Echinacea purpurea (L.) Moench: Biological and Pharmacological Properties. A Review
Abstract
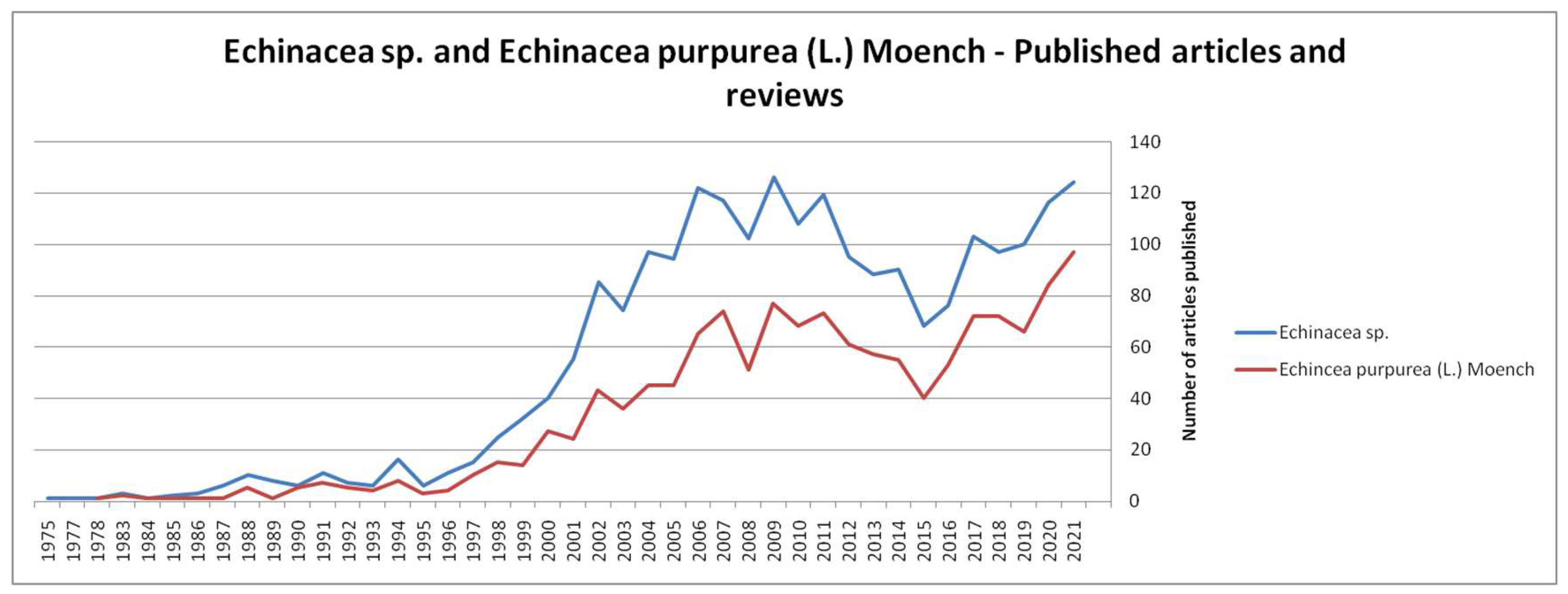
1. Introduction
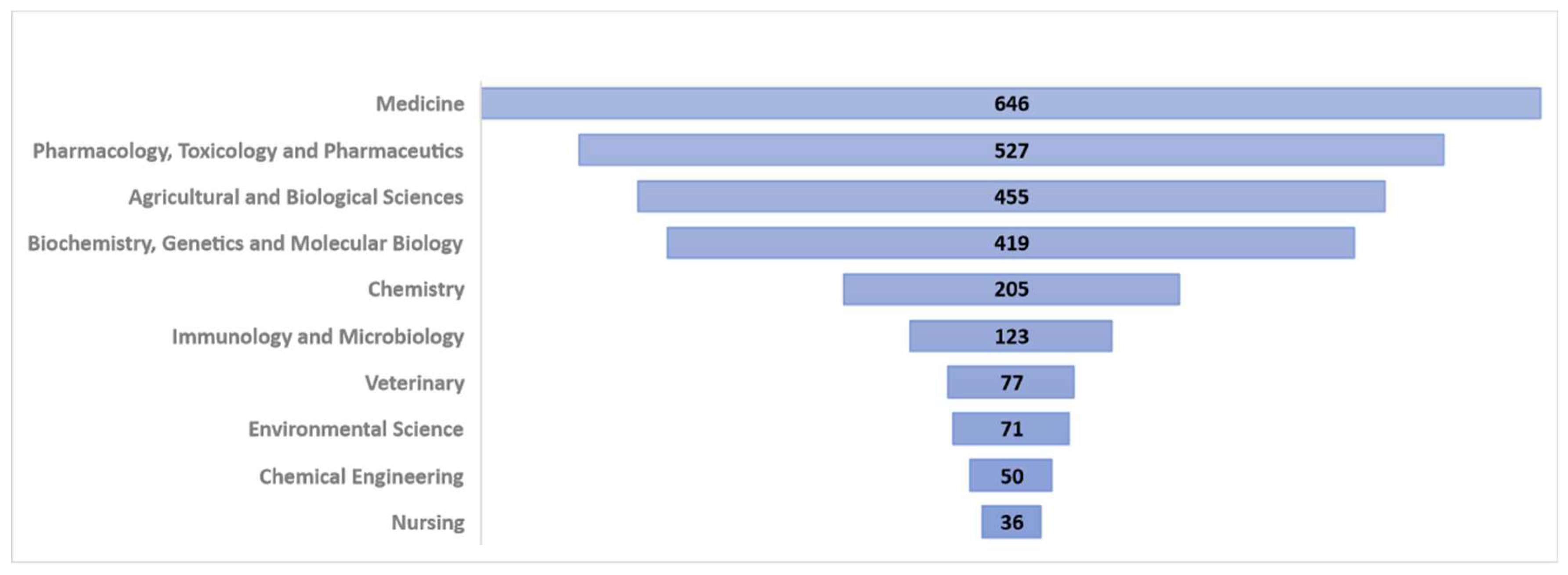
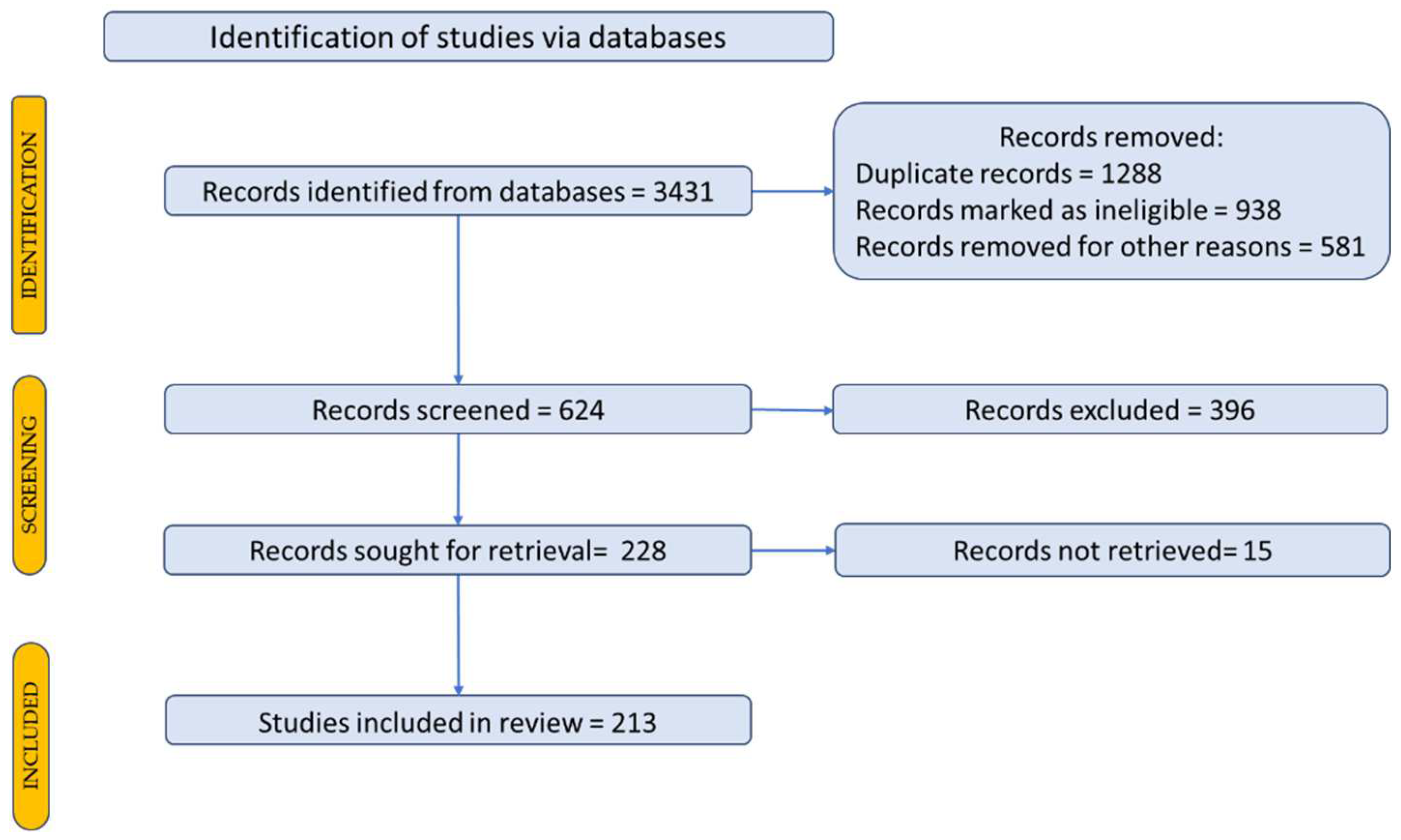
2. Methodology
2.1. Bioactive Compounds of Echinacea purpurea (L.) Moench
2.2. Biological and Pharmacological Effects of Echinacea purpurea (L.) Moench
2.2.1. Immunomodulating and Anti-Inflammatory Effect
2.2.2. Cannabinomimetic Properties
2.2.3. Antiviral Activity
2.2.4. Antimicrobial Effect
2.2.5. Antioxidant Activity
2.2.6. Antiosteoporotic Activity
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Salmeron-Manzano, E.; Garrido-Cardenas, J.A.; Manzano-Agugliaro, F. Worldwide Research Trends on Medicinal Plants. Int. J. Environ. Res. Public Health 2020, 17, 3376. [Google Scholar] [CrossRef] [PubMed]
- Pazyar, N.; Yaghoobi, R.; Rafiee, E.; Mehrabian, A.; Feily, A. Skin Wound Healing and Phytomedicine: A Review. Ski. Pharmacol. Physiol. 2014, 27, 303–310. [Google Scholar] [CrossRef]
- Hegedűs, C.; Muresan, M.; Badale, A.; Bombicz, M.; Varga, B.; Szilágyi, A.; Sinka, D.; Bácskay, I.; Popoviciu, M.; Magyar, I.; et al. SIRT1 Activation by Equisetum arvense L. (Horsetail) Modulates Insulin Sensitivity in Streptozotocin Induced Diabetic Rats. Molecules 2020, 25, 2541. [Google Scholar] [CrossRef] [PubMed]
- Stanisavljević, I.; Stojičević, S.; Veličković, D.; Veljković, V.; Lazic, M. Antioxidant and Antimicrobial Activities of Echinacea (Echinacea purpurea L.) Extracts Obtained by Classical and Ultrasound Extraction. Chin. J. Chem. Eng. 2009, 17, 478–483. [Google Scholar] [CrossRef]
- Kaya, M.; Merdivan, M.; Tashakkori, P.; Erdem, P.; Anderson, J.L. Analysis of Echinacea flower volatile constituents by HS-SPME-GC/MS using laboratory-prepared and commercial SPME fibers. J. Essent. Oil Res. 2018, 31, 91–98. [Google Scholar] [CrossRef]
- Lekar’, A.V.; Borisenko, S.N.; Filonova, O.V.; Vetrova, E.V.; Maksimenko, E.V.; Borisenko, N.I.; Minkin, V.I. Extraction of caftaric and cichoric acids from Echinacea purpurea L. in subcritical water. Russ. J. Phys. Chem. B 2013, 7, 968–975. [Google Scholar] [CrossRef]
- Nadaf, M.; Joharchi, M.R.; Amiri, M.S. Ethnomedicinal uses of plants for the treatment of nervous disorders at the herbal markets of Bojnord, North Khorasan Province, Iran. J. Phytomedicine 2018, 9, 153–163. [Google Scholar] [CrossRef]
- Cao, C.; Kindscher, K. The Medicinal Chemistry of Echinacea Species. Echinacea 2016, 127–145. [Google Scholar] [CrossRef]
- Scopus Database. Available online: https://www-scopus-com.am.e-nformation.ro/results/results.uri?sort=plf-f&src=s&st1=Echinacea&sid=1265d6dbcfd9127a51f79e0747ea36dd&sot=b&sdt=b&sl=24&s=TITLE-ABS-KEY%28Echinacea%29&origin=searchbasic&editSaveSearch=&yearFrom=Before+1960&yearTo=Present (accessed on 9 March 2022).
- Web of Science Database. Available online: https://www-webofscience-com.am.e-nformation.ro/wos/woscc/summary/cc619678-4982-4090-8c17-ca37d828cff1-3348de15/relevance/1 (accessed on 10 March 2022).
- Mistríková, I.; Vaverková, Š. Morphology and anatomy of Echinacea purpurea, E. angustifolia, E. pallida and Parthenium integrifolium. Biologia 2007, 62, 2–5. [Google Scholar] [CrossRef]
- Awang, D.V.C.; Kindack, D.G. Herbal medicine: Echinacea. Can. Pharm. J. 1991, 124, 512–516. [Google Scholar]
- McGregor, R. The taxonomy of the genus Echinacea (Compositae). University of Kansas. Sci. Bull. 1968, 48, 113–142. [Google Scholar]
- Keller, H.W. An anatomical study of the genus Echinacea. Univ. Kans. 1962, 3, 1–97. [Google Scholar]
- Upton, R.; Graff, A.; Jolliffe, G.; Länger, R.; Williamson, E. American Herbal Pharmacopoeia: Botanical Pharmacognosy—Microscopic Characterization of Botanical Medicines, 1st ed.; CRC Press: Boca Raton, FL, USA, 2011; 800p. [Google Scholar] [CrossRef]
- Kindscher, K. (Ed.) Echinacea: Herbal Medicine with a Wild History; Springer International Publishing: Cham, Switzerland, 2016; pp. 47–54. [Google Scholar]
- Belaeva, T.N.; Butenkova, A.N. Comparative analysis of the leaf anatomy of Echinacea purpurea and E. pallida. Biosyst. Divers. 2018, 26, 77–84. [Google Scholar] [CrossRef]
- Committee on Herbal Medicinal Products. Assessment Report on Echinacea purpurea (L.) Moench, Herba; European Medicines Agency: London, UK, 2008; Available online: https://www.ema.europa.eu/en/documents/herbal-report/superseded-assessment-report-echinacea-purpurea-l-moench-herba-recens_en.pdf (accessed on 10 March 2022).
- Karimmojeni, H.; Rezaei, M.; Tseng, T.-M.; Mastinu, A. Effects of Metribuzin Herbicide on Some Morpho-Physiological Characteristics of Two Echinacea Species. Horticulturae 2022, 8, 169. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Moher, D. Updating guidance for reporting systematic reviews: Development of the PRISMA 2020 statement. J. Clin. Epidemiol. 2021, 134, 103–112. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Phytochemistry of the Genus Echinacea, Echinacea; CRC Press: Boca Raton, FL, USA, 2004; pp. 71–88. [Google Scholar]
- Attarzadeh, M.; Balouchi, H.; Rajaie, M.; Dehnavi, M.M.; Salehi, A. Improving growth and phenolic compounds of Echinacea purpurea root by integrating biological and chemical resources of phosphorus under water deficit stress. Ind. Crop. Prod. 2020, 154, 112763. [Google Scholar] [CrossRef]
- Bruni, R.; Brighenti, V.; Caesar, L.K.; Bertelli, D.; Cech, N.B.; Pellati, F. Analytical methods for the study of bioactive compounds from medicinally used Echinacea species. J. Pharm. Biomed. Anal. 2018, 160, 443–477. [Google Scholar] [CrossRef] [PubMed]
- Pallag, A.; Bungau, S.; Tit, M.D.; Jurca, T.M.; Sirbu, V.; Honiges, A.; Horhogea, C. Comparative Study of Polyphenols, Flavonoids and Chlorophylls in Equisetum arvense L. Populations. Revista Chimie 2016, 67, 350–353. [Google Scholar]
- Banica, F.; Bungau, S.; Tit, D.M.; Behl, T.; Otrisal, P.; Nechifor, A.C.; Gitea, D.; Pavel, F.-M.; Nemeth, S. Determination of the Total Polyphenols Content and Antioxidant Activity of Echinacea Purpurea Extracts Using Newly Manufactured Glassy Carbon Electrodes Modified with Carbon Nanotubes. Processes 2020, 8, 833. [Google Scholar] [CrossRef]
- Bauer, R.; Wagner, H.; Hikano, H.; Farnsworth, N.R. Echinacea species as potential immunostimulatory drugs. Econ. Med. Plant Res. 1991, 5, 253–321. [Google Scholar]
- Abdelmohsen, M.M.; Nafiz, N.M.; Seif el Nasr, M.M. Microwave assisted extraction of bio-active compounds (phenolics and alkamides) from Echinacea purpurea. Int. J. Pharma Pharma Sci. 2014, 6, 265–268. [Google Scholar]
