Lamina Cell Shape and Cell Wall Thickness Are Useful Indicators for Metal Tolerance—An Example in Bryophytes
Abstract
1. Introduction
2. Results
2.1. Lamina Cell Measurements
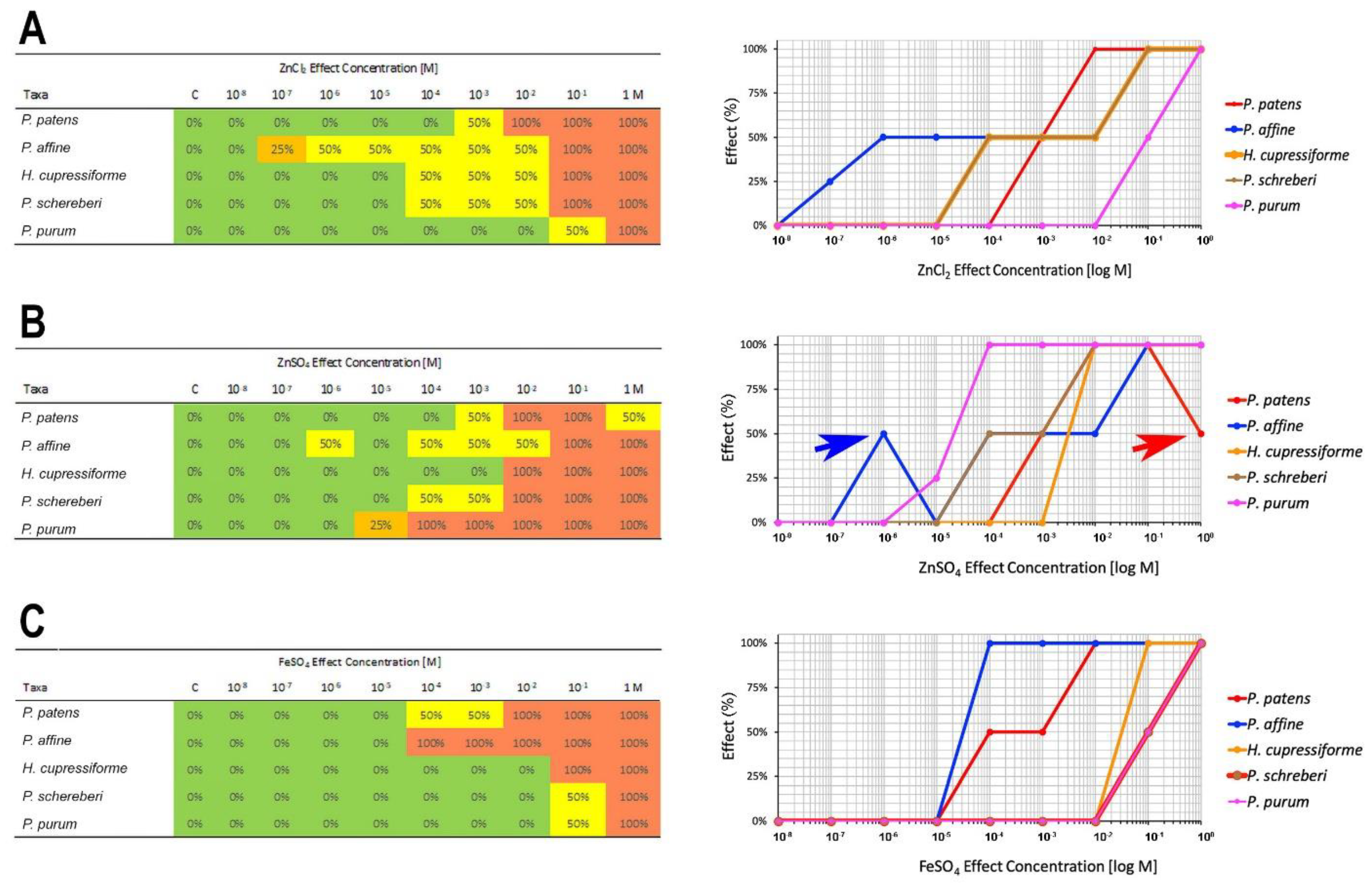
2.2. Metal Tolerance
2.3. Correlations between Cell Shape and Metal Tolerance
3. Discussion
4. Material and Methods
4.1. Plant Species
4.2. Tolerance Tests
4.3. Cell Measurements
4.4. Microscopy
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Harmens, H.; Norris, D.A.; Sharps, K.; Mills, G.; Alber, R.; Aleksiayenak, Y.; Blum, O.; Cucu-Man, S.M.; Dam, M.; De Temmerman, L.; et al. Heavy metal and nitrogen concentrations in mosses are declining across Europe whilst some “hotspots” remain in 2010. Environ. Pollut. 2015, 200, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Zechmeister, H.; Hohenwallner, D.; Riss, A.; Hanus-Illar, I. Estimation of element deposition derived from road traffic sources by using mosses. Environ. Pollut. 2005, 138, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Zechmeister, H.; Hohenwallner, D.; Riss, A.; Hanus-Illnar, A. Variations in heavy metal concentrations in the moss species Abietinella abietina (Hedw.) Fleisch according to sampling time, within site variability and increase in biomass. Sci. Total Environ. 2003, 301, 55–65. [Google Scholar] [CrossRef]
- Danninger, E. Inspektionsbericht: Luftgüteüberwachung. DVR: 0069264. 2017. Available online: https://www.land-oberoesterreich.gv.at/.
- Particulate Matter (PM) Basics. Available online: https://www.epa.gov/pm-pollution/particulate-matter-pm-basics (accessed on 23 January 2020).
- Xing, Y.-F.; Xu, Y.-H.; Shi, M.-H.; Lian, Y.-X. The impact of PM2.5 on the human respiratory system. J. Thorac. Dis. 2016, 8, E69–E74. [Google Scholar]
- Pongratz, T. Luftgütemessungen in der Steiermark, Jahresbericht. 2017. Bericht Nr. Lu-07-2018. Available online: https://www.umwelt.steiermark.at.
- Sassmann, S.; Adlassnig, W.; Puschenreiter, M.; Cadenas, E.J.P.; Leyvas, M.; Lichtscheidl, I.K.; Lang, I. Free metal ion availability is a major factor for tolerance and growth in Physcomitrella patens. Environ. Exp. Bot. 2015, 110, 1–10. [Google Scholar] [CrossRef]
- Sassmann, S.; Weidinger, M.; Adlassnig, W.; Hofhansl, F.; Bock, B.; Lang, I. Zinc and copper uptake in Physcomitrella patens: Limitations and effects on growth and morphology. Environ. Exp. Bot. 2015, 118, 12–20. [Google Scholar] [CrossRef]
- Vanderpoorten, A.; Goffinet, B. Introduction to Bryophytes; Cambridge University Press: Cambridge, UK, 2009. [Google Scholar]
