Pioglitazone Modulates p65-Mediated Mitochondrial Bioenergetics: Implications for Acetaldehyde-Induced HIV Replication in Alveolar Macrophages
Abstract
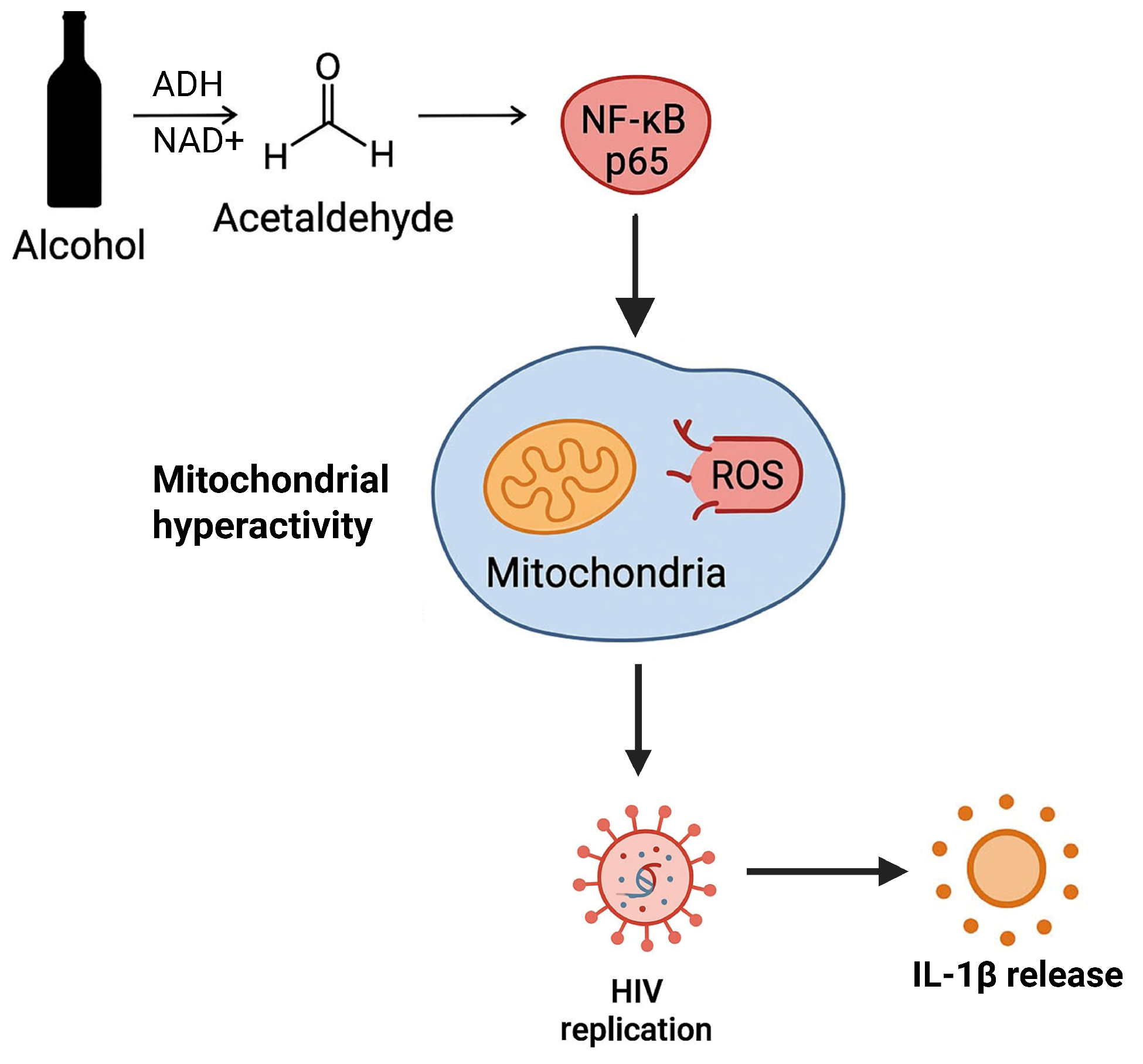
1. Introduction
2. Materials and Methods
2.1. Reagents and Antibodies
2.2. Human Samples
2.2.1. Human Bronchoalveolar Lavage Fluid
2.2.2. Primary Human AMs
2.3. Acetaldehyde Generating System
2.4. EcoHIV Production
2.5. In Vitro Studies
2.5.1. Primary Mouse AMs
2.5.2. MH-S Cells and THP-1-Derived Macrophages
2.6. Extracellular Flux Assays
2.7. RNA Isolation, Real-Time Polymerase Chain Reaction
2.8. Immunoblotting
2.9. Immunofluorescence
2.10. Enzyme-Linked Immunosorbent Assay
2.11. Statistics
3. Results
3.1. The Alveoli of PWH on ART Harbor Replication-Competent HIV, Which Is Potentiated by AGS
3.2. AGS Potentiates EcoHIV Expression and IL-1β Activation in Primary Mouse AMs
3.3. AGS Enhances Mitochondrial Bioenergetics in EcoHIV-Infected MH-S Cells
3.4. AGS Increases Mitochondrial ATP Production and Mitochondrial ROS in MH-S Cells
3.5. Pyruvate and Glutamine Fuel AGS-Induced Mitochondrial Respiration in EcoHIV-Infected MH-S Cells
3.6. AGS-Induced Mitochondrial Respiration Supports EcoHIV Replication and IL-1β Activation in Primary mAMs and MH-S Cells
3.7. Increased p65 Expression in Primary Human AMs (hAMs) from Individuals with AUD and AGS Drives p65 Activation in EcoHIV-Infected Primary mAMs
3.8. Inhibition of p65 Suppresses AGS-Induced Mitochondrial Bioenergetics in EcoHIV-Infected MH-S Cells
3.9. PIO, Which Inhibits p65 Activation, Restores Mitochondrial Bioenergetics in EcoHIV-Infected MH-S Cells Exposed to AGS
3.10. PIO Modulates Alcohol Metabolism-Induced ATP and ROS Production in MH-S Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Abbreviations | Definitions |
| PWH | People living with HIV |
| p65 | Nuclear Factor Kappa B p65 |
| IL-1β | Interleukin-1 beta |
| AMs | Alveolar macrophages |
| PIO | Pioglitazone |
| ROS | Reactive oxygen species |
| ART | Antiretroviral therapy |
| BALF | Bronchoalveolar lavage fluid |
| AIDS | Acquired immune deficiency syndrome |
| ATP | Adenosine triphosphate |
| AUD | Alcohol use disorder |
| PPARγ | Peroxisome proliferator-activated receptor gamma |
| FBS | fetal bovine serum |
| NOX4 | nicotinamide adenine dinucleotide phosphate oxidase 4 |
| PWoH | People living without HIV |
| IRB | Institutional review board |
| CD4 | Cluster differentiation 4 |
| MAA | Malondialdehyde-acetaldehyde |
| USA | United States of America |
| NAD+ | Nicotinamide adenine dinucleotide |
| ADH | Alcohol dehydrogenase |
| AGS | Acetaldehyde generating system |
| mAMs | Mouse alveolar macrophages |
| PFA | Paraformaldehyde |
| ELISA | Enzyme linked immunosorbent assay |
| PMA | Phorbol 12-myristate 13-acetate |
| OCR | Oxygen consumption rate |
| FCCP | carbonilcyanide p-triflouromethoxyphenylhydrazone |
| MLV | Murine leukemia virus |
| qRTPCR | Quantitative real-time polymerase chain reaction |
| GAPDH | glyceraldehyde-3-phosphate dehydrogenase |
| mRNA | Messenger ribonucleic acids |
| Gp20 | Glycoprotein 120 |
| Gag | Group-specific antigen |
| Tat | Trans-activator of transcription |
| NIH | National Institute of Health |
| PBS | Phosphate-buffered saline |
| BSA | Bovine serum albumin |
| DAPI | 4′,6-diamidino-2-phenylindole |
