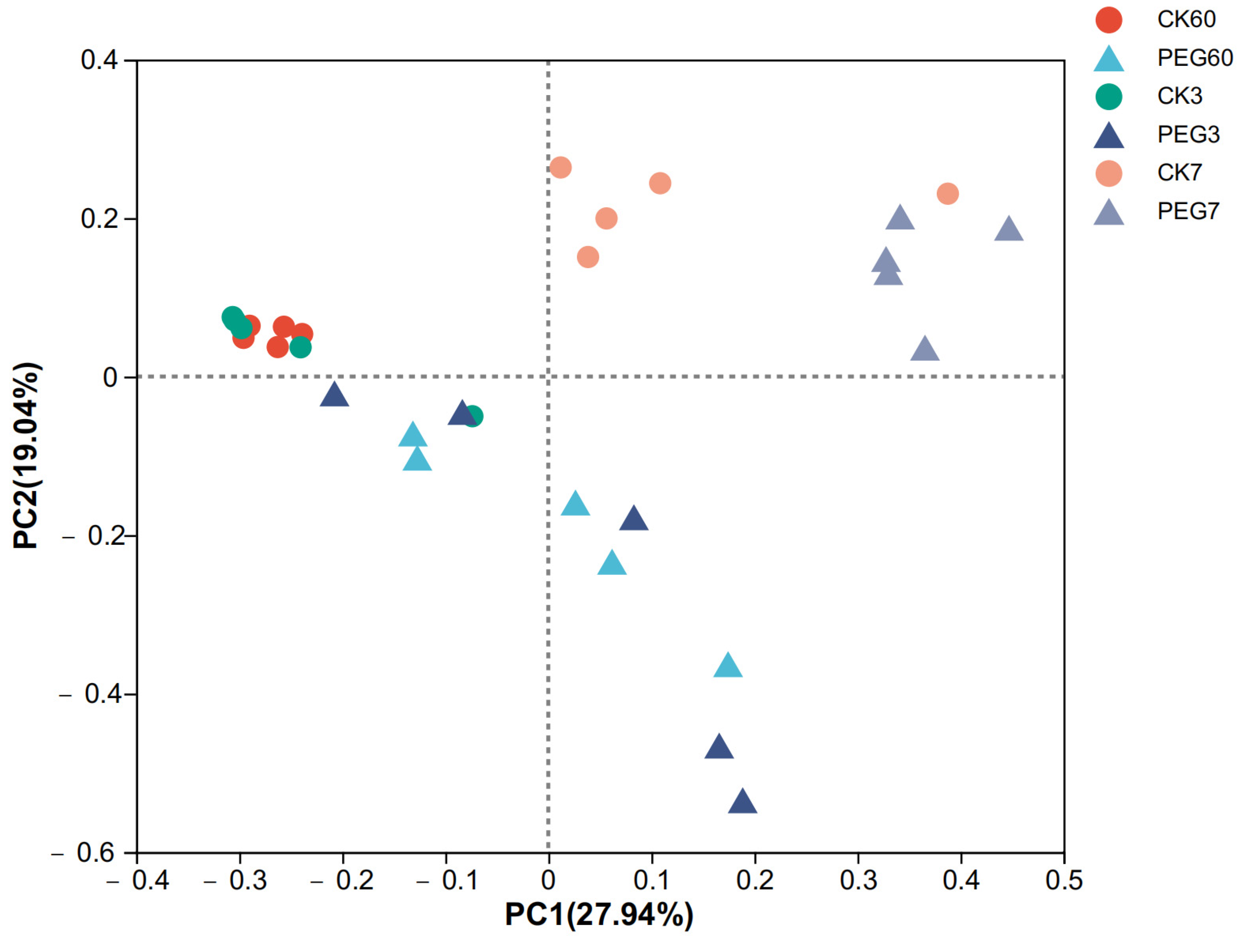
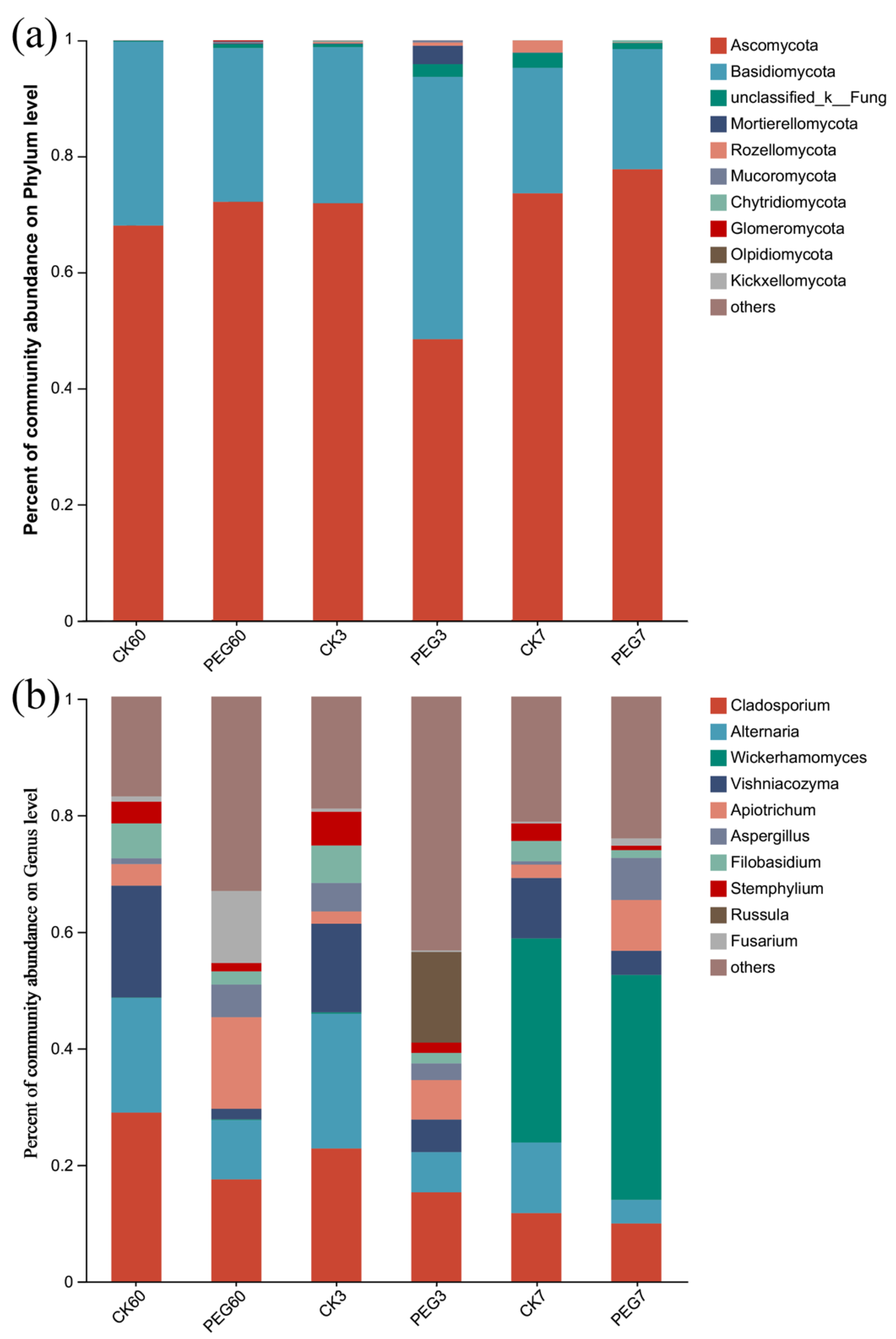
2.3. Fungal Community during Aerobic Exposure
3. Discussion
3.1. Aerobic Stability and Fermentation
3.2. Bacterial Community
3.3. Fungal Community
4. Materials and Methods
4.1. Silage Preparation
4.2. Aerobic Exposure of Silage
4.3. Characteristics Analysis of Silage
4.4. DNA Extraction and Sequence Analysis of Bacteria and Fungi
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Carbonero, C.H.; Mueller-Harvey, I.; Brown, T.A.; Smith, L. Sainfoin (Onobrychis viciifolia): A Beneficial Forage Legume. Plant Genet. Resour. -Charact. Util. 2011, 9, 70–85. [Google Scholar] [CrossRef]
- Mora-Ortiz, M.; Smith, L.M.J. Onobrychis viciifolia; A Comprehensive Literature Review of Its History, Etymology, Taxonomy, Genetics, Agronomy and Botany. Plant Genet. Resour. 2018, 16, 403–418. [Google Scholar] [CrossRef]
- Zeller, W.E. Activity, Purification, and Analysis of Condensed Tannins: Current State of Affairs and Future Endeavors. Crop Sci. 2019, 59, 886–904. [Google Scholar] [CrossRef]
- McAllister, T.A.; Martinez, T.; Bae, H.; Muir, A.; Yanke, L.; Jones, G. Characterization of Condensed Tannins Purified from Legume Forages: Chromophore Production, Protein Precipitation, and Inhibitory Effects on Cellulose Digestion. J. Chem. Ecol. 2005, 31, 2049–2068. [Google Scholar] [CrossRef] [PubMed]
- Jayanegara, A.; Sujarnoko, T.U.P.; Ridla, M.; Kondo, M.; Kreuzer, M. Silage Quality as Influenced by Concentration and Type of Tannins Present in the Material Ensiled: A Meta-Analysis. J. Anim. Physiol. Anim. Nutr. 2019, 103, 456–465. [Google Scholar] [CrossRef]
- Cai, Y.; Benno, Y. Influence of Lactobacillus spp. from an Inoculant and of Weissella and Leuconostoc spp. from Forage Crops on Silage Fermentation. Appled Environ. Microbiol. 1998, 64, 2982–2987. [Google Scholar] [CrossRef]
- Guo, X.S.; Xu, D.M.; Li, F.H.; Bai, J.; Su, R.A. Current Approaches on the Roles of Lactic Acid Bacteria in Crop Silage. Microb. Biotechnol. 2023, 16, 67–87. [Google Scholar] [CrossRef]
- McDonald, P.; Henderson, A.R.; Herson, S. The Biochemistry of Silage; Chalcombe Publications: Kingston, UK; Kent, UK, 1991. [Google Scholar]
