Photosynthetic Apparatus of Hydrocharis morsus-ranae in Different Solar Lighting
Abstract
1. Introduction
2. Results
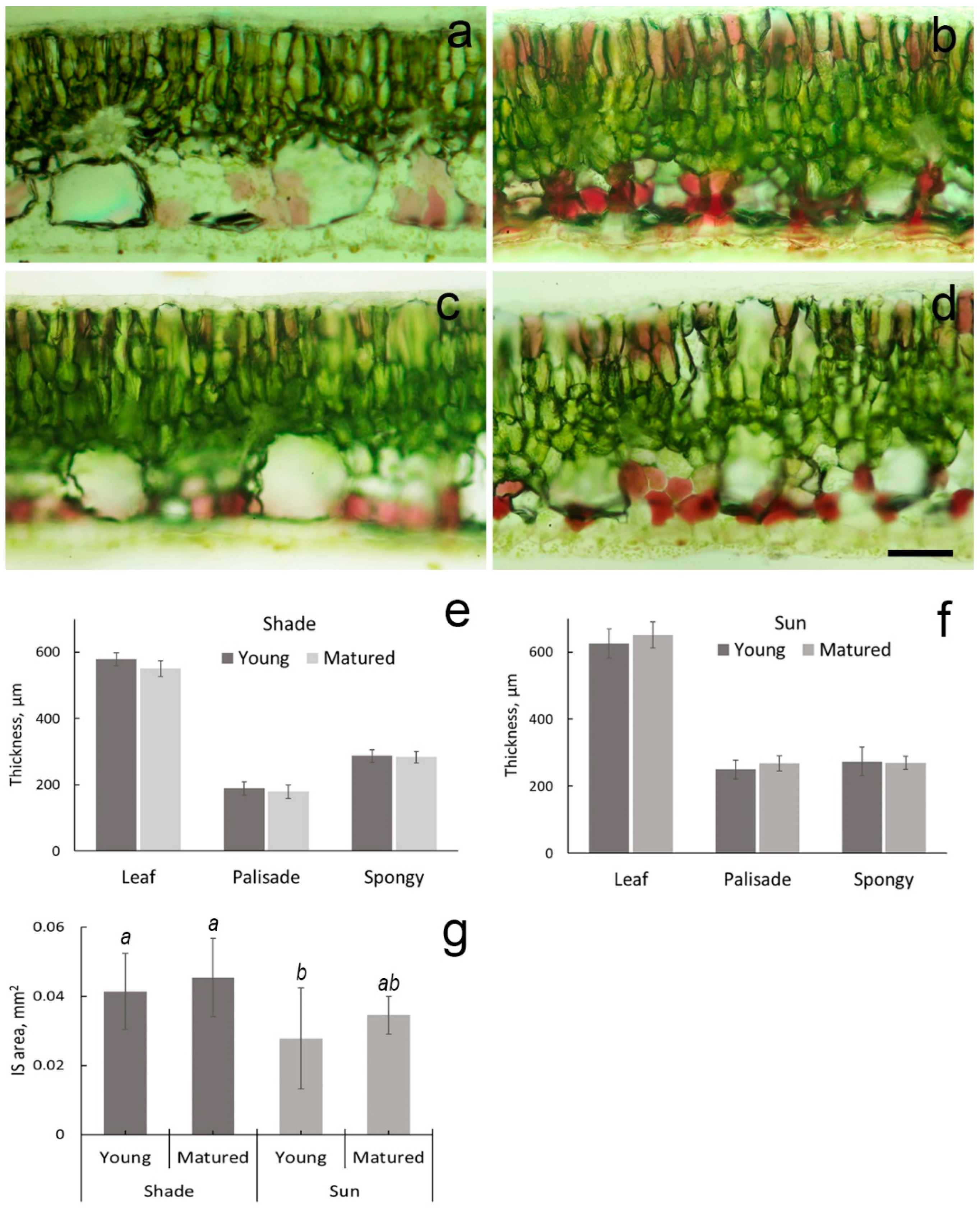
2.1. Leaf Micromorphology
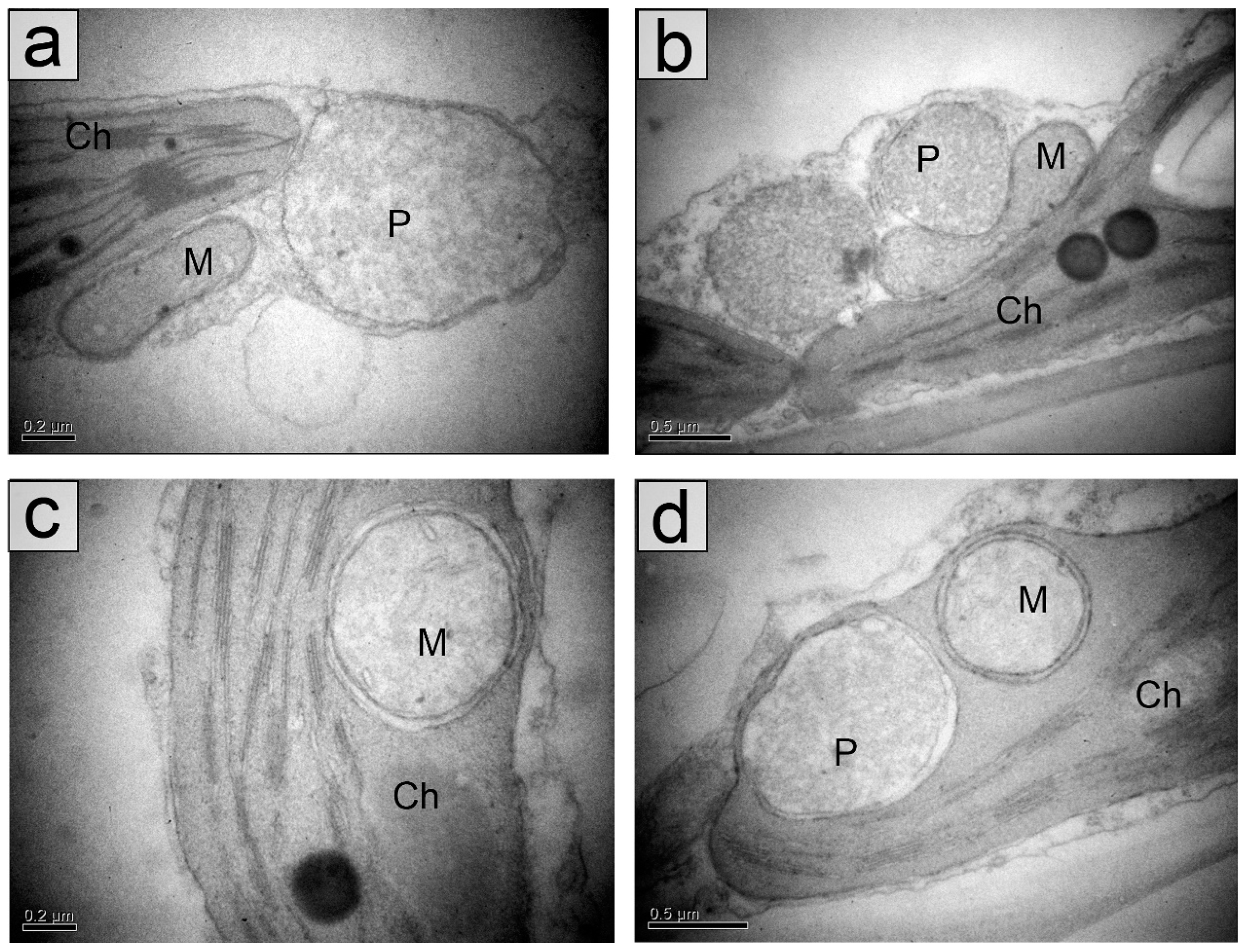
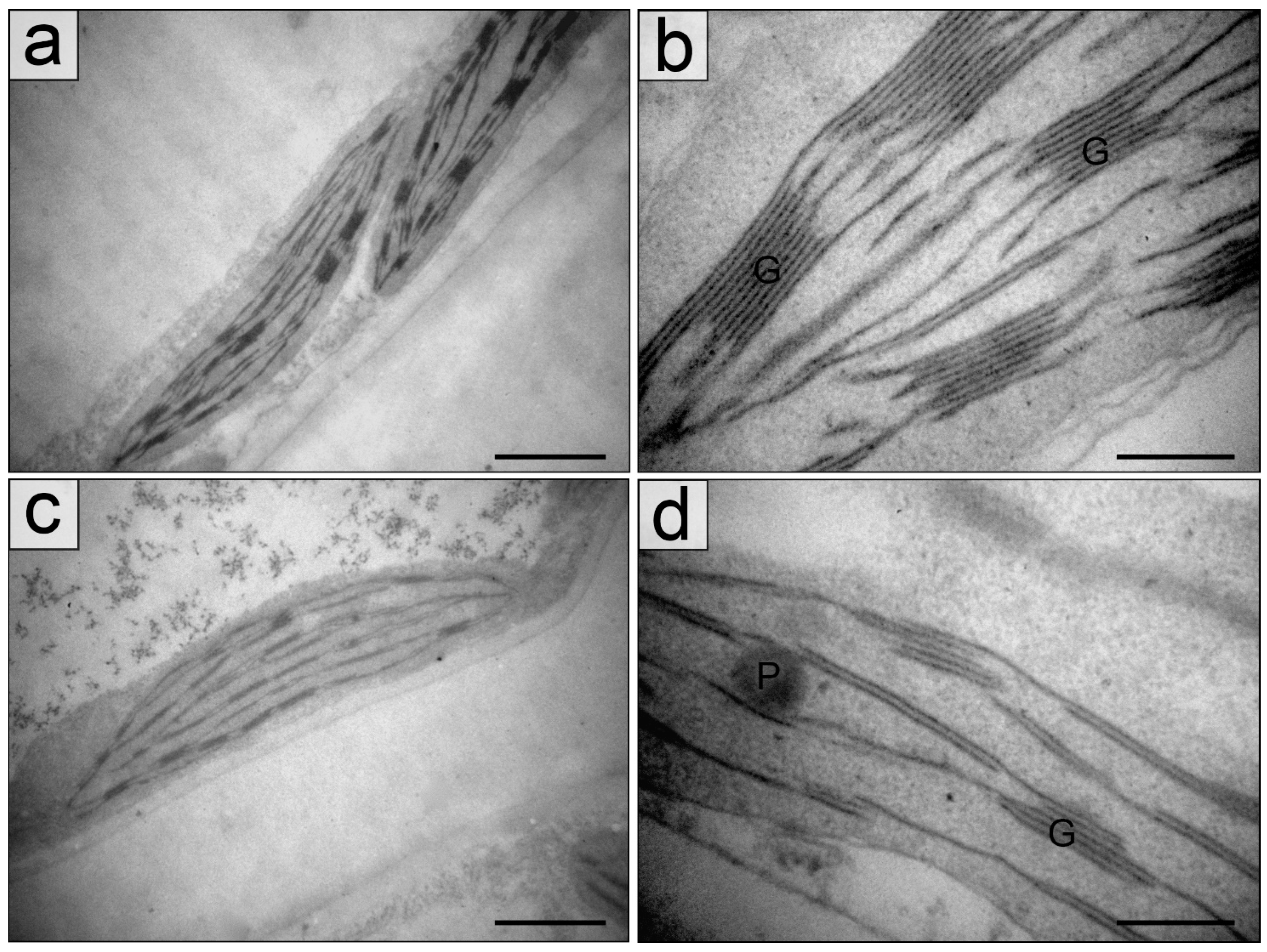
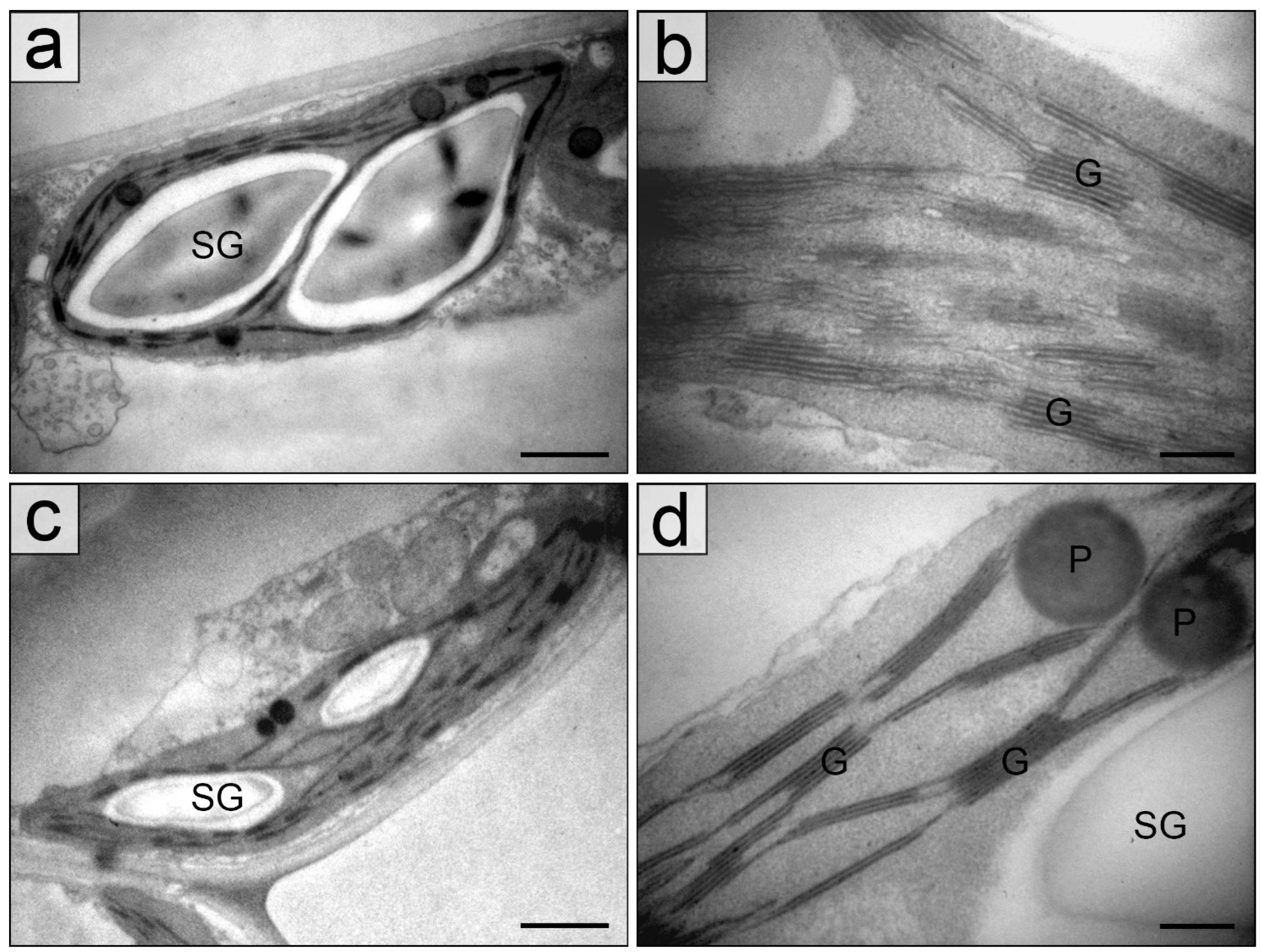
2.2. Chloroplasts in the Palisade Parenchyma Cells
2.3. Photosynthetic Pigments
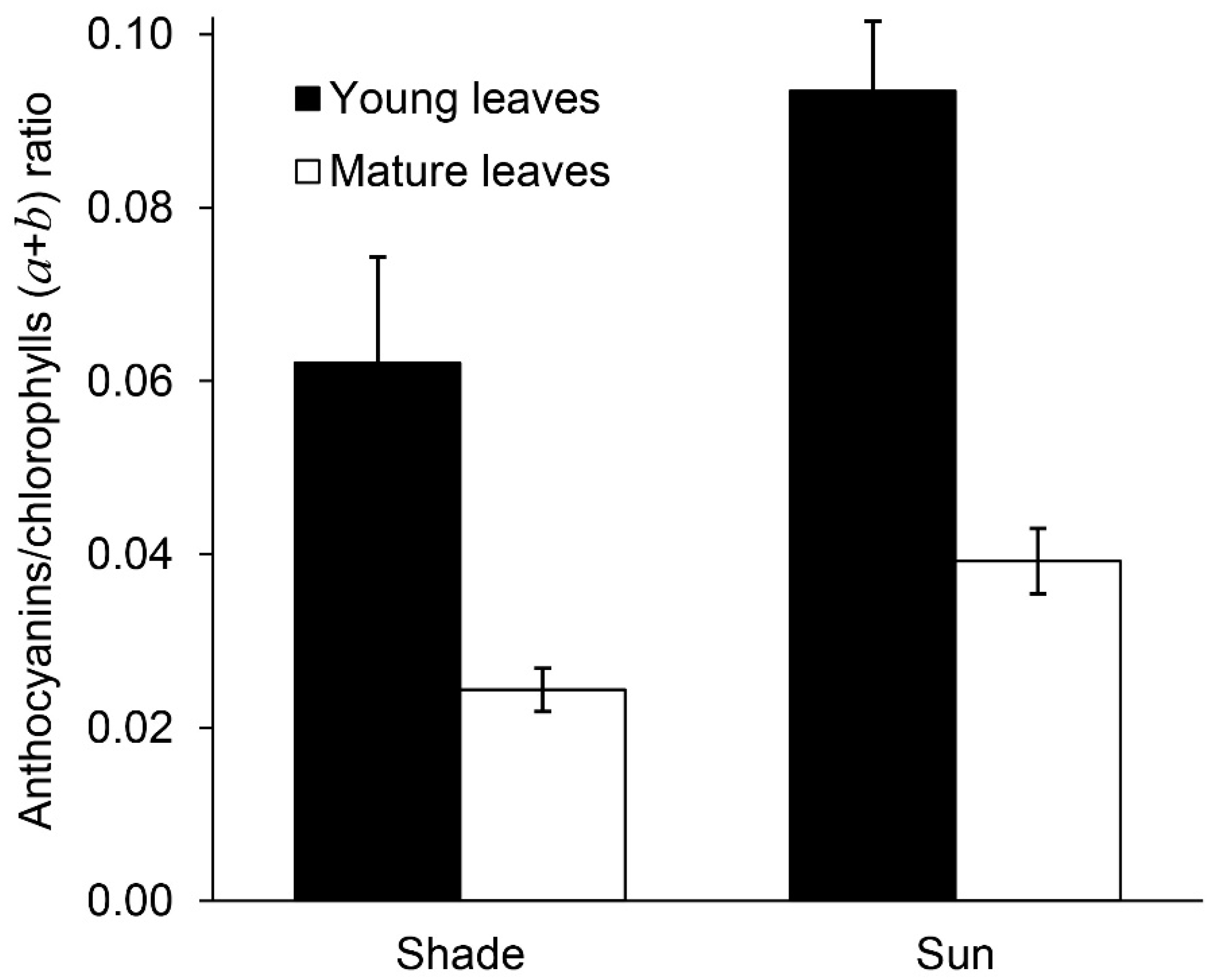
2.4. Anthocyanins and Phenylpropanoids
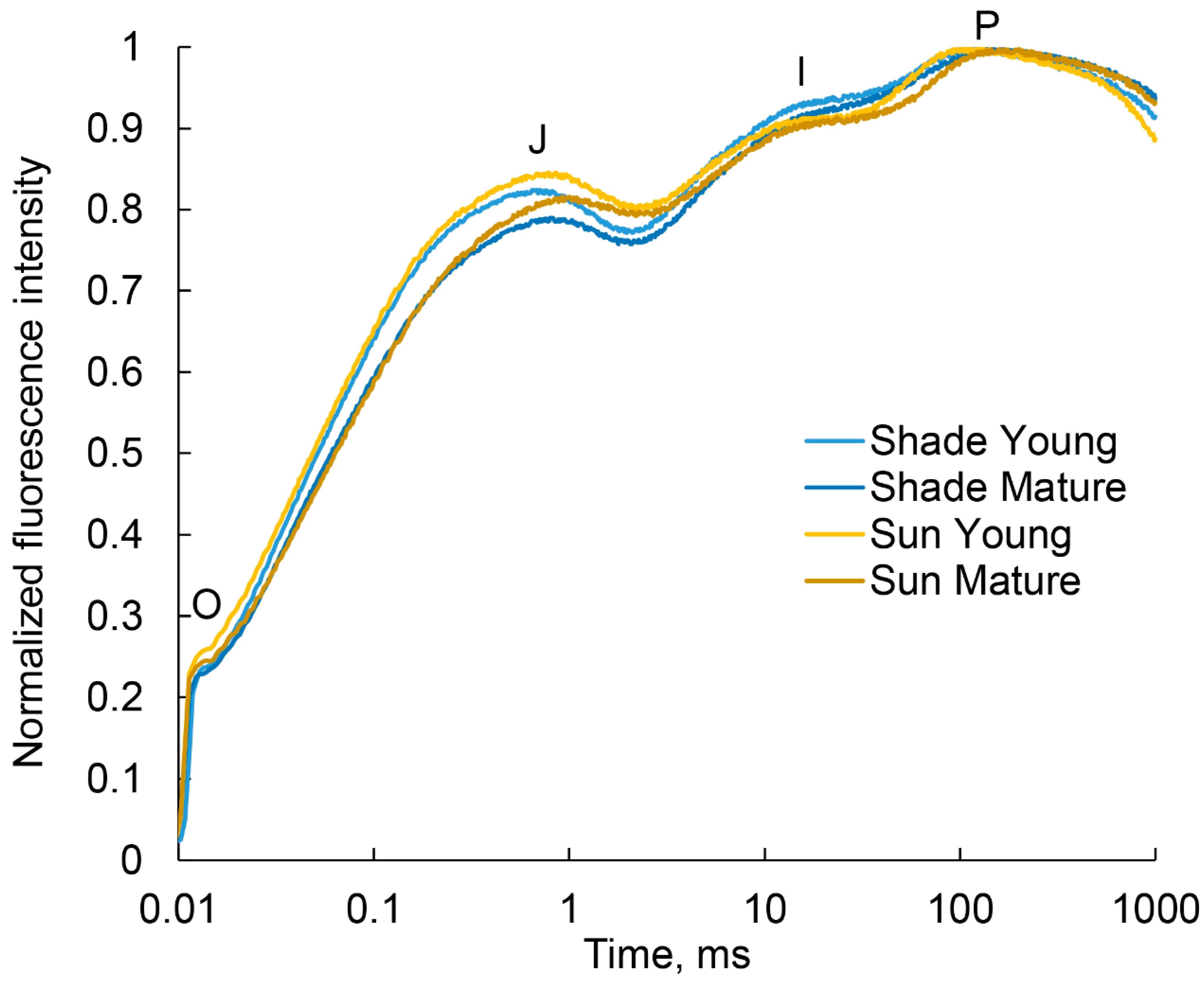
2.5. Analysis of the Photosynthetic Apparatus State by Quantum Yields of Electron Transport
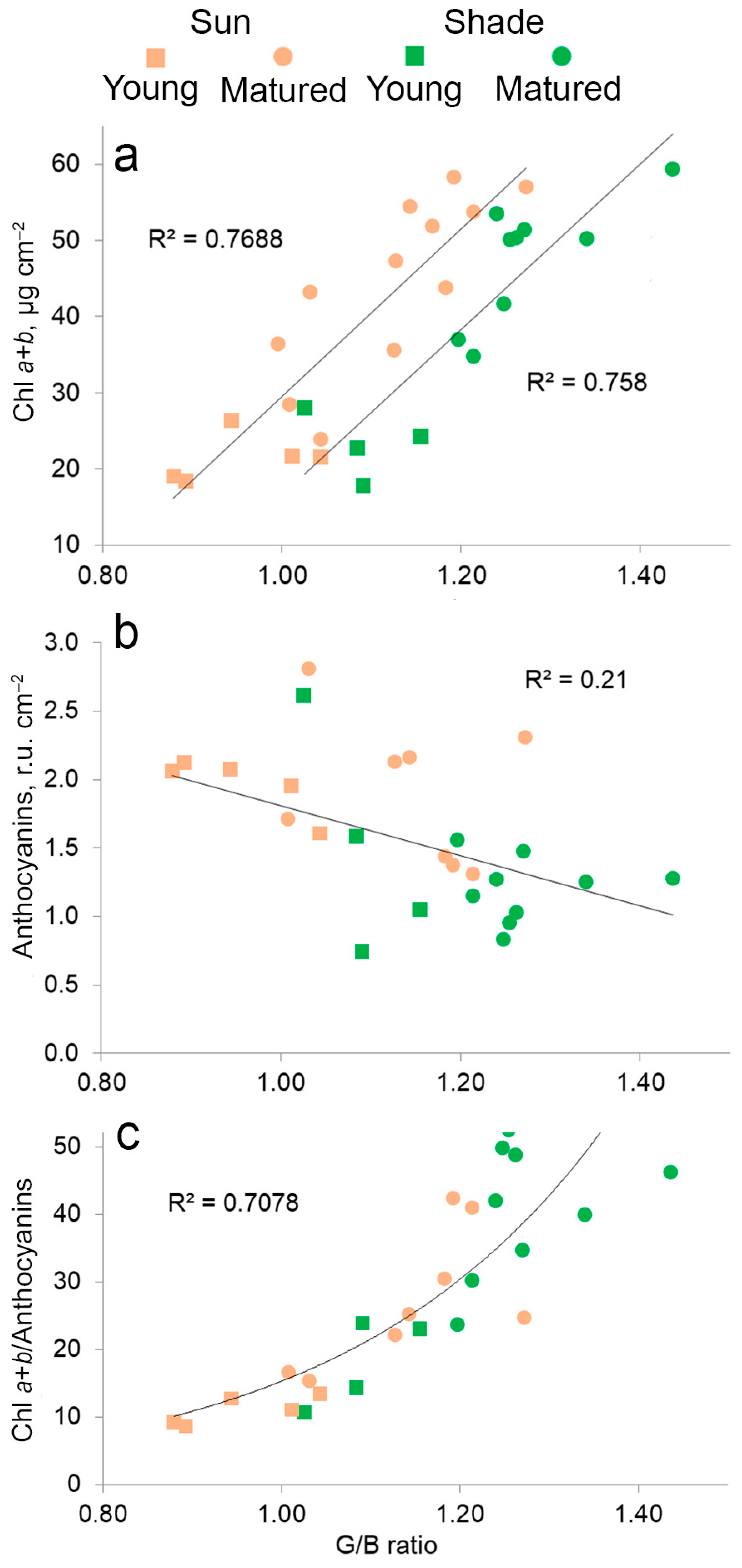
2.6. The Relationship between the Content of Pigments, Anthocyanins and Color in Leaves in Different Lighting
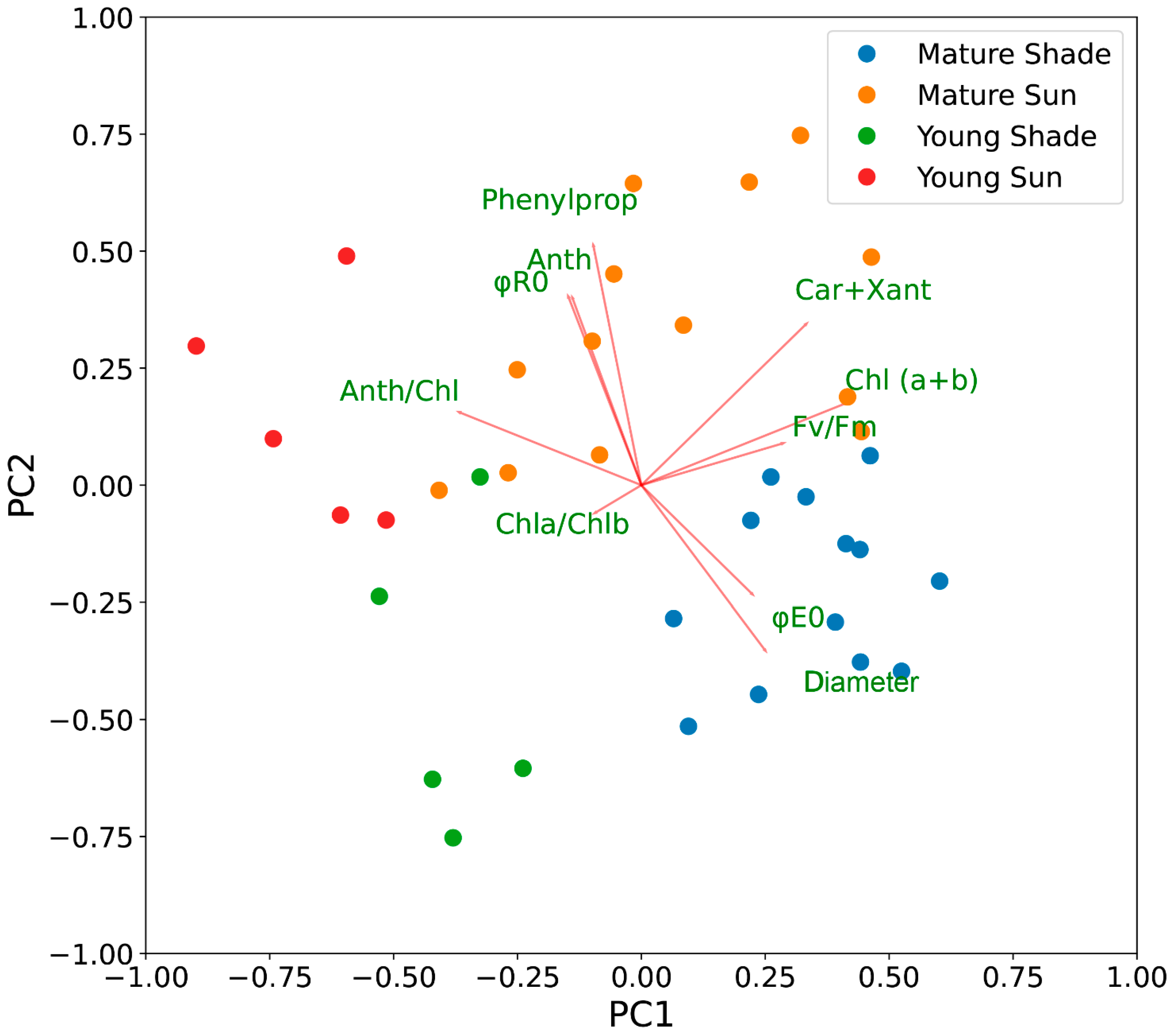
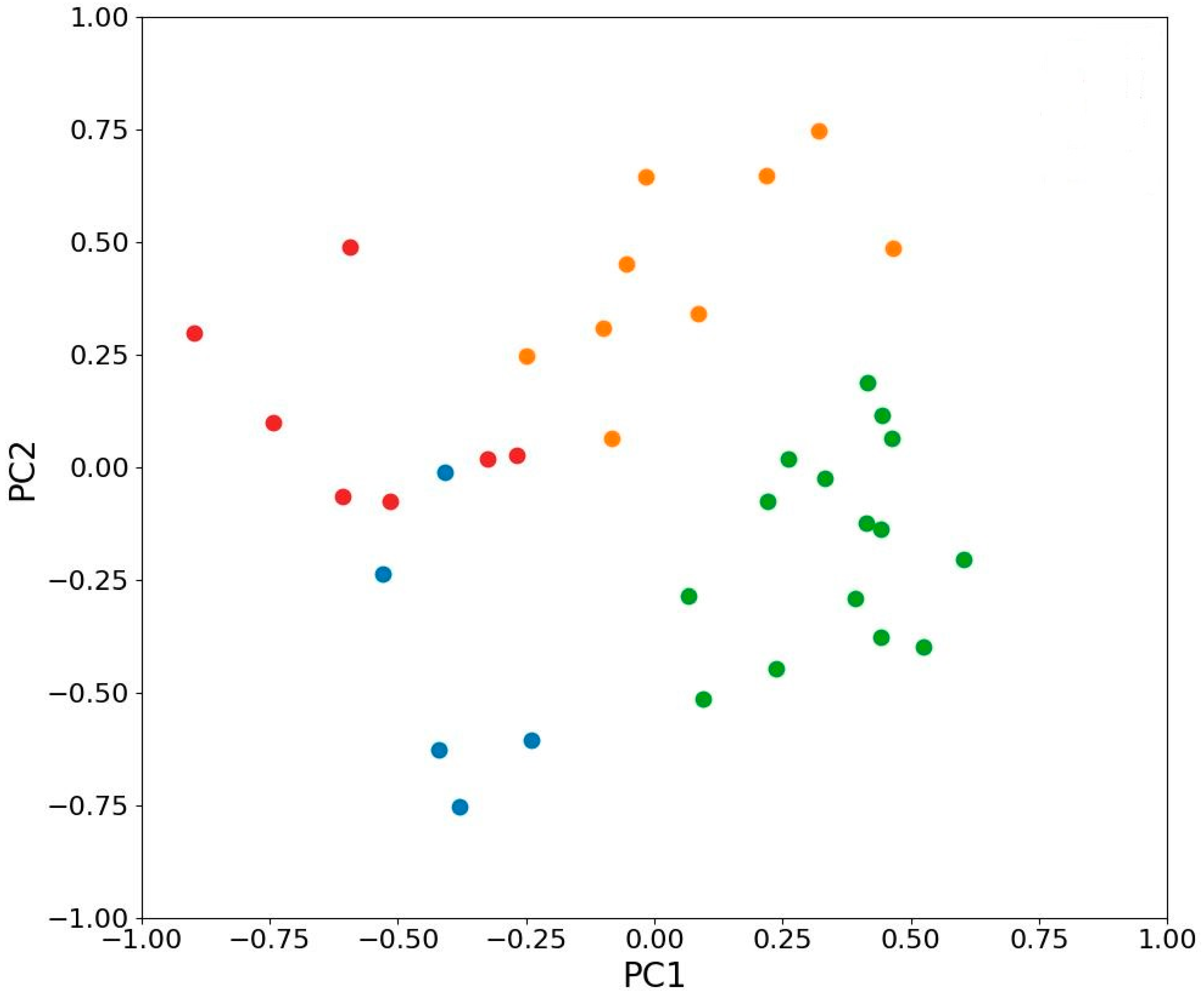
2.7. Principal Component Analysis of the Variability of Photosynthetic Traits
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Light Microscopy
4.3. Transmission Electron Microscopy
4.4. Quantification of Leaf Blade and Chloroplast Structure
4.5. Leaf Color Analysis
4.6. Anthocyanins and Phenylpropanoids Analyses
4.7. Pigment Analysis
4.8. Chlorophyll a Fluorescence Induction and JIP-Test
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cook, C.D.K.; Luond, R. A revision of the genus Hydrocharis (Hydrocharitaceae). Aquat. Bot. 1982, 14, 177–204. [Google Scholar] [CrossRef]
- Scribailo, R.W.; Posluszny, U. The reproductive biology of Hydrocharis morsus-ranae L. Floral biology. Canad. J. Bot. 1984, 62, 2779–2787. [Google Scholar] [CrossRef]
- Catling, P.M.; Miltrow, G.; Haber, E.; Posluszny, U.; Charlton, W.A. The biology of Canadian weeds Hydrocharis morsus-ranae L. Can. J. Plant Sci. 2003, 83, 1001–1016. [Google Scholar] [CrossRef]
- Toma, C. Reproduction of Hydrocharis morsus-ranae taxa in an oxbow lake of the River Vistula. Limnol. Rev. 2013, 3, 171–179. [Google Scholar] [CrossRef]
