A Small Gtp-Binding Protein GhROP3 Interacts with GhGGB Protein and Negatively Regulates Drought Tolerance in Cotton (Gossypium hirsutum L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Stress Treatments
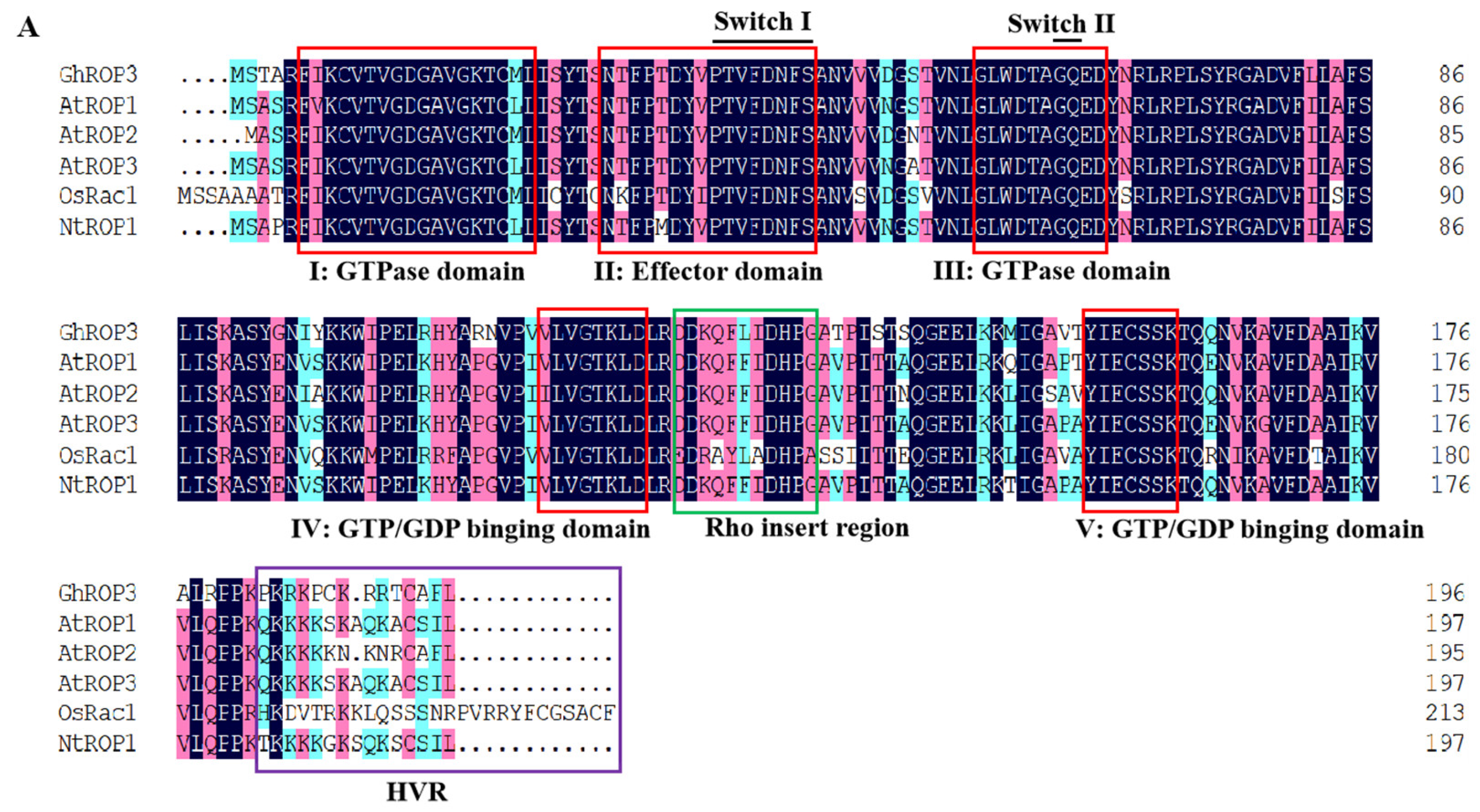
2.2. GhROP3 Bioinformatics Analysis
2.3. RNA Isolation and qRT-PCR
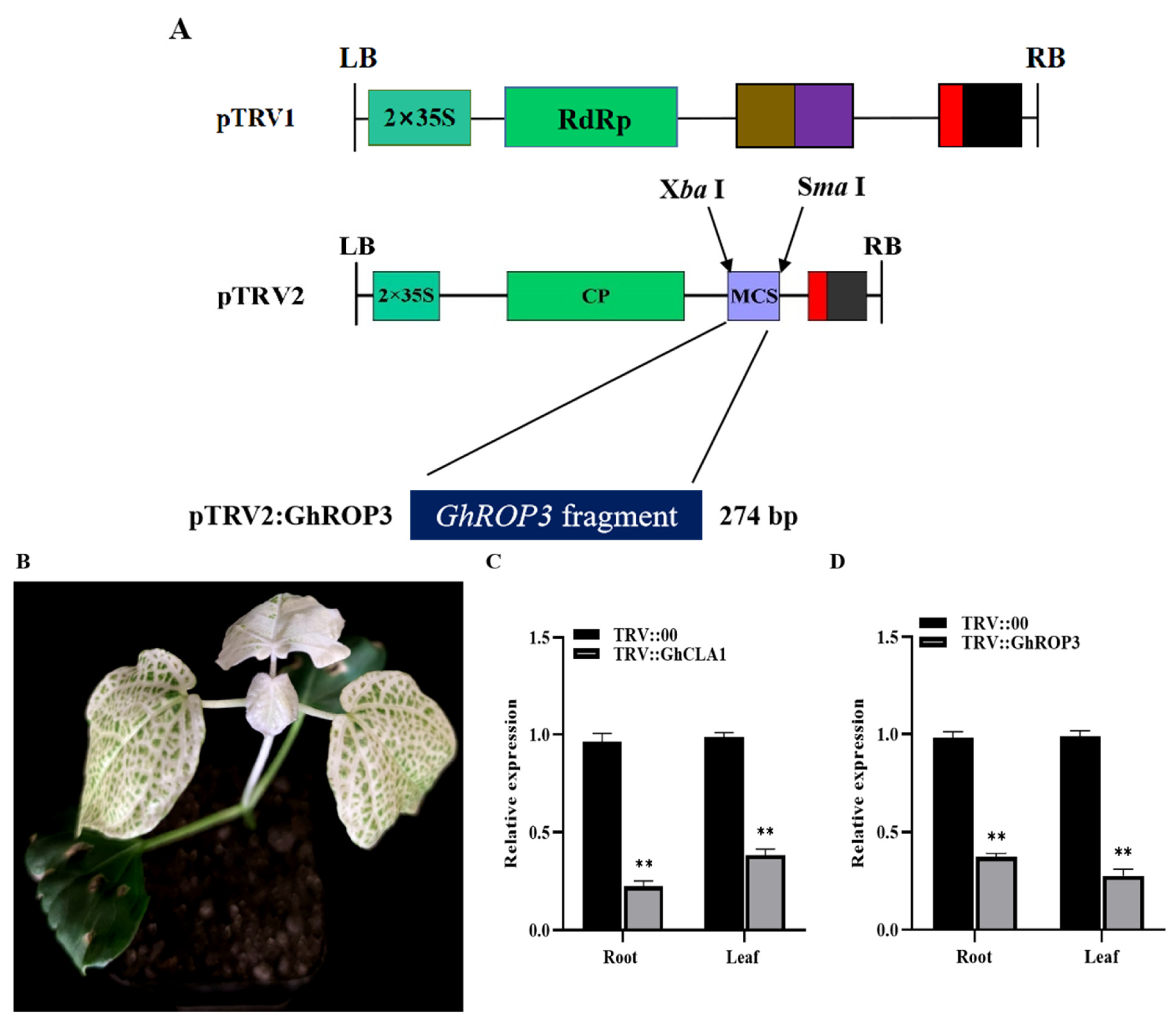
2.4. Construction of a Silencing Vector and VIGS in Cotton
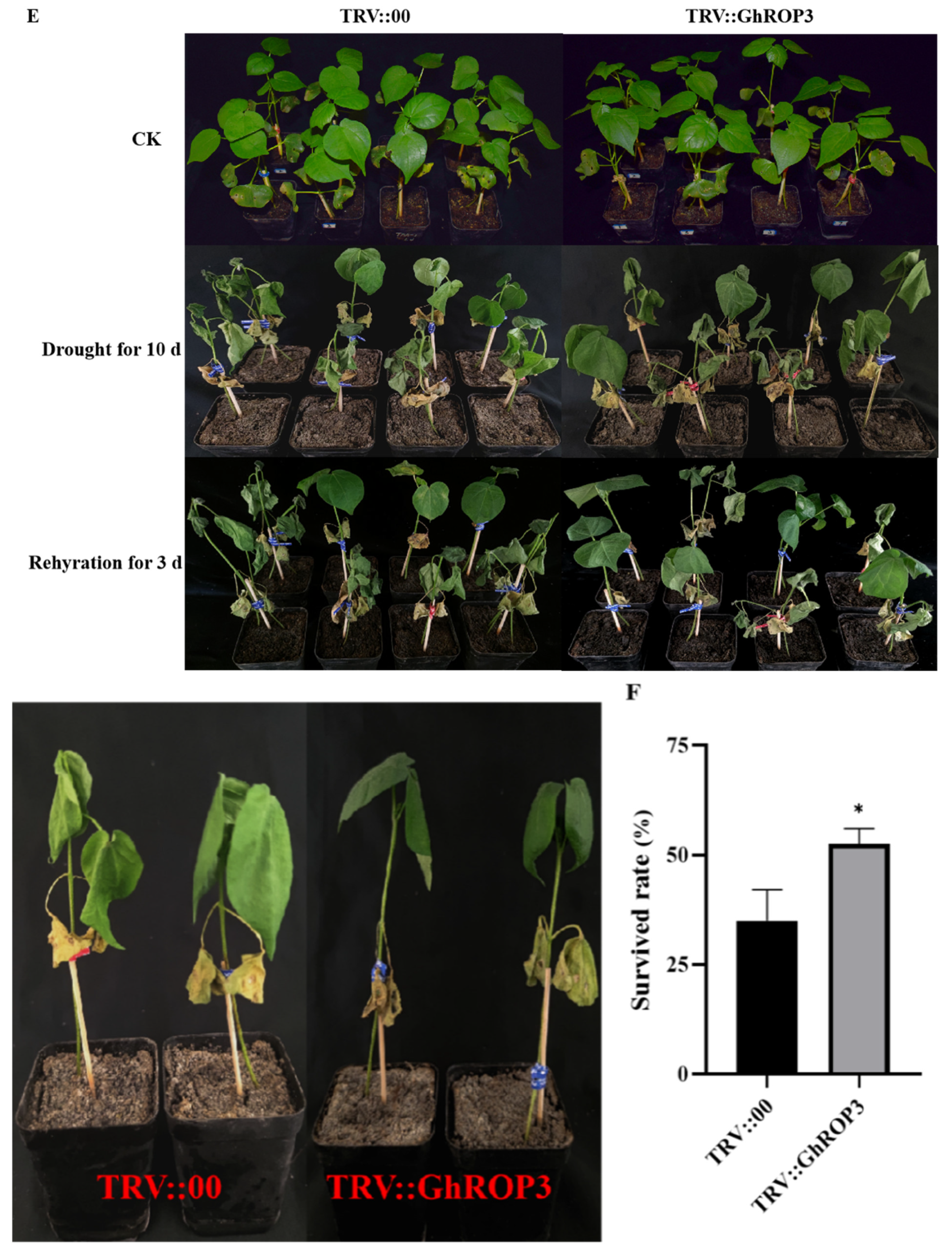
2.5. Drought Treatment and Determination of Physiological and Biochemical Indicators
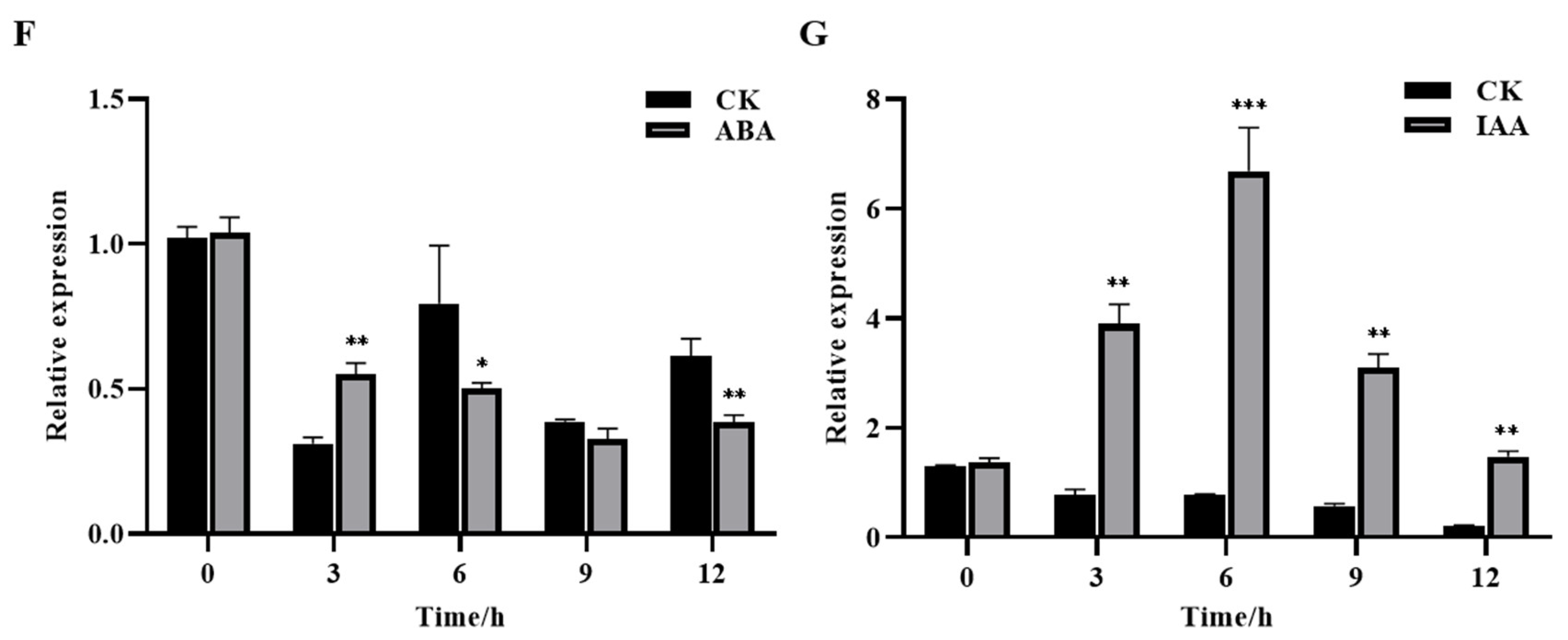
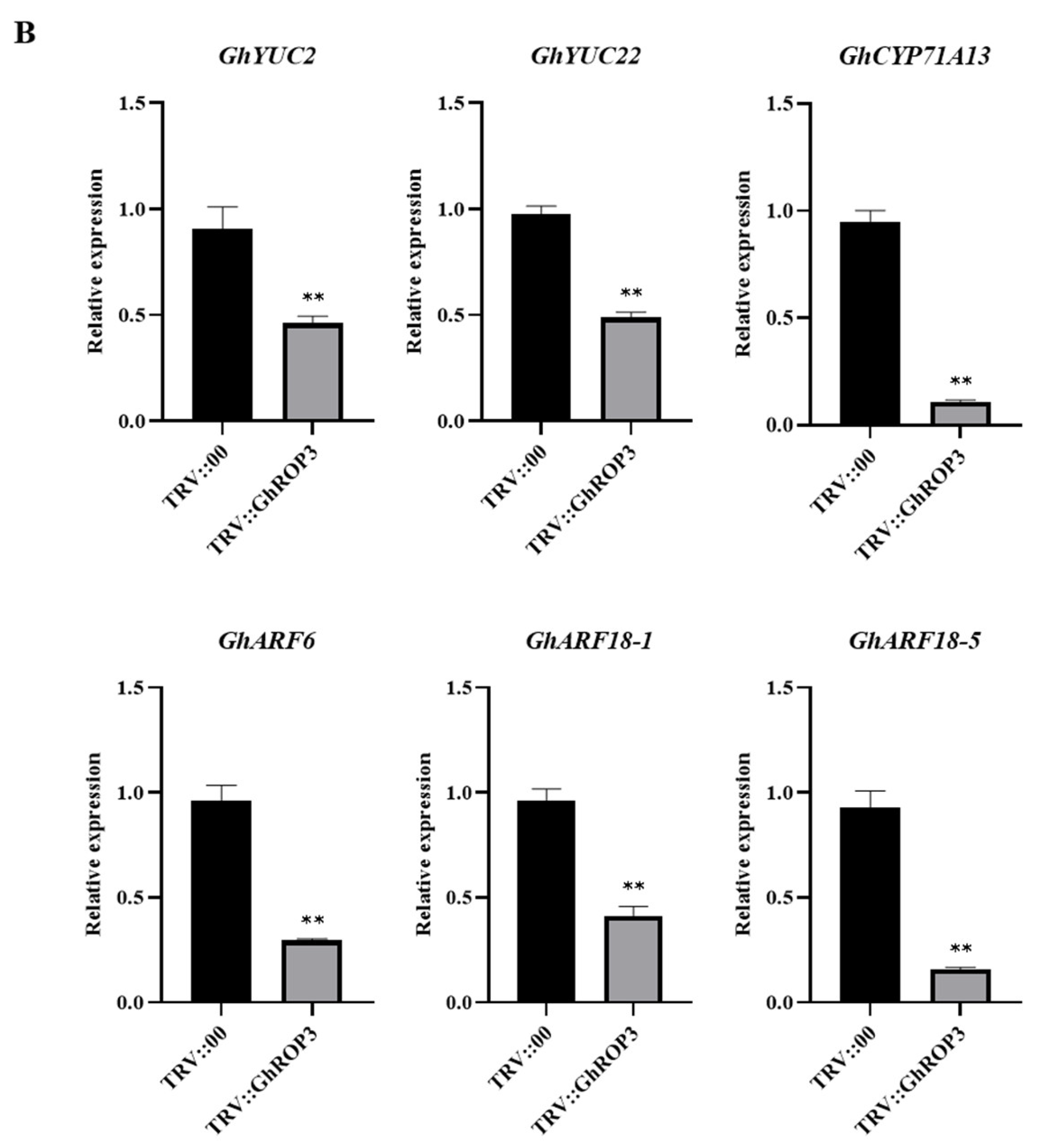
2.6. Expression Analysis of ABA and IAA Metabolism- and Signaling Pathway-Related Marker Genes after VIGS Treatment
2.7. Yeast Two-Hybrid Assay
2.8. Bimolecular Fluorescence Complementation Experiments
2.9. Statistical Analysis
3. Results
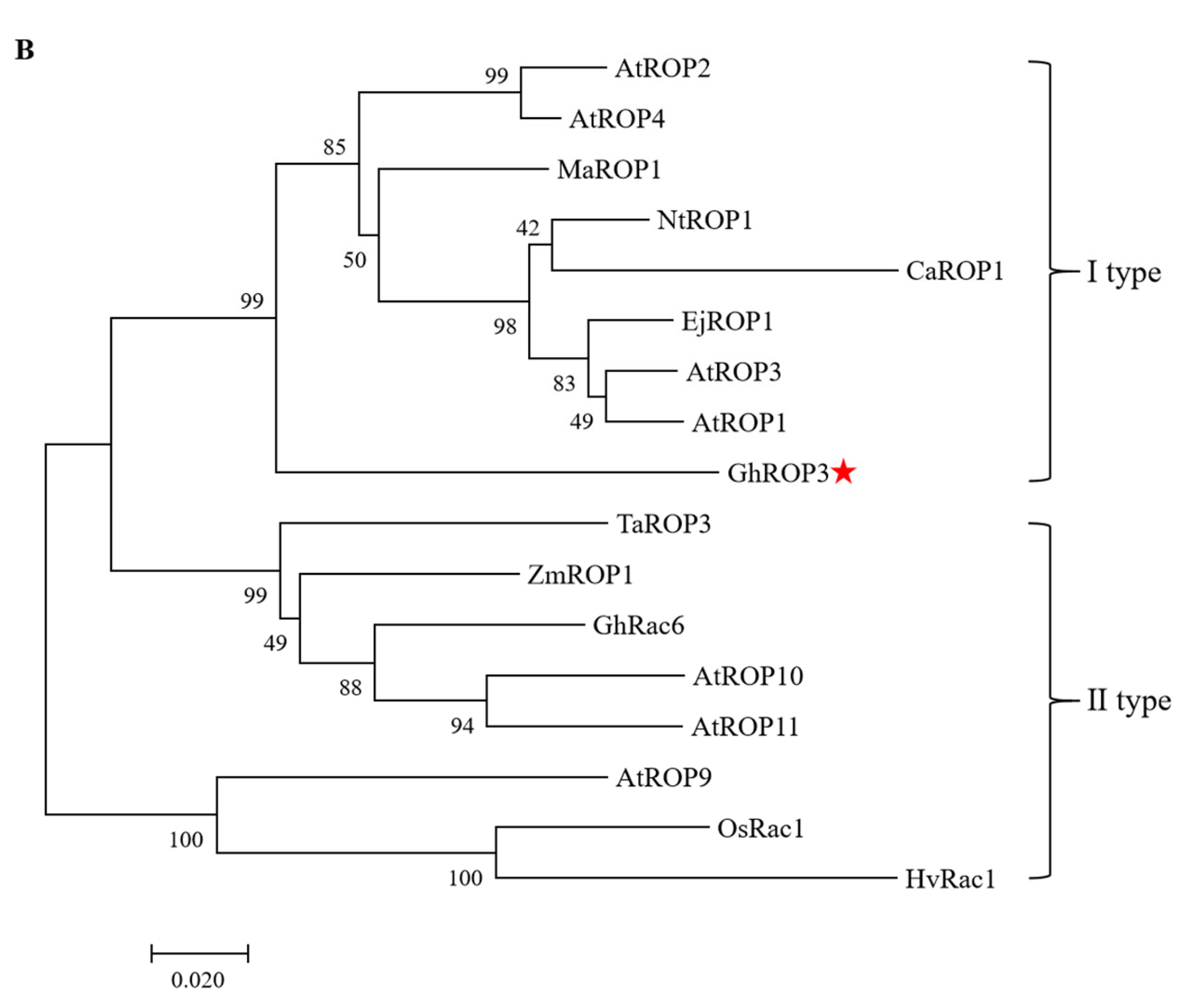
3.1. Phylogenetic Analysis of GhROP3
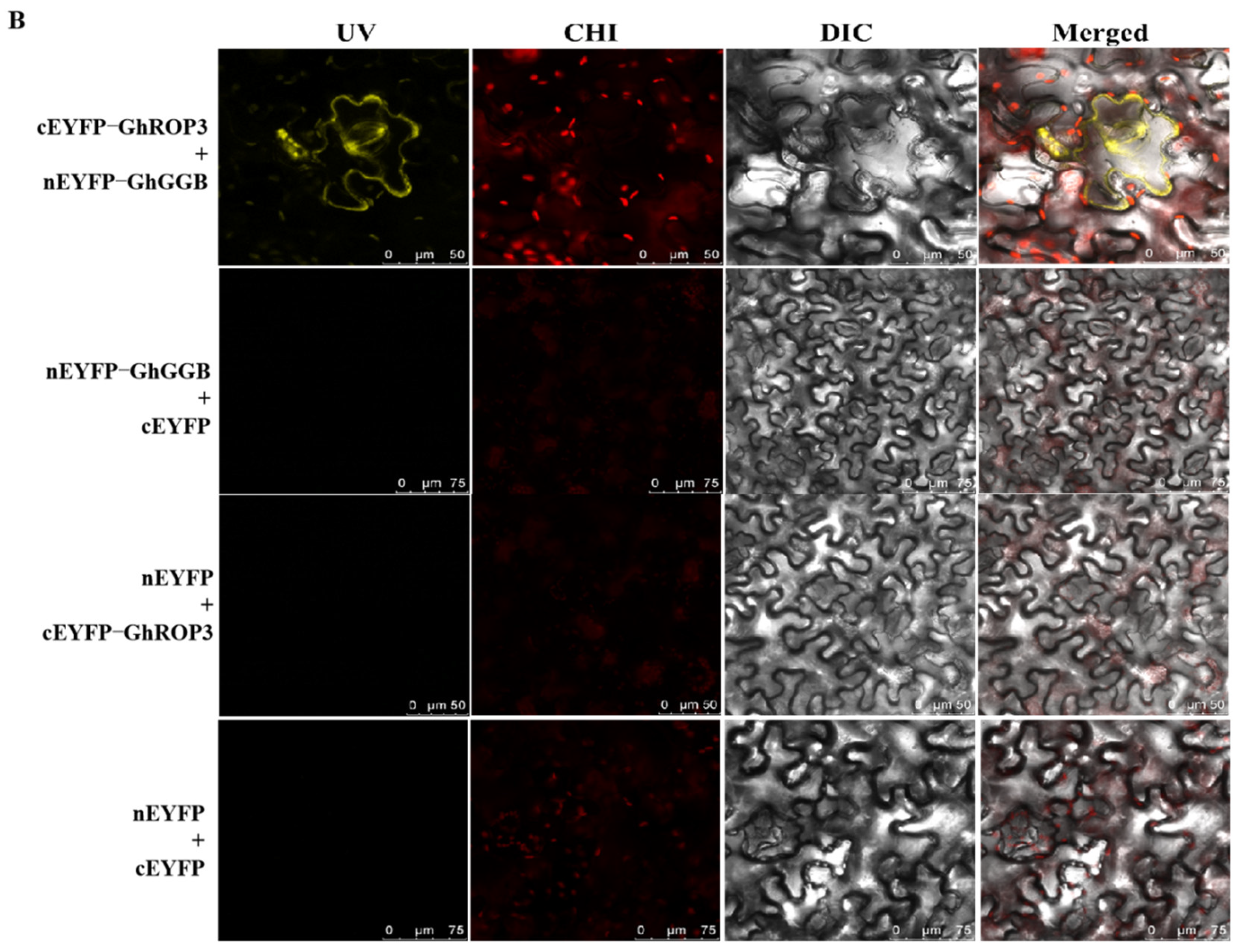
3.2. GhROP3 Protein Interacted with the GhGGB Protein
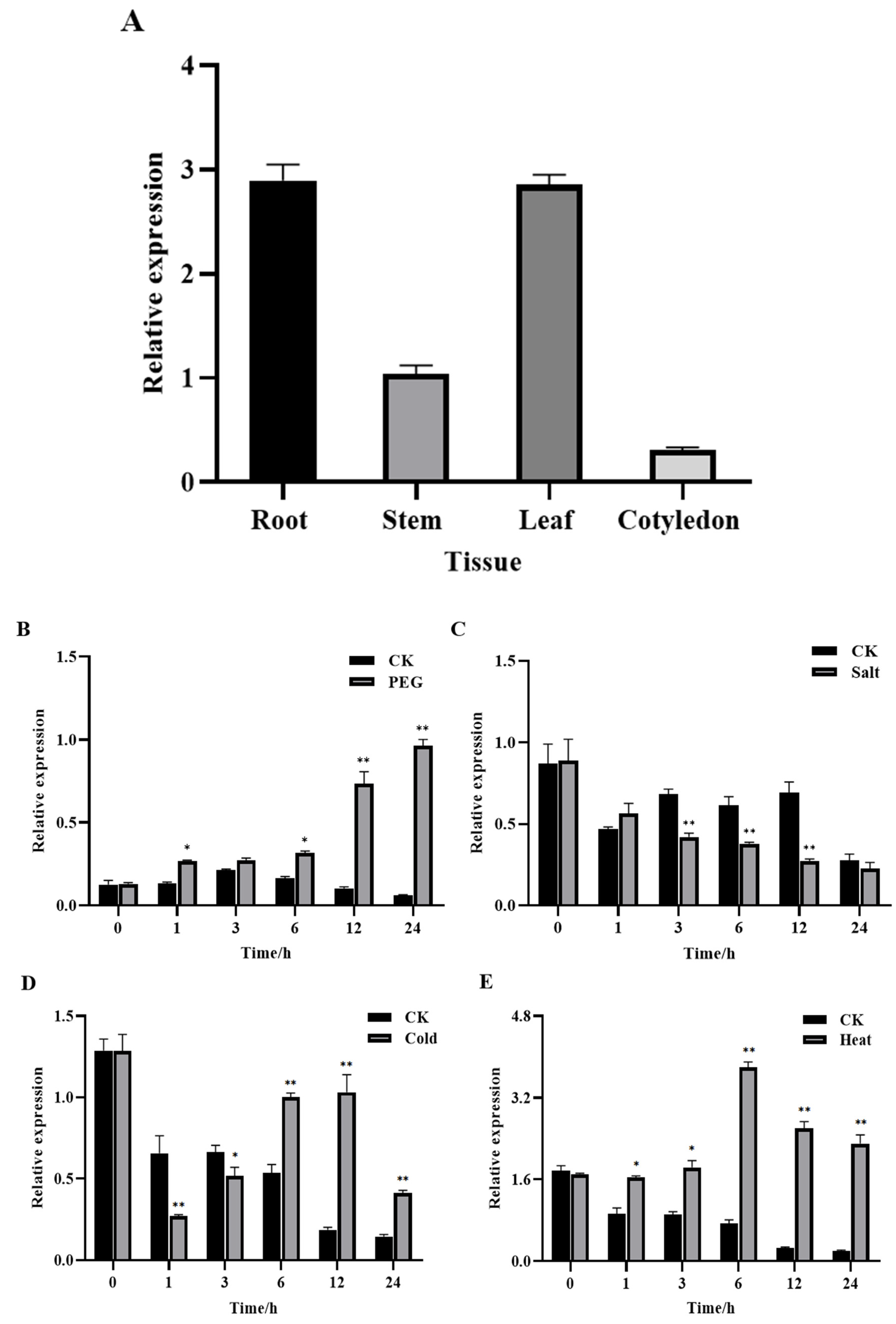
3.3. GhROP3 Was Expressed in Different Tissues of Cotton and Responded to Different Treatments
3.4. Silencing of the GhROP3 Gene Improved Cotton Resistance against Drought Stress
3.5. GhROP3 Negatively Regulated ABA Signaling Pathway, and Positively Regulated IAA Signaling Pathway
4. Discussion
4.1. GhROP3 Is a Prenylation Modification Substrate of GhGGB Protein
4.2. GhROP3 Played a Negative Role in the Response of Plants to Drought Stress
4.3. GhROP3 Mediated Drought Tolerance in Cotton by Participating in the Regulation of ABA and IAA Signaling Pathways
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lu, X.R.; Jia, X.Y.; Niu, J.H. The present situation and prospects of cotton industry development in China. Sci. Agric. Sin. 2018, 51, 26–36. [Google Scholar]
- Bhusal, N.; Lee, M.; Han, A.R.; Han, A.; Kim, H.S. Responses to drought stress in Prunus sargentii and Larix kaempferi seedlings using morphological and physiological parameters. For. Ecol. Manag. 2020, 465, 118099. [Google Scholar]
- Seleiman, M.F.; AlSuhaibani, N.; Ali, N.; Akmal, M.; Alotaibi, M.; Refay, Y.; Dindaroglu, T.; AbdulWajid, H.H.; Battaglia, M.L. Drought stress impacts on plants and different approaches to alleviate its adverse effects. Plants 2021, 10, 259. [Google Scholar] [CrossRef]
- Takai, Y.; Sasaki, T.; Matozaki, T. Small GTP-binding proteins. Phys. Rev. 2001, 81, 153–208. [Google Scholar] [CrossRef]
