Metabolomic Profiling in Combination with Data Association Analysis Provide Insights about Potential Metabolic Regulation Networks among Non-Volatile and Volatile Metabolites in Camellia sinensis cv Baijiguan
Abstract
1. Introduction
2. Results
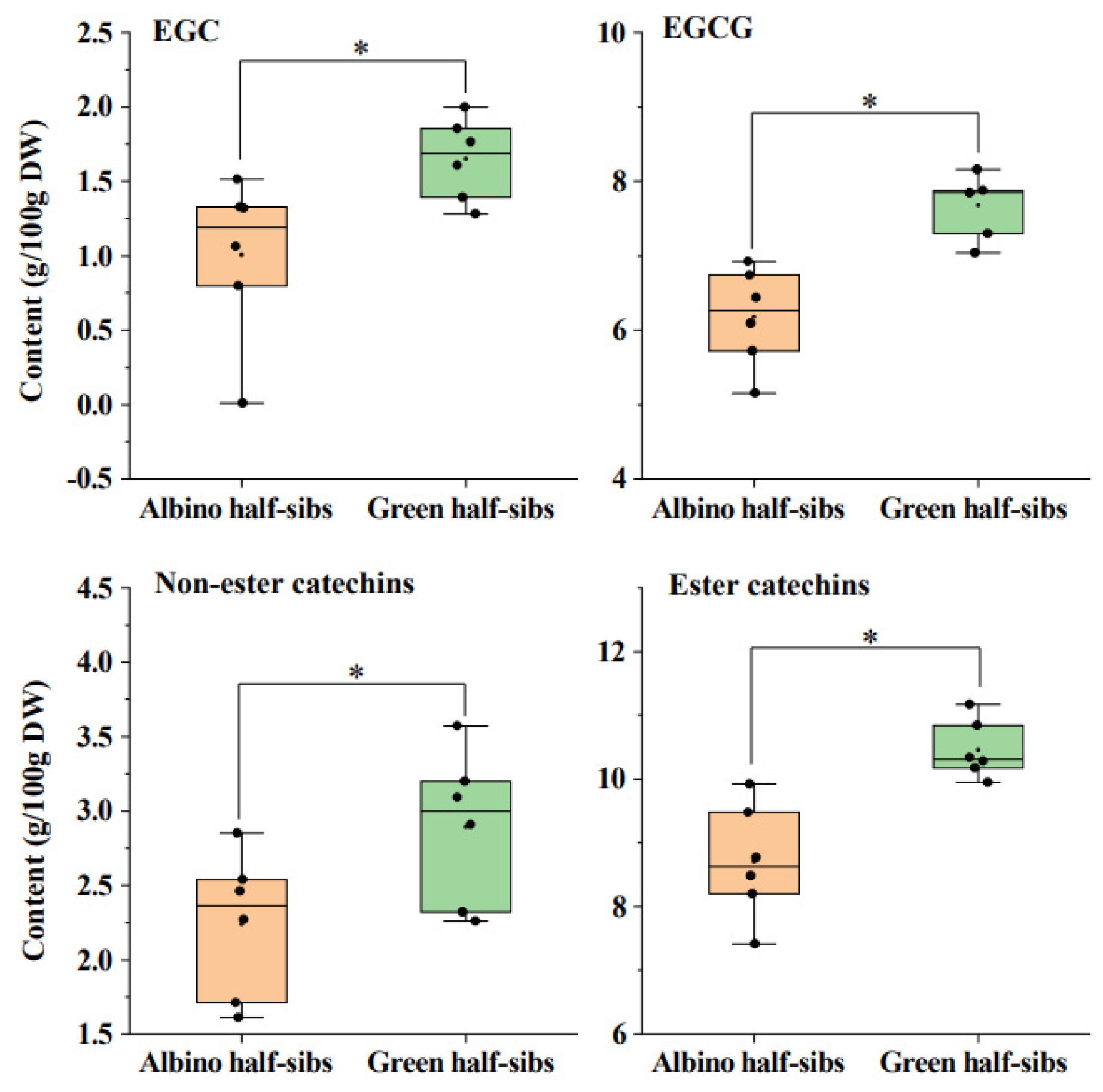
2.1. Non-Volatile Contents of the Albino Half-Sibs and the Green Half-Sibs of Baijiguan
2.2. Volatile Contents of the Albino Half-Sibs and the Green Half-Sibs of Baijiguan
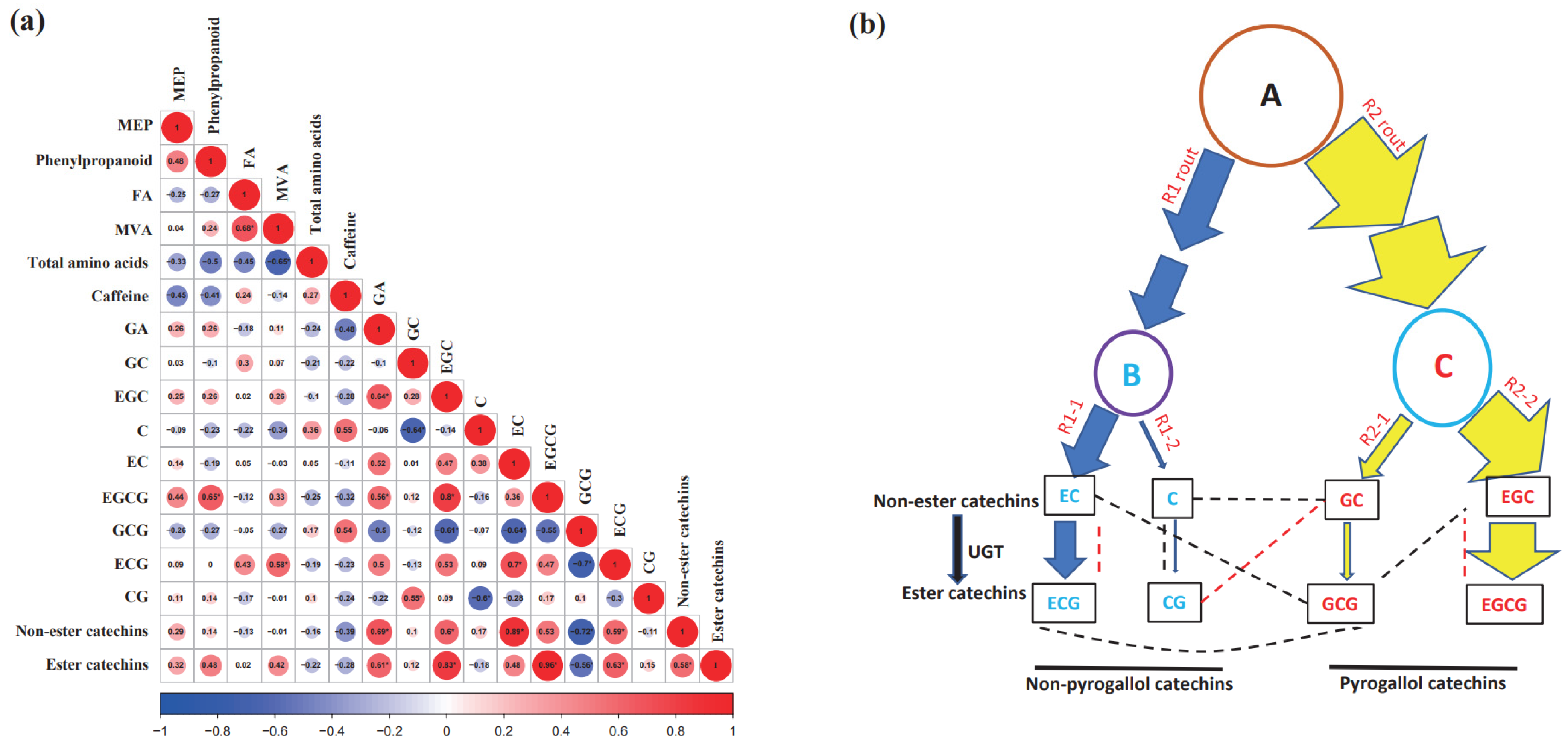
2.3. Correlations among Non-Volatile Metabolites of Baijiguan Half-Sibs
2.4. Correlations among the Four Volatile Synthesis Pathways in Baijiguan Half-Sibs
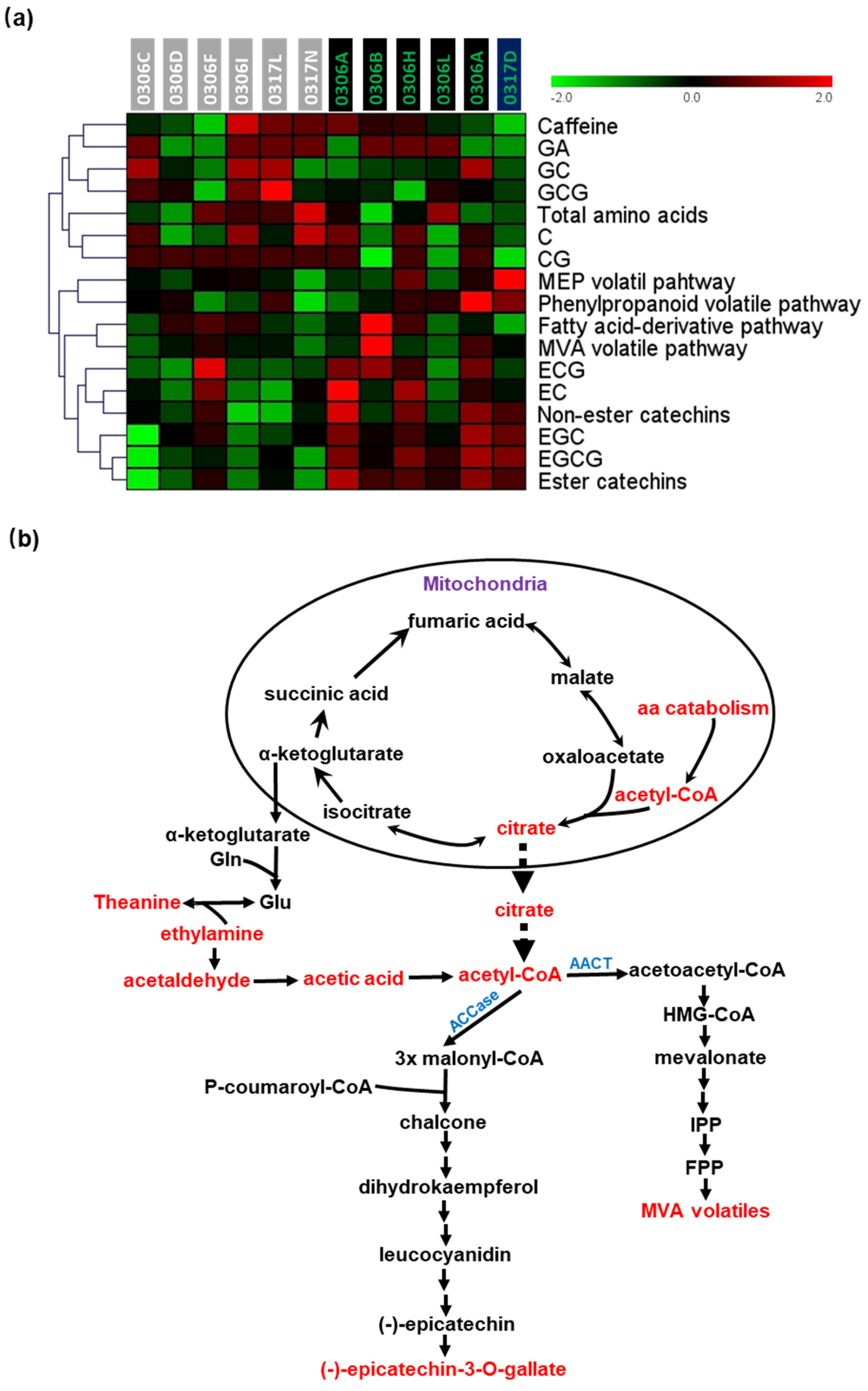
2.5. Correlations between Volatile Synthesis Pathways and Non-Volatile Metabolites in Baijiguan Half-Sibs
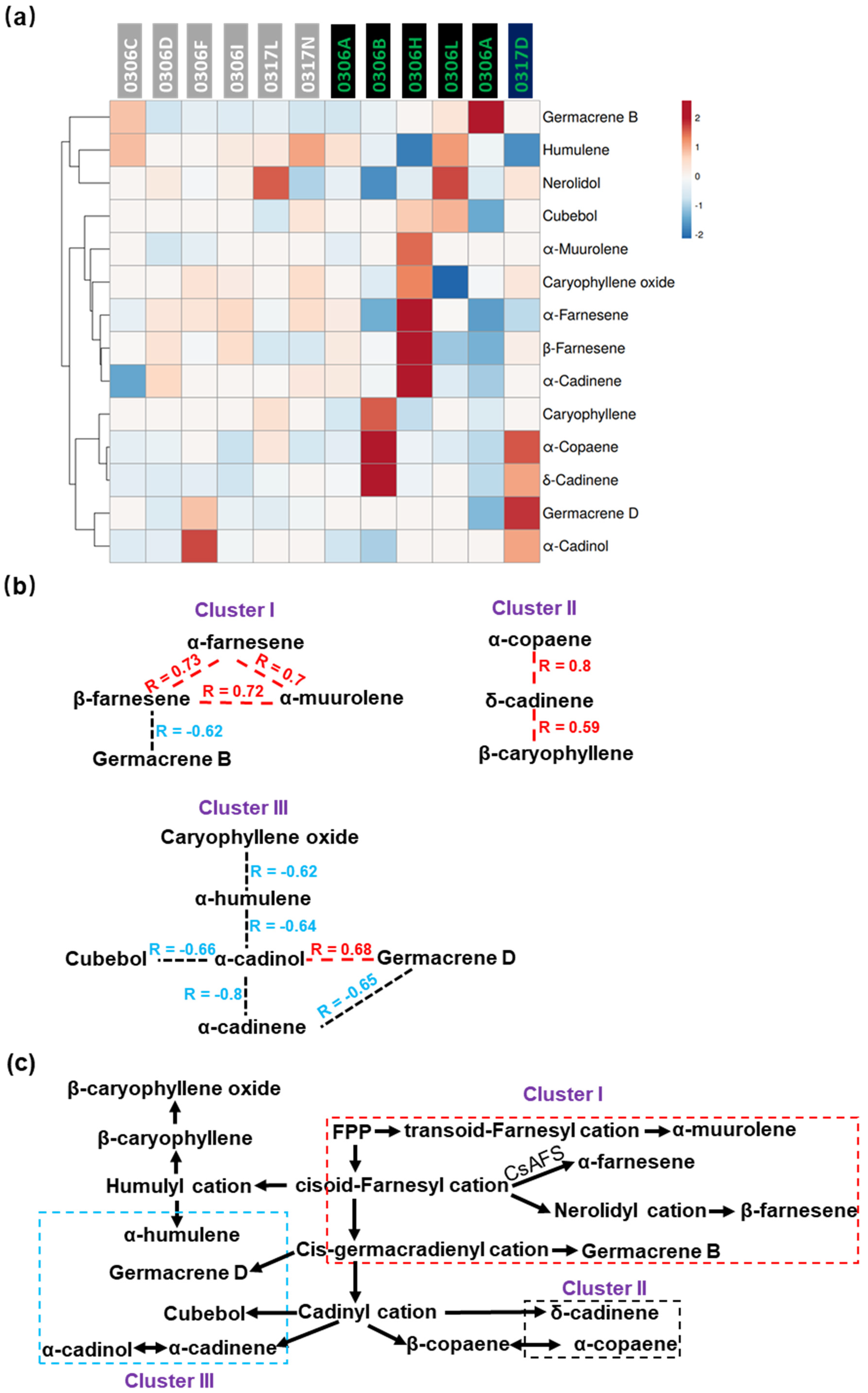
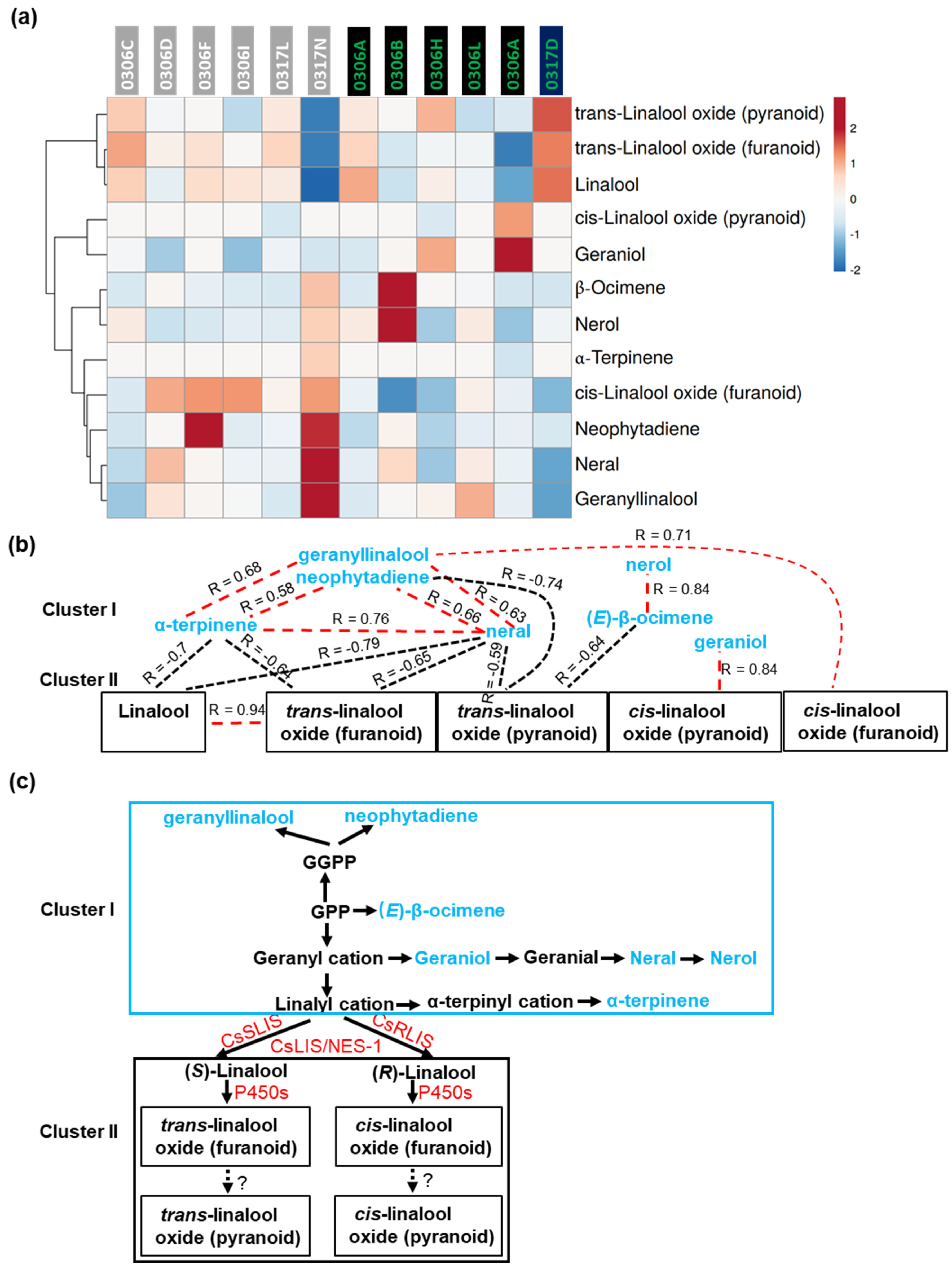
2.6. Correlations among the Volatiles Derived from MVA Pathway in Baijiguan Half-Sibs
2.7. Correlations among the Volatiles Derived from MEP Pathway in Baijiguan Half-Sibs
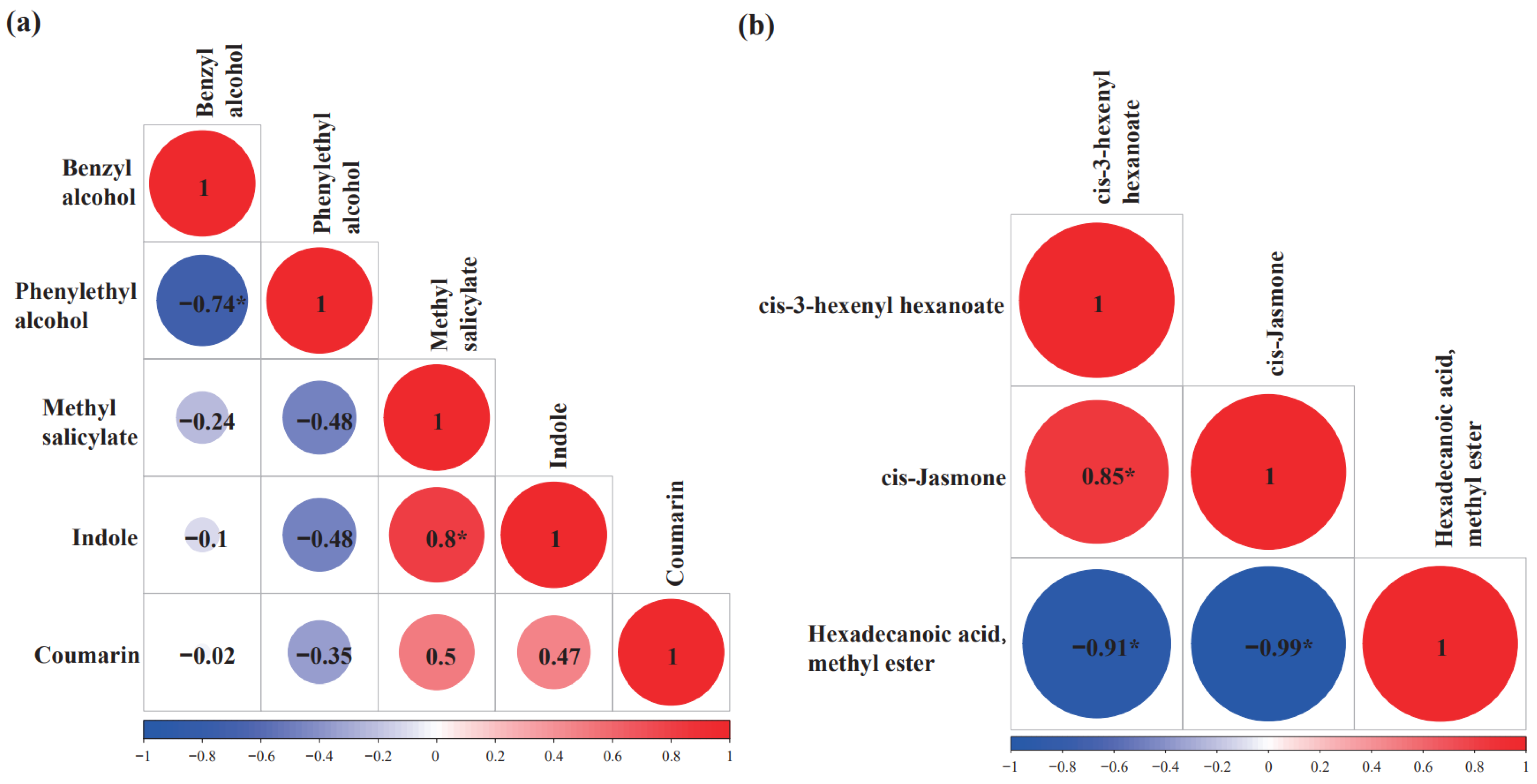
2.8. Correlations among the Volatiles Derived from the Shikimate–Phenylpropanoid Pathway and Fatty Acid-Derivative Pathway
3. Discussion
3.1. The Synthesis of Total Amino Acids, Caffeine, and Catechins Likely Is Independently Regulated in Tender Tea Leaves
3.2. Volatile Synthesis Pathways Were Metabolically Connected with Catechins or Amino Acid Metabolism
3.3. Potential Opportunities to Alter Volatile Contents and Compositions in Tender Tea Leaves
4. Materials and Methods
4.1. Plant Material
4.2. Tea Sample Preparation
4.3. Free Amino Acid Measurement
4.4. Caffeine Quantification
4.5. Catechin Measurement
4.6. Tea Volatile Isolation and Quantification
4.7. Correlation Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Chaturvedula, V.S.P.; Prakash, I. The aroma, taste, color and bioactive constituents of tea. J. Med. Plants Res. 2020, 5, 2110–2124. [Google Scholar]
- Cabrera, C.; Artacho, R.; Gimênez, R. Beneficial effects of green tea—A review. J. Am. Coll. Nutr. 2006, 25, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Gao, M.J.; Hou, R.Y.; Hu, X.Y.; Zhang, L.; Wan, X.C.; Wei, S. Determination of quality constituents in the young leaves of albino tea cultivars. Food Chem. 2014, 155, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Li, C.F.; Xu, Y.X.; Ma, J.Q.; Jin, J.Q.; Huang, D.J.; Yao, M.Z.; Ma, C.L.; Chen, L. Biochemical and transcriptomic analyses reveal different metabolite biosynthesis profiles among three color and developmental stages in ‘Anji Baicha’ (Camellia sinensis). BMC Plant Biol. 2016, 16, 195. [Google Scholar] [CrossRef]
- Robinson, J.; Owuor, P. Tea aroma. In Tea; Willson, K.C., Clifford, M.N., Eds.; Chapman & Hall: London, UK, 1992; pp. 603–647. [Google Scholar]
- Schuh, C.; Schieberle, P. Characterization of the key aroma compounds in the beverage prepared from Darjeeling black tea: Quantitative differences between tea leaves and infusion. J. Agric. Food Chem. 2006, 54, 916–924. [Google Scholar] [CrossRef]
- Feng, Z.H.; Li, Y.F.; Li, M.; Wang, W.J.; Zhang, L.; Wan, X.C.; Yang, X.G. Tea aroma formation from six model manufacturing processes. Food Chem. 2019, 285, 347–354. [Google Scholar] [CrossRef]
- Chen, X.B.; Zhang, Y.; Du, Z.H.; Liu, R.M.; Guo, L.; Chen, C.S.; Wu, H.L.; Chen, M.J. Establishing a quantitative volatile measurement method in tea by integrating sample extraction method optimization and data calibration. Flavour Fragr. J. 2020, 36, 64–74. [Google Scholar] [CrossRef]
