Phloem-Feeding Herbivores Affect Floral Development and Reproduction in the Etruscan Honeysuckle (Lonicera etrusca Santi)
Abstract
1. Introduction
2. Results
2.1. Frequency of Herbivory by Aphids
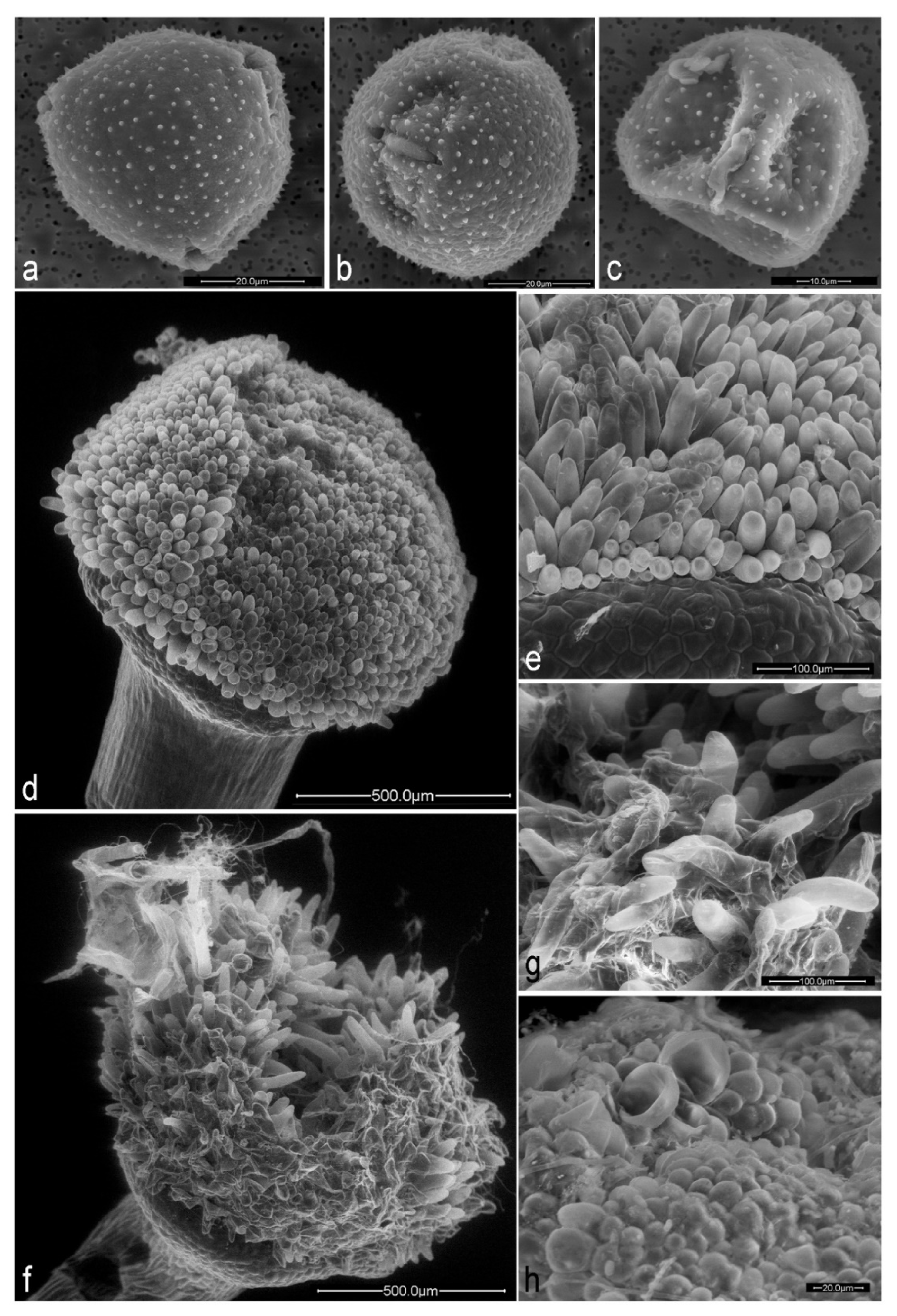
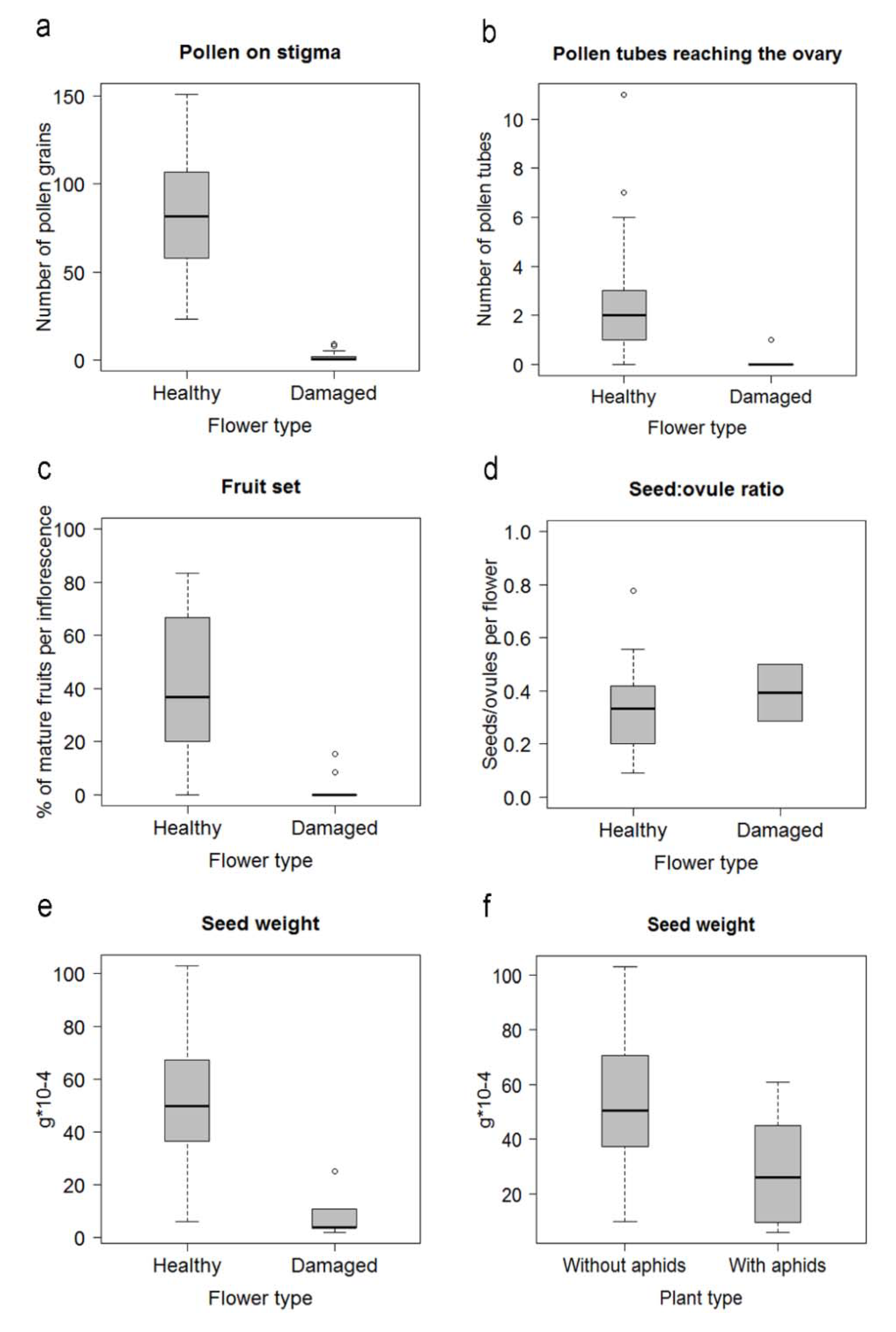
2.2. Effects of Herbivory by Aphids on Floral Traits
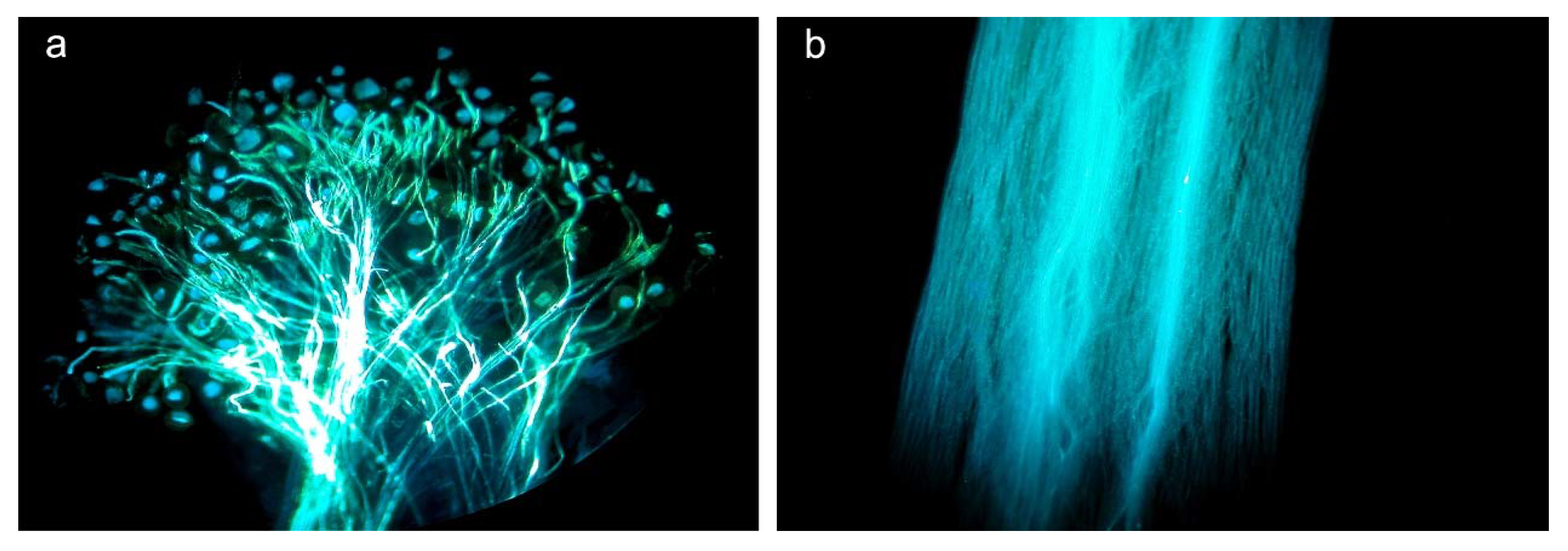
2.3. Effects of Herbivory on Plant Reproduction
3. Discussion
3.1. Effects of Herbivory on Floral Development
3.2. Effects of Herbivory on Plant Reproduction
4. Materials and Methods
4.1. Study Sites
4.2. Study System
4.3. Frequency of Herbivory by Aphids
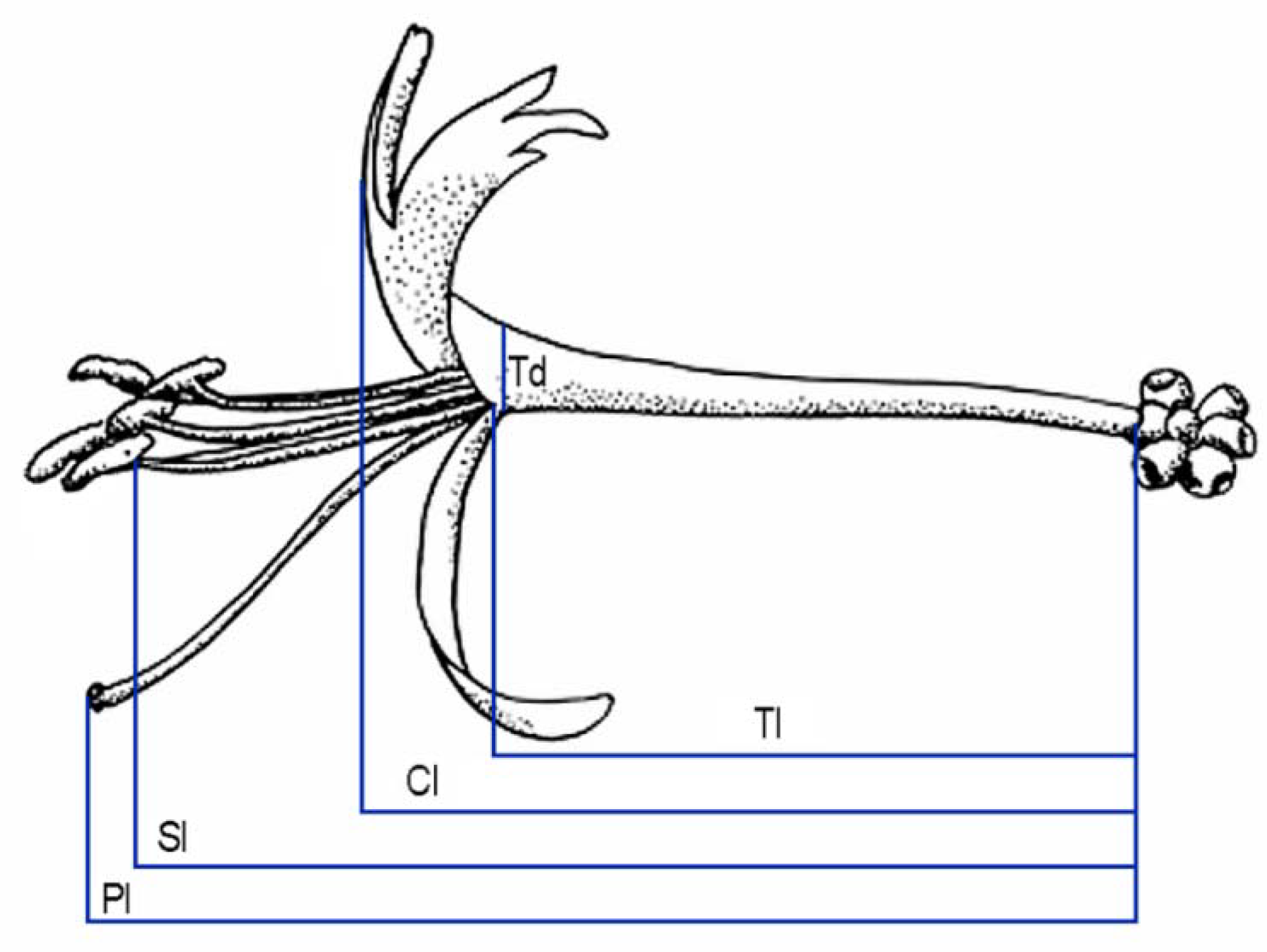
4.4. Effect of Herbivory on Floral Development
4.5. Effect of Herbivory on Plant Reproduction
4.6. Data Analyses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Strauss, S.Y.; Irwin, R.E. Ecological and evolutionary consequences of multispecies plant-animal interactions. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 435–466. [Google Scholar] [CrossRef]
- Genini, J.; Morellato, L.P.C.; Guimarães, P.R., Jr.; Olesen, J.M. Cheaters in mutualism networks. Biol. Lett. 2010, 6, 494–497. [Google Scholar] [CrossRef] [PubMed]
- Faegri, K.; van der Pijl, L. The Principles of Pollination Ecology; Pergamon Press: Oxford, UK, 1979. [Google Scholar]
- Ollerton, J.; Winfree, R.; Tarrant, S. How many flowering plants are pollinated by animals? Oikos 2011, 120, 321–326. [Google Scholar] [CrossRef]
