Chemical Profile and Antimicrobial Activity of the Essential Oils of Helichrysum arenarium (L.) Moench. and Helichrysum italicum (Roth.) G. Don
Abstract
:1. Introduction
2. Results
2.1. Qualitative Composition of the Essential Oil (EO)
2.1.1. Helichrysum arenarium
2.1.2. Helichrysum italicum
2.1.3. Antimicrobial Activity of the H. italicum EO
3. Discussion
3.1. Helichrysum arenarium
3.2. Helichrysum italicum
3.3. Comparing the EOs between H. arenarium and H. italicum
3.4. Antimicrobial Activity of the H. italicum EO
4. Materials and Methods
4.1. Plant Material
4.2. Essential Oil (EO) Extraction
4.3. Gas Chromatography (GC) Flame Ionization Detection (FID) and Gas Chromatography–Mass Spectroscopy (MS) Analyses of the Essential Oils (EO)
4.4. Method for Testing Antimicrobial Activity
Microorganisms
4.5. Statistical Analyses of the Data
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Akaberi, M.; Sahebkar, A.; Azizi, M.; Emami, S.M. Everlasting flowers: Phytochemistry and pharmacology of the genus Helichrysum. Ind. Crops Prod. 2019, 138, 111471. [Google Scholar] [CrossRef]
- Talić, S.; Odak, I.; Lukic, T.; Brkljaca, M.; Bevanda, A.M.; Lasić, A. Chemodiversity of Helichrysum italicum (Roth) G. Don subsp. italicum essential oils from Bosnia and Herzegovina. Fresenius Environ. Bull. 2021, 30, 2492–2502. [Google Scholar]
- Kramberger, K.; Jenko Pražnikar, Z.; Baruca Arbeiter, A.; Petelin, A.; Bandelj, D.; Kenig, S. A Comparative Study of the Antioxidative Effects of Helichrysum italicum and Helichrysum arenarium Infusions. Antioxidants 2021, 10, 380. [Google Scholar] [CrossRef] [PubMed]
- Rančić, A.; Soković, M.; Vukojević, J.; Simić, A.; Marin, P.; Duletić-Laušević, S.; Djoković, D. Chemical composition and antimicrobial activities of essential oils of Myrrhis odorata (L.) Scop, Hypericum perforatum L and Helichrysum arenarium (L.) Moench. J. Essent. Oil Res. 2005, 17, 341–345. [Google Scholar] [CrossRef]
- Mao, Z.; Gan, C.; Zhu, J.; Ma, N.; Wu, L.; Wang, L.; Wang, X. Antiatherosclerotic activities of flavonoids from the flowers of Helichrysum arenarium L. MOENCH through the pathway of anti-inflammation. Bioorg. Med. Chem. Lett. 2017, 27, 2812–2817. [Google Scholar] [CrossRef]
- Les, F.; Venditti, A.; Cásedas, G.; Frezza, C.; Guiso, M.; Sciubba, F.; Serafini, M.; Bianco, A.; Valero, M.S.; López, V. Everlasting flower (Helichrysum stoechas Moench) as a potential source of bioactive molecules with antiproliferative, antioxidant, antidiabetic and neuroprotective properties. Ind. Crops Prod. 2017, 108, 295–302. [Google Scholar] [CrossRef]
- Clapham, A.R. Genus Helichrysum. In Flora Europaea; Tutin, T.G., Heywood, V.H., Burges, N.A., Moore, D.M., Valentine, D.H., Walters, S.M., Webb, D.A., Eds.; Cambridge University Press: London, UK; New York, NY, USA; Melbourne, Australia, 1976; Volume 4, pp. 128–131. [Google Scholar]
- Kuzmanov, B.; Gussev, C. Genus Helichrysum. In Flora Flora in Bulgaria; Anchev, M., Kozhuharov, S., Eds.; Acad. Publ. House Prof. Marin Drinov: Sofia, Bulgaria, 2013; Volume 11, p. 525. ISBN 978-954-322-522-4. [Google Scholar]
- Viegas, D.A.; Palmeira-de-Oliveira, A.; Salgueiro, L.; Martinez-de-Oliveira, J.; Palmeira-de-Oliveira, R. Helichrysum italicum: From traditional use to scientific data. J. Ethnopharmacol. 2014, 151, 54–65. [Google Scholar] [CrossRef]
- Aćimović, M.; Ljujić, J.; Vulić, J.; Zheljazkov, V.D.; Pezo, L.; Varga, A.; Tumbas Šaponjac, V. Helichrysum italicum (Roth) G. Don essential oil from Serbia: Chemical composition, classification and biological activity—May it be a suitable new crop for Serbia? Agronomy 2021, 11, 1282. [Google Scholar] [CrossRef]
- Morone-Fortunato, I.; Montemurro, C.; Ruta, C.; Perrini, R.; Sabetta, W.; Blanco, A.; Lorusso, E.; Avato, P. Essentialoils, genetic relationship sand in vitro establishment of Helichrysum italicum (Roth) G. Don ssp. italicum from wild Mediterrane an germplasm. Ind. Crops Prod. 2010, 32, 639–649. [Google Scholar] [CrossRef]
