The Response of Beech (Fagus sylvatica L.) Populations to Climate in the Easternmost Sites of Its European Distribution
Abstract
1. Introduction
2. Results and Discussions
2.1. Chronology Description
2.2. Beech Response to Climatic Conditions
2.3. Spatio-Temporal Stability of the Climate–Growth Relationship
3. Materials and Methods
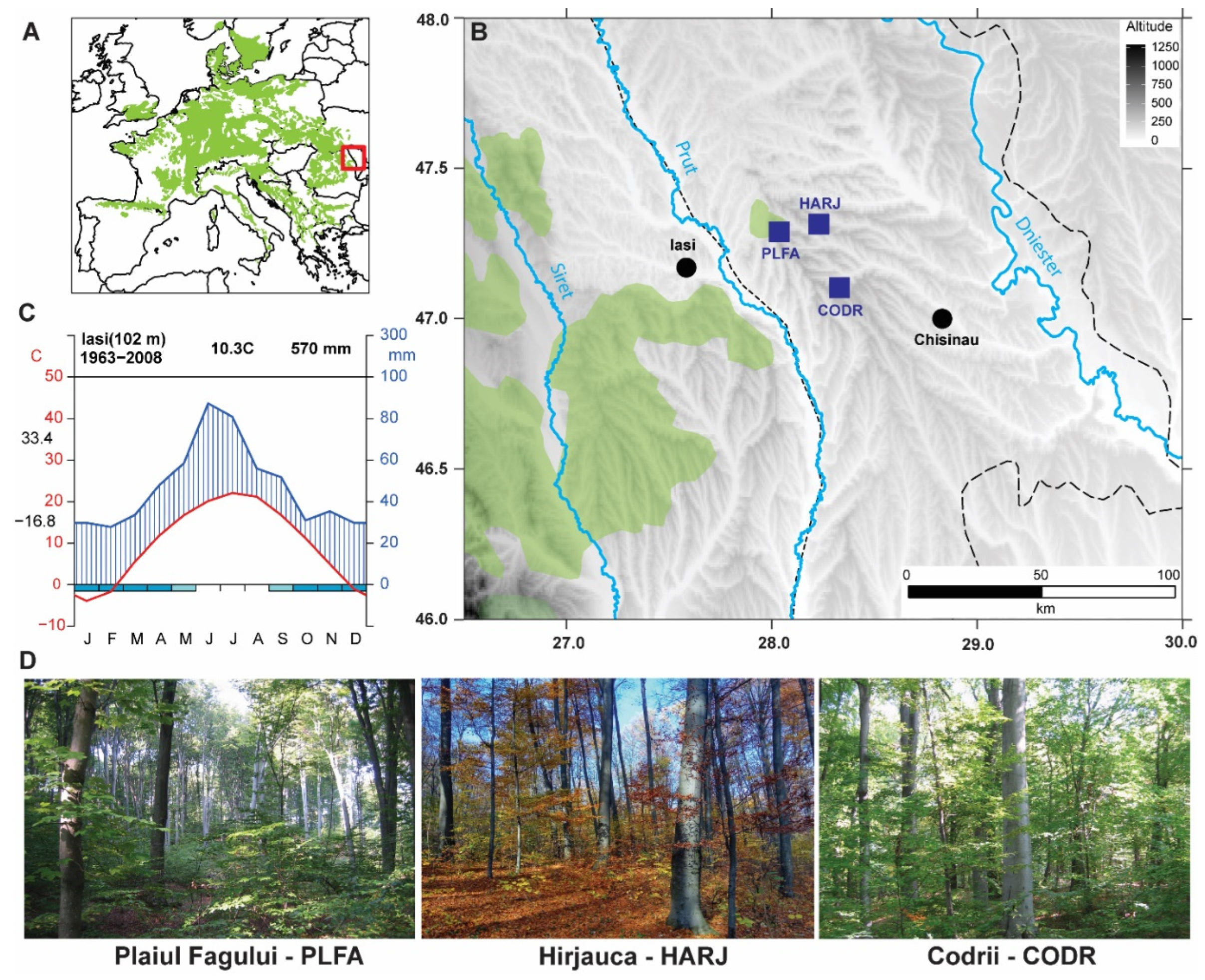
3.1. Site Description
3.2. Chronology Development
3.3. Climate–Growth Relationship
3.4. Stability Maps
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- IPCC. Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Genève, Switzerland, 2021; Available online: https://www.ipcc.ch/report/ar6/wg1/ (accessed on 24 October 2022).
- Lesnikowski, A.; Biesbroek, R.; Ford, J.D.; Berrang-Ford, L. Policy Implementation Styles and Local Governments: The Case of Climate Change Adaptation. Environ. Politics 2020, 30, 753–790. [Google Scholar] [CrossRef]
- Elomina, J.; Pülzl, H. How Are Forests Framed? An Analysis of EU Forest Policy. For. Policy Econ. 2021, 127, 102448. [Google Scholar] [CrossRef]
- Sutanto, S.J.; Vitolo, C.; di Napoli, C.; D’Andrea, M.; van Lanen, H.A.J. Heatwaves, Droughts, and Fires: Exploring Compound and Cascading Dry Hazards at the Pan-European Scale. Environ. Int. 2020, 134, 105276. [Google Scholar] [CrossRef]
- Hlásny, T.; Krokene, P.; Liebhold, A.; Montagné-Huck, C.; Müller, J.; Qui, H.; Raffa, K.; Schelhaas, M.-J.; Seidl, R.; Svoboda, M.; et al. Living with Bark Beetles Impacts, Outlook and Management Options; European Forest Institute: Joensuu, Finland, 2019. [Google Scholar]
- Moravec, V.; Markonis, Y.; Rakovec, O.; Svoboda, M.; Trnka, M.; Kumar, R.; Hanel, M. Europe under Multi-Year Droughts: How Severe Was the 2014–2018 Drought Period? Environ. Res. Lett. 2021, 16, 034062. [Google Scholar] [CrossRef]
- George, J.P.; Neumann, M.; Vogt, J.; Cammalleri, C.; Lang, M. Assessing Effects of Drought on Tree Mortality and Productivity in European Forests across Two Decades: A Conceptual Framework and Preliminary Results. IOP Conf. Ser. Earth Environ. Sci. 2021, 932, 012009. [Google Scholar] [CrossRef]
- Gazol, A.; Camarero, J.J. Compound Climate Events Increase Tree Drought Mortality across European Forests. Sci. Total Environ. 2022, 816, 151604. [Google Scholar] [CrossRef]
- Senf, C.; Buras, A.; Zang, C.S.; Rammig, A.; Seidl, R. Excess Forest Mortality Is Consistently Linked to Drought across Europe. Nat. Commun. 2020, 11, 6200. [Google Scholar] [CrossRef]
- Potop, V.; Soukup, J. Spatiotemporal Characteristics of Dryness and Drought in the Republic of Moldova. Theor. Appl. Climatol. 2009, 96, 305–318. [Google Scholar] [CrossRef]
- Potop, V.; Boroneant, C.; Caian, M. Assessing the Changes in Drought Conditions during Summer in the Republic of Moldova Based on RegCM Simulations. J. Econ. Dev. Environ. People 2013, 2, 63–76. [Google Scholar]
- Olesea, C. Drought in The Republic of Moldova Becomes More Common and Intensive. Lucr. Ştiinţifice. Ser. Agron. 2020, 63, 157–162. [Google Scholar]
- Páscoa, P.; Gouveia, C.M.; Russo, A.C.; Bojariu, R.; Vicente-Serrano, S.M.; Trigo, R.M. Drought Impacts on Vegetation in Southeastern Europe. Remote Sens. 2020, 12, 2156. [Google Scholar] [CrossRef]
- Potop, V. Evolution of Drought Severity and Its Impact on Corn in the Republic of Moldova. Theor. Appl. Climatol. 2011, 105, 469–483. [Google Scholar] [CrossRef]
- Peters, R. Beech Forests; Geobotany; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2013; Volume 24, ISBN 0792344855. [Google Scholar]