- Zhu, B.; Ottaviani, C.C.; Naddafi, R.; Dai, Z.; Du, D. Invasive European frogbit (Hydrocharis morsus-ranae L.) in North America: An updated review 2003–2016. J. Plant Ecol. 2018, 11, 17–25. [Google Scholar] [CrossRef]
- Cutter, E.G.; Feldman, L.J. Trichoblasts in Hydrocharis. I. Origin, differentiation, dimensions and growth. Am. J. Bot. 1970, 57, 190–201. [Google Scholar] [CrossRef]
- Seago, J.L.; Marsh, L.C.; Stevens, K.J.; Soucup, A.; Votrubova, O.; Enstone, D.E. A re-examination of the root cortex in wetland flowering plants with respect to aerenchyma. Ann. Bot. 2005, 96, 565–579. [Google Scholar] [CrossRef]
- Tsyrenova, D.J.; Varfolomeeva, A.S.; Goroshko, J.M. Micromorphology of Hydrocharis dubia (Blume) Backer, L. (Hydrocharitaceae) plants from the Far East of Russia.). Inland Water Biol. 2018, 11, 264–268. [Google Scholar] [CrossRef]
- Efremov, A.N.; Grishina, V.S.; Toma, C.; Mesterhazy, A.; Tchatchouang, E.N. Comparative morphology of the aquatic plant, genus Hydrocharis morsus-ranae L. Can. J. Bot. 2021, 77, 113–121. [Google Scholar]
- Zhu, B.; Ellis, M.S.; Fancher, K.L.; Rudstam, L.G. Shading as a control method for invasive European frogbit (Hydrocharis morsus-ranae L.). PLoS ONE 2014, 9, e98488. [Google Scholar] [CrossRef]
- Gamulya, Y.G. Plants of Ukraine; Factor: Kharkiv, Ukraine, 2011; 210p. [Google Scholar]
- Pindel, Z.; Wozniak, L. Natural conditions for presence of some ornamental water and peatbog plants. Agricultura 1998, 70, 83–87. [Google Scholar]
- Kordyum, E.; Mosyakin, S.; Ivanenko, G.; Ovcharenko, Y.; Brykov, V. Hydropotes of young and mature leaves in Nuphar lutea and Nymphaea alba (Nymphaeaceae): Formation, functions and phylogeny. Aquatic Bot. 2021, 169, 103342. [Google Scholar] [CrossRef]
- Kordyum, E.L.; Bluma, D.A.; Ivanenko, G.F.; Kozeko, E.L.; Artemenko, O.A.; Vedenicheva, N.P. Plasticity of morphophysiological and oxidative metabolism patterns of Sium species (Apiaceae) at different soil moisture. J. Plant Physiol. Pathol. 2019, 7, 12. [Google Scholar]