- Peng, K.; Jin, L.; Niu, Y.D.; Huang, Q.; McAllister, T.A.; Yang, H.E.; Denise, H.; Xu, Z.; Acharya, S.; Wang, S.; et al. Condensed Tannins Affect Bacterial and Fungal Microbiomes and Mycotoxin Production during Ensiling and upon Aerobic Exposure. Appl. Environ. Microbiol. 2018, 84, e02274. [Google Scholar] [CrossRef]
- He, L.; Lv, H.; Xing, Y.; Chen, X.; Zhang, Q. Intrinsic Tannins Affect Ensiling Characteristics and Proteolysis of Neolamarckia cadamba Leaf Silage by Largely Altering Bacterial Community. Bioresour. Technol. 2020, 311, 123496. [Google Scholar] [CrossRef]
- Bueno, A.V.I.; Jobim, C.C.; Daniel, J.L.P.; Gierus, M. Fermentation Profile and Hygienic Quality of Rehydrated Corn Grains Treated with Condensed Tannins from Quebracho Plant Extract. Anim. Feed. Sci. Technol. 2020, 267, 114559. [Google Scholar] [CrossRef]
- Liu, X.L.; Hao, Y.Q.; Jin, L.; Xu, Z.J.; McAllister, T.A. Anti-Escherichia Coli O157:H7 Properties of Purple Prairie Clover and Sainfoin Condensed Tannins. Molecules 2013, 18, 2183–2199. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.Z.; Zhang, F.F.; Wang, T.; Zhang, Y.L.; Li, X.; Chen, Y.C.; Ma, C.H. Effect of Intrinsic Tannins on the Fermentation Quality and Associated with the Bacterial and Fungal Community of Sainfoin Silage. Microorganisms 2022, 10, 844. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.Z.; Wang, X.Z.; Ma, C.H.; Zhang, F.F. Effects of Intrinsic Tannins on Proteolysis Dynamics, Protease Activity, and Metabolome during Sainfoin Ensiling. Front. Microbiol. 2022, 13, 976118. [Google Scholar] [CrossRef]
- Wilkinson, J.M.; Davies, D.R. The Aerobic Stability of Silage: Key Findings and Recent Developments. Grass Forage Sci. 2013, 68, 1–19. [Google Scholar] [CrossRef]
- Niu, D.Z.; Zheng, M.L.; Zuo, S.S.; Jiang, D.; Xu, C.C. Effects of Maize Meal and Limestone on the Fermentation Profile and Aerobic Stability of Smooth Bromegrass (Bromus inermis Leyss) Silage. Grass Forage Sci. 2018, 73, 622–629. [Google Scholar] [CrossRef]
- Merry, R.J.; Davies, D.R. Propionibacteria and Their Role in the Biological Control of Aerobic Spoilage in Silage. Lait 1999, 79, 149–164. [Google Scholar] [CrossRef]