- Geßler, A.; Keitel, C.; Kreuzwieser, J.; Matyssek, R.; Seiler, W.; Rennenberg, H. Potential Risks for European Beech (Fagus Sylvatica L.) in a Changing Climate. Trees-Struct. Funct. 2007, 21, 1–11. [Google Scholar] [CrossRef]
- Dittmar, C.; Zech, W.; Elling, W. Growth Variations of Common Beech (Fagus Sylvatica L.) under Different Climatic and Environmental Conditions in Europe- a Dendroecological Study. For. Ecol. Manag. 2003, 173, 63–78. [Google Scholar]
- Martinez del Castillo, E.; Zang, C.S.; Buras, A.; Hacket-Pain, A.; Esper, J.; Serrano-Notivoli, R.; Hartl, C.; Weigel, R.; Klesse, S.; Resco de Dios, V.; et al. Climate-Change-Driven Growth Decline of European Beech Forests. Commun. Biol. 2022, 5, 163. [Google Scholar] [CrossRef]
- Neycken, A.; Scheggia, M.; Bigler, C.; Lévesque, M. Long-Term Growth Decline Precedes Sudden Crown Dieback of European Beech. Agric. For. Meteorol. 2022, 324, 109103. [Google Scholar] [CrossRef]
- Dulamsuren, C.; Hauck, M.; Kopp, G.; Ruff, M.; Leuschner, C. European Beech Responds to Climate Change with Growth Decline at Lower, and Growth Increase at Higher Elevations in the Center of Its Distribution Range (SW Germany). Trees-Struct. Funct. 2017, 31, 673–686. [Google Scholar] [CrossRef]
- Kolzenburg, R. The Direct Influence of Climate Change on Marginal Populations: A Review. Aquat. Sci. 2022, 84, 24. [Google Scholar] [CrossRef]
- Kitamura, K.; Matsui, T.; Kobayashi, M.; Saitou, H.; Namikawa, K.; Tsuda, Y. Decline in Gene Diversity and Strong Genetic Drift in the Northward-Expanding Marginal Populations of Fagus Crenata. Tree Genet. Genomes 2015, 11, 36. [Google Scholar] [CrossRef]
- Wortemann, R.; Herbette, S.; Barigah, T.S.; Fumanal, B.; Alia, R.; Ducousso, A.; Gomory, D.; Roeckel-Drevet, P.; Cochard, H. Genotypic Variability and Phenotypic Plasticity of Cavitation Resistance in Fagus Sylvatica L. across Europe. Tree Physiol. 2011, 31, 1175–1182. [Google Scholar] [CrossRef]
- Harter, D.E.V.; Nagy, L.; Backhaus, S.; Beierkuhnlein, C.; Fussi, B.; Huber, G.; Jentsch, A.; Konnert, M.; Thiel, D.; Kreyling, J. A Comparison of Genetic Diversity and Phenotypic Plasticity among European Beech (Fagus Sylvatica L.) Populations from Bulgaria and Germany under Drought and Temperature Manipulation. Int. J. Plant Sci. 2015, 176, 232–244. [Google Scholar] [CrossRef]
- Frank, A.; Pluess, A.R.; Howe, G.T.; Sperisen, C.; Heiri, C. Quantitative Genetic Differentiation and Phenotypic Plasticity of European Beech in a Heterogeneous Landscape: Indications for Past Climate Adaptation. Perspect. Plant Ecol. Evol. Syst. 2017, 26, 1–13. [Google Scholar] [CrossRef]
- Dieler, J.; Pretzsch, H. Morphological Plasticity of European Beech (Fagus Sylvatica L.) in Pure and Mixed-Species Stands. For. Ecol. Manag. 2013, 295, 97–108. [Google Scholar] [CrossRef]
- Cavin, L.; Jump, A.S. Highest Drought Sensitivity and Lowest Resistance to Growth Suppression Are Found in the Range Core of the Tree Fagus Sylvatica L. Not the Equatorial Range Edge. Glob. Chang. Biol. 2017, 23, 362–379. [Google Scholar] [CrossRef]
- Zimmermann, J.; Hauck, M.; Dulamsuren, C.; Leuschner, C. Climate Warming-Related Growth Decline Affects Fagus Sylvatica, But Not Other Broad-Leaved Tree Species in Central European Mixed Forests. Ecosystems 2015, 18, 560–572. [Google Scholar] [CrossRef]
- Falk, W.; Hempelmann, N. Species Favourability Shift in Europe Due to Climate Change: A Case Study for Fagus Sylvatica L. and Picea Abies (L.) Karst. Based on an Ensemble of Climate Models. J. Climatol. 2013, 2013, 787250. [Google Scholar] [CrossRef]