- Costa, P.; Miguel Loureiro, J.; Teixeira, M.A.; Rodrigues, A.E. Extraction of aromatic volatiles by hydrodistillation and super critical fluid extraction with CO2 from Helichrysum italicum subsp. picardii growingin Portugal. Ind. Crops Prod. 2015, 77, 680–683. [Google Scholar] [CrossRef]
- Ninčević, T.; Grdiša, M.; Šatović, Z.; Jug-Dujaković, M. Helichrysum italicum (Roth) G. Don: Taxonomy, biological activity, biochemical and genetic diversity. Ind. Crops Prod. 2019, 138, 111487. [Google Scholar] [CrossRef]
- Paolini, J.; Desjobert, J.M.; Costa, J.; Bernardini, A.F.; Castellini, C.B.; Cioni, P.L.; Guido Flamini, G.; Morelli, I. Composition of essential oils of Helichrysum italicum (Roth) G. Don fil subsp. italicum from Tuscan archipelago islands. Flavour Frag. J. 2006, 21, 805–808. [Google Scholar] [CrossRef]
- Leonardi, M.; Ambryszewska, K.E.; Melai, B.; Flamini, G.; Cioni, P.L.; Parri, F.; Pistelli, L. Essential oil composition of Helichrysum italicum (Roth) G.Don ssp. italicum from Elba Island (Tuscany, Italy). Chem. Biodivers. 2013, 10, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Mastelić, J.; Politeo, O.; Jerkovic, I.; Radosevic, N. Composition and antimicrobial activity of Helichrysum italicum essential oil and its terpene and terpenoid fractions. Chem. Nat. Compd. 2005, 41, 35–40. [Google Scholar] [CrossRef]
- Mastelić, J.; Politeo, O.; Jerković, I. Contribution to the analysis of the essential oil of Helichrysum italicum (Roth) G. Don.–Determination of ester bonded acids and phenols. Molecules 2008, 13, 795–803. [Google Scholar] [CrossRef] [Green Version]
- Gismondi, A.; Di Marco, G.; Antonella Canini, A. Helichrysum italicum (Roth) G. Don essential oil: Composition and potential antineoplastic effect. S. Afr. J. Bot. 2020, 133, 222–226. [Google Scholar] [CrossRef]
- Pljevljakušić, D.; Bigović, D.; Janković, T.; Jelačić, S.; Šavikin, K. Sandy everlasting (Helichrysum arenarium (L.) Moench): Botanical, chemical and biological properties. Front. Plant Sci. 2018, 9, 1123. [Google Scholar] [CrossRef] [Green Version]
- The State Pharmacopoeia of the USSR, 11th ed.; Part 2; USSR: Moscow, Russia, 1999.
- Pharmacopoeia Helvetica, 7th ed.; Département Fédéral de L’intérieur: Berne, Switzerland, 1987; Volume 1.
- Farmakopea Polska–Wydanie VI/Polish Pharmacopoeia, 6th ed.; Rzeczpospolita Polska Minister Zdrowa, Urzad Rejestracji Produktów Leczniczych: Warszawa, Poland, 2002; Wyrobów Medycznych i Produktów Biobójczych.
- WHO Monographs on Medicinal Plants Commonly Used in the Newly Independent States (NIS); World Health Organization: Geneva, Switzerland, 2015.
- Butorac, B. Helichrysum arenarium (L.) Moench subsp. arenarium. In Red Data Book of Flora of Serbia; 1. Extinct and critically endangered taxa; Stevanović, V., Ed.; Ministry of Environment of the Republic of Serbia: Belgrade, Serbia, 1999; pp. 294–296. [Google Scholar]
- Olsson, K.; Pihlik, U.; Radušiene, J.; Wedelsbäck, B.K. Helichrysum arenarium (L.) Moench (Everlasting) in Spice and Medicinal Plants in the Nordic and Baltic Countries Conservation of Genetic Resources; The SPIMED-Project Group at the Nordic Gene Bank: Alnarp, Sweden, 2005; pp. 55–65. [Google Scholar]
- Sawilska, A.K.; Jendrzejczak, E. Efficiency of Sandy Everlasting [Helichrysum arenarium (L.) Moench] cultivation from in vitro seedlings and achenes. Ind. Crops Prod. 2013, 43, 50–55. [Google Scholar] [CrossRef]
- Biological Diversity Act (Act on Amending and Supplementing). Decree No 354 accepted by the 40th National Assembly on 1st November 2007–State Gazette, No 94/16.11.2007, 2–44 2007. Available online: https://www.lex.bg/bg/laws/ldoc/2135456926 (accessed on 12 June 2021). (In Bulgarian).