- Ono, E.; Wong, H.L.; Kawasaki, T.; Hasegawa, M.; Kodama, O.; Shimamoto, K. Essential role of the small GTPase Rac in disease resistance of rice. Proc. Natl. Acad. Sci. USA 2001, 98, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Bustelo, X.R.; Sauzeau, V.; Berenjeno, I.M. GTP-binding proteins of the Rho/Rac family: Regulation, effectors and functions in vivo. Bioessays 2007, 29, 356–370. [Google Scholar] [CrossRef] [PubMed]
- Sahai, E.; Marshall, C.J. RHO-GTPases and cancer. Nat. Rev. Cancer 2002, 2, 133–142. [Google Scholar] [CrossRef]
- Jaffe, A.B.; Hall, A. Rho GTPases: Biochemistry and biology. Annu. Rev. Cell Dev. Biol. 2005, 21, 247–269. [Google Scholar] [CrossRef]
- Ridley, A.J. Rho GTPases and actin dynamics in membrane protrusions and vesicle trafficking. Trends Cell. Biol. 2006, 16, 522–529. [Google Scholar] [CrossRef]
- Berken, A.; Wittinghofer, A. Structure and function of Rho-type molecular switches in plants. Plant Physiol. Biochem. 2008, 46, 380–393. [Google Scholar] [CrossRef]
- Winge, P.; Brembu, T.; Kristensen, R.; Bones, A.M. Genetic structure and evolution of RAC-GTPases in Arabidopsis thaliana. Genetics 2000, 156, 1959–1971. [Google Scholar] [CrossRef] [PubMed]
- Feiquelman, G.; Fu, Y.; Yalovsky, S. Rop GTPases structure-function and signaling pathways. Plant Physiol. 2018, 176, 57–79. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Watson, J. Molecular cloning and characterization of rho, aras-related small GTP-binding protein from the garden pea. Proc. Natl. Acad. Sci. USA 1993, 90, 8732–8736. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Kaneko-Kawano, T.; Shimamoto, K. Rho family GTPase dependent immunity in plants and animals. Front. Plant Sci. 2014, 5, 522. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.D.; Tang, R.J.; Zhang, X.; Luan, S.; Yu, F. Feronia receptor kinase at the crossroads of hormone signaling and stress responses. Plant Cell Physiol. 2017, 58, 1143–1150. [Google Scholar] [CrossRef]
- Zhang, B.; Yang, G.H.; Chen, Y.; Zhao, Y.H.; Gao, P.; Liu, B.; Wang, H.Y.; Zheng, Z.L. C-terminal domain (CTD) phosphatase links Rho GTPase signaling to Pol II CTD phosphorylation in Arabidopsis and yeast. Proc. Natl. Acad. Sci. USA 2016, 113, 8197–8206. [Google Scholar] [CrossRef]
- Schepetilnikov, M.; Makarian, J.; Srour, O.A.; Geldreich, A.; Yang, Z.Z.; Chicher, J.; Hammann, P.; Ryabova, L.A. GTPase ROP2 binds and promotes activation of target of rapamycin, TOR, in response to auxin. EMBO J. 2017, 36, 886–903. [Google Scholar] [CrossRef]