- Bugmann, H.; Pfister, C. Impacts of Interannual Climate Variability on Past and Future Forest Composition. Reg. Environ. Chang. 2000, 1, 112–125. [Google Scholar] [CrossRef]
- Rasche, L.; Fahse, L.; Zingg, A.; Bugmann, H. Enhancing Gap Model Accuracy by Modeling Dynamic Height Growth and Dynamic Maximum Tree Height. Ecol. Model. 2012, 232, 133–143. [Google Scholar] [CrossRef]
- Dorado-Liñán, I.; Akhmetzyanov, L.; Menzel, A. Climate Threats on Growth of Rear-Edge European Beech Peripheral Populations in Spain. Int. J. Biometeorol. 2017, 61, 2097–2110. [Google Scholar] [CrossRef]
- Parmesan, C. Climate and Species’ Range. Nature 1996, 382, 765–766. [Google Scholar] [CrossRef]
- Rose, L.; Leuschner, C.; Köckemann, B.; Buschmann, H. Are Marginal Beech (Fagus Sylvatica L.) Provenances a Source for Drought Tolerant Ecotypes? Eur. J. For. Res. 2009, 128, 335–343. [Google Scholar] [CrossRef]
- di Filippo, A.; Biondi, F.; Čufar, K.; de Luis, M.; Grabner, M.; Maugeri, M.; Presutti Saba, E.; Schirone, B.; Piovesan, G. Bioclimatology of Beech (Fagus Sylvatica L.) in the Eastern Alps: Spatial and Altitudinal Climatic Signals Identified through a Tree-Ring Network. J. Biogeogr. 2007, 34, 1873–1892. [Google Scholar] [CrossRef]
- Roibu, C.C.; Popa, I.; Kirchhefer, A.J.; Palaghianu, C. Growth Responses to Climate in a Tree-Ring Network of European Beech (Fagus Sylvatica L.) from the Eastern Limit of Its Natural Distribution Area. Dendrochronologia 2017, 42, 104–116. [Google Scholar] [CrossRef]
- Roibu, C.-C.; Nagavciuc, V.; Ionita, M.; Popa, I.; Horodnic, S.-A.; Mursa, A.; Büntgen, U. A Tree Ring-Based Hydroclimate Reconstruction for Eastern Europe Reveals Large-Scale Teleconnection Patterns. Clim. Dyn. 2022, 59, 2979–2994. [Google Scholar] [CrossRef]
- Tegel, W.; Seim, A.; Hakelberg, D.; Hoffmann, S.; Panev, M.; Westphal, T.; Büntgen, U. A Recent Growth Increase of European Beech (Fagus Sylvatica L.) at Its Mediterranean Distribution Limit Contradicts Drought Stress. Eur. J. For. Res. 2014, 133, 61–71. [Google Scholar] [CrossRef]
- Hacket-Pain, A.J.; Cavin, L.; Friend, A.D.; Jump, A.S. Consistent Limitation of Growth by High Temperature and Low Precipitation from Range Core to Southern Edge of European Beech Indicates Widespread Vulnerability to Changing Climate. Eur. J. For. Res. 2016, 135, 897–909. [Google Scholar] [CrossRef]
- Farahat, E.; Linderholm, H.W. Growth–Climate Relationship of European Beech at Its Northern Distribution Limit. Eur. J. For. Res. 2018, 137, 619–629. [Google Scholar] [CrossRef]
- Piovesan, G.; Biondi, F.; Bernabei, M.; di Filippo, A.; Schirone, B. Spatial and Altitudinal Bioclimatic Zone of the Italian Peninsula Identified from a Beech (Fagus Sylvatica) Tree Network. Acta Oecol. 2005, 27, 197–210. [Google Scholar]
- Piovesan, G.; Biondi, F.; di Filippo, A.; Alessandrini, A.; Maugeri, M. Drought-Driven Growth Reduction in Old Beech (Fagus Sylvatica L.) Forests of the Central Apennines, Italy. Glob. Chang. Biol. 2008, 14, 1265–1281. [Google Scholar] [CrossRef]
- Büntgen, U.; Krusic, P.J.; Piermattei, A.; Coomes, D.A.; Esper, J.; Myglan, V.S.; Kirdyanov, A.V.; Camarero, J.J.; Crivellaro, A.; Körner, C. Limited Capacity of Tree Growth to Mitigate the Global Greenhouse Effect under Predicted Warming. Nat. Commun. 2019, 10, 2171. [Google Scholar] [CrossRef]