- Barnes, J.; Anderson, L.A.; Gibbons, S.; Phillipson, J.D. Echinacea species (Echinacea angustifolia (DC.) Hell., Echinacea pallida (Nutt.) Nutt., Echinacea purpurea (L.) Moench): A review of their chemistry, pharmacology and clinical properties. J. Pharm. Pharmacol. 2005, 57, 929–954. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, B.G.; Santos, L.F.F.; Pianetti, G.A.; César, I.C. A Rapid UPLC Method for the Simultaneous Quantitation of Caffeic Acid Derivatives in Dried Extracts of Echinacea Purpurea. J. Chromatogr. Sci. 2021, 59, 439–444. [Google Scholar] [CrossRef]
- Vendramin, V.; Viel, A.; Vincenzi, S. Caftaric Acid Isolation from Unripe Grape: A “Green” Alternative for Hydroxycinnamic Acids Recovery. Molecules 2021, 26, 1148. [Google Scholar] [CrossRef] [PubMed]
- Ramezannezhad, R.; Aghdasi, M.; Fatemi, M. Enhanced production of cichoric acid in cell suspension culture of Echinacea purpurea by silver nanoparticle elicitation. Plant Cell Tissue Organ Cult. (PCTOC) 2019, 139, 261–273. [Google Scholar] [CrossRef]
- Attarzadeh, M.; Balouchi, H.; Rajaie, M.; Dehnavi, M.M.; Salehi, A. Growth and nutrient content of Echinacea purpurea as affected by the combination of phosphorus with arbuscular mycorrhizal fungus and Pseudomonas florescent bacterium under different irrigation regimes. J. Environ. Manag. 2018, 231, 182–188. [Google Scholar] [CrossRef]
- Maggini, V.; De Leo, M.; Granchi, C.; Tuccinardi, T.; Mengoni, A.; Gallo, E.R.; Biffi, S.; Fani, R.; Pistelli, L.; Firenzuoli, F.; et al. The influence of Echinacea purpurea leaf microbiota on chicoric acid level. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Tabar, R.S.; Moieni, A.; Monfared, S.R. Improving biomass and chicoric acid content in hairy roots of Echinacea purpurea L. Biologia 2019, 74, 941–951. [Google Scholar] [CrossRef]
- Dalby-Brown, L.; Barsett, H.; Landbo, A.-K.R.; Meyer, A.A.S.; Mølgaard, P. Synergistic Antioxidative Effects of Alkamides, Caffeic Acid Derivatives, and Polysaccharide Fractions from Echinacea purpurea on in Vitro Oxidation of Human Low-Density Lipoproteins. J. Agric. Food Chem. 2005, 53, 9413–9423. [Google Scholar] [CrossRef] [PubMed]
- Kakimov, A.; Muratbayev, A.; Zharykbasova, K.; Amanzholov, S.; Mirasheva, G.; Kassymov, S.; Utegenova, A.; Jumazhanova, M.; Shariati, M.A. Heavy metals analysis, GCMS-QP quantification of flavonoids, amino acids and saponins, analysis of tannins and organoleptic properties of powder and tincture of Echinacea purpurea (L.) and Rhapónticum carthamoídes. Potravinarstvo Slovak J. Food Sci. 2021, 15, 330–339. [Google Scholar] [CrossRef]
- Nyalambisa, M.; Oyemitan, I.; Matewu, R.; Oyedeji, O.; Oluwafemi, O.; Songca, S.P.; Nkeh-Chungag, B.N. Volatile constituents and biological activities of the leaf and root of Echinacea species from South Africa. Saudi Pharm. J. 2016, 25, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Balciunaite, G.; Haimi, P.-J.; Mikniene, Z.; Savickas, G.; Ragazinskiene, O.; Juodziukyniene, N.; Baniulis, D.; Pangonyte, D. Identification of Echinacea Purpurea (L.) Moench Root LysM Lectin with Nephrotoxic Properties. Toxins 2020, 12, 88. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.M.; Ramaiah, S. Pharmacological importance of Echinacea purpurea. Int. J. Pharm. Biol. Sci. 2011, 2, 305–314. [Google Scholar]
- Liu, C.-Z.; Abbasi, B.H.; Gao, M.; Murch, A.S.J.; Saxena, P.K. Caffeic Acid Derivatives Production by Hairy Root Cultures of Echinacea purpurea. J. Agric. Food Chem. 2006, 54, 8456–8460. [Google Scholar] [CrossRef]
- Erkoyuncu, M.T.; Yorgancilar, M. Optimization of callus cultures at Echinacea purpurea L. for the amount of caffeic acid derivatives. Electron. J. Biotechnol. 2021, 51, 17–27. [Google Scholar] [CrossRef]
- Pellati, F.; Benvenuti, S.; Magro, L.; Melegari, M.; Soragni, F. Analysis of phenolic compounds and radical scavenging activity of Echinacea spp. J. Pharm. Biomed. Anal. 2004, 35, 289–301. [Google Scholar] [CrossRef]
- Thygesen, L.; Thulin, J.; Mortensen, A.; Skibsted, L.H.; Molgaard, P. Antioxidant activity of cichoric acid and alkamides from Echinacea purpurea, alone and in combination. Food Chem. 2007, 101, 74–81. [Google Scholar] [CrossRef]
- Jukić, H.; Habeš, S.; Aldžić, A.; Durgo, K.; Kosalec, I. Antioxidant and prooxidant activities of phenolic compounds of the extracts of Echinacea purpurea (L.). Bull. Chem. Technol. Bosnia Herzeg. 2015, 44, 43–52. [Google Scholar]
- Kim, H.-O.; Durance, T.D.; Scaman, C.H.; Kitts, D.D. Retention of Caffeic Acid Derivatives in Dried Echinacea purpurea. J. Agric. Food Chem. 2000, 48, 4182–4186. [Google Scholar] [CrossRef]
- Bergeron, C.; Livesey, J.F.; Awang, D.V.C.; Arnason, J.T.; Rana, J.; Baum, B.R.; Letchamo, W. A quantitative HPLC method for the quality assurance of Echinacea Products on the North American market. Phytochem. Anal. 2000, 11, 207–215. [Google Scholar] [CrossRef]
- Brown, P.N.; Chan, M.; Betz, J.M. Optimization and single-laboratory validation study of a high-performance liquid chromatography (HPLC) method for the determination of phenolic Echinacea constituents. Anal. Bioanal. Chem. 2010, 397, 1883–1892. [Google Scholar] [CrossRef] [PubMed]
- Chiou, S.-Y.; Sung, J.-M.; Huang, P.-W.; Lin, S.-D. Antioxidant, Antidiabetic, and Antihypertensive Properties of Echinacea purpurea Flower Extract and Caffeic Acid Derivatives Using In Vitro Models. J. Med. Food 2017, 20, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Geng, X.; Tian, X.; Tu, P.; Pu, X. Neuroprotective effects of echinacoside in the mouse MPTP model of Parkinson's disease. Eur. J. Pharmacol. 2007, 564, 66–74. [Google Scholar] [CrossRef]
- Bauer, R. [Echinacea drugs—Effects and active ingredients]. Z. fur arztliche Fortbild. 1996, 90. [Google Scholar]
- Hou, R.; Xu, T.; Li, Q.; Yang, F.; Wang, C.; Huang, T.; Hao, Z. Polysaccharide from Echinacea purpurea reduce the oxidant stress in vitro and in vivo. Int. J. Biol. Macromol. 2020, 149, 41–50. [Google Scholar] [CrossRef]
- Karg, C.A.; Wang, P.; Vollmar, A.M.; Moser, S. Re-opening the stage for Echinacea research—Characterization of phylloxanthobilins as a novel anti-oxidative compound class in Echinacea purpurea. Phytomedicine 2019, 60, 152969. [Google Scholar] [CrossRef]
- Karg, C.A.; Wang, P.; Kluibenschedl, F.; Müller, A.P.D.T.; Allmendinger, L.; Vollmar, A.M.; Moser, S. Phylloxanthobilins are Abundant Linear Tetrapyrroles from Chlorophyll Breakdown with Activities Against Cancer Cells. Eur. J. Org. Chem. 2020, 2020, 4499–4509. [Google Scholar] [CrossRef]
- Mazza, G.; Cottrell, T. Volatile Components of Roots, Stems, Leaves, and Flowers of Echinacea Species. J. Agric. Food Chem. 1999, 47, 3081–3085. [Google Scholar] [CrossRef]
- Luo, X.-B.; Chen, B.; Yao, S.-Z.; Zeng, J.-G. Simultaneous analysis of caffeic acid derivatives and alkamides in roots and extracts of Echinacea purpurea by high-performance liquid chromatography–photodiode array detection–electrospray mass spectrometry. J. Chromatogr. A 2002, 986, 73–81. [Google Scholar] [CrossRef]
