EEG & Eye Tracking User Experiments for Spatial Memory Task on Maps
Abstract
1. Introduction
2. Methodology
2.1. Experiment 1
2.2. Experiment 2
2.1.1. Participants
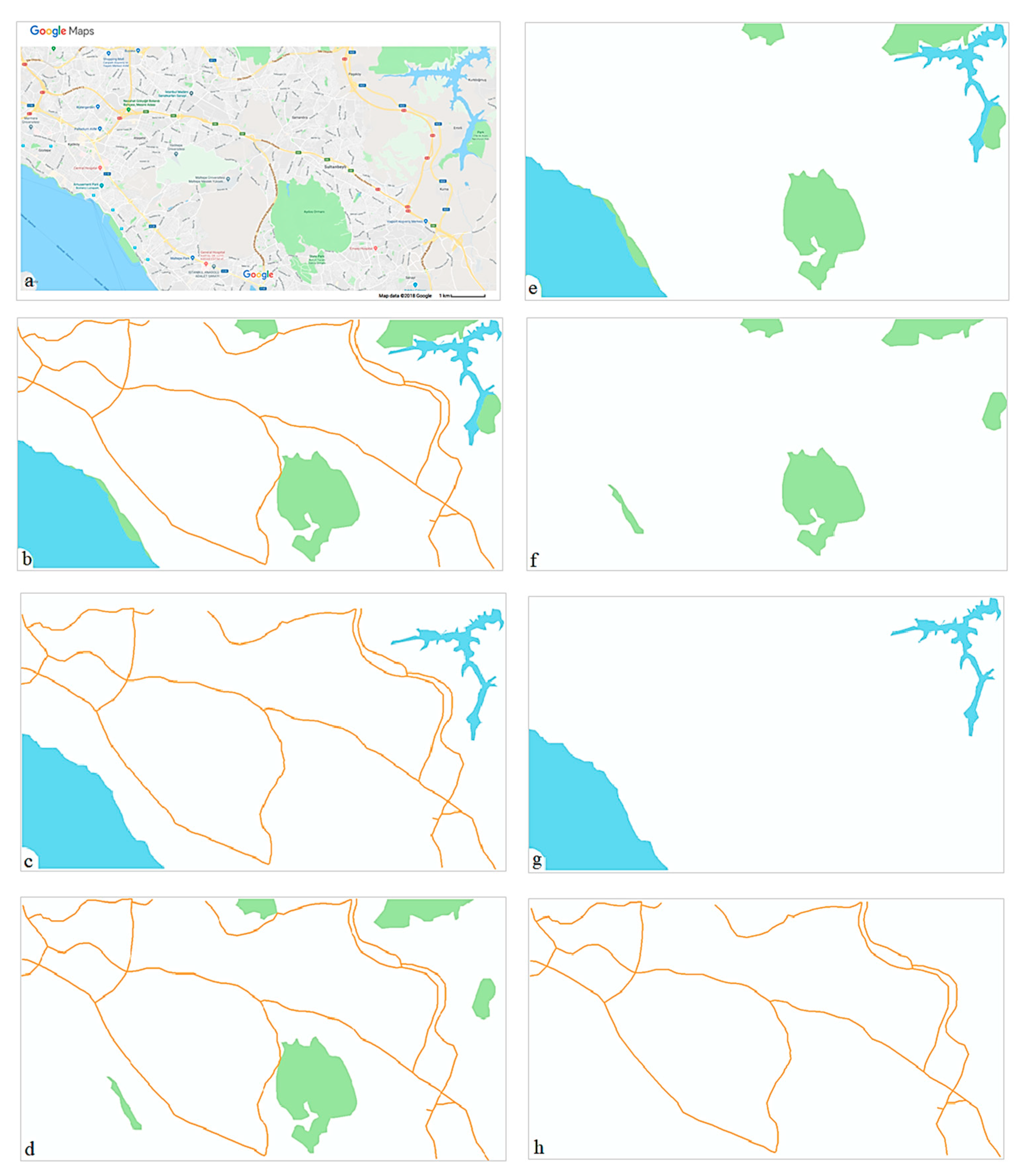
2.2.2. Task and Stimuli
2.2.3. Procedures
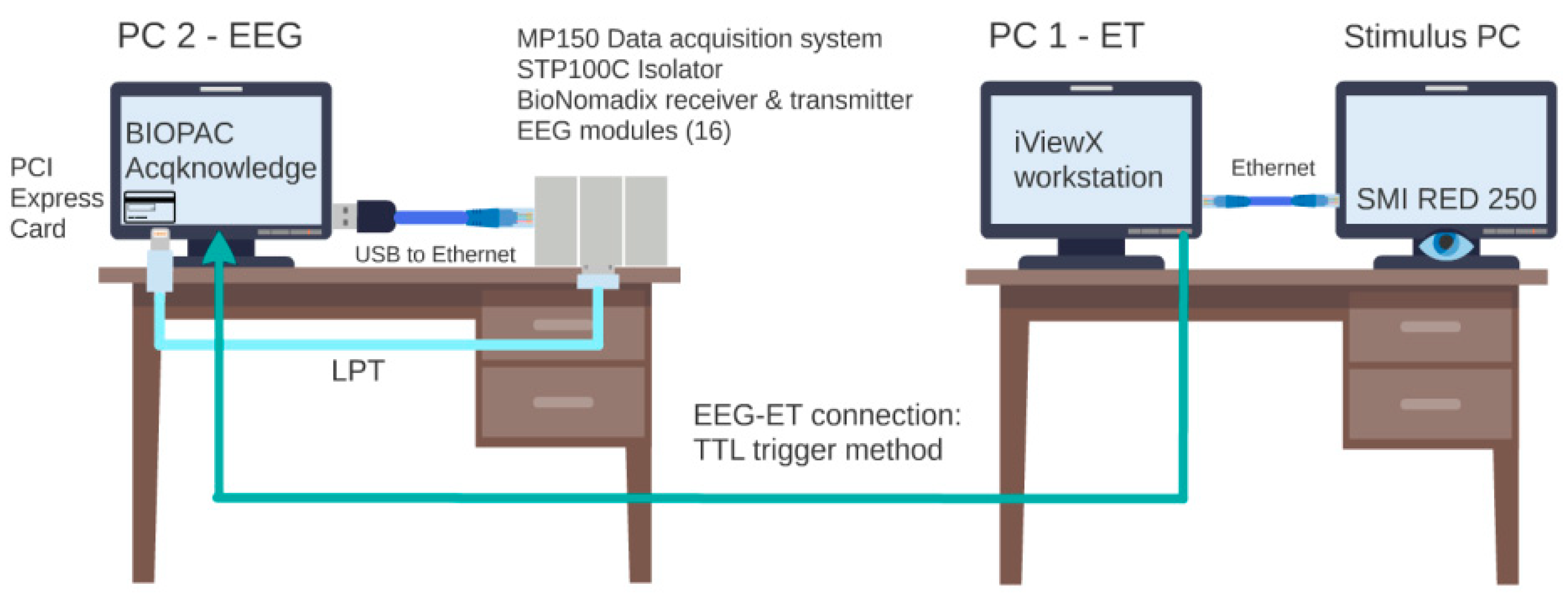
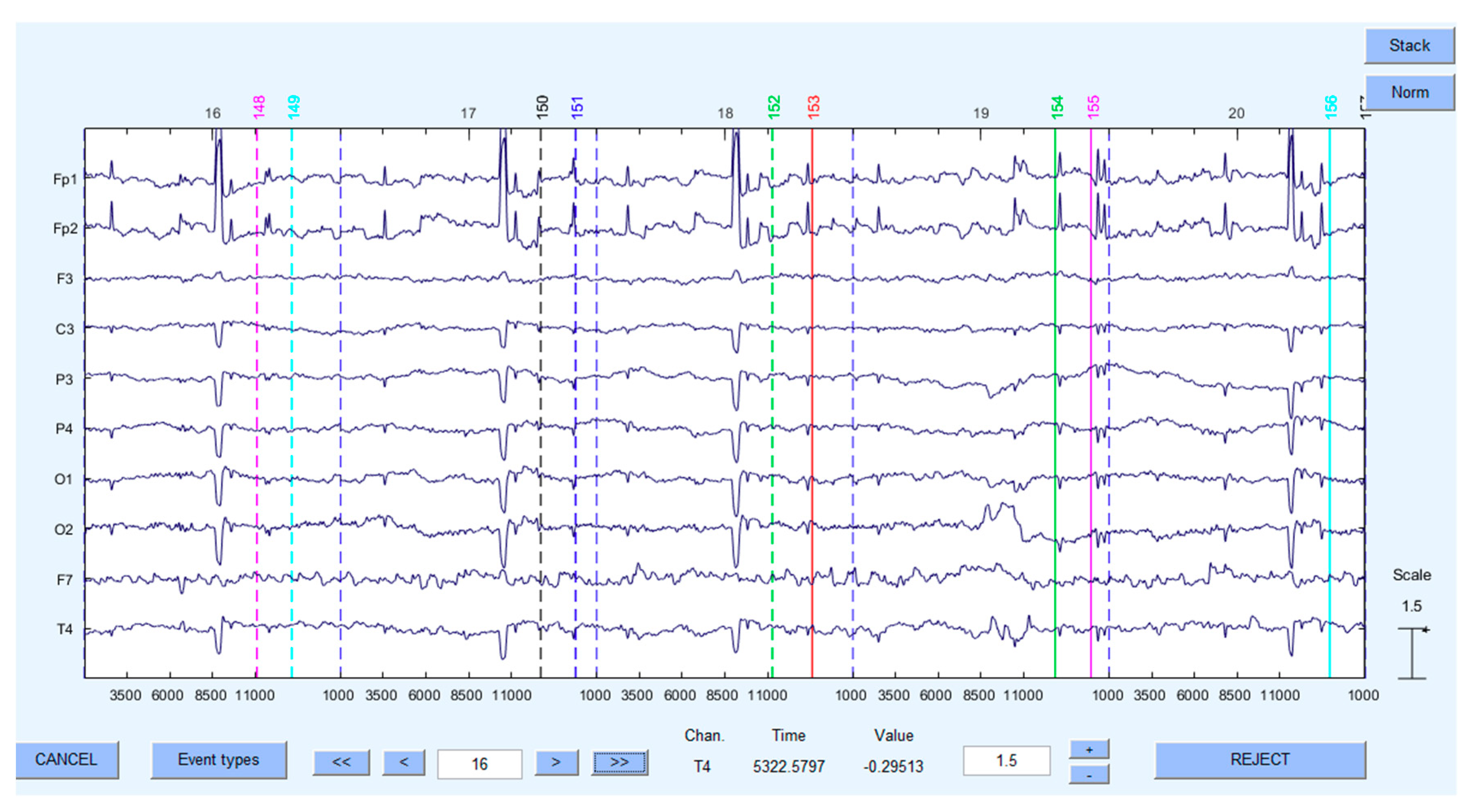
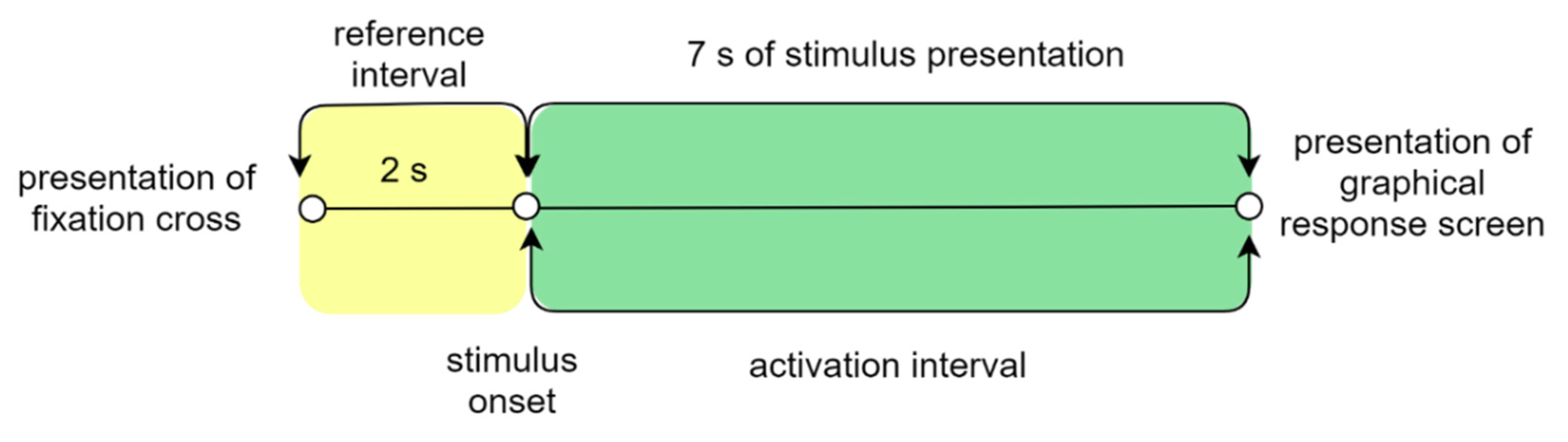
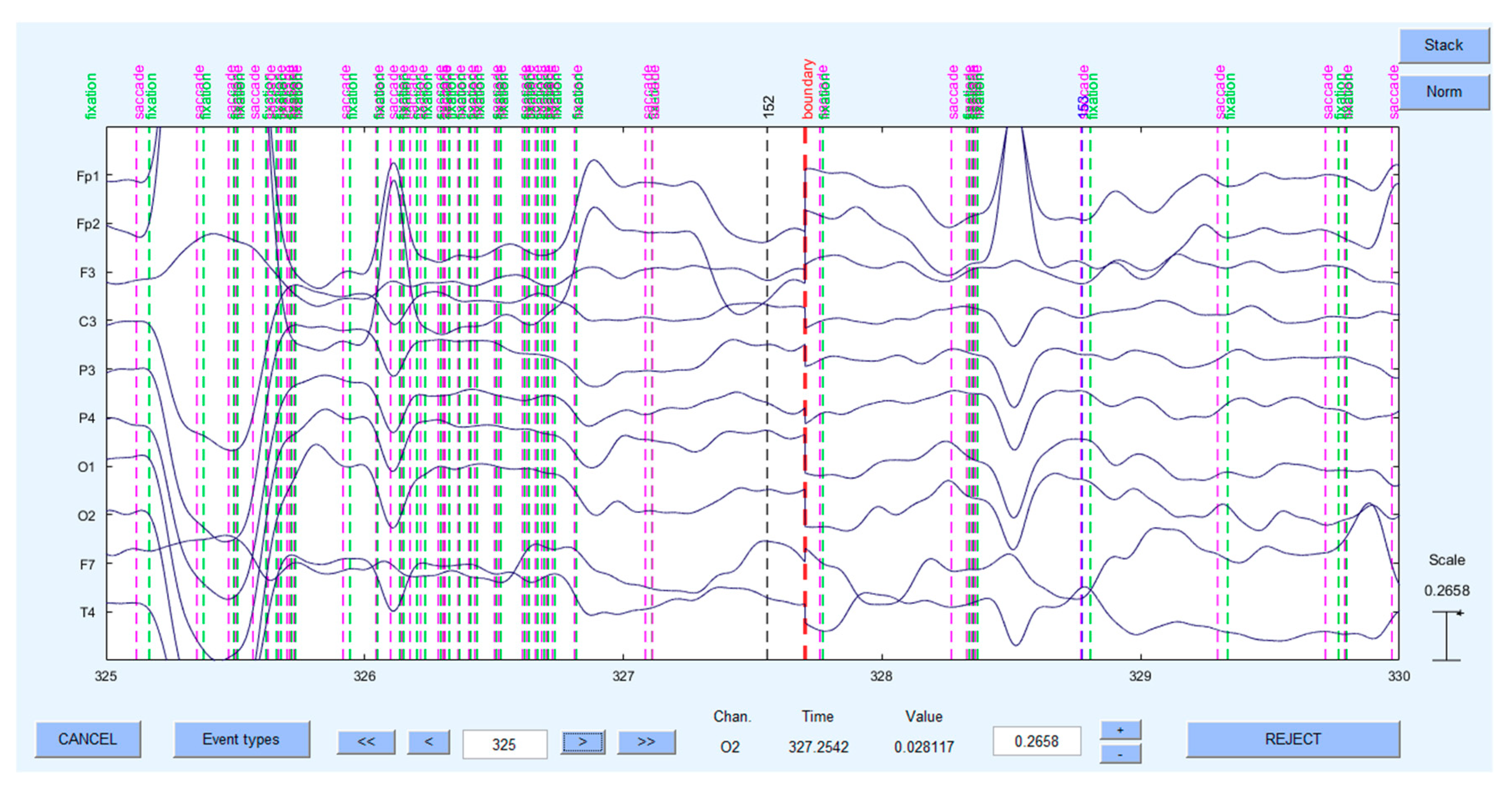
2.2.4. Psychological Measures to Use: ET & EEG Metrics
- [0 2] s for the events in the reference interval—fixation crosses
- [0 7] s for the events in the activation interval—map stimuli
3. Results
3.1. Experiment 1
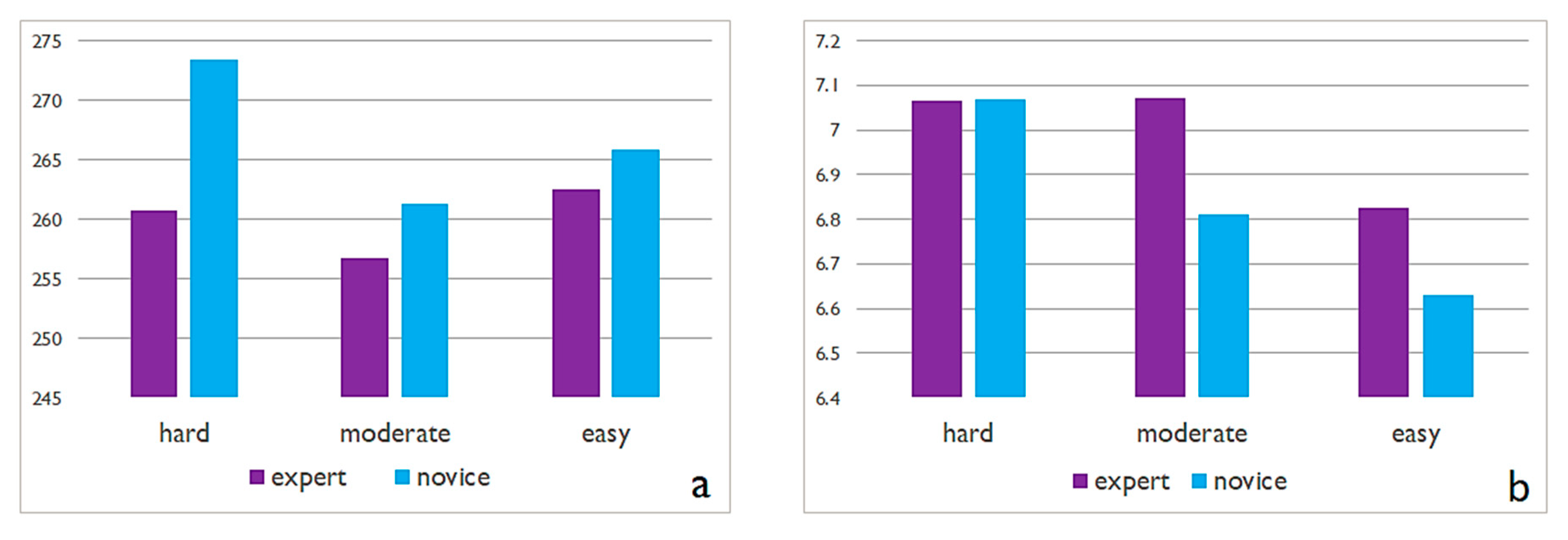
3.2. Experiment 2
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Griffin, A. Cartography, Visual Perception and Cognitive Psychology. In The Routledge Handbook of Mapping and Cartography; Kent, A.J., Vujakovic, P., Eds.; Taylor & Francis: London, UK, 2017; pp. 44–54. [Google Scholar]
- Eckert, D.M. On the Nature of Maps and Map Logic. Cartogr. Int. J. Geogr. Inf. Geovisualization 1977, 14, 01–07. [Google Scholar] [CrossRef]