| PAMP | Pathogen-associated molecular pattern |
| hAM | Human alveolar macrophages |
| RFU | Relative fluorescence unit |
| ANOVA | Analysis of variance |
| MitoSOX | Mitochondrial superoxide |
| H2O2 | Hydrogen peroxide |
Appendix A


References
- Alum, E.U.; Uti, D.E.; Ugwu, O.P.; Alum, B.N. Toward a cure—Advancing HIV/AIDs treatment modalities beyond antiretroviral therapy: A Review. Medicine 2024, 103, e38768. [Google Scholar] [CrossRef]
- Foka, F.E.T.; Mufhandu, H.T. Current ARTs, Virologic Failure, and Implications for AIDS Management: A Systematic Review. Viruses 2023, 15, 1732. [Google Scholar] [CrossRef]
- Webel, A.R.; Schexnayder, J.; Cioe, P.A.; Zuñiga, J.A. A Review of Chronic Comorbidities in Adults Living With HIV: State of the Science. J. Assoc. Nurses AIDS Care 2021, 32, 322–346. [Google Scholar] [CrossRef]
- Pipitò, L.; Zinna, G.; Trizzino, M.; Gioè, C.; Tolomeo, M.; Di Carlo, P.; Colomba, C.; Gibaldi, L.; Iaria, C.; Almasio, P.; et al. Causes of hospitalization and predictors of in-hospital mortality among people living with HIV in Sicily-Italy between 2010 and 2021. J. Infect. Public Health 2023, 16, 1703–1708. [Google Scholar] [CrossRef]
- Bhattarai, P.; Karki, M.; Mehari, A. HIV and Chronic Lower Respiratory Diseases Mortality Trend From 1999 to 2020. Am. J. Respir. Crit. Care Med. 2025, 211, A4992. [Google Scholar] [CrossRef]
- Kalmin, M.M.; Westreich, D.; Drummond, B.M.; Sun, J.; Mehta, S.H.; Kirk, G.D. Incident obstructive lung disease and mortality among people with HIV and a history of injecting drugs. Aids 2021, 35, 1451–1460. [Google Scholar] [CrossRef] [PubMed]
- Heidari, S.L.; Hove-Skovsgaard, M.; Arentoft, N.S.; Svartstein, A.W.; Møller, D.L.; Jensen, C.S.; Benfield, T.; Jensen, J.S.; Thudium, R.F.; Nielsen, S.D. Incidence of bacterial respiratory infection and pneumonia in people with HIV with and without airflow limitation. Int. J. Infect. Dis. 2024, 139, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Trickey, A.; McGinnis, K.; Gill, M.J.; Abgrall, S.; Berenguer, J.; Wyen, C.; Hessamfar, M.; Reiss, P.; Kusejko, K.; Silverberg, M.J.; et al. Longitudinal trends in causes of death among adults with HIV on antiretroviral therapy in Europe and North America from 1996 to 2020: A collaboration of cohort studies. Lancet HIV 2024, 11, e176–e185. [Google Scholar] [CrossRef]
- Stanzel, F. Bronchoalveolar lavage. In Principles and Practice of Interventional Pulmonology; Springer: Berlin/Heidelberg, Germany, 2012; pp. 165–176. [Google Scholar]
- Schiff, A.E.; Linder, A.H.; Luhembo, S.N.; Banning, S.; Deymier, M.J.; Diefenbach, T.J.; Dickey, A.K.; Tsibris, A.M.; Balazs, A.B.; Cho, J.L.; et al. T cell-tropic HIV efficiently infects alveolar macrophages through contact with infected CD4+ T cells. Sci. Rep. 2021, 11, 3890. [Google Scholar] [CrossRef]
- Twigg, H.L.; Schnizlein-Bick, C.T.; Weiden, M.; Valentine, F.; Wheat, J.; Day, R.B.; Rominger, H.; Zheng, L.; Collman, R.G.; Coombs, R.W.; et al. Measurement of antiretroviral drugs in the lungs of HIV-infected patients. HIV Ther. 2010, 4, 247–251. [Google Scholar] [CrossRef]
- Collini, P.J.; Bewley, M.A.; Mohasin, M.; Marriott, H.M.; Miller, R.F.; Geretti, A.M.; Beloukas, A.; Papadimitropoulos, A.; Read, R.C.; Noursadeghi, M.; et al. HIV gp120 in the Lungs of Antiretroviral Therapy-treated Individuals Impairs Alveolar Macrophage Responses to Pneumococci. Am. J. Respir. Crit. Care Med. 2018, 197, 1604–1615. [Google Scholar] [CrossRef]
