Time-Series of Vegetation Indices (VNIR/SWIR) Derived from Sentinel-2 (A/B) to Assess Turgor Pressure in Kiwifruit
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
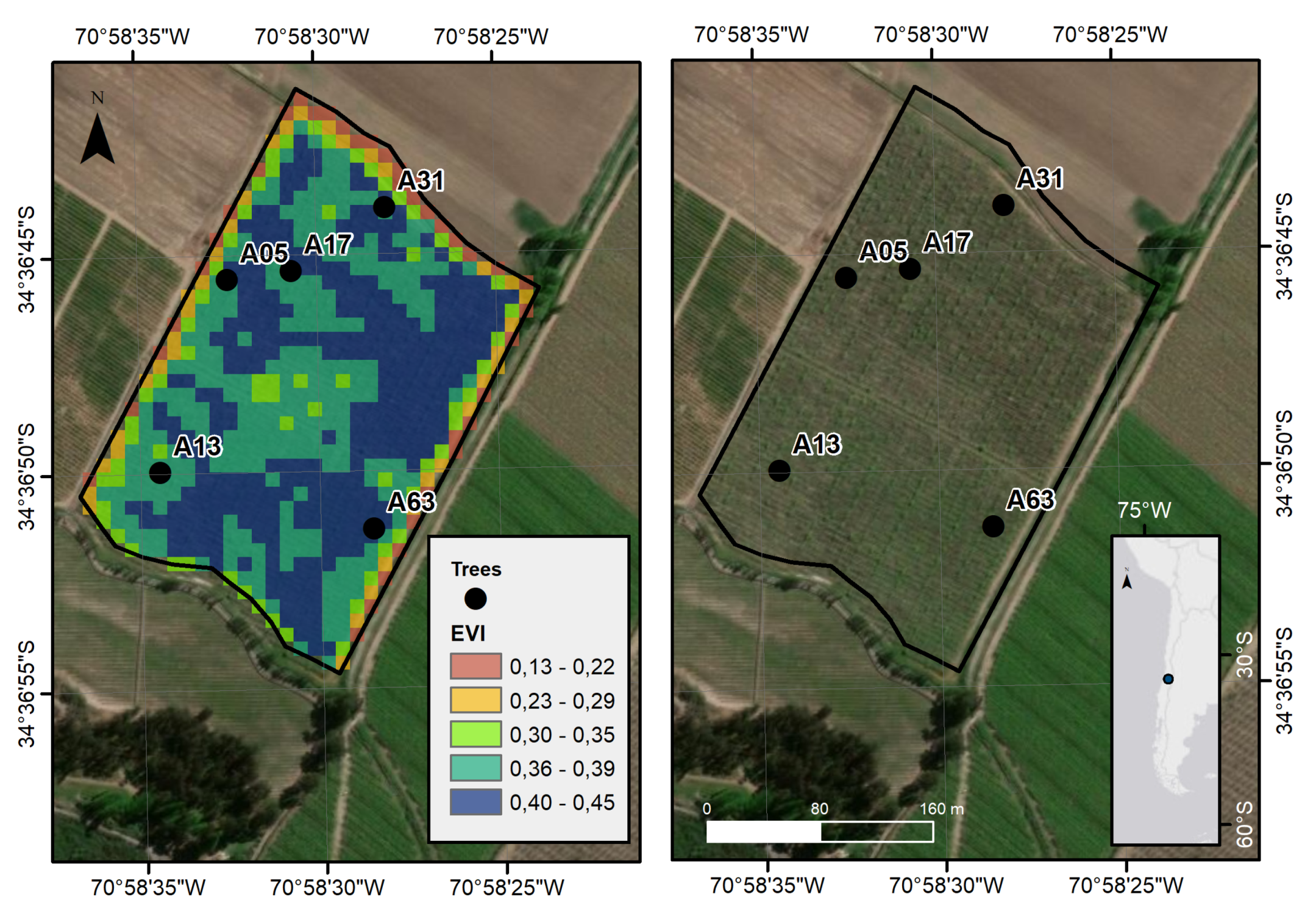
2.1. Study Area
2.2. Data
Sentinel-2
2.3. Patch Pressure (Pp) Yara Water-Sensor
2.4. Spectrals Vegetation Indices
2.5. Exploratory Analysis
2.6. Correlation Analysis
3. Results
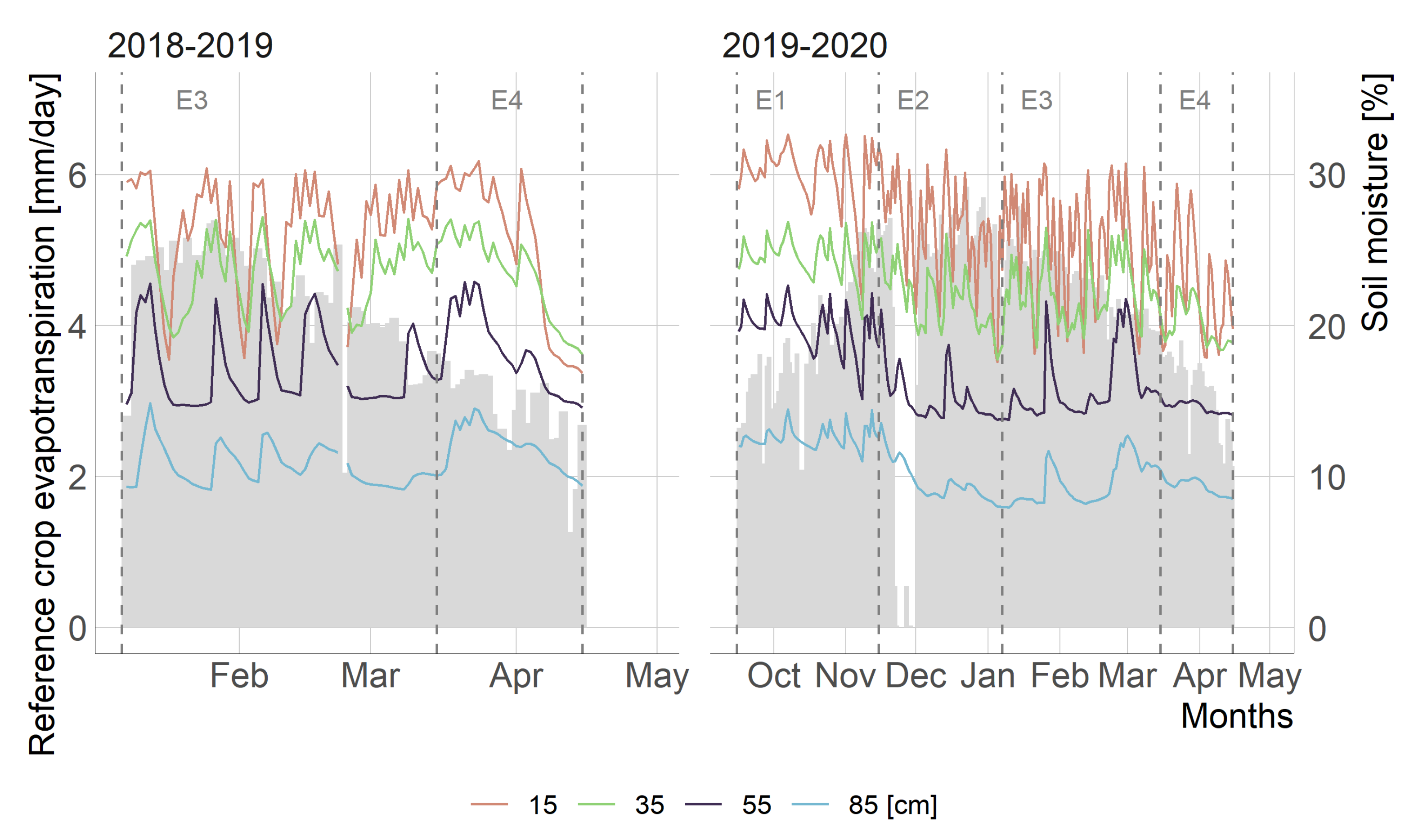
