Herbal Extracts Incorporated into Shortbread Cookies: Impact on Color and Fat Quality of the Cookies
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.2. Shortbread Cookies Preparation
2.3. Fatty Acid Analysis
2.4. Color Measurement
2.5. Sensory Evaluation
2.6. Fat Extraction
2.7. Chemical Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Color Measurement
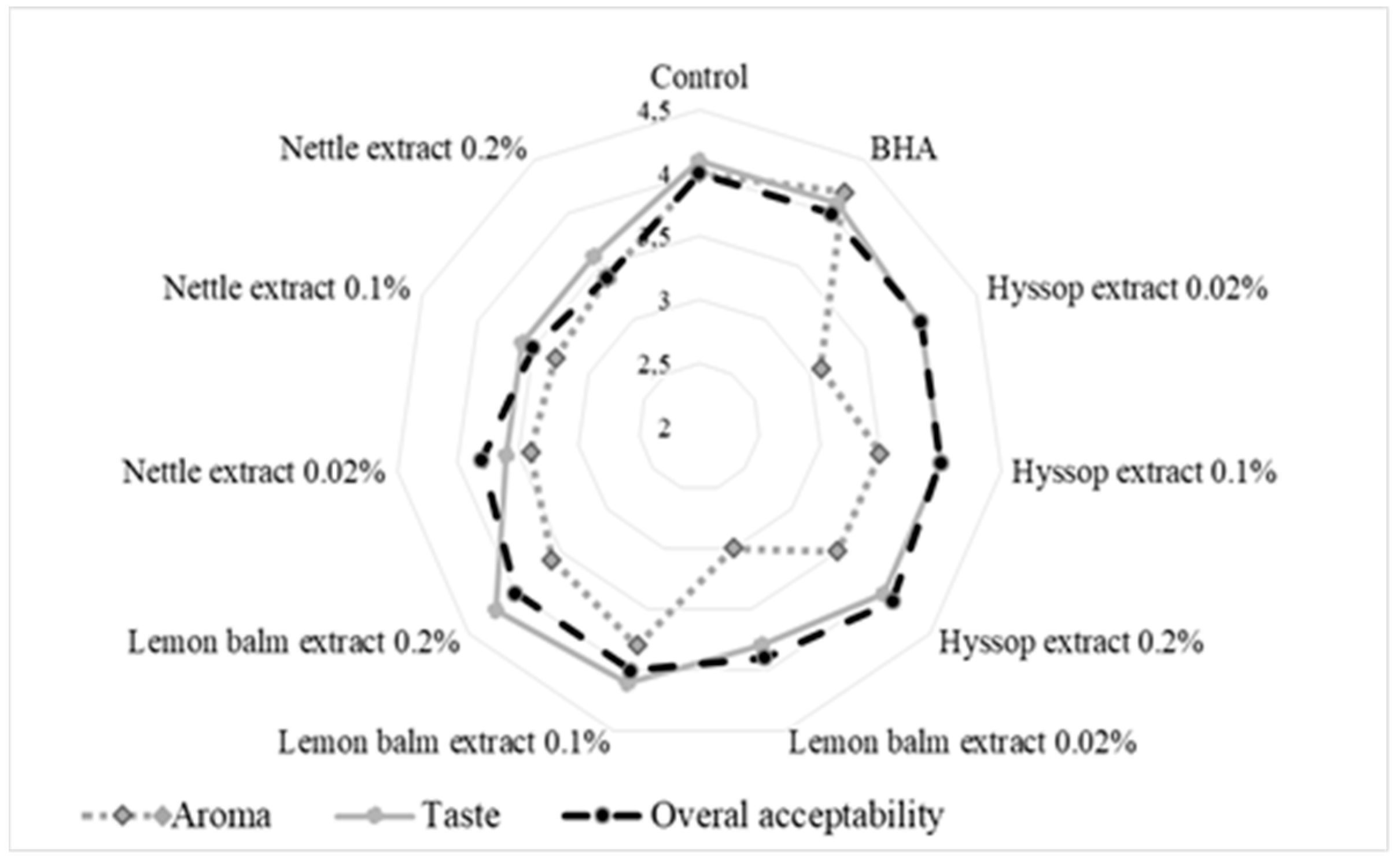
3.2. Sensory Evaluation
3.3. Chemical Analysis
3.3.1. Changes in Peroxide (PV) and p-Anisidine (p-AnV) Values
3.3.2. Specific Extinction Coefficients Measurement
3.3.3. Antioxidant Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zbikowska, A.; Kowalska, M.; Pieniowska, J. Assessment of shortcrust biscuits with reduced fat content of microcrystalline cellulose and psyllium as fat replacements. J. Food Process. Preserv. 2018, 42, e13675. [Google Scholar] [CrossRef]
- Manohar, R.S.; Rao, P.H. Effect of emulsifiers, fat level and type on the rheological characteristics of biscuit dough and quality of biscuits. J. Sci. Food Agric. 1999, 79, 1223–1231. [Google Scholar] [CrossRef]
- Onacik-Gür, S.; Żbikowska, A.; Jaroszewska, A. Effect of high-oleic sunflower oil and other pro-health ingredients on physical and sensory properties of biscuits. CyTA–J. Food 2015, 13, 621–628. [Google Scholar] [CrossRef]
- Marconi, O.; Martini, R.; Mangione, A.; Falconi, C.; Pepe, C.; Perretti, G. Palatability and stability of shortbread made with low saturated fat content. J. Food Sci. 2014, 79, C469–C475. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S.; Ribeiro-Santos, R.; Ramos, F.; Castilho, M.C.; Sanches-Silva, A. UHPLC-DAD multi-method for determination of phenolic in aromatic palnts. Food Anal. Methods 2018, 11, 440–450. [Google Scholar] [CrossRef]
- Babović, N.; Žižović, I.; Saićić, S.; Ivanović, J.; Petrović, S. Oxidative stabilization of sunflower oil by antioxidant fractions from selected Lamiaceae herbs. Chem. Ind. Chem. Eng. Q. 2010, 16, 287–293. [Google Scholar] [CrossRef]
- Ondrejovič, M.; Kraic, F.; Benkovičová, H.; Šilhár, S. Optimization of antioxidant extraction from lemon balm (Melissa officinalis). Czech J. Food Sci. 2012, 30, 385–393. [Google Scholar] [CrossRef]
- Verma, P.P.S.; Singh, A.; Rahaman, L.; Bahl, J.R. Lemon balm (Melissa officinalis L.) an herbal medicinal plant with broad therapeutic uses and cultivation practices: A review. Int. J. Recent Adv. Multidiscip. Res. 2015, 2, 0928–0933. [Google Scholar]
- Moradkhani, H.; Sargsyan, E.; Bibak, H.; Naseri, B.; Sadat-Hosseini, M.; Fayazi-Barjin, A.; Meftahizade, H. Melissa officinalis L., a valuable medicine plant: A review. J. Med. Plants Res. 2010, 4, 2753–2759. [Google Scholar]