- Cribbs, S.K.; Lennox, J.; Caliendo, A.M.; Brown, L.A.; Guidot, D.M. Healthy HIV-1-Infected Individuals on Highly Active Antiretroviral Therapy Harbor HIV-1 in Their Alveolar Macrophages. AIDS Res. Hum. Retroviruses 2015, 31, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Costiniuk, C.T.; Salahuddin, S.; Farnos, O.; Olivenstein, R.; Pagliuzza, A.; Orlova, M.; Schurr, E.; De Castro, C.; Bourbeau, J.; Routy, J.P.; et al. HIV persistence in mucosal CD4+ T cells within the lungs of adults receiving long-term suppressive antiretroviral therapy. Aids 2018, 32, 2279–2289. [Google Scholar] [CrossRef] [PubMed]
- Alexandrova, Y.; Costiniuk, C.T.; Jenabian, M.A. Pulmonary Immune Dysregulation and Viral Persistence During HIV Infection. Front. Immunol. 2021, 12, 808722. [Google Scholar] [CrossRef]
- Duko, B.; Ayalew, M.; Ayano, G. The prevalence of alcohol use disorders among people living with HIV/AIDS: A systematic review and meta-analysis. Subst. Abus. Treat. Prev. Policy 2019, 14, 52. [Google Scholar] [CrossRef] [PubMed]
- New-Aaron, M.; Kang, M.; Yeligar, S.M. Pulmonary symptoms associated with heavy alcohol consumption among people living with HIV: An analysis of the NHANES 1999–2010. Alcohol Alcohol. 2024, 59, agae021. [Google Scholar] [CrossRef] [PubMed]
- New-Aaron, M.; Chang, S.S.; Fan, X.; Staitieh, B.S.; Koval, M.; Yeligar, S.M. Pioglitazone Reverses Alcohol-induced HIV Replication and IL-1β Expression in Alveolar Macrophages. Am. J. Respir. Cell Mol. Biol. 2025, 73, 713–724. [Google Scholar] [CrossRef]
- Castellano, P.; Prevedel, L.; Valdebenito, S.; Eugenin, E.A. HIV infection and latency induce a unique metabolic signature in human macrophages. Sci. Rep. 2019, 9, 3941. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, D.; Chiozzi, P.; Falzoni, S.; Dal Susino, M.; Melchiorri, L.; Baricordi, O.R.; Di Virgilio, F. Extracellular ATP triggers IL-1 beta release by activating the purinergic P2Z receptor of human macrophages. J. Immunol. 1997, 159, 1451–1458. [Google Scholar] [CrossRef]
- Tavares, L.A.; Januário, Y.C.; daSilva, L.L.P. HIV-1 Hijacking of Host ATPases and GTPases That Control Protein Trafficking. Front. Cell Dev. Biol. 2021, 9, 622610. [Google Scholar] [CrossRef]
- Wang, X.; Yang, Z.; Ye, X.; McGuinness, O.P.; Yang, H.; Lu, H.; Ye, J. NF-κB inactivation in myeloid cell leads to reprogramming of whole-body energy metabolism in response to high-fat diet. Cell Death Discov. 2025, 11, 367. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Ybanez, M.D.; Johnson, H.S.; McDonald, J.N.; Mesropyan, L.; Sancheti, H.; Martin, G.; Martin, A.; Lim, A.M.; Dara, L.; et al. Dynamic adaptation of liver mitochondria to chronic alcohol feeding in mice: Biogenesis, remodeling, and functional alterations. J. Biol. Chem. 2012, 287, 42165–42179. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Alvarez, R.; Martinez-Outschoorn, U.E.; Lin, Z.; Lamb, R.; Hulit, J.; Howell, A.; Sotgia, F.; Rubin, E.; Lisanti, M.P. Ethanol exposure induces the cancer-associated fibroblast phenotype and lethal tumor metabolism: Implications for breast cancer prevention. Cell Cycle 2013, 12, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Crotty, K.M.; Kabir, S.A.; Chang, S.S.; Mehta, A.J.; Yeligar, S.M. Pioglitazone reverses alcohol-induced alterations in alveolar macrophage mitochondrial phenotype. Alcohol. Clin. Exp. Res. 2024, 48, 810–826. [Google Scholar] [CrossRef] [PubMed]
