Herbicide Options to Control Naturalised Infestations of Cereus uruguayanus in Rangeland Environments of Australia
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Details
2.2. Experiment 1. Screening for Suitable Herbicides and Application Techniques
2.3. Experiment 2. Evaluating Efficacy of Soil Applied Residual Herbicides
2.4. Experiment 3. Optimising Glyphosate Dose for Cut Stump Application
2.5. Statistical Analysis
3. Results
3.1. Weather
3.2. Experiment 1. Screening for Suitable Herbicides and Application Techniques
3.3. Experiment 2. Evaluating Efficacy of Soil Applied Residual Herbicides
3.4. Experiment 3. Optimising Glyphosate Dose for Cut Stump Application
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anderson, E.F. The Cactus Family; Timber Press Inc.: Portland, OR, USA, 2001; p. 776. [Google Scholar]
- Nobel, P.S. Cacti: Biology and Uses; University of California Press: Berkeley, CA, USA, 2002; p. 291. [Google Scholar]
- Sheehan, M.R.; Potter, S. Managing Opuntioid Cacti in Australia: Best Practice Control Manual for Austrocylindropuntia, Cylindropuntia and Opuntia Species; Department of Primary Industries and Regional Development: Perth, Australia, 2017; p. 158. [Google Scholar]
- Nobel, P.S. Desert Wisdom, Agaves and Cacti; CO2, Water, Climate Change; Universe Inc.: Bloomington, NY, USA, 2009; p. 182. [Google Scholar]
- Hosking, J.R.; McFadyen, R.E.; Murray, N.D. Distribution and biological control of cactus species in eastern Australia. Plant Prot. Q. 1988, 3, 115–123. [Google Scholar]
- Chuk, M. Invasive cacti—A threat to the rangelands of Australia. In Proceedings of the 16th Biennial Conference of the Australian Rangeland Society, Bourke, Australia, 26–30 September 2010; Eldridge, D.J., Waters, C., Eds.; Australian Rangeland Society: Perth, Australia, 2010. [Google Scholar]
- Belnap, J.; Ludwig, J.A.; Wilcox, B.P.; Betancourt, J.L.; Dean, W.R.J.; Hoffmann, B.D.; Milton, S.J. Introduced and invasive species in novel rangeland ecosystems: Friends or foes? Rangel. Ecol. Manag. 2012, 65, 569–578. [Google Scholar] [CrossRef]
- Wilcox, D.; Fitzgerald, D. Unintended Consequences of an Early Mining Boom. Range Manag. Newsl. 2009, 9, 3–4. [Google Scholar]
- Forster, P.I.; Schmeider, M. Cereus uruguayanus (Cactaceae) and its naturalised occurrence in Queensland, Australia. Austrobaileya 2000, 5, 671–677. [Google Scholar]
- Taylor, N.P. Nomenclatural adjustments in various genera of Cactaceae (subfam. Cactoideae). Cactaceae Consens. Initiat. 1998, 6, 15–16. [Google Scholar]
- Queensland Department of Agriculture and Fisheries. Willows Cactus. 2020. Available online: https://www.daf.qld.gov.au/__data/assets/pdf_file/0004/1474069/willows-cactus.pdf (accessed on 30 May 2020).
- Hosking, J.R.; Conn, B.J.; Lepschi, B.J.; Barker, C.H. Plant species first recognised as naturalised for New South Wales in 2002 and 2003, with additional comments on species recognised as naturalised in 2000–2001. Cunninghamia 2007, 10, 139–166. [Google Scholar]
- Sutton, G.F.; Klein, H.; Paterson, I.D. Evaluating the efficacy of Hypogeococcus sp. as a biological control agent of the cactaceous weed Cereus jamacaru in South Africa. BioControl 2018, 63, 493–503. [Google Scholar] [CrossRef]
- Moran, V.C.; Zimmermann, H.G. Biological control of cactus weeds of minor importance in South Africa. Agric. Ecosyst. Environ. 1991, 37, 37–55. [Google Scholar] [CrossRef]
- Jordaan, J.; Mantji, T. Biological control of Cereus jamacaru (queen of the night cactus) in the Thornveld of the Limpopo Province, South Africa. Grassroots 2012, 12, 36–40. [Google Scholar]
- Bureau of Meteorology. Monthly Rainfall for the Willow Gemfields, Queensland, Australia. Available online: http://www.bom.gov.au/jsp/ncc/cdio/weatherData/av?p_nccObsCode=139&p_display_type=dataFile&p_startYear=&p_c=&p_stn_num=035215 (accessed on 4 September 2021).