- Arceusz, A.; Wesolowski, M. Quality consistency evaluation of Melissa officinalis L. commercial herbs by HPLC fingerprint and quantitation of selected phenolic acids. J. Pharm. Biomed. Anal. 2013, 83, 215–220. [Google Scholar] [CrossRef]
- Carnat, A.P.; Carnat, A.; Fraisse, D.; Lamaison, J.L. The aromatic and polyphenolic composition of lemon balm (Melissa officinalis L. subsp. officinalis) tea. Pharma. Acta Helv. 1998, 72, 301–305. [Google Scholar] [CrossRef]
- Dias, M.I.; Barros, L.; Sousa, M.J.; Ferreira, I.C.F.R. Systematic comparison of nutraceuticals and antioxidant potential of cultivated, in vitro cultured and commercial Melissa officinalis samples. Food Chem. Toxicol. 2012, 50, 1866–1873. [Google Scholar] [CrossRef] [PubMed]
- Murakami, Y.; Omoto, T.; Asai, I.; Shimomura, K.; Yoshihira, K.; Ishimaru, K. Rosmarinic acid and related phenolics in transformed root cultures of Hyssopus officinalis. Plant Cell Tiss. Org. Cult. 1998, 53, 75–78. [Google Scholar] [CrossRef]
- Hatipoğlu, G.; Sökmen, M.; Bektaş, E.; Daferera, D.; Sökmen, A.; Demir, E.; Şahin, H. Automated and standard extraction of antioxidant phenolic compounds of Hyssopus officinalis L. ssp. angustifolius. Ind. Crop. Prod. 2013, 43, 427–433. [Google Scholar] [CrossRef]
- Moro, A.; Zalacain, A.; Hurtado de Mendoza, J.; Carmona, M. Effects of agronomic practices on volatile composition of Hyssopus officinalis L. essential oils. Molecules 2011, 16, 4131–4139. [Google Scholar] [CrossRef]
- Fathiazad, F.; Hamedeyazdan, S. A review on Hyssopus officinalis L.: Composition and biological activities. Afr. J. Pharm. Pharmacol. 2011, 5, 1959–1966. [Google Scholar] [CrossRef]
- Sirvastava, A.; Awasthi, K.; Kumar, B.; Misra, A.; Srivastava, S. Pharmacognostic and pharmacological evaluation of Hyssopus officinalis L. (Lamiaceae) collected from Kashmir Himalayas, India. Pharmacogn. J. 2018, 10, 690–693. [Google Scholar] [CrossRef]
- Vlase, L.; Benedec, D.; Hanganu, D.; Damian, G.; Csillag, I.; Sevaster, B.; Mot, A.C.; Silaghi-Dumitrescu, R.; Tilea, I. Evaluation of antioxidant and antimicrobial activities and phenolic profile for Hyssopus officinalis, Ocimum basilicum and Teucrium chamaedrys. Molecules 2014, 19, 5490–5507. [Google Scholar] [CrossRef]
- Chrpová, D.; Kouřimská, L.; Gordon, M.H.; Heřmanová, V.; Roubičková, I.; Pánek, J. Antioxidant activity of selected phenols and herbs used in diets for medical conditions. Czech J. Food Sci. 2010, 28, 317–325. [Google Scholar] [CrossRef]