- Kordyum, E.; Kozeko, L.; Ovcharenko, Y.; Brykov, V. Assessment of alcohol dehydrogenase synthesis and aerenchyma formation in the tolerance of Sium L. species (Apiaceae) to waterlogging. Aquat. Bot. 2017, 142, 71–77. [Google Scholar] [CrossRef]
- Miner, B.; Sultan, S.; Morgan, S.G.; Padilla, D.K.; Relyeae, R.A. Ecological consequences of phenotypic plasticity. Trend Ecol. Evol. 2006, 20, 686–692. [Google Scholar] [CrossRef]
- Dubyna, D.V.; Kordyum, E. Ontogenesis plasticity of vascular plants: Molecular, cellular, population and cenotic aspects. Visn. Nac. Akad. Nauk Ukr. 2015, 7, 32–40. [Google Scholar] [CrossRef]
- Lloyd, S.P. Least squares quantization in PCM. IEEE Transact Inform Theory 1982, 28, 129–137. [Google Scholar] [CrossRef]
- Sultan, S.E. Phenotypic plasticity for plant development, function and life history. Trends Plant Sci. 2000, 5, 537–542. [Google Scholar] [CrossRef]
- Sultan, S.E. Plant developmental responses to the environment: Eco-devo insights. Curr. Opin. Blant Biol. 2010, 13, 96–101. [Google Scholar] [CrossRef]
- Kordyum, E.L.; Sytnik, K.M.; Baranenko, V.V.; Belyavskaya, N.A.; Klymchuk, D.A.; Nedukha, E.M. Cellular Mechanisms of Plant Adaptation to Unfavourable Influences of Ecological Factors in Natural Conditions; Naukova Dumka: Kyiv, Ukraine, 2003; 273p. [Google Scholar]
- Kelly, S.A.; Panhuis, T.M.; Stoehr, A.M. Phenotypic Plasticity: Molecular Mechanisms and Adaptive Significance. Compr. Physiol. 2012, 2, 1417–1439. [Google Scholar]
- Bradshow, A.D. Evolutionary significance of phenotypic plasticity in plants. Adv. Genet. 1965, 13, 115–155. [Google Scholar]
- Frenkel, M.; Bellafiore, S.; Rochaix, J.-D.; Jansson, S. Hierarchy amongst photosynthetic acclimation responses for plant fitness. Physiol. Plant. 2007, 129, 455–459. [Google Scholar] [CrossRef]