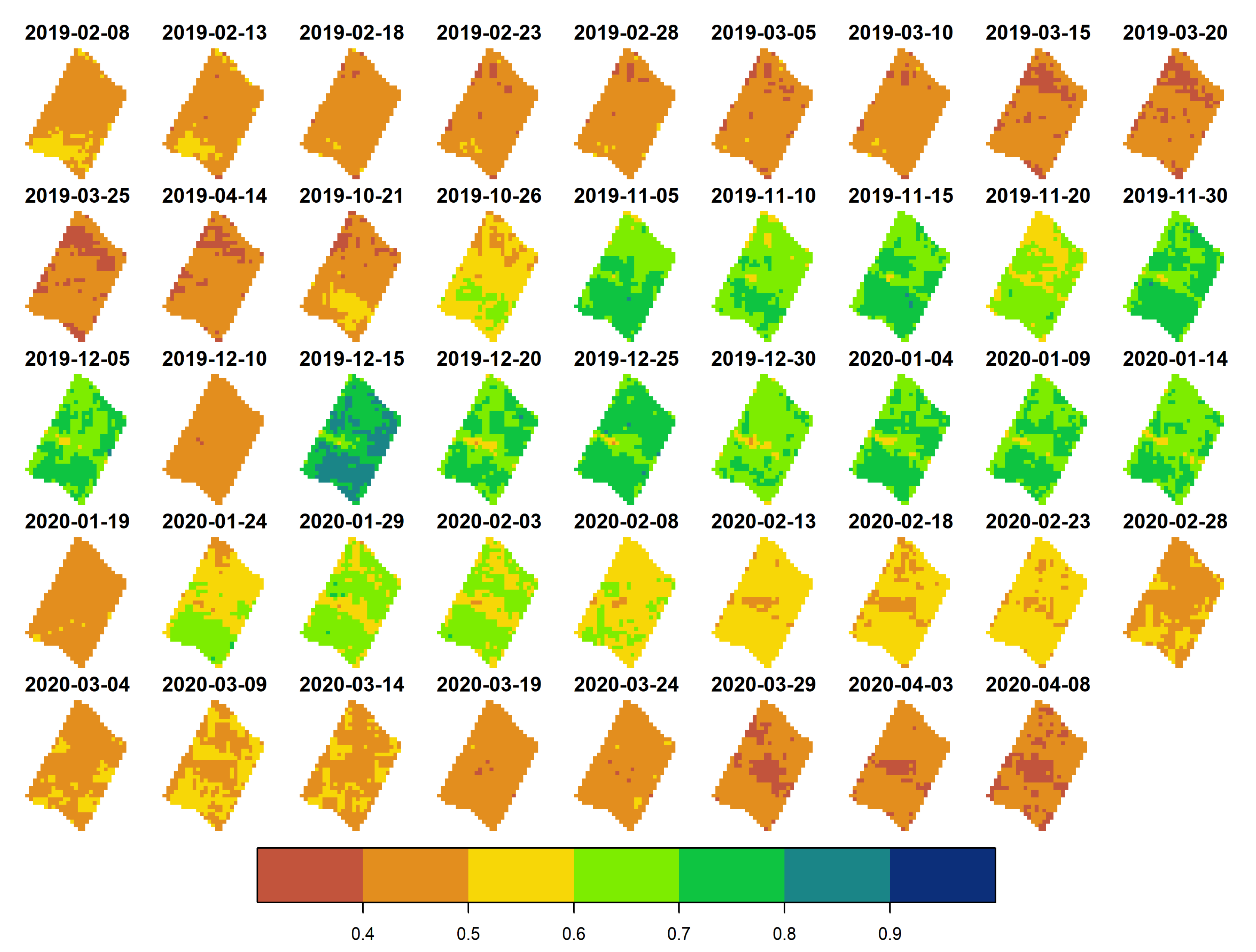
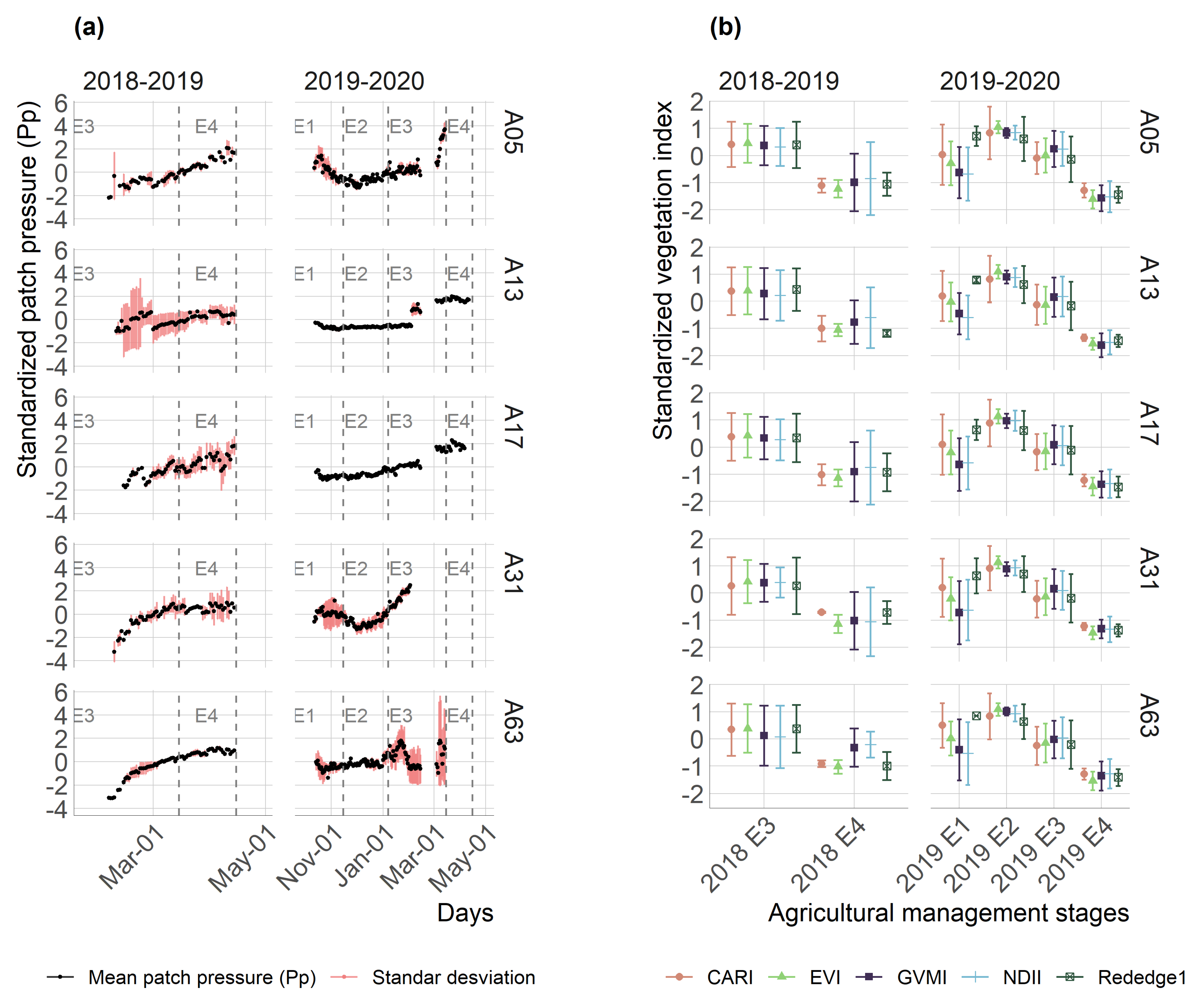
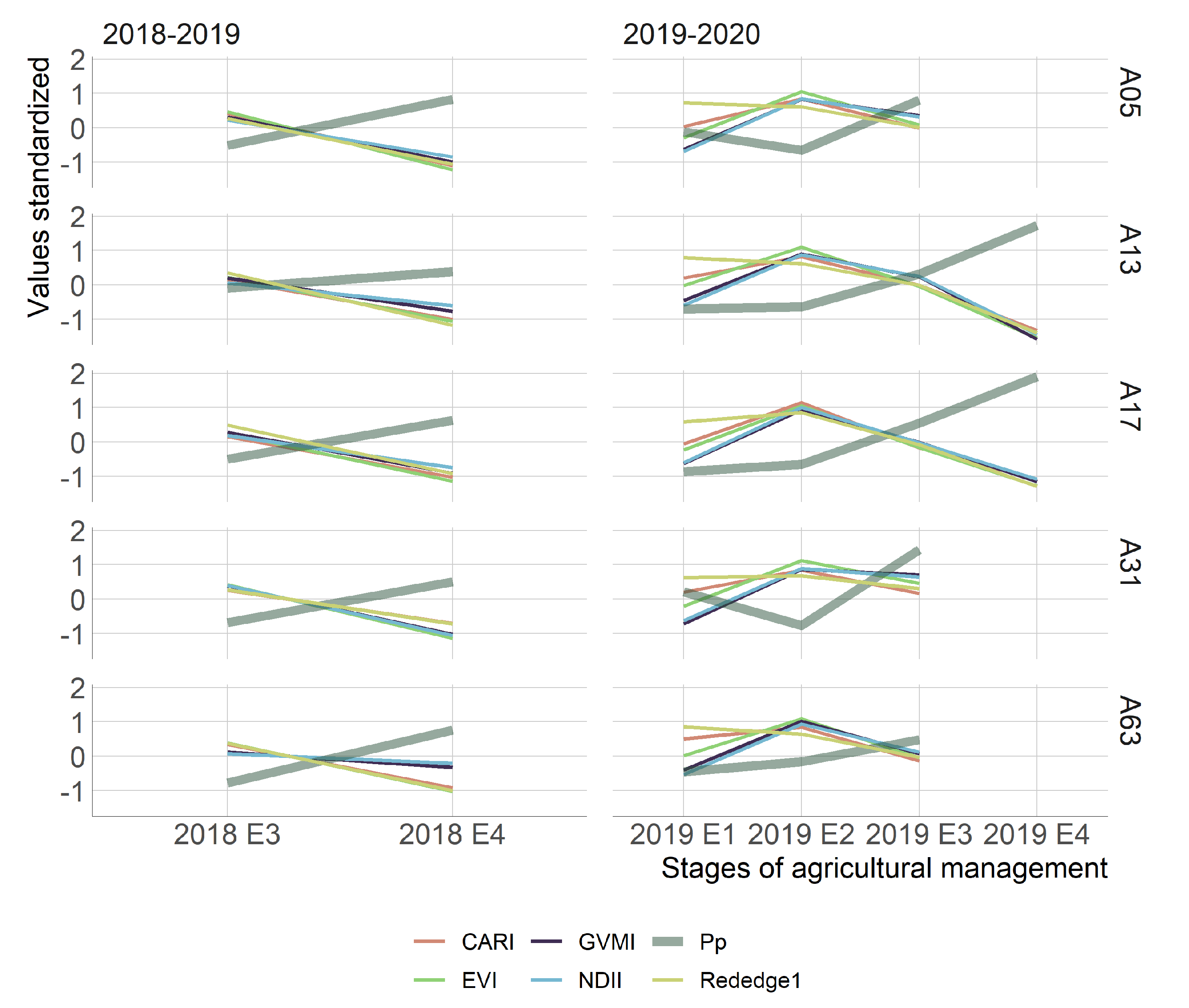
3.1. Exploratory Analysis
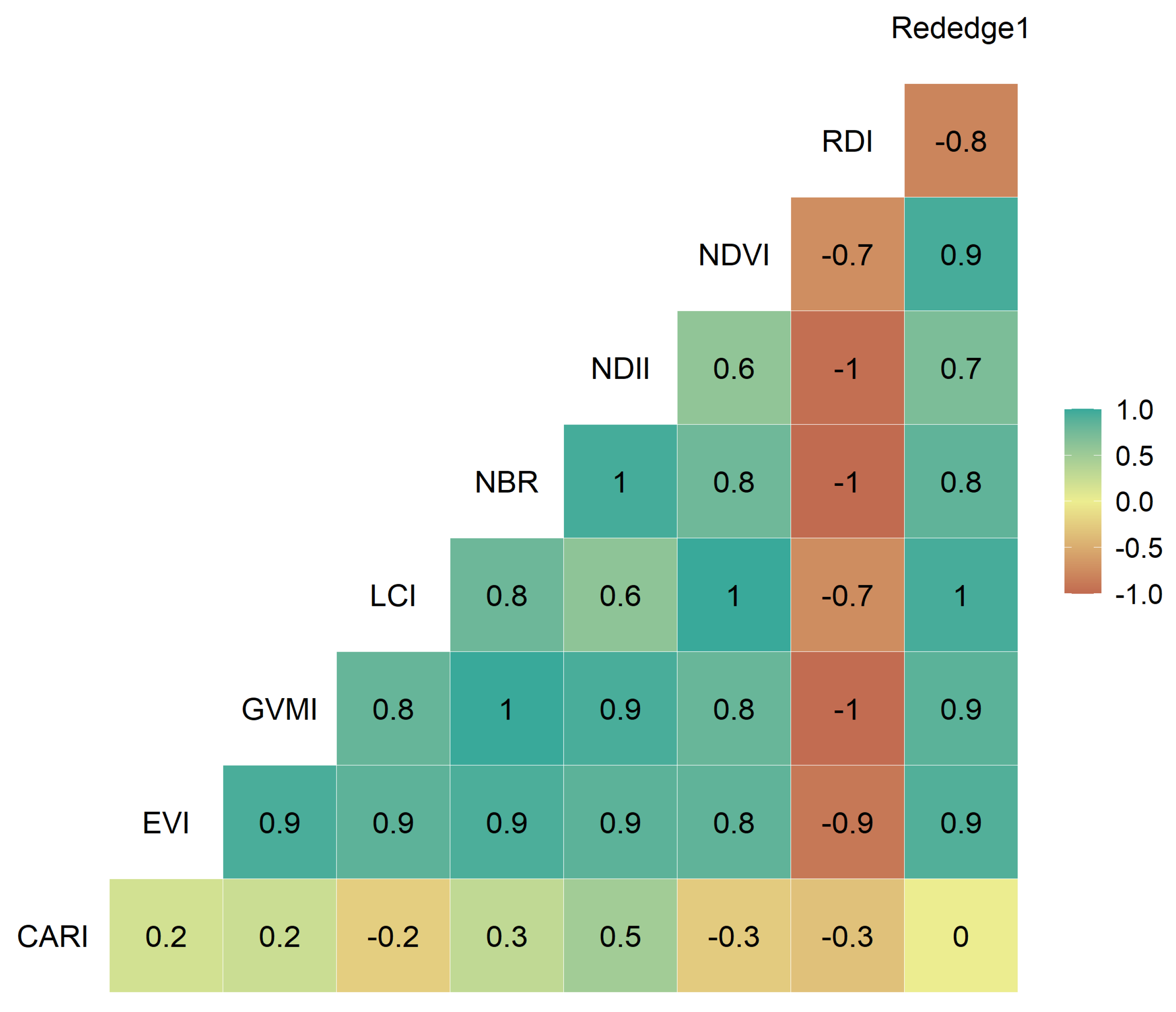
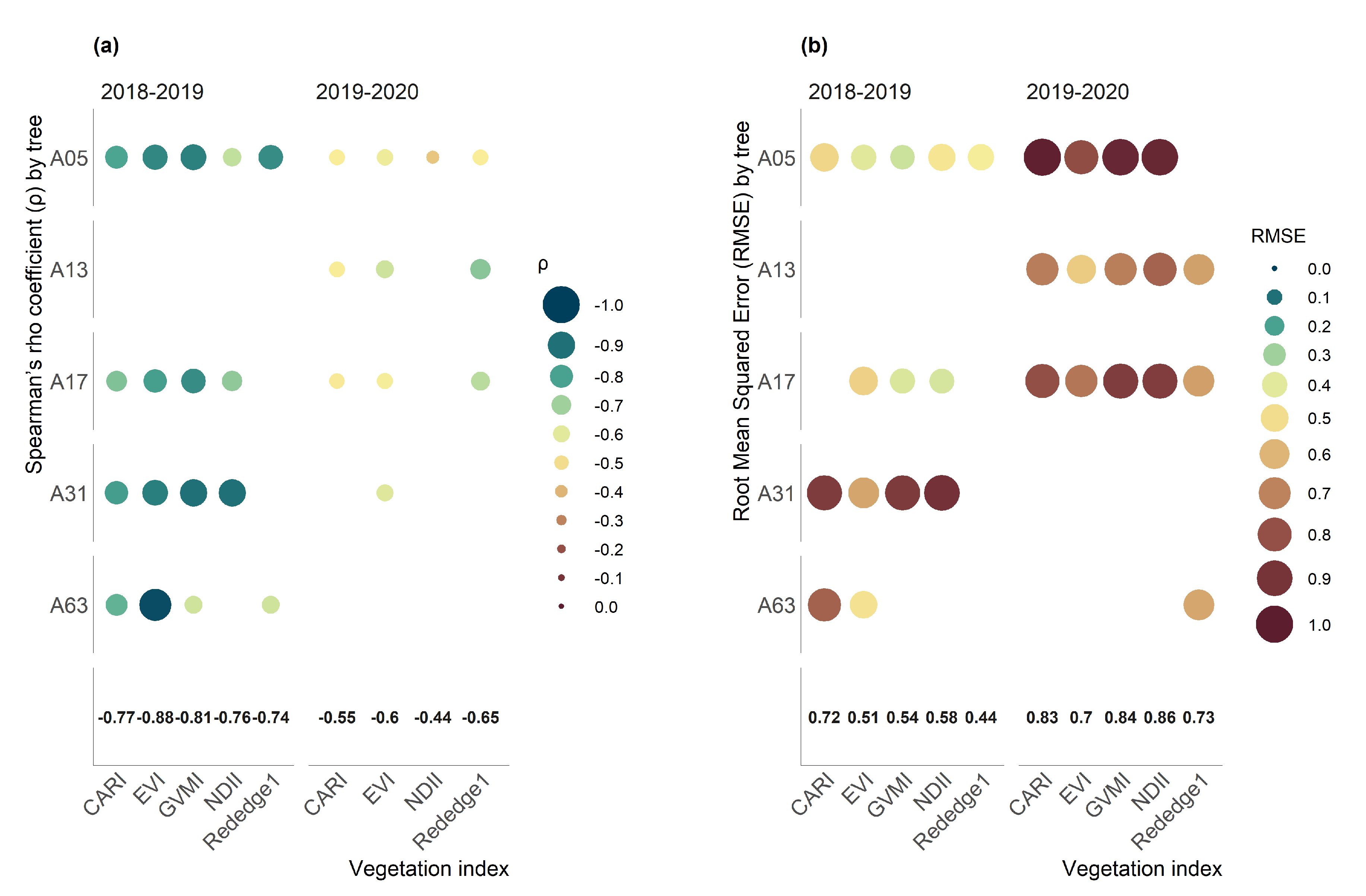
3.2. Correlation Analysis
4. Discussion
5. Conclusions
- From the nine vegetation indices studies, the CARI index was the one with the lowest temporal correlation (r = 0.17) between indices over the kiwifruit trees, which would be explained by the spectral behavior of the green band that is not found in the other indices.
- It was evidenced that continuous measurements of patch pressure (Pp) detected the temporary changes in the leaf’s hydric state, which were attributable both to the phenological behavior of the vegetation and agricultural management.
- The crop development highly influenced the performance of the vegetation indices through the season, which explains most of the changes in the water status on the canopy of kiwifruit. Nevertheless, the time-series of vegetation indices that obtained the highest Pp relationship were EVI and Rededge1 for the 2018–2019 and 2019–2020 seasons, respectively.
- The Rededge1 index was less sensitive to vegetative development, and its capacity to monitor the hydric status of the vegetation based on leaf turgor needs to be further investigated.
