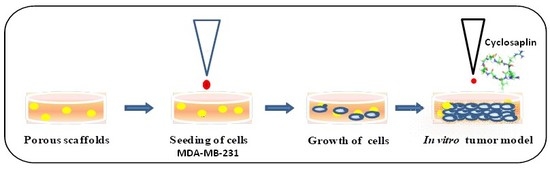
Evaluation of Cyclosaplin Efficacy Using a Silk Based 3D Tumor Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Fibroin from A. mylitta Silkworm
2.3. Fabrication of 2D and 3D Silk Matrices
2.4. Culture, Maintenance and Seeding of MDA-MB-231 Cells
2.5. Cell Proliferation Assay
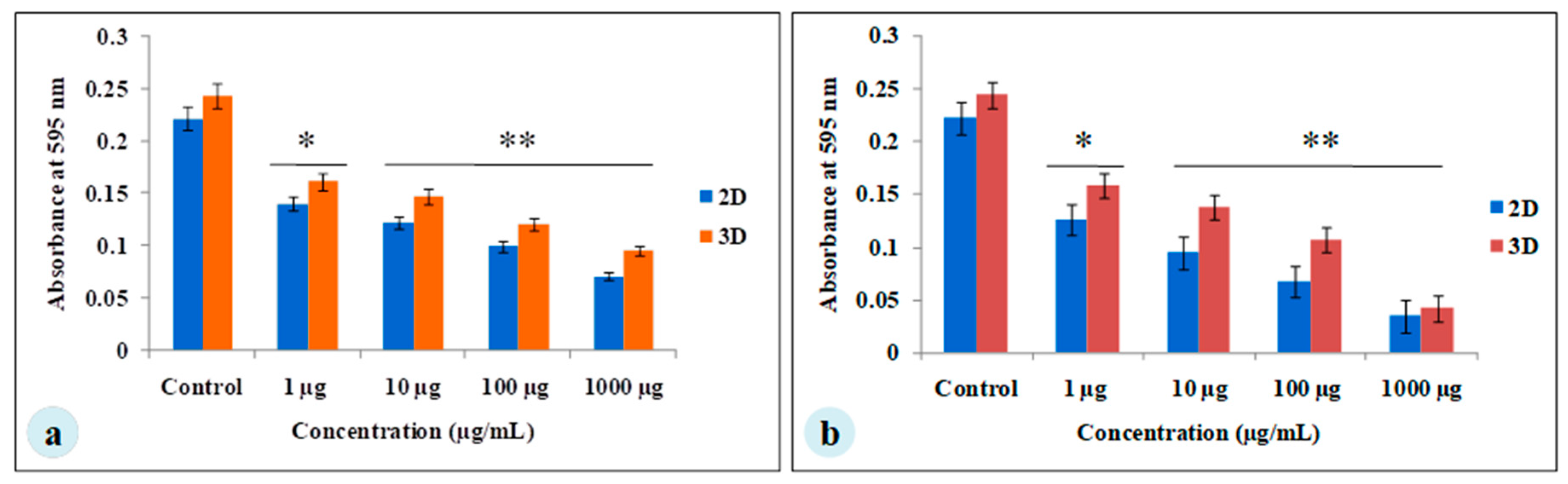
2.6. Chemotherapeutic Studies
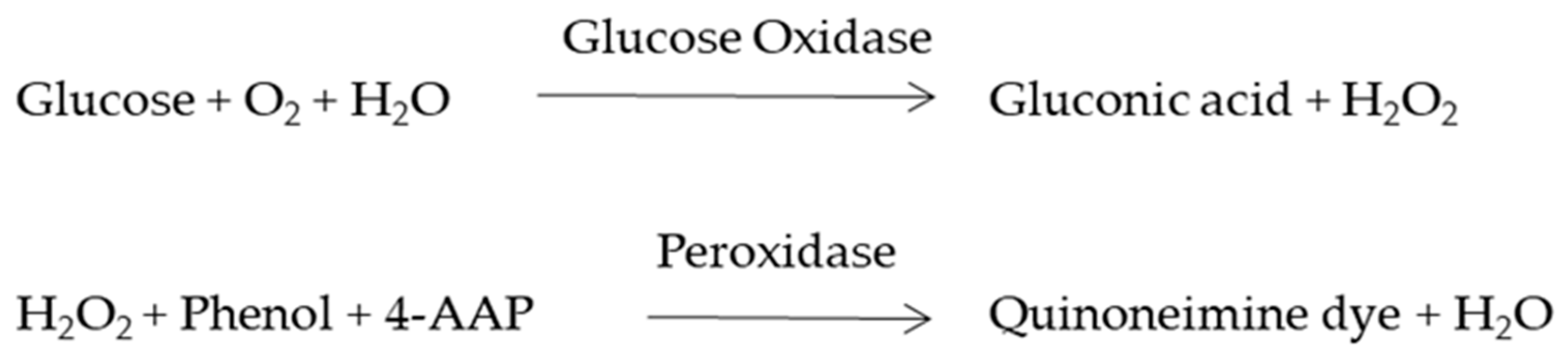
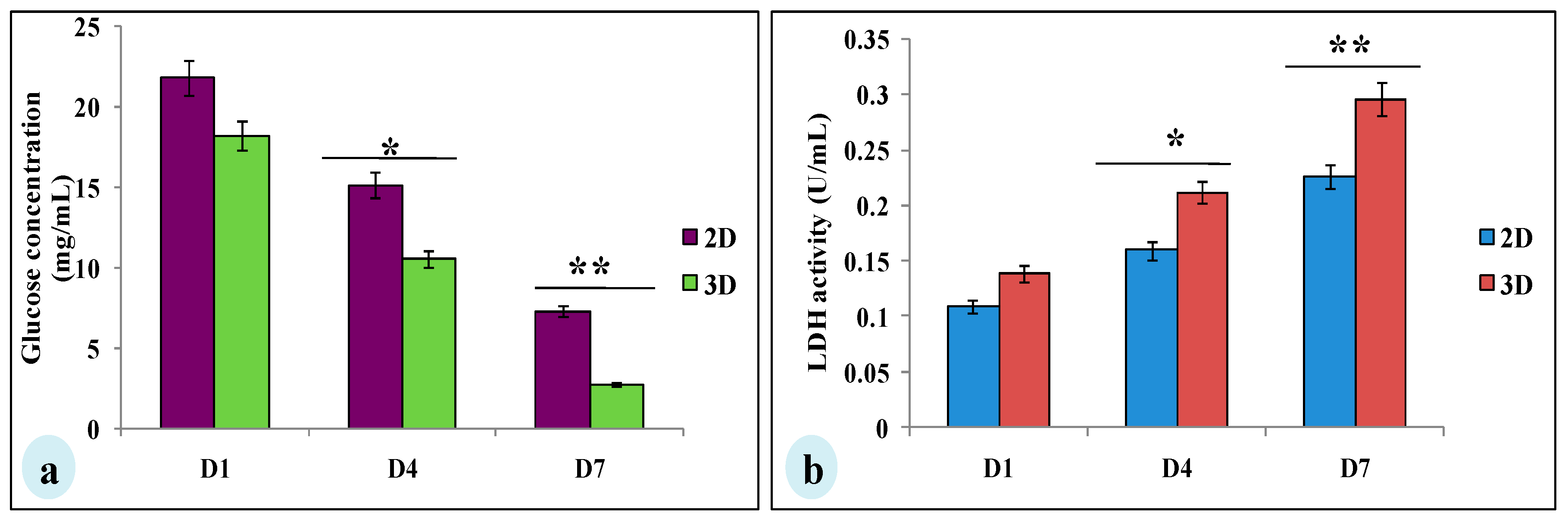
2.7. Glucose Consumption and Lactic Acid Production Assay

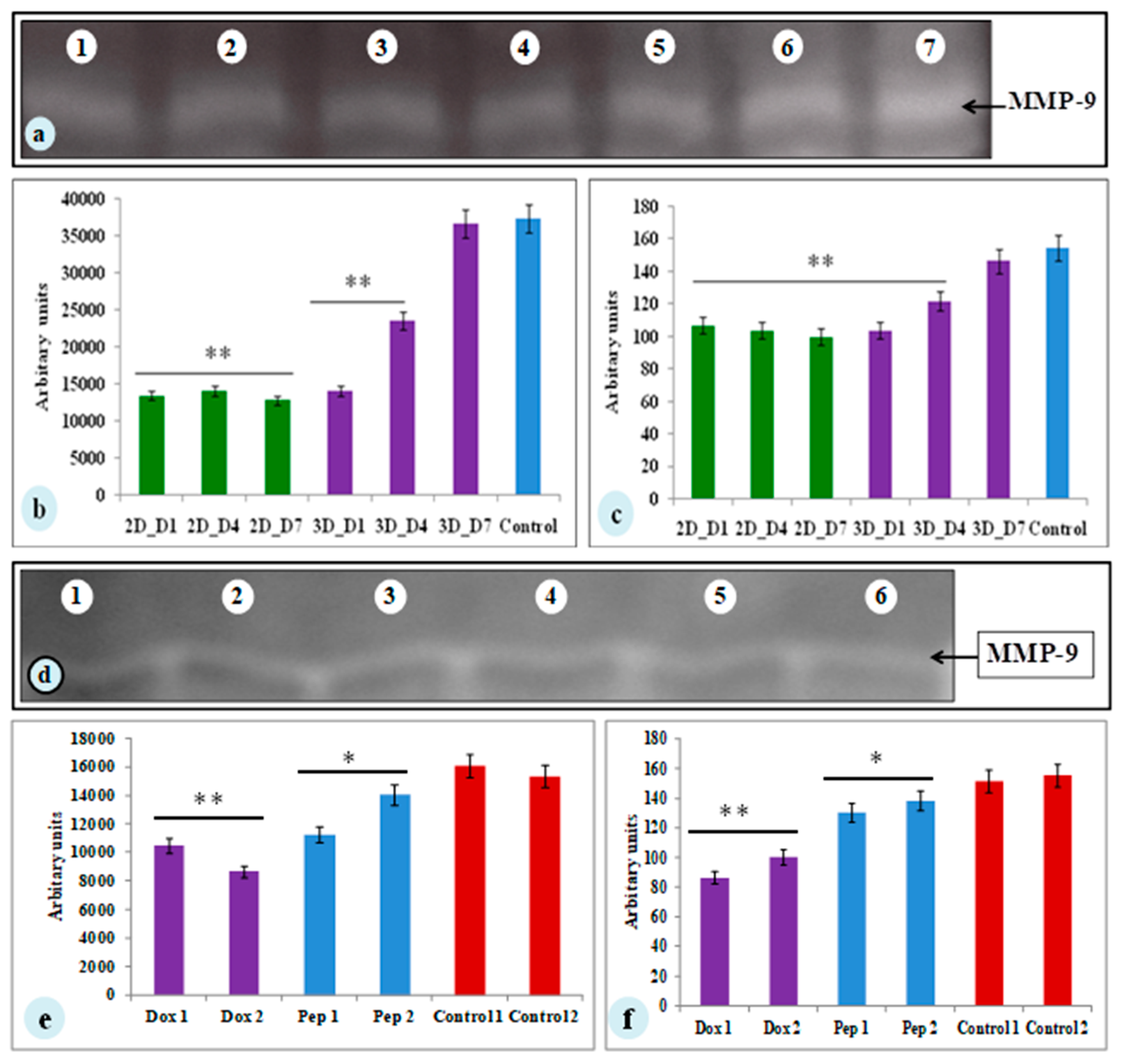
2.8. Matrix Metalloproteinase 9 (MMP-9) Activity of MDA-MB-231 Cells on Silk Constructs
2.9. Scanning Electron Microscopy
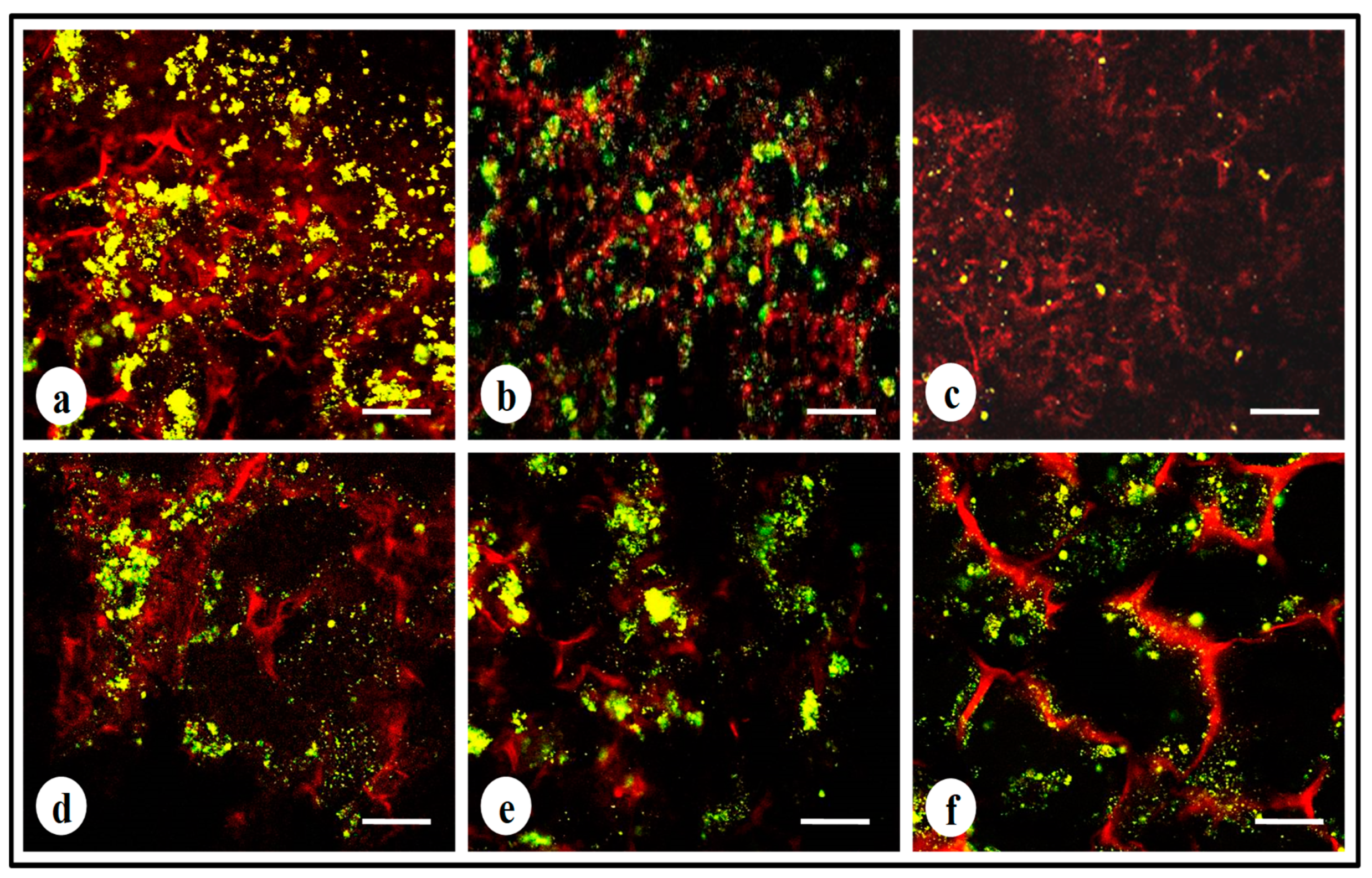
2.10. Live–Dead Assay
2.11. Analysis of Cytoskeletal Organization
2.12. Statistical Analysis
3. Results
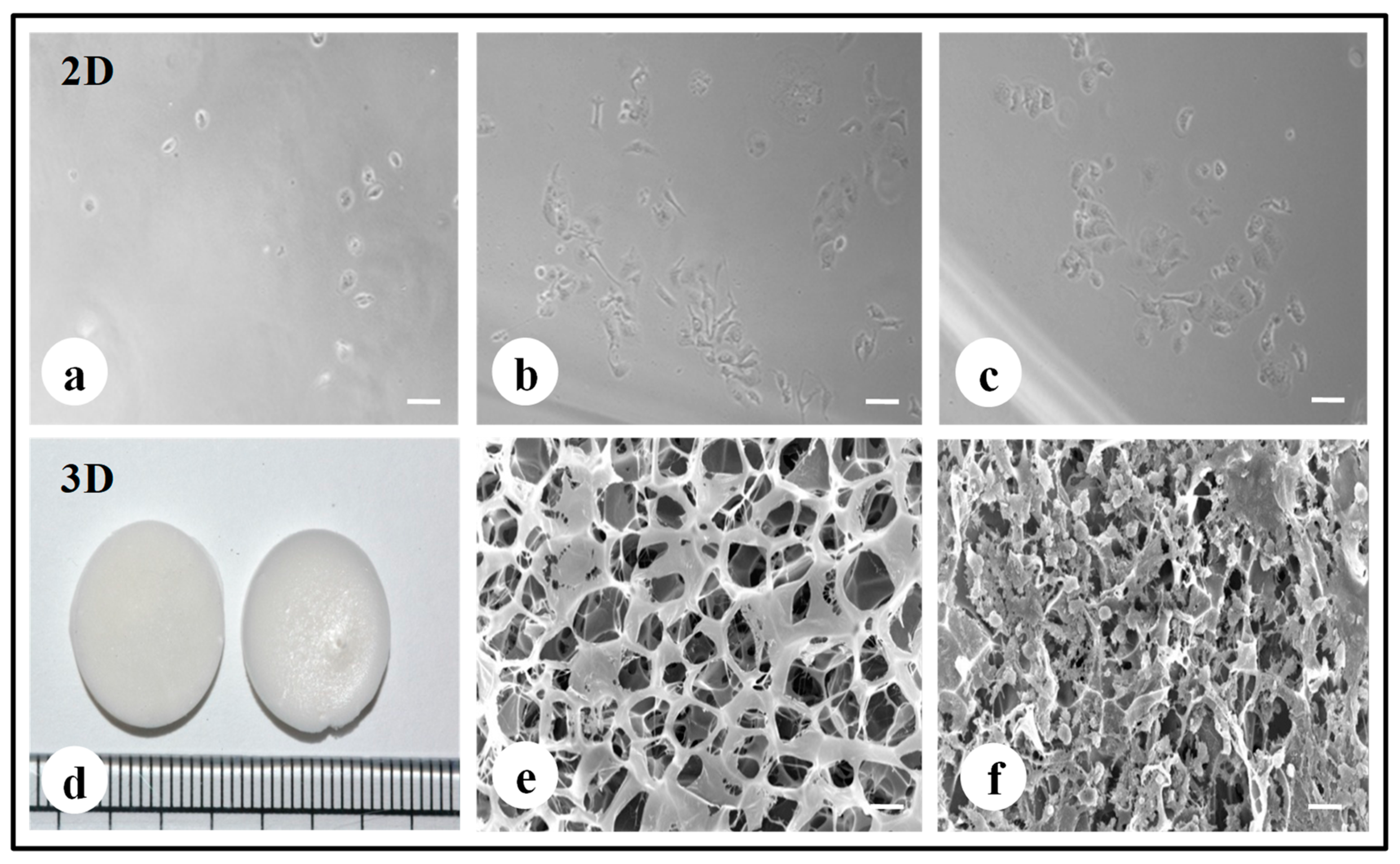
3.1. Preparation and Fabrication of 2D/3D Silk Constructs
3.2. Cell Proliferation Assay
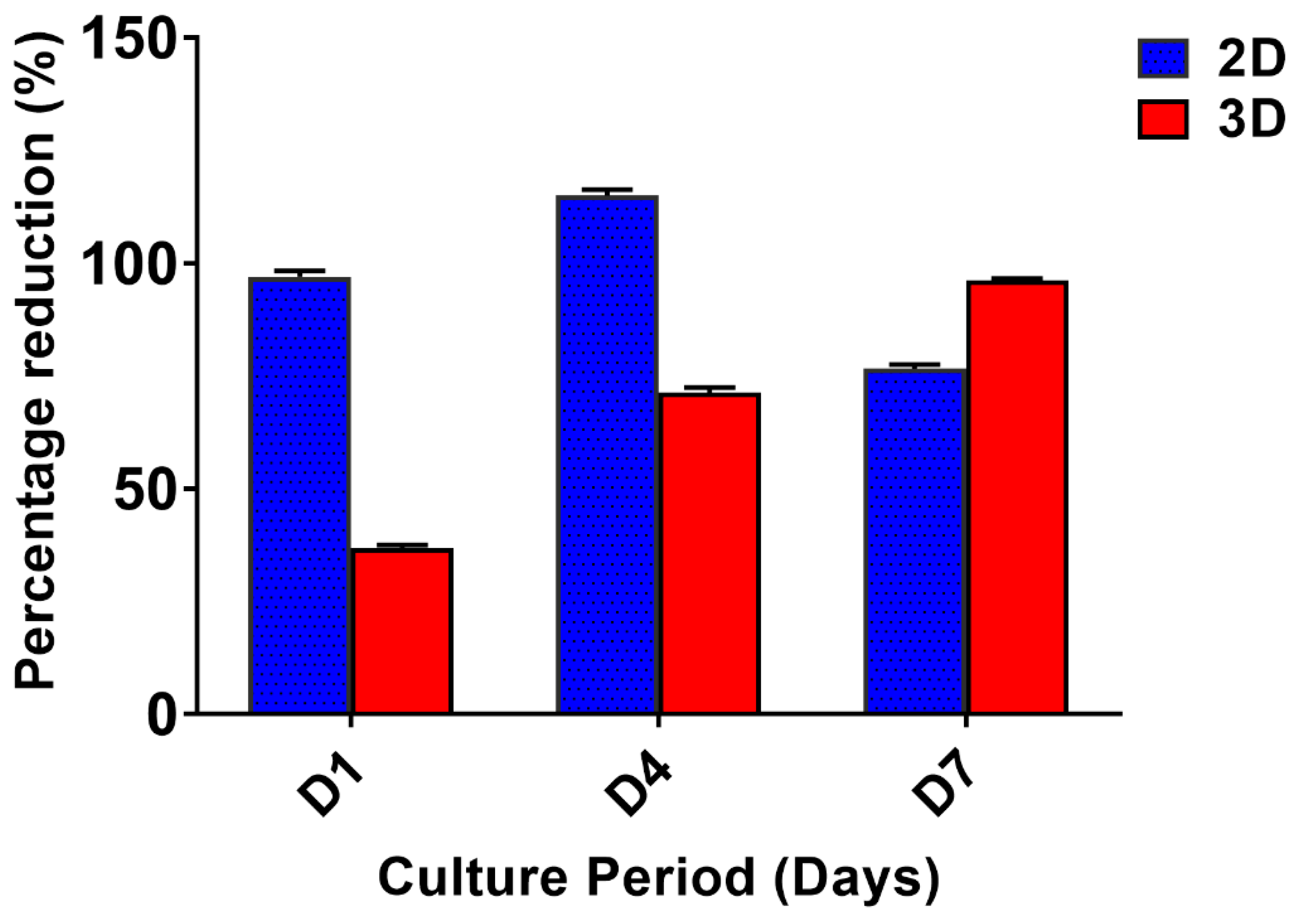
3.3. Chemotherapeutic Studies
3.4. Glucose Consumption and Lactate Dehydrogenase Assay
3.5. MMP-9 Activity of MDA-MB-231 Cells on Silk Constructs
3.6. Scanning Electron Microscopy
3.7. Live–Dead Assay
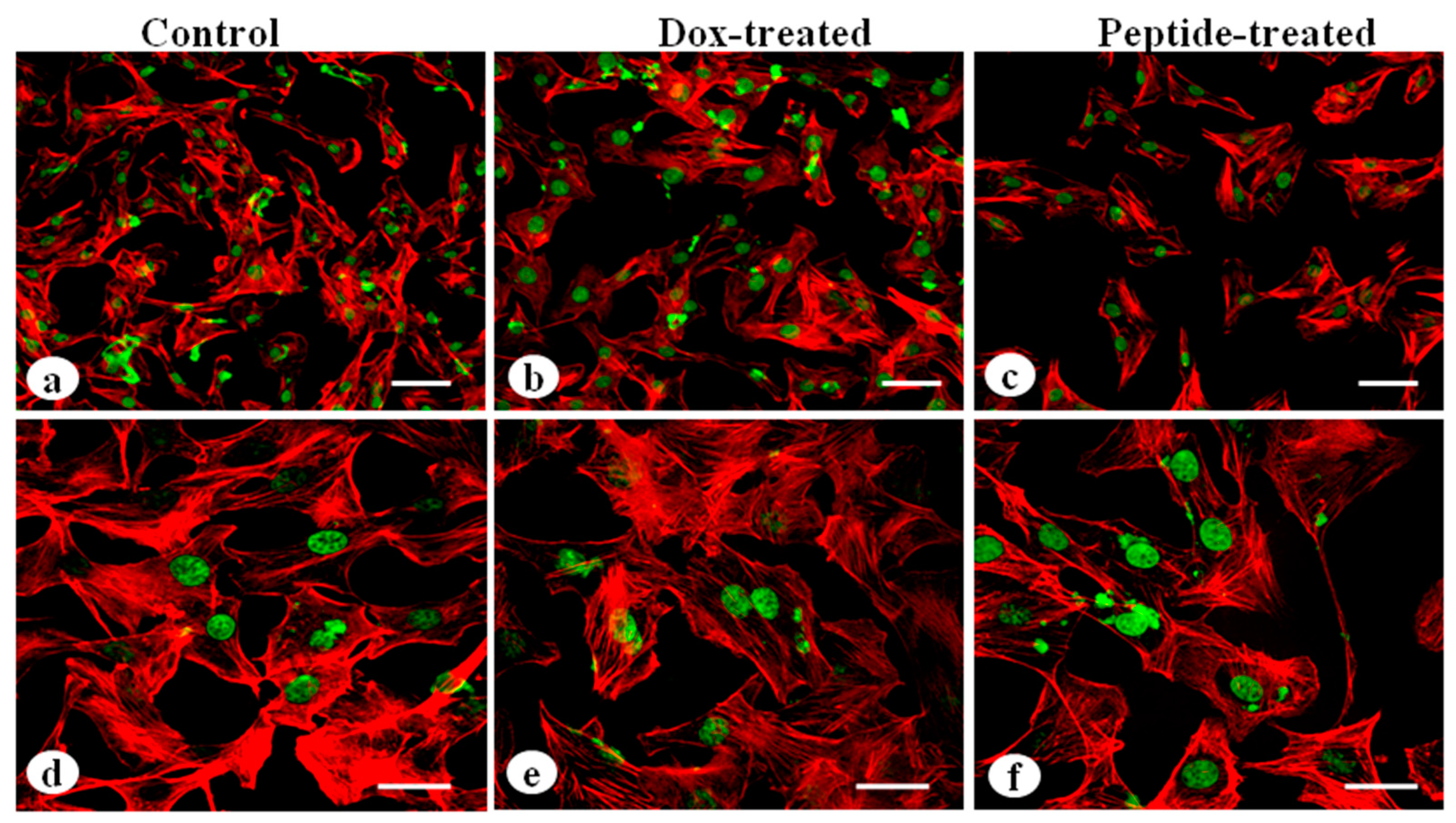
3.8. Analysis of Cytoskeletal Organization
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Talukdar, S.; Kundu, S.C. Engineered 3D Silk-Based Metastasis Models: Interactions Between Human Breast Adenocarcinoma, Mesenchymal Stem Cells and Osteoblast-Like Cells. Adv. Funct. Mater. 2013, 23, 5249–5260. [Google Scholar] [CrossRef]