- Atkinson, R.C.; Shiffrin, R.M. Human Memory: A Proposed System and Its Control Processes. In Psychology of Learning and Motivation; Spence, K.W., Spence, J.T., Eds.; Academic Press: New York, USA, 1968; Volume 2, pp. 89–195. [Google Scholar] [CrossRef]
- Keskin, M.; Ooms, K.; Dogru, A.O.; De Maeyer, P. Digital Sketch Maps and Eye Tracking Statistics as Instruments to Obtain Insights into Spatial Cognition. J. Eye Mov. Res. 2018, 11. [Google Scholar] [CrossRef]
- Bertin, J. Semiology of Graphics (Translation by WJ Berg 1983); University of Wisconsin Press: Madison, WI, USA, 1967. [Google Scholar]
- Herbert, G.; Chen, X. A Comparison of Usefulness of 2D and 3D Representations of Urban Planning. Cartogr. Geogr. Inf. Sci. 2015, 42, 22–32. [Google Scholar] [CrossRef]
- Kveladze, I.; Kraak, M.-J.; van Elzakker, C.P.J.M. The Usability of a GeoVisual Analytics Environment for the Exploration and Analysis of Different Datasets. In Advances in Cartography and GIScience; Peterson, M.P., Ed.; Lecture Notes in Geoinformation and Cartography; Springer International Publishing: New York City, NY, USA, 2017; pp. 207–221. [Google Scholar]
