Coupling Traditional Monitoring and Citizen Science to Disentangle the Invasion of Halyomorpha halys
Abstract
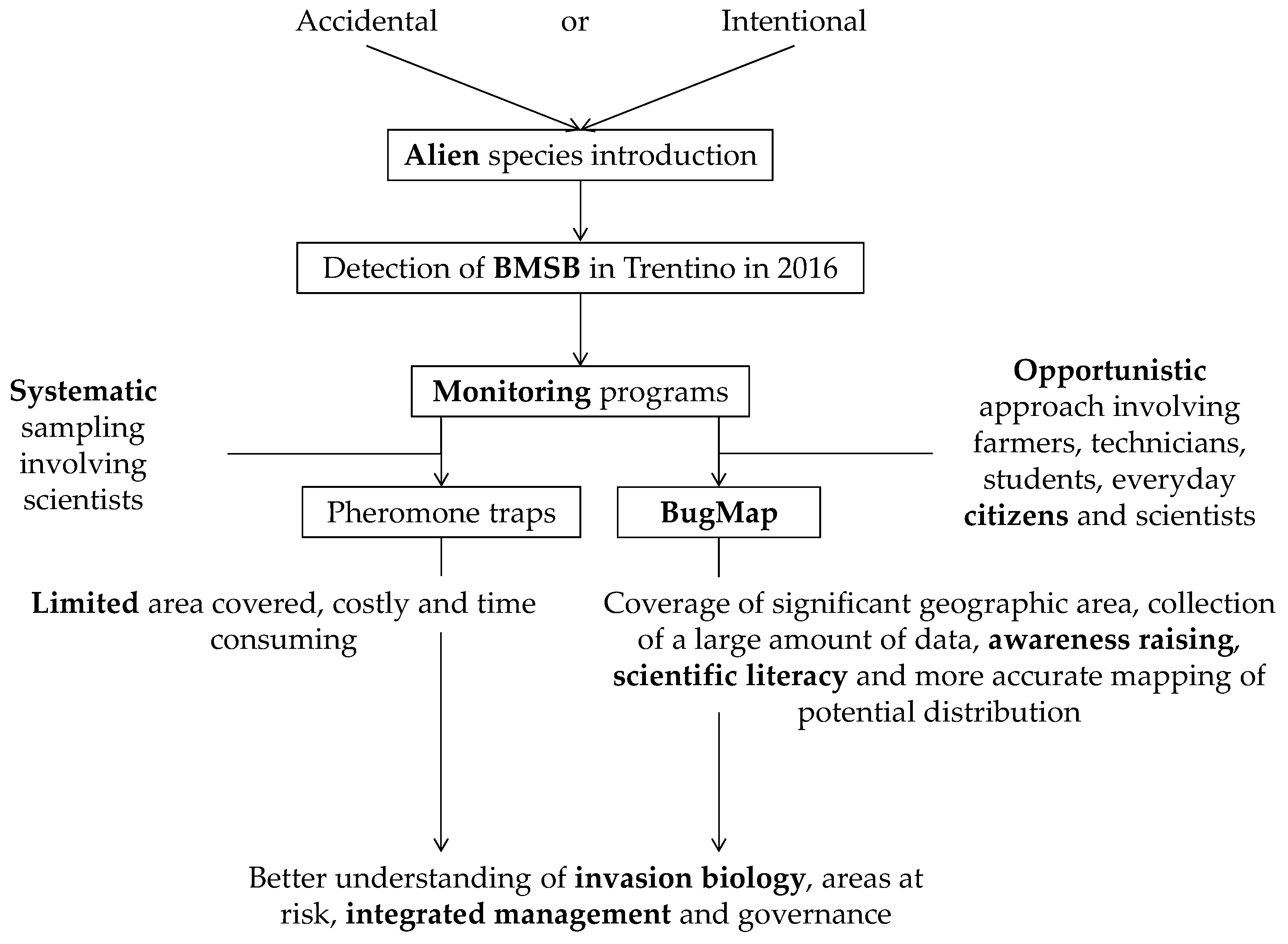
1. Introduction
2. Materials and Methods
2.1. Data Acquisition
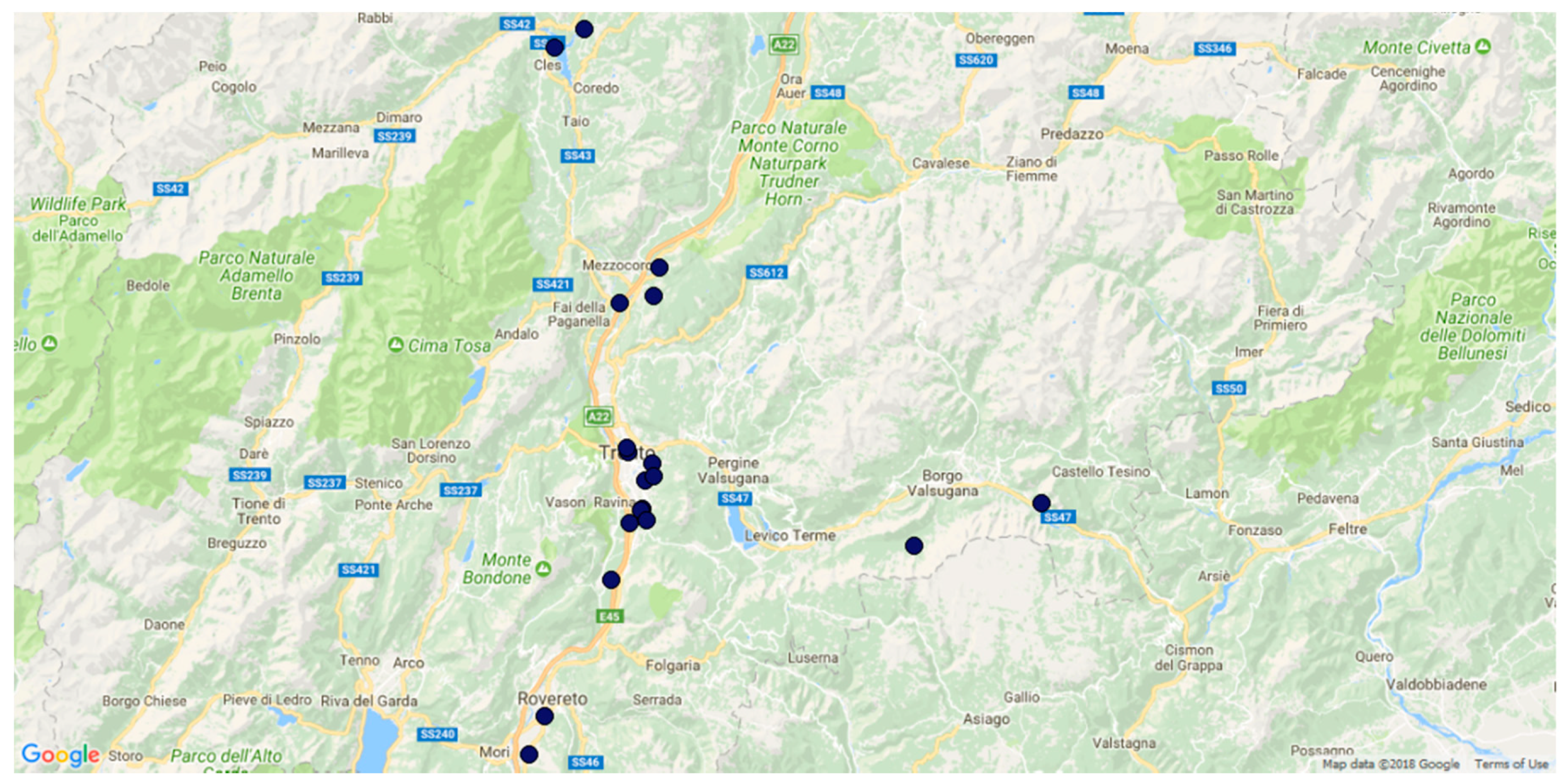
2.1.1. Study Area
2.1.2. BugMap, a Mobile-Based Application for Crowdsourced BMSB Reports
2.1.3. BugMap Campaign
2.1.4. Pheromone Traps
2.2. Modeling Current and Potential Distribution of BMSB in Trentino
2.2.1. Environmental Predictors
2.2.2. BMSB MaxEnt Distribution Modeling
2.2.3. Accounting for BugMap Sampling Bias
2.2.4. Setting the Threshold of BMSB Distribution Model
3. Results and Discussion
3.1. Citizens’ Impact
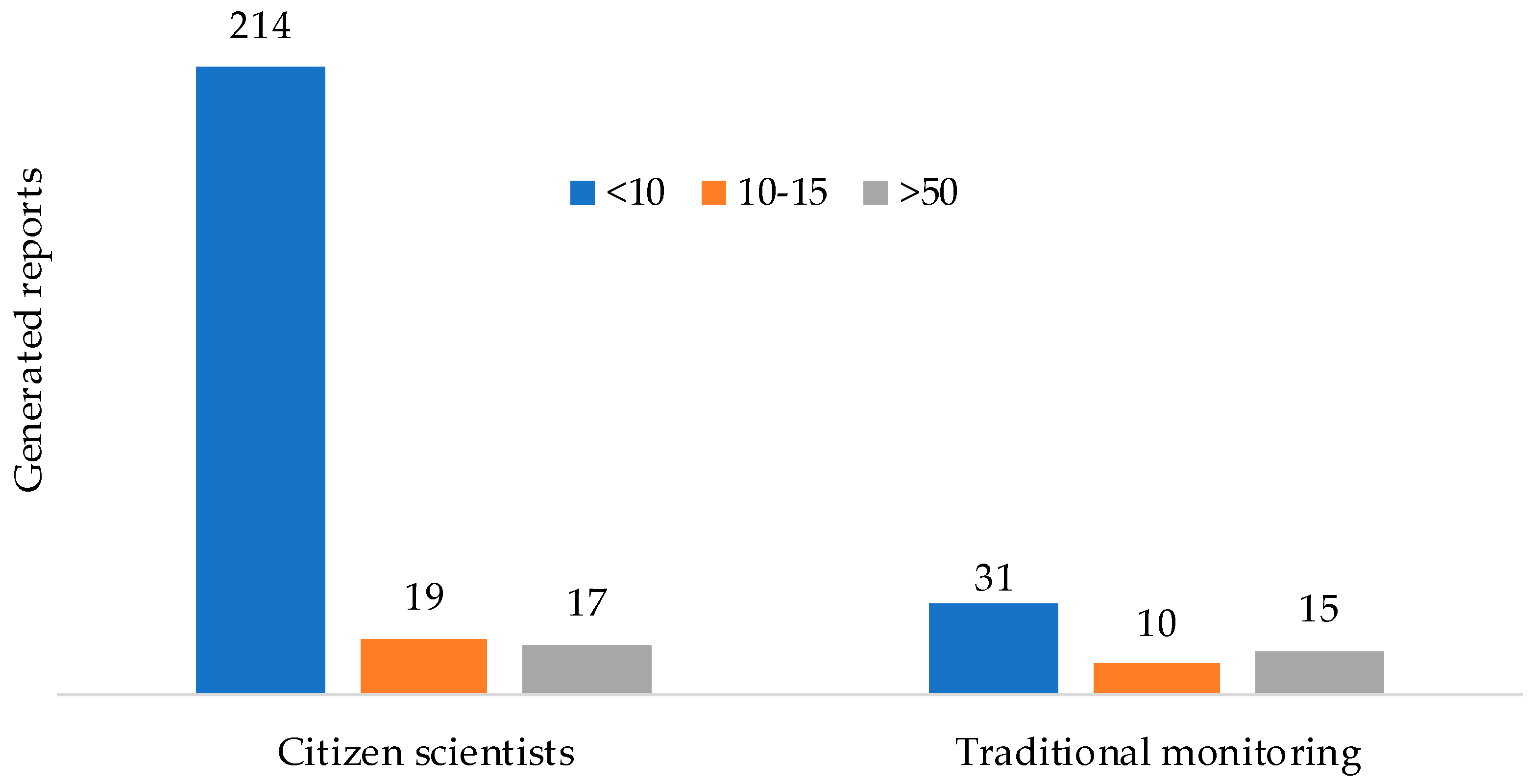
3.1.1. Citizen Science VS. Traditional Monitoring
3.1.2. The Effect of Training on User Performance
3.2. BMSB Invasion
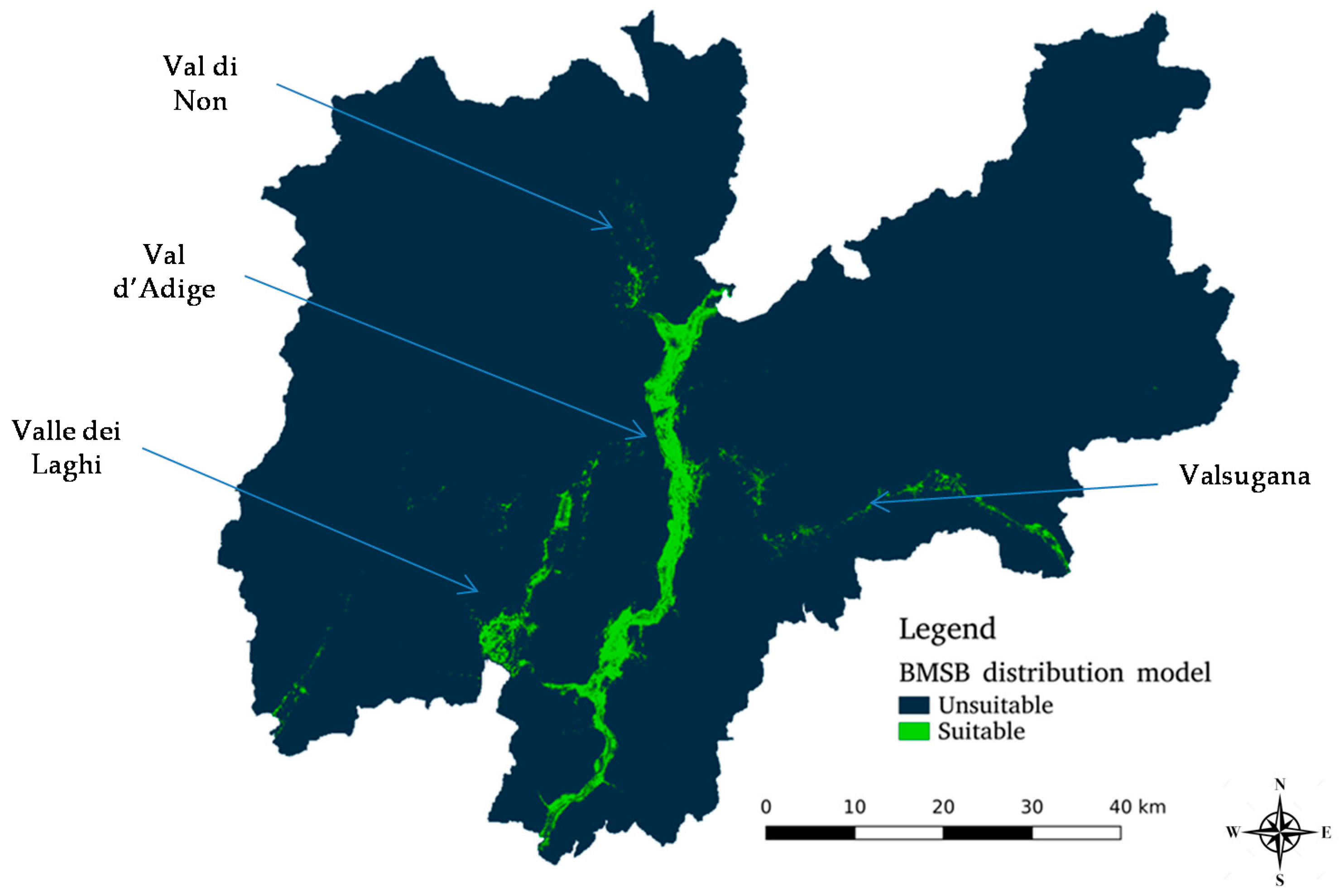
3.2.1. Invasion Dynamics in Trentino
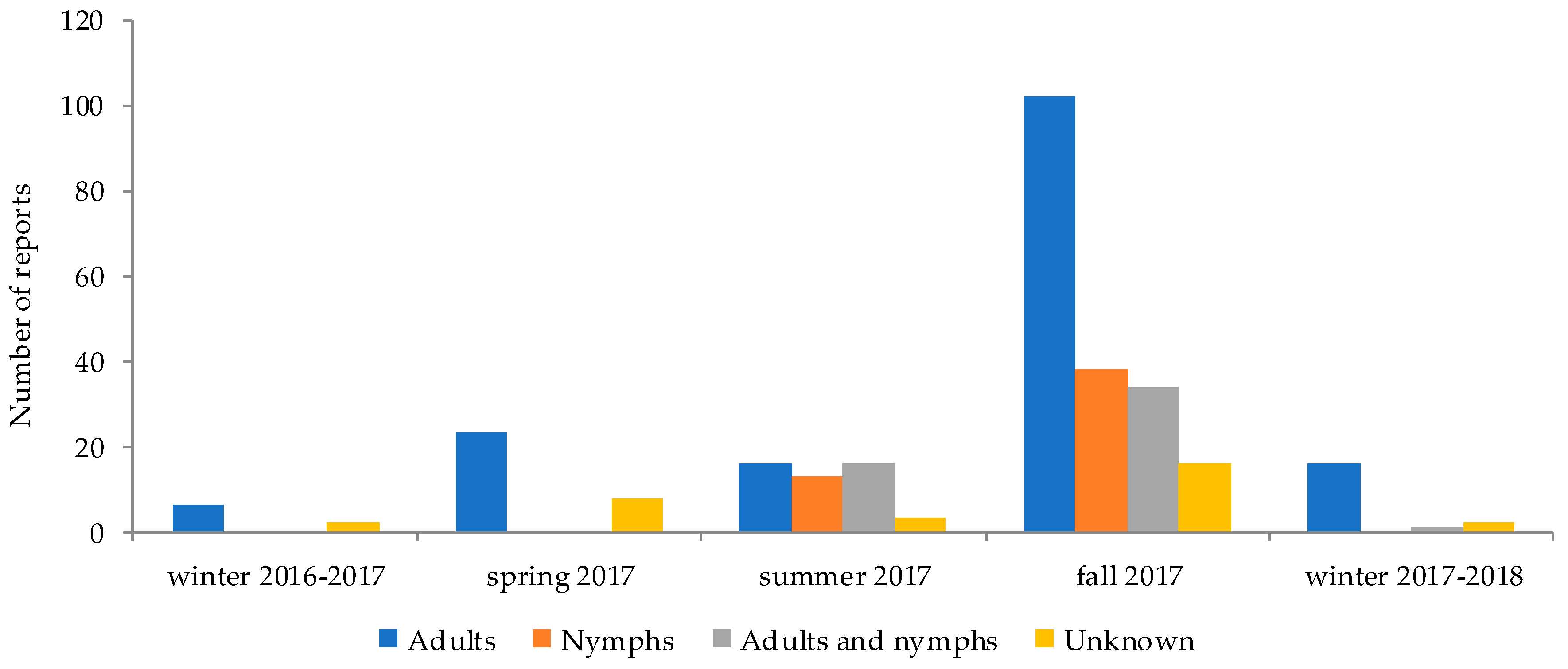