- Dondajewska, E.; Juzwa, W.; Mackiewicz, A.; Dams-Kozlowska, H. 3 Heterotypic breast cancer model based on a silk fibroin scaffold to study the tumor microenvironment. Oncotarget 2017, 9, 4935–4950. [Google Scholar] [CrossRef]
- Iivascu, A.; Makubbies, K. Rapid generation of single-tumor spheroids for high throughput cell function and toxicity analysis. J. Biomol. Screen. 2006, 11, 8. [Google Scholar] [CrossRef] [PubMed]
- Breslin, S.; O’Driscoll, L. Three-dimensional cell culture: The missing link in drug discovery. Drug Discov. Today 2013, 18, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Cukierman, E.; Roumen, P.; Kenneth, M.Y. Cell interactions with three-dimensional matrices. Curr. Opin. Cell Biol. 2002, 14, 633–639. [Google Scholar] [CrossRef]
- Inman, J.L.; Bissell, J.M. Apical polarity in three-dimensional culture systems: Where to now. J. Biol. 2010, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- Martin-Belmonte, F.; Yu, W.; Rodriguez-Fraticelli, A.E.; Ewald, A.; Werb, Z.; Alonso, M.A. Cell polarity dynamics controls the mechanism of lumen formation in epithelial morphogenesis. Curr. Biol. 2008, 18, 507–513. [Google Scholar] [CrossRef]
- Kunz-Schughar, L.A.; Freyer, J.P.; Hofstaedter, F.; Ebner, R. The use of 3D cultures for high-throughput screening: The multicellular spheroid model. J. Biomol. Screen. 2004, 9, 273–285. [Google Scholar] [CrossRef]
- Shin, C.S.; Kwak, B.; Han, B.; Park, K. Development of an in vitro 3D tumor model to study therapeutic efficiency of an anticancer drug. Mol. Pharm. 2013, 10, 2167–2175. [Google Scholar] [CrossRef]
- Gurski, L.A.; Petrelli, N.J.; Jia, X.; Farach-Carson, M.C. 3D matrices for anti-cancer Drug testing and Development. Oncol. Issues 2010, 25, 20–25. [Google Scholar] [CrossRef]