- Shevock, J.R. The amazing design of a moss leaf. BryoString 2015, 3, 9–19. [Google Scholar]
- Flowers, S. Mosses: Utah and the West; University Press: Provo, UT, USA, 1973. [Google Scholar]
- Probst, W. Biologie der Moos- und Farnpflanzen, 2nd ed.; Quelle & Meyer: Heidelberg, Germany, 1987. [Google Scholar]
- Boquete, T.; Lang, I.; Weidinger, M.; Richards, C.L.; Alonso, C. Patterns and mechanisms of heavy metal accumulation and tolerance in two terrestrial moss species with contrasting habitat specialization. Environ. Exp. Bot. 2021, 182, 104336. [Google Scholar] [CrossRef]
- Stanković, J.D.; Sabovljević, A.D.; Sabovljević, M.S. Bryophytes and heavy metals: A review. Acta Bot. Croat. 2018, 77, 109–118. [Google Scholar] [CrossRef]
- Sarkar, P.; Bosneaga, E.; Auer, M. Plant cell walls throughout evolution: Towards a molecular understanding of their design principles. J. Exp. Bot. 2009, 60, 3615–3635. [Google Scholar] [CrossRef]
- Fangel, J.U.; Ulvskov, P.; Knox, J.P.; Mikkelsen, M.D.; Harholt, J.; Popper, Z.A.; Willats, W.G.T. Cell wall evolution and diversity. Front. Plant Sci. 2012, 3, 152. [Google Scholar] [CrossRef] [PubMed]
- Sassmann, S.; Wernitznig, S.; Lichtscheidl, I.K.; Lang, I. Comparing copper resistance in two bryophytes: Mielichhoferia elongata Hornsch. versus Physcomitrella patens Hedw. Protoplasma 2010, 246, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Wolterbeek, H.T.; Verburg, T.G. Judging Survey Quality: Local Variances. Environ. Monit. Assess 2002, 73, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Tyler, G. Bryophytes and heavy metals: A literature review. Bot. J. Linn. Soc. 1990, 104, 231–253. [Google Scholar] [CrossRef]
- Lang, I.; Wernitznig, S. Sequestration at the cell wall and plasma membrane facilitates zinc tolerance in the moss Pohlia drummondii. Environ. Exp. Bot. 2011, 74, 186–193. [Google Scholar] [CrossRef]
- Sabovljević, M.S.; Weidinger, M.; Sabovljević, A.D.; Adlassnig, W.; Lang, I. Is the Binding Pattern of Zinc(II) Equal in Different Bryophyte Species? Microsc. Microanal. 2018, 24, 69–74. [Google Scholar] [CrossRef]
- Sabovljevic, M.S.; Weidinger, M.; Sabovljevic, A.D.; Stankovic, J.; Adlassnig, W.; Lang, I. Metal accumulation in the acrocarp moss Atrichum undulatum undercontrolled conditions. Environ. Pollut. 2020, 256, 113397. [Google Scholar] [CrossRef]
- Zechmeister, H.; Kropic, M.; Moser, D.; Denner, M.; Hohenwallner, D.; Hanus-Illnar, A.; Scharf, S.; Riss, A.; Mirtl, M. Moos-Monitoring in Österreich. Aufsammlung 2015. Umweltbundesamt Report REP-0595:1-180; Umweltbundesamt GmbH: Vienna, Austria, 2016. [Google Scholar]