- Bureau of Meteorology. Monthly Mean Minimum Temperature Data for Emerald Airport, Queensland, Australia. Available online: http://www.bom.gov.au/jsp/ncc/cdio/weatherData/av?p_nccObsCode=38&p_display_type=dataFile&p_startYear=&p_c=&p_stn_num=035264 (accessed on 4 September 2021).
- Bureau of Meteorology. Monthly Mean Maximum Temperature Data for Emerald Airport, Queensland, Australia. Available online: http://www.bom.gov.au/jsp/ncc/cdio/weatherData/av?p_nccObsCode=36&p_display_type=dataFile&p_startYear=&p_c=&p_stn_num=035264 (accessed on 4 September 2021).
- CropLife Australia. Herbicide Mode of Action Table. Available online: https://www.croplife.org.au/resources/programs/resistance-management/herbicide-moa-table-4-draft-2/ (accessed on 4 September 2021).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 14 May 2021).
- De Mendiburu, F. Agricolae: Statistical Procedures for Agricultural Research. R Package Version 1.3-3. Available online: https://CRAN.R-project.org/package=agricolae (accessed on 14 May 2021).
- Fogliatto, S.; Milan, M.; Vidotto, F. Control of Ailanthus altissima using cut stump and basal bark herbicide applications in an eighteenth-century fortress. Weed Res. 2020, 60, 425–434. [Google Scholar] [CrossRef]
- Oneto, S.R.; Kyser, G.B.; DiTomaso, J.M. Efficacy of mechanical and herbicide control methods for Scotch Broom (Cytisus scoparius) and cost analysis of chemical control options. Invasive Plant Sci. Manag. 2010, 3, 421–428. [Google Scholar] [CrossRef]
- Enloe, S.F.; O’Sullivan, S.E.; Loewenstein, N.J.; Brantley, E.F.; Dwight, K.L. Triclopyr application timing and concentration influence low-volume basal bark efficacy on Chinese privet (Ligustrum sinense). Invasive Plant Sci. Manag. 2016, 9, 235–241. [Google Scholar] [CrossRef]
- Campbell, S.; Vogler, W.; Brazier, D.; Vitelli, J.; Brooks, S. Weed Leucaena and its significance, implications and control. Trop. Grassl. Forrajes 2019, 7, 280–289. [Google Scholar] [CrossRef]