- Future research needs to address two main issues: (i) to be able to separate the temporal behavior of Vis due to vegetation development to aisle the changes due to the variation of water status in the plant, and (ii) explore with restricted levels of water supply on kiwifruit, which could be implemented with controlled deficit irrigation treatments.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CARI | Chlorophyll Absorption Ratio IndeX |
| Coefficient of determination | |
| EVI | Enhanced Vegetation Index (EVI) |
| ESA | European Space Agency (ESA) |
| ET | Evapotranspiration (ET) |
| GVMI | Global Vegetation Moisture Index (GVMI) |
| LAI | leaf area index (LAI) |
| LCI | Leaf Chlorophyll Index (LCI) |
| NIR | Near infrared (NIR) |
| NBR | Normalized burn ratio Index (NBR) |
| NDII | Normalized Difference Infrared Index (NDII) |
| NDVI | Normalized Difference Vegetation Index (NDVI) |
| NDWI | Normalized Difference Water Index (NDWI) |
| Pp | Patch pressure (Pp) |
| r | Pearson’s coefficient |
| RTM | Radiative transfer model (RTM) |
| RMSE | Root mean square error (RMSE) |
| SWIR | Short-wave infrared (SWIR) |
| SIWSI | Shortwave Infrared Water Stress Index |
| RDI | Simple Ratio MIR/NIR Ratio Drought Index |
| SPAC | Soil-plant-atmosphere continuum |
| Spearman’s non-parametric coefficient rho | |
| BOA | Bottom of the Atmosphere |
| VNIR | Visible and near infrared |
| WUE | Water use efficiency |
References
- Misra, A.K. Climate change and challenges of water and food security. Int. J. Sustain. Built Environ. 2014, 3, 153–165. [Google Scholar] [CrossRef]
- Lipper, L.; Thornton, P.; Campbell, B.M.; Baedeker, T.; Braimoh, A.; Bwalya, M.; Caron, P.; Cattaneo, A.; Garrity, D.; Henry, K.; et al. Climate-smart agriculture for food security. Nat. Clim. Chang. 2014, 4, 1068–1072. [Google Scholar] [CrossRef]
- Garreaud, R.D.; Alvarez-Garreton, C.; Barichivich, J.; Boisier, J.P.; Christie, D.; Galleguillos, M.; LeQuesne, C.; McPhee, J.; Zambrano-Bigiarini, M. The 2010–2015 megadrought in central Chile: Impacts on regional hydroclimate and vegetation. Hydrol. Earth Syst. Sci. 2017, 21, 6307–6327. [Google Scholar] [CrossRef]
- Zambrano, F.; Lillo-Saavedra, M.; Verbist, K.; Lagos, O. Sixteen years of agricultural drought assessment of the biobío region in chile using a 250 m resolution vegetation condition index (VCI). Remote Sens. 2016, 8, 530. [Google Scholar] [CrossRef]
- Zambrano, F.; Wardlow, B.; Tadesse, T.; Lillo-Saavedra, M.; Lagos, O. Evaluating satellite-derived long-term historical precipitation datasets for drought monitoring in Chile. Atmos. Res. 2017, 186, 26–42. [Google Scholar] [CrossRef]
- Zambrano, F.; Vrieling, A.; Nelson, A.; Meroni, M.; Tadesse, T. Prediction of drought-induced reduction of agricultural productivity in Chile from MODIS, rainfall estimates, and climate oscillation indices. Remote Sens. Environ. 2018, 219, 15–30. [Google Scholar] [CrossRef]
- Boisier, J.P.; Alvarez-Garretón, C.; Cordero, R.R.; Damiani, A.; Gallardo, L.; Garreaud, R.D.; Lambert, F.; Ramallo, C.; Rojas, M.; Rondanelli, R. Anthropogenic drying in central-southern Chile evidenced by long-term observations and climate model simulations. Elem. Sci. Anth. 2018, 6, 74. [Google Scholar] [CrossRef]
- Zambrano, F.; Molina, M.; Venegas, A.; Molina, J.; Vidal, P. Impact of Megadrought on Vegetation Productivity in Chile: Forest Lesser Resistant than Crops and Grassland. 2020. Available online: https://www.researchgate.net/publication/338801833_IMPACT_OF_MEGADROUGHT_ON_VEGETATION_PRODUCTIVITY_IN_CHILE_FOREST_LESSER_RESISTANT_THAN_CROPS_AND_GRASSLAND (accessed on 26 October 2020).
- Kirkham, M.B. Principles of Soil and Plant Water Relations; Elsevier Inc., 2005; Available online: https://www.sciencedirect.com/book/9780124200227/principles-of-soil-and-plant-water-relations (accessed on 26 October 2020). [CrossRef]
- Doorenbos, J.; Kassam, A.H. Yield Response to Water, FAO Irrigation and Drainage Paper 33; Food and Agriculture Organization of the United Nations: Rome, Italy, 1986. [Google Scholar]
- Nobel, P.S. Physicochemical and Environmental Plant Physiology; Elsevier Inc., 2009; Available online: https://www.sciencedirect.com/book/9780123741431/physicochemical-and-environmental-plant-physiology (accessed on 26 October 2020). [CrossRef]
- Scholander, P.F.; Hammel, H.T.; Bradstreet, E.D.; Hemmingsen, E.A. Sap pressure in vascular plants. Science 1965, 148, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Ehrenberger, W.; Rüger, S.; Rodríguez-Domínguez, C.M.; Díaz-Espejo, A.; Fernández, J.; Moreno, J.; Zimmermann, D.; Sukhorukov, V.L.; Zimmermann, U. Leaf patch clamp pressure probe measurements on olive leaves in a nearly turgorless state. Plant Biol. 2012, 14, 666–674. [Google Scholar] [CrossRef]
- Fernández, J.E. Plant-based sensing to monitor water stress: Applicability to commercial orchards. Agric. Water Manag. 2014, 142, 99–109. [Google Scholar] [CrossRef]
- Rodriguez-Dominguez, C.M.; Hernandez-Santana, V.; Buckley, T.N.; Fernández, J.E.; Diaz-Espejo, A. Sensitivity of olive leaf turgor to air vapour pressure deficit correlates with diurnal maximum stomatal conductance. Agric. For. Meteorol. 2019, 272–273, 156–165. [Google Scholar] [CrossRef]
- Westhoff, M.; Schneider, H.; Zimmermann, D.; Mimietz, S.; Stinzing, A.; Wegner, L.; Kaiser, W.; Krohne, G.; Shirley, S.; Jakob, P.; et al. The mechanisms of refilling of xylem conduits and bleeding of tall birch during spring. Plant Biol. 2008, 10, 604–623. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, U.; Bitter, R.; Marchiori, P.E.R.; Rüger, S.; Ehrenberger, W.; Sukhorukov, V.L.; Schüttler, A.; Ribeiro, R.V. A non-invasive plant-based probe for continuous monitoring of water stress in real time: A new tool for irrigation scheduling and deeper insight into drought and salinity stress physiology. Theor. Exp. Plant Physiol. 2013, 25, 2–11. [Google Scholar] [CrossRef]