- Brienen, R.J.W.; Caldwell, L.; Duchesne, L.; Voelker, S.; Barichivich, J.; Baliva, M.; Ceccantini, G.; di Filippo, A.; Helama, S.; Locosselli, G.M.; et al. Forest Carbon Sink Neutralized by Pervasive Growth-Lifespan Trade-Offs. Nat. Commun. 2020, 11, 4241. [Google Scholar] [CrossRef]
- Dorado-Liñán, I.; Ayarzagüena, B.; Babst, F.; Xu, G.; Gil, L.; Battipaglia, G.; Buras, A.; Čada, V.; Camarero, J.J.; Cavin, L.; et al. Jet Stream Position Explains Regional Anomalies in European Beech Forest Productivity and Tree Growth. Nat. Commun. 2022, 13, 2015. [Google Scholar] [CrossRef]
- Muffler, L.; Weigel, R.; Hacket-Pain, A.J.; Klisz, M.; van der Maaten, E.; Wilmking, M.; Kreyling, J.; van der Maaten-Theunissen, M. Lowest Drought Sensitivity and Decreasing Growth Synchrony towards the Dry Distribution Margin of European Beech. J. Biogeogr. 2020, 47, 1910–1921. [Google Scholar] [CrossRef]
- Robson, T.M.; Sánchez-Gómez, D.; Cano, F.J.; Aranda, I. Variation in Functional Leaf Traits among Beech Provenances during a Spanish Summer Reflects the Differences in Their Origin. Tree Genet. Genomes 2012, 8, 1111–1121. [Google Scholar] [CrossRef]
- Esper, J.; Benz, M.; Pederson, N. Influence of Wood Harvest on Tree-Ring Time-Series of Picea Abies in a Temperate Forest. For. Ecol. Manag. 2012, 284, 86–92. [Google Scholar] [CrossRef]
- Cescatti, A.; Piutti, E. Silvicultural Alternatives, Competition Regime and Sensitivity to Climate in a European Beech Forest. For. Ecol. Manag. 1998, 102, 213–223. [Google Scholar] [CrossRef]
- Čufar, K.; Prislan, P.; de Luis, M.; Gričar, J. Tree-Ring Variation, Wood Formation and Phenology of Beech (“Fagus Sylvatica”) from a Representative Site in Slovenia, SE Central Europe. Trees-Struct. Funct. 2008, 22, 749–758. [Google Scholar] [CrossRef]
- Arnič, D.; Gričar, J.; Jevšenak, J.; Božič, G.; von Arx, G.; Prislan, P. Different Wood Anatomical and Growth Responses in European Beech (Fagus Sylvatica L.) at Three Forest Sites in Slovenia. Front. Plant Sci. 2021, 12, 669229. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gazol, A.; Sangüesa-Barreda, G.; Vergarechea, M.; Alfaro-Sánchez, R.; Cattaneo, N.; Vicente-Serrano, S.M. Tree Growth Is More Limited by Drought in Rear-Edge Forests Most of the Times. For. Ecosyst. 2021, 8, 25. [Google Scholar] [CrossRef]
- Meyer, B.F.; Buras, A.; Rammig, A.; Zang, C.S. Higher Susceptibility of Beech to Drought in Comparison to Oak. Dendrochronologia 2020, 64, 125780. [Google Scholar] [CrossRef]
- Mette, T.; Dolos, K.; Meinardus, C.; Bräuning, A.; Reineking, B.; Blaschke, M.; Pretzsch, H.; Beierkuhnlein, C.; Gohlke, A.; Wellstein, C. Climatic Turning Point for Beech and Oak under Climate Change in Central Europe. Ecosphere 2013, 4, 1–9. [Google Scholar] [CrossRef]
- Allen, C.D.; Breshears, D.D.; McDowell, N.G. On Underestimation of Global Vulnerability to Tree Mortality and Forest Die-off from Hotter Drought in the Anthropocene. Ecosphere 2015, 6, 1–55. [Google Scholar] [CrossRef]
- Leuschner, C. Drought Response of European Beech (Fagus Sylvatica L.)—A Review. Perspect. Plant Ecol. Evol. Syst. 2020, 47, 125576. [Google Scholar] [CrossRef]
- Kozlowski, T.T.; Pallardy, S.G. Growth Control in Woody Plants; Elsevier: Amsterdam, The Netherlands, 1997; ISBN 0080532683. [Google Scholar]
- Pallardy, S. Physiology of Woody Plants, 3rd ed.; Elsevier Academic Press: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Bolte, A.; Czajkowski, T.; Kompa, T. The North-Eastern Distribution Range of European Beech—A Review. Forestry 2007, 80, 413–429. [Google Scholar] [CrossRef]
