Molecular Antioxidant Properties and In Vitro Cell Toxicity of the p-Aminobenzoic Acid (PABA) Functionalized Peptide Dendrimers §
Abstract
:1. Introduction
2. Materials and Methods
2.1. General Procedures
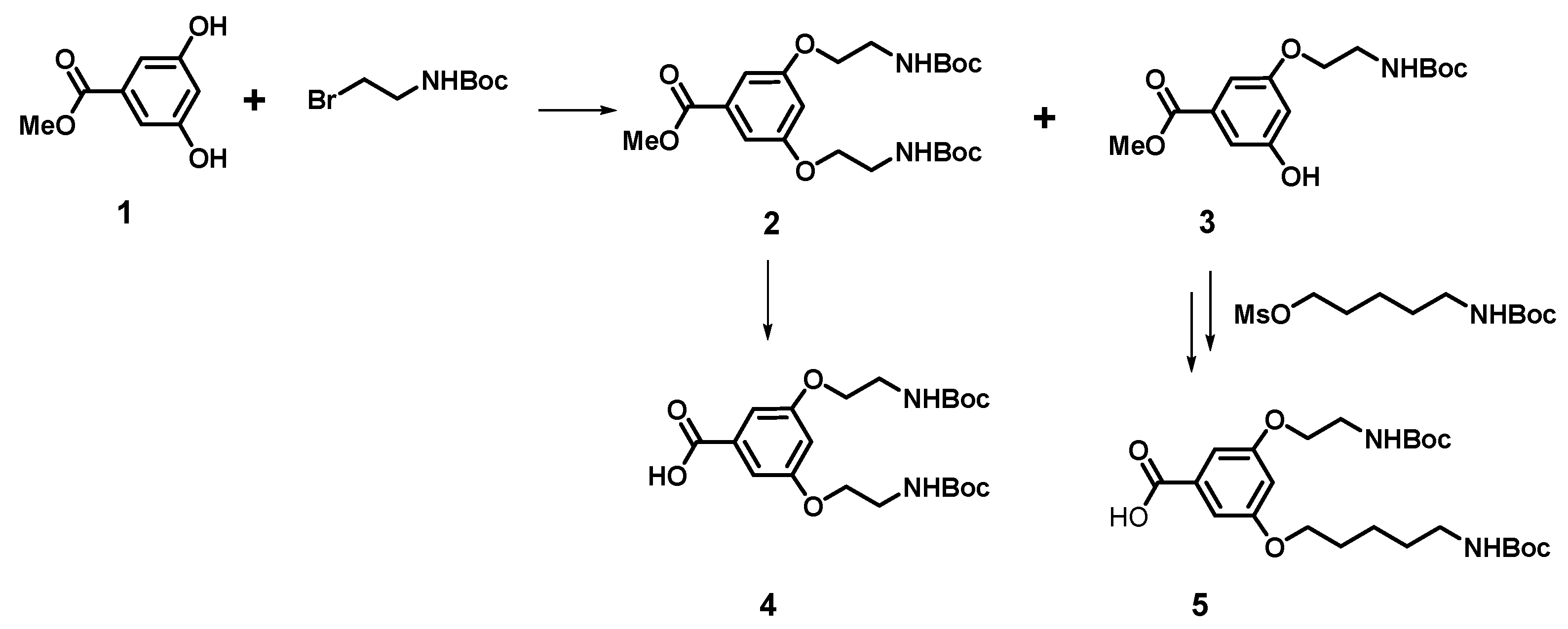
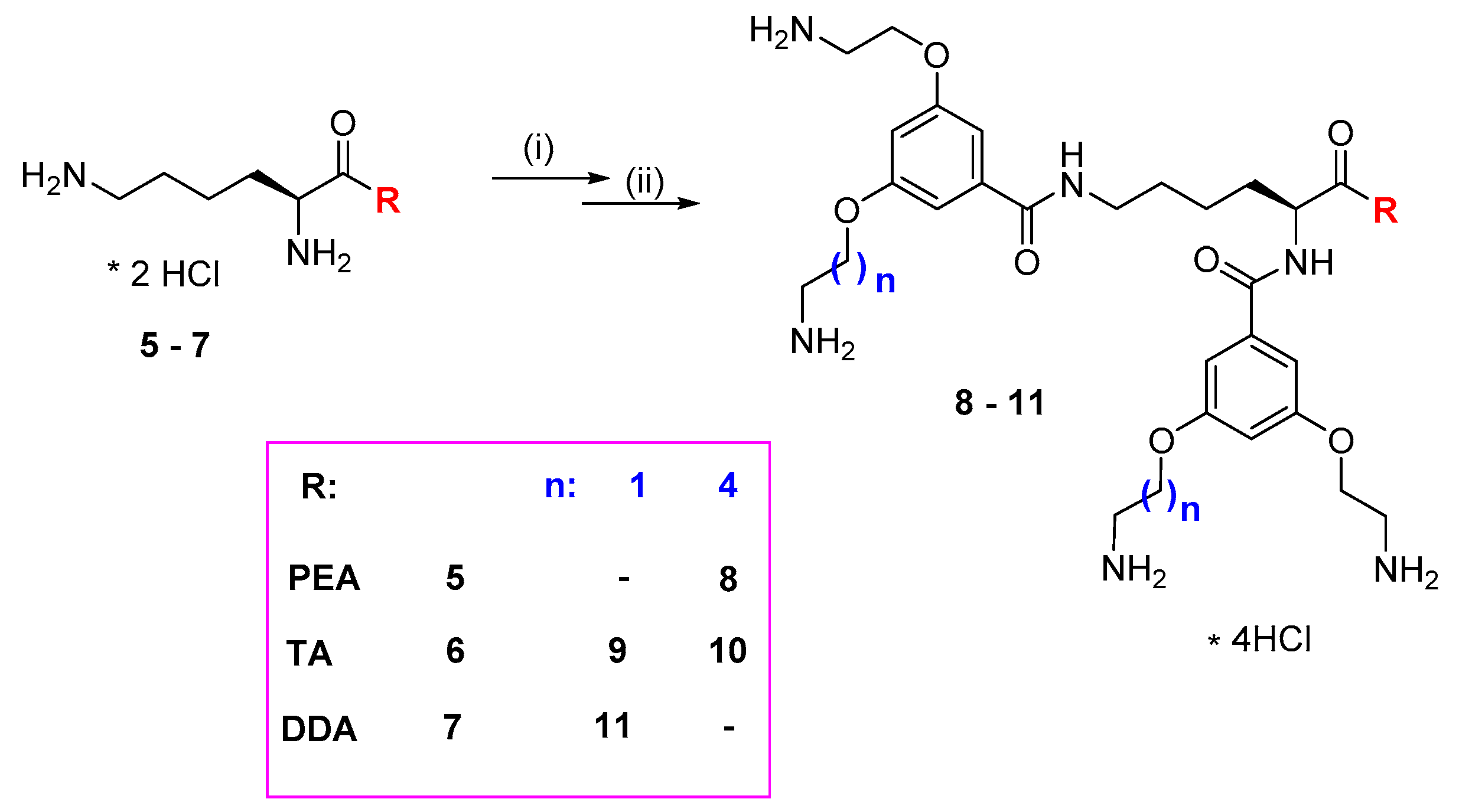
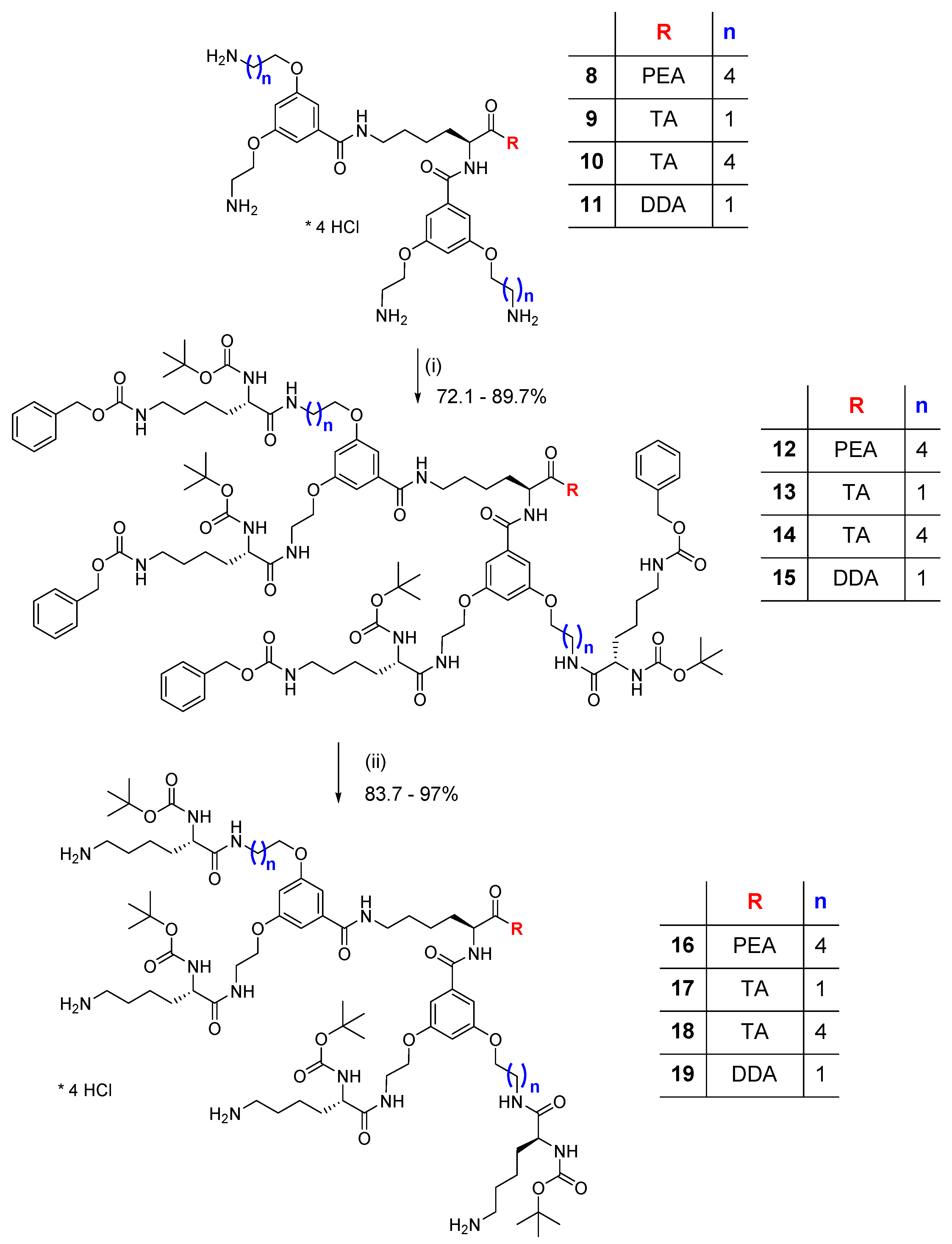
2.2. Synthesis and Characterization of p-Aminobenzoic (PABA)-Terminated Dendrimers
2.3. Spectrocscopic Data of New Dendrimers (16–27)
2.3.1. Dendrimer 16 (Obtained from 12)
2.3.2. Dendrimer 17 (Obtained from 13)
2.3.3. Dendrimer 18 (Obtained from 14)
2.3.4. Dendrimer 19 (Obtained from 15)
2.3.5. Dendrimer 20 (Obtained from 16)
2.3.6. Dendrimer 21 (Obtained from 17)
2.3.7. Dendrimer 22 (Obtained from 18)
2.3.8. Dendrimer 23 (Obtained from 19)
2.3.9. Boc-Deprotected Dendrimers
2.3.10. Dendrimer 24 (Obtained from 20)
2.3.11. Dendrimer 25 (Obtained from 21)
2.3.12. Dendrimer 26 (Obtained from 22)
2.3.13. Dendrimer 27 (Obtained from 24)
2.4. Primary Cultures of Cerebellar Granule Cells (CGC)
2.4.1. Cerebellar Granule Cells Viability
2.4.2. Reactive Oxygen Species (ROS) Measurement
2.5. Cell Proliferation Assays
2.5.1. DPPH Assay
2.5.2. ABTS Assay
3. Results
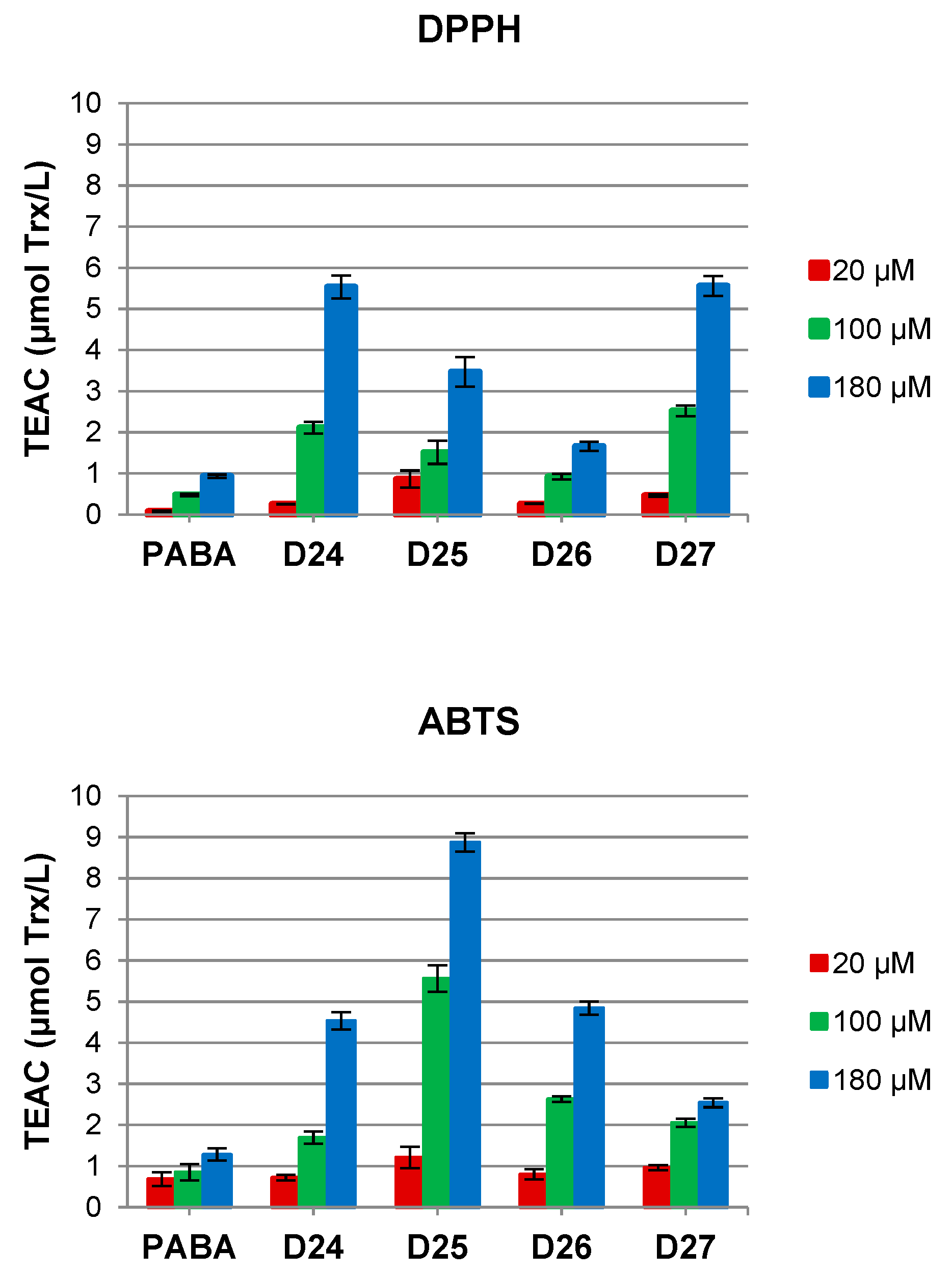