- Medicinal Plant Act. Decree No 29 accepted by the National Assembly on 23 March 2000, State Gazette, No 29/07.04.2000, 9–21. Available online: https://lex.bg/laws/ldoc/2134916096 (accessed on 12 June 2021). (In Bulgarian).
- Smirnova, L.P.; Pervykh, L.N. Quantitative determination of the total content of flavonoids in the flowers of immortelle Helichrysum arenarium. Pharm. Chem. J. 1998, 32, 321–324. [Google Scholar] [CrossRef]
- Czinner, E.; Hagymási, K.; Blázovics, A.; Kéry, A.; Szoke, E.; Lemberkovics, A. In vitro antioxidant properties of Helichrysum arenarium (L.) Moench. J. Ethnopharmacol. 2000, 73, 437–443. [Google Scholar] [CrossRef]
- Czinner, E.; Lemberkovics, E.; Bihátsi-Karsai, E.; Vitányi, G.; Lelik, L. Composition of the essential oil from the Inflorescence of Helichrysum arenarium (L.) Moench. J. Essent. Oil Res. 2000, 12, 728–730. [Google Scholar] [CrossRef]
- Lemberkovics, E.; Czinner, E.; Balázs, A.; Bihátsi-Karsai, E.; Vitányi, G.; Lelik, L.; Bernáth, J.; Szóke, E. New data on composition of esssential oil from inflorescence of everlasting (Helichrysum arenarium (L.) Moench.). Acta Pharm. Hung. 2001, 71, 187–191. [Google Scholar] [PubMed]
- Judzentiene, A.; Butkiene, R. Chemical composition of the essential oils of wild Helichrysum arenarium (L.) with differently colored inflorescences from eastern Lithuania. J. Essent. Oil Res. 2006, 18, 80–83. [Google Scholar] [CrossRef]
- Bryksa-Godzisz, M.; Weglarz, Z.; Przybyl, J. Phenolic compounds in yellow everlasting (Helichrysum arenarium (L.) Moench) growing wild in the middle part of the Bug river valley. Herba Pol. 2006, 52, 26–31. [Google Scholar]
- Radušienė, J.; Judžentienė, A. Volatile composition of Helichrysum arenarium field accessions with differently coloured inflorescences. Biologija 2008, 54, 116–120. [Google Scholar] [CrossRef]
- Yang, Y.; Huang, Y.; Gu, D.; Yili, A.; Sabir, G.; Aisa, H.A. Separation and purification of three flavonoids from Helichrysum arenarium (L.) Moench by HSCCC. Chromatographia 2009, 69, 963–967. [Google Scholar] [CrossRef]
- Lv, H.; Sabir, G.; Kungurhan, B.; Liu, Y.; Aisa, H.A. New phthalide glycosides from Helichrysum arenarium (L.) Moench. J. Asian Nat. Prod. Res. 2009, 11, 352–356. [Google Scholar] [CrossRef]
- Zhang, Y.W.; Sun, W.X.; Li, X.; Zhao, C.C.; Meng, D.L.; Li, N. Two new compounds from Helichrysum arenarium (L.). J. Asian Nat. Prod. Res. 2009, 11, 289–293. [Google Scholar] [CrossRef]
- Eshbakova, K.A.; Aisa, N.A. Component of Helichrysum arenarium. Chem. Nat. Compd. 2009, 45, 929–930. [Google Scholar] [CrossRef]
- Morikawa, T.; Wang, L.-B.; Nakamura, S.; Ninomiya, K.; Yokoyama, E.; Matsuda, H.; Muraoka, O.; Wu, L.-J.; Yoshikawa, M. Medicinal flowers. XXVII. new flavanone and chalcone glycosides, arenariumosides I, II, III, and IV, and tumor necrosis factor-α inhibitors from everlasting, flowers of Helichrysum arenarium. Chem. Pharm. Bull. 2009, 57, 361–367. [Google Scholar] [CrossRef] [Green Version]
- Albayrak, S.; Aksoy, A.; Saǧdiç, O.; Budak, U. Phenolic compounds and antioxidant and antimicrobial properties of helichrysum species collected from eastern Anatolia, Turkey. Turk. J. Biol. 2010, 34, 463–473. [Google Scholar] [CrossRef]