- Strauss, S.Y.; Conner, J.K.; Rush, S.L. Foliar herbivory affects floral characters and plant attractiveness to pollinators: Implications for male and female plant fitness. Am. Nat. 1996, 147, 1098–1107. [Google Scholar] [CrossRef]
- Wise, M.J.; Cummins, J.J. Strategies of Solanum carolinense for regulating maternal investment in response to foliar and floral herbivory. J. Ecol. 2006, 94, 629–636. [Google Scholar] [CrossRef]
- Lehtilä, K.; Strauss, S.Y. Leaf damage by herbivores affects attractiveness to pollinators in wild radish. Raphanus raphanistrum. Oecologia 1997, 111, 396–403. [Google Scholar]
- Strauss, S.Y. Floral characters link herbivores, pollinators, and plant fitness. Ecology 1997, 78, 1640–1645. [Google Scholar] [CrossRef]
- Krupnick, G.A.; Weis, A.E.; Campbell, D.R. The consequences of floral herbivory for pollinator service to Isomeris arborea. Ecology 1999, 80, 125–134. [Google Scholar] [CrossRef]
- Strauss, S.Y.; Agrawal, A.A. The ecology and evolution of plant tolerance to herbivory. Trends Ecol. Evol. 1999, 14, 179–185. [Google Scholar] [CrossRef]
- Brody, A.K.; Irwin, R.E. When resources don’t rescue: Flowering phenology and species interactions affect compensation to herbivory in Ipomopsis aggregata. Oikos 2012, 121, 1424–1434. [Google Scholar] [CrossRef]
- Strauss, S.Y. Direct, indirect, and cumulative effects of three native herbivores on a shared host plant. Ecology 1991, 72, 543–558. [Google Scholar] [CrossRef]
- Poveda, K.; Steffan-Dewenter, I.; Scheu, S.; Tscharntke, T. Effects of below-and above-ground herbivores on plant growth, flower visitation and seed set. Oecologia 2003, 135, 601–605. [Google Scholar] [CrossRef]