- Ooms, K.; Maeyer, P.D.; Dupont, L.; Veken, N.V.D.; de Weghe, N.V.; Verplaetse, S. Education in Cartography: What Is the Status of Young People’s Map-Reading Skills? Cartogr. Geogr. Inf. Sci. 2016, 43, 134–153. [Google Scholar] [CrossRef]
- Duchowski, A.T. Case Studies. In Eye Tracking Methodology: Theory and Practice; Duchowski, A.T., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 225–244. [Google Scholar] [CrossRef]
- Jacob, R.J.K.; Karn, K.S. Commentary on Section 4—Eye Tracking in Human-Computer Interaction and Usability Research: Ready to Deliver the Promises. In The Mind’s Eye; Hyönä, J., Radach, R., Deubel, H., Eds.; North-Holland: Amsterdam, The Netherlands, 2003; pp. 573–605. [Google Scholar] [CrossRef]
- Çöltekin, A.; Fabrikant, S.I.; Lacayo, M. Exploring the Efficiency of Users’ Visual Analytics Strategies Based on Sequence Analysis of Eye Movement Recordings. Int. J. Geogr. Inf. Sci. 2010, 24, 1559–1575. [Google Scholar] [CrossRef]
- Ooms, K. Maps, How Do Users See Them?: An in Depth Investigation of the Map Users’ Cognitive Processes. Doctoral Thesis, Ghent University: Ghent, Belgium, 2012. [Google Scholar]