- Biebl, R. Über die gegensätzliche Wirkung der Spurenelemente Zink und Bor auf die Blattzellen von Mnium rostratum. Oesterreichische Bot. Z. 1947, 4, 61–73. [Google Scholar] [CrossRef]
- Shaw, A.J. Metal tolerance in bryophytes. In Heavy Metal Tolerance in Plants: Evolutionary Aspects; Shaw, A.J., Ed.; CRC Press Inc.: Boca Raton, FL, USA, 1990; pp. 133–152. [Google Scholar]
- Url, W. Zur Kenntnis der Todeszonen im konzentrationsgestuften Resistenzversuch. Physiol. Plant 1957, 10, 318–327. [Google Scholar] [CrossRef]
- Url, W.G. Über Schwermetalle-, zumal Kupferresistenz einiger Moose. Protoplasma 1956, 46, 768–793. [Google Scholar] [CrossRef]
- Büscher, P.; Koedam, N.; Van Speybroeck, D. Cation-exchange properties and adaption to soil acidity in bryophytes. New Phytol. 1990, 115, 177–186. [Google Scholar] [CrossRef]
- Soudzilovskaia, N.A.; Cornelissen, J.H.C.; During, H.J.; van Logtestijn, R.S.P.; Lang, S.I.; Aerts, R. Similar cation exchange capacities among bryophyte species refute a presumed mechanism of peatland acidification. Ecology 2010, 91, 2716–2726. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, J.A.; Vazquez, M.D.; Lopez, J.; Carballeira, A. Modelling the extra and intracellular uptake and discharge of heavy metals in Fontinalis antipyretica transplanted along a heavy metal and pH contamination gradient. Environ. Pollut. 2006, 139, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, M.D.; Lopez, J.; Carballeira, A. Modification of the sequential elution technique for the extraction of heavy metals from bryophytes. Sci. Total Environ. 1999, 241, 53–62. [Google Scholar] [CrossRef]
- Vidali, L.; Bezanilla, M. Physcomitrella patens: A model for tip cell growth and differentiation. Curr. Opin. Plant Biol. 2012, 15, 625–631. [Google Scholar] [CrossRef]
- Adlassnig, W.; Weiss, Y.S.; Sassmann, S.; Steinhauser, G.; Hofhansl, F.; Baumann, N.; Lichtscheidl, I.K.; Lang, I. The copper spoil heap Knappenberg, Austria, as a model for metal habitats—Vegetation, substrate and contamination. Sci. Total Environ. 2016, 563–564, 1037–1049. [Google Scholar] [CrossRef]
- Sirka, P.; Kubesova, S.; Miskova, K. Bryophytes of spoil heaps rich in toxic metals in Central Slovakia. Thaiszia-J. Bot. 2018, 28, 059–077. [Google Scholar]
- Hnilica, E.; Kohout, R. Mathe-Lexicon.at. 2018. Available online: https.//:www.mathe-lexikon.at (accessed on 10 June 2019).