- Draoui, N.; Feron, O. Lactate shuttles at a glance: From physiological paradigms to anti-cancer treatments. Dis. Models Mech. 2011, 4, 727–732. [Google Scholar] [CrossRef]
- Cairns, R.A.; Harris, I.S.; Mak, T.W. Regulation of cancer cell metabolism. Nat. Rev. Cancer 2011, 11, 85–95. [Google Scholar] [CrossRef]
- Coussens, L.M.; Fingleton, B.; Matrisian, L.M. Matrix metalloproteinase inhibitors and cancer: Trials and tribulations. Science 2002, 295, 2387–2392. [Google Scholar] [CrossRef]
- Egeblad, M.; Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nat. Rev. Cancer 2002, 2, 161–174. [Google Scholar] [CrossRef]
- Rundhaug, J.E. Matrix metalloproteinases and angiogenesis. J. Cell. Mol. Med. 2005, 9, 267–285. [Google Scholar] [CrossRef]
- Mueller, M.M.; Fusenig, N.E. Friends or foes—Bipolar effects of the tumor stroma in cancer. Nat. Rev. Cancer 2004, 4, 839–849. [Google Scholar] [CrossRef]
- Lee, J.; Cuddihy, M.J.; Kotov, N.A. Three-dimensional cell culture matrices: State of the art. Tissue Eng. Part B Rev. 2008, 14, 61–86. [Google Scholar] [CrossRef]
- Vepari, C.; Kaplan, D.L. Silk as a biomaterial. Prog. Polym. Sci. 2007, 32, 991–1007. [Google Scholar] [CrossRef]
- Subia, B.; Kundu, S.C. Drug loading and release on tumor cells using silk fibroinalbumin nanoparticles as carriers. Nanotechnology 2013, 24, 035103. [Google Scholar] [CrossRef]
- Zhang, X.H.; Baughman, C.B.; Kaplan, D.L. In vitro evaluation of electrospun silk fibroin scaffolds for vascular cell growth. Biomaterials 2008, 29, 2217–2227. [Google Scholar] [CrossRef]
- Talukdar, S.; Mandal, M.; Dietmar, W.; Hutmacher, P.J.; Soekmadji, C.; Kundu, S.C. Engineered silk fibroin protein 3D matrices for in vitro tumor model. Biomaterials 2011, 32, 2149–2159. [Google Scholar] [CrossRef]