- Chen, M.J.; Guo, L.; Zhou, H.W.; Guo, Y.L.; Zhang, Y.; Lin, Z.; Sun, M.; Zeng, W.; Wu, H.L. Absolute quantitative volatile measurement from fresh tea leaves and the derived teas revealed contributions of postharvest synthesis of endogenous volatiles for the aroma quality of made teas. Appl. Sci. 2021, 11, 613. [Google Scholar] [CrossRef]
- Chen, Z.H.; Lin, Z.-H.; You, X.-M.; Zhong, Q.-S.; Shan, R.-Y.; Chen, C.-S.; Yu, W.-Q. Genetics on color of Baijiguan tea leaves. Acta Tea Sin. 2019, 60, 99–105. [Google Scholar]
- Du, Y.Y.; Liang, Y.R.; Wang, H.; Wang, K.R.; Lu, J.L.; Zhang, G.H.; Lin, W.P.; Li, M.; Fang, Q.Y. A study on the chemical composition of albino tea cultivars. J. Hortic. Sci. Biotechnol. 2006, 81, 809–812. [Google Scholar] [CrossRef]
- Li, Q.; Huang, J.N.; Liu, S.Q.; Li, J.; Yang, X.H.; Liu, Y.S.; Liu, Z.H. Proteomic analysis of young leaves at three developmental stages in an albino tea cultivar. Proteome Sci. 2011, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.L.; Chen, L.; Wang, X.C.; Jin, J.Q.; Ma, J.Q.; Yao, M.Z.; Wang, Z.L. Differential expression analysis of different albescent stages of Baiye 1 (Camellia sinensis (L.) O. Kuntze) using cDNA microarray. Sci. Hortic. 2012, 148, 246–254. [Google Scholar] [CrossRef]
- Ma, Q.P.; Li, H.; Zou, Z.G.; Arkorful, E.; Lv, Q.R.; Zhou, Q.Q.; Chen, X.; Sun, K.; Li, X.H. Transcriptomic analyses identify albino associated genes of a novel albino tea germplasm ‘Huabai 1’. Hortic. Res. 2018, 5, 54. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.K.; Li, N.N.; Du, Y.Y.; Liang, Y.R. Effect of sunlight shielding on leaf structure and amino acids concentration of light sensitive albino tea plan. Afr. J. Biotechnol. 2013, 12, 5535–5539. [Google Scholar]
- Wu, Q.J.; Chen, Z.D.; Sun, W.J.; Deng, T.T.; Chen, M.J. De novo sequencing of the leaf transcriptome reveals complex light-responsive regulatory networks in Camellia sinensis cv. Baijiguan. Front. Plant Sci. 2016, 7, 332. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.F.; Han, Z.X.; Feng, L.; Gao, L.P.; Gao, M.J.; Gruber, M.Y.; Zhang, Z.L.; Xia, T.; Wan, X.C.; Wei, S. Metabolic flux redirection and transcriptomic reprogramming in the albino tea cultivar ‘Yu-Jin-Xiang’ with an emphasis on catechin production. Sci. Rep. 2017, 7, 45062. [Google Scholar] [CrossRef] [PubMed]
- Song, L.B.; Ma, Q.P.; Zou, Z.W.; Sun, K.; Yao, Y.T.; Tao, J.H.; Kaleri, N.A.; Li, X.H. Molecular link between leaf coloration and gene expression of flavonoid and carotenoid biosynthesis in Camellia sinensis cultivar ‘Huangjinya’. Front. Plant Sci. 2017, 8, 803. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Zeng, L.T.; Yu, Z.M.; Li, J.L.; Tang, J.C.; Su, X.G.; Yang, Z.Y. Differential accumulation of aroma compounds in normal green and albino-induced yellow tea (Camellia sinensis) leaves. Molecules 2018, 23, 2677. [Google Scholar] [CrossRef]
- Liu, G.F.; Liu, J.J.; He, Z.R.; Wang, F.M.; Yang, H.; Yan, Y.F.; Gao, M.J.; Gruber, M.Y.; Wan, X.C.; Wei, S. Implementation of CsLIS/NES in linalool biosynthesis involves transcript splicing regulation in Camellia sinensis. Plant Cell Environ. 2017, 41, 176–186. [Google Scholar] [CrossRef]
- Li, C.F.; Ma, J.Q.; Huang, D.J.; Ma, C.L.; Jin, J.Q.; Yao, M.Z.; Chen, L. Comprehensive dissection of metabolic changes in albino and green tea cultivars. J. Agric. Food Chem. 2018, 66, 2040–2048. [Google Scholar] [CrossRef]