- Dixon, A.F.G. The role of aphids in wood formation. I. The effect of the sycamore aphid, Dreopanosiphum platanoides (Schr.)(Aphididae), on the growth of sycamore, Acer pseudoplatanus (L.). J. Appl. Ecol. 1971, 8, 165–179. [Google Scholar] [CrossRef]
- Larson, K.C. The impact of two gall-forming arthropods on the photosynthetic rates of their hosts. Oecologia 1998, 115, 161–166. [Google Scholar] [CrossRef]
- Power, A.G.; Flecker, A.S. Virus specificity in disease systems: Are species redundant? In The Importance of Species: Perspectives on Expendability and Triage; Kareiva, P., Levin, S.A., Eds.; Princeton University Press: Princeton, NJ, USA, 2003; pp. 330–346. [Google Scholar]
- Van Emden, H.F.; Harrington, R. Aphids as Crop Pests; CABI: Wallingford, UK, 2007. [Google Scholar]
- Silveira, T.A.; Sanches, P.A.; Zazycki, L.C.F.; Costa-Lima, T.C.; Cabezas-Guerrero, M.F.; Favaris, A.P.; Goulart, J.M.; Bento, S.; Santana, A.E.G. Phloem-feeding herbivory on flowering melon plants enhances attraction of parasitoids by shifting floral to defensive volatiles. Arthropod Plant Interact. 2018, 12, 751–760. [Google Scholar] [CrossRef]
- Watanabe, S.; Murakami, Y.; Hasegawa, E. Effects of aphid parasitism on host plant fitness in an aphid-host relationship. PLoS ONE 2018, 13, e0202411. [Google Scholar] [CrossRef] [PubMed]
- Snow, A.A.; Stanton, M.L. Aphids limit fecundity of a weedy annual (Raphanus sativus). Am. J. Bot. 1988, 75, 589–593. [Google Scholar] [CrossRef]