- Yong, F.; Aisa, H.A.; Mukhamatkhanova, R.F.; Shamyanov, I.D.; Levkovich, M.G. New flavanone and other constituens of Helichrysum arenarium to China. Chem. Nat. Compd. 2011, 46, 872–875. [Google Scholar] [CrossRef]
- Oji, K.A.; Shafaghat, A. Constituents and antimicrobial activity of the essential oils from flower, leaf and stem of Helichrysum armenium in NPC. Nat. Prod. Commun. 2012, 7, 671–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gradinaru, A.C.; Silion, M.; Trifan, A.; Miron, A.; Aprotosoaie, A.C. Helichrysum arenarium subsp. arenarium phenolic composition and antibacterial activity against lower respiratory tract pathogens. Nat. Prod. Res. 2014, 28, 2076–2080. [Google Scholar] [CrossRef]
- Moghadam, H.D.; Sani, A.; Sangatash, M.M. Inhibitory effect of Helichrysum arenarium essential oil on the growth of food contaminated microorganisms. J. Essent. Oil Bear. Plants 2014, 17, 911–921. [Google Scholar] [CrossRef]
- Bandeira Reidel, R.V.; Cioni, P.; Cervelli, C.; Ruffoni, B.; Pistelli, L. Essential oil composition from three Helichrysum species. Planta Med. 2016, 82 (Suppl. 1), S1–S381. [Google Scholar] [CrossRef]
- Bandeira Reidel, R.V.; Cioni, P.L.; Ruffoni, B.; Cervelli, C.; Pistelli, L. Aroma profile and essential oil composition of Helichrysum species. Nat. Prod. Commun. 2017, 12, 1507–1512. [Google Scholar] [CrossRef] [Green Version]
- Babotă, M.; Mocan, A.; Vlase, L.; Crișan, O.; Ielciu, I.; Gheldiu, A.-M.; Vodnar, D.C.; Crișan, G.; Păltinean, R. 2018. Phytochemical analysis, antioxidant and antimicrobial activities of Helichrysum arenarium (L.) Moench. and Antennaria dioica (L.) Gaertn. Flowers. Molecules 2018, 23, 409. [Google Scholar] [CrossRef] [Green Version]
- Judzentiene, A.; Charkova, T.; Misiūnas, A. Chemical composition of the essential oils from Helichrysum arenarium (L.) plants growing in Lithuanian forests. J. Essent. Oil Res. 2019, 31, 305–311. [Google Scholar] [CrossRef]
- Liu, X.; Xuemin Jing, X.; Li, G. A process to acquire essential oil by distillation concatenated liquid-liquid extraction and flavonoids by solid-liquid extraction simultaneously from Helichrysum arenarium (L.) Moench inflorescences under ionic liquid-microwave mediated. Sep. Purif. Technol. 2019, 209, 164–174. [Google Scholar] [CrossRef]
- Stankov, S.; Fidan, H.; Petkova, N.; Stoyanova, A.; Dincheva, I.; Dogan, H.; Cosge, B.; Senkal, C.B.; Uskutoglu, T.; Bas, H.; et al. Phytochemical composition of Helichrysum arenarium (L.) Moench essential oil (aerial parts) from Turkey. Food Technol. Ukr. Food J. 2020, 9, 503–512. [Google Scholar] [CrossRef]
- Ivanović, M.; Albreht, A.; Krajnc, P.; Vovk, I.; Razboršek, M.I. Sustainable ultrasound-assisted extraction of valuable phenolics from inflorescences of Helichrysum arenarium L. using natural deep eutectic solvents. Ind. Crops Prod. 2021, 160, 113102. [Google Scholar] [CrossRef]
- Melito, S.; Petretto, G.L.; Podani, J.; Foddai, M.; Maldini, M.; Chessa, M.; Pintore, G. Altitude and climate influence Helichrysum italicum subsp. microphyllum essential oils com-position. Ind. Crops Prod. 2016, 80, 242–250. [Google Scholar] [CrossRef]