3.1. Antioxidant Properties—Neutral (DPPH) and Cationic Radicals (ABTS) Quenching Potency
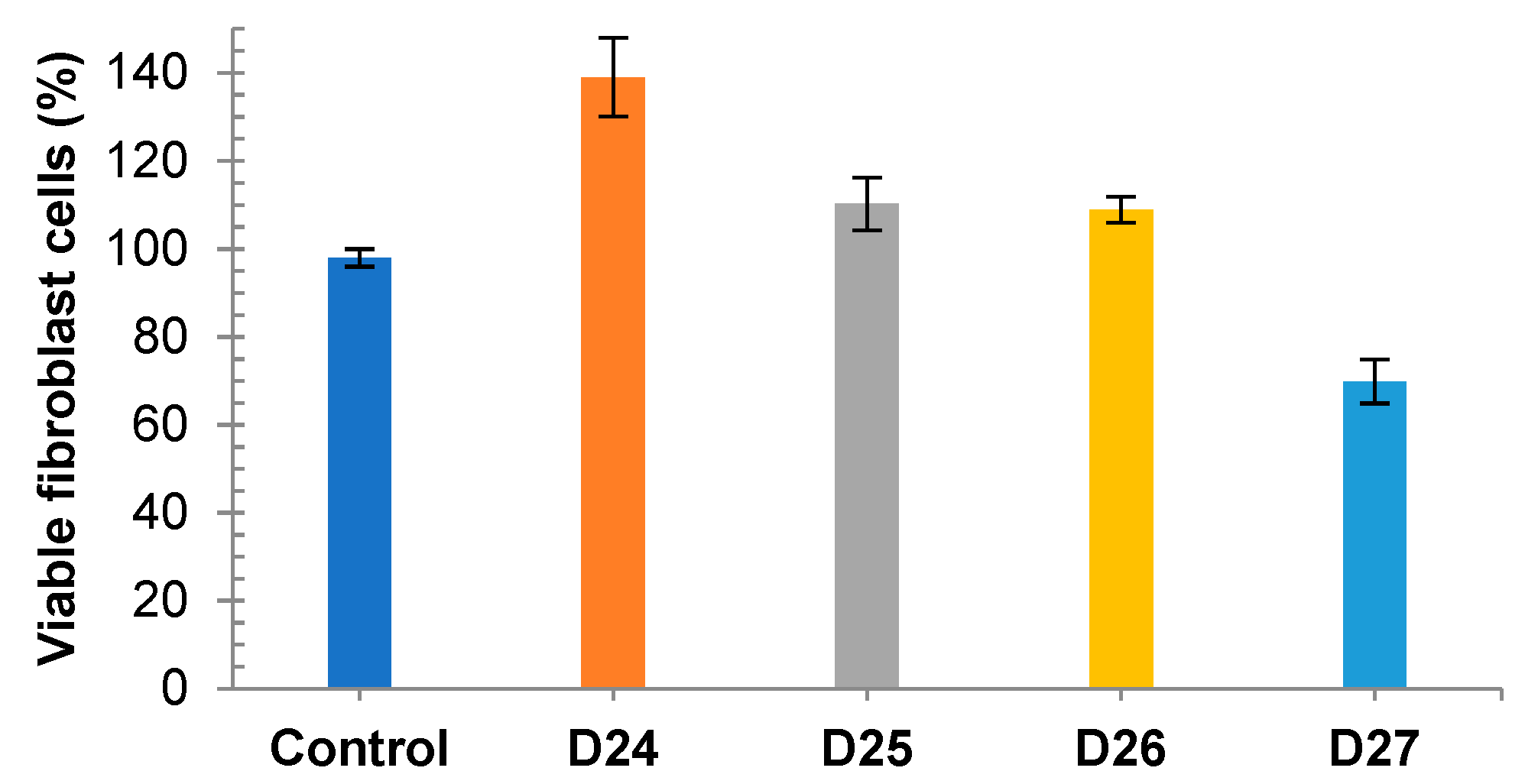
3.2. Cytotoxicity on Melanoma and Fibroblast Cell Lines
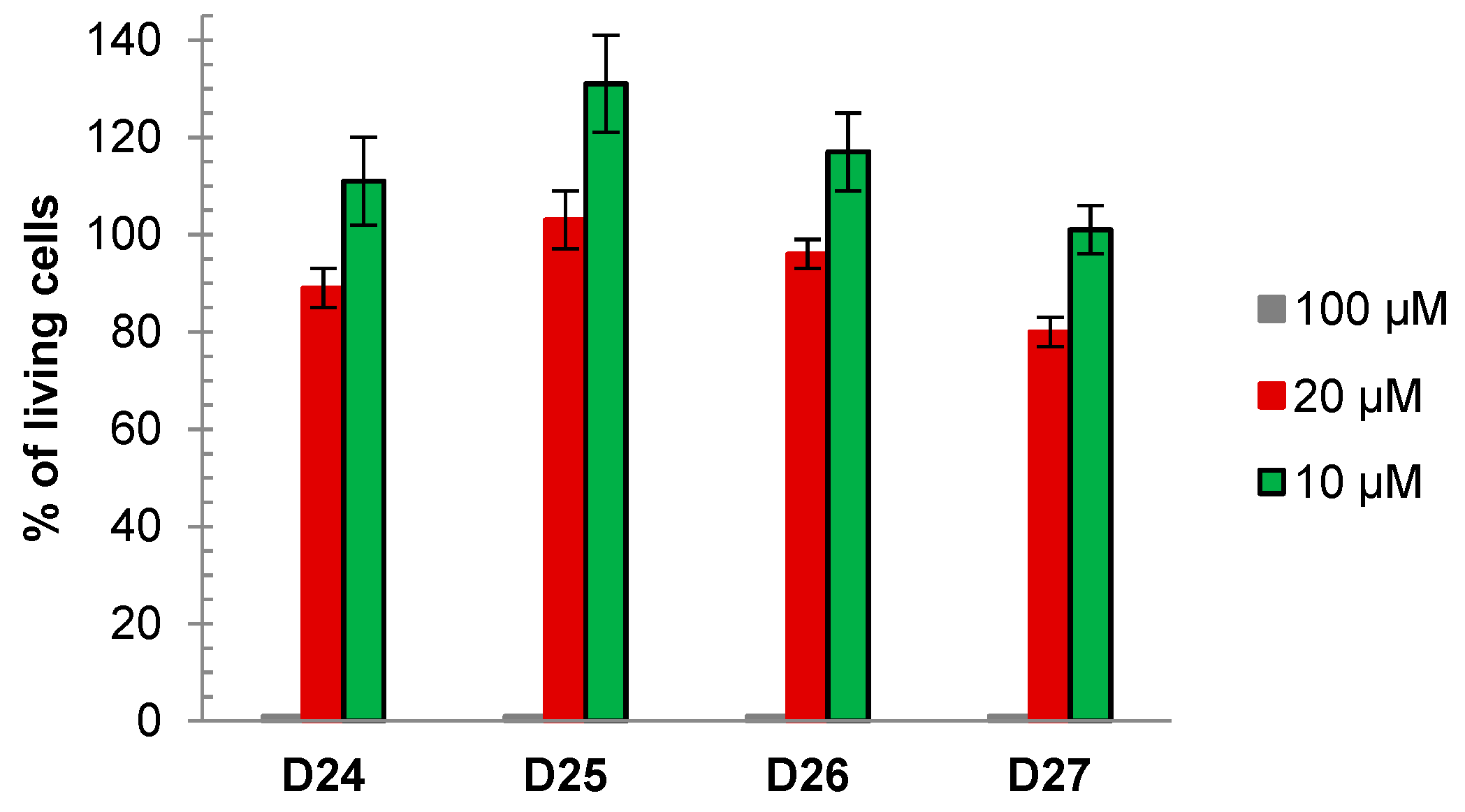
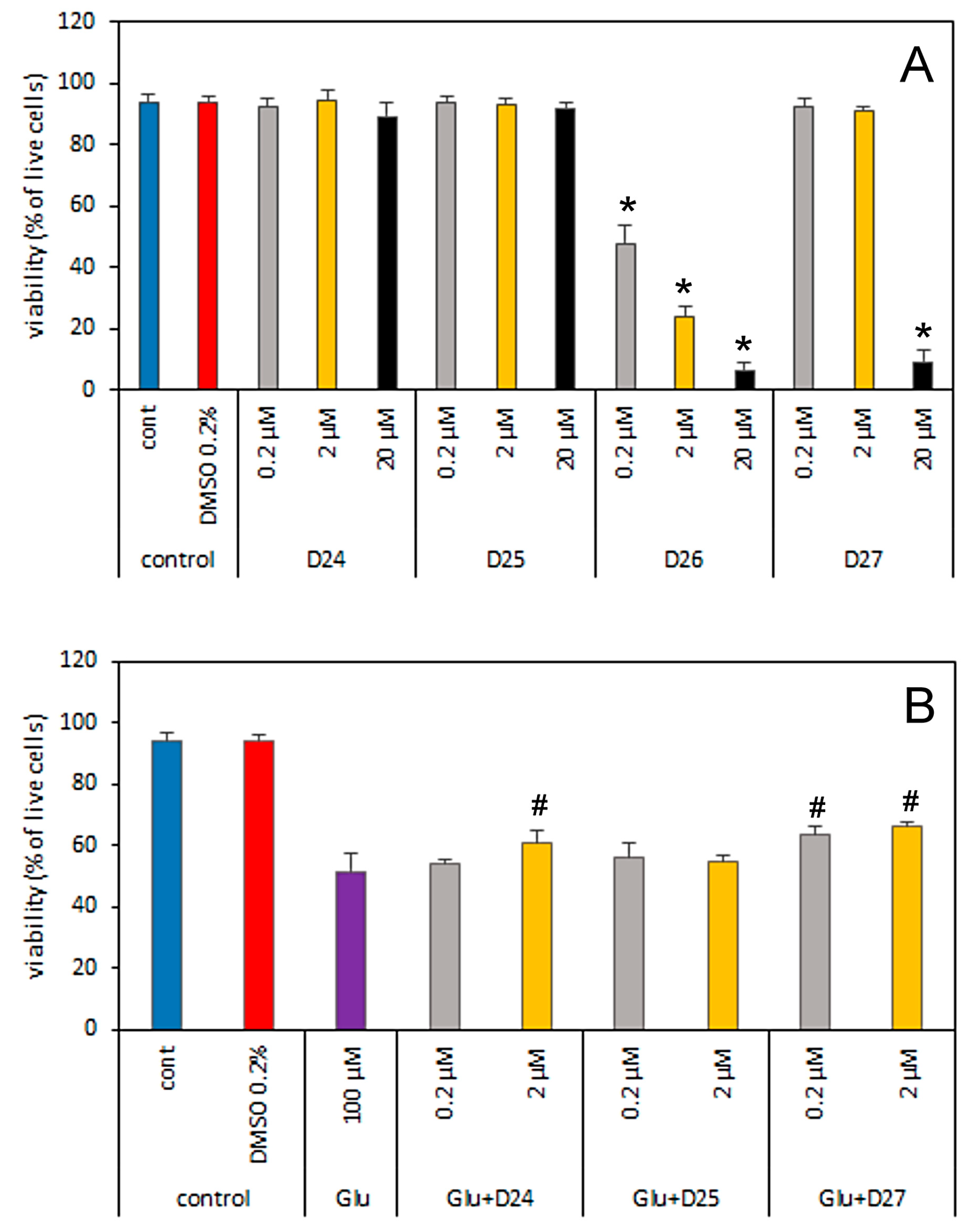
3.3. Toxicity of Dendrimers on Primary Cultures of Cerebellar Granule Cells
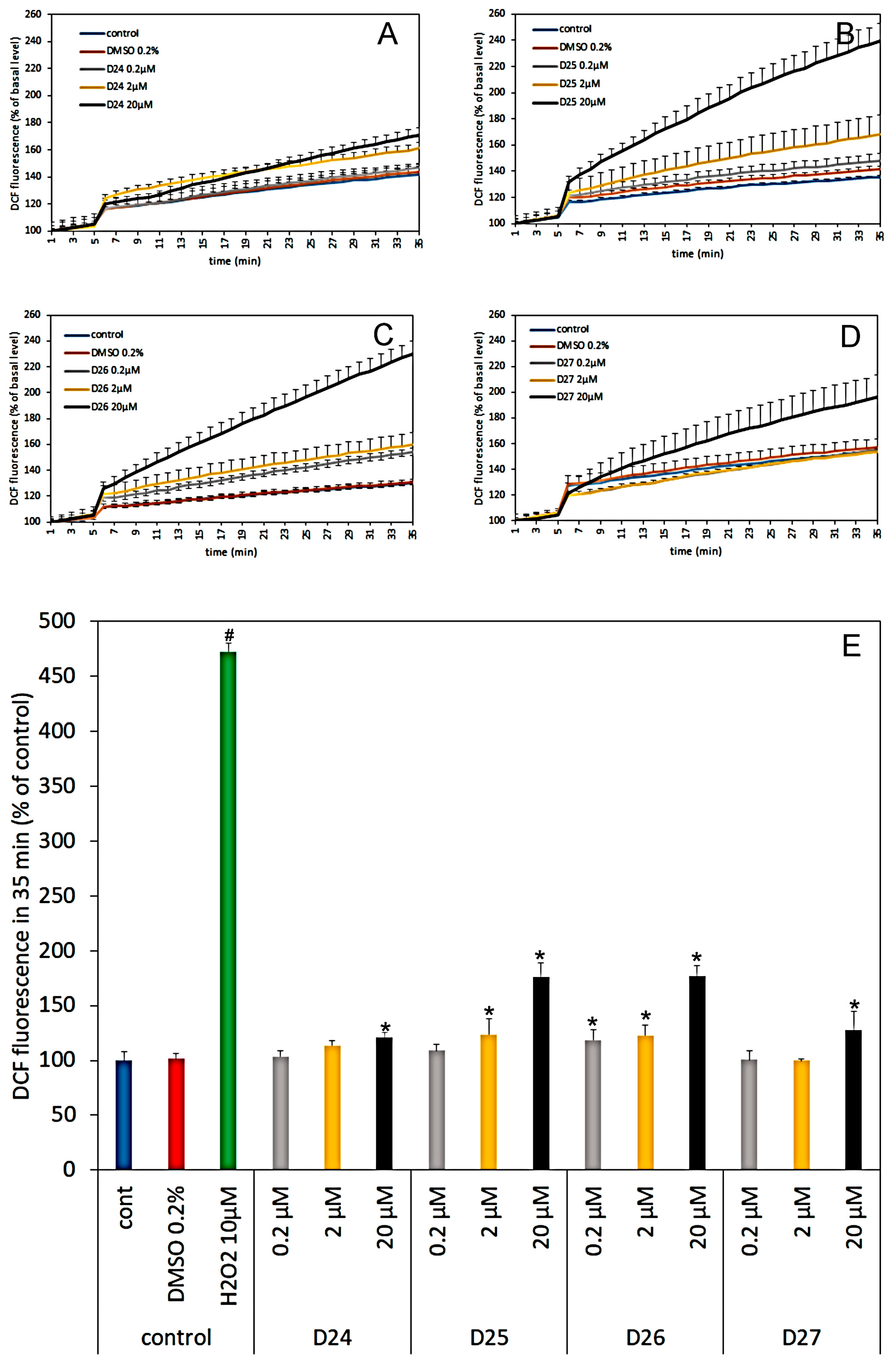
3.4. Effect of Dendrimers on the Reactive Oxygen Species Production in Cerebral Granule Cells Cultures
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Tomalia, D.A.; Baker, H.; Dewald, J.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P.A. New Class of Polymers: Starburst-Dendritic Macromolecules. Polym. J. 1985, 17, 117–132. [Google Scholar] [CrossRef]
- Kannan, R.M.; Nance, E.; Kannan, S.; Tomalia, D.A. Emerging concepts in dendrimer-based nanomedicine: From design principles to clinical applications. J. Intern. Med. 2014, 276, 579–617. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Outschoorn, U.E.; Peiris-Pages, M.; Pestell, R.G.; Sotgia, F.; Lisanti, M.P. Cancer metabolism: A therapeutic perspective. Nat. Rev. Clin. Oncol. 2017, 14, 11–31. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.M.; Salgado, A.J.; Sousa, N.; Mano, J.F.; Reis, R.L. Dendrimers and derivatives as a potential therapeutic tool in regenerative medicine strategies—A review. Prog. Polym. Sci. 2010, 35, 1163–1194. [Google Scholar] [CrossRef]
- Sun, L.; Li, X.; Wei, X.; Luo, Q.; Guan, P.; Wu, M.; Zhu, H.; Luo, K.; Gong, Q. Stimuli-Responsive Biodegradable Hyperbranched Polymer-Gadolinium Conjugates as Efficient and Biocompatible Nanoscale Magnetic Resonance Imaging Contrast Agents. ACS Appl. Mater. Interfaces 2016, 8, 10499–10512. [Google Scholar] [CrossRef]
- Ke, W.; Shao, K.; Huang, R.; Han, L.; Liu, Y.; Li, J.; Kuang, Y.; Ye, L.; Lou, J.; Jiang, C. Gene delivery targeted to the brain using an Angiopep-conjugated polyethyleneglycol-modified polyamidoamine dendrimer. Biomaterials 2009, 30, 6976–6985. [Google Scholar] [CrossRef]
- Li, Y.; He, H.; Jia, X.; Lu, W.L.; Lou, J.; Wei, Y. A dual-targeting nanocarrier based on poly(amidoamine) dendrimers conjugated with transferrin and tamoxifen for treating brain gliomas. Biomaterials 2012, 33, 3899–3908. [Google Scholar] [CrossRef]
- Yam, J.C.; Kwok, A.K. Ultraviolet light and ocular diseases. Int. Ophthalmol. 2014, 34, 383–400. [Google Scholar] [CrossRef]