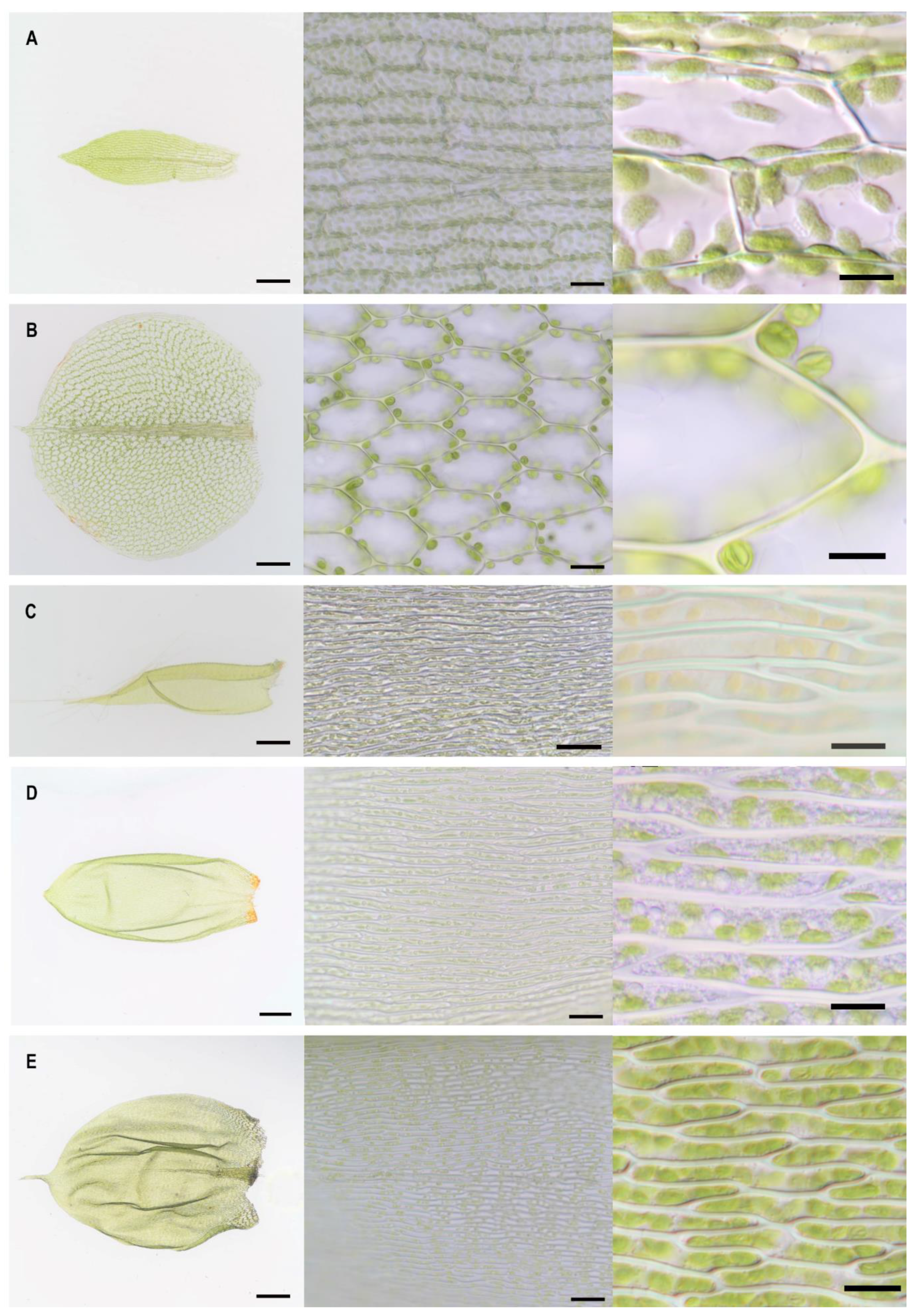

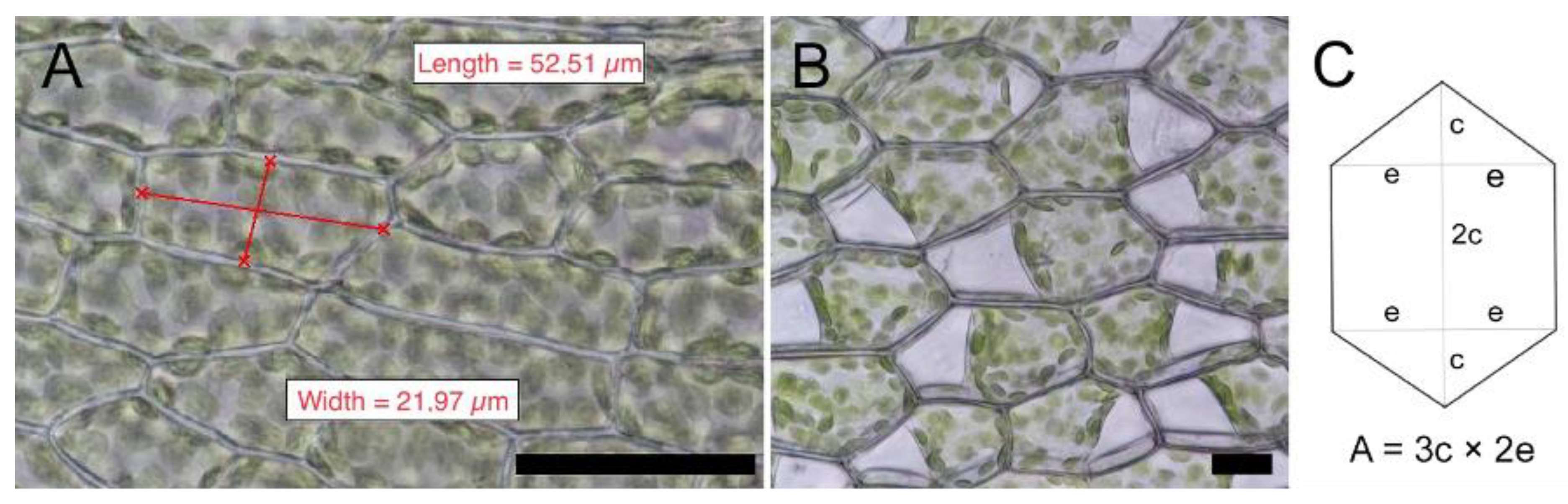
| Moss Species | µ Cell Length (SD) (µM) | µ Cell Width (SD) (µM) | µ Cell Length to Width Ratio | Shape of Mid Lamina Cells | µ Cell Area (SD) (µM2) | µ Cell Wall Thickness (SD) (µM) |
|---|---|---|---|---|---|---|
| Plagiomnium affine | 51 (4) | 37 (3) | 1 | hexagonal | 1402 (150) | 0.65 (0.12) |
| Physcomitrium patens | 63 (12) | 31 (6) | 2 | rectangular | 1979 (569) | 0.26 (0.05) |
| Pseudoscleropodium purum | 65 (8) | 5 (1) | 12 | rectangular, longish | 354 (73) | 0.46 (0.07) |
| Hypnum cupressiforme | 72 (11) | 3 (1) | 25 | rectangular, longish | 220 (53) | 0.88 (0.17) |
| Pleurozium schreberi | 94 (12) | 8 (1) | 12 | rectangular, longish | 735 (140) | 0.86 (0.18) |
| NOEC Median | LOEC Median | |||||
|---|---|---|---|---|---|---|
| Species | ZnCl2 | ZnSO4 | FeSO4 | ZnCl2 | ZnSO4 | FeSO4 |
| P. patens | 10−4 | 10−4 | 10−5 | 10−3 | 10−3 | 10−4 |
| P. affine | 5.5 × 10−8 | 5.55 × 10−6 | 10−5 | 5.5 × 10−7 | 10−6 | 10−4 |
| H. cupressiforme | 10−5 | 10−3 | 10−2 | 10−4 | 10−2 | 10−1 |
| P. schreberi | 10−5 | 10−5 | 10−2 | 10−4 | 10−4 | 10−1 |