- Thomsen, M.O.; Fretté, X.C.; Christensen, K.B.; Christensen, L.P.; Grevsen, K. Seasonal Variations in the Concentrations of Lipophilic Compounds and Phenolic Acids in the Roots of Echinacea purpurea and Echinacea pallida. J. Agric. Food Chem. 2012, 60, 12131–12141. [Google Scholar] [CrossRef] [PubMed]
- Coelho, J.; Barros, L.; Dias, M.I.; Finimundy, T.C.; Amaral, J.S.; Alves, M.J.; Calhelha, R.C.; Santos, P.F.; Ferreira, I.C. Echinacea purpurea (L.) Moench: Chemical Characterization and Bioactivity of Its Extracts and Fractions. Pharmaceuticals 2020, 13, 125. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.A. Echinacea. Elsevier 2005, 2, 116. [Google Scholar]
- Billah, M.; Hosen, B.; Khan, F.; Niaz, K. Echinacea Book: Nonvitamin and Nonmineral Nutritional Supplements, 1st ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 205–210. [Google Scholar] [CrossRef]
- Birt, D.F.; Widrlechner, M.P.; Lalone, C.; Wu, L.; Bae, J.; Solco, A.K.; Kraus, G.; Murphy, P.; Wurtele, E.; Leng, Q.; et al. Echinacea in infection. Am. J. Clin. Nutr. 2008, 87, 488S–492S. [Google Scholar] [CrossRef]
- Rahman, A.N.A.; Khalil, A.A.; Abdallah, H.; ElHady, M. The effects of the dietary supplementation of Echinacea purpurea extract and/or vitamin C on the intestinal histomorphology, phagocytic activity, and gene expression of the Nile tilapia. Fish Shellfish Immunol. 2018, 82, 312–318. [Google Scholar] [CrossRef]
- Barrett, B. Medicinal properties of Echinacea: A critical review. Phytomedicine 2003, 10, 66–86. [Google Scholar] [CrossRef]
- Senchina, D.S.; Martin, A.E.; Buss, J.E.; Kohut, M.L. Effects of Echinacea extracts on macrophage antiviral activities. Phytotherapy Res. 2009, 24, 810–816. [Google Scholar] [CrossRef]
- Soon, S.L.; Crawford, R.I. Recurrent erythema nodosum associated with echinacea herbal therapy. J. Am. Acad. Dermatol. 2001, 44, 298–299. [Google Scholar] [CrossRef]
- Cupp, M.J.; Davis, J. Toxicology and Clinical Pharmacology of Herbal Products; Humana Press: Totowa, NJ, SUA, 2000; pp. 85–93. [Google Scholar]
- Aucoin, M.; Cooley, K.; Saunders, P.R.; Carè, J.; Anheyer, D.; Medina, D.N.; Cardozo, V.; Remy, D.; Hannan, N.; Garber, A. The effect of Echinacea spp. on the prevention or treatment of COVID-19 and other respiratory tract infections in humans: A rapid review. Adv. Integr. Med. 2020, 7, 203–217. [Google Scholar] [CrossRef]
- Weishaupt, R.; Bächler, A.; Feldhaus, S.; Lang, G.; Klein, P.; Schoop, R. Safety and Dose-Dependent Effects of Echinacea for the Treatment of Acute Cold Episodes in Children: A Multicenter, Randomized, Open-Label Clinical Trial. Children 2020, 7, 292. [Google Scholar] [CrossRef]
- Elek, F.; Eszter, D.; Rebeka, K.; Szende, V.; Melinda, U.; Eszter, L.-Z. Mapping of Echinacea-based food supplements on the Romania market and qualitative evaluation of the most commonly used products. Bull. Med Sci. 2020, 93, 111–123. [Google Scholar] [CrossRef]
- Chen, X.-L.; Zhang, J.-J.; Chen, R.; Li, Q.-L.; Yang, Y.-S.; Wu, H. An Uncommon Plant Growth Regulator, Diethyl Aminoethyl Hexanoate, Is Highly Effective in Tissue Cultures of the Important Medicinal Plant Purple Coneflower (Echinacea purpurea L.). BioMed Res. Int. 2013, 2013, 1–12. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stuart, D.L.; Wills, R.B.H. Alkylamide and Cichoric Acid Levels inEchinacea purpureaTissues During Plant Growth. J. Herbs Spices Med. Plants 2000, 7, 91–101. [Google Scholar] [CrossRef]
- Thomas, A. Regulatory Aspects of Herbal Medicine; Academic Press: Cambridge, MA, USA, 2017; pp. 165–178. [Google Scholar] [CrossRef]
- Waidyanatha, S.; Pierfelice, J.; Cristy, T.; Mutlu, E.; Burback, B.; Rider, C.V.; Ryan, K. A strategy for test article selection and phytochemical characterization of Echinacea purpurea extract for safety testing. Food Chem. Toxicol. 2020, 137, 111125. [Google Scholar] [CrossRef] [PubMed]
- Rios, M.Y.; Olivo, H.F. Natural and Synthetic Alkamides. Stud. Nat. Prod. Chem. 2014, 43, 79–121. [Google Scholar] [CrossRef]
- Woelkart, K.; Koidl, C.; Grisold, A.; Gangemi, J.D.; Turner, R.B.; Marth, E.; Bauer, R. Bioavailability and Pharmacokinetics of Alkamides From the Roots of Echinacea angustifolia in Humans. J. Clin. Pharmacol. 2005, 45, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Woelkart, K.; Dittrich, P.; Beubler, E.; Pinl, F.; Schoop, R.; Suter, A.; Bauer, R. Pharmacokinetics of the Main Alkamides after Administration of three Different Echinacea purpurea Preparations in Humans. Planta Medica 2008, 74, 651–656. [Google Scholar] [CrossRef]
- Saeidnia, S.; Manayi, A.; Vazirian, M. Echinacea purpurea: Pharmacology, phytochemistry and analysis methods. Pharmacogn. Rev. 2015, 9, 63–72. [Google Scholar] [CrossRef]
- Matthias, A.; Banbury, L.; Stevenson, L.M.; Bone, K.M.; Leach, D.N.; Lehmann, R.P. Alkylamides from Echinacea Modulate Induced Immune Responses in Macrophages. Immunol. Investig. 2007, 36, 117–130. [Google Scholar] [CrossRef]
- Mudge, E.; Lopes-Lutz, D.; Brown, P.; Schieber, A. Analysis of Alkylamides in Echinacea Plant Materials and Dietary Supplements by Ultrafast Liquid Chromatography with Diode Array and Mass Spectrometric Detection. J. Agric. Food Chem. 2011, 59, 8086–8094. [Google Scholar] [CrossRef]
- Cech, N.B.; Kandhi, V.; Davis, J.M.; Hamilton, A.; Eads, D.; Laster, S.M. Echinacea and its alkylamides: Effects on the influenza A-induced secretion of cytokines, chemokines, and PGE2 from RAW 264.7 macrophage-like cells. Int. Immunopharmacol. 2010, 10, 1268–1278. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, A.; Friedberg, K. Untersuchungen zur Wirkung von Echinacea angustifolia auf die unspezifische zelluläre Immunantwort der Maus [The effect of Echinacea angustifolia on non-specific cellular immunity in the mouse]. Arzneimmittel-Forschung 1991, 41, 141–147. [Google Scholar]
- Shariatinia, Z. Natural Polysaccharides in Drug Delivery and Biomedical Applications; Academic Press: London, UK, 2019; Chapter 2; pp. 15–57. [Google Scholar]
- Balciunaite, G.; Juodsnukyte, J.; Savickas, A.; Ragazinskiene, O.; Siatkute, L.; Zvirblyte, G.; Mistiniene, E.; Savickiene, N. Fractionation and evaluation of proteins in roots of Echinacea purpurea (L.) Moench. Acta Pharm. 2015, 65, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Chen, Y.; Zhong, S.; Ji, B.; Wang, J.; Bai, X.; Shi, G. Anti-Inflammatory Activity of N-Butanol Extract from Ipomoea stolonifera In Vivo and In Vitro. PLoS ONE 2014, 9, e95931. [Google Scholar] [CrossRef]