- Fernández-López, J.; Sevilla, L.; Sayas-Barberá, E.; Navarro, C.; Marín, F.; Pérez-Alvarez, J.A. Evaluation of the antioxidant potential of hyssop (Hyssopus officinalis L.) and rosemary (Rosmarinus officinalis L.) extracts in cooked pork meat. J. Food Sci. 2003, 68, 660–664. [Google Scholar] [CrossRef]
- Rutto, L.K.; Xu, Y.; Ramirez, E.; Brandt, M. Mineral properties and dietary value of raw and processed stinging nettle (Urtica dioica L.). Int. J. Food Sci. 2013, 2013, 857120. [Google Scholar] [CrossRef] [PubMed]
- Biesiada, A.; Wołoszczak, E.; Sokół-Łętowska, A.; Kucharska, A.Z.; Nawirska-Olszańska, A. The effect of nitrogen form and dose on yield, chemical composition and antioxidant activity of stinging nettle (Urtica dioica L.). Herba Pol. 2009, 55, 84–93. [Google Scholar]
- Hojnik, M.; Škerget, M.; Knez, Ž. Isolation of chlorophylls from stinging nettle (Urtica dioica L.). Sep. Purif. Technol. 2007, 57, 37–46. [Google Scholar] [CrossRef]
- Kregiel, D.; Pawlikowska, E.; Antolak, H. Urtica spp.: Ordinary plants with extraordinary properties. Molecules 2018, 23, 1664. [Google Scholar] [CrossRef] [PubMed]
- Gülçin, I.; Küfrevioğlu, I.; Oktay, M.; Büyükokuroğlu, M.E. Antioxidant, antimicrobial, antiulcer and analgesic activities of nettle (Urtica dioica L.). J. Ethnopharmacol. 2004, 90, 205–215. [Google Scholar] [CrossRef]
- Turhan, S.; Sagir, I.; Temiz, H. Oxidative stability of brined anchovies (Engraulis encrasicholus) with plant extracts. Int. J. Food Sci. Technol. 2009, 44, 386–393. [Google Scholar] [CrossRef]
- Kozłowska, M.; Laudy, A.E.; Przybył, J.; Ziarno, M.; Majewska, E. Chemical composition and antibacterial activity of some medicinal plants from Lamiaceae family. Acta Pol. Pharm. 2015, 72, 757–767. [Google Scholar]
- International Organization for Standardization (ISO). ISO 12966-2:2017 Animal and Vegetable Fats and Oils–Gas Chromatography of Fatty Acid Methyl Esters–Part 2: Preparation of Methyl Esters of Fatty Acids; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- International Organization for Standardization (ISO). ISO 3960:2009 Animal and Vegetable Fats and Oils–Determination of Peroxide Value–Iodometric Visual Endpoint Determination; ISO: Geneva, Switzerland, 2009. [Google Scholar]
- International Organization for Standardization (ISO). ISO 6885:2008 Animal and Vegetable Fats and Oils–Determination of Anisidine Value; ISO: Geneva, Switzerland, 2008. [Google Scholar]
- International Organization for Standardization (ISO). ISO 3656:2011 Animal and Vegetable Fats and Oils–Determination of Ultraviolet Absorbance Expressed as Specific UV Extinction; ISO: Geneva, Switzerland, 2011. [Google Scholar]