- Moran, P.J.; Thompson, G.A. Molecular responses to aphid feeding in Arabidopsis in relation to plant defence pathways. Plant Physiol. 2001, 125, 1074–1085. [Google Scholar] [CrossRef]
- Goggin, F.L. Plant-aphid interactions: Molecular and ecological perspectives. Curr. Opin. Plant Biol. 2007, 10, 399–408. [Google Scholar] [CrossRef]
- Guitián, P.; Guitián, J.; Navarro, L. Pollen transfer and diurnal versus nocturnal pollination in Lonicera etrusca. Acta Oecol. 1993, 14, 219–227. [Google Scholar]
- Jordano, P. Reproductive biology of three species of the genus Lonicera (Caprifoliaceae) in the Sierra de Cazorla. Anales del Jardín Botánico de Madrid 1990, 48, 31–52. [Google Scholar]
- Rojas-Nossa, S.V.; Sánchez, J.M.; Navarro, L. Nectar robbing and plant reproduction: An interplay of positive and negative effects. Oikos 2021. [Google Scholar] [CrossRef]
- Douglas, A.E. Phloem-sap feeding by animals: Problems and solutions. J. Exp. Bot. 2006, 57, 747–754. [Google Scholar] [CrossRef]
- Züst, T.; Agrawal, A.A. Trade-offs between plant growth and defense against insect herbivory: An emerging mechanistic synthesis. Ann. Rev. Plant. Biol. 2017, 68, 513–534. [Google Scholar] [CrossRef]
- Lucas-Barbosa, D.; van Loon, J.J.; Dicke, M. The effects of herbivore-induced plant volatiles on interactions between plants and flower-visiting insects. Phytochemistry 2011, 72, 1647–1654. [Google Scholar] [CrossRef] [PubMed]
- Mothershead, K.; Marquis, R.J. Fitness impacts of herbivory through indirect effects on plant-pollinator interactions in Oenothera macrocarpa. Ecology 2000, 81, 30–40. [Google Scholar] [CrossRef]