- Usai, M.; Foddai, M.; Bernardini, A.F.; Muselli, A.; Costa, J.; Marchetti, M. Chemical composition and variation of the essential oil of wild Sardinian Helichrysum italicum G. Don subsp. microphyllum (Willd.) Nym from vegetative period to post-blooming. J. Essent. Oil Res. 2010, 22, 373–380. [Google Scholar] [CrossRef]
- Benítez, G.; González-Tejero, M.R.; Molero-Mesa, J. Pharmaceutical ethnobotany in the western part of granada province (southern Spain): Ethnopharmacological synthesis. J. Ethnopharmacol. 2010, 129, 87–105. [Google Scholar] [CrossRef] [PubMed]
- Djihane, B.; Wafa, N.; Elkhamssa, S.; Pedro, H.J.; Maria, A.E.; Mohamed Mihoub, Z. Chemical constituents of Helichrysum italicum (Roth) G. Don essential oil and their antimicrobial activity against Gram-positive and Gram-negative bacteria, filamentous fungi and Candida albicans. Saudi Pharm. J. 2017, 25, 780–787. [Google Scholar] [CrossRef]
- Oliva, A.; Garzoli, S.; Sabatino, M.; Tadić, V.; Costantini, S.; Ragno, R.; Božović, M. Chemical composition and antimicrobial activity of essential oil of Helichrysum italicum (Roth) G. Don fil. (Asteraceae) from Montenegro. Nat. Prod. Res. 2020, 34, 445–448. [Google Scholar] [CrossRef]
- Staver, M.M.; Gobin, I.; Ratkaj, I.; Petrovic, M.; Vulinovic, A.; Sablic, D.M.; Broznic, D. In vitro antiproliferative and antimicrobial activity of the essential oil from the flowers and leaves of Helichrysum italicum (Roth) G. Don growing in Central Dalmatia (Croatia). J. Essent. Oil Bear. Plants 2018, 21, 77–91. [Google Scholar] [CrossRef]
- Dzamic, A.M.; Mileski, K.S.; Ciric, A.D.; Ristic, M.S.; Sokovic, M.D.; Marin, P.D. Essential oil composition, antioxidant and antimicrobial properties of essential oil and deodorized extracts of Helichrysum italicum (Roth) G. Don. J. Essent. Oil Bear. Plants 2019, 22, 493–503. [Google Scholar] [CrossRef]
- Mollova, S.; Fidan, H.; Antonova, D.; Bozhilov, D.; Stanev, S.; Kostova, I.; Stoyanova, A. Chemical composition and antimicrobial and antioxidant activity of Helichrysum italicum (Roth) G.Don subspecies essential oils. Turk. J. Agric. For. 2020, 44, 371–378. [Google Scholar] [CrossRef]
- Cantore, L.P.; Iacobellis, S.N.; Marco, D.A.; Capasso, F.; Senatore, F. Antibacterial activity of Coriandrum sativum L. and Foeniculum vulgare Miller essential oil. J. Agric. Food Chem. 2004, 52, 7862–7866. [Google Scholar] [CrossRef] [PubMed]
- Mesic, A.; Mahmutović-Dizdarević, I.; Tahirović, E.; Durmišević, I.; Eminovic, I.; Jerković-Mujkić, A.; Bešta-Gajević, R. Evaluation of toxicological and antimicrobial activity of lavender and immortelle essential oils. Drug Chem. Toxicol. 2021, 44, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Rossi, P.G.; Berti, L.; Panighi, J.; Luciani, A.; Maury, J.; Muselli, A.; De Rocca Serra, D.; Gonny, M.; Bolla, J.M. Antibacterial action of essential oils from Corsica. J. Essen. Oils Res. 2007, 19, 176–182. [Google Scholar] [CrossRef]
- Combes, C.; Legrix, M.; Rouquet, V.; Rivoire, S.; Grasset, S.; Cenizo, V.; Moga, A.; Portes, P. 166 Helichrysum italicum essential oil prevents skin lipids peroxidation caused by pollution and UV. J. Investig. Dermatol. 2017, 137, S221. [Google Scholar] [CrossRef]