- Kimerling, A.J.; Buckley, A.R.; Muehrcke, P.C.; Muehrcke, J.O. Map Use: Reading and Analysis, 6th ed.; Esri Press: Redlands, CA, USA, 2009. [Google Scholar]
- MacEachren, A.M. How Maps Work: Representation, Visualization, and Design; Guilford Press: New York, NY, USA, 2004. [Google Scholar]
- Gedminas, L. Evaluating Hurricane Advisories Using Eye-Tracking and Biometric Data. Master’s Thesis, East Carolina University, Greenville, NC, USA, 2011. [Google Scholar]
- Togami, H. Affects on visual search performance of individual differences in fixation time and number of fixations. Ergonomics 1984, 27, 789–799. [Google Scholar] [CrossRef]
- Bigne, E.; Llinares, C.; Torrecilla, C. Elapsed time on first buying triggers brand choices within a category: A virtual reality-based study. J. Bus. Res. 2016, 69, 1423–1427. [Google Scholar] [CrossRef]
- Gevins, A.; Smith, M.E. Neurophysiological Measures of Working Memory and Individual Differences in Cognitive Ability and Cognitive Style. Cereb. Cortex. 2000, 10, 829–839. [Google Scholar] [CrossRef]
- Witvoet, J. Does Cognitive Load Influence Performance in a Game-Based Learning Task? Available online: https://essay.utwente.nl/64203/ (accessed on 30 September 2019).