3.2.2. Seasonal Phenology of BMSB
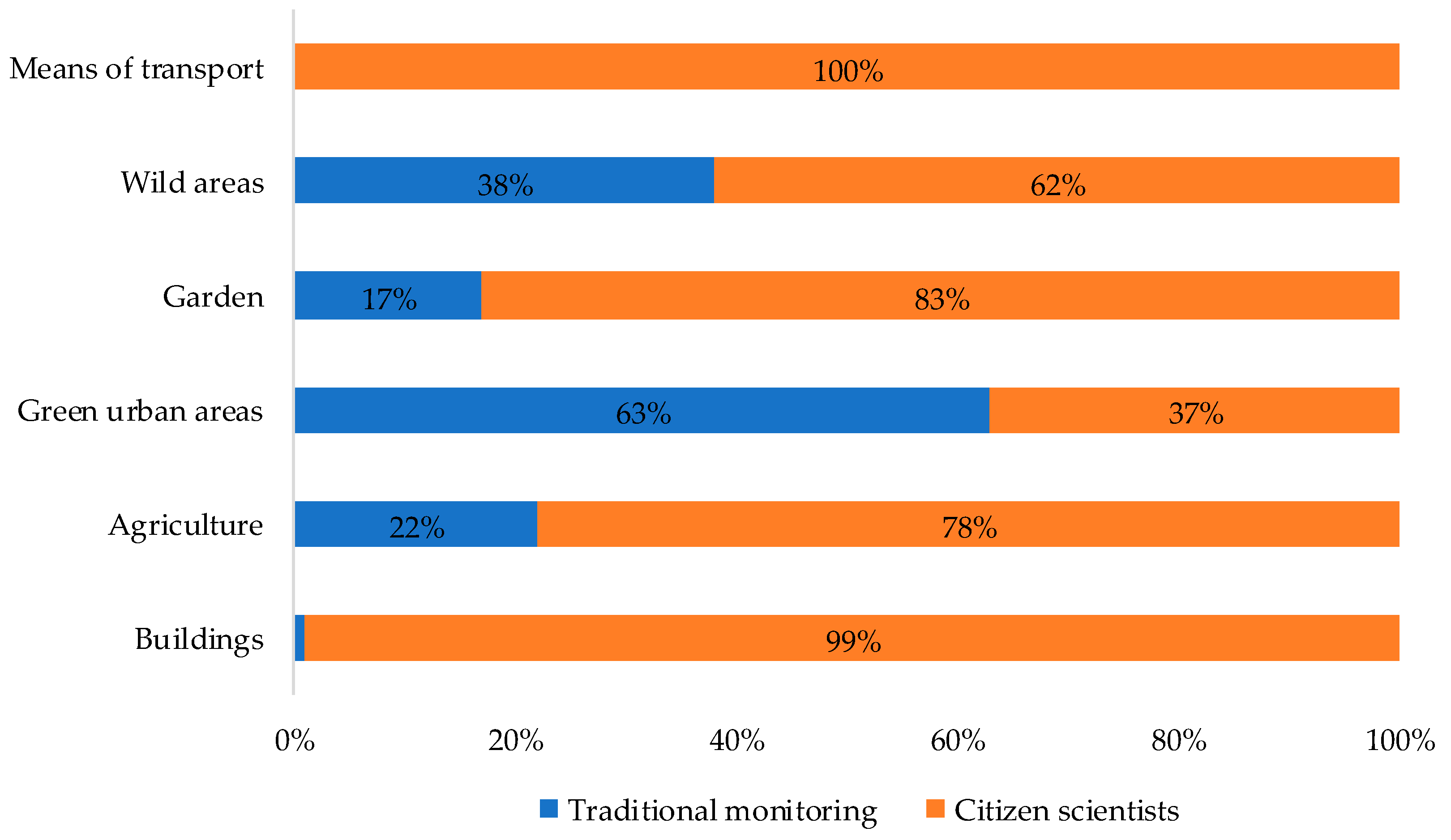
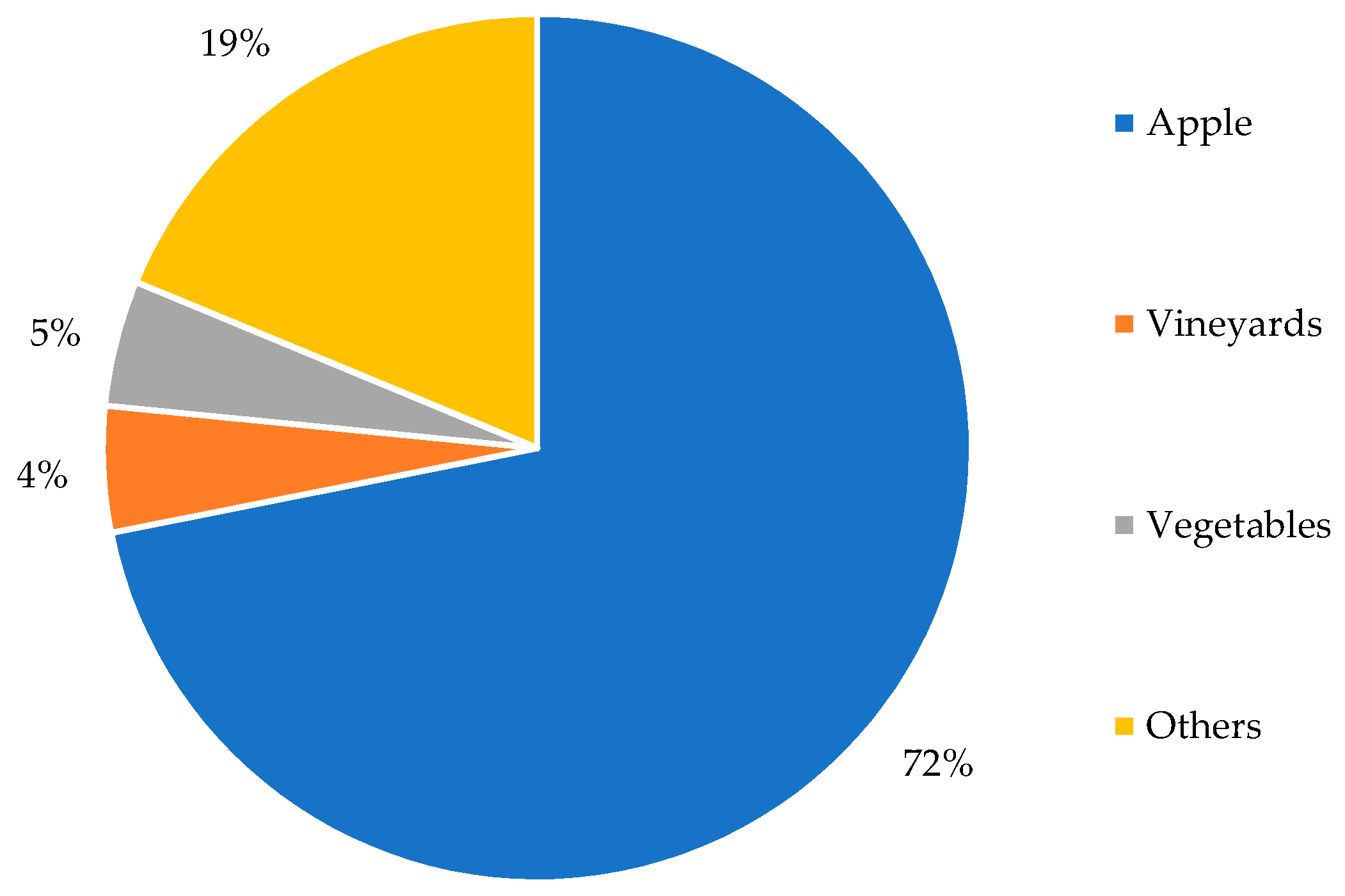
3.2.3. Menace in Agricultural Areas
3.3. BMSB Projected Distribution in Trentino
3.3.1. MaxEnt-Generated Suitability Map
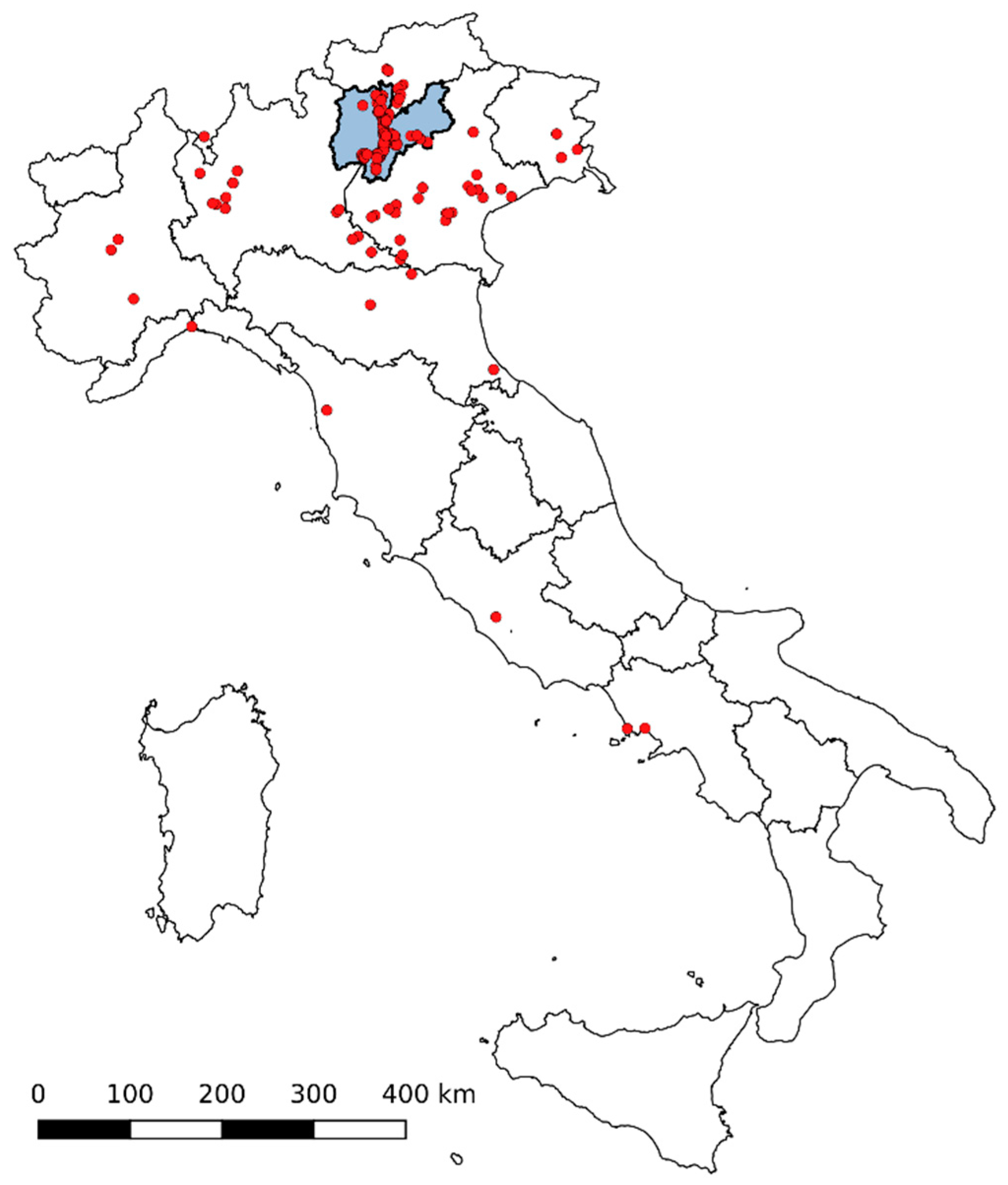
3.3.2. Nationwide Involvement
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Oxford English Dictionary. “Art, n.1.” OED Online. Oxford University Press: Oxford, UK, January 2018. Available online: www.oed.com/viewdictionaryentry/Entry/11125 (accessed on 21 January 2018).
- Dickinson, J.L.; Zuckerberg, B.; Bonter, D.N. Citizen Science as an ecological research tool: Challenges and benefits. Annu. Rev. Ecol. Evol. Syst. 2010, 41, 149–172. [Google Scholar] [CrossRef]
- Danielsen, F.; Burgess, N.D.; Balmford, A. Monitoring matters: Examining the potential of locally-based approaches. Biodivers. Conserv. 2005, 14, 2507–2542. [Google Scholar] [CrossRef]