- Vitelli, J.; Madigan, B.; Wilkinson, P.; Van Haaren, P. Calotrope (Calotropis procera) control. Rangel. J. 2008, 30, 339–348. [Google Scholar] [CrossRef]
- Enloe, S.F.; O’Sullivan, S.E.; Loewenstein, N.J.; Brantley, E.; Lauer, D.K. The influence of treatment timing and shrub size on Chinese privet (Ligustrum sinense) control with cut stump herbicide treatments in the southeastern United States. Invasive Plant. Sci. Manag. 2018, 11, 49–55. [Google Scholar] [CrossRef]
- Burch, P.L.; Zedaker, S.M. Removing the invasive tree Ailanthus altissima and restoring natural cover. J. Arboric. 2003, 29, 18–24. [Google Scholar]
- Carmona, R.; Neto, S.B.C.A.; Pereira, R.C. Control of Acacia farnesiana and of Mimosa pteridofita in pastures. Pesqui. Agropecu. Bras. 2001, 36, 1301–1307. [Google Scholar] [CrossRef]
- Novoa, A.; Brundu, G.; Day, M.D.; Deltoro, V.; Essl, F.; Foxcroft, L.C.; Fried, G.; Kaplin, H.; Kumschick, S.; Lloyd, S.; et al. Global actions for managing cactus invasions. Plants 2019, 8, 421. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, H.G. Global invasions of cacti (Opuntia sp.): Control, management and conflicts of interest. In Crop Ecology, Cultivation and Uses of Cactus Pear; Inglese, P., Mondragon, C., Nefzaoui, A., Saenz, C., Eds.; Food and Agriculture Organisation of the United Nations and International Center for Agricultural Research in the Dry Areas: Rome, Italy, 2017; pp. 171–184. ISBN 978-92-5-109860-8. [Google Scholar]
- Lewis, K.A.; Tzilivakis, J.; Warner, D.; Green, A. An international database for pesticide risk assessments and management. Hum. Ecol. Risk Assess. An. Intern. J. 2016, 22, 1050–1064. [Google Scholar] [CrossRef]
- Lewis, K.; Tzilivakis, J. Development of a data set of pesticide dissipation rates in/on various plant matrices for the Pesticide Properties DataBase (PPDB). Data 2017, 2, 28. [Google Scholar] [CrossRef]
- Van Bruggen, A.H.C.; He, M.M.; Shin, K.; Mai, V.; Jeong, K.C.; Finckh, M.R.; Morris, J.G. Environmental and health effects of the herbicide glyphosate. Sci. Total Environ. 2018, 616/617, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Vitelli, J.S.; Pitt, J.L. Assessment of current weed control methods relevant to the management of the biodiversity of Australian rangelands. Rangel. J. 2006, 28, 37–46. [Google Scholar] [CrossRef]