- Beauzamy, L.; Nakayama, N.; Boudaoud, A. Flowers under Pressure: Ins and Outs of Turgor Regulation in Development. 2014. Available online: https://academic.oup.com/aob/article/114/7/1517/2769111 (accessed on 26 October 2020). [CrossRef]
- Jones, H.G. Plants and Microclimate: A Quantitative Approach to Environmental Plant Physiology; Cambridge University Press, 2013; Volume 9780521279, pp. 1–407. Available online: https://www.researchgate.net/publication/287238047_Plants_and_Microclimate_A_Quantitative_Approach_to_Environmental_Plant_Physiology (accessed on 26 October 2020). [CrossRef]
- Zimmermann, D.; Reuss, R.; Westhoff, M.; Geßner, P.; Bauer, W.; Bamberg, E.; Bentrup, F.W.; Zimmermann, U. A novel, non-invasive, online-monitoring, versatile and easy plant-based probe for measuring leaf water status. J. Exp. Bot. 2008, 59, 3157–3167. [Google Scholar] [CrossRef] [PubMed]
- Rüger, S.; Ehrenberger, W.; Arend, M.; Geßner, P.; Zimmermann, G.; Zimmerann, D.; Bentrup, F.W.; Nadler, A.; Raveh, E.; Sukhorukv, V.L.; et al. Comparative monitoring of temporal and spatial changes in tree water status using the non-invasive leaf patch clamp pressure probe and the pressure bomb. Agric. Water Manag. 2010, 98, 283–290. [Google Scholar] [CrossRef]
- Jacquemoud, S.; Ustin, S.L.; Verdebout, J.; Schmuck, G.; Andreoli, G.; Hosgood, B. Estimating leaf biochemistry using the PROSPECT leaf optical properties model. Remote Sens. Environ. 1996, 56, 194–202. [Google Scholar] [CrossRef]
- Penuelas, J.; Filella, I.; Biel, C.; Serrano, L.; Save, R. The reflectance at the 950–970 nm region as an indicator of plant water status. Int. J. Remote Sens. 1993, 14, 1887–1905. [Google Scholar] [CrossRef]
- Chávez, R.O.; Clevers, J.G.; Herold, M.; Ortiz, M.; Acevedo, E. Modelling the spectral response of the desert tree prosopis tamarugo to water stress. Int. J. Appl. Earth Obs. Geoinf. 2012, 21, 53–65. [Google Scholar] [CrossRef]
- Knapp, A.K.; Carter, G.A. Variability in leaf optical properties among 26 species from a broad range of habitats. Am. J. Botany 1998, 85, 940–946. [Google Scholar] [CrossRef]
- Ourcival, J.M.; Joffre, R.; Rambal, S. Exploring the relationships between reflectance and anatomical and biochemical properties in Quercus ilex leaves. New Phytol. 1999, 143, 351–364. [Google Scholar] [CrossRef]
- Colombo, R.; Meroni, M.; Marchesi, A.; Busetto, L.; Rossini, M.; Giardino, C.; Panigada, C. Estimation of leaf and canopy water content in poplar plantations by means of hyperspectral indices and inverse modeling. Remote Sens. Environ. 2008, 112, 1820–1834. [Google Scholar] [CrossRef]
- Bai, T.; Zhang, N.; Mercatoris, B.; Chen, Y. Jujube yield prediction method combining Landsat 8 Vegetation Index and the phenological length. Comput. Electron. Agric. 2019, 162, 1011–1027. [Google Scholar] [CrossRef]
- Karkauskaite, P.; Tagesson, T.; Fensholt, R. Evaluation of the plant phenology index (PPI), NDVI and EVI for start-of-season trend analysis of the Northern Hemisphere boreal zone. Remote Sens. 2017, 9, 485. [Google Scholar] [CrossRef]
- Raghavendra, B.R.; Mohammed Aslam, M.A. Sensitivity of vegetation indices of MODIS data for the monitoring of rice crops in Raichur district, Karnataka, India. Egypt. J. Remote Sens. Space Sci. 2017, 20, 187–195. [Google Scholar] [CrossRef]
- Xe, J.; Su, B. Significant remote sensing vegetation indices: A review of developments and applications. J. Sens. 2017, 2017. [Google Scholar] [CrossRef]
- Rouse, J.W.; Haas, R.H.; Schell, J.A.; Deering, D.W. Monitoring Vegetation Systems in the Great Plains with ERTS. 1973. Available online: https://ntrs.nasa.gov/citations/19740022614 (accessed on 26 October 2020).
- Huete, A.; Didan, K.; Miura, H.; Rodriguez, E.; Gao, X.; Ferreira, L. Overview of the radiometric and biopyhsical performance of the MODIS vegetation indices. Remote Sens. Environ. 2002, 83, 195–213. [Google Scholar] [CrossRef]
- Gerhards, M.; Schlerf, M.; Rascher, U.; Udelhoven, T.; Juszczak, R.; Alberti, G.; Miglietta, F.; Inoue, Y. Analysis of Airborne Optical and Thermal Imagery for Detection of Water Stress Symptoms. Remote Sens. 2018, 10, 1139. [Google Scholar] [CrossRef]
- Gerhards, M.; Schlerf, M.; Mallick, K.; Udelhoven, T. Challenges and Future Perspectives of Multi-/Hyperspectral Thermal Infrared Remote Sensing for Crop Water-Stress Detection: A Review. Remote Sens. 2019, 11, 1240. [Google Scholar] [CrossRef]
- Ji, L.; Zhang, L.; Wylie, B.K.; Rover, J. On the terminology of the spectral vegetation index (NIR − SWIR)/(NIR+SWIR). Int. J. Remote Sens. 2011, 32, 6901–6909. [Google Scholar] [CrossRef]
- Kim, D.M.; Zhang, H.; Zhou, H.; Du, T.; Wu, Q.; Mockler, T.C.; Berezin, M.Y. Highly sensitive image-derived indices of water-stressed plants using hyperspectral imaging in SWIR and histogram analysis. Sci. Rep. 2015. [Google Scholar] [CrossRef]
- Hardisky, M.A.; Klemas, V.; Smart, R.M. The Influence of Soil Salinity, Growth Form, and Leaf Moisture on the Spectral Radiance of Spartina alterniflora Canopies. Photogramm. Eng. Remote Sens. 1983, 49, 77–83. [Google Scholar]
- Gao, B.C. NDWI—A normalized difference water index for remote sensing of vegetation liquid water from space. Remote Sens. Environ. 1996, 58, 257–266. [Google Scholar] [CrossRef]
- Fensholt, R.; Sandholt, I. Derivation of a shortwave infrared water stress index from MODIS near- and shortwave infrared data in a semiarid environment. Remote Sens. Environ. 2003, 87, 111–121. [Google Scholar] [CrossRef]
- Xiao, X.; Hollinger, D.; Aber, J.; Goltz, M.; Davidson, E.A.; Zhang, Q.; Moore, B. Satellite-based modeling of gross primary production in an evergreen needleleaf forest. Remote Sens. Environ. 2004, 89, 519–534. [Google Scholar] [CrossRef]
- ESA. ESA - SENTINEL 2. 2015. Available online: http://www.esa.int/Applications/Observing_the_Earth/Copernicus/Sentinel-2 (accessed on 26 October 2020).