- Talukdar, S.; Kundu, S.C. A non-mulberry silk fibroin protein based 3D in vitro tumor model for evaluation of anticancer drug activity. Adv. Funct. Mater. 2012, 22, 4778–4788. [Google Scholar] [CrossRef]
- Mishra, A.; Gauri, S.S.; Mukhopadhyay, S.K.; Chatterjee, S.; Das, S.S.; Mandal, S.M.; Dey, S. Identification and structural characterization of a new pro-apoptotic cyclic octapeptide cyclosaplin from somatic seedling of Santalum album L. Peptides 2014, 54, 148–158. [Google Scholar] [CrossRef]
- Kundu, B.; Kundu, S.C. Silk sericin/polyacrylamide in situ forming hydrogels for dermal reconstruction. Biomaterials 2012, 33, 7456–7467. [Google Scholar] [CrossRef]
- Kundu, B.; Saha, P.; Datta, K.; Kundu, S.C. A silk fibroin based hepatocarcinoma model and the assessment of the drug response in hyaluronan-binding protein 1 over expressed HepG2 cells. Biomaterials 2013, 34, 9462–9474. [Google Scholar] [CrossRef]
- Fornari, F.A.; Randolph, J.K.; Yalowich, J.C.; Ritke, M.K.; Gewirtz, D.A. Interference by doxorubicin with DNA unwinding in MCF-7 breast tumor cells. Mol. Pharmacol. 1994, 45, 649–656. [Google Scholar]
- Tacar, O.; Sriamornsak, P.; Das, C.R. Doxorubicin: An update on anticancer molecular action, toxicity and novel drug delivery systems. J. Toxicol. Pharmacol. 2013, 65, 157–170. [Google Scholar] [CrossRef]
- Kleiner, D.E.; Stetler-Stevenson, W.G. Quantitative zymography: detection of picogram quantities of gelatinases. Anal. Biochem. 1994, 218, 325–329. [Google Scholar] [CrossRef]