- Thiers, B. Index Herbariorum: A Global Directory of Public Herbaria and Associated Staff; New York Botanical Garden’s Virtual Herbarium: New York, NY, USA, 2019; Available online: http://sweetgum.nybg.org/ih (accessed on 25 June 2021).
- Zheljazkov, V.D.; Semerdjieva, I.B.; Dincheva, I.; Kacaniova, M.; Astatkie, T.; Radoukova, T.; Schlegel, V. Antimicrobial and antioxidant activity of Juniper galbuli essential oil constituents eluted at different times. Ind. Crops Prod. 2017, 109, 529–537. [Google Scholar] [CrossRef]
- Montgomery, D.C. Design and Analysis of Experiments, 10th ed.; Wiley & Sons: New York, NY, USA, 2020. [Google Scholar]
- SAS Institute Inc. SAS/STAT® 9.4 User’s Guide; SAS Institute Inc.: Cary, NC, USA, 2014. [Google Scholar]
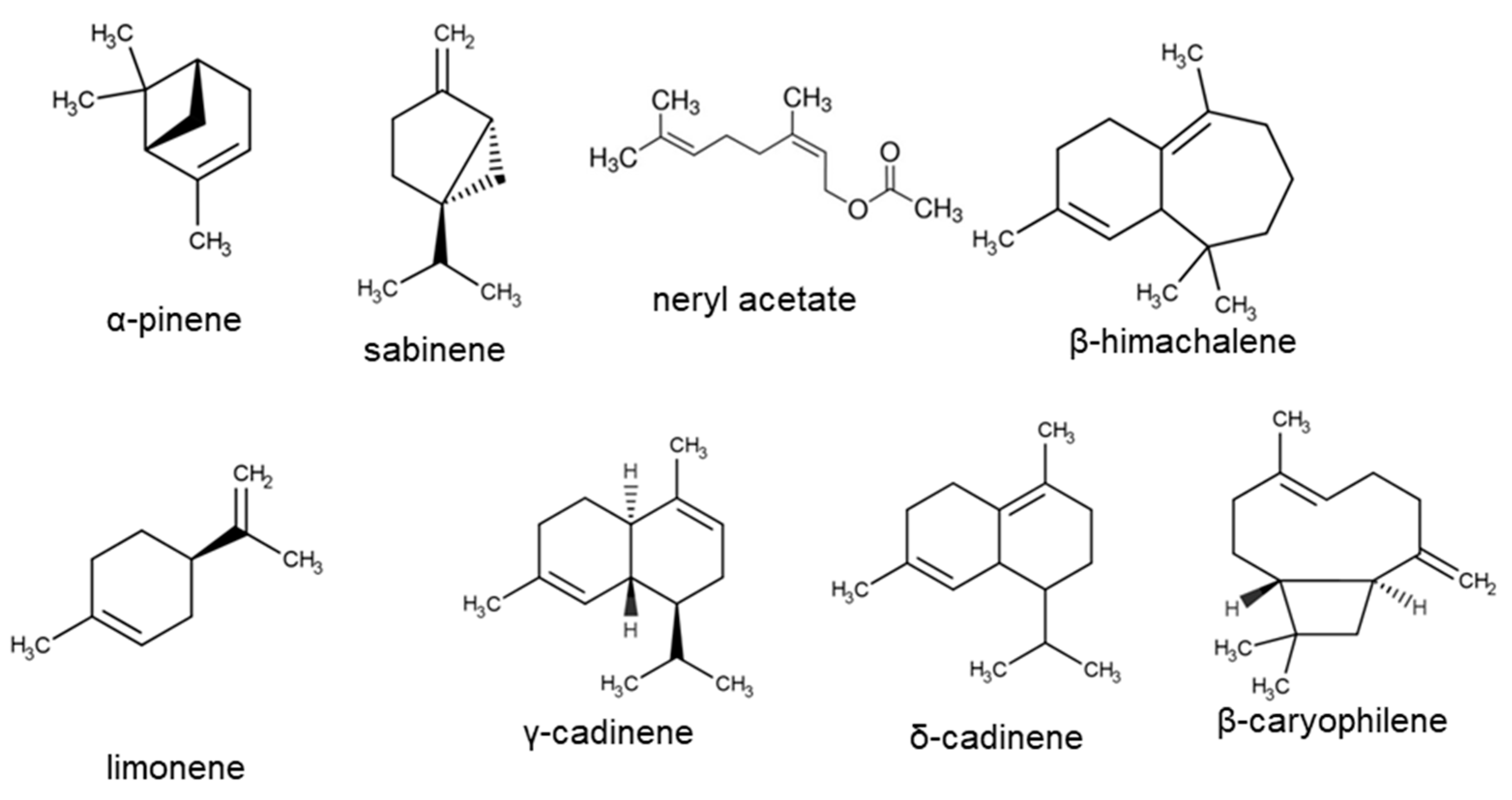

| Reference | Main Compounds | Country |
|---|---|---|
| Rančić et al. [4] | diepi-α-cedrene (17.9%), α-ylangene (13.9%), cyclosativene (11.9%), limonene (11.4%) | Serbia |
| Mao et al. [5] | narirutin, naringin, eriodictyol, luteolin, galuteolin, astragalin, kaempferol | China |
| Smirnova and Pervykh [29] | flavonoids-astragalin, luteolin, kaempferol | Russian Federation |
| Czinner et al. [30] | phenolic compound | Hungary |
| Czinner et al. [31] | linalool (1.7%), anethole (3.2%), carvacrol (3.6%), α-muurolol (1.3%), 1.5% of β-asarone | Hungary |
| Lemberkovics et al. [32] | linalool, alpha-terpineol, carvone monoterpenes; anethole, anisaldehyde, thymol, carvacrol, eugenol, beta-asarone, butylhydroxyanisole aromatic components; alpha-humulene, beta-caryophyllene, gamma-muurolene, delta-cadinene, copaene, alpha-gurjunene, caryophyllenol, delta-cadinol and globulol sesquiterpenes, caprylic acid, pelargonic, caprinic, lauric acids, methyl palmitate | Hungary |
| Judzentiene and Butkiene [33] | β-caryophyllene; δ-cadinene; octadecane; heneicosane | Lithuania |
| Bryksa-Godzisz et al. [34] | phenolic compounds | Poland |
| Radušienė and Judžentienė [35] | 1.8-cineole (2.3–7%); α-copaene (2.2–3.6%); trans-caryophyllene (4.4–8.8%); epi-a-cadinol (2–4%); m/z-149 (phthalide)(0.6–5.6%); heneicosane (1.5–5.1%) | Lithuania |
| Yang et al. [36] | Flavonoids (naringenin-7-O-β-d-glycoside, isoquercitrin, astragalin) | China |
| Lv et al. [37] | prenylated phthalide glycosides | China |
| Zhang et al. [38] | 6,7-dimethoxy-4-hydroxy-1-naphthoic acid (1),(Z)-5-hydroxy-7-methoxy-4-[3-methyl-4-(O-β-d-xylopyranosyl)but-2-enyl]isobenzofuran-1(3H)-one (2). | China |
| Eshbakova and Aisa [39] | naringenin, helichrysum phthalide, diosmin, oleanolic acid | Republic of Uzbekistan |
| Morikawa et al. [40] | naringenin 7-O-β-D-glucopyranoside, apigenin 7-O-β-D-glucopyranoside, apigenin 7-O-gentiobioside, apigenin 7,4′-di-O-β-D-glucopyranoside | cultivated in Poland purchased Tochimoto Tenkaido Co., Ltd., Osaka, Japan |
| Albayrak et al. [41] | phenolic compounds | Turkey |
| Yong et al. [42] | β-sitosterol, stigmasterol, β-sitosterol, β-D-glucopyranoside, stigmasterol, caffeic acid ethyl ester. | China |