- Staitieh, B.S.; Ding, L.; Neveu, W.A.; Spearman, P.; Guidot, D.M.; Fan, X. HIV-1 decreases Nrf2/ARE activity and phagocytic function in alveolar macrophages. J. Leukoc. Biol. 2017, 102, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Ten, V.S.; Ratner, V. Mitochondrial bioenergetics and pulmonary dysfunction: Current progress and future directions. Paediatr. Respir. Rev. 2020, 34, 37–45. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, B.; Wang, Y.; Zhang, H.; He, L.; Wang, P.; Dong, M. Mitochondrial dysfunction in pulmonary arterial hypertension. Front. Physiol. 2022, 13, 1079989. [Google Scholar] [CrossRef] [PubMed]
- Lewis, W.; Copeland, W.C.; Day, B.J. Mitochondrial DNA Depletion, Oxidative Stress, and Mutation: Mechanisms 0f Dysfunction from Nucleoside Reverse Transcriptase Inhibitors. Lab. Investig. 2001, 81, 777–790. [Google Scholar] [CrossRef]
- Walker, U.A.; Bäuerle, J.; Laguno, M.; Murillas, J.; Mauss, S.; Schmutz, G.; Setzer, B.; Miquel, R.; Gatell, J.M.; Mallolas, J. Depletion of mitochondrial DNA in liver under antiretroviral therapy with didanosine, stavudine, or zalcitabine. Hepatology 2004, 39, 311–317. [Google Scholar] [CrossRef]
- Selvaraj, S.; Ghebremichael, M.; Li, M.; Foli, Y.; Langs-Barlow, A.; Ogbuagu, A.; Barakat, L.; Tubridy, E.; Edifor, R.; Lam, W.; et al. Antiretroviral therapy-induced mitochondrial toxicity: Potential mechanisms beyond polymerase-γ inhibition. Clin. Pharmacol. Ther. 2014, 96, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Venhoff, N.; Setzer, B.; Melkaoui, K.; Walker, U.A. Mitochondrial toxicity of tenofovir, emtricitabine and abacavir alone and in combination with additional nucleoside reverse transcriptase inhibitors. Antivir. Ther. 2007, 12, 1075–1085. [Google Scholar] [CrossRef]
- Young, M.J. Off-Target Effects of Drugs that Disrupt Human Mitochondrial DNA Maintenance. Front. Mol. Biosci. 2017, 4, 74. [Google Scholar] [CrossRef]
- Barroso, S.; Guitart-Mampel, M.; García-García, F.J.; Cantó-Santos, J.; Valls-Roca, L.; Andújar-Sánchez, F.; Vilaseca-Capel, A.; Tobías, E.; Arias-Dimas, A.; Quesada-López, T.; et al. Metabolic, Mitochondrial, and Inflammatory Effects of Efavirenz, Emtricitabine, and Tenofovir Disoproxil Fumarate in Asymptomatic Antiretroviral-Naïve People with HIV. Int. J. Mol. Sci. 2024, 25, 8418. [Google Scholar] [CrossRef]
- Morris, N.L.; Harris, F.L.; Brown, L.A.S.; Yeligar, S.M. Alcohol induces mitochondrial derangements in alveolar macrophages by upregulating NADPH oxidase 4. Alcohol 2021, 90, 27–38. [Google Scholar] [CrossRef]
- Staitieh, B.S.; Hu, X.; Yeligar, S.M.; Auld, S.C. Paired ATAC- and RNA-seq offer insight into the impact of HIV on alveolar macrophages: A pilot study. Sci. Rep. 2023, 13, 15276. [Google Scholar] [CrossRef] [PubMed]