- Shi, B.B.; Wang, J.; Gao, H.F.; Yang, Q.C.; Wang, Y.; Day, B.; Ma, Q. The small GTP-binding protein TaRop10 interacts with TaTrxh9 and functions as a negative regulator of wheat resistance against the stripe rust. Plant Sci. 2021, 309, 110937. [Google Scholar] [CrossRef]
- Ma, Q.H.; Zhu, H.H.; Han, J.Q. Wheat ROP proteins modulate defense response through lignin metabolism. Plant Sci. 2017, 262, 32–38. [Google Scholar] [CrossRef]
- Qiu, A.L.; Liu, Z.Q.; Li, J.Z.; Chen, Y.S.; Guan, D.Y.; He, S.L. The ectopic expression of CaRop1 modulates the response of tobacco plants to Ralstonia solanacearum and aphids. Front. Plant Sci. 2016, 7, 1177. [Google Scholar] [CrossRef]
- Kiirika, L.M.; Bergmann, H.F.; Schikowsky, C.; Wimmer, D.; Korte, J.; Schmitz, U.; Niehaus, K.; Colditz, F. Silencing of the Rac1 GTPase MtROP9 in Medicago truncatula stimulates early mycorrhizal and oomycete root colonizations but negatively affects rhizobial infection. Plant Physiol. 2012, 159, 501–516. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, X.M.; Zhuang, R.; Wei, Z.T.; Shu, W.X.; Wang, X.J.; Kang, Z.S. TaRac6 is a potential susceptibility factor by regulating the ROS burst negatively in the Wheat-Puccinia striiformis f. sp. tritici interaction. Front. Plant Sci. 2020, 11, 716. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.W.; Zhang, X.L.; Na, R.; Yang, S.Q.; Tian, Z.M.; Zhao, Y.; Zhao, J. StRac1 plays an important role in potato resistance against Phytophthora infestans via regulating H2O2 production. J. Plant Physiol. 2020, 253, 153249. [Google Scholar] [CrossRef] [PubMed]
- Lemichez, E.; Wu, Y.; Sanchez, J.P.; Mettouchi, A.; Mathur, J.; Chua, N.H. Inactivation of AtRac1 by abscisic acid is essential for stomatal closure. Genes Dev. 2001, 15, 1808–1816. [Google Scholar] [CrossRef]
- Li, Z.X.; Kang, J.; Sui, N.; Liu, D. ROP11 GTPase is a Negative regulator of multiple ABA responses in Arabidopsis. J. Integr. Plant Biol. 2012, 54, 169–179. [Google Scholar] [CrossRef]
- Li, C.J.; Lu, H.M.; Li, W.; Yuan, M.; Fu, Y. A ROP2-RIC1 pathway fine-tunes microtubule reorganization for salt tolerance in Arabidopsis. Plant Cell Environ. 2017, 40, 1127–1142. [Google Scholar] [CrossRef]
- Zermiani, M.; Zonin, E.; Nonis, A.; Begheldo, M.; Ceccato, L.; Vezzaro, A.; Baldan, B.; Trentin, A.; Masi, A.; Pegoraro, M.; et al. Ethylene negatively regulates transcript abundance of ROP-GAP rheostat-encoding genes and affects apoplastic reactive oxygen species homeostasis in epicarps of cold stored apple fruits. J. Exp. Bot. 2015, 66, 7255–7270. [Google Scholar] [CrossRef]