- Nikitaki, Z.; Hellweg, C.E.; Georgakilas, A.G.; Ravanat, J.L. Stress-induced DNA damage biomarkers: Applications and limitations. Front. Chem. 2015, 3, 35. [Google Scholar] [CrossRef]
- Canto, C.; Menzies, K.J.; Auwerx, J. NAD(+) Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metab. 2015, 22, 31–53. [Google Scholar] [CrossRef]
- Li, H.; Horke, S.; Forstermann, U. Vascular oxidative stress, nitric oxide and atherosclerosis. Atherosclerosis 2014, 237, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Panieri, E.; Santoro, M.M. ROS homeostasis and metabolism: A dangerous liason in cancer cells. Cell Death Dis. 2016, 7, e2253. [Google Scholar] [CrossRef] [PubMed]
- Gach, K.; Dlugosz, A.; Janecka, A. The role of oxidative stress in anticancer activity of sesquiterpene lactones. Naunyn Schmiedebergs Arch. Pharmacol. 2015, 388, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Kim, J.H.; Gupta, S.C.; Aggarwal, B.B. Targeting death receptors for TRAIL by agents designed by Mother Nature. Trends Pharmacol. Sci. 2014, 35, 520–536. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Chang, H.; Li, H.; Wang, S. Induction of reactive oxygen species: An emerging approach for cancer therapy. Apoptosis 2017, 22, 1321–1335. [Google Scholar] [CrossRef] [PubMed]