- Kaiser, E.; Morales, A.; Harbinson, J.; Kromdijk, J.; Heuvelink, E.; Marcelis, L.F.M. Dynamic photosynthesis in different environmental conditions. J. Exp. Bot. 2015, 66, 2415–2426. [Google Scholar] [CrossRef] [PubMed]
- Schneider, T.; Bolger, A.; Zeier, J.; Preiskowski, S.; Benes, V.; Trenkamp, S.; Usadel, B.; Farré, E.M.; Matsubara, S. Fluctuating light interacts with time of day and leaf development stage to reprogram gene expression. Plant Physiol. 2019, 179, 1632–1657. [Google Scholar] [CrossRef] [PubMed]
- Morales, A.; Kaiser, E. Photosynthetic acclimation to fluctuating irradiance in plants. Front. Plant Sci. 2020, 11, 268. [Google Scholar] [CrossRef]
- Dietzel, L.; Pfannschmidt, T. Photosynthetic acclimation to light gradients in plant stands comes out of shade. Plant Signal. Behav. 2008, 3, 1116–1118. [Google Scholar] [CrossRef]
- Murchie, E.H.; Norton, P. Acclimation of photosynthesis to irradiance and spectral quality in British plant species: Chlorophyll content, photosynthetic capacity and habitat preference. Plant Cell Environ. 1997, 20, 438–448. [Google Scholar] [CrossRef]
- Yano, S.; Terashima, I. Separate localization of light signal perception for sun or shade type chloroplast and palisade tissue differentiation in Chenopodium album. Plant Cell Physiol. 2001, 42, 1303–1310. [Google Scholar] [CrossRef]
- Jia, H.; Liggins, J.R.; Chow, W.S. Acclimation of leaves to low light produces large grana: The origin of the predominant attractive force at work. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2012, 367, 3494–3502. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Buschmann, C.; Doll, M.; Fietz, H.-J.; Bach, T.; Kozel, U.; Meier, D.; Rahmsdorf, U. Photosynthetic activity, chloroplast ultrastructure, and leaf characteristics of high-light and low-light plants and of sun and shade leaves. Photosynthesis Res. 1981, 2, 115–141. [Google Scholar] [CrossRef]
- Pearcy, R.W.; Franceschp, V.R. Photosynthetic characteristics and chloroplast ultrastructure of C3 and C4 tree species grown in high- and low-light environments. Photosynthesis Res. 1986, 9, 317–331. [Google Scholar] [CrossRef]