- Klimesch, W. EEG Alpha and Theta Oscillations Reflect Cognitive and Memory Performance: A Review and Analysis. Brain Res. Rev. 1999, 29, 169–195. [Google Scholar] [CrossRef]
- Kumar, N.; Kumar, J. Measurement of Cognitive Load in HCI Systems Using EEG Power Spectrum: An Experimental Study. Procedia Comput. Science 2016, 84, 70–78. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Da Silva, F.L. Event-Related EEG/MEG Synchronization and Desynchronization: Basic Principles. Clin. Neurophysiol. 1999, 110, 1842–1857. [Google Scholar] [CrossRef]
- Guay, S.; De Beaumont, L.; Drisdelle, B.L.; Lina, J.-M.; Jolicoeur, P. Electrophysiological Impact of Multiple Concussions in Asymptomatic Athletes: A Re-Analysis Based on Alpha Activity during a Visual-Spatial Attention Task. Neuropsychologia 2018, 108, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Coan, J.A.; Allen, J.J. State and Trait of Frontal EEG Asymmetry in Emotion. In The Asymmetrical Brain; Hugdahl, K., Davidson, R.J., Eds.; MIT Press: Cambridge, MA, USA, 2003; pp. 566–615. [Google Scholar]
- Harmon-Jones, E. Clarifying the Emotive Functions of Asymmetrical Frontal Cortical Activity. Psychophysiology 2003, 40, 838–848. [Google Scholar] [CrossRef] [PubMed]
- Lanini-Maggi, S. Depicting Movement Data with Animations for Embodied and Real-Time Decision-Making: A User Study with Air Traffic Control Displays and Real-Time Movement Data. Ph.D. Thesis, University of Zurich, Zurich, Switzerland, 2017. [Google Scholar]
- Quaedflieg, C.W.E.M.; Smulders, F.T.Y.; Meyer, T.; Peeters, F.; Merckelbach, H.; Smeets, T. The Validity of Individual Frontal Alpha Asymmetry EEG Neurofeedback. Soc. Cogn. Affect. Neurosci. 2016, 11, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Adolph, D.; von Glischinski, M.; Wannemüller, A.; Margraf, J. The Influence of Frontal Alpha-Asymmetry on the Processing of Approach- and Withdrawal-Related Stimuli—A Multichannel Psychophysiology Study. Psychophysiology 2017, 54, 1295–1310. [Google Scholar] [CrossRef]
- Smith, E.E.; Reznik, S.J.; Stewart, J.L.; Allen, J.J.B. Assessing and Conceptualizing Frontal EEG Asymmetry: An Updated Primer on Recording, Processing, Analyzing, and Interpreting Frontal Alpha Asymmetry. Int. J. Psychophysiol. 2017, 111, 98–114. [Google Scholar] [CrossRef] [PubMed]
- Davidson, R.J. Affect, cognition, and hemispheric specialization. In Emotion, Cognition, and Behavior; Izard, C.E., Kagan, J., Zajonc, R.B., Eds.; Cambridge University Press: Cambridge, UK, 1984; pp. 320–365. [Google Scholar]
- Tomarken, A.J.; Davidson, R.J.; Wheeler, R.E.; Doss, R.C. Individual Differences in Anterior Brain Asymmetry and Fundamental Dimensions of Emotion. J. Pers. Soc. Psychol. 1992, 62, 676. [Google Scholar] [CrossRef]