- Praticò, S.; Di Fazio, S.; Modica, G. Multi Temporal Analysis of Sentinel-2 Imagery for Mapping Forestry Vegetation Types: A Google Earth Engine Approach; Springer: Cham, Switzerland, 2021; pp. 1650–1659. [Google Scholar] [CrossRef]
- Vogelmann, J.E.; Xian, G.; Homer, C.; Tolk, B. Monitoring gradual ecosystem change using Landsat time series analyses: Case studies in selected forest and rangeland ecosystems. Remote Sens. Environ. 2012, 122, 92–105. [Google Scholar] [CrossRef]
- Zhang, M.; Gong, P.; Qi, S.; Liu, C.; Xiong, T. Mapping bamboo with regional phenological characteristics derived from dense Landsat time series using Google Earth Engine. Int. J. Remote Sens. 2019, 40, 9541–9555. [Google Scholar] [CrossRef]
- Frampton, W.J.; Dash, J.; Watmough, G.; Milton, E.J. Evaluating the capabilities of Sentinel-2 for quantitative estimation of biophysical variables in vegetation. ISPRS J. Photogramm. Remote Sens. 2013, 82, 83–92. [Google Scholar] [CrossRef]
- Beck, H.E.; Zimmermann, N.E.; McVicar, T.R.; Vergopolan, N.; Berg, A.; Wood, E.F. Present and future köppen-geiger climate classification maps at 1-km resolution. Sci. Data 2018, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- DGA. Pronóstico de Caudales de Deshielo Temporada de Riego 2019–2020; Technical Report; Dirección General de Aguas. Ministerio de Obras Públicas: Gobierno de Chile, Santiago, Chile, 2019. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Evapotranspiración del cultivo. arXiv 2006, arXiv:1011.1669v3. [Google Scholar]
- Sabaini, C.; Goecke, P. Hacia la produccion de un kiwi hayward más homogenéo y dulce. Fruticola 2013, 2, 17–23. [Google Scholar]
- Sabaini, C. Manejo Productivo del Kiwi Orientado a Obtener un Producto Rico y Homogéneo; Technical Report; Fedefruta, ASOEX, 2012; Available online: https://www.asoex.cl/seminario-kiwis-agosto-2012/finish/30-seminario-kiwis-agosto/223-manejo-productivo-del-kiwi-orientado-a-obtener-un-producto-rico-y-homogeneo.html (accessed on 26 October 2020).
- Ranghetti, L.; Boschetti, M.; Nutini, F.; Busetto, L. “sen2r”: An R toolbox for automatically downloading and preprocessing Sentinel-2 satellite data. Comput. Geosci. 2020, 139. [Google Scholar] [CrossRef]
- R Core Team. R: The R Project for Statistical Computing. 2020. Available online: https://www.r-project.org/ (accessed on 26 October 2020).
- Cloutis, E.A.; Connery, D.R.; Dover, F.J.; Major, D.J. Airborne multi-spectral monitoring of agricultural crop status: Effect of time of year, crop type and crop condition parameter. Int. J. Remote Sens. 1996, 17, 2579–2601. [Google Scholar] [CrossRef]
- Datt, B. Remote sensing of water content in Eucalyptus leaves. Aust. J. Bot. 1999, 47, 909–923. [Google Scholar] [CrossRef]
- Kim, M.S.; Daughtry, C.S.T.; Chappelle, E.W.; Mcmurtrey, J.E.; Walthall, C.L. The Use of High Spectral Resolution Bands for Estimating Absorbed Photosynthetically Active Radiation (A Par). Technical Report; 1994. Available online: https://ntrs.nasa.gov/citations/19950010604 (accessed on 26 October 2020).
- Key, C.H.; Benson, N.; Ohlen, D.; Howard, S.; McKinley, R.; Zhu, Z. The Normalized Burn Ratio and Relationships to Burn Severity. 2002. Available online: https://www.yumpu.com/en/document/view/24226870/the-normalized-burn-ratio-and-relationships-to-burn-severity- (accessed on 26 October 2020).
- Ceccato, P.; Flasse, S.; Grégoire, J.M. Designing a spectral index to estimate vegetation water content from remote sensing data: Part 1: Theoretical approach. Remote Sens. Environ. 2002, 82, 188–197. [Google Scholar] [CrossRef]
- Pinder, J.E., III; Mcleod, K.W. Indications of Relative Drought Stress in Longleaf Pine from Thematic Mapper Data. Photogramm. Eng. Remote Sens. 1999, 65, 495–501. [Google Scholar]
- Hijmans, R.J. Geographic Data Analysis and Modeling [R Package Raster Version 3.3-13]. 2020. Available online: https://rdrr.io/cran/raster/ (accessed on 26 October 2020).
- Becker, R.A.; Chambers, J.M.; Wilks, A.R. The New S Language: A Programming Environment for Data Analysis and Graphics; Wadsworth and Brooks/Cole Advanced Books & Software: Monterey, CA, USA, 1988. [Google Scholar]
- Pearson, K. Notes on the history of correlation. Biometrika 1920, 13, 25–45. [Google Scholar] [CrossRef]
- Spearman, C. The proof and measurement of association between two things. Am. J. Psychol. 1904, 15, 72–101. [Google Scholar] [CrossRef]
- Hahn, G.J. The Coefficient of Determination Exposed. Chem. Technol. 1973, 3, 609–612. [Google Scholar]
- Wilks, D.S. Statistical Methods in the Atmospheric Sciences, 2nd ed.; 2006; p. 649. Available online: https://www.scirp.org/(S(i43dyn45teexjx455qlt3d2q))/reference/ReferencesPapers.aspx?ReferenceID=1432882 (accessed on 26 October 2020).
- Keller, M. Phenology and Growth Cycle; 2020; pp. 61–103. Available online: https://www.sciencedirect.com/science/article/pii/B9780128163658000026?via%3Dihub (accessed on 26 October 2020). [CrossRef]
- Jensen, J.R. Remote Sensing of the Environment: An Earth Resource Perspective, 2nd ed.; 2014; Volume 1, pp. 333–378. Available online: https://www.amazon.com/Remote-Sensing-Environment-Resource-Perspective/dp/0131889508 (accessed on 26 October 2020).