- Boardman, N.K. Comparative photosynthesis of sun and shade plants. Annu. Rev. Plant Physiol. 2003, 28, 355–377. [Google Scholar] [CrossRef]
- Mathur, S.; Jain, L.; Jajoo, A. Photosynthetic efficiency in sun and shade plants. Photosynthetica 2018, 56, 354–365. [Google Scholar] [CrossRef]
- Yamori, W. Photosynthetic response to fluctuating environments and photoprotective strategies under abiotic stress. J. Plant Res. 2016, 129, 379–395. [Google Scholar] [CrossRef]
- Tian, Y.; Zhong, R.; Wei, J.; Luo, H.; Eyal, Y.; Jin, H.; Wu, L.; Liang, K.; Li, Y.; Chen, S.; et al. Arabidopsis CHLOROPHYLLASE 1 Protects young leaves from long-term photodamage by facilitating FtsH-mediated D1 degradation in photosystem II Repair. Molecular Plant 2021, 14, 1149–1167. [Google Scholar] [CrossRef]
- Cogdell, R.J.; Gardiner, A.T. Functions of carotenoids in photosynthesis. Methods Enzymol. 1993, 214, 185–193. [Google Scholar]
- Bartley, G.E.; Scolnik, P.A. Plant carotenoids: Pigments for photoprotection, visual attraction, and human health. Plant Cell 1995, 7, 1027–1038. [Google Scholar]
- Sun, T.; Rao, S.; Zhou, X.; Li, L. Plant carotenoids: Recent advances and future perspectives. Molec. Horticult. 2022, 2, 1–21. [Google Scholar] [CrossRef]
- Krause, G.H.; Virgo, A.; Winter, K. High susceptibility to photoinhibition of young leaves of tropical forest trees. Planta 1995, 197, 591–683. [Google Scholar] [CrossRef]
- Kržič, N.Š.; Gaberščik, A. Photochemical efficiency of amphibious plants in an intermittent lake. Aqua. Bot. 2005, 83, 281–288. [Google Scholar] [CrossRef]
- Stirbet, A.; Lazár, D.; Kromdijk, J. Chlorophyll a fluorescence induction: Can just a one-second measurement be used to quantify abiotic stress responses? Photosynthetica 2018, 56, 86–104. [Google Scholar] [CrossRef]
- Takahashi, S.; Badger, M.R. Photoprotection in plants: A new light on photosystem II damage. Trends Plant Sci. 2011, 16, 53–60. [Google Scholar] [CrossRef]