- Partanen, J.I. Calculation of Stoichiometric Dissociation Constants of Monoprotic Carboxylic Acids in Dilute Aqueous Sodium or Potassium Chloride Solutions and p[m(H+)] Values for Acetate and Formate Buffers at 25 °C. Talanta 2000, 52, 863–871. [Google Scholar] [CrossRef]
- Woolford, M.K. Microbiological Screening of the Straight Chain Fatty Acids (C1–C12) as Potential Silage Additives. J. Sci. Food Agric. 1975, 26, 219–228. [Google Scholar] [CrossRef]
- Weinberg, Z.G.; Ashbell, G.; Hen, Y.; Azrieli, A. The Effect of Applying Lactic Acid Bacteria at Ensiling on the Aerobic Stability of Silages. J. Appl. Bacteriol. 1993, 75, 512–518. [Google Scholar] [CrossRef]
- Keshri, J.; Chen, Y.R.; Pinto, R.; Kroupitski, Y.; Weinberg, Z.G.; Saldinger, S.S. Bacterial Dynamics of Wheat Silage. Front. Microbiol. 2019, 10, 1532. [Google Scholar] [CrossRef]
- Schmidt, R.J.; Kung, L. The Effects of Lactobacillus Buchneri with or without a Homolactic Bacterium on the Fermentation and Aerobic Stability of Corn Silages Made at Different Locations. J. Dairy Sci. 2010, 93, 1616–1624. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.S.; Wang, C.; Sun, L.; Xu, H.W.; Jiang, Y.; Na, N.; Yin, G.M.; Liu, S.B.; Xue, Y.L. Dynamics of Bacterial and Fungal Communities and Metabolites during Aerobic Exposure in Whole-Plant Corn Silages with Two Different Moisture Levels. Front. Microbiol. 2021, 12, 663895. [Google Scholar] [CrossRef] [PubMed]
- Danner, H.; Holzer, M.; Mayrhuber, E.; Braun, R. Acetic Acid Increases Stability of Silage under Aerobic Conditions. Appl. Environ. Microbiol. 2003, 69, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Li, X.X.; Xu, W.B.; Yang, J.S.; Zhao, H.B.; Xin, H.S.; Zhang, Y.G. Effect of Different Levels of Corn Steep Liquor Addition on Fermentation Characteristics and Aerobic Stability of Fresh Rice Straw Silage. Anim. Nutr. 2016, 2, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yu, Z.; Na, R.S. Effects of Different Additives on Fermentation Quality and Aerobic Stability of Leymus chinensis Silage. Grass Forage Sci. 2018, 73, 413–419. [Google Scholar] [CrossRef]