- Aranda, I.; Cano, F.J.; Gascó, A.; Cochard, H.; Nardini, A.; Mancha, J.A.; López, R.; Sánchez-Gómez, D. Variation in Photosynthetic Performance and Hydraulic Architecture across European Beech (Fagus Sylvatica L.) Populations Supports the Case for Local Adaptation to Water Stress. Tree Physiol. 2015, 35, 34–46. [Google Scholar] [CrossRef]
- Grossiord, C.; Buckley, T.N.; Cernusak, L.A.; Novick, K.A.; Poulter, B.; Siegwolf, R.T.W.; Sperry, J.S.; McDowell, N.G. Plant Responses to Rising Vapor Pressure Deficit. New Phytol. 2020, 226, 1550–1566. [Google Scholar] [CrossRef]
- McDowell, N.G.; Sapes, G.; Pivovaroff, A.; Adams, H.D.; Allen, C.D.; Anderegg, W.R.L.; Arend, M.; Breshears, D.D.; Brodribb, T.; Choat, B.; et al. Mechanisms of Woody-Plant Mortality under Rising Drought, CO2 and Vapour Pressure Deficit. Nat. Rev. Earth Environ. 2022, 3, 294–308. [Google Scholar]
- Sanginés de Cárcer, P.; Vitasse, Y.; Peñuelas, J.; Jassey, V.E.J.; Buttler, A.; Signarbieux, C. Vapor–Pressure Deficit and Extreme Climatic Variables Limit Tree Growth. Glob. Chang. Biol. 2018, 24, 1108–1122. [Google Scholar] [CrossRef]
- Lens, F.; Gleason, S.M.; Bortolami, G.; Brodersen, C.; Delzon, S.; Jansen, S. Functional Xylem Characteristics Associated with Drought-induced Embolism in Angiosperms. New Phytol. 2022, 236, 2019–2036. [Google Scholar] [CrossRef]
- Elliott, K.J.; Miniat, C.F.; Pederson, N.; Laseter, S.H. Forest Tree Growth Response to Hydroclimate Variability in the Southern Appalachians. Glob. Chang. Biol. 2015, 21, 4627–4641. [Google Scholar] [CrossRef]
- Kannenberg, S.A.; Novick, K.A.; Alexander, M.R.; Maxwell, J.T.; Moore, D.J.P.; Phillips, R.P.; Anderegg, W.R.L. Linking Drought Legacy Effects across Scales: From Leaves to Tree Rings to Ecosystems. Glob. Chang. Biol. 2019, 25, 2978–2992. [Google Scholar] [CrossRef]
- Roibu, C.C.; Sfecla, V.; Mursa, A.; Ionita, M.; Nagavciuc, V.; Chiriloaei, F.; Lesan, I.; Popa, I. The Climatic Response of Tree Ring Width Components of Ash (Fraxinus Excelsior L.) and Common Oak (Quercus Robur L.) from Eastern Europe. Forests 2020, 11, 600. [Google Scholar] [CrossRef]
- Weigel, R.; Muffler, L.; Klisz, M.; Kreyling, J.; van der Maaten-Theunissen, M.; Wilmking, M.; van der Maaten, E. Winter Matters: Sensitivity to Winter Climate and Cold Events Increases towards the Cold Distribution Margin of European Beech (Fagus Sylvatica L.). J. Biogeogr. 2018, 45, 2779–2790. [Google Scholar] [CrossRef]
- Bosela, M.; Lukac, M.; Castagneri, D.; Sedmák, R.; Biber, P.; Carrer, M.; Konôpka, B.; Nola, P.; Nagel, T.A.; Popa, I.; et al. Contrasting Effects of Environmental Change on the Radial Growth of Co-Occurring Beech and Fir Trees across Europe. Sci. Total Environ. 2018, 615, 1460–1469. [Google Scholar] [CrossRef]
- Drobyshev, I.; Övergaard, R.; Saygin, I.; Niklasson, M.; Hickler, T.; Karlsson, M.; Sykes, M.T. Masting Behaviour and Dendrochronology of European Beech (Fagus Sylvatica L.) in Southern Sweden. For. Ecol. Manag. 2010, 259, 2160–2171. [Google Scholar] [CrossRef]
- Obladen, N.; Dechering, P.; Skiadaresis, G.; Tegel, W.; Keßler, J.; Höllerl, S.; Kaps, S.; Hertel, M.; Dulamsuren, C.; Seifert, T.; et al. Tree Mortality of European Beech and Norway Spruce Induced by 2018-2019 Hot Droughts in Central Germany. Agric. For. Meteorol. 2021, 307, 108482. [Google Scholar] [CrossRef]
- Popa, I.; Caisin, V. Răspunsul Comparativ al Fagului Şi Stejarului La Secetă În Rezervaţia Naturală Codrii (R. Moldova). Bucov. For. 2015, 15, 45–53. [Google Scholar]