- Zhang, T.-J.; Chow, W.S.; Liu, X.-T.; Zhang, P.; Liu, N.; Peng, C.-L. A magic red coat on the surface of young leaves: Anthocyanins distributed in trichome layer protect Castanopsis fissa leaves from photoinhibition. Tree Physiol. 2016, 36, 1296–1306. [Google Scholar] [CrossRef] [PubMed]
- Gould, K.S. Nature’s swiss army knife: The diverse protective roles of anthocyanins in leaves. J. Biomed. Biotechnol. 2004, 5, 314–320. [Google Scholar] [CrossRef]
- Close, D.C.; Beadle, C.L. The ecophysiology of foliar anthocyanin. Botanical Rev. 2003, 69, 149–161. [Google Scholar] [CrossRef]
- Steyn, W.J.; Wand, S.J.E.; Holcroft, D.M.; Jacobs, G. Anthocyanins in vegetative tissues: A proposed unified function in photoprotection. New Phytol. 2002, 155, 349–361. [Google Scholar] [CrossRef]
- Manetas, Y.; Petropoulou, Y.; Psaras, G.K.; Drinia, A. Exposed red (anthocyanic) leaves of Quercus coccifera display shade characteristics. Functional Plant Biol. 2003, 30, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Neill, S.O.; Gould, K.S. Anthocyanins in leaves: Light attenuators or antioxidants? Functional Plant Biol. 2003, 30, 865–873. [Google Scholar] [CrossRef]
- Liakopoulos, G.; Nikolopoulos, D.; Klouvatou, A.; Vekkos, K.A.; Manetas, Y.; Karabourniotis, G. The photoprotective role of epidermal anthocyanins and surface pubescence in young leaves of grapevine (Vitis vinifera). Ann. Bot. 2006, 98, 257–265. [Google Scholar] [CrossRef]
- van den Berg, A.K.; Perkins, T.D. Do anthocyanins function as photoprotective light screens in senescing sugar maple leaves? J. Plant Sci. Curr. Res. 2017, 1, 002. [Google Scholar] [CrossRef]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef]
- Dong, N.Q.; Lin, H.X. Contribution of phenylpropanoid metabolism to plant development and plant–environment interactions. J. Integrative Plant Biol. 2021, 63, 180–209. [Google Scholar] [CrossRef]