- Jones, W.T.; Mangan, J.L. Complexes of the Condensed Tannins of Sainfoin (Onobrychis viciifolia Scop.) with Fraction 1 Leaf Protein and with Submaxillary Mucoprotein, and Their Reversal by Polyethylene Glycol and PH. J. Sci. Food Agric. 1977, 28, 126–136. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, X.K.; Gu, Q.C.; Liang, M.Z.; Mu, S.L.; Zhou, B.; Huang, F.; Lin, B.; Zou, C.X. Analysis of the Correlation between Bacteria and Fungi in Sugarcane Tops Silage Prior to and after Aerobic Exposure. Bioresour. Technol. 2019, 291, 121835. [Google Scholar] [CrossRef]
- Muck, R.E. Silage Microbiology and Its Control through Additives. Rev. Bras. Zootec. 2010, 39, 183–191. [Google Scholar] [CrossRef]
- Keshri, J.; Chen, Y.R.; Pinto, R.; Kroupitski, Y.; Weinberg, Z.G.; Sela, S. Microbiome Dynamics during Ensiling of Corn with and without Lactobacillus plantarum Inoculant. Appl. Microbiol. Biotechnol. 2018, 102, 4025–4037. [Google Scholar] [CrossRef]
- Wang, M.S.; Chen, M.Y.; Bai, J.; Zhang, J.Y.; Su, R.N.; Franco, M.; Ding, Z.T.; Zhang, X.; Zhang, Y.; Guo, X.S. Ensiling Characteristics, in Vitro Rumen Fermentation Profile, Methane Emission and Archaeal and Protozoal Community of Silage Prepared with Alfalfa, Sainfoin and Their Mixture. Anim. Feed. Sci. Technol. 2021, 284, 115154. [Google Scholar] [CrossRef]
- Xu, D.M.; Ding, Z.T.; Wang, M.S.; Bai, J.; Ke, W.C.; Zhang, Y.X.; Guo, X.S. Characterization of the Microbial Community, Metabolome and Biotransformation of Phenolic Compounds of Sainfoin (Onobrychis viciifolia) Silage Ensiled with or without Inoculation of Lactobacillus plantarum. Bioresour. Technol. 2020, 316, 123910. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.W.; Tan, Z.F.; Gu, L.B.; Ma, H.; Wang, Z.Y.; Wang, L.; Wu, G.F.; Qin, G.Y.; Wang, Y.P. Pang Variation of Microbial Community and Fermentation Quality in Corn Silage Treated with Lactic Acid Bacteria and Artemisia Argyi during Aerobic Exposure. Toxins 2022, 14, 349. [Google Scholar] [CrossRef] [PubMed]
- Daniel, H.M.; Moons, M.C.; Huret, S.; Vrancken, G.; De Vuyst, L. Wickerhamomyces Anomalus in the Sourdough Microbial Ecosystem. Antonie Van Leeuwenhoek 2011, 99, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Ravasio, D.; Carlin, S.; Boekhout, T.; Groenewald, M.; Vrhovsek, U.; Walther, A.; Wendland, J. Adding Flavor to Beverages with Non-Conventional Yeasts. Fermentation 2018, 4, 15. [Google Scholar] [CrossRef]