| Technique | Herbicide Active Ingredient(s) | Mode of Action (Group) | Herbicide Trade Name | Rate (g a.i. L−1) | Carrier | Treatment Date |
|---|---|---|---|---|---|---|
| Basal bark (traditional) | Triclopyr (240 g L−1)/picloram (120 g L−1) | 4 | Access™ | 4/2 | Diesel | May 2016 |
| Basal bark (thinline) | Triclopyr (240 g L−1)/picloram (120 g L−1) | 4 | Access™ | 24/12 | Diesel | May 2016 |
| Cut stump | Aminopyralid (375 g kg−1)/metsulfuron-methyl (300 g kg−1) | 4/2 | Stinger™ | 1.5/1.2 | Water 1 | May 2016 |
| Cut stump | Aminopyralid (4.47 g L−1)/picloram (44.7 g L−1) | 4 | Vigilant™ II | 4.47/44.7 | - 2 | Oct 2016 |
| Cut stump | Glyphosate (360 g L−1) | 9 | Roundup® | 360 | - 2 | May 2016 |
| Cut stump | Metsulfuron-methyl (600 g kg−1) | 2 | Brush-Off® | 1.2 | Water 1 | May 2016 |
| Cut stump | Triclopyr (240 g L−1)/picloram (120 g L−1) | 4 | Access™ | 4/2 | Diesel | May 2016 |
| Cut stump | Triclopyr (200 g L−1)/picloram (100 g L−1)/aminopyralid (25 g L−1) | 4 | Tordon™ RegrowthMaster | 10/5/1.25 | Water 1 | May 2016 |
| Foliar | Aminopyralid (375 g kg−1)/metsulfuron-methyl (300 g kg−1) | 4/2 | Stinger™ | 0.15/0.12 | Water 1 | Oct 2016 |
| Foliar | Amitrole (250 g L−1)/ammonium thiocyanate (220 g L−1) | 34 | Amitrole T | 10/8.8 | Water 1 | Oct 2016 |
| Foliar | Metsulfuron-methyl (600 g kg−1) | 2 | Brush-Off® | 0.12 | Water 1 | Oct 2016 |
| Foliar | MSMA (720 g L−1) | 0 | Daconate 720® | 18 | Water 1 | Oct 2016 |
| Foliar | Picloram (240 g L−1) + fluroxypyr (333 g L−1) | 4 | Stuka Flexi + Starane™ Advanced | 0.6/0.5 | Water 1 | Oct 2016 |
| Foliar | Triclopyr (600 g L−1) | 4 | Garlon™ 600 | 18 | Water 1 | Oct 2016 |
| Foliar | Triclopyr (300 g L−1)/picloram (100 g L−1)/aminopyralid (8 g L−1) | 4 | Grazon™ Extra | 1.5/0.5/0.04 | Water 1 | Oct 2016 |