- Dong, F.; Shi, Y.Z.; Liu, M.Y.; Fan, K.; Zhang, Q.F.; Ruan, J.F. iTRAQ-based quantitative proteomics analysis reveals the mechanism underlying the weakening of carbon metabolism in chlorotic tea leaves. Int. J. Mol. Sci. 2018, 19, 3943. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.Y.; Zhang, W.F.; Liu, Y.; Zhang, H.J.; Ren, H.Z.; Chen, Y.; Wang, L.; Zeng, J.M.; Yang, Y.J.; Wang, X.C. Pale green mutant analyses reveal the importance of CsGLKs in chloroplast developmental regulation and their effects on flavonoid biosynthesis in tea plant. Plant Physiol. Biochem. 2020, 146, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.F.; Zhao, H.; Guo, F.; Shi, X.P.; Ye, C.; Yang, P.X.; Liu, B.Y.; Ni, D.J. Transcriptomic analysis reveals mechanism of light-sensitive albinism in tea plant Camellia sinensis ‘Huangjinju’. BMC Plant Biol. 2020, 20, 216. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yue, C.; Cao, H.L.; Zhou, Y.H.; Zeng, J.M.; Yang, Y.J.; Wang, X.C. Biochemical and transcriptome analyses of a novel chlorophyll-deficient chlorina tea plant cultivar. BMC Plant Biol. 2014, 14, 352. [Google Scholar] [CrossRef]
- Li, N.N.; Yang, Y.P.; Ye, J.H.; Lu, J.L.; Zheng, X.Q.; Liang, Y.R. Effects of sunlight on gene expression and chemical composition of light-sensitive albino tea plant. Plant Growth Regul. 2016, 78, 253–262. [Google Scholar] [CrossRef]
- Cheng, S.H.; Fu, X.M.; Liao, Y.Y.; Xu, X.L.; Zeng, L.T.; Tang, J.C.; Li, J.L.; Lai, J.H.; Yang, Z.Y. Differential accumulation of specialized metabolite l-theanine in green and albino induced yellow tea (Camellia sinensis) leaves. Food Chem. 2019, 276, 93–100. [Google Scholar] [CrossRef]
- Lu, M.Q.; Han, J.Y.; Zhu, B.Y.; Jia, H.Y.; Yang, T.Y.; Wang, R.J.; Deng, W.W.; Zhang, Z.Z. Significantly increased amino acid accumulation in a novel albino branch of the tea plant (Camellia sinensis). Planta 2019, 249, 363–376. [Google Scholar] [CrossRef]
- Li, C.F.; Yao, M.Z.; Ma, C.L.; Ma, J.Q.; Jin, J.Q.; Chen, L. Differential metabolic profiles during the albescent stages of ‘Anji Baicha’ (Camellia sinensis). PLoS ONE 2015, 10, e0139996. [Google Scholar] [CrossRef]
- Li, J.L.; Zeng, L.T.; Liao, Y.Y.; Gu, D.C.; Tang, J.C.; Yang, Z.Y. Influence of chloroplast defects on formation of jasmonic acid and characteristic aroma compounds in tea (Camellia sinensis) leaves exposed to postharvest stresses. Int. J. Mol. Sci. 2019, 20, 1044. [Google Scholar] [CrossRef]
- Gachon, C.M.; Langlois-Meurinne, M.; Henry, Y.; Saindrenan, P. Transcriptional co-regulation of secondary metabolism enzymes in Arabidopsis: Functional and evolutionary implications. Plant Mol. Biol. 2005, 58, 229–245. [Google Scholar] [CrossRef]
- Wille, A.; Zimmermann, P.; Vranová, E.; Fürholz, A.; Laule, O.; Bleuler, S.; Hennig, L.; Prelic, A.; von Rohr, P.; Thiele, L.; et al. Sparse graphical Gaussian modeling of the isoprenoid gene network in Arabidopsis thaliana. Genome Biol. 2004, 5, R92. [Google Scholar] [CrossRef] [PubMed]
- Jian, W.; Cao, H.H.; Yuan, S.; Liu, Y.D.; Lu, J.F.; Lu, W.; Li, N.; Wang, J.H.; Zou, J.; Tang, N.; et al. SlMYB75, an MYB-type transcription factor, promotes anthocyanin accumulation and enhances volatile aroma production in tomato fruits. Hortic. Res. 2019, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Wei, K.; Wang, L.Y.; Zhou, J.; He, W.; Zeng, J.M.; Jiang, Y.W.; Cheng, H. Catechin contents in tea (Camellia sinensis) as affected by cultivar and environment and their relation to chlorophyll contents. Food Chem. 2011, 125, 44–48. [Google Scholar] [CrossRef]