- Miao, H.; Sun, P.; Liu, J.; Wang, J.Y.; Xu, B.Y.; Jin, Z.Q. Overexpression of a novel ROP gene from the banana (MaROP5g) confers increased salt stress tolerance. Int. J. Mol. Sci. 2018, 19, 3108. [Google Scholar] [CrossRef]
- Xu, X.H.; Ye, X.L.; Xing, A.Q.; Wu, Z.C.; Li, X.Y.; Shu, Z.F.; Wang, Y.H. Camellia sinensis small GTPase gene (CsRAC1) involves in response to salt stress, drought stress and ABA signaling pathway. Gene 2022, 821, 146318. [Google Scholar] [CrossRef]
- Wang, T.Z.; Xia, X.Z.; Zhao, M.G.; Tian, Q.Y.; Zhang, W.H. Expression of a Medicago falcata small GTPase gene, MfARL1 enhanced tolerance to salt stress in Arabidopsis thaliana. Plant Physiol. Biochem. 2013, 63, 227–235. [Google Scholar] [CrossRef]
- Cao, Y.R.; Li, Z.G.; Tao, C.; Zhang, Z.G.; Zhang, J.S.; Chen, S.Y. Overexpression of a tobacco small G protein gene NtRop1 caluses salt sensitivity and hydrogen peroxide productionin transgenic plants. Sci. China Ser. C Life Sci. 2008, 51, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Li, X.B.; Xiao, Y.H.; Luo, M.; Hou, L.; Li, D.M.; Luo, X.Y.; Pei, Y. Cloning and expression analysis of two Rac genes from cotton (Gossypium hirsutum L.). Acta Genet. Sin. 2005, 32, 72–78. [Google Scholar]
- Delmer, D.P.; Pear, J.R.; Andrawis, A.; Stalker, D.M. Genes encoding small GTP-binding proteins analogous to mammalian rac are preferentially expressed in developing cotton fibers. Mol. Gen. Genet. MGG 1995, 248, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Triplett, B.A. Characterization of GhRac1 GTPase expressed in developing cotton (Gossypium hirsutum L.) fibers. Biochim. Biophys. Acta 2004, 1679, 214–221. [Google Scholar] [CrossRef]
- Wang, Y.J. Functional Analysis of GhROP6 in Cotton Responsive to Verticillium Dahliae; Huazhong Agricultural University: Wuhan, China, 2017. [Google Scholar]
- Yang, Z.; Liu, J.J.; Luo, L.; Ye, S.; Yang, Y.Z.; Zhang, G.H.; Wang, X.P.; Zhang, J.M. The cotton GhRac6 gene encoding a plant ROP/RAC protein improves the plant defense response to aphid feeding. Plant Mol. Biol. Rep. 2018, 36, 888–896. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, Y.J.; Dai, P.H.; Ren, Y.P.; Wang, Q.; Liu, X.D. Cotton Bsr-k1 modulates lignin deposition participating in plant resistance against Verticillium dahliae and Fusarium oxysporum. J. Plant Growth Regul. 2021, 95, 283–292. [Google Scholar] [CrossRef]
- Ni, Z.Y.; Liu, N.; Yu, Y.H.; Bi, C.X.; Chen, Q.J.; Qu, Y.Y. The cotton 70-kDa heat shock protein GhHSP70–26 plays a positive role in the drought stress response. Environ. Exp. Bot. 2021, 191, 104628. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Mandel, M.A.; Feldmann, K.A.; Herrera-Estrella, L.; Rocha-Sosa, M.; León, P. CLA1, a novel gene required for chloroplast development, is highly conserved in evolution. Plant J. 2010, 9, 649–658. [Google Scholar] [CrossRef]