- Measham, T.G. Building Capacity for Environmental Management: Local knowledge and rehabilitation on the Gippsland Red Gum Plains. Aust. Geogr. 2007, 38, 145–159. [Google Scholar] [CrossRef]
- Brossard, D.; Lewenstein, B.; Bonney, R. Scientific knowledge and attitude change: The impact of a citizen science project. Int. J. Sci. Educ. 2005, 27, 1099–1121. [Google Scholar] [CrossRef]
- Lepczyk, C.A. Integrating published data and citizen science to describe bird diversity across a landscape. J. Appl. Ecol. 2005, 42, 672–677. [Google Scholar] [CrossRef]
- Greenwood, J.J.D. Citizens, science and bird conservation. J. Ornithol. 2007, 148, 77–124. [Google Scholar] [CrossRef]
- Cooley, J. The distribution of periodical cicada (Magicicada) Brood I in 2012 with previously unreported disjunct populations (Hemiptera: Cicadadae, Magicicada). Bull. Entomol. Soc. Am. 2015, 61, 51–56. [Google Scholar] [CrossRef][Green Version]
- Palmer, J.R.B.; Oltra, A.; Collantes, F.; Delgado, J.A.; Lucientes, J.; Delacour, S.; Bengoa, M.; Eritja, R.; Bartumeus, F. Citizen science provides a reliable and scalable tool to track disease-carrying mosquitoes. Nat. Commun. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Hulme, P.E.; Bacher, S.; Kenis, M.; Klotz, S.; Kuhn, I.; Minchin, D.; Nentwig, W.; Olenin, S.; Panov, V.; Pergl, J.; et al. Grasping at the routes of biological invasions: A framework for integrating pathways into policy. J. Appl. Ecol. 2008, 45, 403–414. [Google Scholar] [CrossRef]
- Cini, A.; Anfora, G.; Escudero-Colomar, L.A.; Grassi, A.; Santosuosso, U.; Seljak, G.; Papini, A. Tracking the invasion of the alien fruit pest Drosophila suzukii in Europe. J. Pest Sci. 2014, 87, 559–566. [Google Scholar] [CrossRef]
- Geslin, B.; Gauzens, B.; Baude, M.; Dajoz, I.; Fontaine, C.; Henry, M.; Ropars, L.; Rollin, O.; Thébault, E.; Vereecken, N.J. Massively introduced managed species and their consequences for plant-pollinator interactions. In Networks of Invasion: Empirical Evidence and Case Studies, 1st ed.; Bohan, D.A., Dumbrell, A.J., Massol, F., Eds.; Academic Press: Cambridge, MA, USA, 2017; Volume 57, pp. 147–199. [Google Scholar]
- Leskey, T.C.; Nielsen, A.L. Impact of the invasive brown marmorated stink bug in North America and Europe: History, biology, ecology, and management. Annu. Rev. Entomol. 2018, 63, 599–618. [Google Scholar] [CrossRef] [PubMed]
- Fogain, R.; Graff, S. First records of the invasive pest, Halyomorpha halys (Hemiptera: Pentatomidae), in Ontario and Quebec. J. Entomol. Soc. Ont. 2011, 142, 45–48. [Google Scholar]
- Wermelinger, B.; Wyniger, D.; Forster, B. First records of an invasive bug in Europe: Halyomorpha halys Stål (Heteroptera: Pentatomidae), a new pest on woody ornamentals and fruit trees? Bull. Soc. Entomol. Suiss. 2008, 81, 1–8. [Google Scholar]
- EPPO Global Database. Available online: https://gd.eppo.int (accessed on 13 January 2018).