| P. purum | 5.5 × 10−3 | 5.5 × 10−6 | 10−2 | 10−1 | 5.5 × 10−5 | 10−1 |
| ZnCl2 | ZnSO4 | FeSO4 | |
|---|---|---|---|
| Cell Length | 0.16 (0.023) | 0.27 (<0.001) | 0.60 (<0.001) |
| Cell Width | −0.34 (<0.001) | −0.24 (<0.001) | −0.85 (<0.001) |
| Ratio Length/Width | 0.24 (<0.001) | 0.36 (<0.001) | 0.85 (<0.001) |
| Cell Wall Thickness | −0.57 (<0.001) | 0.24 (0.001) | 0.50 (<0.001) |
| Ratio Length/Cell Wall Thickness | 0.68 (<0.001) | −0.06 (0.406) | −0.17 (0.017) |
| Ratio width/Cell Wall Thickness | 0.09 (0.202) | −0.25 (<0.001) | −0.85 (<0.001) |
| Species | Collection | Culture Conditions |
|---|---|---|
| Plagiomnium affine (Blandow ex Funck) T.J. Kop. (Mniaceae) | laboratory | Non-sterile culture 20 °C, 14/10 h day/night |
| Physcomitrium patens (Hedw.) Bruch and Schimp. (Funariaceae) | laboratory | sterile culture 20 °C, 14/10 h day/night |
| Pseudoscleropodium purum (Hedw.) M. Fleisch (Brachytheciaceae) | 48.183470, 16.067139 | Natural habitat |
| Hypnum cupressiforme Hedw. (Hypnaceae) | 48.183470, 16.044465 | Natural habitat |