- Wei, T.L.; Wang, Y.; Xie, Z.Z.; Guo, D.Y.; Chen, C.W. Enhanced ROS scavenging and sugar accumulation contribute to drought tolerance of naturally occurring autotetraploids in Poncirus trifoliata. Plant Biotechnol. J. 2019, 17, 1394–1407. [Google Scholar] [CrossRef]
- Wang, X.Y.; Chen, B.Z.; Ma, C.K.; Qiao, K.K.; Li, Z.S.; Wang, J.S.; Peng, R.H.; Fan, S.L.; Ma, Q.F. Systematical characterization of YUCCA gene family in five cotton species, and potential functions of YUCCA22 gene in drought resistance of cotton. Ind. Crop. Prod. 2021, 162, 113290. [Google Scholar] [CrossRef]
- Li, Y.; Wugalihan, A.; Dai, P.H.; Liu, C.; Yao, Z.P.; Guo, W.Z.; Liu, X.D. Interaction between cotton small GTP binding protein GhROP4 and protein geranyl acyltransferase GhGGB. Mol. Plant Breed. 2020, 18, 8126–8130. [Google Scholar]
- Yang, M.; Hu, Y.; Li, J.Y.; Li, Y.; Liu, C.; Dai, P.H.; Liu, X.D. Analysis of interaction between ROP2 and GGB Proteins of Arabidopsis thaliana. Mol. Plant Breed. 2020, 18, 2216–2224. [Google Scholar]
- Crowell, D.N.; Huizinga, D.H. Protein isoprenylation: The fat of the matter. Trends Plant Sci. 2009, 14, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.D.; Chary, S.N.; Chernoff, E.A.; Zeng, Q.; Running, M.P.; Crowell, D.N. Protein geranylgeranyltransferase I is involved in specific aspects of abscisic acid and auxin signaling in Arabidopsis. J. Plant Physiol. 2005, 139, 722–733. [Google Scholar] [CrossRef] [PubMed]
- Andrews, M.; Huizinga, D.H.; Crowell, D.N. The CaaX specificities of Arabidopsis protein prenyltransferases explain era1 and ggb phenotypes. BMC Plant Biol. 2010, 10, 118. [Google Scholar] [CrossRef] [PubMed]
- Trainin, T.; Shmuel, M.; Delmer, D.P. In vitro prenylation of the small GTPase Rac13 of cotton. Plant Physiol. 1996, 112, 1491–1497. [Google Scholar] [CrossRef][Green Version]
- Li, J.B.; Luan, Y.S.; Liu, Z. Overexpression of SpWRKY1 promotes resistance to Phytophthora nicotianae and tolerance to salt and drought stress in transgenic tobacco. Physiol. Plant. 2015, 155, 248–266. [Google Scholar] [CrossRef]
- Khan, M.A.; Iqbal, H.S.M.A.; Akram, M.W. Characterization of drought tolerance in bread wheat genotypes using physiological indices. Gesunde Pflanz. 2022, 74, 467–475. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Xi, M.; Li, Y.; Cheng, Z.W.; Wang, S.; Kong, F.L. Improvement in salt tolerance of Iris pseudacorus L. in constructed wetland by exogenous application of salicylic acid and calcium chloride. J. Environ. Manag. 2021, 300, 113703. [Google Scholar] [CrossRef]