- Bariselli, M.; Bugiani, R.; Maistrello, L. Distribution and damage caused by Halyomorpha halys in Italy. Bull. OEPP 2016, 46, 332–334. [Google Scholar] [CrossRef]
- Nielsen, A.L.; Hamilton, G.C. Life history of the invasive species Halyomorpha halys (Hemiptera: Pentatomidae) in northeastern United States. Ann. Entomol. Soc. Am. 2009, 102, 608–616. [Google Scholar] [CrossRef]
- Oregon Department of Agriculture: Pest Alert: Brown Marmorated Stink Bug Halyomorpha halys. Available online: http://www.oregon.gov/ODA/Pages/default.aspx (accessed on 4 December 2017).
- Inkley, D. Characteristics of home invasion by the brown marmorated stink bug (Hemiptera: Pentatomidae). J. Entomol. Sci. 2012, 47, 125–130. [Google Scholar] [CrossRef]
- Cesari, M.; Maistrello, L.; Piemontese, L.; Bonini, R.; Dioli, P.; Lee, W.; Park, C.G.; Partsinevelos, G.K.; Rebecchi, L.; Guidetti, R. Genetic diversity of the brown marmorated stink bug Halyomorpha halys in the invaded territories of Europe and its patterns of diffusion in Italy. Biol. Invasions 2017, 1–20. [Google Scholar] [CrossRef]
- Trentino Agricoltura. Available online: http://www.trentinoagricoltura.it/Trentino-Agricoltura/Settori2/Ortofrutticoltura/Mela (accessed on 9 January 2018).
- Roura-Pascual, N.; Krug, R.M.; Richardson, D.M.; Hui, C. Spatially-explicit sensitivity analysis for conservation management exploring the influence of decisions in invasive alien plant management. Divers. Distrib. 2010, 16, 426–438. [Google Scholar] [CrossRef]
- Jackson, S.T.; Overpeck, J.T. Responses of plant populations and communities to environmental changes of the late Quaternary. Paleobiology 2000, 26, 194–220. [Google Scholar] [CrossRef]
- Anderson, R.P.; Lew, D.; Peterson, A.T. Evaluating predictive models of species’ distributions: Criteria for selecting optimal models. Ecol. Model. 2003, 162, 211–232. [Google Scholar] [CrossRef]
- Merow, C.; Smith, M.J.; Silander, J.A., Jr. A practical guide to MaxEnt for modeling species’ distributions: What it does, and why inputs and settings matter. Ecocraphy 2013, 36, 1058–1069. [Google Scholar] [CrossRef]
- Hahn, N.G.; Kaufman, A.J.; Rodriguez-Saona, C.; Nielsen, A.L.; Laforest, J.; Hamilton, G.C. Exploring the spread of the brown marmorated stink bug in New Jersey through the use of crowdsourced reports. Am. Entomol. 2016, 62, 36–45. [Google Scholar] [CrossRef]
- Silverton, J. A new dawn for citizen science. Trends Ecol. Evolut. 2009, 24, 467–471. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, R.M. Remote Sensing and GIS Studies on the Spatial Distribution and Management of Japanese Beetle Adults and Grubs. Ph.D. Dissertation, Purdue University, West Lafayette, IN, USA, 2003. [Google Scholar]
- Dminić, I.; Kozina, A.; Bažok, R.; Barčić, J.I. Geographic information system (GIS) and entomological research: A Review. J. Food Agric. Environ. 2010, 8, 1193–1198. [Google Scholar]
- Istat-Istituto Nazionale di Statistica. Available online: http://www.istat.it/en/ (accessed on 16 February 2018).
- Provincia di Trento. Available online: http://www.statweb.provincia.tn.it/ (accessed on 20 February 2018).
- Capinha, C.; Anastácio, P. Assessing the environmental requirements of invaders using ensembles of distribution models. Divers. Distrib. 2011, 17, 13–24. [Google Scholar] [CrossRef]
- Autonomous Province of Trento. Available online: http://www.territorio.provincia.tn.it/portal/server.pt/community/cartografia_di_base/260/cartografia_di_base/19024 (accessed on 6 August 2017).
- Philips, S.J.; Anderson, R.P.; Schapire, R.E. Maximum entropy modeling of species geographic distributions. Ecol. Model. 2006, 190, 231–359. [Google Scholar] [CrossRef]
- Elith, J.; Philips, S.J.; Hastie, T.; Dudík, M.; Chee, Y.E.; Yates, C.J. A statistical explanation of MaxEnt for ecologists. Divers. Distrib. 2011, 17, 43–57. [Google Scholar] [CrossRef]
- Zhu, G.; Bu, W.; Gao, Y.; Liu, G. Potential geographic distribution of brown marmorated stink bug invasion (Halyomorpha halys). PLoS ONE 2012, 7, e31246. [Google Scholar] [CrossRef] [PubMed]
- Fourcade, Y.; Engler, J.O.; Rödder, D.; Secondi, J. Mapping Species Distributions with MAXENT Using a Geographically Biased Sample of Presence Data: A Performance Assessment of Methods for Correcting Sampling Bias. PLoS ONE 2014, 9, e97122. [Google Scholar] [CrossRef] [PubMed]
- GRASS Development Team. Geographic Resources Analysis Support System (GRASS) Software, Version 7.0; Open Source Geospatial Foundation: Chicago, IL, USA, 2015. [Google Scholar]
- Phillips, S.J.; Dudík, M.; Elith, J.; Graham, C.H.; Lehmann, A.; Leathwick, J.; Ferrier, S. Sample selection bias and presence-only distribution models: Implications for background and pseudo-absence data. Ecol. Appl. 2009, 19, 181–197. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Cantor, S.B.; Sun, C.C.; Tortolero-Luna, G.; Richards-Kortum, R.; Follen, M. A comparison of C/B ratios from studies using receiver operating characteristic curve analysis. J. Clin. Epidemiol. 1999, 52, 885–892. [Google Scholar] [CrossRef]
- Liu, C.; Berry, P.; Dawson, T.; Pearson, R. Selecting thresholds of occurrence in the prediction of species distributions. Ecography 2005, 28, 385–393. [Google Scholar] [CrossRef]
- Wallner, A.M.; Hamilton, G.C.; Nielsen, A.L.; Hahn, N.; Green, E.J.; Rodriguez-Saona, C.R. Landscape factors facilitating the invasive dynamics and distribution of the brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae), after arrival in the United States. PLoS ONE 2014, 9, e95691. [Google Scholar] [CrossRef] [PubMed]
- Maistrello, L.; Dioli, P.; Bariselli, M.; Mazzoli, G.L.; Giacalone-Forini, I. Citizen science and early detection of invasive species: Phenology of first occurrences of Halyomorpha halys in Southern Europe. Biol. Invasions 2016, 18, 3109–3116. [Google Scholar] [CrossRef]
- Sargent, C.; Martinson, H.M.; Raupp, M.J. Traps and trap placement may affect location of brown marmorated stink bug (Hemiptera: Pentatomidae) and increase injury to tomato fruits in home gardens. Environ. Entomol. 2014, 43, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Rossi-Stacconi, M.V.; Rupinder, K.; Mazzoni, V.; Ometto, L.; Grassi, A.; Gottardello, A.; Rota-Stabelli, O.; Anfora, G. Multiple lines of evidence for reproductive winter diapause in the invasive pest Drosophila suzukii: Useful clues for control strategies. J. Pest Sci. 2016, 89, 689–700. [Google Scholar] [CrossRef]
- Holtz, T.; Kamminga, K. Qualitative Analysis of the Pest Risk Potential of the Brown Marmorated Stink Bug (BMSB), Halyomorpha halys (Stål), in the United States. United States Department of Agriculture: APHIS 2010. Available online: https://www.michigan.gov/documents/mda/BMSB_Pest_Risk_Potential_-_USDA_APHIS_Nov_2011_344862_7.pdf (accessed on 6 January 2018).