- James, S.A.; Bond, C.J.; Stanley, R.; Ravella, S.R.; Peter, G.; Dlauchy, D.; Roberts, I.N. Apiotrichum terrigenum Sp Nov., a Soil-Associated Yeast Found in Both the UK and Mainland Europe. Int. J. Syst. Evol. Microbiol. 2016, 66, 5046–5050. [Google Scholar] [CrossRef]
- Qian, X.J.; Zhou, X.H.; Chen, L.; Zhang, X.Y.; Xin, F.X.; Dong, W.L.; Zhang, W.M.; Oshsenreither, K.; Jiang, M. Bioconversion of Volatile Fatty Acids into Lipids by the Oleaginous Yeast Apiotrichum porosum DSM27194. Fuel 2021, 290, 119811. [Google Scholar] [CrossRef]
- Xiao, Y.Z.; Sun, L.; Wang, Z.J.; Wang, W.; Xin, X.P.; Xu, L.J.; Du, S. Fermentation Characteristics, Microbial Compositions, and Predicted Functional Profiles of Forage Oat Ensiled with Lactiplantibacillus plantarum or Lentilactobacillus buchneri. Fermentation 2022, 8, 707. [Google Scholar] [CrossRef]
- Ranjit, N.K.; Kung, L. The Effect of Lactobacillus buchneri, Lactobacillus plantarum, or a Chemical Preservative on the Fermentation and Aerobic Stability of Corn Silage. J. Dairy Sci. 2000, 83, 526–535. [Google Scholar] [CrossRef]
- McDonald, P.; Henderson, A.R. Determination of Water-Soluble Carbohydrates in Grass. J. Sci. Food Agric. 1964, 15, 395–398. [Google Scholar] [CrossRef]
- Weatherburn, M.W. Phenol-Hypochlorite Reaction for Determination of Ammonia. Anal. Chem. 1967, 39, 971–974. [Google Scholar] [CrossRef]
- Zahiroddini, H.; Baah, J.; Absalom, W.; McAllister, T.A. Effect of an Inoculant and Hydrolytic Enzymes on Fermentation and Nutritive Value of Whole Crop Barley Silage. Anim. Feed. Sci. Technol. 2004, 117, 317–330. [Google Scholar] [CrossRef]
- Su, R.N.; Ni, K.K.; Wang, T.W.; Yang, X.P.; Zhang, J.; Liu, Y.Y.; Shi, W.X.; Yan, L.; Jie, C.; Zhong, J. Effects of Ferulic Acid Esterase-Producing Lactobacillus fermentum and Cellulase Additives on the Fermentation Quality and Microbial Community of Alfalfa Silage. PeerJ 2019, 7, e7712. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME Allows Analysis of High-Throughput Community Sequencing Data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Madden, T.L.; Schaffer, A.A.; Zhang, J.H.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A New Generation of Protein Database Search Programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed]