- Davidson, R.J. Affective Style and Affective Disorders: Perspectives from Affective Neuroscience. Cogn. Emot. 1998, 12, 307–330. [Google Scholar] [CrossRef]
- Coan, J.A.; Allen, J.J.B.; McKnight, P.E. A Capability Model of Individual Differences in Frontal EEG Asymmetry. Biol. Psychol. 2006, 72, 198–207. [Google Scholar] [CrossRef]
- Edler, D.; Bestgen, A.K.; Kuchinke, L.; Dickmann, F. Grids in topographic maps reduce distortions in the recall of learned object locations. PLoS ONE 2014, 9, e98148. [Google Scholar] [CrossRef]
- Ooms, K.; De Maeyer, P.; Fack, V. Listen to the map user: Cognition, memory, and expertise. Cartogr. J. 2015, 52, 3–19. [Google Scholar] [CrossRef]
- Anderson, E.W.; Potter, K.C.; Matzen, L.E.; Shepherd, J.F.; Preston, G.A.; Silva, C.T. A User Study of Visualization Effectiveness Using EEG and Cognitive Load. In Computer Graphics Forum; Blackwell Publishing: Oxford, UK, 2011; Volume 30, pp. 791–800. [Google Scholar]
- Gevins, A.; Smith, M.E.; McEvoy, L.; Yu, D. High-Resolution EEG Mapping of Cortical Activation Related to Working Memory: Effects of Task Difficulty, Type of Processing, and Practice. Cereb. Cortex. 1997, 7, 374–385. [Google Scholar] [CrossRef] [PubMed]
- Gevins, A.; Smith, M.E. Neurophysiological Measures of Cognitive Workload during Human-Computer Interaction. Theor. Issues Ergon. Sci. 2003, 4, 113–131. [Google Scholar] [CrossRef]
- Sauseng, P.; Klimesch, W.; Schabus, M.; Doppelmayr, M. Fronto-Parietal EEG Coherence in Theta and Upper Alpha Reflect Central Executive Functions of Working Memory. Int. J. Psychophysiol. 2005, 57, 97–103. [Google Scholar] [CrossRef]
- Fink, A.; Grabner, R.H.; Benedek, M.; Neubauer, A.C. Divergent Thinking Training Is Related to Frontal Electroencephalogram Alpha Synchronization. Eur. J. Neurosci. 2006, 23, 2241–2246. [Google Scholar] [CrossRef]
- Neubauer, A.C.; Fink, A. Intelligence and Neural Efficiency. Neurosci. Biobehav. Rev. 2009, 33, 1004–1023. [Google Scholar] [CrossRef]
- Delorme, A.; Makeig, S. EEGLAB Tutorial Outline Chapter 1: Loading Data in EEGLAB; Swartz Center for Computational Neuroscience (SCCN): San Diego, CA, USA, 2012. [Google Scholar]
- Dimigen, O.; Sommer, W.; Hohlfeld, A.; Jacobs, A.M.; Kliegl, R. Coregistration of Eye Movements and EEG in Natural Reading: Analyses and Review. J. Exp. Psychol. Gen. 2011, 140, 552–572. [Google Scholar] [CrossRef]
- Chuang, C.-H.; Cao, Z.; King, J.-T.; Wu, B.-S.; Wang, Y.-K.; Lin, C.-T. Brain Electrodynamic and Hemodynamic Signatures Against Fatigue During Driving. Front. Neurosci. 2018, 12, 181. [Google Scholar] [CrossRef]