- Stimpel, M.; Proksch, A.; Wagner, H.; Lohmann-Matthes, M.L. Macrophage activation and induction of macrophage cytotoxicity by purified polysaccharide fractions from the plant Echinacea purpurea. Infect. Immun. 1984, 46, 845–849. [Google Scholar] [CrossRef]
- Sharma, S.; Anderson, M.; Schoop, S.; Hudson, J. Bactericidal and anti-inflammatory properties of a standardized Echinacea extract (Echinaforce®): Dual actions against respiratory bacteria. Phytomedicine 2010, 17, 563–568. [Google Scholar] [CrossRef]
- Vazirian, M.; Dianat, S.; Manayi, A.; Ziari, R.; Mousazadeh, A.; Habibi, E.; Saeidnia, S.; Amanzadeh, Y. Anti-inflammatory effect, total polysaccharide, total phenolics content and antioxidant activity of the aqueous extract of three basidiomycetes. Res. J. Pharmacogn. 2014, 1, 15–21. [Google Scholar]
- Mazzio, E.A.; Soliman, K.F.A. In vitro screening for the tumoricidal properties of international medicinal herbs. Phytotherapy Res. 2008, 23, 385–398. [Google Scholar] [CrossRef]
- Vickers, A. Botanical Medicines for the Treatment of Cancer: Rationale, Overview of Current Data, and Methodological Considerations for Phase I and II Trials. Cancer Investig. 2002, 20, 1069–1079. [Google Scholar] [CrossRef]
- Voaden, D.J.; Jacobson, M. Tumor inhibitors. 3. Identification and synthesis of a oncolytic hydrocarbon from American coneflower roots. J. Med. Chem. 1972, 15, 619–623. [Google Scholar] [CrossRef]
- Yang, G.; Li, K.; Liu, C.; Peng, P.; Bai, M.; Sun, J.; Li, Q.; Yang, Z.; Yang, Y.; Wu, H. A Comparison of the Immunostimulatory Effects of Polysaccharides from Tetraploid and Diploid Echinacea purpurea. BioMed Res. Int. 2018, 2018, 1–12. [Google Scholar] [CrossRef]
- Yao, L.; Bai, L.; Tan, Y.; Sun, J.; Qu, Q.; Shi, D.; Guo, S.; Liu, C. The immunoregulatory effect of sulfated Echinacea purpurea polysaccharide on chicken bone marrow-derived dendritic cells. Int. J. Biol. Macromol. 2019, 139, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.-L.; Chiu, C.-C.; Chen, J.Y.-F.; Chan, K.-C.; Lin, S.-D. Cytotoxic effects of Echinacea purpurea flower extracts and cichoric acid on human colon cancer cells through induction of apoptosis. J. Ethnopharmacol. 2012, 143, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Abreu, R.M.; Ferreira, I.; Calhelha, R.C.; Lima, R.T.; Vasconcelos, M.H.; Adega, F.; Chaves, R.; Queiroz, M.-J.R. Anti-hepatocellular carcinoma activity using human HepG2 cells and hepatotoxicity of 6-substituted methyl 3-aminothieno[3,2-b]pyridine-2-carboxylate derivatives: In vitro evaluation, cell cycle analysis and QSAR studies. Eur. J. Med. Chem. 2011, 46, 5800–5806. [Google Scholar] [CrossRef] [PubMed]
- Luettig, B.; Steinmüller, C.; Gifford, G.E.; Wagner, H.; Lohmann-Matthes, M.-L. Macrophage Activation by the Polysaccharide Arabinogalactan Isolated From Plant Cell Cultures of Echinacea purpurea. JNCI: J. Natl. Cancer Inst. 1989, 81, 669–675. [Google Scholar] [CrossRef]
- Sharif, K.O.M.; Tufekci, E.F.; Ustaoglu, B.; Altunoglu, Y.C.; Zengin, G.; Llorent-Martínez, E.; Guney, K.; Baloglu, M.C. Anticancer and biological properties of leaf and flower extracts of Echinacea purpurea (L.) Moench. Food Biosci. 2021, 41, 101005. [Google Scholar] [CrossRef]
- Jiang, W.; Zhu, H.; Xu, W.; Liu, C.; Hu, B.; Guo, Y.; Cheng, Y.; Qian, H. Echinacea purpurea polysaccharide prepared by fractional precipitation prevents alcoholic liver injury in mice by protecting the intestinal barrier and regulating liver-related pathways. Int. J. Biol. Macromol. 2021, 187, 143–156. [Google Scholar] [CrossRef]
- Xu, W.; Hu, B.; Cheng, Y.; Guo, Y.; Yao, W.; Qian, H. Echinacea purpurea suppresses the cell survival and metastasis of hepatocellular carcinoma through regulating the PI3K/Akt pathway. Int. J. Biochem. Cell Biol. 2021, 142, 106115. [Google Scholar] [CrossRef]
- Guiotto, P.; Woelkart, K.; Grabnar, I.; Voinovich, D.; Perissutti, B.; Invernizzi, S.; Granzotto, M.; Bauer, R. Pharmacokinetics and immunomodulatory effects of phytotherapeutic lozenges (bonbons) with Echinacea purpurea extract. Phytomedicine 2008, 15, 547–554. [Google Scholar] [CrossRef]
- Del-Río-Navarro, B.E.; Espinosa-Rosales, F.J.; Flenady, V.; Sienra-Monge, J.J. Immunostimulants for preventing respiratory tract infection in children. Cochrane Database Syst. Rev. 2006, CD004974. [Google Scholar] [CrossRef]
- Kim, H.-R.; Oh, S.-K.; Lim, W.; Lee, H.K.; Moon, B.-I.; Seoh, J.-Y. Immune Enhancing Effects of Echinacea purpurea Root Extract by Reducing Regulatory T Cell Number and Function. Nat. Prod. Commun. 2014, 9, 511–514. [Google Scholar] [CrossRef] [PubMed]
- Bodinet, C.; Beuscher, N. Antiviral and Immunological Activity of Glycoproteins from Echinacea purpurea Radix. Planta Medica 1991, 57, A33–A34. [Google Scholar] [CrossRef]
- Bergeron, C.; Gafner, S. Quantitative Analysis of the Polysaccharide and Glycoprotein Fractions in Echinacea purpurea. And Echinacea angustifolia. by HPLC-ELSD for Quality Control of Raw Material. Pharm. Biol. 2007, 45, 98–105. [Google Scholar] [CrossRef]
- Kurkin, V.A.; Akushskaya, A.S.; Avdeeva, E.V.; Velmyaikina, E.I.; Daeva, E.D.; Kadentsev, V.I. Flavonoids from Echinacea purpurea. Russ. J. Bioorganic Chem. 2011, 37, 905–906. [Google Scholar] [CrossRef]
- Agrawal, A.D. Pharmacological Activities of Flavonoids: A Review. Int. J. Pharm. Sci. Nanotechnol. 2011, 4, 1394–1398. [Google Scholar] [CrossRef]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef]
- Dogan, Z.; Ergul, B.; Sarikaya, M.; Filik, L.; Gonultaş, M.A.; Hucumenoglu, S.; Can, M. The protective effect of Echinacea spp. (Echinacea angustifolia and Echinacea purpurea) in a rat colitis model induced by acetic acid. Pak. J. Pharm Sci. 2014, 27, 1827–1835. [Google Scholar]
- Lee, T.T.; Huang, C.C.; Shieh, X.H.; Chen, C.L.; Chen, L.J.; Yu, B. Flavonoid, Phenol and Polysaccharide Contents of Echinacea Purpurea, L. and Its Immunostimulant Capacity In Vitro. Int. J. Environ. Sci. Dev. 2010, 1. [Google Scholar] [CrossRef]
- Speroni, E.; Govoni, P.; Guizzardi, S.; Renzulli, C.; Guerra, M. Anti-inflammatory and cicatrizing activity of Echinacea pallida Nutt. root extract. J. Ethnopharmacol. 2001, 79, 265–272. [Google Scholar] [CrossRef]
- Ekeuku, S.O.; Pang, K.-L.; Chin, K.-Y. Effects of Caffeic Acid and Its Derivatives on Bone: A Systematic Review. Drug Des. Devel Ther. 2021, 15, 259–275. [Google Scholar] [CrossRef]
- Zhang, P.; Tang, Y.; Li, N.-G.; Zhu, Y.; Duan, J.-A. Bioactivity and Chemical Synthesis of Caffeic Acid Phenethyl Ester and Its Derivatives. Molecules 2014, 19, 16458–16476. [Google Scholar] [CrossRef] [PubMed]
- Yeğin, M.E.; Bilkay, U.; Tiftikçioğlu, Y.; Uyanikgil, Y.; Çavuşoğlu, T.; Ercan, G.; Gürdal, M. Altering effects of caffeic acid phenethyl ester (CAPE) and ischemia/reperfusion injury: An experimental study in a rat TRAM flap model. Eur. J. Plast. Surg. 2020, 43, 527–534. [Google Scholar] [CrossRef]