- Corada, M.; Mariotti, M.; Thurston, G.; Smith, K.; Kunkel, R.; Brockhaus, M.; Lampugnani, M.G.; Martin-Padura, I.; Stoppacciaro, A.; Ruco, L.; et al. Vascular endothelial–cadherin is an important determinant of microvascular integrity in vivo. Proc. Natl. Acad. Sci. USA 1999, 96, 9815–9820. [Google Scholar] [CrossRef]
- Hersel, U.; Dahmen, C.; Kessler, H. RGD modified polymers: Biomaterials for stimulated cell adhesion and beyond. Biomaterials 2003, 24, 4385–4415. [Google Scholar] [CrossRef]
- Feder-Mengus, C.; Ghosh, S.; Reschner, A.; Martin, I.; Spagnoli, G.C. New dimensions in tumor immunology: What does 3D culture reveal? Trends Mol. Med. 2008, 14, 333–340. [Google Scholar] [CrossRef]
- Horning, J.L.; Sahoo, S.K.; Vijayaraghavalu, S. 3D tumor model for in vitro evaluation of anticancer drugs. Mol. Pharmacol. 2008, 5, 849–862. [Google Scholar] [CrossRef]
- Park, C.C.; Bissell, M.J.; Hoff, M.H.B. The influence of the microenvironment on the malignant phenotype. Mol. Med. Today 2000, 6, 324–329. [Google Scholar] [CrossRef]
- Fischbach, C.; Chen, R.; Polverini, P.J.; Mooney, D.J. Engineering tumors with 3D scaffolds. Nat. Methods 2007, 4, 855–860. [Google Scholar] [CrossRef]
- Nirmalanandhan, V.S.; Duren, A.P.; Vielhauer, H.G.; Sittampalam, G.S. Activity of anticancer agents in a three-dimensional cell culture model. Assay Drug Dev. Technol. 2010, 8, 581–590. [Google Scholar] [CrossRef]
- Quanwen, L.; Albert, B.C.; Mattingly, R.R. Three-Dimensional Overlay Culture Models of Human Breast Cancer Reveal a Critical Sensitivity to Mitogen-Activated Protein Kinase Kinase Inhibitors. J. Pharmacol. Exp. Ther. 2010, 332, 821–828. [Google Scholar]
- Starzec, A.; Briane, D.; Kraemer, N.; Kouyoumdjian, J.C.; Moretti, J.L.; Beaupain, R.; Oudar, O. Spatial organization of three-dimensional co-cultures of adriamycin-sensitive and -resistant human breast cancer MCF-7 cells. Biol. Cell 2003, 95, 257–264. [Google Scholar] [CrossRef]