- Sapkota, M.; Burnham, E.L.; DeVasure, J.M.; Sweeter, J.M.; Hunter, C.D.; Duryee, M.J.; Klassen, L.W.; Kharbanda, K.K.; Sisson, J.H.; Thiele, G.M.; et al. Malondialdehyde-Acetaldehyde (MAA) Protein Adducts Are Found Exclusively in the Lungs of Smokers with Alcohol Use Disorders and Are Associated with Systemic Anti-MAA Antibodies. Alcohol. Clin. Exp. Res. 2017, 41, 2093–2099. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, T.A.; Warren, K.J.; Wetzel, T.J.; Suwondo, T.; Rensch, G.P.; DeVasure, J.M.; Mosley, D.D.; Kharbanda, K.K.; Thiele, G.M.; Burnham, E.L.; et al. Malondialdehyde-Acetaldehyde Adduct Formation Decreases Immunoglobulin A Transport across Airway Epithelium in Smokers Who Abuse Alcohol. Am. J. Pathol. 2021, 191, 1732–1742. [Google Scholar] [CrossRef] [PubMed]
- Kaphalia, L.; Calhoun, W.J. Alcoholic lung injury: Metabolic, biochemical and immunological aspects. Toxicol. Lett. 2013, 222, 171–179. [Google Scholar] [CrossRef]
- Shaskan, E.G.; Dolinsky, Z.S. Elevated endogenous breath acetaldehyde levels among abusers of alcohol and cigarettes. Prog. Neuropsychopharmacol. Biol. Psychiatry 1985, 9, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Lindros, K.O.; Stowell, A.; Pikkarainen, P.; Salaspuro, M. Elevated blood acetaldehyde in alcoholics with accelerated ethanol elimination. Pharmacol. Biochem. Behav. 1980, 13, 119–124. [Google Scholar] [CrossRef]
- New-Aaron, M.; Thomes, P.G.; Ganesan, M.; Dagur, R.S.; Donohue, T.M.; Kusum, K.K.; Poluektova, L.Y.; Osna, N.A. Alcohol-Induced Lysosomal Damage and Suppression of Lysosome Biogenesis Contribute to Hepatotoxicity in HIV-Exposed Liver Cells. Biomolecules 2021, 11, 1497. [Google Scholar] [CrossRef]
- Potash, M.J.; Chao, W.; Bentsman, G.; Paris, N.; Saini, M.; Nitkiewicz, J.; Belem, P.; Sharer, L.; Brooks, A.I.; Volsky, D.J. A mouse model for study of systemic HIV-1 infection, antiviral immune responses, and neuroinvasiveness. Proc. Natl. Acad. Sci. USA 2005, 102, 3760–3765. [Google Scholar] [CrossRef]
- Gu, C.J.; Borjabad, A.; Hadas, E.; Kelschenbach, J.; Kim, B.H.; Chao, W.; Arancio, O.; Suh, J.; Polsky, B.; McMillan, J.; et al. EcoHIV infection of mice establishes latent viral reservoirs in T cells and active viral reservoirs in macrophages that are sufficient for induction of neurocognitive impairment. PLoS Pathog. 2018, 14, e1007061. [Google Scholar] [CrossRef]
- New-Aaron, M.; Dagur, R.S.; Koganti, S.S.; Ganesan, M.; Wang, W.; Makarov, E.; Ogunnaike, M.; Kharbanda, K.K.; Poluektova, L.Y.; Osna, N.A. Alcohol and HIV-Derived Hepatocyte Apoptotic Bodies Induce Hepatic Stellate Cell Activation. Biology 2022, 11, 1059. [Google Scholar] [CrossRef]
- Banga, R.; Perreau, M. The multifaceted nature of HIV tissue reservoirs. Curr. Opin. HIV AIDS 2024, 19, 116–123. [Google Scholar] [CrossRef]
- Wu, G.; Zuck, P.; Goh, S.L.; Milush, J.M.; Vohra, P.; Wong, J.K.; Somsouk, M.; Yukl, S.A.; Shacklett, B.L.; Chomont, N.; et al. Gag p24 Is a Marker of Human Immunodeficiency Virus Expression in Tissues and Correlates With Immune Response. J. Infect. Dis. 2021, 224, 1593–1598. [Google Scholar] [CrossRef] [PubMed]
- Akiso, M.; Ameka, M.; Naidoo, K.; Langat, R.; Kombo, J.; Sikuku, D.; Ndung’u, T.; Altfeld, M.; Anzala, O.; Mureithi, M. Metabolic and mitochondrial dysregulation in CD4+ T cells from HIV-positive women on combination anti-retroviral therapy. PLoS ONE 2023, 18, e0286436. [Google Scholar] [CrossRef] [PubMed]