- Wu, S.; Zhang, K.; Qin, H.; Niu, M.; Zhao, W.; Ye, M.; Zou, H.; Yang, Y. Caffeic acid phenethyl ester (CAPE) revisited: Covalent modulation of XPO1/CRM1 activities and implication for its mechanism of action. Chem. Biol. Drug Des. 2016, 89, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yang, J. A comparative study of caffeic acid and a novel caffeic acid conjugate SMND-309 on antioxidant properties in vitro. LWT 2012, 46, 239–244. [Google Scholar] [CrossRef]
- Silva, T.; Oliveira, C.; Borges, F. Caffeic acid derivatives, analogs and applications: A patent review (2009–2013). Expert Opin. Ther. Patents 2014, 24, 1257–1270. [Google Scholar] [CrossRef]
- Chicca, A.; Adinolfi, B.; Martinotti, E.; Fogli, S.; Breschi, M.; Pellati, F.; Benvenuti, S.; Nieri, P. Cytotoxic effects of Echinacea root hexanic extracts on human cancer cell lines. J. Ethnopharmacol. 2007, 110, 148–153. [Google Scholar] [CrossRef]
- Senica, M.; Mlinsek, G.; Veberic, R.; Mikulic-Petkovsek, M. Which Plant Part of Purple Coneflower (Echinacea purpurea (L.) Moench) Should be Used for Tea and Which for Tincture? J. Med. Food 2019, 22, 102–108. [Google Scholar] [CrossRef]
- Wu, C.-H.; Murthy, H.N.; Hahn, E.-J.; Paek, K.-Y. Enhanced production of caftaric acid, chlorogenic acid and cichoric acid in suspension cultures of Echinacea purpurea by the manipulation of incubation temperature and photoperiod. Biochem. Eng. J. 2007, 36, 301–303. [Google Scholar] [CrossRef]
- Chiellini, C.; Maida, I.; Maggini, V.; Bosi, E.; Mocali, S.; Emiliani, G.; Perrin, E.; Firenzuoli, F.; Mengoni, A.; Fani, R. Preliminary data on antibacterial activity of Echinacea purpurea-associated bacterial communities against Burkholderia cepacia complex strains, opportunistic pathogens of Cystic Fibrosis patients. Microbiol. Res. 2017, 196, 34–43. [Google Scholar] [CrossRef]
- Tsai, Y.-L.; Chiou, S.-Y.; Chan, K.-C.; Sung, J.-M.; Lin, S.-D. Caffeic acid derivatives, total phenols, antioxidant and antimutagenic activities of Echinacea purpurea flower extracts. LWT 2012, 46, 169–176. [Google Scholar] [CrossRef]
- Jiang, Z.; Wang, J.; Li, X.; Zhang, X. Echinacoside and Cistanche tubulosa (Schenk) R. wight ameliorate bisphenol A-induced testicular and sperm damage in rats through gonad axis regulated steroidogenic enzymes. J. Ethnopharmacol. 2016, 193, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Xing, X.X.; Liu, Z.J.; Han, B. Effects of Acteoside and Echinacoside on the Expression of the BMP2 in Rat Osteoblast. Prog. Vet. Med. 2011, 32, 45–48. [Google Scholar]
- Liu, J.; Yang, L.; Dong, Y.; Zhang, B.; Ma, X. Echinacoside, an Inestimable Natural Product in Treatment of Neurological and other Disorders. Molecules 2018, 23, 1213. [Google Scholar] [CrossRef] [PubMed]
- Jia, C.; Shi, H.; Jin, W.; Zhang, K.; Jiang, Y.; Zhao, M.; Tu, P. Metabolism of Echinacoside, a Good Antioxidant, in Rats: Isolation and Identification of Its Biliary Metabolites. Drug Metab. Dispos. 2008, 37, 431–438. [Google Scholar] [CrossRef]
- Pires, C.; Martins, N.; Carvalho, A.M.; Barros, L.; Ferreira, I.C. Phytopharmacologic preparations as predictors of plant bioactivity: A particular approach to Echinacea purpurea (L.) Moench antioxidant properties. Nutrition 2016, 32, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Dong, L.; Wang, H.; Niu, J.; Zou, M.; Wu, N.; Yu, D.; Wang, Y. Echinacoside induces apoptotic cancer cell death by inhibiting the nucleotide pool sanitizing enzyme MTH1. OncoTargets Ther. 2015, 8, 3649–3664. [Google Scholar] [CrossRef]
- World Health Organization. WHO Monographs on Selected Medicinal Plants; World Health Organization: Geneva, Switzerland, 1999; pp. 136–145. [Google Scholar]
- Foster, S. Echinacea: Nature's Immune Enhancer; Healing Arts Press: Rochester, VT, USA, 1991; pp. 13–43. [Google Scholar]
- Khalaf, A.A.; Hussein, S.; Tohamy, A.F.; Marouf, S.; Yassa, H.D.; Zaki, A.R.; Bishayee, A. Protective effect of Echinacea purpurea (Immulant) against cisplatin-induced immunotoxicity in rats. DARU J. Pharm. Sci. 2019, 27, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Sousa, S.G.; Oliveira, L.A.; Magalhães, D.D.A.; de Brito, T.V.; Batista, J.A.; Pereira, C.M.C.; Costa, M.D.S.; Mazulo, J.C.R.; Filgueiras, M.D.C.; Vasconselos, D.F.P.; et al. Chemical structure and anti-inflammatory effect of polysaccharide extracted from Morinda citrifolia Linn (Noni). Carbohydr. Polym. 2018, 197, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, M.O.; Christensen, L.P.; Grevsen, K. Harvest Strategies for Optimization of the Content of Bioactive Alkamides and Caffeic Acid Derivatives in Aerial Parts and in Roots of Echinacea purpurea. J. Agric. Food Chem. 2018, 66, 11630–11639. [Google Scholar] [CrossRef]
- Sultan, M.T.; Buttxs, M.S.; Qayyum, M.M.N.; Suleria, H.A.R. Immunity: Plants as Effective Mediators. Crit. Rev. Food Sci. Nutr. 2014, 54, 1298–1308. [Google Scholar] [CrossRef]
- Pillai, S.; Pillai, C.; Mitscher, L.A.; Cooper, R. Use of Quantitative Flow Cytometry to Measure Ex Vivo Immunostimulant Activity of Echinacea: The Case for Polysaccharides. J. Altern. Complement. Med. 2007, 13, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vetvickova, J. Natural immunomodulators and their stimulation of immune reaction: True or false? Anticancer Res. 2014, 34. [Google Scholar]
- Aarland, R.C.; Bañuelos-Hernández, A.E.; Fragoso-Serrano, M.; Sierra-Palacios, E.D.C.; de León-Sánchez, F.D.; Pérez-Flores, L.J.; Rivera-Cabrera, F.; Mendoza-Espinoza, J.A. Studies on phytochemical, antioxidant, anti-inflammatory, hypoglycaemic and antiproliferative activities of Echinacea purpurea and Echinacea angustifolia extracts. Pharm. Biol. 2016, 55, 649–656. [Google Scholar] [CrossRef] [PubMed]
- McCann, D.A.; Solco, A.; Liu, Y.; Macaluso, F.; Murphy, P.A.; Kohut, M.L.; Senchina, D.S. Cytokine- and interferon-modulating properties of Echinacea spp. root tinctures stored at -20 degrees C for 2 years. J. Interferon Cytokine Res. 2007, 27, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Baron, S.E.; Cohen, S.N.; Archer, C.B.; on behalf of British Association of Dermatologists and Royal College of General Practitioners. Guidance on the diagnosis and clinical management of atopic eczema. Clin. Exp. Dermatol. 2012, 37, 7–12. [Google Scholar] [CrossRef]
- Kapur, S.; Watson, W.; Carr, S. Atopic dermatitis. Allergy Asthma Clin. Immunol. 2018, 14, 1–10. [Google Scholar] [CrossRef]
- Bíró, T.; Tóth, B.I.; Haskó, G.; Paus, R.; Pacher, P. The endocannabinoid system of the skin in health and disease: Novel perspectives and therapeutic opportunities. Trends Pharmacol. Sci. 2009, 30, 411–420. [Google Scholar] [CrossRef]