- Rubio-Cuadrado, Á.; Camarero, J.J.; del Río, M.; Sánchez-González, M.; Ruiz-Peinado, R.; Bravo-Oviedo, A.; Gil, L.; Montes, F. Long-Term Impacts of Drought on Growth and Forest Dynamics in a Temperate Beech-Oak-Birch Forest. Agric. For. Meteorol. 2018, 259, 48–59. [Google Scholar] [CrossRef]
- Liese, R.; Leuschner, C.; Meier, I.C. The Effect of Drought and Season on Root Life Span in Temperate Arbuscular Mycorrhizal and Ectomycorrhizal Tree Species. J. Ecol. 2019, 107, 2226–2239. [Google Scholar] [CrossRef]
- Fritts, H.C. Tree Rings and Climate; Academic Press: London, UK, 1976. [Google Scholar]
- Brázdil, R.; Stepánková, P.; Kyncl, T.; Kyncl, J. Fir Tree-Ring Reconstruction of March-July Precipitation in Southern Moravia (Czech Republic), 1376–1996. Clim. Res. 2002, 20, 223–239. [Google Scholar] [CrossRef][Green Version]
- Wilson, R.J.S.; Luckman, B.H.; Esper, J. A 500 Year Dendroclimatic Reconstruction of Spring-Summer Precipitation from the Lower Bavarian Forest Region, Germany. Int. J. Climatol. 2005, 25, 611–630. [Google Scholar] [CrossRef]
- Büntgen, U.; Brázdil, R.; Frank, D.; Esper, J. Three Centuries of Slovakian Drought Dynamics. Clim. Dyn. 2010, 35, 315–329. [Google Scholar] [CrossRef]
- Büntgen, U.; Brázdil, R.; Dobrovolný, P.; Trnka, M.; Kyncl, T. Five Centuries of Southern Moravian Drought Variations Revealed from Living and Historic Tree Rings. Theor. Appl. Climatol. 2011, 105, 167–180. [Google Scholar] [CrossRef]
- Tumajer, J.; Begović, K.; Čada, V.; Jenicek, M.; Lange, J.; Mašek, J.; Kaczka, R.J.; Rydval, M.; Svoboda, M.; Vlček, L.; et al. Ecological and Methodological Drivers of Non-Stationarity in Tree Growth Response to Climate. Glob. Chang. Biol. 2022, 1–15. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Chang; Shukla, P.R., Skea, J., Slade, R., Khourdajie, A.A., van Diemen, R., McCollum, D., Pathak, M., Some, S., Vyas, P., Fradera, R., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2022. [Google Scholar]
- Yuan, W.; Zheng, Y.; Piao, S.; Ciais, P.; Lombardozzi, D.; Wang, Y.; Ryu, Y.; Chen, G.; Dong, W.; Hu, Z.; et al. Increased Atmospheric Vapor Pressure Deficit Reduces Global Vegetation Growth. Sci. Adv. 2019, 5, eaax1396. [Google Scholar] [CrossRef]
- Balting, D.F.; AghaKouchak, A.; Lohmann, G.; Ionita, M. Northern Hemisphere Drought Risk in a Warming Climate. NPJ Clim. Atmos. Sci. 2021, 4, e2020JC017127. [Google Scholar] [CrossRef]
- Will, R.E.; Wilson, S.M.; Zou, C.B.; Hennessey, T.C. Increased Vapor Pressure Deficit Due to Higher Temperature Leads to Greater Transpiration and Faster Mortality during Drought for Tree Seedlings Common to the Forest–Grassland Ecotone. New Phytol. 2013, 200, 366–374. [Google Scholar] [CrossRef]
- ICAS. Raport Privind Starea Fondului Forestier Şi Rezultatele Activităţii Agenţiei “Moldsilva” În Perioada 2010–2015; ICAS: Chișinau, Moldova, 2016. [Google Scholar]
- Euforgen Distribution Map of Beech (Fagus Sylvatica). Available online: http://www.euforgen.org (accessed on 24 October 2022).
- Fang, J.; Lechowicz, M.J. Climatic Limits for the Present Distribution of Beech (Fagus L.) Species in the World. J. Biogeogr. 2006, 33, 1804–1819. [Google Scholar] [CrossRef]
- Gärtner, H.; Nievergelt, D. The Core-Microtome: A New Tool for Surface Preparation on Cores and Time Series Analysis of Varying Cell Parameters. Dendrochronologia 2010, 28, 85–92. [Google Scholar] [CrossRef]