- Van Beek, J.; Tits, L.; Somers, B.; Coppin, P. Stem Water Potential Monitoring in Pear Orchards through worldview-2 Multispectral Imagery. Remote Sens. 2013, 5, 6647–6666. [Google Scholar] [CrossRef]
- Lin, Y.; Zhu, Z.; Guo, W.; Sun, Y.; Yang, X.; Kovalskyy, V. Continuous Monitoring of Cotton Stem Water Potential using Sentinel-2 Imagery. Remote Sens. 2020, 12, 1176. [Google Scholar] [CrossRef]
- Ollinger, S.V.; Richardson, A.D.; Martin, M.E.; Hollinger, D.Y.; Frolking, S.E.; Reich, P.B.; Plourde, L.C.; Katul, G.G.; Munger, J.W.; Oren, R.; et al. Canopy nitrogen, carbon assimilation, and albedo in temperate and boreal forests: Functional relations and potential climate feedbacks. Proc. Natl. Acad. Sci. USA 2008, 105, 19336–19341. [Google Scholar] [CrossRef] [PubMed]
- Soria-Ruiz, J.; Fernandez-Ordonez, Y.; McNair, H. Corn Monitoring and Crop Yield Using Optical and Microwave Remote Sensing. Geosci. Remote Sens. 2009. [Google Scholar] [CrossRef]
- Vreugdenhil, M.; Wagner, W.; Bauer-Marschallinger, B.; Pfeil, I.; Teubner, I.; Rüdiger, C.; Strauss, P. Sensitivity of Sentinel-1 backscatter to vegetation dynamics: An Austrian case study. Remote Sens. 2018, 10, 1396. [Google Scholar] [CrossRef]
- Ihuoma, S.O.; Madramootoo, C.A. Recent Advances in Crop Water Stress Detection. 2017. Available online: https://www.sciencedirect.com/science/article/abs/pii/S0168169916310766 (accessed on 26 October 2020). [CrossRef]
- Clarke, T.R.; Moran, M.S.; Inoue, Y.; Vidal, A. Estimating crop water defficiency using the relation between surface minus air temperature and spectral vegetation index. Remote Sens. Environ. 1994, 49, 246–263. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Pons-Fernández, X.; Cuadrat-Prats, J.M. Mapping soil moisture in the central Ebro river valley (northeast Spain) with Landsat and NOAA satellite imagery: A comparison with meteorological data. Int. J. Remote Sens. 2004, 25, 4325–4350. [Google Scholar] [CrossRef]
- Wang, K.; Li, Z.; Cribb, M. Estimation of evaporative fraction from a combination of day and night land surface temperatures and NDVI: A new method to determine the Priestley-Taylor parameter. Remote Sens. Environ. 2006, 102, 293–305. [Google Scholar] [CrossRef]


| Management Stage | Start | End | Description |
|---|---|---|---|
| E1: Load regulation | 15 September | 15 November | It begins about 10 days before budding with the start of floral differentiation. Flower buds develop until flowering. Pruning and thinning work is carried out |
| E2: Pollination | 15 November | 7 January | It begins with the pollination of the flower. The fruit develops and grows at high rates reaching 50% of its weight and 70% of its final volume. In this stage the vegetation reaches its highest water demand and the temperature reaches its maximum |
| E3: Vegetation management | 7 January | 15 March | Green pruning is carried out, corresponding to the removal of shoots and foliage in order to improve the distribution and penetration of light in the crown |
| E4: Fruit harvest | 15 March | 15 April | The maturity and harvest of fruit is ensured, it is the shortest stage and extends from the third week of March to mid-April. |
| Bands | Spatial Resolution (m) | Central Wavelength (m) (A;B) |
|---|---|---|
| B1 - Coastal aerosol | 60 | 442.7; 442.2 |
| B2 - Blue | 10 | 492.4; 492.1 * |
| B3 - Green | 10 | 559.8; 559.0 * |
| B4 - Red | 10 | 664.6; 664.9 * |
| B5 - Red Edge | 20 | 704.1; 703.8 * |
| B6 - Red Edge | 20 | 740.5; 739.1 |
| B7 - Red Edge | 20 | 782.8; 779.7 |
| B8 - NIR | 10 | 832.8; 832.9 * |
| B8A - Red Edge | 20 | 864.7; 864.0 |
| B9 - Water vapour | 60 | 945.1; 943.2 * |
| B10 - SWIR - Cirrus | 60 | 1373.5; 1376.9 |
| B11 - SWIR | 20 | 1613.7; 1610.4 * |
| B12 - SWIR | 20 | 2202.4; 2185.7 * |
| Tree | Number of Yara Water Sensors | |
|---|---|---|
| 2018–2019 | 2019–2020 | |
| A05 | 2 | 2 |
| A13 | 2 | 2 |
| A17 | 3 | 1 |
| A31 | 2 | 2 |
| A63 | 2 | 2 |
| Wavelength | Vegetation Index | Formula | Reference |
|---|---|---|---|
| VNIR | Enhanced Vegetation Index (EVI) | [33] | |
| VNIR | Red edge 1 (Rededge1) | [54] | |
| VNIR | Leaf Chlorophyll Index (LCI) | [55] | |
| VNIR | Normalized Difference Vegetation Index (NDVI) | [32] | |
| VNIR | Chlorophyll Absorption Ratio Index (CARI) | [56] | |
| VNIR-SWIR | Normalized Difference Infrared Index (NDII) | [38] | |
| VNIR-SWIR | Normalized burn ratio Index (NBR) | [57] | |
| VNIR-SWIR | Global Vegetation Moisture Index (GVMI) | [58] | |
| VNIR-SWIR | Simple Ratio MIR/NIR Ratio Drought Index (RDI) | [59] |
| 2018–2019 | 2019–2020 | |||||
|---|---|---|---|---|---|---|
| Tree | E3 | E4 | E1 | E2 | E3 | E4 |
| A05 | 0.42 | 0.25 | 0.56 | 0.14 | 0.31 | - |
| A13 | 1.34 | 0.64 | 0.11 | 0.04 | 0.12 | 0.05 |
| A17 | 0.38 | 0.90 | - | - | - | - |
| A31 | 0.40 | 0.54 | 0.89 | 0.42 | 0.31 | - |
| A63 | 0.30 | 0.14 | 0.51 | 0.22 | 1.43 | - |
| Mean | 0.54 | 0.49 | 0.52 | 0.20 | 0.54 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jopia, A.; Zambrano, F.; Pérez-Martínez, W.; Vidal-Páez, P.; Molina, J.; de la Hoz Mardones, F. Time-Series of Vegetation Indices (VNIR/SWIR) Derived from Sentinel-2 (A/B) to Assess Turgor Pressure in Kiwifruit. ISPRS Int. J. Geo-Inf. 2020, 9, 641. https://doi.org/10.3390/ijgi9110641
Jopia A, Zambrano F, Pérez-Martínez W, Vidal-Páez P, Molina J, de la Hoz Mardones F. Time-Series of Vegetation Indices (VNIR/SWIR) Derived from Sentinel-2 (A/B) to Assess Turgor Pressure in Kiwifruit. ISPRS International Journal of Geo-Information. 2020; 9(11):641. https://doi.org/10.3390/ijgi9110641
Chicago/Turabian StyleJopia, Alberto, Francisco Zambrano, Waldo Pérez-Martínez, Paulina Vidal-Páez, Julio Molina, and Felipe de la Hoz Mardones. 2020. "Time-Series of Vegetation Indices (VNIR/SWIR) Derived from Sentinel-2 (A/B) to Assess Turgor Pressure in Kiwifruit" ISPRS International Journal of Geo-Information 9, no. 11: 641. https://doi.org/10.3390/ijgi9110641
APA StyleJopia, A., Zambrano, F., Pérez-Martínez, W., Vidal-Páez, P., Molina, J., & de la Hoz Mardones, F. (2020). Time-Series of Vegetation Indices (VNIR/SWIR) Derived from Sentinel-2 (A/B) to Assess Turgor Pressure in Kiwifruit. ISPRS International Journal of Geo-Information, 9(11), 641. https://doi.org/10.3390/ijgi9110641