- Oláh, A.; Szabó-Papp, J.; Soeberdt, M.; Knie, U.; Dähnhardt-Pfeiffer, S.; Abels, C.; Bíró, T. Echinacea purpurea -derived alkylamides exhibit potent anti-inflammatory effects and alleviate clinical symptoms of atopic eczema. J. Dermatol. Sci. 2017, 88, 67–77. [Google Scholar] [CrossRef]
- Greaves, M.W. Pathogenesis and Treatment of Pruritus. Curr. Allergy Asthma Rep. 2010, 10, 236–242. [Google Scholar] [CrossRef]
- Kilic, A.; Harder, A.; Reich, H.; Knie, U.; Masur, C.; Abels, C. Efficacy of hydrophilic or lipophilic emulsions containing Echinacea purpurea extract in treatment of different types of pruritus. Clin. Cosmet. Investig. Dermatol. 2018, 11, 591–602. [Google Scholar] [CrossRef]
- Lee, Y.-S.; Wysocki, A.; Warburton, D.; Tuan, T.-L. Wound healing in development. Birth Defects Res. Part C: Embryo Today: Rev. 2012, 96, 213–222. [Google Scholar] [CrossRef]
- Rousseau, B.; Tateya, I.; Lim, X.; Munoz-Del-Rio, A.; Bless, D.M. Investigation of Anti-Hyaluronidase Treatment on Vocal Fold Wound Healing. J. Voice 2006, 20, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Zhao, X.; Zhao, C.; Pu, X.; Liu, H.; Li, H.; Fu, L.; Tang, Y. Hydrocarbon Charging and Accumulation in the Permian Reservoirs of theWumaying Buried Hill, Huanghua Depression, Bohai Bay Basin, China. Energies 2021, 14, 8109. [Google Scholar] [CrossRef]
- Glevitzky, I.; Dumitrel, G.A.; Glevitzky, M.; Pasca, B.; Otřísal, P.; Bungau, S.; Cioca, G.; Pantis, C.; Popa, M. Statistical Analysis of the Relationship Between Antioxidant Activity and the Structure of Flavonoid Compounds. Rev. Chim. 2019, 70, 3103–3107. [Google Scholar] [CrossRef]
- Meng, H.; Li, J.; Dong, Y.; He, Y.; Ren, H.; Liu, Y.; Qu, Z.; Zhang, W.; Zhang, L.; Bao, T.; et al. Poly traditional Chinese medicine formulation prepared with skin moisturizing properties. Dermatol. Ther. 2020, 33. [Google Scholar] [CrossRef] [PubMed]
- Assessment Report on Echinacea purpurea (L.) Moench, Herbarecens, European Medicines Agency 2015. Available online: https://www.ema.europa.eu/en/documents/herbal-report/final-assessment-report-echinacea-purpurea-l-moench-herba-recens_en.pdf (accessed on 15 March 2022).
- Raduner, S.; Majewska, A.; Chen, J.Z.; Xie, X.Q.; Hamon, J.; Faller, B.; Altmann, K.H.; Gertsch, J. Alkylamides from Echinacea are a new class of cannabinomimetics. Cannabinoid type 2 receptor-dependent and -independent immunomodulatory effects. J. Biol. Chem. 2006, 281, 14192–14206. [Google Scholar] [CrossRef]
- Woelkart, K.; Bauer, R. The Role of Alkamides as an Active Principle of Echinacea. Planta Medica 2007, 73, 615–623. [Google Scholar] [CrossRef]
- Chicca, A.; Raduner, S.; Pellati, F.; Strompen, T.; Altmann, K.-H.; Schoop, R.; Gertsch, J. Synergistic immunomopharmacological effects of N-alkylamides in Echinacea purpurea herbal extracts. Int. Immunopharmacol. 2009, 9, 850–858. [Google Scholar] [CrossRef]
- Woelkart, K.; Xu, W.; Pei, Y.; Makriyannis, A.; Picone, R.P.; Bauer, R. The Endocannabinoid System as a Target for Alkamides fromEchinacea angustifoliaRoots. Planta Medica 2005, 71, 701–705. [Google Scholar] [CrossRef]
- Kumar, A.; Premoli, M.; Aria, F.; Bonini, S.A.; Maccarinelli, G.; Gianoncelli, A.; Memo, M.; Mastinu, A. Cannabimimetic plants: Are they new cannabinoidergic modulators? Planta 2019, 249, 1681–1694. [Google Scholar] [CrossRef]
- Maccarrone, M.; Bab, I.; Bíró, T.; Cabral, G.A.; Dey, S.K.; Di Marzo, V.; Konje, J.C.; Kunos, G.; Mechoulam, R.; Pacher, P.; et al. Endocannabinoid signaling at the periphery: 50 years after THC. Trends Pharmacol. Sci. 2015, 36, 277–296. [Google Scholar] [CrossRef] [PubMed]
- Navarro, G.; Morales, P.; Cueto, C.R.; Fernández-Ruiz, J.; Jagerovic, N.; Franco, R. Targeting Cannabinoid CB2 Receptors in the Central Nervous System. Medicinal Chemistry Approaches with Focus on Neurodegenerative Disorders. Front. Neurosci. 2016, 10, 406. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Caram-Salas, N.L.; Li, W.; Wang, L.; Arnason, J.T.; Harris, C.S. Interactions of Echinacea spp. Root Extracts and Alkylamides With the Endocannabinoid System and Peripheral Inflammatory Pain. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Gertsch, J.; Schoop, R.; Kuenzle, U.; Suter, A. Echinacea alkylamides modulate TNF-α gene expression via cannabinoid receptor CB2 and multiple signal transduction pathways. FEBS Lett. 2004, 577, 563–569. [Google Scholar] [CrossRef]
- Matthias, A.; Blanchfield, J.T.; Penman, K.G.; Toth, I.; Lang, C.-S.; De Voss, J.J.; Lehmann, R.P. Permeability studies of alkylamides and caffeic acid conjugates from echinacea using a Caco-2 cell monolayer model. J. Clin. Pharm. Ther. 2004, 29, 7–13. [Google Scholar] [CrossRef]
- Haller, J.; Hohmann, J.; Freund, T.F. The effect of Echinacea preparations in three laboratory tests of anxiety: Comparison with chlordiazepoxide. Phytotherapy Res. 2010, 24, 1605–1613. [Google Scholar] [CrossRef]
- Pacher, P.; Bátkai, S.; Kunos, G. The Endocannabinoid System as an Emerging Target of Pharmacotherapy. Pharmacol. Rev. 2006, 58, 389–462. [Google Scholar] [CrossRef]
- Greger, H. Alkamides: Structural Relationships, Distribution and Biological Activity1. Planta Medica 1984, 50, 366–375. [Google Scholar] [CrossRef]
- Hohmann, J.; Rédei, D.; Forgo, P.; Szabó, P.; Freund, T.F.; Haller, J.; Bojnik, E.; Benyhe, S. Alkamides and a neolignan from Echinacea purpurea roots and the interaction of alkamides with G-protein-coupled cannabinoid receptors. Phytochemistry 2011, 72, 1848–1853. [Google Scholar] [CrossRef]
- Orhan, I.; Şenol, F.; Gülpinar, A.; Kartal, M.; Şekeroglu, N.; Deveci, M.; Kan, Y.; Şener, B. Acetylcholinesterase inhibitory and antioxidant properties of Cyclotrichium niveum, Thymus praecox subsp. caucasicus var. caucasicus, Echinacea purpurea and E. pallida. Food Chem. Toxicol. 2009, 47, 1304–1310. [Google Scholar] [CrossRef]
- Kurkin, V.A.; Dubishchev, A.V.; Ezhkov, V.N.; Titova, I.N.; Avdeeva, E.V. Antidepressant activity of some phytopharmaceuticals and phenylpropanoids. Pharm. Chem. J. 2006, 40, 614–619. [Google Scholar] [CrossRef]
- Thompson, K.D. Antiviral activity of Viracea® against acyclovir susceptible and acyclovir resistant strains of herpes simplex virus. Antivir. Res. 1998, 39, 55–61. [Google Scholar] [CrossRef]
- Binns, S.E.; Hudson, J.; Merali, S.; Arnason, J.T. Antiviral Activity of Characterized Extracts from Echinacea spp. (Heliantheae: Asteraceae) against Herpes simplex Virus (HSV-I). Planta Medica 2002, 68, 780–783. [Google Scholar] [CrossRef] [PubMed]
- McDougall, B.; King, P.J.; Wu, B.W.; Hostomsky, Z.; Reinecke, M.G.; Robinson, W.E. Dicaffeoylquinic and Dicaffeoyltartaric Acids Are Selective Inhibitors of Human Immunodeficiency Virus Type 1 Integrase. Antimicrob. Agents Chemother. 1998, 42, 140–146. [Google Scholar] [CrossRef]