- Morris, N.L.; Michael, D.N.; Crotty, K.M.; Chang, S.S.; Yeligar, S.M. Alcohol-Induced Glycolytic Shift in Alveolar Macrophages Is Mediated by Hypoxia-Inducible Factor-1 Alpha. Front. Immunol. 2022, 13, 865492. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Kuwabara, R.; de Haan, B.J.; Smink, A.M.; de Vos, P. Acetate and Butyrate Improve β-cell Metabolism and Mitochondrial Respiration under Oxidative Stress. Int. J. Mol. Sci. 2020, 21, 1542. [Google Scholar] [CrossRef]
- Maruta, H.; Abe, R.; Yamashita, H. Effect of Long-Term Supplementation with Acetic Acid on the Skeletal Muscle of Aging Sprague Dawley Rats. Int. J. Mol. Sci. 2022, 23, 4691. [Google Scholar] [CrossRef]
- Sahuri-Arisoylu, M.; Mould, R.R.; Shinjyo, N.; Bligh, S.W.A.; Nunn, A.V.W.; Guy, G.W.; Thomas, E.L.; Bell, J.D. Acetate Induces Growth Arrest in Colon Cancer Cells Through Modulation of Mitochondrial Function. Front. Nutr. 2021, 8, 588466. [Google Scholar] [CrossRef] [PubMed]
- Foo, J.; Bellot, G.; Pervaiz, S.; Alonso, S. Mitochondria-mediated oxidative stress during viral infection. Trends Microbiol. 2022, 30, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.J.; Lee, W.; Ku, K.B.; Yoon, G.Y.; Moon, H.W.; Kim, C.; Kim, M.H.; Yi, Y.S.; Jun, S.; Kim, B.T.; et al. SARS-CoV-2 aberrantly elevates mitochondrial bioenergetics to induce robust virus propagation. Signal Transduct. Target. Ther. 2024, 9, 125. [Google Scholar] [CrossRef]
- Sorbara, M.T.; Girardin, S.E. Mitochondrial ROS fuel the inflammasome. Cell Res. 2011, 21, 558–560. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak, K.; Lukasiak, A.; Grel, H.; Dworakowska, B.; Jakiela, S.; Stobiecka, M. Monitoring of dynamic ATP level changes by oligomycin-modulated ATP synthase inhibition in SW480 cancer cells using fluorescent “On-Off” switching DNA aptamer. Anal. Bioanal. Chem. 2019, 411, 6899–6911. [Google Scholar] [CrossRef]
- Mauro, C.; Leow, S.C.; Anso, E.; Rocha, S.; Thotakura, A.K.; Tornatore, L.; Moretti, M.; De Smaele, E.; Beg, A.A.; Tergaonkar, V.; et al. NF-κB controls energy homeostasis and metabolic adaptation by upregulating mitochondrial respiration. Nat. Cell Biol. 2011, 13, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Assefa, Y.; Gilks, C.F. Ending the epidemic of HIV/AIDS by 2030: Will there be an endgame to HIV, or an endemic HIV requiring an integrated health systems response in many countries? Int. J. Infect. Dis. 2020, 100, 273–277. [Google Scholar] [CrossRef]
- Lorenzo-Redondo, R.; Fryer, H.R.; Bedford, T.; Kim, E.-Y.; Archer, J.; Kosakovsky Pond, S.L.; Chung, Y.-S.; Penugonda, S.; Chipman, J.G.; Fletcher, C.V.; et al. Persistent HIV-1 replication maintains the tissue reservoir during therapy. Nature 2016, 530, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Sadowski, I.; Hashemi, F.B. Strategies to eradicate HIV from infected patients: Elimination of latent provirus reservoirs. Cell Mol. Life Sci. 2019, 76, 3583–3600. [Google Scholar] [CrossRef]
- Braithwaite, R.S.; Bryant, K.J. Influence of alcohol consumption on adherence to and toxicity of antiretroviral therapy and survival. Alcohol. Res. Health 2010, 33, 280–287. [Google Scholar]
- Baum, M.K.; Rafie, C.; Lai, S.; Sales, S.; Page, J.B.; Campa, A. Alcohol use accelerates HIV disease progression. AIDS Res. Hum. Retroviruses 2010, 26, 511–518. [Google Scholar] [CrossRef]