- Kozłowska, M.; Gruczyńska, E.; Ścibisz, I.; Rudzińska, M. Fatty acids and sterols composition, and antioxidant activity of oils extracted from the plant seeds. Food Chem. 2016, 213, 450–456. [Google Scholar] [CrossRef]
- Kozłowska, M.; Krzyczkowska, J. Effect of oils extracted from plant seeds on the growth and lipolytic activity of Yarrowia lipolytica yeast. J. Am. Oil Chem. Soc. 2017, 94, 661–671. [Google Scholar] [CrossRef]
- Mildner-Szkudlarz, S.; Zawirska-Wojtasiak, R.; Obuchowski, W.; Gośliński, M. Evaluation of antioxidant activity of green tea extract and its effect on the biscuits lipid fraction oxidative stability. J. Food Sci. 2009, 74, S362–S370. [Google Scholar] [CrossRef]
- Kozłowska, M.; Żbikowska, A.; Gruczyńska, E.; Żontała, K.; Półtorak, A. Effects of spice extracts on lipid fraction oxidative stability of cookies investigated by DSC. J. Therm. Anal. Calorim. 2014, 118, 1697–1705. [Google Scholar] [CrossRef]
- Balestra, F.; Cocci, E.; Pinnavaia, G.G.; Romani, S. Evaluation of antioxidant, rheological and sensorial properties of wheat dough and bread containing ginger powder. LWT-Food Sci. Technol. 2011, 44, 700–705. [Google Scholar] [CrossRef]
- Boz, H.; Karaoğlu, M.M. Improving the quality of whole wheat bread by using various plant origin materials. Czech J. Food Sci. 2013, 31, 457–466. [Google Scholar] [CrossRef]
- Caleja, C.; Barros, L.; Barreira, J.C.M.; Ciric, A.; Sokovic, M.; Calhelha, R.C.; Beatriz, M.; Oliveira, P.P.; Ferreira, I.C.F.R. Suitability of lemon balm (Melissa officinalis L.) extract rich in rosmarinic acid as a potential enhancer of functional properties in cupcakes. Food Chem. 2018, 250, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Latoch, A.; Stasiak, D.M. Effect of water extract of Urtica dioica L. on lipid oxidation and color of cooked pork sausage. J. Food Process. Pres. 2017, 41, e12818. [Google Scholar] [CrossRef]
- Nanditha, B.R.; Jena, B.S.; Prabhasankar, P. Influence of natural antioxidants and their carry-through property in biscuit processing. J. Sci. Food Agric. 2009, 89, 288–298. [Google Scholar] [CrossRef]
- Cheng, Y.F.; Bhat, R. Functional, physicochemical and sensory properties of novel cookies produced by utilizing underutilized (Pithecellobium jiringa Jack.) legume flour. Food Biosci. 2016, 14, 54–61. [Google Scholar] [CrossRef]
- Riyazi, S.S.; Asefi, N. Antioxidant effect of Urtica dioica on the stability of rapeseed oil during deep frying of French fries. Int. J. Biosci. 2015, 6, 20–28. [Google Scholar]