- Gärtner, H.; Schweingruber, F. Microscopic Preparation Techniques for Plant Stem Analysis; Verlag Dr. Kessel: Remagen-Oberwinter, Germany, 2013; ISBN 9783941300767. [Google Scholar]
- Rinn, F. TSAP-Win User Reference; Rinntech: Heidelberg, Germany, 2003. [Google Scholar]
- Holmes, R.L. Computer-Assisted Quality Control in Tree-Ring Dating and Measurement. Tree-Ring Bull. 1983, 43, 69–75. [Google Scholar]
- Bunn, A.G. A Dendrochronology Program Library in R (DplR). Dendrochronologia 2008, 26, 115–124. [Google Scholar] [CrossRef]
- Cook, E.R.; Peters, K. Calculating Unbiased Tree-Ring Indices for the Study of Climatic and Environmental Change. Holocene 1997, 7, 361–370. [Google Scholar] [CrossRef]
- Frank, D.; Esper, J.; Cook, E.R. Adjustment for Proxy Number and Coherence in a Large-Scale Temperature Reconstruction. Geophys. Res. Lett. 2007, 34, L16709. [Google Scholar] [CrossRef]
- Wigley, T.M.L.; Briffa, K.R.; Jones, P.D. On the Average Value of Correlated Time Series, with Applications in Dendroclimatology and Hydrometeorology. J. Clim. Appl. Meteorol. 1984, 23, 201–213. [Google Scholar] [CrossRef]
- Eckstein, D.; Bauch, J. Beitrag Zur Rationalisierung Eines Dendrochronologischen Verfahrens Und Zur Analyse Seiner Aussagesicherheit. Forstwiss. Cent. 1969, 88, 230–250. [Google Scholar] [CrossRef]
- Baillie, M.G.L.; Pilcher, J.R. A simple crossdating program for tree-ring research. Tree Ring Bull. 1973, 33, 7–14. [Google Scholar]
- Abatzoglou, J.T.; Dobrowski, S.Z.; Parks, S.A.; Hegewisch, K.C. TerraClimate, a High-Resolution Global Dataset of Monthly Climate and Climatic Water Balance from 1958–2015. Sci. Data 2018, 5, 170191. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Beguería, S.; López-Moreno, J.I. A Multiscalar Drought Index Sensitive to Global Warming: The Standardized Precipitation Evapotranspiration Index. J. Clim. 2010, 23, 1696–1718. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Beguería, S.; López-Moreno, J.I.; Angulo, M.; Kenawy, A.E.L. A New Global 0.5° Gridded Dataset (1901–2006) of a Multiscalar Drought Index: Comparison with Current Drought Index Datasets Based on the Palmer Drought Severity Index. J. Hydrometeorol. 2010, 11, 1033–1043. [Google Scholar] [CrossRef]
- Cook, B.I.; Smerdon, J.E.; Cook, E.R.; Williams, A.P.; Anchukaitis, K.J.; Mankin, J.S.; Allen, K.; Andreu-Hayles, L.; Ault, T.R.; Belmecheri, S.; et al. Megadroughts in the Common Era and the Anthropocene. Nat. Rev. Earth Environ. 2022, 3, 741–757. [Google Scholar] [CrossRef]
- Ionita, M.; Nagavciuc, V. Changes in Drought Features at the European Level over the Last 120 Years. Nat. Hazards Earth Syst. Sci. 2021, 21, 1685–1701. [Google Scholar] [CrossRef]
- Rebetez, M.; Mayer, H.; Dupont, O.; Schindler, D.; Gartner, K.; Kropp, J.P.; Menzel, A. Heat and Drought 2003 in Europe: A Climate Synthesis. Ann. For. Sci. 2006, 63, 569–577. [Google Scholar] [CrossRef]
- Bakke, S.J.; Ionita, M.; Tallaksen, L.M. The 2018 Northern European Hydrological Drought and Its Drivers in a Historical Perspective. Hydrol. Earth Syst. Sci. 2020, 24, 5621–5653. [Google Scholar] [CrossRef]
- Laaha, G.; Gauster, T.; Tallaksen, L.M.; Vidal, J.P.; Stahl, K.; Prudhomme, C.; Heudorfer, B.; Vlnas, R.; Ionita, M.; van Lanen, H.A.J.; et al. The European 2015 Drought from a Hydrological Perspective. Hydrol. Earth Syst. Sci. 2017, 21, 3001–3024. [Google Scholar] [CrossRef]
- Zang, C.; Biondi, F. Treeclim: An R Package for the Numerical Calibration of Proxy-Climate Relationships. Ecography 2015, 38, 431–436. [Google Scholar] [CrossRef]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer Publishing Company, Incorporated: Berlin/Heidelberg, Germany, 2009; ISBN 0387981403. [Google Scholar]