- Polle, A. Dissecting the superoxide dismutase-ascorbate glutathione pathway in chloroplasts by metabolic modeling. Computer simulations as a step towards flux analysis. Plant Physiol. 2001, 126, 445–462. [Google Scholar] [CrossRef] [PubMed]
- Bhusal, N.; Lee, M.; Lee, H.; Adhikari, A.; Han, A.R.; Han, A.; Kim, H.S. Evaluation of morphological, physiological, and biochemical traits for assessing drought resistance in eleven tree species. Sci. Total Environ. 2021, 779, 146466. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.A.D.; Suzuki, N.; Ciftci-Yilmaz, S.; Mittler, R. Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ. 2010, 33, 453–467. [Google Scholar] [CrossRef] [PubMed]
- Khan, T.A.; Fariduddin, Q.; Yusuf, M. Low-temperature stress: Is phytohormones application a remedy? Environ. Sci. Pollut. Res. 2017, 24, 21574–21590. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Dominguez, C.M.; Buckley, T.N.; Egea, G.; De, C.A.; Hernandez-Santana, V.; Martorell, S.; Diaz-Espejo, A. Most stomatal closure in woody species under moderate drought can be explained by stomatal responses to leaf turgor. Plant Cell Environ. 2016, 39, 2014–2016. [Google Scholar] [CrossRef]
- Meher, P.S.; Shivakrishna, P.; Reddy, K.A.; Rao, D.M. Effect of PEG-6000 imposed drought stress on RNA content, relative water content (RWC), and chlorophyll content in peanut leaves and roots. Saudi J. Biol Sci. 2018, 25, 285–289. [Google Scholar] [CrossRef]
- Drake, P.L.; Froend, R.H.; Franks, P.J. Smaller, faster stomata: Scaling of stomatal size, rate of response, and stomatal conductance. J. Exp. Bot. 2013, 64, 495–505. [Google Scholar] [CrossRef]
- Bhusal, N.; Han, S.G.; Yoon, T.M. Impact of drought stress on photosynthetic response, leaf water potential, and stem sap flow in two cultivars of bi-leader apple trees (Malus×domestica Borkh.). Sci. Hortic. 2019, 246, 535–543. [Google Scholar] [CrossRef]
- Li, H.; Shen, J.J.; Zheng, Z.L.; Lin, Y.; Yang, Z. The Rop GTPase switch controls multiple developmental processes in Arabidopsis. J. Plant Physiol. 2001, 126, 670–684. [Google Scholar] [CrossRef]
- Zheng, Z.L.; Nafisi, M.; Tam, A.; Li, H.; Crowell, D.N.; Chary, S.N.; Schroeder, J.I.; Shen, J.J.; Yang, Z.B. Plasma membrane-associated ROP10 small GTPase is a specific negative regulator of abscisic acid responses in Arabidopsis. Plant Cell. 2002, 14, 2787–2797. [Google Scholar] [CrossRef]