- Belščak-Cvitanović, A.; Komes, D.; Durgo, K.; Vojvodić, A.; Bušić, A. Nettle (Urtica dioica L.) extracts as functional ingredients for production of chocolates with improved bioactive composition and sensory properties. J. Food Sci. Technol. 2015, 52, 7723–7734. [Google Scholar] [CrossRef]
- Davidov-Pardo, G.; Moreno, M.; Marin-Arroyo, M.R.; Bleibaum, R.N.; Bruhn, C.M. Sensory and consumer perception of the addition of grape seed extracts in cookies. J. Food Sci. 2012, 77, S430–S438. [Google Scholar] [CrossRef]
- Lee, H.-J.; Choi, Y.-J.; Choi, Y., II; Lee, J.-J. Effects of lemon balm on the oxidative stability and the quality properties of hamburger patties during refrigerated storage. Korean J. Food Sci. Anim. Resour. 2014, 34, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Kozłowska, M.; Żbikowska, A.; Szpicer, A.; Półtorak, A. Oxidative stability of lipid fractions of sponge-fat cakes after green tea extracts application. J. Food Sci. Technol. 2019, 56, 2628–2638. [Google Scholar] [CrossRef] [PubMed]
- Soleimani, H.; Barzegar, M.; Sahari, M.A.; Naghdi, B.H. An investigation on the antioxidant activities of Hyssopus officinalis L. and Echinacea purpurea L. plant extracts in oil model system. J. Med. Plants 2011, 10, 61–72. [Google Scholar]
- Buta, N.; Popa, N.; Roman, L.; Bordea, G.; Bordea, A.; Bordea, N.; Poiană, M.A.; Traşcă, T.-I. The antioxidant effect of Melissa officinalis extract regarding the sunflower oil used in food thermal applications. J. Agroaliment. Proc. Technol. 2013, 19, 276–279. [Google Scholar]
- Farahmandfar, R.; Naeil, M.H.; Naderi, M.; Asnaashari, M. Stabilizing corn oil using the lemon balm (Melissa officinalis) antioxidants extracted by subcritical water. J. Food Sci. Technol. 2019, 56, 695–704. [Google Scholar] [CrossRef]
- Kozłowska, M.; Ścibisz, I.; Zaręba, D.; Ziarno, M. Antioxidant properties and effect on lactic acid bacterial growth of spice extracts. CyTA-J. Food 2015, 13, 573–577. [Google Scholar] [CrossRef]
- Chrubasik, J.E.; Roufogalis, B.D.; Wagner, H.; Chrubasik, S.A. A comprehensive review on nettle effect and efficacy profiles, Part I: Herba urticae. Phytomedicine 2007, 14, 423–435. [Google Scholar] [CrossRef]
- Hanczakowska, E.; Świątkiewicz, M.; Szewczyk, A. Effect of dietary nettle extract on pig meat quality. Med. Wet. 2007, 63, 525–527. [Google Scholar]
- Loetscher, Y.; Kreuzer, M.; Messikommer, R.E. Oxidative stability of the meat of broilers supplemented with rosemary leaves, rosehip fruits, chokeberry pomace, and entire nettle, and effects on performance and meat quality. Poult. Sci. 2013, 92, 2938–2948. [Google Scholar] [CrossRef]