- Ionita, M.; Lohmann, G.; Rimbu, N. Prediction of Spring Elbe Discharge Based on Stable Teleconnections with Winter Global Temperature and Precipitation. J. Clim. 2008, 21, 6215–6226. [Google Scholar] [CrossRef]
- Ionita, M. Mid Range Forecasting of the German Waterways Streamflow Based on Hydrologic, Atmospheric and Oceanic Data; Bornemann, H., Ed.; Alfred-Wegener-Institut Helmholtz-Zentrum für Polar- und Meeresforschung: Bremerhaven, Germany, 2017. [Google Scholar]
- Nagavciuc, V.; Ionita, M.; Perșoiu, A.; Popa, I.; Loader, N.J.; McCarroll, D. Stable Oxygen Isotopes in Romanian Oak Tree Rings Record Summer Droughts and Associated Large-Scale Circulation Patterns over Europe. Clim. Dyn. 2019, 52, 6557–6568. [Google Scholar] [CrossRef]
- Nagavciuc, V.; Kern, Z.; Ionita, M.; Hartl, C.; Konter, O.; Esper, J.; Popa, I.; Ionel, P.; Popa, I. Climate Signals in Carbon and Oxygen Isotope Ratios of Pinus Cembra Tree-Ring Cellulose from the Călimani Mountains, Romania. Int. J. Climatol. 2020, 40, 2539–2556. [Google Scholar] [CrossRef]
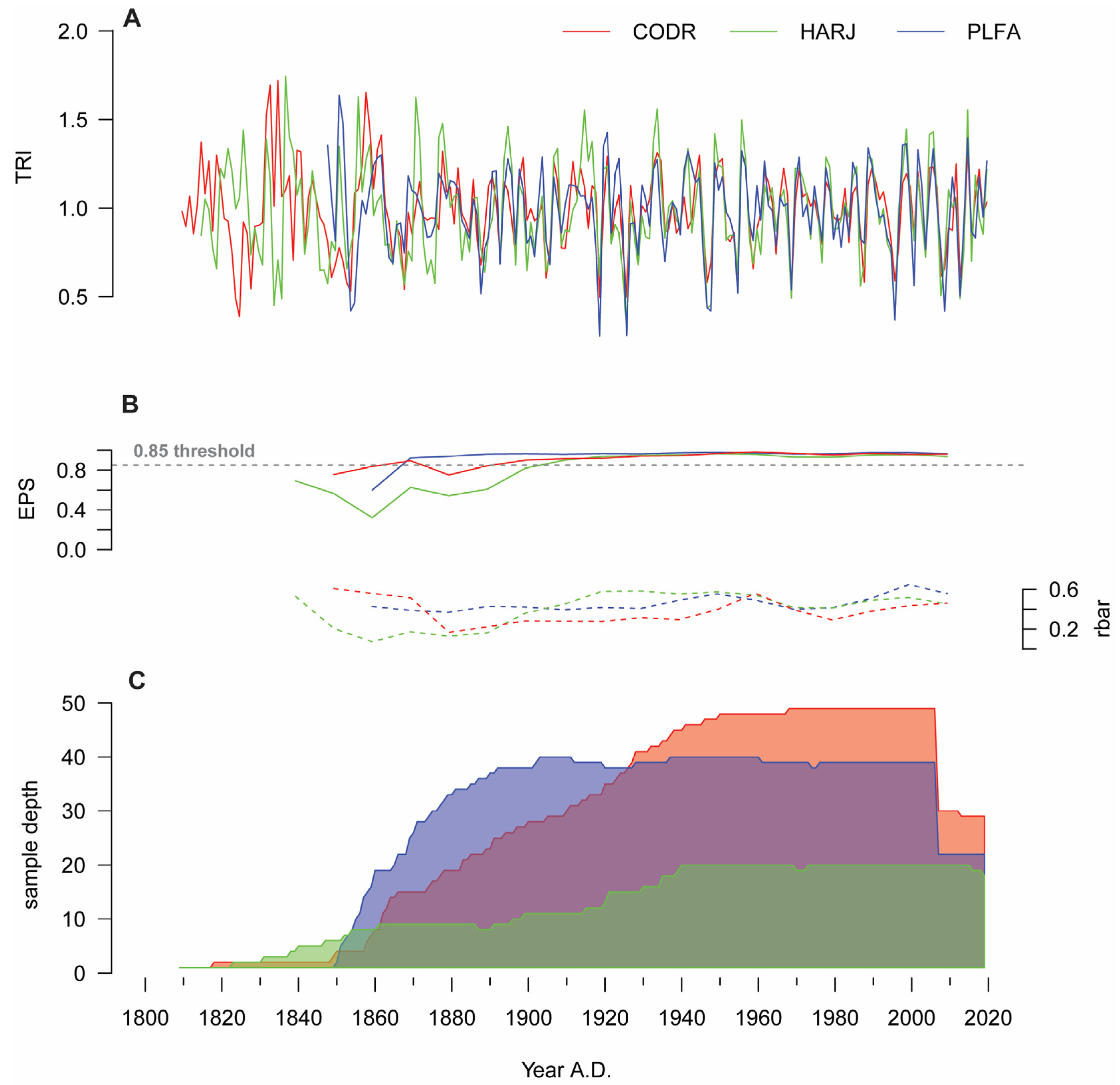
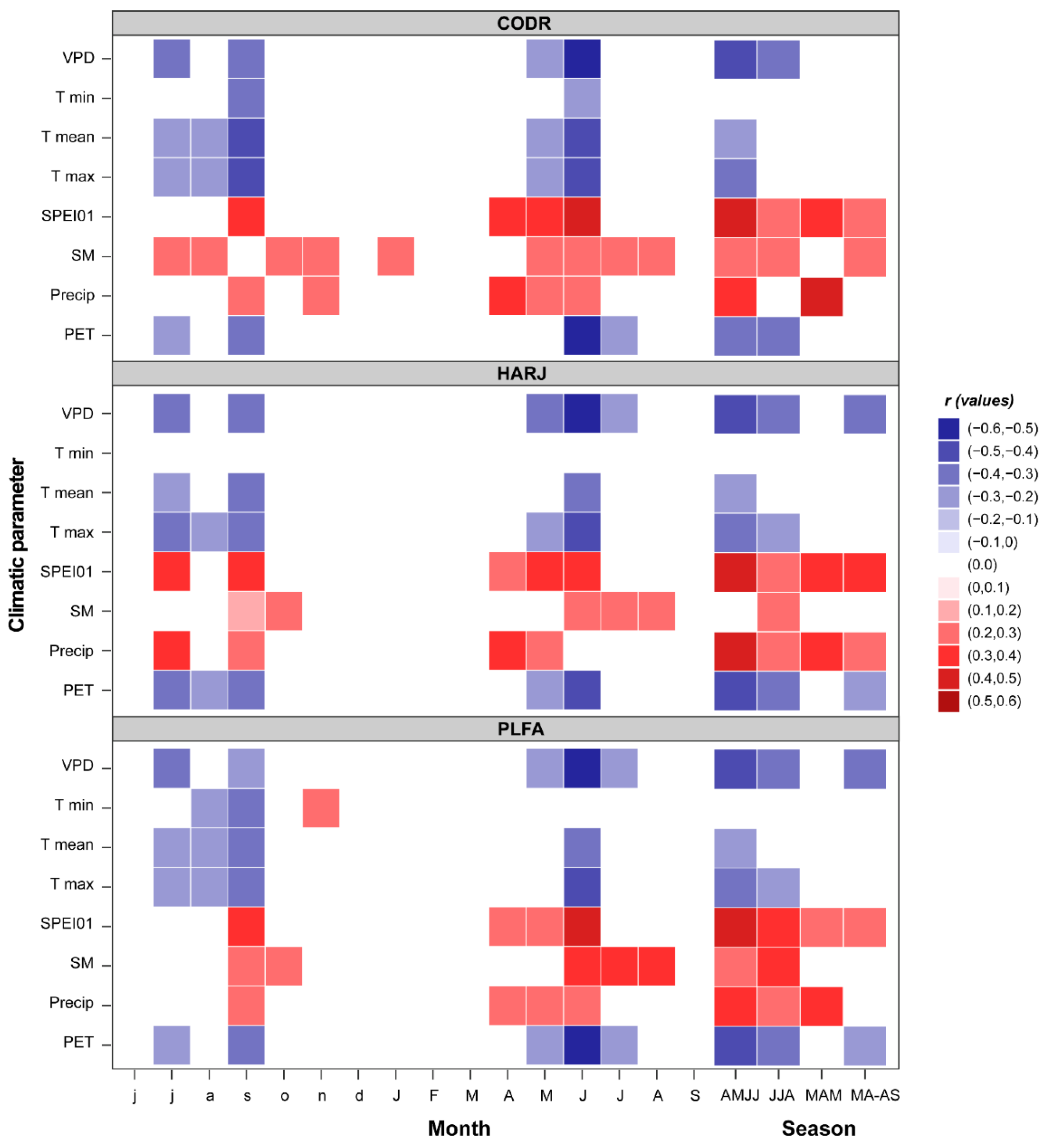

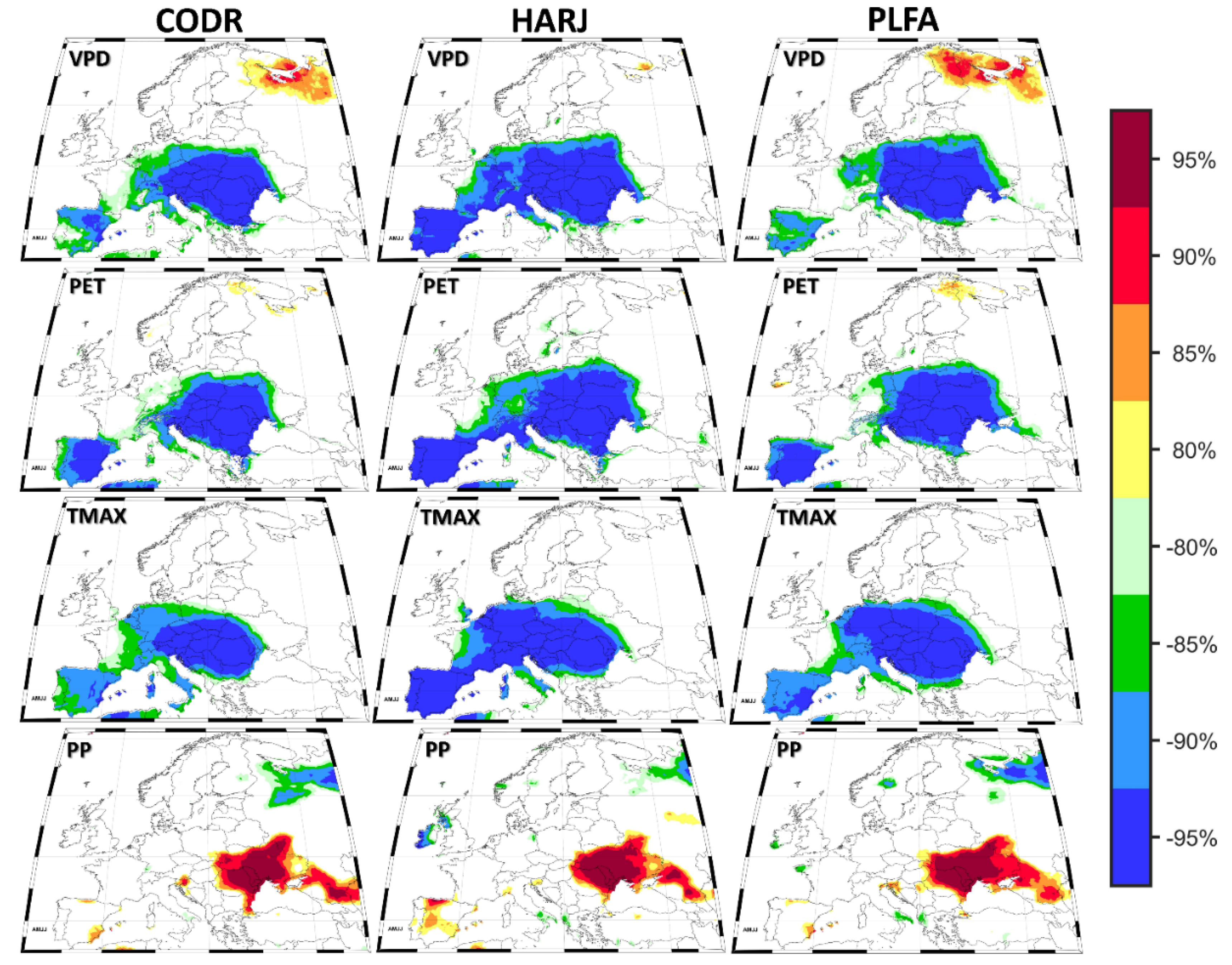
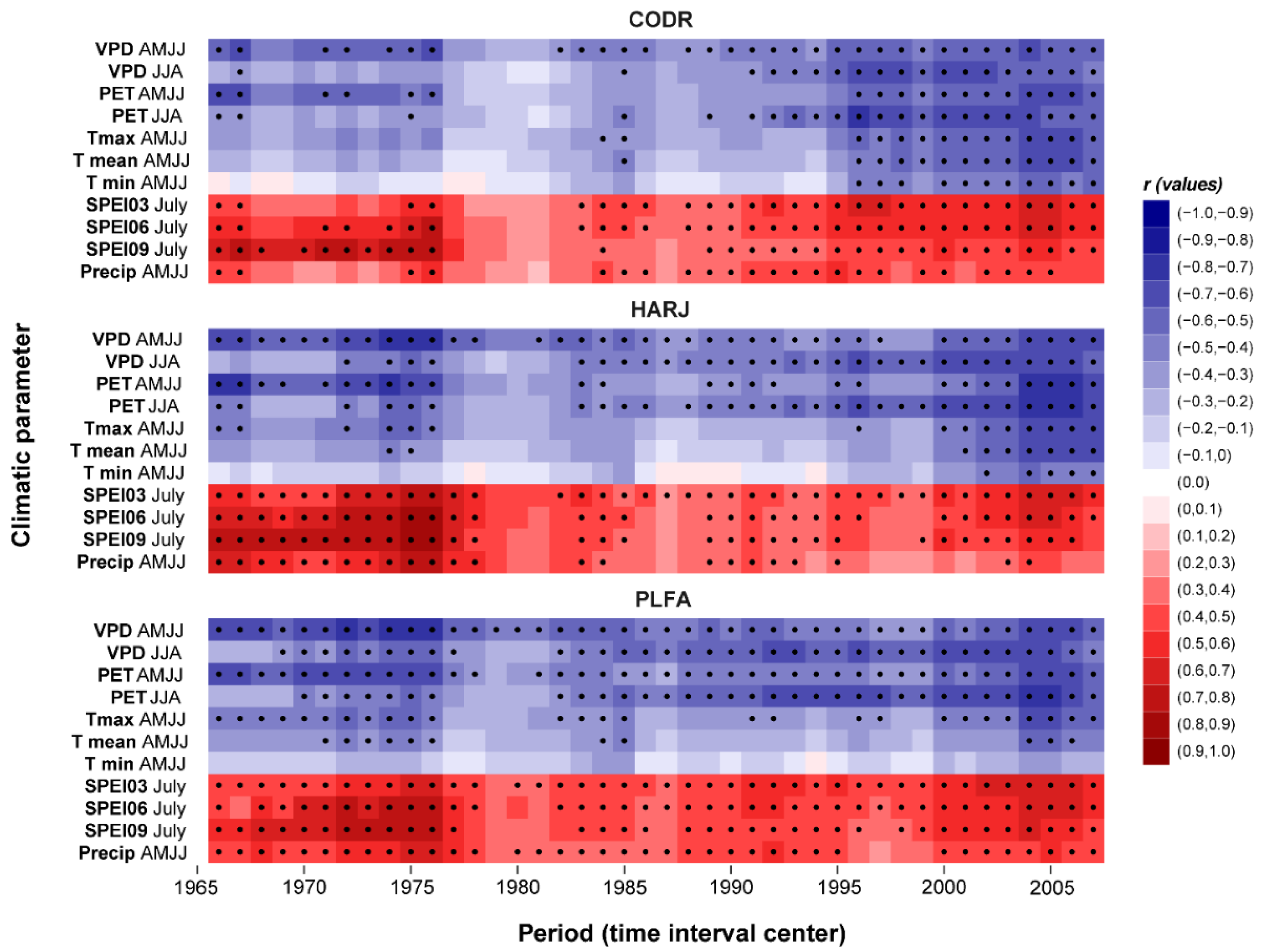
| Site | Raw Data | Detrended Data | ||||||
|---|---|---|---|---|---|---|---|---|
| Time Span | MSL ± SD | MGR ± SD | AC1 | rbar | EPS | MS | SNR | |
| CODR | 1809–2019 | 121 ± 30 | 2.56 ± 0.53 | 0.58 | 0.364 | 0.966 | 0.33 | 28.1 |
| HARJ | 1814–2019 | 122 ± 49 | 2.68 ± 0.61 | 0.57 | 0.453 | 0.948 | 0.42 | 18.22 |
| PLFA | 1847–2019 | 135 ± 35 | 2.23 ± 0.74 | 0.58 | 0.452 | 0.972 | 0.35 | 34.64 |
| Code | Location | Number of Samples | Latitude (Degrees) | Longitude (Degrees) | Altitude (m a.s.l) |
|---|---|---|---|---|---|
| CODR | Codrii | 49 | 47.10 N | 28.32 E | 244 |
| HARJ | Hîrjauca | 22 | 47.31 N | 28.23 E | 250 |
| PLFA | Plaiul Fagului | 43 | 47.28 N | 28.02 E | 225 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roibu, C.-C.; Palaghianu, C.; Nagavciuc, V.; Ionita, M.; Sfecla, V.; Mursa, A.; Crivellaro, A.; Stirbu, M.-I.; Cotos, M.-G.; Popa, A.; et al. The Response of Beech (Fagus sylvatica L.) Populations to Climate in the Easternmost Sites of Its European Distribution. Plants 2022, 11, 3310. https://doi.org/10.3390/plants11233310
Roibu C-C, Palaghianu C, Nagavciuc V, Ionita M, Sfecla V, Mursa A, Crivellaro A, Stirbu M-I, Cotos M-G, Popa A, et al. The Response of Beech (Fagus sylvatica L.) Populations to Climate in the Easternmost Sites of Its European Distribution. Plants. 2022; 11(23):3310. https://doi.org/10.3390/plants11233310
Chicago/Turabian StyleRoibu, Cătălin-Constantin, Ciprian Palaghianu, Viorica Nagavciuc, Monica Ionita, Victor Sfecla, Andrei Mursa, Alan Crivellaro, Marian-Ionut Stirbu, Mihai-Gabriel Cotos, Andrei Popa, and et al. 2022. "The Response of Beech (Fagus sylvatica L.) Populations to Climate in the Easternmost Sites of Its European Distribution" Plants 11, no. 23: 3310. https://doi.org/10.3390/plants11233310
APA StyleRoibu, C.-C., Palaghianu, C., Nagavciuc, V., Ionita, M., Sfecla, V., Mursa, A., Crivellaro, A., Stirbu, M.-I., Cotos, M.-G., Popa, A., Sfecla, I., & Popa, I. (2022). The Response of Beech (Fagus sylvatica L.) Populations to Climate in the Easternmost Sites of Its European Distribution. Plants, 11(23), 3310. https://doi.org/10.3390/plants11233310