- Zinoviev, A.Y. Visualization of Multidimensional Data; KSTU: Krasnoyarsk, Russia, 2000; 180p. [Google Scholar]
- Lever, J.; Krzywinski, M.; Altman, N. Principal component analysis. Nat. Methods 2017, 14, 641–642. [Google Scholar] [CrossRef]
- Hussner, A.; Lösch, R. Growth and photosynthesis of Hydrocotyle ranunculoides L. fil. in Central Europe. Flora 2007, 202, 653–660. [Google Scholar] [CrossRef]
- Chen, Z.Y.; Peng, Z.S.; Yang, J.; Chen, W.Y.; Ou-Yang, Z.M. A mathematical model for describing light-response curves in Nicotiana tabacum L. Photosynthetica 2011, 49, 467–471. [Google Scholar] [CrossRef]
- Lin, K.-H.; Shih, F.-C.; Huang, M.-Y.; Weng, J.-H. Physiological characteristics of photosynthesis in yellow-green, green and dark-green chinese kale (Brassica oleracea L. var. Alboglabra Musil.) under varying light intensities. Plants 2020, 9, 960. [Google Scholar] [CrossRef]
- Eitel, J.U.; Vierling, L.A.; Long, D.S.; Litvak, M.; Eitel, K.C. Simple assessment of needleleaf and broadleaf chlorophyll content using a flatbed color scanner. Can. J. For. Res. 2011, 41, 1445–1451. [Google Scholar] [CrossRef]
- Murray, J.R.; Hackett, W.P. Dihydroflavonol reductase activity in relation to differential anthocyanin accumulation in juvenile and mature phase Hedera helix L. Plant Physiol. 1991, 97, 343–351. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Buschmann, C. Extraction of phtosynthetic tissues: Chlorophylls and carotenoids. Curr. Protocols Food Analyt. Chem. 2001, 1, F4-2. [Google Scholar]

| Lighting | Leaves | Chloroplast | |||
|---|---|---|---|---|---|
| Length, µm | Width, µm | Number of Thylakoids per Granum | Plastoglobule Diameter, µm | ||
| Shade | Young | 9.24 ± 0.38 a | 1.46 ± 0.05 a | 6.77 ± 0.41 a | 0.09 ± 0.023 a |
| Mature | 11.63 ± 0.37 b | 3.48 ± 0.19 b | 7.67 ± 0.45 a | 0.29 ± 0.051 b | |
| Sun | Young | 6,98 ± 0,29 a | 1.39 ± 0.06 a | 4.76 ± 0.31 b | 0.12 ± 0.021 a |
| Mature | 9.73 ± 0.27 b | 1.64 ± 0.08 a | 5.28 ± 0.38 b | 0.23 ± 0.067 b | |