- Akberova, S.Y. New biological properties of p-aminobenzoic acid. Biol. Bull. 2002, 29, 390–393. [Google Scholar] [CrossRef]
- LeBlanc, J.G.; Milani, C.; de Giori, G.S.; Sesma, F.; van Sinderen, D.; Ventura, M. Bacteria as vitamin suppliers to their host: A gut microbiota perspective. Curr. Opin. Biotechnol. 2013, 24, 160–168. [Google Scholar] [CrossRef]
- Low, P.S.; Henne, W.A.; Doorneweerd, D.D. Discovery and Development of Folic-Acid-Based Receptor Targeting for Imaging and Therapy of Cancer and Inflammatory Diseases. Acc. Chem. Res. 2008, 41, 20–129. [Google Scholar] [CrossRef]
- Song, G.C.; Choi, H.K.; Ryu, C.M. The folate precursor para-aminobenzoic acid elicits induced resistance against Cucumber mosaic virus and Xanthomonas axonopodis. Ann. Bot. 2013, 111, 925–934. [Google Scholar] [CrossRef]
- Güçlü, K.; Kıbrıslıoğlu, G.; Özyürek, M.; Apak, R. Development of a Fluorescent Probe for Measurement of Peroxyl Radical Scavenging Activity in Biological Samples. J. Agric. Food Chem. 2014, 62, 1839–1845. [Google Scholar] [CrossRef]