- Hongisto, V.; Fey, V.; Mpindi, J.P. High-Throughput 3D Screening Reveals Differences in Drug Sensitivities between Culture Models of JIMT1 Breast Cancer Cells. PLoS ONE 2013, 8, e77232. [Google Scholar] [CrossRef]
- Weaver, V.M.; Lelievre, S.; Lakins, J.N.; Chrenek, M.A.; Jones, J.C.; Giancotti, F.; Werb, Z.; Bissell, M.J. β4integrin-dependent formation of polarized three-dimensional architecture confers resistance to apoptosis in normal and malignant mammary epithelium. Cancer Cell 2002, 2, 205–216. [Google Scholar] [CrossRef]
- Salani, B.; Rio, A.D.; Massollo, M. Metformin impairs glucose consumption and survival in calu 1 cells by direct inhibition of Hexokinase-II. Sci. Rep. 2013, 3, 2070. [Google Scholar] [CrossRef]
- Warburg, O. On the origin of cancer cells. Science 1956, 123, 309–311. [Google Scholar] [CrossRef]
- Weber, S.; Wolf, K. Two changes of the same nucleotide confer resistance to diuron and antimycin in the mitochondrial cytochrome b gene of Schizosaccharomyces pombe. FEBS Lett. 1977, 237, 31–34. [Google Scholar] [CrossRef]
- Koukourakis, G.; Kelekis, N.; Armonis, V.; Kouloulias, V. Brachytherapy for prostate cancer: A systematic review. Adv. Urol. 2009, 2009, 327945. [Google Scholar] [CrossRef]
- McCleland, S.E.; Marini, C.; Ravera, S.; Maggi, D.; Sambuceti, G. Replication stress links structural and numerical cancer chromosomal instability. Nature 2013, 494, 492–496. [Google Scholar]
- Walenta, S.; Schroeder, T.; Mueller-Klieser, W. Lactate in solid malignant tumors: Potential basis of a metabolic classification in clinical oncology. Curr. Med. Chem. 2004, 11, 2195–2204. [Google Scholar] [CrossRef]