- Zhang, Q.F.; Tang, D.D.; Liu, M.Y.; Ruan, J.Y. Integrated analyses of the transcriptome and metabolome of the leaves of albino tea cultivars reveal coordinated regulation of the carbon and nitrogen metabolism. Sci. Hortic. 2018, 231, 272–281. [Google Scholar] [CrossRef]
- Li, N.N.; Lu, J.L.; Li, Q.S.; Zheng, X.Q.; Wang, X.C.; Wang, L.; Wang, Y.C.; Ding, C.Q.; Liang, Y.R.; Yang, Y.J. Dissection of chemical composition and associated gene expression in the pigment deficient tea cultivar ‘Xiaoxueya’ reveals an albino phenotype and metabolite formation. Front. Plant Sci. 2019, 10, 1543. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.G.; Li, J.; Li, Y.H.; Yuan, L.; Liu, S.Q.; Huang, J.A.; Liu, Z.H. Dynamic changes in catechin levels and catechin biosynthesis-related gene expression in albino tea plants (Camellia sinensis L.). Plant Physiol. Biochem. 2013, 71, 132–143. [Google Scholar] [CrossRef]
- Zhu, X.J.; Zhao, Z.; Xin, H.H.; Wang, M.L.; Wang, W.D.; Chen, X.; Li, X.-H. Isolation and dynamic expression of four genes involving in shikimic acid pathway in Camellia sinensis ‘Baicha 1’ during periodic albinism. Mol. Biol. Rep. 2016, 43, 1119–1127. [Google Scholar] [CrossRef]
- Xu, Y.X.; Chen, W.; Ma, C.L.; Shen, S.Y.; Zhou, Y.Y.; Zhou, L.Q.; Chen, L. Proteome and acetyl-proteome profiling of Camellia sinensis cv. ‘Anji Baicha’ during periodic albinism reveals alterations in photosynthetic and secondary metabolite biosynthetic pathways. Front. Plant Sci. 2017, 8, 2104. [Google Scholar] [CrossRef]
- Xu, Y.X.; Shen, C.J.; Ma, J.Q.; Chen, W.; Mao, J.; Zhou, Y.Y.; Chen, L. Quantitative succinyl-proteome profiling of Camellia sinensis cv. ‘Anji Baicha’ during periodic albinism. Sci. Rep. 2017, 7, 1873. [Google Scholar] [CrossRef]
- Zeng, C.Z.; Lin, H.Y.; Liu, Z.X.; Liu, Z.H. Analysis of young shoots of ‘Anji Baicha’ (Camellia sinensis) at three developmental stages using nontargeted LC-MS-based metabolomics. J. Food Sci. 2019, 84, 1746–1757. [Google Scholar] [CrossRef]
- Xu, P.; Su, H.; Jin, R.; Mao, Y.X.; Xu, A.A.; Cheng, H.Y.; Wang, Y.F.; Meng, Q. Shading effects on leaf color conversion and biosynthesis of the major secondary metabolites in the albino tea cultivar “Yujinxiang”. J. Agric. Food Chem. 2020, 68, 2528–2538. [Google Scholar] [CrossRef] [PubMed]
- Werner, R.A.; Rossmann, A.; Schwarz, C.; Bacher, A.; Schmidt, H.L.; Eisenreich, W. Biosynthesis of gallic acid in Rhus typhina: Discrimination between alternative pathways from natural oxygen isotope abundance. Phytochemistry 2004, 65, 2809–2813. [Google Scholar] [CrossRef] [PubMed]
- Tohge, T.; Watanabe, M.; Hoefgen, R.; Fernie, A.R. Shikimate and phenylalanine biosynthesis in the green lineage. Front. Plant Sci. 2013, 4, 62. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, E.; Schultz, G. Localization, purification, and characterization of shikimate oxidoreductase-dehydroquinate hydrolase from stroma of spinachchloroplasts. Plant Physiol. 1985, 79, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Ossipov, V.; Salminen, J.P.; Ossipova, S.; Haukioja, E.; Pihlaja, K. Gallic acid and hydrolysable tannins are formed in birch leaves from an intermediate compound of the shikimate pathway. Biochem. Syst. Ecol. 2003, 31, 3–16. [Google Scholar] [CrossRef]