| Oji et al. [43] | limonene (21.2%), alpha-cadinol (18.2%), borneol (11.9%), delta-cadinene (9%), bornyl acetate (8%), alpha-humulene (7.3%). | Iran |
| Gradinaru et al. [44] | caffeic acid; flavonoids (apigenin, naringenin, apigenin-7-O-glucoside, naringenin-O-hexosides) | Romania |
| Moghadam et al. [45] | spathulenol (36.6%), β-pinene (12.5%) | Iran |
| Bandeira Reidel et al. [46] | β-caryophyllene (27–46%); (E)-2-hexenal; β-pinene (7.4%); | Italy |
| Bandeira Reidel et al. [47] | β-pinene (7.4%); β-caryophyllene (27.5%); δ-cadinene (3.2%); pentadecanoic acid, methyl ester (31%) | Italy |
| Babotă et al. [48] | phenolic compound; methoxylated flavone; sterolic compound; | Romania |
| Judzentiene et al. [49] | 1,8-cineole (8.9%, one sample), β-caryophyllene (5.8–36.2%, 14 oils), γ- and δ-cadinene (5.8% and 9%); octadecane (7.1–22.3%). | Lithuania |
| Liu et al. [50] | linalool (2.81%); 4-acetyl-1-methylcyclohexene (1.88%); β-spathulenol (24.03%); caryophyllene oxide (3.05%); ledol (6.22%); hinesol (3.86%); β-eudesmol (2.56%); α-eudesmol (4.37%); α-cadinol (7.76%); α-bisabolol (5.71%) | Inner Mongolia, China |
| Stankov et al. [51] | oleic acid (30.28%), ethyl hexadecanoate (20.19%), linoleic acid (18.89%), sclareol (4.22%) | Turkey |
| Ivanović et al. [52] | phenolic compounds | Slovenia |
| Constituent | p-Value | Constituent | p-Value | Constituent | p-Value |
|---|---|---|---|---|---|
| α-pinene | 0.033 * | β-gurjunene | 0.322 | 1-terpinen-4-ol | 0.002 |
| sabinene | 0.716 | germacrene D | 0.027 | long-chain alkane | 0.017 |
| β-pinene | 0.073 | germacra-4(15),5,10(14)-trien-1 | 0.089 | n-tetradecane | 0.138 |
| D-limonene | 0.403 | monoterpenes | 0.004 | diterpenoids | 0.355 |
| trans-verbenol | 0.400 | sesquiterpenes | 0.084 |
| Collection | α-Pinene | β-Pinene | 1-Terpinen-4-ol | Germacrene D | Germacra-4(15),5,10(14)-trien |
|---|---|---|---|---|---|
| 689 Location 1 | 44.35 a * | 2.85 a | 0.92 c | 3.56 b | 1.14 ab |
| 691 Location 2 | 36.60 b | 2.30 b | 2.11 a | 5.33 a | 1.09 b |
| 699 Location 3 | 34.64 b | 2.67 ab | 1.26 b | 4.83 a | 1.37 a |
| Collection | Monoterpenes | Sesquiterpenes | Long-chain alkane | Diterpenoids | |
| 689 Location 1 | 73.99 a | 16.08 b | 4.33 b | 3.25 b | |
| 691 Location 2 | 68.96 b | 18.01 ab | 6.23 a | 3.45 ab | |
| 699 Location 3 | 65.72 c | 19.41 a | 5.69 a | 4.27 a |
| Constituent | Overall Mean Concentration | |
|---|---|---|
| Sabinene | 10.80 | 0.607 |
| D-limonene | 2.11 | 0.151 |
| trans-verbenol | 3.18 | 0.217 |
| n-tetradecane | 2.36 | 0.135 |
| β-gurjunene | 3.61 | 0.259 |
| Constituent | p-Value | Constituent | p-Value |
|---|---|---|---|
| α-Pinene | <0.001 * | β-Caryophyllene | 0.001 |
| D-Limonene | 0.019 | p-Cymen-7-ol acetate | 0.004 |
| 2-Methyl butyl-2-methyl butyrate | <0.001 | α-Guaiene | <0.001 |
| Isoamyl tiglate | 0.004 | γ-Curcumene | 0.002 |
| 1-Terpinen-4-ol | 0.001 | β-Himachalene | 0.467 |
| Nerol | 0.001 | β-Curcumene | 0.232 |
| Neryl acetate | 0.001 | Germacrene D-4-ol | 0.025 |
| α-Copaene | 0.049 | γ-Eudesmol | 0.027 |
| Italicene | 0.016 | tau.-Muurolol | 0.751 |
| α-cis-Bergamotene | 0.002 | β-Eudesmol | 0.120 |