- Tindall, K.V.; Fothergill, K.; McCormack, B. Halyomorpha halys (Hemiptera: Pentatomidae): A first Kansas record. J. Kansas Entomol. Soc. 2012, 85, 169. [Google Scholar] [CrossRef]
- Anfora, G. (Trento University, Trento, Trentino, Italy). Personal Communication, 2018.
- Crall, A.W.; Newman, G.J.; Jarnevich, C.S.; Stohlgren, T.J.; Waller, D.M.; Graham, J. Improving and integrating data on invasive species collected by citizen scientists. Biol. Invasions 2010, 12, 3419–3428. [Google Scholar] [CrossRef]
- Kolar, C.S.; Lodge, D.M. Progress in invasion biology: Predicting invaders. Trends Ecol. Evolut. 2001, 16, 199–204. [Google Scholar] [CrossRef]
- Keane, R.M.; Crawley, M.J. Exotic plant invasions and the enemy release hypothesis. Trends Ecol. Evol. 2002, 17, 164–170. [Google Scholar] [CrossRef]
- Nielsen, A.L.; Holmstrom, K.; Hamilton, G.C.; Cambridge, J.; Ingerson–Mahar, J. Use of black light traps to monitor the abundance, spread, and flight behavior of Halyomorpha halys (Hemiptera: Pentatomidae). J. Econ. Entomol. 2013, 106, 1495–1502. [Google Scholar] [CrossRef] [PubMed]
- Blossey, B.; Notzold, R. Evolution of increased competitive ability in invasive nonindigenous plants: A hypothesis. J. Ecol. 1995, 83, 887–889. [Google Scholar] [CrossRef]
- Lee, K.A.; Klasing, K.C. A role for immunology in invasion biology. Trends Ecol. Evolut. 2004, 19, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, E.J.; Venugopal, P.D.; Martinson, H.M.; Raupp, M.J.; Shrewsbury, P.M. Host plant use by the invasive Halyomorpha halys (Stål) on woody ornamental trees and shrubs. PLoS ONE 2016, 11, e0149975. [Google Scholar] [CrossRef] [PubMed]
- Philips, C.R.; Kuhar, T.P.; Dively, G.P.; Hamilton, G.; Whalen, J.; Kamminga, K. Seasonal abundance and phenology of Halyomorpha halys (Hemiptera: Pentatomidae) on different pepper cultivars in the Mid-Atlantic (United States). J. Econ. Entomol. 2016, 110, 192–200. [Google Scholar] [CrossRef]
- Nielsen, A.L.; Fleischer, S.; Hamilton, G.C.; Hancock, T.; Krawczyk, G.; Lee, J.C.; Ogburn, E.; Pote, J.M.; Raudenbush, A.; Rucker, A.; et al. Phenology of brown marmorated stink bug described using female reproductive development. Ecol. Evol. 2017, 7, 6680–6690. [Google Scholar] [CrossRef] [PubMed]
- Haye, T.; Abdallah, S.; Gariepy, T.; Wyniger, D. Phenology, life table analysis and temperature requirements of the invasive brown marmorated stink bug, Halyomorpha halys, in Europe. J. Pest Sci. 2014, 87, 407–418. [Google Scholar] [CrossRef]
- Morrison, W.R.; Lee, D.H.; Short, B.D.; Khrimian, A.; Leskey, T.C. Establishing the behavioral basis for an attract-and-kill strategy to manage the invasive Halyomorpha halys in apple orchards. J. Pest Sci. 2016, 89, 81–96. [Google Scholar] [CrossRef]
- Bergh, J.C.; Morrison, W.R.; Joseph, S.V.; Leskey, T.C. Characterizing spring emergence of adult Halyomorpha halys using experimental overwintering shelters and commercial pheromone traps. Entomol. Exp. Appl. 2017, 162, 336–345. [Google Scholar] [CrossRef]
- Wiman, N.G.; Walton, V.M.; Shearer, P.W.; Rondon, S.I. Electronically monitored labial dabbing and stylet ‘probing’ behaviors of brown marmorated stink bug, Halyomorpha halys, in simulated environments. PLoS ONE 2014, 9, e113514. [Google Scholar] [CrossRef] [PubMed]
- Acebes-Doria, A.L.; Leskey, T.C.; Bergh, J.C. Host plant effects on Halyomorpha halys (Hemiptera: Pentatomidae) nymphal development and survivorship. Environ. Entomol. 2016, 45, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Joseph, S.V.; Nita, M.; Leskey, T.C.; Bergh, J.C. Temporal effects on the incidence and severity of brown marmorated stink bug (Hemiptera: Pentatomidae) feeding injury to peaches and apples during the fruiting period in Virginia. J. Econ. Entomol. 2015, 108, 592–599. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, K.P.; Fletcher, R.J.; Rota, C.T.; Hutto, R.L. Predicting species distributions from samples collected along roadsides. Conserv. Biol. 2012, 26, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, P.D.; Coffey, P.L.; Dively, G.P.; Lamp, W.O. Adjacent habitat influence on stink bug (Hemiptera: Pentatomidae) densities and the associated damage at field corn and soybean edges. PLoS ONE 2014, 9, e109917. [Google Scholar] [CrossRef] [PubMed]