- Kennedy, J.S.; Stroyan, H.L.G. Biology of aphids. Annu. Rev. Entomol. 1959, 4, 139–160. [Google Scholar] [CrossRef]
- Pickett, J.A.; Wadhams, L.J.; Woodcock, C.M.; Hardie, J. The chemical ecology of aphids. Annu. Rev. Entomol. 1992, 37, 67–90. [Google Scholar] [CrossRef]
- Smith, C.M.; Boyko, E.V. The molecular bases of plant resistance and defense responses to aphid feeding: Current status. Entomol. Exp. Appl. 2007, 122, 1–16. [Google Scholar] [CrossRef]
- Rojas-Nossa, S.V. Ecological and Evolutionary Implications of Nectar Robbing. Ph.D. Thesis, University of Vigo, Vigo, Spain, 2015. [Google Scholar]
- Koricheva, J. Meta-analysis of sources of variation in fitness costs of plant antiherbivore defenses. Ecology 2002, 83, 176–190. [Google Scholar] [CrossRef]
- Ohm, J.R.; Miller, T.E. Balancing anti-herbivore benefits and anti-pollinator costs of defensive mutualists. Ecology 2014, 95, 2924–2935. [Google Scholar] [CrossRef]
- Marden, J.H. Remote perception of floral nectar by bumblebees. Oecologia 1984, 64, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Goulson, D.; Hawson, S.A.; Stout, J.C. Foraging bumblebees avoid flowers already visited by conspecifics or by other bumblebee species. Anim. Behav. 1998, 55, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Jersáková, J.; Johnson, S.D.; Kindlmann, P. Mechanisms and evolution of deceptive pollination in orchids. Biol. Rev. 2006, 81, 219–235. [Google Scholar] [CrossRef]