- Walenta, S.; Wetterling, M.; Lehrke, M. High lactate levels predict likelihood of metastases, tumor recurrence, and restricted patient survival in human cervical cancers. Cancer Res. 2000, 60, 916–921. [Google Scholar]
- Chitcholtan, K.; Sykes, P.H.; Evans, J.J. The resistance of intracellular mediators to doxorubicin and cisplatin are distinct in 3D and 2D endometrial cancer. J. Transl. Med. 2012, 10, 38. [Google Scholar] [CrossRef]
- Bauvois, B. New facets of matrix metalloproteinases MMP-2 and MMP-9 as cell surface transducers: Outside-in signaling and relationship to tumor progression. Biochim. Biophys. Acta 2012, 1825, 29–36. [Google Scholar] [CrossRef]
- Decock, J.; Hendrickx, W.; Vanleeuw, U.; Van, B.V.; Van, H.S.; Christiaens, M.R.; Ye, S.; Paridaens, R. Plasma MMP1 and MMP8 expression in breast cancer: Protective role of MMP8 against lymph node metastasis. BMC Cancer 2008, 20, 8–77. [Google Scholar]
- Patel, S.; Sumitra, G.; Koner, B.C.; Saxena, A. Role of serum matrix metalloproteinase 2 and 9 to predict breast cancer progression. Clin. Biochem. 2011, 44, 869–872. [Google Scholar] [CrossRef]
- Hall, A. The cytoskeleton and cancer. Cancer Metastasis Rev. 2009, 28, 5–14. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Condeelis, J. Regulation of the actin cytoskeleton in cancer cell migration and invasion. Biochim. Biophys. Acta 2007, 1773, 642–652. [Google Scholar] [CrossRef]
- Stehn, J.R.; Nikolas, K.H.; Teresa, B. A Novel class of anticancer compounds targets the actin cytoskeleton in tumor cells. Cancer Res. 2013, 73, 5169–5182. [Google Scholar] [CrossRef]