- Niu, J. Preliminary Study of Banana Small GTPases Gene MaROP1 in Transgenic Arabidopsis; College of Agricalture Hainan University: Haikou, China, 2011. [Google Scholar]
- Pei, X.X.; Wang, X.Y.; Fu, G.Y.; Chen, B.J.; Nazir, M.F.; Pan, Z.E.; He, S.P.; Du, X.M. Identification and functional analysis of 9-cis-epoxy carotenoid dioxygenase (NCED) homologs in G. hirsutum. Int. J. Biol. Macromol. 2021, 182, 298–310. [Google Scholar] [CrossRef] [PubMed]
- He, R.R.; Zhuang, Y.; Cai, Y.M.; Agüero, C.B.; Liu, S.L.; Wu, J.; Deng, S.H.; Walker, M.A.; Lu, J.; Zhang, Y.L. Overexpression of 9-cis-epoxycarotenoid dioxygenase cisgene in grapevine increases drought tolerance and results in pleiotropic effects. Front. Plant Sci. 2018, 9, 970. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Andújar, C.; Martínez-Pérez, A.; Ferrández-Ayela, A.; Albacete, A.; Martínez-Melgarejo, P.A.; Dodd, I.C.; Thompson, A.J.; Pérez-Pérez, J.M.; Pérez-Alfocea, F. Impact of overexpression of 9-cis-epoxycarotenoid dioxygenase on growth and gene expression under salinity stress. Plant Sci. 2020, 295, 110268. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, R. Abscisic acid synthesis and response. Arab. Book 2013, 11, e0166. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Xiong, L.M.; Li, W.B.; Zhu, J.K.; Zhu, J.H. The plant cuticle is required for osmotic stress regulation of abscisic acid biosynthesis and osmotic stress tolerance in Arabidopsis. Plant Cell 2011, 23, 1971–1984. [Google Scholar] [CrossRef]
- Robert, H.S.; Chulmin, P.; Loreto, G.C.; Wójcikowska, B.; Pěnčík, A.; Novák, O.; Chen, J.Y.; Grunewald, W.; Dresselhaus, T.; Friml, J.; et al. Maternal auxin supply contributes to early embryo patterning in Arabidopsis. Nat. Plants. 2018, 4, 548–553. [Google Scholar] [CrossRef]
- Bielach, A.; Hrtyan, M.; Tognetti, V.B. Plants under stress: Involvement of auxin and cytokinin. Int. J. Mol. Sci. 2017, 18, 1427. [Google Scholar] [CrossRef]
- Won, C.; Shen, X.L.; Mashiguchi, K.; Zheng, Z.Y.; Dai, X.H.; Cheng, Y.F.; Kasahara, H.; Kamiya, Y.J.; Chory, J.; Zhao, Y.D. Conversion of tryptophan to indole-3-acetic acid by TRYPTOPHAN AMINOTRANSFERASES OF ARABIDOPSIS and YUCCAs in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 18518–18523. [Google Scholar] [CrossRef]
- Tiwari, S.B.; Hagen, G.; Guilfoyle, T. The roles of auxin response factor domains in auxin-responsive transcription. Plant Cell 2003, 15, 533–543. [Google Scholar] [CrossRef]