- Benedek, M.; Schickel, R.J.; Jauk, E.; Fink, A.; Neubauer, A.C. Alpha Power Increases in Right Parietal Cortex Reflects Focused Internal Attention. Neuropsychologia 2014, 56, 393–400. [Google Scholar] [CrossRef]
- Vlaskamp, B.N.S.; Hooge, I.T.C. Crowding degrades saccadic search performance. Vis. Res. 2006, 46, 417–425. [Google Scholar] [CrossRef]
- Dimigen, O.; Reinacher, U. A matlab tool for simultaneous eye tracking and eeg. In Journal of Cognitive Neuroscience; MIT Press: Cambridge, MA, USA, 2013; pp. 143–144. [Google Scholar]
- Nikolaev, A.R.; Meghanathan, R.N.; van Leeuwen, C. Combining EEG and Eye Movement Recording in Free Viewing: Pitfalls and Possibilities. Brain Cogn. 2016, 107, 55–83. [Google Scholar] [CrossRef] [PubMed]
| Experiment 1 | Experiment 2 | |
|---|---|---|
| Research Question | How does cognitive load vary between experts and novices while memorizing the main structuring elements of a map stimulus without any time constraints? | How does cognitive load vary between experts and novices while memorizing a (part of) map content in a limited study period? How does the complexity/difficulty of the task influence the cognitive load? |
| Goal | To evaluate the cognitive processes, abilities and/or limitations of map users when they first study a 2D static map and retrieve this information later. | To test the effect of task difficulty on behavior, which is the retrieval of the main structuring elements with varying levels. |
| Hypothesis | We expect that the spatial memory task will cause higher cognitive load in novices compared to experts. | The tasks involving the retrieval of only linear features will cause less cognitive load for both groups compared to the other features. We additionally expect that experts would perform better at tasks demanding higher cognitive load. |
| Participants | 56 participants: 24 experts (13 females, 11 males) 30 novices (7 females, 23 males) Age range: 18–35 | 22 participants: 11 experts (5 females, 6 males) 11 novices (6 females, 5 males) Age range: 25–35 |
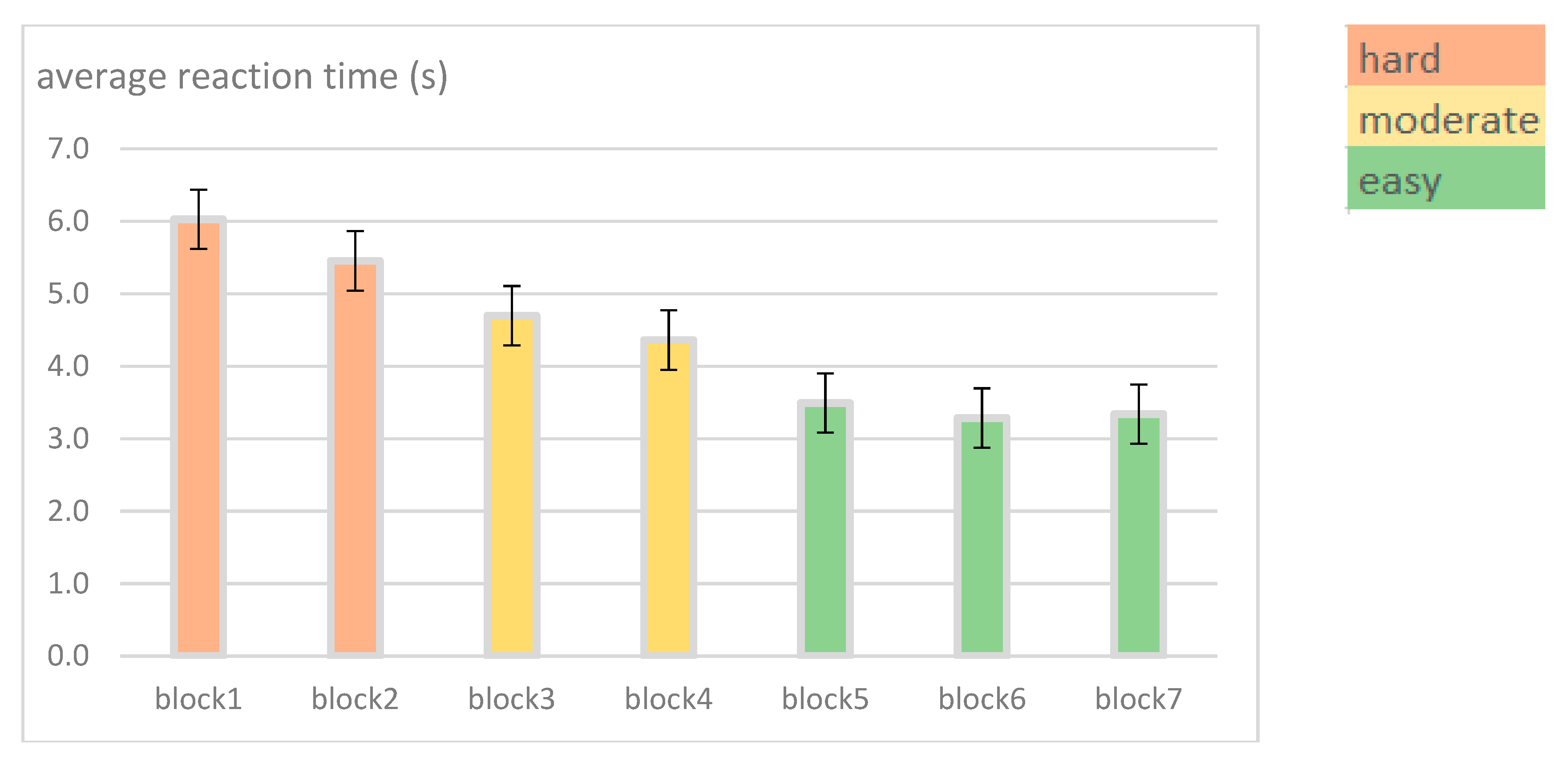
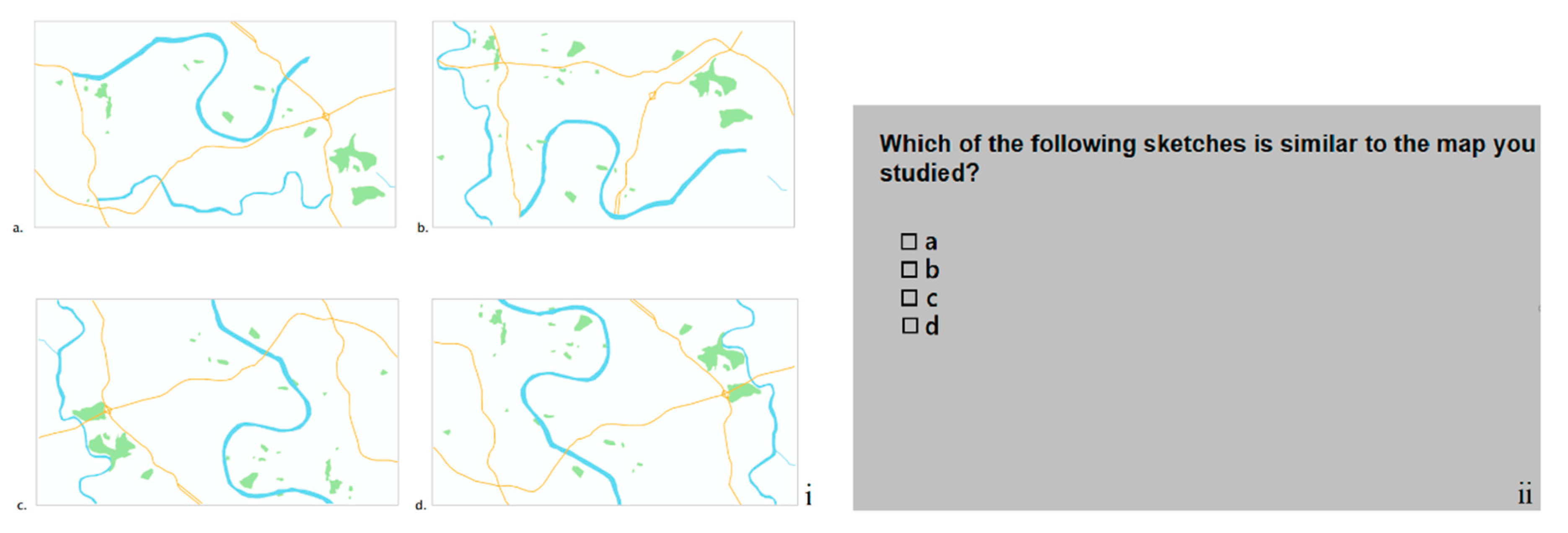
| Task procedures | Participants studied one map stimulus for as long as they wanted to memorize all the main structuring elements included in the map they studied. Once they thought they had studied the map long enough, they pressed a certain key and then they had to draw this map from memory by using MS Paint. After drawing the sketch map, participants used a special key to terminate the task. | Randomized block design: Seven blocks representing seven difficulty types. Each block includes 50 trials (i.e., one for each stimulus) focusing on the similarity of: Block 1: The whole map Block 2: Roads and hydrography Block 3: Roads and green areas Block 4: Green areas and hydrography Block 5: Green areas Block 6: Hydrography Block 7: Roads |