| Sesquiterpenes | 0.048 | Monoterpenes | <0.001 |
| Ester | 0.669 | Long-chain alkane | 0.069 |
| Country | α-Pinene | D-Limonene | 2-Methyl butyl-2-methyl butyr | Isoamyl tiglate | 1-Terpinen-4-ol | Nerol | Neryl acetate |
| Bosnia | 13.74 a * | 3.37 b | 0.087 c | 0.83 b | 0.29 c | 0.19 b | 4.04 c |
| France | 4.84 b | 5.23 a | 4.31 a | 1.74 a | 1.37 b | 2.26 a | 14.87 a |
| Corsica | 2.83 c | 4.94 a | 3.44 b | 1.93 a | 1.67 a | 2.50 a | 12.37 b |
| Country | α-Copaene | Italicene | α-cis-Bergamotene | β-Caryophyllene | p-Cymen-7-ol acetate | α-Guaiene | γ-Curcumene |
| Bosnia | 2.38 a | 2.93 b | 0.38 b | 3.65 a | 5.27 a | 1.71 b | 2.46 a |
| France | 1.99 ab | 4.67 a | 1.19 a | 0.38 b | 2.47 b | 3.98 a | 0.93 b |
| Corsica | 1.71 b | 4.23 a | 1.19 a | 0.48 b | 2.50 b | 4.08 a | 0.65 b |
| Country | Germacrene D-4-ol | γ-Eudesmol | β-Himachalene | Monoterpenes | Sesquiterpenes | Long-chain alkane | Ester |
| Bosnia | 2.49 a | 1.71 b | 10.80 ab | 29.83 c | 54.80 a | 0.74 b | 7.84 ab |
| France | 0.80 b | 3.35 a | 9.90 b | 37.30 a | 45.23 b | 0.84 ab | 7.96 a |
| Corsica | 1.81 a | 3.47 a | 10.99 a | 35.29 b | 47.90 ab | 0.99 a | 7.09 c |
| Constituent | Overall Mean Concentration | |
|---|---|---|
| β-Himachalene | 10.57 | 0.832 |
| β-Curcumene | 2.05 | 0.411 |
| tau.-Muurolol | 1.13 | 0.083 |
| β-Eudesmol | 1.44 | 0.208 |
| Ester | 7.63 | 0.982 |
| Location | SA | EF | SP | PA | YE | SE | CA | CK | CT |
|---|---|---|---|---|---|---|---|---|---|
| Bosnia | 9.33 b | 4.00 b | 8.33 b | 2.33 b | 2.33 b | 5.67 a | 5.33 a | 4.67 b | 5.67 a |
| Corsica | 14.67 a | 1.67 c | 6.67 c | 2.67 b | 5.33 a | 3.33 b | 5.33 a | 5.67 b | 4.33 a |
| France | 14.67 a | 12.33 a | 10.67 a | 5.33 a | 5.00 a | 4.00 b | 5.67 a | 10.67 a | 6.33 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheljazkov, V.D.; Semerdjieva, I.; Yankova-Tsvetkova, E.; Astatkie, T.; Stanev, S.; Dincheva, I.; Kačániová, M. Chemical Profile and Antimicrobial Activity of the Essential Oils of Helichrysum arenarium (L.) Moench. and Helichrysum italicum (Roth.) G. Don. Plants 2022, 11, 951. https://doi.org/10.3390/plants11070951
Zheljazkov VD, Semerdjieva I, Yankova-Tsvetkova E, Astatkie T, Stanev S, Dincheva I, Kačániová M. Chemical Profile and Antimicrobial Activity of the Essential Oils of Helichrysum arenarium (L.) Moench. and Helichrysum italicum (Roth.) G. Don. Plants. 2022; 11(7):951. https://doi.org/10.3390/plants11070951
Chicago/Turabian StyleZheljazkov, Valtcho D., Ivanka Semerdjieva, Elina Yankova-Tsvetkova, Tess Astatkie, Stanko Stanev, Ivayla Dincheva, and Miroslava Kačániová. 2022. "Chemical Profile and Antimicrobial Activity of the Essential Oils of Helichrysum arenarium (L.) Moench. and Helichrysum italicum (Roth.) G. Don" Plants 11, no. 7: 951. https://doi.org/10.3390/plants11070951
APA StyleZheljazkov, V. D., Semerdjieva, I., Yankova-Tsvetkova, E., Astatkie, T., Stanev, S., Dincheva, I., & Kačániová, M. (2022). Chemical Profile and Antimicrobial Activity of the Essential Oils of Helichrysum arenarium (L.) Moench. and Helichrysum italicum (Roth.) G. Don. Plants, 11(7), 951. https://doi.org/10.3390/plants11070951