- Sowińska, M.; Laskowska, A.; Guśpiel, A.; Solecka, J.; Bochynska-Czyż, M.; Lipkowski, A.W.; Trzeciak, K.; Urbanczyk-Lipkowska, Z. Bioinspired Amphiphilic Peptide Dendrimers as Specific and Effective Compounds against Drug Resistant Clinical Isolates of E. coli. Bioconjugate Chem. 2018, 29, 3571–3585. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, A.J.; Liskamp, R.M.J. Synthesis of Novel Dendrimeric Systems Containing NLO Ligands. Eur. J. Org. Chem. 2005, 487–495. [Google Scholar] [CrossRef]
- Zieminska, E.; Ruszczynska, A.; Lazarewicz, J.W. Tetrabromobisphenol A disturbs zinc homeostasis in cultured cerebellar granule cells: A dual role in neurotoxicity. Food Chem. Toxicol. 2017, 109, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Lenart, J.; Zieminska, E.; Diamandakis, D.; Lazarewicz, J.W. Altered expression of genes involved in programmed cell death in primary cultured rat cerebellar granule cells acutely challenged with tetrabromobisphenol A. Neurotoxicology 2017, 63, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Zieminska, E.; Lenart, J.; Lazarewicz, J.W. Select putative neurodevelopmental toxins modify SNAP-25 expression in primary cultures of rat cerebellar granule cells. Toxicology 2016, 370, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Bickers, D.R.; Athar, M. Oxidative Stress in the Pathogenesis of Skin Disease. J. Investig. Dermatol. 2006, 126, 2565–2575. [Google Scholar] [CrossRef]