- Bousquet, P.F.; Paulsen, L.A.; Fondy, C.; Lipski, K.M.; Loucy, K.J.; Fondy, T.P. Effects of cytochalasin B in culture and in vivo on murine Madison 109 lung carcinoma and on B16 melanoma. Cancer Res. 1990, 50, 1431–1439. [Google Scholar]
- Senderowicz, M.J. Jasplakinolide’s Inhibition of the Growth of Prostate Carcinoma cells In vitro with disruption of the actin cytoskeleton. J. Natl. Cancer Inst. 1995, 87, 46–51. [Google Scholar] [CrossRef]
- Barkan, D.; Justin, L.S.; Kamaraju, A.K.; Mark, J.; Cho, E.C.; Lockett, S.; Khanna, C.; Chambers, A.F.; Green, J.E. Inhibition of Metastatic Outgrowth from Single Dormant tumor Cells by targeting the Cytoskeleton. Cancer Res. 2008, 68, 6241–6250. [Google Scholar] [CrossRef]

| Drug | IC 50 (μg/mL) | Fold Change in IC50 | |
|---|---|---|---|
| 2D Monolayer | 3D Model | ||
| Cyclosaplin | 16.8 6 ± 0.09 | 89.27 ± 0.2 | 5.3 |
| Doxorubicin | 2.8 ± 0.06 | 16.44 ± 0.1 | 5.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mishra, A.; Mukhopadhyay, S.K.; Dey, S. Evaluation of Cyclosaplin Efficacy Using a Silk Based 3D Tumor Model. Biomolecules 2019, 9, 123. https://doi.org/10.3390/biom9040123
Mishra A, Mukhopadhyay SK, Dey S. Evaluation of Cyclosaplin Efficacy Using a Silk Based 3D Tumor Model. Biomolecules. 2019; 9(4):123. https://doi.org/10.3390/biom9040123
Chicago/Turabian StyleMishra, Abheepsa, Sourav K. Mukhopadhyay, and Satyahari Dey. 2019. "Evaluation of Cyclosaplin Efficacy Using a Silk Based 3D Tumor Model" Biomolecules 9, no. 4: 123. https://doi.org/10.3390/biom9040123
APA StyleMishra, A., Mukhopadhyay, S. K., & Dey, S. (2019). Evaluation of Cyclosaplin Efficacy Using a Silk Based 3D Tumor Model. Biomolecules, 9(4), 123. https://doi.org/10.3390/biom9040123