| Foliar | Triclopyr (200 g L−1)/picloram (100 g L−1)/aminopyralid (25 g L−1) | 4 | Tordon™ RegrowthMaster | 5/2.5/0.625 | Water 1 | Oct 2016 |
| Stem injection | Amitrole (250 g L−1)/ammonium thiocyanate (220 g L−1) | 34 | Amitrole T | 250/220 | - 2 | Oct 2016 |
| Stem injection | Glyphosate (360 g L−1) | 9 | Roundup® | 360 | - 2 | Oct 2016 |
| Stem injection | MSMA (720 g L−1) | 0 | Daconate 720® | 240 | Water | Oct 2016 |
| Stem injection | Triclopyr (200 g L−1)/picloram (100 g L−1)/aminopyralid (25 g L−1) | 4 | Tordon™ RegrowthMaster | 40/20/5 | Water | Oct 2016 |
| Control | none | - | - | - | - |
| Year | Jan. | Feb. | Mar. | Apr. | May | June | July | Aug. | Sept. | Oct. | Nov. | Dec. | Tot. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2016 | 161.0 | 183.8 | 26.4 | 3.8 | 0.0 | 116.2 | 132.0 | 22.2 | 93.0 | 11.4 | 43.3 | 38.0 | 831.1 |
| 2017 | 233.4 | 3.0 | 117.6 | 0.0 | 3.0 | 2.8 | 22.6 | 5.8 | 0.0 | 94.4 | 109.6 | 75.6 | 667.8 |
| 2018 | 33.0 | 107.6 | 31.0 | 4.8 | 4.0 | 5.4 | 1.0 | 0.0 | 0.0 | 126.6 | 20.2 | 26.6 | 360.2 |
| 2019 | 2.2 | 0.0 | 163.4 | 212.2 | 0.0 | 15.2 | 17.4 | 12.0 | 0.0 | 28.8 | 23.0 | 8.6 | 482.8 |
| Mean | 104.0 | 91.7 | 56.0 | 33.7 | 36.4 | 24.5 | 21.5 | 24.2 | 26 | 40.2 | 57.9 | 84.1 | 589.0 |
| Herbicide Treatment | Plant Mortality (%) 1 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 6 MAT | 12 MAT | 17 MAT | 25 MAT | 31 MAT 2 | 42 MAT 2 | |||||
| S | L | S | L | S | L | S | L | |||
| Control 3 | 0b | 0b | 0d | 0d | 0d | 0d | 0d | 0d | 2b | 5b |
| Basal bark | ||||||||||
| Triclopyr/picloram (Traditional) | 4b | 0b | 58b | 6c | 71b | 19c | 100a | 71c | 96a | 98a |
| Triclopyr/picloram (Thinline) | 5b | 0b | 56b | 7c | 80b | 17c | 100a | 85b | 95a | 100a |
| Cut stump 4 | ||||||||||
| Aminopyralid/metsulfuron-methyl | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a |
| Glyphosate | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a |
| Metsulfuron-methyl | 100a | 96a | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a |
| Triclopyr/picloram | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a |