- Robinson, W.E. l-Chicoric acid, an inhibitor of human immunodeficiency virus type 1 (HIV-1) integrase, improves on the in vitro anti-HIV-1 effect of Zidovudine plus a protease inhibitor (AG1350). Antivir. Res. 1998, 39, 101–111. [Google Scholar] [CrossRef]
- Wacker, A.; Hilbig, W. Virushemmung mit Echinacea purpurea. Planta Medica 1978, 33, 89–102. [Google Scholar] [CrossRef]
- Pleschka, S.; Stein, M.; Schoop, R.; Hudson, J.B. Anti-viral properties and mode of action of standardized Echinacea purpurea extract against highly pathogenic avian Influenza virus (H5N1, H7N7) and swine-origin H1N1 (S-OIV). Virol. J. 2009, 6, 197. [Google Scholar] [CrossRef]
- Fusco, D.; Liu, X.; Savage, C.; Taur, Y.; Xiao, W.; Kennelly, E.; Yuan, J.; Cassileth, B.; Salvatore, M.; Papanicolaou, G.A. Echinacea purpurea aerial extract alters course of influenza infection in mice. Vaccine 2010, 28, 3956–3962. [Google Scholar] [CrossRef]
- Vimalanathan, S.; Kang, L.; Amiguet, V.T.; Livesey, J.; Arnason, J.T.; Hudson, J. Echinacea purpurea. Aerial Parts Contain Multiple Antiviral Compounds. Pharm. Biol. 2005, 43, 740–745. [Google Scholar] [CrossRef]
- Behl, T.; Kaur, I.; Sehgal, A.; Singh, S.; Sharma, N.; Anwer, K.; Makeen, H.A.; Albratty, M.; Alhazmi, H.A.; Bhatia, S.; et al. There is nothing exempt from the peril of mutation — The Omicron spike. Biomed. Pharmacother. 2022, 148, 112756. [Google Scholar] [CrossRef]
- Behl, T.; Kaur, I.; Aleya, L.; Sehgal, A.; Singh, S.; Sharma, N.; Bhatia, S.; Al-Harrasi, A.; Bungau, S. CD147-spike protein interaction in COVID-19: Get the ball rolling with a novel receptor and therapeutic target. Sci. Total Environ. 2021, 808, 152072. [Google Scholar] [CrossRef] [PubMed]
- Aucoin, M.; Cardozo, V.; McLaren, M.D.; Garber, A.; Remy, D.; Baker, J.; Gratton, A.; Kala, M.A.; Monteiro, S.; Warder, C.; et al. A systematic review on the effects of Echinacea supplementation on cytokine levels: Is there a role in COVID-19? Metab. Open 2021, 11, 100115. [Google Scholar] [CrossRef] [PubMed]
- Percival, S.S. Use of echinacea in medicine. Biochem. Pharmacol. 2000, 60, 155–158. [Google Scholar] [CrossRef]
- Pascarella, G.; Strumia, A.; Piliego, C.; Bruno, F.; Del Buono, R.; Costa, F.; Scarlata, S.; Agrò, F.E. COVID-19 diagnosis and management: A comprehensive review. J. Intern. Med. 2020, 288, 192–206. [Google Scholar] [CrossRef]
- Coperchini, F.; Chiovato, L.; Croce, L.; Magri, F.; Rotondi, M. The cytokine storm in COVID-19: An overview of the involvement of the chemokine/chemokine-receptor system. Cytokine Growth Factor Rev. 2020, 53, 25–32. [Google Scholar] [CrossRef]
- Keller, A.C. Swiss Echinacea extract shown safe and effective in preventing colds in largest Echinacea clinical trial. Herb. Gram 2012, 96, 28–29. [Google Scholar]
- Jan, J.-T.; Cheng, T.-J.R.; Juang, Y.-P.; Ma, H.-H.; Wu, Y.-T.; Yang, W.-B.; Cheng, C.-W.; Chen, X.; Chou, T.-H.; Shie, J.-J.; et al. Identification of existing pharmaceuticals and herbal medicines as inhibitors of SARS-CoV-2 infection. Proc. Natl. Acad. Sci. USA 2021, 118. [Google Scholar] [CrossRef]
- Sharma, M.; Shawn, A.; Anderson, S.A.; Schoop, R.; Hudson, J.B. Induction of multiple pro-inflammatory cytokines by respiratory viruses and reversal by standardized Echinacea, a potent antiviral herbal extract. Antivir. Res. 2009, 83, 165–170. [Google Scholar] [CrossRef]
- Signer, J.; Jonsdottir, H.R.; Albrich, W.C.; Strasser, M.; Züst, R.; Ryter, S.; Ackermann-Gäumann, R.; Lenz, N.; Siegrist, D.; Suter, A. In vitro virucidal activity of Echinaforce®, an Echinacea purpurea preparation, against coronaviruses, including common cold coronavirus 229E and SARS-CoV-2. Virol J. 2020, 17, 136. [Google Scholar] [CrossRef]
- Nicolussi, S.; Ardjomand-Woelkart, K.; Stange, R.; Gancitano, G.; Klein, P.; Ogal, M. Echinacea as a Potential Force against Coronavirus Infections? A Mini-Review of Randomized Controlled Trials in Adults and Children. Microorganisms 2022, 10, 211. [Google Scholar] [CrossRef]
- Hudson, J.B. Applications of the Phytomedicine Echinacea purpurea (Purple Coneflower) in Infectious Diseases. J. Biomed. Biotechnol. 2011, 2012, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Vohra, S.; Arnason, J.T.; Hudson, J.B. Echinacea. Extracts Contain Significant and Selective Activities Against Human Pathogenic Bacteria*. Pharm. Biol. 2008, 46, 111–116. [Google Scholar] [CrossRef]
- Burns, J.; Zhao, L.; Taylor, E.W.; Spelman, K. The influence of traditional herbal formulas on cytokine activity. Toxicology 2010, 278, 140–159. [Google Scholar] [CrossRef] [PubMed]
- Maggini, V.; Miceli, E.; Fagorzi, C.; Maida, I.; Fondi, M.; Perrin, E.; Mengoni, A.; Bogani, P.; Chiellini, C.; Mocali, S.; et al. Antagonism and antibiotic resistance drive a species-specific plant microbiota differentiation in Echinacea spp. FEMS Microbiol. Ecol. 2018, 94. [Google Scholar] [CrossRef]
- Duțu, L.E.; Popescu, M.L.; Purdel, C.N.; Ilie, E.I.; Luță, E.-A.; Costea, L.; Gîrd, C.E. Traditional Medicinal Plants—A Possible Source of Antibacterial Activity on Respiratory Diseases Induced by Chlamydia pneumoniae, Haemophilus influenzae, Klebsiella pneumoniae and Moraxella catarrhalis. Diversity 2022, 14, 145. [Google Scholar] [CrossRef]
- Alves, M.; Ferreira, I.; Martins, A.; Pintado, M. Antimicrobial activity of wild mushroom extracts against clinical isolates resistant to different antibiotics. J. Appl. Microbiol. 2012, 113, 466–475. [Google Scholar] [CrossRef]
- Chen, X.-M.; Hu, C.; Raghubeer, E.; Kitts, D.D. Effect of High Pressure Pasteurization on Bacterial Load and Bioactivity of Echinacea Purpurea. J. Food Sci. 2010, 75, C613–C618. [Google Scholar] [CrossRef]
- Merali, S.; Binns, S.; Paulin-Levasseur, M.; Ficker, C.; Smith, M.; Baum, B.; Brovelli, E.; Arnason, J. Antifungal and Anti-inflammatory Activity of the Genus Echinacea. Pharm. Biol. 2003, 41, 412–420. [Google Scholar] [CrossRef]
- Choi, C.M.; Berson, D.S. Cosmeceuticals. Semin. Cutan. Med. Surg. 2006, 25, 163–168. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Mnayer, D.; Morais-Braga, M.F.B.; Carneiro, J.N.P.; Bezerra, C.F.; Coutinho, H.D.M.; Salehi, B.; Martorell, M.; Contreras, M.D.M.; Soltani-Nejad, A.; et al. Echinacea plants as antioxidant and antibacterial agents: From traditional medicine to biotechnological applications. Phytotherapy Res. 2018, 32, 1653–1663. [Google Scholar] [CrossRef]
- Sloley, B.D.; Urichuk, L.J.; Tywin, C.; Coutts, R.T.; Pang, P.K.T.; Shan, J.J. Comparison of chemical components and antioxidant capacity of different Echinacea species. J. Pharm. Pharmacol. 2001, 53, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Badale, A. Fenugreek seed and its active agent diosgenin treatment effects on different metabolic parameters in rats. Farmacia 2019, 67, 92–98. [Google Scholar] [CrossRef]