| Independent variables | 1 map design type (i.e., 2D static topographic map) 1 task difficulty level (i.e., retrieval of the main structuring elements of the whole map stimulus) 2 expertise levels (i.e., experts vs. novices) | 1 map design type (i.e., Google maps stimuli) 7 task difficulty levels (i.e., classified as easy, moderate, hard) ~ linear & polygon features within blocks 2 expertise levels (i.e., experts vs. novices) |
| Dependent variables | Trial durations *, eye movements, EEG (alpha power, FAA), self-reported metrics (i.e., questionnaire) * | Response time of correct answers, eye movements, EEG metrics (ERD-ERS), self-reported metrics (i.e., questionnaire) * |
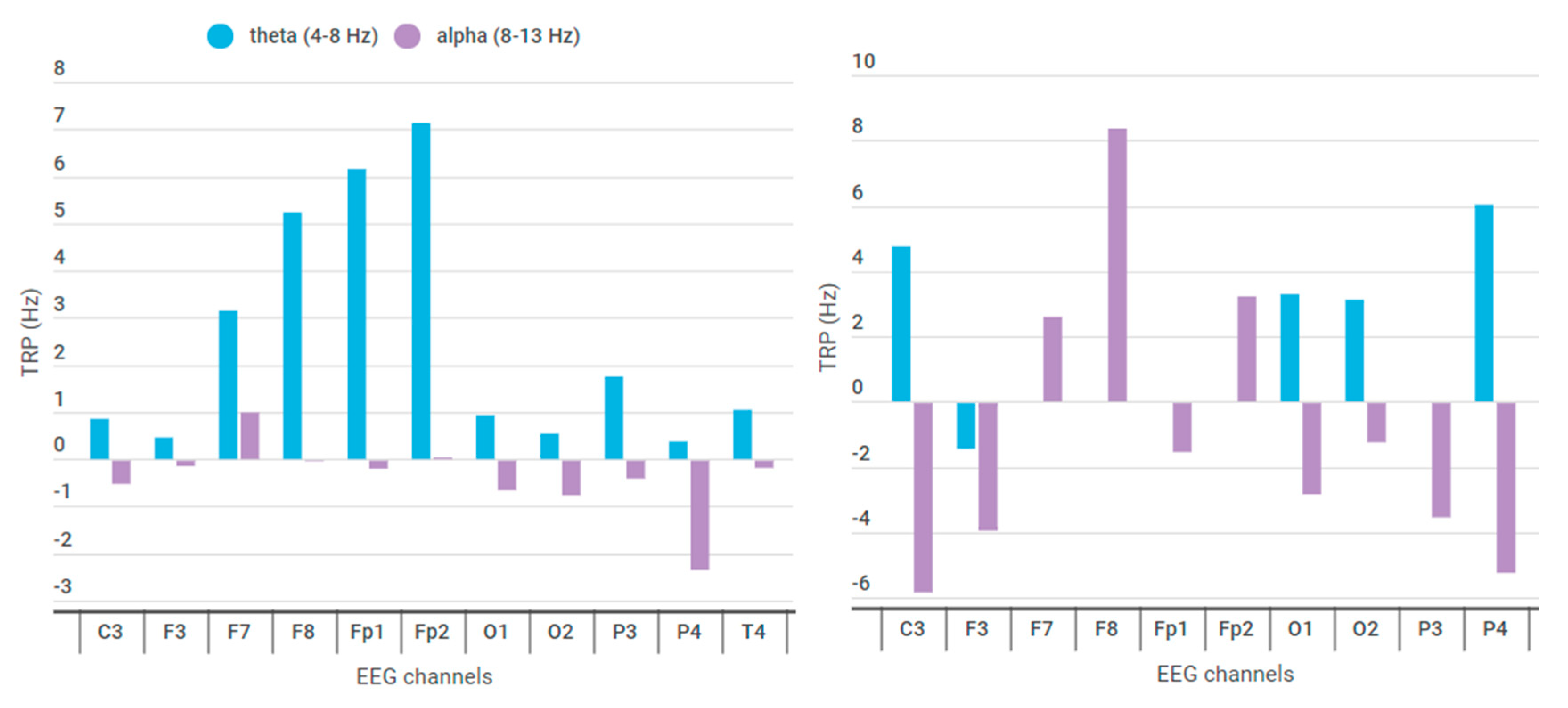
| EEG Channels | ERP Theta (4–8 Hz) | ERP Alpha (8–13 Hz) | EEG Channels | ERP Theta (4–8 Hz) | ERP Alpha (8–13 Hz) |
|---|---|---|---|---|---|
| Block1 | Block 2 | ||||
| C3 | 4.76 × 10−5 | −5.83 × 10−6 | C3 | 8.57 × 10−1 | −4.92 × 10−2 |
| F3 | −1.37 × 10−5 | −3.87 × 10−6 | F3 | 4.58 × 10−1 | −1.15 × 10−2 |
| F7 | 2.58 × 10−4 | 2.59 × 10−7 | F7 | 3.15 | 9.89 × 10−2 |
| F8 | 3.25 × 10−4 | 8.36 × 10−6 | F8 | 5.23 | −2.18 × 10−3 |
| Fp1 | 5.42 × 10−4 | −1.54 × 10−6 | Fp1 | 6.15 | −1.7 × 10−2 |
| Fp2 | 7.15 × 10−4 | 3.22 × 10−6 | Fp2 | 7.13 | 3.49 × 10−3 |
| O1 | 3.29 × 10−5 | −2.76 × 10−6 | O1 | 9.32 × 10−1 | −6.22 × 10−2 |
| O2 | 3.11 × 10−5 | −1.23 × 10−5 | O2 | 5.41 × 10−1 | −7.39 × 10−2 |
| P3 | 1.11 × 10−4 | −3.49 × 10−6 | P3 | 1.75 | −3.83 × 10−2 |
| P4 | 6.03 × 10−5 | −5.18 × 10−6 | P4 | 3.72 × 10−1 | −2.32 × 10−1 |
| T4 | 9.72 × 10−5 | −5.30 × 10−6 | T4 | 1.05 | −1.54 × 10−2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keskin, M.; Ooms, K.; Dogru, A.O.; De Maeyer, P. EEG & Eye Tracking User Experiments for Spatial Memory Task on Maps. ISPRS Int. J. Geo-Inf. 2019, 8, 546. https://doi.org/10.3390/ijgi8120546
Keskin M, Ooms K, Dogru AO, De Maeyer P. EEG & Eye Tracking User Experiments for Spatial Memory Task on Maps. ISPRS International Journal of Geo-Information. 2019; 8(12):546. https://doi.org/10.3390/ijgi8120546
Chicago/Turabian StyleKeskin, Merve, Kristien Ooms, Ahmet Ozgur Dogru, and Philippe De Maeyer. 2019. "EEG & Eye Tracking User Experiments for Spatial Memory Task on Maps" ISPRS International Journal of Geo-Information 8, no. 12: 546. https://doi.org/10.3390/ijgi8120546
APA StyleKeskin, M., Ooms, K., Dogru, A. O., & De Maeyer, P. (2019). EEG & Eye Tracking User Experiments for Spatial Memory Task on Maps. ISPRS International Journal of Geo-Information, 8(12), 546. https://doi.org/10.3390/ijgi8120546