| Triclopyr/picloram/aminopyralid | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a | 100a |
| Herbicide Treatment | Plant Mortality (%) 1 | |||||||
|---|---|---|---|---|---|---|---|---|
| 1 MAT | 7 MAT | 13 MAT 2 | 20 MAT 2 | 26 MAT 2 | 38 MAT 2 | |||
| S | L | S | L | |||||
| Control 3 | 0f | 0f | 0i | 0i | 0f | 0e | 0.5d | 2e |
| Foliar | ||||||||
| Aminopyralid/metsulfuron-methyl | 0f | 0f | 11g–i | 18f–h | 42d | 74b | 82b | 93bc |
| Amitrole/ammonium thiocyanate | 0f | 0f | 25d–h | 0i | 17e | 32d | 38c | 40d |
| Metsulfuron-methyl | 0f | 0f | 0i | 0i | 0f | 0e | 0.5d | 5e |
| MSMA | 86b | 21d | 100a | 84bc | 99ab | 99a | 99a | 99ab |
| Picloram + fluroxypyr (tank mix) | 0f | 0f | 29d–g | 0i | 31de | 51cd | 73b | 83c |
| Triclopyr | 2ef | 0f | 90ab | 58cd | 99ab | 100a | 100a | 100a |
| Triclopyr/picloram/aminopyralid 4 | 0f | 0f | 53c–e | 10g–i | 76c | 100a | 100a | 100a |
| Tricloypr/picloram/aminopyralid 5 | 0f | 0f | 47d–f | 22e–h | 88c | 100a | 100a | 100a |
| Stem injection | ||||||||
| Amitrole/ammonium thiocyanate | 6e | 0f | 44d–f | 0i | 38de | 66bc | 83b | 96a–c |
| Glyphosate | 35cd | 0f | 95ab | 28d–g | 91bc | 99a | 100a | 100a |
| MSMA | 100a | 42c | 100a | 96ab | 100a | 100a | 100a | 100a |
| Tricloypr/picloram/aminopyralid 5 | 0f | 0f | 45d–f | 3h–i | 22de | 79b | 99a | 99ab |
| Cut stump 6 | ||||||||
| Aminopyralid/picloram | 0f | 0f | 100a | 100a | 100a | 100a | 100a | 100a |
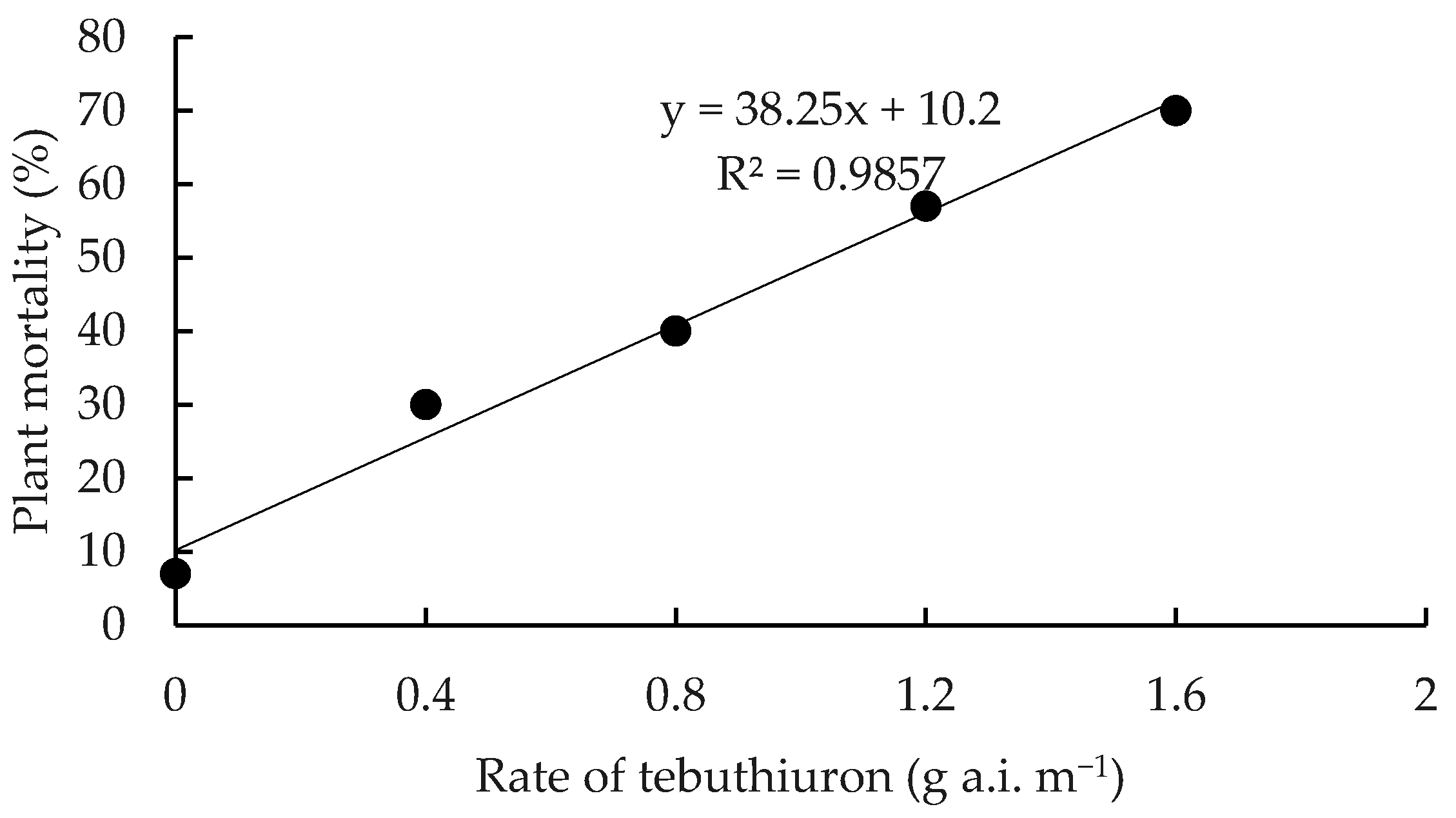
| Treatment | Plant Mortality (%) 1 | ||||
|---|---|---|---|---|---|
| 6 MAT | 11 MAT | 19 MAT | 25 MAT | 36 MAT | |
| Control 2 | 0c | 0e | 0e | 3e | 7d |
| Tebuthiuron | |||||
| 0.4 g a.i. m−1 | 20a–c | 27b–d | 30b–d | 30b–e | 30cd |
| 0.8 g a.i. m−1 | 20ab | 27bc | 40b–d | 40b–d | 40bc |
| 1.2 g a.i. m−1 | 17a–c | 23b–d | 47bc | 57bc | 57bc |
| 1.6 g a.i. m−1 | 24ab | 47b | 60b | 63b | 70ab |
| Hexazinone | |||||
| 0.5 g a.i. m−1 | 0c | 3de | 10de | 20de | 27cd |
| 1 g a.i. m−1 | 7bc | 10c–e | 20cd | 20c–e | 20cd |
| Stem injected 3 | 45a | 90a | 93a | 93a | 93a |