| Pleurozium schreberi (Will. ex Brid.) Mitt. (Hylocomiaceae) | 48.183470, 16.066399 | Natural habitat |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petschinger, K.; Adlassnig, W.; Sabovljevic, M.S.; Lang, I. Lamina Cell Shape and Cell Wall Thickness Are Useful Indicators for Metal Tolerance—An Example in Bryophytes. Plants 2021, 10, 274. https://doi.org/10.3390/plants10020274
Petschinger K, Adlassnig W, Sabovljevic MS, Lang I. Lamina Cell Shape and Cell Wall Thickness Are Useful Indicators for Metal Tolerance—An Example in Bryophytes. Plants. 2021; 10(2):274. https://doi.org/10.3390/plants10020274
Chicago/Turabian StylePetschinger, Katharina, Wolfram Adlassnig, Marko S. Sabovljevic, and Ingeborg Lang. 2021. "Lamina Cell Shape and Cell Wall Thickness Are Useful Indicators for Metal Tolerance—An Example in Bryophytes" Plants 10, no. 2: 274. https://doi.org/10.3390/plants10020274
APA StylePetschinger, K., Adlassnig, W., Sabovljevic, M. S., & Lang, I. (2021). Lamina Cell Shape and Cell Wall Thickness Are Useful Indicators for Metal Tolerance—An Example in Bryophytes. Plants, 10(2), 274. https://doi.org/10.3390/plants10020274