- Rojas-Nossa, S.V.; Sánchez, J.M.; Navarro, L. Nectar robbing: A common phenomenon mainly determined by accessibility constraints, nectar volume and density of energy rewards. Oikos 2016, 125, 1044–1055. [Google Scholar] [CrossRef]
- Blackman, R.L.; Eastop, V.F. Aphids on the World’s Herbaceous Plants and Shrubs; Volume 1—Host Lists and Keys; Volume 2—The aphids; John Wiley & Sons: Chichester, UK, 2006. [Google Scholar]
- Alford, D.V. Pests of Ornamental Trees, Shrubs and Flowers: A Colour Handbook; Manson Publishing: London, UK, 2012. [Google Scholar]
- Rojas-Nossa, S.V.; Sánchez, J.M.; Navarro, L. Effects of nectar robbing on male and female reproductive success of a pollinator-dependent plant. Ann. Bot. 2016, 117, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Diggle, P.K. Architectural effects and the interpretation of patterns of fruit and seed development. Ann. Rev. Ecol. Syst. 1995, 26, 531–552. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]

| Floral Trait 1 | Flower Type Mean ± S. D. (Sample Size) | Paired Student’s t-Test | |
|---|---|---|---|
| Healthy | Damaged | ||
| Nectar volume (µL) | 4.7 ± 4.1 (83) | 0 (27) | |
| Sugar concentration (◦Brix) | 21.1 ± 6.3 (83) | - | |
| Total corolla length | 30.9 ± 2.6 (83) | 12.5 ± 2.3 (63) | t = −14.2, p = 0.001 |
| Tube length | 25.6 ± 1.8 (83) | 9.2 ± 3.3 (63) | t = −14.3, p < 0.001 |
| Tube diameter | 2.4 ± 0.3 (83) | 1.5 ± 0.3 (55) | t = −10.9, p < 0.001 |
| Pistil length | 42.7 ± 2.8 (83) | 14.8 ± 3.7 (63) | t = −9.5, p < 0.001 |
| Stamens length | 39.4 ± 3.0 (83) | 12.6 ± 3.0 (63) | t = −13.9, p < 0.001 |