- Błoch-Mechkour, A.; Bally, T.; Sikora, A.; Michalski, R.; Marcinek, A.; Gebicki, J. Radicals and Radical Ions Derived from Indole, Indole-3-carbinol and Diindolylmethane. J. Phys. Chem. A 2010, 114, 6787–6794. [Google Scholar] [CrossRef]
- Susan, E.; Walden, S.E.; Wheeler, R.A. Distinguishing Features of Indolyl Radical and Radical Cation: Implications for Tryptophan Radical Studies. J. Phys. Chem. 1996, 100, 1530–1535. [Google Scholar] [CrossRef]
- Ziemińska, E.; Stafiej, A.; Toczyłowska, B.; Lazarewicz, J.W. Synergistic neurotoxicity of oxygen-glucose deprivation and tetrabromobisphenol A in vitro: Role of oxidative stress. Pharmacol. Rep. 2012, 64, 1166–1178. [Google Scholar] [CrossRef]
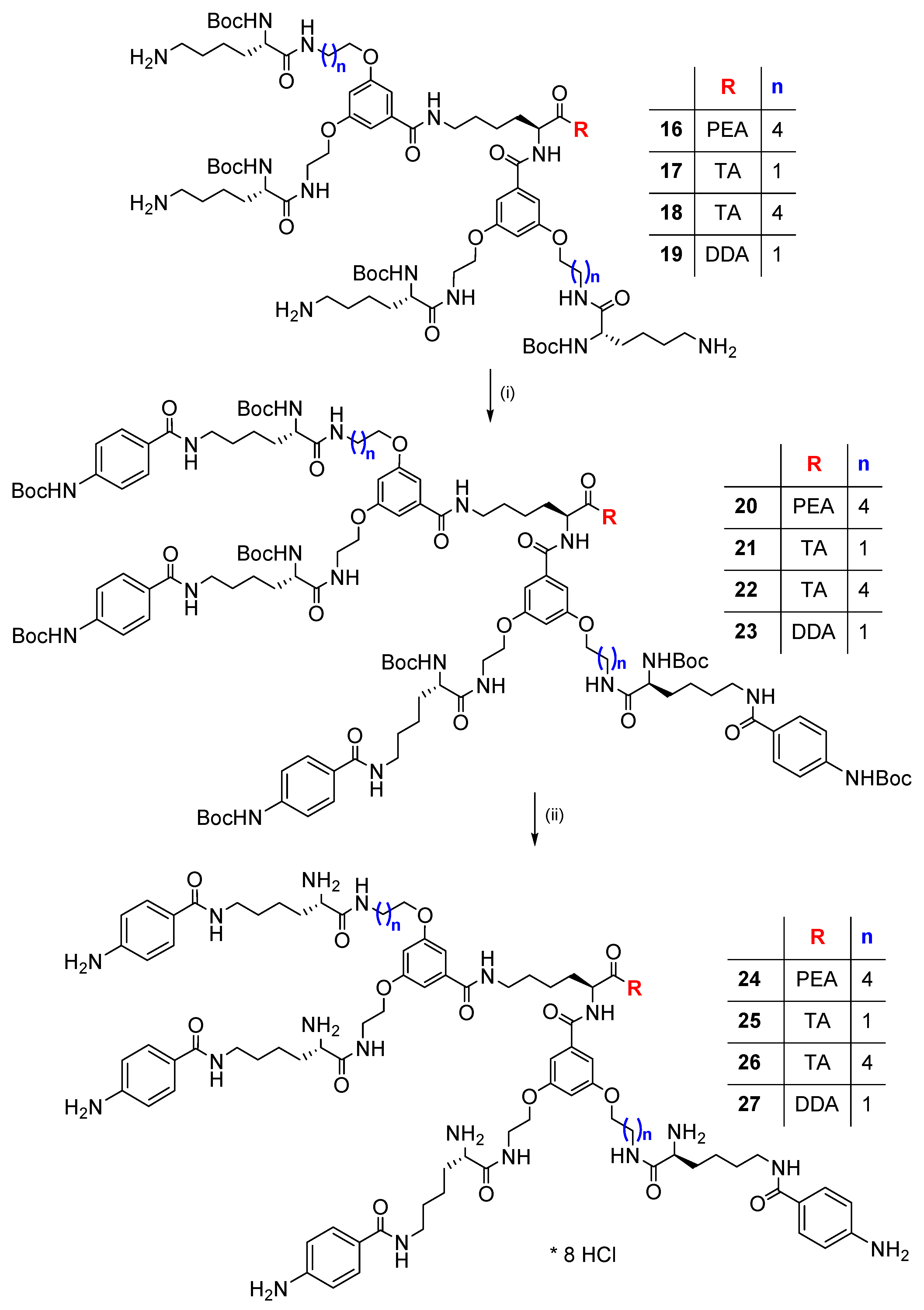
| Protected Dendrimers 20–23 | Deprotected Dendrimers 24–27 | |||||||
|---|---|---|---|---|---|---|---|---|
| No. | Deprotection Yield (%) | M.p. (°C) | [α]D25 (c 1, MeOH) | Rf (CHCl3/MeOH 8:1) | No. | Deprotection yield (%) | M.p. (°C) | [α]D25 (c 1, MeOH) |
| 20 | 60.0 a | 144–150 | −12.7 | 0.38 | 24 | 91.3 | 175–191 | −1.9 |
| 21 | 39.2 a | 150–160 | −14.7 | 0.38 | 25 | 91.7 | 200–211 | −5.5 |