- Zhou, X.C.; Zeng, L.T.; Chen, Y.J.; Wang, X.W.; Liao, Y.Y.; Xiao, Y.Y.; Fu, X.M.; Yang, Z.-Y. Metabolism of gallic acid and its distributions in tea (Camellia sinensis) plants at the tissue and subcellular levels. Int. J. Mol. Sci. 2020, 21, 5684. [Google Scholar] [CrossRef]
- Bao, X.M.; Focke, M.; Pollard, M.; Ohlrogge, J. Understanding in vivo carbon precursor supply for fatty acid synthesis in leaf tissue. Plant J. 2000, 22, 39–50. [Google Scholar] [CrossRef]
- Fan, J.L.; Yan, C.S.; Roston, R.; Shanklin, J.; Xu, C.C. Arabidopsis lipins, PDAT1 acyltransferase, and SDP1 triacylglycerol lipase synergistically direct fatty acids toward β-oxidation, thereby maintaining membrane lipid homeostasis. Plant Cell. 2014, 26, 4119–4134. [Google Scholar] [CrossRef]
- Sapir-Mir, M.; Mett, A.; Belausov, E.; Tal-Meshulam, S.; Frydman, A.; Gidoni, D.; Eyal, Y. Peroxisomal localization of Arabidopsis isopentenyl diphosphate isomerases suggests that part of the plant isoprenoid mevalonic acid pathway is compartmentalized to peroxisomes. Plant Physiol. 2008, 148, 1219–1228. [Google Scholar] [CrossRef]
- Clastre, M.; Papon, N.; Courdavault, V.; GiglioliGuivarch, N.; St-Pierre, B.; Simkin, A.J. Subcellular evidence for the involvement of peroxisomes in plant isoprenoid biosynthesis. Plant Signal. Behav. 2011, 6, 2044–2046. [Google Scholar] [CrossRef][Green Version]
- Ahumada, I.; Cairó, A.; Hemmerlin, A.; González, V.; Pateraki, I.; Bach, T.J.; Rodríguez-Concepción, M.; Campos, N.; Boronat, A. Characterisation of the gene family encoding acetoacetyl-CoA thiolase in Arabidopsis. Funct. Plant Biol. 2008, 35, 1100–1111. [Google Scholar] [CrossRef] [PubMed]
- Reumann, S.; Babujee, L.; Ma, C.; Wienkoop, S.; Siemsen, T.; Antonicelli, G.E.; Rasche, N.; Lüder, F.; Weckwerth, W.; Jahn, O. Proteome analysis of Arabidopsis leaf peroxisomes reveals novel targeting peptides, metabolic pathways, and defense mechanisms. Plant Cell 2007, 19, 3170–3793. [Google Scholar] [CrossRef] [PubMed]
- Corea, O.R.A.; Ki, C.; Cardenas, C.L.; Kim, S.J.; Brewer, S.E.; Patten, A.M. Arogenate dehydratase isoenzymes profoundly and differentially modulate carbon flux into lignins. J. Biol. Chem. 2012, 287, 11446–11459. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Dudareva, N. The shikimate pathway and aromatic Amino acid biosynthesis in plants. Annu. Rev. Plant Biol. 2012, 63, 73–105. [Google Scholar] [CrossRef]
- Kirma, M.; Araújo, W.L.; Fernie, A.R.; Galili, G. The multifacted role of aspartate-family amino acids in plant metabolism. J. Exp. Bot. 2012, 63, 4995–50019. [Google Scholar] [CrossRef]
- Hildebrandt, T.M.; Nesi, A.N.; Araújo, W.L.; Braun, H.P. Amino acid catabolism in plants. Mol. Plant 2015, 8, 1563–1579. [Google Scholar] [CrossRef]
- Tapiero, H.; Mathé, G.; Couvreur, P.; Tew, K.D. Glutamine and glutamate. Biomed. Pharmacother. 2002, 56, 446–457. [Google Scholar] [CrossRef]
- Zhou, P.; Feng, Z.; Chen, M.J.; Ye, N.X.; Lin, Q.; Ouyang, L.Q.; Cai, X.M.; Meng, P.; Gong, X.D.; Wang, Y. Determination of 21 free amino acids in 5 types of tea by ultra-high performance liquid chromatography coupled with tandem mass spectrometry (UHPLC–MS/MS) using a modified 6-aminoquinolyl-Nhydroxysuccinimidyl carbamate (AQC) method. J. Food Compos. Anal. 2019, 81, 46–54. [Google Scholar] [CrossRef]
- Sasaoka, K.; Kito, M.; Onishi, Y. Some properties of the theanine synthesizing enzyme in tea seedlings. Agric. Biol. Chem. 1965, 29, 984–988. [Google Scholar] [CrossRef]