- Yotsawimonwat, S.; Rattanadechsakul, J.; Okonogi, S.; Rattanadechsakul, P. Skin improvement and stability of Echinacea purpurea dermatological formulations. Int. J. Cosmet. Sci. 2010, 32, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
- Oniszczuk, T.; Oniszczuk, A.; Gondek, E.; Guz, L.; Puk, K.; Kocira, A.; Kusz, A.; Kasprzak, K.; Wójtowicz, A. Active polyphenolic compounds, nutrient contents and antioxidant capacity of extruded fish feed containing purple coneflower (Echinacea purpurea (L.) Moench.). Saudi J. Biol. Sci. 2016, 26, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Soleimani, H.; Barzegar, M.; Sahari, M.; Naghdi Badi, H. An Investigation on the Antioxidant Activities of Hyssopus officinalis L. and Echinacea purpurea L. Plant Extracts in Oil Model System. J. Med. Plants. 2011, 10, 61–72. [Google Scholar]
- Murthy, H.N.; Kim, Y.-S.; Park, S.-Y.; Paek, K.-Y. Biotechnological production of caffeic acid derivatives from cell and organ cultures of Echinacea species. Appl. Microbiol. Biotechnol. 2014, 98, 7707–7717. [Google Scholar] [CrossRef]
- Mølgaard, P.; Johnsen, S.; Christensen, A.P.; Cornett§, C. HPLC Method Validated for the Simultaneous Analysis of Cichoric Acid and Alkamides in Echinacea purpurea Plants and Products. J. Agric. Food Chem. 2003, 51, 6922–6933. [Google Scholar] [CrossRef]
- Masteikova, R.; Muselik, J.; Bernatonienė, J.; Bernatonienė, R. Antioxidative activity of Ginkgo, Echinacea, and Ginseng tinctures. Medicina 2007, 43, 306–309. [Google Scholar] [CrossRef]
- Lepojević, I.; Lepojević, Z.; Pavlić, B.; Ristić, M.; Zeković, Z.; Vidović, S. Solid-liquid and high-pressure (liquid and supercritical carbon dioxide) extraction of Echinacea purpurea L. J. Supercrit. Fluids 2017, 119, 159–168. [Google Scholar] [CrossRef]
- Burnaz, N.A.; Küçük, M.; Akar, Z. An on-line HPLC system for detection of antioxidant compounds in some plant extracts by comparing three different methods. J. Chromatogr. B 2017, 1052, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Gadjalova, A.; Mihaylova, D. Ultrasound-assisted extraction of medicinal plants and evaluation of their biological activity. Food Res. 2019, 3, 530–536. [Google Scholar] [CrossRef]
- Fotakis, C.; Tsigrimani, D.; Tsiaka, T.; Lantzouraki, D.Z.; Strati, I.F.; Makris, C.; Tagkouli, D.; Proestos, C.; Sinanoglou, V.J.; Zoumpoulakis, P. Metabolic and antioxidant profiles of herbal infusions and decoctions. Food Chem. 2016, 211, 963–971. [Google Scholar] [CrossRef] [PubMed]
- Świderski, G.; Jabłońska-Trypuć, A.; Kalinowska, M.; Świsłocka, R.; Karpowicz, D.; Magnuszewska, M.; Lewandowski, W. Spectroscopic, Theoretical and Antioxidant Study of 3d-Transition Metals (Co(II), Ni(II), Cu(II), Zn(II) Complexes with Cichoric Acid. Materials 2020, 13, 3102. [Google Scholar] [CrossRef] [PubMed]
- Newair, E.F.; Abdel-Hamid, R.; Kilmartin, P.A. Electrochemical Determination of the Antioxidant Activity inEchinacea PurpureaRoots Using Square Wave Voltammetry. Electroanalysis 2017, 29, 1131–1140. [Google Scholar] [CrossRef]
- Perry, N.B.; Burgess, E.J.; Glennie, V.L. Echinacea Standardization: Analytical Methods for Phenolic Compounds and Typical Levels in Medicinal Species. J. Agric. Food Chem. 2001, 49, 1702–1706. [Google Scholar] [CrossRef] [PubMed]
- Che, C.-T.; Wong, M.S.; Lam, C.W.K. Natural Products from Chinese Medicines with Potential Benefits to Bone Health. Molecules 2016, 21, 239. [Google Scholar] [CrossRef]
- Ghitea, T.C.; El-Kharoubi, A.; Ganea, M.; Bimbo-Szuhai, E.; Nemeth, T.S.; Ciavoi, G.; Foghis, M.; Dobjanschi, L.; Pallag, A.; Micle, O. The Antimicrobial Activity of Origanum vulgare L. Correlated with the Gastrointestinal Perturbation in Patients with Metabolic Syndrome. Molecules 2021, 26, 283. [Google Scholar] [CrossRef]
- Li, F.; Yang, X.; Yang, Y.; Guo, C.; Zhang, C.; Yang, Z.; Li, P. Antiosteoporotic activity of echinacoside in ovariectomized rats. Phytomedicine 2013, 20, 549–557. [Google Scholar] [CrossRef]


| Storage Condition | Storage Temperature | Alchylamides Concentration | Chicoric Acid Concentration | Ref. |
|---|---|---|---|---|
| 60 days in the dark | 5 °C | unchanged | 70% decrease | [71,72] |
| 60 days in the light | 20 °C | 65% decrease | unchanged | [71,72] |
| Bioactive Compounds | Biological and Pharmacological Effects | References |
|---|---|---|
| Alkylamides | Anti-inflammatory | [74,75,76,77,78,79,80,81] |
| Immunomodulatory | ||
| Modulation of macrophages | ||
| Reduction of NO and tumor necrosis factor -α | ||
| Mediators of antiviral immunity | ||
| Cannabinoid receptor type 2 | ||
| Polysaccharides | Antitumoral | [82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98] |
| Antioxidant | ||
| Antimicrobial | ||
| Antifungal | ||
| Antiviral | ||
| Immunomodulatory | ||
| Hypoglycemic | ||
| Hepatoprotective | ||
| Gastrointestinal-protective | ||
| Antidiabetic | ||
| Glycoproteins | Immunomodulatory | [99,100,101,102,103] |
| Flavonoids | Antioxidant activity | [104,105,106,107,108,109] |
| Anti-inflammatory | ||
| Anti-ulcer activity | ||
| Antiallergic | ||
| Antiviral | ||
| Caffeic acid derivatives | Antioxidant activity | [110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126] |
| Antiosteoporotic activity | ||
| Anti-inflammatory | ||
| Antimicrobial | ||
| Anti-tumoral | ||
| Neuroprotective action |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burlou-Nagy, C.; Bănică, F.; Jurca, T.; Vicaș, L.G.; Marian, E.; Muresan, M.E.; Bácskay, I.; Kiss, R.; Fehér, P.; Pallag, A. Echinacea purpurea (L.) Moench: Biological and Pharmacological Properties. A Review. Plants 2022, 11, 1244. https://doi.org/10.3390/plants11091244
Burlou-Nagy C, Bănică F, Jurca T, Vicaș LG, Marian E, Muresan ME, Bácskay I, Kiss R, Fehér P, Pallag A. Echinacea purpurea (L.) Moench: Biological and Pharmacological Properties. A Review. Plants. 2022; 11(9):1244. https://doi.org/10.3390/plants11091244
Chicago/Turabian StyleBurlou-Nagy, Cristina, Florin Bănică, Tünde Jurca, Laura Grațiela Vicaș, Eleonora Marian, Mariana Eugenia Muresan, Ildikó Bácskay, Rita Kiss, Pálma Fehér, and Annamaria Pallag. 2022. "Echinacea purpurea (L.) Moench: Biological and Pharmacological Properties. A Review" Plants 11, no. 9: 1244. https://doi.org/10.3390/plants11091244
APA StyleBurlou-Nagy, C., Bănică, F., Jurca, T., Vicaș, L. G., Marian, E., Muresan, M. E., Bácskay, I., Kiss, R., Fehér, P., & Pallag, A. (2022). Echinacea purpurea (L.) Moench: Biological and Pharmacological Properties. A Review. Plants, 11(9), 1244. https://doi.org/10.3390/plants11091244