| Item | Treatment | Days of Ensiling | Days of Aerobic Exposure | SEM | p Value | |||
|---|---|---|---|---|---|---|---|---|
| 60 | 3 | 7 | Day | Treatment | D × T | |||
| DM | CK | 24.42 | 24.81 B | 24.78 B | 0.8465 | 0.0146 | <0.01 | 0.367 |
| PEG | 25.42 | 26.40 A | 26.30 A | |||||
| CP g/kg DM | CK | 202.93 a | 193.77 Ab | 196.02 Ab | 1.407 | <0.01 | <0.01 | <0.01 |
| PEG | 202.11 a | 182.92 Bb | 183.72 Bb | |||||
| pH | CK | 4.55 Ab | 4.62 Aa | 4.66 Aa | 0.011 | <0.01 | <0.01 | <0.01 |
| PEG | 4.55 A | 4.54 B | 4.54 B | |||||
| WSC g/kg DM | CK | 24.33 Aa | 11.80 b | 10.53 b | 1.137 | <0.01 | <0.01 | <0.01 |
| PEG | 11.24 B | 7.43 | 5.26 | |||||
| AN g/kg DM | CK | 0.67 Ba | 0.71 Ba | 0.32 Ab | 0.0403 | <0.01 | <0.01 | <0.01 |
| PEG | 0.77 Ab | 0.94 Aa | 0.30 Ac | |||||
| LA g/kg DM | CK | 23.21 a | 3.59 b | 3.66 b | 1.567 | <0.01 | 0.132 | 0.08 |
| PEG | 23.67 a | 5.75 b | 3.19 b | |||||
| AA g/kg DM | CK | 16.49 a | 11.41 b | 13.34 b | 0.412 | <0.01 | 0.93 | 0.457 |
| PEG | 16.14 a | 12.33 b | 12.63 b | |||||
| PA g/kg DM | CK | ND | ND | ND | - | - | - | - |
| PEG | ND | ND | ND | |||||
| BA g/kg DM | CK | ND | ND | ND | 1.153 | 0.132 | <0.01 | 0.132 |
| PEG | 14.90 | 12.38 | 10.88 | |||||
| Yeast Log10 CFU/g FM | CK | 2.98 Bb | 3.91a | 4.32 Aa | 0.125 | 0.01 | 0.05 | <0.01 |
| PEG | 3.96 A | 4.16 | 4.08 B | |||||
| LAB Log10 CFU/g FM | CK | 6.48 Bb | 6.77 Bb | 6.95 Aa | 0.1202 | 0.787 | 0.292 | 0.242 |
| PEG | 7.15 Aa | 7.20 Aa | 6.63 Ab | |||||
| Mold Log10 CFU/g FM | CK | 2.73 Bb | 3.08 Aa | 3.16 Aa | 0.064 | <0.01 | 0.029 | <0.01 |
| PEG | 3.41 Aa | 2.68 Bb | 2.42 Bb | |||||
| AB Log10 CFU/g FM | CK | 6.09 b | 6.88 b | 7.19 a | 0.106 | 0.002 | 0.937 | 0.088 |
| PEG | 6.41 | 7.16 | 6.64 | |||||
| Aerobic stability (h) | CK PEG | 156.32 b 182.43 a | 3.74 | - | 0.029 | - | ||
| Days | Treatment | Ace | Chao | Shannon | Simpson | Sobs | Coverage |
|---|---|---|---|---|---|---|---|
| 60 d of ensiling | CK | 121.03 | 114.60 | 1.78 | 0.2967 | 100.00 | 0.9995 |
| PEG | 111.13 | 102.75 | 1.74 | 0.3224 | 84.60 | 0.9996 | |