- Zhang, A.Y.; Xu, Y.; Lu, J.; Song, F.Y.; Sun, J.H.; Wang, C.; Lian, J.; Zhao, L.L.; Zhao, B.C. OsIAA20, an Aux/IAA protein, mediates abiotic stress tolerance in rice through an ABA pathway. Plant Sci. 2021, 308, 110903. [Google Scholar] [CrossRef]
- Wang, F.B.; Niu, H.F.; Xin, D.Q.; Long, Y.; Wang, G.P.; Liu, Z.M.; Li, G.; Zhang, F.; Qi, M.Y.; Ye, Y.X.; et al. OsIAA18, an Aux/IAA transcription factor gene, is involved in salt and drought tolerance in rice. Front. Plant Sci. 2021, 12, 738660. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Claes, A.R.; Grebe, T.; Hermkes, R.; Viotti, C.; Ikeda, Y.; Grebe, M. Auxin and ROP GTPase signaling of polar nuclear migration in root epidermal hair cells. Plant Physiol. 2018, 176, 378–391. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.B.; Liu, H.L.; Chen, M.; Li, X.J.; Wang, M.Y.; Yang, Y.L.; Wang, C.L.; Huang, J.Q.; Liu, G.L.; Liu, Y.T.; et al. ROP3 GTPase contributes to polar auxin transport and auxin responses and is important for embryogenesis and seedling growth in Arabidopsis. Plant Cell 2014, 26, 3501–3518. [Google Scholar] [CrossRef]
- Nagawa, S.; Xu, T.D.; Lin, D.S.; Dhonukshe, P.; Zhang, X.X.; Friml, J.; Scheres, B.; Fu, Y.; Yang, Z.B. ROP GTPase-dependent actin microfilaments promote PIN1 polarization by localized inhibition of clathrin-dependent endocytosis. PLoS Biol. 2012, 10, e1001299. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Z.; Lei, J.; Dai, P.; Liu, C.; Wugalihan, A.; Liu, X.; Li, Y. A Small Gtp-Binding Protein GhROP3 Interacts with GhGGB Protein and Negatively Regulates Drought Tolerance in Cotton (Gossypium hirsutum L.). Plants 2022, 11, 1580. https://doi.org/10.3390/plants11121580
Hu Z, Lei J, Dai P, Liu C, Wugalihan A, Liu X, Li Y. A Small Gtp-Binding Protein GhROP3 Interacts with GhGGB Protein and Negatively Regulates Drought Tolerance in Cotton (Gossypium hirsutum L.). Plants. 2022; 11(12):1580. https://doi.org/10.3390/plants11121580
Chicago/Turabian StyleHu, Ziyao, Jianfeng Lei, Peihong Dai, Chao Liu, Abuduweili Wugalihan, Xiaodong Liu, and Yue Li. 2022. "A Small Gtp-Binding Protein GhROP3 Interacts with GhGGB Protein and Negatively Regulates Drought Tolerance in Cotton (Gossypium hirsutum L.)" Plants 11, no. 12: 1580. https://doi.org/10.3390/plants11121580
APA StyleHu, Z., Lei, J., Dai, P., Liu, C., Wugalihan, A., Liu, X., & Li, Y. (2022). A Small Gtp-Binding Protein GhROP3 Interacts with GhGGB Protein and Negatively Regulates Drought Tolerance in Cotton (Gossypium hirsutum L.). Plants, 11(12), 1580. https://doi.org/10.3390/plants11121580