| 22 | 50.0 a | 149–155 | −9.9 | 0.44 | 26 | 96.8 | 189–196 | +0.06 |
| 23 | 39.1 b | 142–149 | −12.0 | 0.77 | 27 | 97.7 | 182–199 | − 4.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sowinska, M.; Morawiak, M.; Bochyńska-Czyż, M.; Lipkowski, A.W.; Ziemińska, E.; Zabłocka, B.; Urbanczyk-Lipkowska, Z.
Molecular Antioxidant Properties and In Vitro Cell Toxicity of the p-Aminobenzoic Acid (PABA) Functionalized Peptide Dendrimers
Sowinska M, Morawiak M, Bochyńska-Czyż M, Lipkowski AW, Ziemińska E, Zabłocka B, Urbanczyk-Lipkowska Z.
Molecular Antioxidant Properties and In Vitro Cell Toxicity of the p-Aminobenzoic Acid (PABA) Functionalized Peptide Dendrimers
Sowinska, Marta, Maja Morawiak, Marta Bochyńska-Czyż, Andrzej W. Lipkowski, Elżbieta Ziemińska, Barbara Zabłocka, and Zofia Urbanczyk-Lipkowska.
2019. "Molecular Antioxidant Properties and In Vitro Cell Toxicity of the p-Aminobenzoic Acid (PABA) Functionalized Peptide Dendrimers
Sowinska, M., Morawiak, M., Bochyńska-Czyż, M., Lipkowski, A. W., Ziemińska, E., Zabłocka, B., & Urbanczyk-Lipkowska, Z.
(2019). Molecular Antioxidant Properties and In Vitro Cell Toxicity of the p-Aminobenzoic Acid (PABA) Functionalized Peptide Dendrimers