- Wei, C.L.; Yang, H.; Wang, S.B.; Zhao, J.; Liu, C.; Gao, L.P.; Xia, E.H.; Lu, Y.; Tai, Y.L.; She, G.B.; et al. Draft genome sequence of Camellia sinensis var. sinensis provides insights into the evolution of the tea genome and tea quality. Proc. Natl. Acad. Sci. USA 2018, 115, E4151–E4158. [Google Scholar] [CrossRef]
- Huang, R.; Wang, J.Y.; Yao, M.Z.; Ma, C.L.; Chen, L. Quantitative trait loci mapping for free amino acid content using an albino population and SNP markers provides insight into the genetic improvement of tea plants. Hortic. Res. 2022, 9, uhab029. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.W.; Zeng, L.T.; Liao, Y.Y.; Li, J.L.; Tang, J.C.; Yang, Z.Y. Formation of α-farnesene in tea (Camellia sinensis) leaves induced by herbivore-derived wounding and its effect on neighboring tea plants. Int. J. Mol. Sci. 2019, 20, 4151. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Tholl, D.; D’Auria, J.C.; Farooq, A.; Pichersky, E.; Gershenzon, J. Biosynthesis and emission of terpenoid volatiles from Arabidopsis flowers. Plant Cell 2003, 15, 481–494. [Google Scholar] [CrossRef] [PubMed]
- Dueholm, B.; Drew, D.P.; Sweetman, C.; Simonsen, H.T. In planta and in silico characterization of five sesquiterpene synthases from Vitis vinifera (cv. Shiraz) berries. Planta 2019, 249, 59–70. [Google Scholar] [CrossRef]
- Lauchli, R.; Pitzer, J.; Kitto, R.Z.; Ralbarczyk, K.Z.; Rabe, K.S. Improved selectivity of an engineered multi-product terpene synthase. Org. Biomol. Chem. 2014, 12, 4013–4020. [Google Scholar] [CrossRef]
- Meesters, R.J.W.; Duisken, M.; Hollender, J. Study on the cytochrome P450-mediated oxidative metabolism of the terpene alcohol linalool: Indication of biological epoxidation. Xenobiotica 2007, 37, 604–617. [Google Scholar] [CrossRef]
- Ginglinger, J.F.; Boachon, B.; Höfer, R.; Paetz, C.; Köllner, T.G.; Miesch, L.; Lugan, R.; Baltenweck, R.; Mutterer, J.; Ullmann, P.; et al. Gene coexpression analysis reveals complex metabolism of the monoterpene alcohol linalool in Arabidopsis flowers. Plant Cell 2013, 25, 4640–4657. [Google Scholar] [CrossRef]
- Raguso, R.A.; Pichersky, E. New perspectives in pollination biology: Floral fragrances. a day in the life of a linalool molecule: Chemical communication in a plant-pollinator system. Part 1: Linalool biosynthesis in flowering plants. Plant Species Biol. 1999, 14, 95–120. [Google Scholar] [CrossRef]
- Höfer, R.; Boachon, B.; Renault, H.; Gavira, C.; Miesch, L.; Iglesias, J.; Ginglinger, J.F.; Allouche, L.; Miesch, M. Dual function of the cytochrome P450 CYP76 family from Arabidopsis thaliana in the metabolism of monoterpenols and phenylurea herbicides. Plant Physiol. 2014, 116, 1149–1161. [Google Scholar] [CrossRef]
- Boachon, B.; Junker, R.R.; Miesch, L.; Bassard, J.E.; Höfer, R.; Caillieaudeaux, R.; Seidel, D.E.; Lesot, A.; Heinrich, C.; Ginglinger, J.F.; et al. CYP76C1 (cytochrome P450)-mediated linalool metabolism and the formation of volatile and soluble linalool oxides in Arabidopsis flowers: A strategy for defense against floral antagonists. Plant Cell 2015, 27, 2972–2990. [Google Scholar] [CrossRef]