- Bagby, G.J.; Amedee, A.M.; Siggins, R.W.; Molina, P.E.; Nelson, S.; Veazey, R.S. Alcohol and HIV Effects on the Immune System. Alcohol. Res. 2015, 37, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.C.; Hahn, J.A.; Saitz, R.; Bryant, K.; Lira, M.C.; Samet, J.H. Alcohol Use and Human Immunodeficiency Virus (HIV) Infection: Current Knowledge, Implications, and Future Directions. Alcohol. Clin. Exp. Res. 2016, 40, 2056–2072. [Google Scholar] [CrossRef]
- Barrero, C.A.; Datta, P.K.; Sen, S.; Deshmane, S.; Amini, S.; Khalili, K.; Merali, S. HIV-1 Vpr Modulates Macrophage Metabolic Pathways: A SILAC-Based Quantitative Analysis. PLoS ONE 2013, 8, e68376. [Google Scholar] [CrossRef] [PubMed]
- Staitieh, B.S.; Auld, S.C.; Ahmed, M.; Fan, X.; Smirnova, N.; Yeligar, S.M. Granulocyte Macrophage-Colony Stimulating Factor Reverses HIV Protein-Induced Mitochondrial Derangements in Alveolar Macrophages. AIDS Res. Hum. Retroviruses 2021, 37, 224–232. [Google Scholar] [CrossRef]
- Yeligar, S.M.; Harris, F.L.; Hart, C.M.; Brown, L.A. Ethanol induces oxidative stress in alveolar macrophages via upregulation of NADPH oxidases. J. Immunol. 2012, 188, 3648–3657. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Harris, F.L.; Jones, D.P.; Brown, L.A.S. Alcohol induces mitochondrial redox imbalance in alveolar macrophages. Free Radic. Biol. Med. 2013, 65, 1427–1434. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.D.; Brown, L.A. Ethanol (EtOH)-induced TGF-β1 and reactive oxygen species production are necessary for EtOH-induced alveolar macrophage dysfunction and induction of alternative activation. Alcohol. Clin. Exp. Res. 2012, 36, 1952–1962. [Google Scholar] [CrossRef]
- Shytaj, I.L.; Lucic, B.; Forcato, M.; Penzo, C.; Billingsley, J.; Laketa, V.; Bosinger, S.; Stanic, M.; Gregoretti, F.; Antonelli, L.; et al. Alterations of redox and iron metabolism accompany the development of HIV latency. Embo J. 2020, 39, e102209. [Google Scholar] [CrossRef]
- Fan, X.; Joshi, P.C.; Koval, M.; Guidot, D.M. Chronic alcohol ingestion exacerbates lung epithelial barrier dysfunction in HIV-1 transgenic rats. Alcohol. Clin. Exp. Res. 2011, 35, 1866–1875. [Google Scholar] [CrossRef]
- Suresh, D.R.; Annam, V.; Pratibha, K.; Prasad, B.V.M. Total antioxidant capacity—A novel early bio-chemical marker of oxidative stress in HIV infected individuals. J. Biomed. Sci. 2009, 16, 61. [Google Scholar] [CrossRef]
- Bogacka, I.; Xie, H.; Bray, G.A.; Smith, S.R. Pioglitazone Induces Mitochondrial Biogenesis in Human Subcutaneous Adipose Tissue In Vivo. Diabetes 2005, 54, 1392–1399. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, X.; Meng, L.; Gong, M.; Li, J.; Shi, W.; Qiu, J.; Yang, Y.; Zhao, J.; Suo, Y.; et al. Pioglitazone Inhibits Diabetes-Induced Atrial Mitochondrial Oxidative Stress and Improves Mitochondrial Biogenesis, Dynamics, and Function Through the PPAR-γ/PGC-1α Signaling Pathway. Front. Pharmacol. 2021, 12, 658362. [Google Scholar] [CrossRef]
- Abu Shelbayeh, O.; Arroum, T.; Morris, S.; Busch, K.B. PGC-1α Is a Master Regulator of Mitochondrial Lifecycle and ROS Stress Response. Antioxidants 2023, 12, 1075. [Google Scholar] [CrossRef] [PubMed]