| 3 d aerobic exposure | CK | 136.99 | 118.14 | 1.95 | 0.2403 | 90.00 | 0.9994 |
| PEG | 113.79 | 92.52 | 1.70 | 0.3290 | 74.40 | 0.9994 | |
| 7 d aerobic exposure | CK | 149.12 | 140.47 | 2.10 | 0.2958 | 130.40 | 0.9994 |
| PEG | 124.49 | 103.31 | 1.95 | 0.2416 | 82.40 | 0.9995 | |
| SEM | 9.73 | 9.57 | 0.075 | 0.016 | 9.83 | - | |
| p value | Treatment (T) | 0.72 | 0.78 | 0.37 | 0.59 | 0.63 | - |
| Day (D) | 0.36 | 0.23 | 0.36 | 0.54 | 0.21 | - | |
| T × D | 0.95 | 0.88 | 0.86 | 0.23 | 0.75 | - | |
| Days | Treatment | Ace | Chao | Shannon | Simpson | Sobs | Coverage |
|---|---|---|---|---|---|---|---|
| 60 d of ensiling | CK | 189.28 a | 159.31 a | 3.00 | 0.1080 | 146.6 a | 0.9995 |
| PEG | 150.36 | 146.29 a | 2.91 | 0.1067 | 134.8 a | 0.9996 | |
| 3 d aerobic exposure | CK | 124.47 | 119.00 | 2.56 | 0.2001 | 110.6 | 0.9994 |
| PEG | 105.64 | 104.33 | 3.03 | 0.1351 | 102.0 | 0.9994 | |
| 7 d aerobic exposure | CK | 91.63 b | 83.55 b | 2.91 | 0.1479 | 77.0 b | 0.9994 |
| PEG | 99.13 | 78.89 b | 2.68 | 0.1467 | 65.0 b | 0.9995 | |
| SEM | 9.28 | 7.937 | 0.098 | 0.02 | 7.62 | - | |
| p value | Treatment (T) | <0.01 | <0.01 | 0.75 | 0.53 | <0.01 | - |
| Day (D) | 0.274 | 0.396 | 0.82 | 0.62 | 0.37 | - | |
| T × D | 0.458 | 0.940 | 0.35 | 0.79 | 0.99 | - | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, R.; Ma, C.; Zhang, F.; Wang, X. Effects of Condensed Tannins on Bacterial and Fungal Communities during Aerobic Exposure of Sainfoin Silage. Plants 2023, 12, 2967. https://doi.org/10.3390/plants12162967
Huang R, Ma C, Zhang F, Wang X. Effects of Condensed Tannins on Bacterial and Fungal Communities during Aerobic Exposure of Sainfoin Silage. Plants. 2023; 12(16):2967. https://doi.org/10.3390/plants12162967
Chicago/Turabian StyleHuang, Rongzheng, Chunhui Ma, Fanfan Zhang, and Xuzhe Wang. 2023. "Effects of Condensed Tannins on Bacterial and Fungal Communities during Aerobic Exposure of Sainfoin Silage" Plants 12, no. 16: 2967. https://doi.org/10.3390/plants12162967
APA StyleHuang, R., Ma, C., Zhang, F., & Wang, X. (2023). Effects of Condensed Tannins on Bacterial and Fungal Communities during Aerobic Exposure of Sainfoin Silage. Plants, 12(16), 2967. https://doi.org/10.3390/plants12162967