- Ilc, T.; Parage, C.; Boachon, B.; Navrot, N.; Werck-Reichhart, D. Monoterpenol oxidative metabolism: Role in plant adaptation and potential applications. Front. Plant Sci. 2016, 7, 509. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.T.; Zhou, Y.; Gui, J.D.; Fu, X.M.; Mei, X.; Zhen, Y.P.; Ye, T.X.; Du, B.; Dong, F.; Watanabe, N.; et al. Formation of volatile tea constituent indole during the oolong tea manufacturing process. J. Agric. Food Chem. 2016, 64, 5011–5019. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.W.; Wang, R.X.; Yang, T.Y.; Jiang, L.N.; Zhang, Z.Z. Functional characterization of salicylic acid carboxyl methyltransferase from Camellia sinensis, providing the aroma compound of methyl salicylate during withering process of white tea. J. Agric. Food Chem. 2017, 65, 11036–11045. [Google Scholar] [CrossRef]
- Boatright, J.; Negre, F.; Chen, X.; Kish, C.M.; Wood, B.; Peel, G.; Orlova, I.; Gang, D.; Rhodes, D.; Dudareva, N. Understanding in vivo benzenoid metabolism in petunia petal tissue. Plant Physiol. 2004, 135, 1993–2011. [Google Scholar] [CrossRef] [PubMed]
- Dixon, R.A. Natural products and plant disease resistance. Nature 2001, 411, 843–847. [Google Scholar] [CrossRef]
- Chen, M.J.; Vijaykumar, V.; Lu, B.W.; Xia, B.; Li, N. Cis- and trans-cinnamic acids have different effects on the catalytic properties of Arabidopsis phenylalanine ammonia lyases PAL1, PAL2, and PAL4. J. Integr. Plant Biol. 2005, 47, 67–75. [Google Scholar] [CrossRef]
- Fu, X.M.; Chen, Y.Y.; Mei, X.; Katsuno, T.; Kobayashi, E.; Dong, F.; Watanabe, N.; Yang, Z.Y. Regulation of formation of volatile compounds of tea (Camellia sinensis) leaves by single light wavelength. Sci. Rep. 2015, 5, 16858. [Google Scholar] [CrossRef]
- Kolosova, N.; Gorenstein, N.; Kish, C.M.; Dudareva, N. Regulation of circadian methyl benzoate emission in diurnally and nocturnally emitting plants. Plant Cell 2001, 13, 2333–2347. [Google Scholar] [CrossRef]
- Verdonk, J.C.; de Vos, C.H.R.; Verhoeven, H.A.; Haring, M.A.; van Tunen, A.J.; Schuurink, R.C. Regulation of floral scent production in petunia revealed by targeted metabolomics. Phytochemistry 2003, 62, 997–1008. [Google Scholar] [CrossRef]
- Verdonk, J.C.; Haring, M.A.; van Tunen, A.J.; Schuurink, R.C. ODORANT1 regulates fragrance biosynthesis in petunia flowers. Plant Cell 2005, 17, 1612–1624. [Google Scholar] [CrossRef]
- Spitzer-Rimon, B.; Marhevka, E.; Barkai, O.; Marton, I.; Edelbaum, O.; Masci, T.; Prathapani, N.K.; Shklarman, E.; Ovadis, M.; Vainstein, A. EOBII, a gene encoding a flower-specific regulator of phenylpropanoid volatiles’ biosynthesis in petunia. Plant Cell 2010, 22, 1961–1976. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Chen, M.J.; Guo, Y.L.; Lin, Z. Variations in fatty acids affected their derivative volatiles during Tieguanyin tea processing. Foods 2022, 11, 1563. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.Q.; Jin, J.Q.; Yao, M.Z.; Ma, C.L.; Xu, Y.X.; Hao, W.J.; Chen, L. Quantitative trait loci mapping for theobromine and caffeine contents in tea plant (Camellia sinensis). J. Agric. Food Chem. 2018, 66, 13321–13327. [Google Scholar] [CrossRef]
- Yang, Y.S.; Zhao, M.; Liu, Y.; Fang, Z.T.; Li, Q.S.; Zheng, X.Q.; Liang, Y.R.; Lu, J.L. Separation and identification of an abundant trigalloylglucose from special tea genetic resources. Beverage Plant Res. 2022, 2, 11. [Google Scholar] [CrossRef]
- Niemetz, R.; Gross, G.G. Enzymology of gallotannin and ellagitannin biosynthesis. Phytochemistry 2005, 66, 2001–2011. [Google Scholar] [CrossRef]