- Giridharan, S.; Srinivasan, M. Mechanisms of NF-κB p65 and strategies for therapeutic manipulation. J. Inflamm. Res. 2018, 11, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, Y.; Xiao, F.; Liu, Y.; Wang, J.; Gao, H.; Rong, S.; Yao, Y.; Li, J.; Xu, G. The peroxisome proliferator-activated receptor γ agonist pioglitazone prevents NF-κB activation in cisplatin nephrotoxicity through the reduction of p65 acetylation via the AMPK-SIRT1/p300 pathway. Biochem. Pharmacol. 2016, 101, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Al-Muzafar, H.M.; Alshehri, F.S.; Amin, K.A. The role of pioglitazone in antioxidant, anti-inflammatory, and insulin sensitivity in a high fat-carbohydrate diet-induced rat model of insulin resistance. Braz. J. Med. Biol. Res. 2021, 54, e10782. [Google Scholar] [CrossRef]










| Target Genes | Forward Primer | Reverse Primer |
|---|---|---|
| MLV envelope protein | 5′-TGG GAC CAC AGG CTA CAC TAGA-3′ | 5′-TGA TGA CAG CAT GCC AGG GAG TGG-3′ |
| EcoHIV gag | 5′-TGG GAC CAC AGG CTA CAC TAGA-3′ | 5′-CAG CCA AAA CTC TTG CTT TAT GG-3′ |
| EcoHIV Nef | 5′-GAG TGA AAA ATC TCT AGC AGT GGC GC-3′ | 5′-GCT GAA GAG GCA CAG GTT CCT CAG GTCG-3′ |
| EcoHIV Tat | 5′-CCT AGG ACT GCT TGT AAT AAG TGT-3′ | 5′-GTC GGG TCC CCT CGG GAC TGG GAG-3′ |
| EcoHIV Vif | 5′-AAG AGG CGA GGG GCA GCGA-3′ | 5′-TCT TTA CTT TTC TTC TTG GTA CTA CCT TTA TG-3′ |
| HIV 1 gp120 | 5′-TCC TGC TCA ACT TCC TGT CGA G-3′ | 5′-CAC AGG TCA AAC CTC CTA GGA ATG-3′ |
| HIV 1 gag | 5′-GAG GAT CCC CCA TAG TGC AGA ACC TC-3′ | 5′ CCG GTA CCT TAG AAA ACT CTT GCT TTA TG-3′ |
| HIV 1 Tat | 5′-GAA GCA TCC AGG AAG TCA GC-3′ | 5′-GGA GGT GGG TGC TTT GAT AG-3′ |
| IL-1β (human) | 5′-AGC TAC GAA TCT CCG ACCAC-3′ | 5′-CGT TAT CCC ATG TGT CGA AGA A-3′ |
| GAPDH (mouse, human, or rabbit) | 5′-AGC TTG TCA TCA ACG GGA AG-3′ | 5′-TTT GAT GTT AGT GGG GTC TCG-3′ |
| Total | Non-AUD (n = 6) | AUD (n = 8) | |
|---|---|---|---|
| Age in years (mean(SD)) | 39.78 (11.29) | 39.75 (13.19) | 41.33 (9.1) |
| Gender (%) | |||
| Male | 50% | 50% | 50% |
| Female | 50% | 50% | 50% |
| Race (%) | |||
| Black | 79% | 83% | 75% |
| White | 7% | 17% | 0 |
| More than one race | 14% | 0 | 25% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
New-Aaron, M.; Chang, S.; Fan, X.; Mehta, A.; Auld, S.C.; Staitieh, B.S.; Koval, M.; Yeligar, S.M. Pioglitazone Modulates p65-Mediated Mitochondrial Bioenergetics: Implications for Acetaldehyde-Induced HIV Replication in Alveolar Macrophages. Biomolecules 2025, 15, 1737. https://doi.org/10.3390/biom15121737
New-Aaron M, Chang S, Fan X, Mehta A, Auld SC, Staitieh BS, Koval M, Yeligar SM. Pioglitazone Modulates p65-Mediated Mitochondrial Bioenergetics: Implications for Acetaldehyde-Induced HIV Replication in Alveolar Macrophages. Biomolecules. 2025; 15(12):1737. https://doi.org/10.3390/biom15121737
Chicago/Turabian StyleNew-Aaron, Moses, Sarah Chang, Xian Fan, Ashish Mehta, Sara C. Auld, Bashar S. Staitieh, Michael Koval, and Samantha M. Yeligar. 2025. "Pioglitazone Modulates p65-Mediated Mitochondrial Bioenergetics: Implications for Acetaldehyde-Induced HIV Replication in Alveolar Macrophages" Biomolecules 15, no. 12: 1737. https://doi.org/10.3390/biom15121737
APA StyleNew-Aaron, M., Chang, S., Fan, X., Mehta, A., Auld, S. C., Staitieh, B. S., Koval, M., & Yeligar, S. M. (2025). Pioglitazone Modulates p65-Mediated Mitochondrial Bioenergetics: Implications for Acetaldehyde-Induced HIV Replication in Alveolar Macrophages. Biomolecules, 15(12), 1737. https://doi.org/10.3390/biom15121737