- Morrison, W.R.; Milonas, P.; Kapantaidaki, D.E.; Cesari, M.; Bella, E.; Guidetti, R.; Haye, T.; Maistrello, L.; Moraglio, S.T.; Piemontese, L.; et al. Attraction of Halyomorpha halys (Hemiptera: Pentatomidae) haplotypes in North America and Europe to baited traps. Sci. Rep. 2017, 7, 16941. [Google Scholar] [CrossRef] [PubMed]
- Cira, T.M.; Venette, R.C.; Aigner, J.; Kuhar, T.; Mullins, D.E.; Gabbert, S.E.; Hutchison, W.D. Cold tolerance of Halyomorpha halys (Hemiptera: Pentatomidae) across geographic and temporal scales. Environ. Entomol. 2016, 45, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Dosdall, L.M. Evidence for successful overwintering of diamondback moth, Plutella xylostella (L.) (Lepidoptera: Plutellidae), in Alberta. Can. Entomol. 1994, 126, 183–185. [Google Scholar] [CrossRef]
- Leskey, T.C.; Short, B.D.; Lee, D.H. Efficacy of insecticide residues on adult Halyomorpha halys (Stål) (Hemiptera: Pentatomidae) mortality and injury in apple and peach orchards. Pest Manag. Sci. 2014, 70, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Kistner, E.J. Climate Change Impacts on the Potential Distribution and Abundance of the Brown Marmorated Stink Bug (Hemiptera: Pentatomidae) with Special Reference to North America and Europe. Environ. Entomol. 2017, 46, 1212–1224. [Google Scholar] [CrossRef] [PubMed]
- Conrad, C.C.; Hilchey, K.G. A review of citizen science and community-based environmental monitoring: Issues and opportunities. Environ. Monit. Assess. 2011, 176, 273–291. [Google Scholar] [CrossRef] [PubMed]
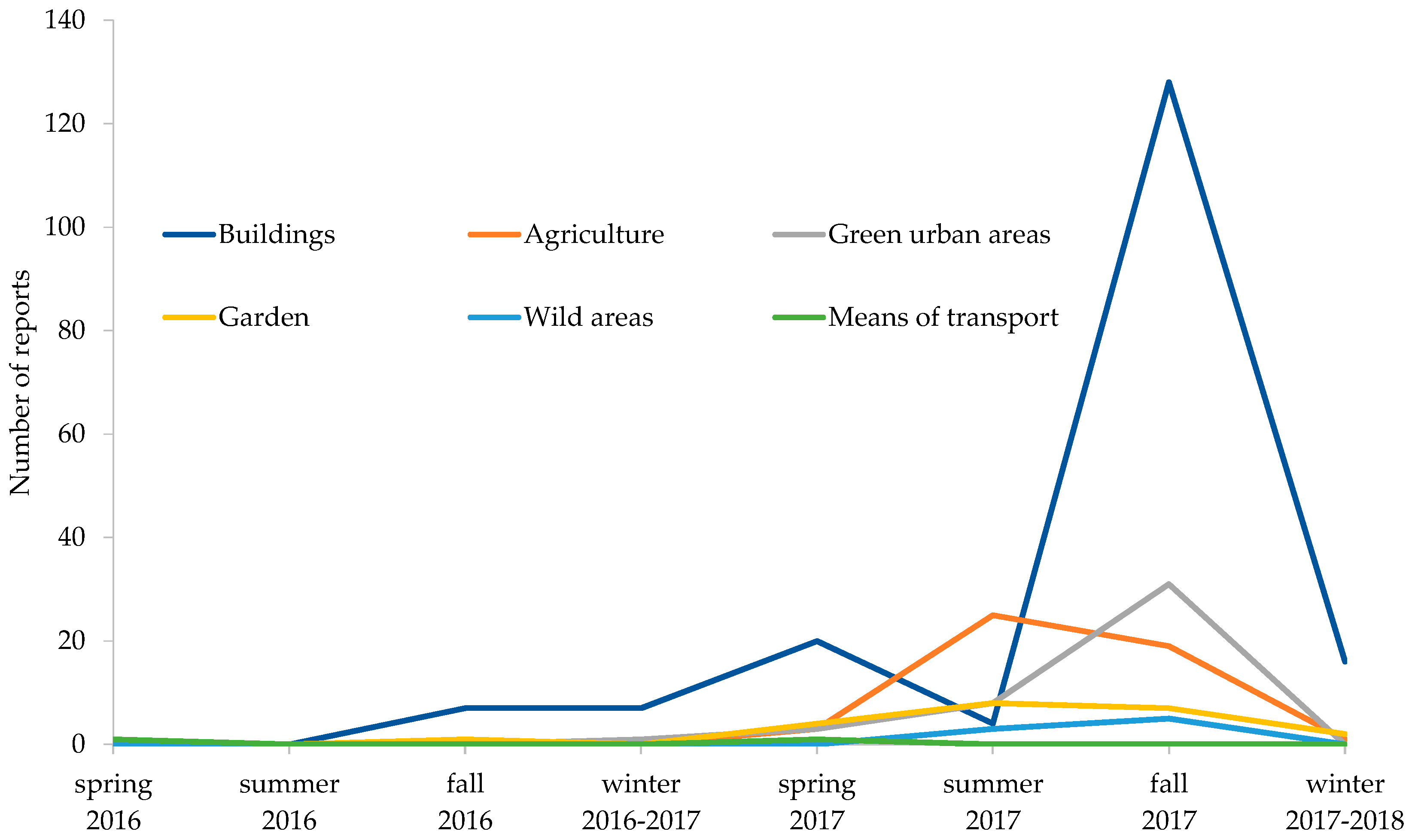
| Index | Spatial Resolution | Parameter |
|---|---|---|
| Digital elevation model (DEM) a | 10 m | Slope |
| Aspect | ||
| Average hours of sun per season | ||
| Land use b | 10 m | Continuous urban fabric |
| Green urban areas | ||
| Fruit trees and berry plantations | ||
| Hydrography | 10 m | Distance from rivers |
| Distance from lakes |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malek, R.; Tattoni, C.; Ciolli, M.; Corradini, S.; Andreis, D.; Ibrahim, A.; Mazzoni, V.; Eriksson, A.; Anfora, G. Coupling Traditional Monitoring and Citizen Science to Disentangle the Invasion of Halyomorpha halys. ISPRS Int. J. Geo-Inf. 2018, 7, 171. https://doi.org/10.3390/ijgi7050171
Malek R, Tattoni C, Ciolli M, Corradini S, Andreis D, Ibrahim A, Mazzoni V, Eriksson A, Anfora G. Coupling Traditional Monitoring and Citizen Science to Disentangle the Invasion of Halyomorpha halys. ISPRS International Journal of Geo-Information. 2018; 7(5):171. https://doi.org/10.3390/ijgi7050171
Chicago/Turabian StyleMalek, Robert, Clara Tattoni, Marco Ciolli, Stefano Corradini, Daniele Andreis, Aya Ibrahim, Valerio Mazzoni, Anna Eriksson, and Gianfranco Anfora. 2018. "Coupling Traditional Monitoring and Citizen Science to Disentangle the Invasion of Halyomorpha halys" ISPRS International Journal of Geo-Information 7, no. 5: 171. https://doi.org/10.3390/ijgi7050171
APA StyleMalek, R., Tattoni, C., Ciolli, M., Corradini, S., Andreis, D., Ibrahim, A., Mazzoni, V., Eriksson, A., & Anfora, G. (2018). Coupling Traditional Monitoring and Citizen Science to Disentangle the Invasion of Halyomorpha halys. ISPRS International Journal of Geo-Information, 7(5), 171. https://doi.org/10.3390/ijgi7050171