- Liu, Y.J.; Gao, L.P.; Liu, L.; Yang, Q.; Lu, Z.W.; Nie, Z.Y.; Wang, Y.S.; Xia, T. Purification and characterization of a novel galloyltransferase involved in catechin galloylation in the tea plant (Camellia sinensis). J. Biol. Chem. 2012, 287, 44406–44417. [Google Scholar] [CrossRef]
- GB/T 8314-2013; Tea—Determination of Free Amino Acids Content. National Standards of People’s Republic of China: Beijing, China, 2013. Available online: https://www.nssi.org.cn/nssi/front/84568010.html (accessed on 13 September 2022).
- GB/T 8312-2013; Tea—Determination of Caffeine Content. National Standards of People’s Republic of China: Beijing, China, 2013. Available online: https://www.nssi.org.cn/nssi/front/84568003.html (accessed on 13 September 2022).
- GB/T 8313-2018; Determination of Total Polyphenols and Catechins Content in Tea. National Standards of People’s Republic of China: Beijing, China, 2018. Available online: https://www.nssi.org.cn/nssi/front/108563683.html (accessed on 13 September 2022).
| Leaf Type | Germplasm Name | Phenylpropanoids | Monoterpenes/ Diterpenes | Sesquiterpenes | Fatty Acid Derivatives |
|---|---|---|---|---|---|
| Albino half-sibs | 0306C | 19.56 ± 1.54 | 12.18 ± 0.43 | 0.91 ± 0.12 | 0.19 ± 0.01 |
| 0306D | 21.18 ± 1.55 | 9.02 ± 0.38 | 1.27 ± 0.17 | 0.36 ± 0.02 | |
| 0306F | 9.53 ± 0.88 | 13.21 ± 1.73 | 1.57 ± 0.70 | 0.40 ± 0.05 | |
| 0306I | 14.53 ± 0.76 | 14.01 ± 0.45 | 1.26 ± 0.12 | 0.36 ± 0.04 | |
| 0317L | 23.72 ± 1.41 | 11.24 ± 0.52 | 1.28 ± 0.09 | 0.23 ± 0.01 | |
| 0317N | 4.35 ± 0.11 | 3.06 ± 0.12 | 0.75 ± 0.03 | 0.15 ± 0.01 | |
| Green half-sibs | 0306A | 11.48 ± 0.61 | 10.24 ± 0.57 | 1.14 ± 0.06 | 0.26 ± 0.01 |
| 0306B | 17.58 ± 0.58 | 8.79 ± 0.78 | 3.19 ± 0.32 | 0.72 ± 0.05 | |
| 0306H | 23.06 ± 0.59 | 18.53 ± 0.67 | 1.11 ± 0.09 | 0.39 ± 0.02 | |
| 0306L | 22.72 ± 1.61 | 7.24 ± 0.60 | 0.89 ± 0.04 | 0.15 ± 0.00 | |
| 0309A | 37.24 ± 0.56 | 14.57 ± 0.39 | 1.71 ± 0.15 | 0.27 ± 0.02 | |
| 0317D | 28.26 ± 1.35 | 31.00 ± 1.82 | 1.33 ± 0.33 | 0.06 ± 0.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, M.; Kong, X.; Zhang, Y.; Wang, S.; Zhou, H.; Fang, D.; Yue, W.; Chen, C. Metabolomic Profiling in Combination with Data Association Analysis Provide Insights about Potential Metabolic Regulation Networks among Non-Volatile and Volatile Metabolites in Camellia sinensis cv Baijiguan. Plants 2022, 11, 2557. https://doi.org/10.3390/plants11192557
Chen M, Kong X, Zhang Y, Wang S, Zhou H, Fang D, Yue W, Chen C. Metabolomic Profiling in Combination with Data Association Analysis Provide Insights about Potential Metabolic Regulation Networks among Non-Volatile and Volatile Metabolites in Camellia sinensis cv Baijiguan. Plants. 2022; 11(19):2557. https://doi.org/10.3390/plants11192557
Chicago/Turabian StyleChen, Mingjie, Xiangrui Kong, Yi Zhang, Shiya Wang, Huiwen Zhou, Dongsheng Fang, Wenjie Yue, and Changsong Chen. 2022. "Metabolomic Profiling in Combination with Data Association Analysis Provide Insights about Potential Metabolic Regulation Networks among Non-Volatile and Volatile Metabolites in Camellia sinensis cv Baijiguan" Plants 11, no. 19: 2557. https://doi.org/10.3390/plants11192557
APA StyleChen, M., Kong, X., Zhang, Y., Wang, S., Zhou, H., Fang, D., Yue, W., & Chen, C. (2022). Metabolomic Profiling in Combination with Data Association Analysis Provide Insights about Potential Metabolic Regulation Networks among Non-Volatile and Volatile Metabolites in Camellia sinensis cv Baijiguan. Plants, 11(19), 2557. https://doi.org/10.3390/plants11192557