| Ovules/flower | 11 ± 1.2 (20) | 8.8 ± 2.3 (20) | t = 3.67, p = 0.001 |
| Ovule length | 1.3 ± 0.1 (100) | 1.1 ± 0.1 (100) | t = 5.1, p < 0.001 |
| Ovule width | 0.8 ± 0.1 (100) | 0.6 ± 0.1 (100) | t = 4.9, p < 0.001 |
| Pollen grains per anther | 2362 ± 366 (20) | 1447.4 ± 757 (20) | t = −4.8, p < 0.001 |
| Pollen equatorial axis (µm) | 71.1 ± 3.5 (50) | 59.4 ± 5.3 (50) | t = 9.7, p < 0.001 |
| Pollen polar axis (µm) | 63.2 ± 3.4 (39) | 55.2 ± 5.2 (50) | t = 7.7, p < 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rojas-Nossa, S.V.; Sánchez, J.M.; Navarro, L. Phloem-Feeding Herbivores Affect Floral Development and Reproduction in the Etruscan Honeysuckle (Lonicera etrusca Santi). Plants 2021, 10, 815. https://doi.org/10.3390/plants10040815
Rojas-Nossa SV, Sánchez JM, Navarro L. Phloem-Feeding Herbivores Affect Floral Development and Reproduction in the Etruscan Honeysuckle (Lonicera etrusca Santi). Plants. 2021; 10(4):815. https://doi.org/10.3390/plants10040815
Chicago/Turabian StyleRojas-Nossa, Sandra V., José María Sánchez, and Luis Navarro. 2021. "Phloem-Feeding Herbivores Affect Floral Development and Reproduction in the Etruscan Honeysuckle (Lonicera etrusca Santi)" Plants 10, no. 4: 815. https://doi.org/10.3390/plants10040815
APA StyleRojas-Nossa, S. V., Sánchez, J. M., & Navarro, L. (2021). Phloem-Feeding Herbivores Affect Floral Development and Reproduction in the Etruscan Honeysuckle (Lonicera etrusca Santi). Plants, 10(4), 815. https://doi.org/10.3390/plants10040815