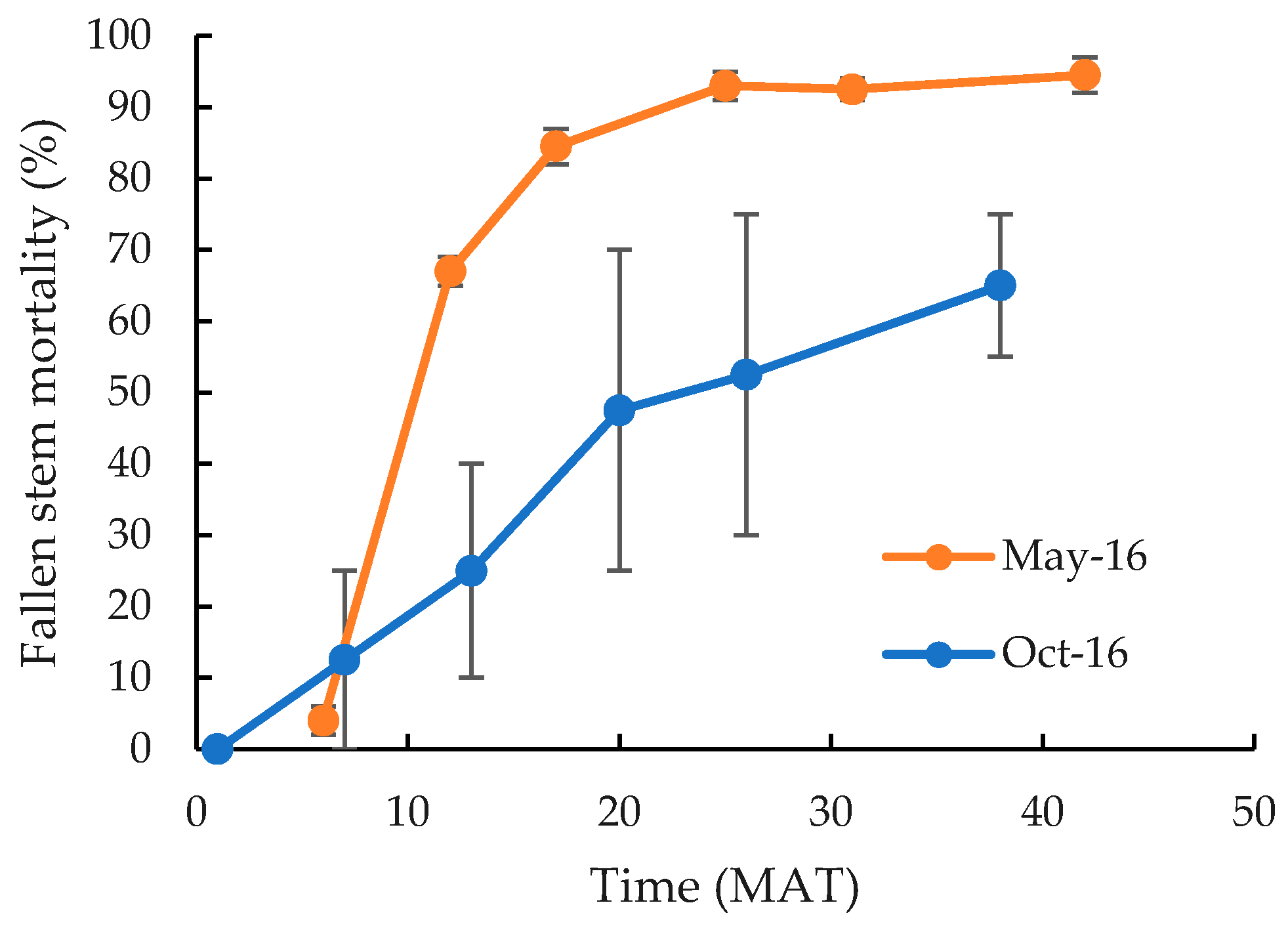
| Herbicide Treatment | Cut Stump Mortality (%) 1 | Fallen Stem Mortality (%) 1 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 7 MAT | 13 MAT | 25 MAT | 7 MAT | 13 MAT | 25 MAT | |||||||
| S | L | S | L | S | L | S | L | S | L | S | L | |
| Control | 4g | 14fg | 17c | 27c | 31d | 58c | 44c–e | 23e | 50e–g | 27g | 71b | 72b |
| Triclopyr/picloram | 100a | 95ab | 100a | 100a | 100a | 100a | 76a–c | 20e | 82b–d | 49e–g | 100a | 93ab |
| Glyphosate | ||||||||||||
| 45 g a.i. L−1 | 84b–d | 35ef | 87a | 60b | 97ab | 90b | 79ab | 37de | 88a–c | 44fg | 100a | 92ab |
| 90 g a.i. L−1 | 77cd | 60de | 95a | 87a | 97ab | 95ab | 82ab | 32e | 94a | 50e–g | 100a | 81ab |
| 180 g a.i. L−1 | 88bc | 69cd | 100a | 93a | 100a | 97ab | 63b–d | 59b–d | 85a–d | 71c–e | 100a | 86ab |
| 360 g a.i. L−1 | 97ab | 85b–d | 100a | 96a | 100a | 100a | 91a | 35de | 94ab | 62d–f | 100a | 88ab |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campbell, S.; Bajwa, A.; Hosking, K.; Brazier, D.; Mellor, V.; Perkins, M. Herbicide Options to Control Naturalised Infestations of Cereus uruguayanus in Rangeland Environments of Australia. Plants 2021, 10, 2227. https://doi.org/10.3390/plants10102227
Campbell S, Bajwa A, Hosking K, Brazier D, Mellor V, Perkins M. Herbicide Options to Control Naturalised Infestations of Cereus uruguayanus in Rangeland Environments of Australia. Plants. 2021; 10(10):2227. https://doi.org/10.3390/plants10102227
Chicago/Turabian StyleCampbell, Shane, Ali Bajwa, Kelsey Hosking, Dannielle Brazier, Vincent Mellor, and Melinda Perkins. 2021. "Herbicide Options to Control Naturalised Infestations of Cereus uruguayanus in Rangeland Environments of Australia" Plants 10, no. 10: 2227. https://doi.org/10.3390/plants10102227
APA StyleCampbell, S., Bajwa, A., Hosking, K., Brazier, D., Mellor, V., & Perkins, M. (2021). Herbicide Options to Control Naturalised Infestations of Cereus uruguayanus in Rangeland Environments of Australia. Plants, 10(10), 2227. https://doi.org/10.3390/plants10102227