| Pigment | Shade Leaves | Sun Leaves | ||
|---|---|---|---|---|
| Young | Mature | Young | Mature | |
| Chlorophyll a | 4.09 ± 0.35 a | 9.95 ± 0.38 b | 5.12 ± 0.33 c | 4.82 ± 0.36 c |
| Chlorophyll b | 1.71 ± 0.19 a | 4.35 ± 0.19 b | 2.01 ± 0.16 c | 2.08 ± 0.14 c |
| Chlorophylls (a + b) | 5.79 ± 0.53 a | 14.29 ± 0.56 b | 7.13 ± 0.47 c | 6.90 ± 0.50 c |
| Carotenoids | 1.34 ± 0.12 a | 2.86 ± 0.11 b | 2.00 ± 0.12 c | 1.78 ± 0.11 c |
| Substance | Shade Leaves | Sun Leaves | ||
|---|---|---|---|---|
| Young | Mature | Young | Mature | |
| Anthocyanins | 0.37 ± 0.10 a | 0.34 ± 0.03 a | 0.65 ± 0.03 b | 0.26 ± 0.03 a |
| Phenylpropanoids | 0.35 ± 0.06 a | 0.40 ± 0.03 a | 0.77 ± 0.07 b | 0.40 ± 0.02 a |
| Parameter | Shade Leaves | Sun Leaves | ||
|---|---|---|---|---|
| Young | Mature | Young | Mature | |
| Fv/Fm | 0.70 ± 0.02 a | 0.73 ± 0.01 b | 0.69 ± 0.01 a | 0.73 ± 0.01 b |
| φE0 | 0.35 ± 0.01 a | 0.37 ± 0.00 b | 0.34 ± 0.01 a | 0.34 ± 0.01 a |
| φR0 | 0.25 ± 0.01 a | 0.25 ± 0.00 a | 0.27 ± 0.01 a | 0.26 ± 0.00 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kordyum, E.; Polishchuk, O.; Akimov, Y.; Brykov, V. Photosynthetic Apparatus of Hydrocharis morsus-ranae in Different Solar Lighting. Plants 2022, 11, 2658. https://doi.org/10.3390/plants11192658
Kordyum E, Polishchuk O, Akimov Y, Brykov V. Photosynthetic Apparatus of Hydrocharis morsus-ranae in Different Solar Lighting. Plants. 2022; 11(19):2658. https://doi.org/10.3390/plants11192658
Chicago/Turabian StyleKordyum, Elizabeth, Oleksandr Polishchuk, Yuri Akimov, and Vasyl Brykov. 2022. "Photosynthetic Apparatus of Hydrocharis morsus-ranae in Different Solar Lighting" Plants 11, no. 19: 2658. https://doi.org/10.3390/plants11192658
APA StyleKordyum, E., Polishchuk, O., Akimov, Y., & Brykov, V. (2022). Photosynthetic Apparatus of Hydrocharis morsus-ranae in Different Solar Lighting. Plants, 11(19), 2658. https://doi.org/10.3390/plants11192658