- Ma, R.; Gao, T.; Song, L.; Zhang, L.; Jiang, Y.; Li, J.; Zhang, X.; Gao, F.; Zhou, G. Effects of oil-water mixed frying and pure-oil frying on the quality characteristics of soybean oil and chicken chop. Food Sci. Technol. Camp. 2016, 36, 329–336. [Google Scholar] [CrossRef]
- Cagdas, E.; Kumcuoglu, S. Effect of grape seed powder on oxidative stability of precooked chicken nuggets during frozen storage. J. Food Sci. Technol. 2015, 52, 2918–2925. [Google Scholar] [CrossRef] [PubMed]
- Budryn, G.; Nebesny, E. Effect of green and roasted coffee antioxidants on quality and shelf life of cookies and chocolates. J. Food Process. Pres. 2013, 37, 835–845. [Google Scholar] [CrossRef]
- Economu, K.D.; Oreopoulos, V.; Thomas, C.D. Antioxidant activity of some plant extracts of the family Labiatae. J. Am. Oil Chem. Soc. 1991, 68, 109–113. [Google Scholar] [CrossRef]
- Kahn, M.A.; Shahidi, F. Effects of natural and synthetic antioxidants on the oxidative stability of borage and evening primrose triacylglycerols. Food Chem. 2001, 75, 431–437. [Google Scholar] [CrossRef]
- Nogala-Kalucka, M.; Korczak, J.; Dratwia, M.; Lampart-Szczapa, E.; Siger, A.; Buchowski, M. Changes in antioxidant activity and free radical scavenging potential of rosemary extract and tocopherols in isolated rapeseed oil triacylglycerols during accelerated tests. Food Chem. 2005, 93, 227–235. [Google Scholar] [CrossRef]
- Konsoula, Z.; Liakopoulou-Kyriakides, M. Effect of endogenous antioxidants of sesame seeds and sesame oil to the thermal stability of edible vegetable oils. LWT-Food Sci. Technol. 2010, 43, 1379–1386. [Google Scholar] [CrossRef]
- Mišan, A.; Mimica-Dukić, N.; Sakač, M.; Mandić, A.; Sedej, I.; Šimurina, O.; Tumbas, V. Antioxidant activity of medicinal plant extracts in cookies. J. Food Sci. 2011, 76, C1239–C1244. [Google Scholar] [CrossRef]
| Sample | Parameter | |||
|---|---|---|---|---|
| L* | a* | b* | ||
| Control | 65.28 ± 1.34 e | 5.45 ± 0.68 h | 24.50 ± 1.05 f,g | |
| BHA | 0.02% | 63.83 ± 2.12 c | 4.25 ± 0.89 e | 23.76 ± 1.12 d |
| Hyssop extract | 0.02% | 66.81 ± 1.12 g | 4.15 ± 1.11 e | 24.38 ± 1.12 f |
| 0.1% | 66.45 ± 1.45 f,g | 3.30 ± 0.87 d | 23.08 ± 1.32 b | |
| 0.2% | 63.26 ± 1.54 b | 4.45 ± 1.34 f | 22.91 ± 1.34 b | |
| Lemon balm extract | 0.02% | 66.31 ± 1.23 f | 2.84 ± 0.99 c | 22.66 ± 0.78 a |
| 0.1% | 66.64 ± 1.45 f,g | 2.83 ± 1.23 c | 23.58 ± 0.67 d | |
| 0.2% | 64.46 ± 1.57 d | 2.68 ± 1.09 c | 25.47 ± 0.87 h | |
| Nettle extract | 0.02% | 64.82 ± 1.78 d | 4.68 ± 0.95 g | 23.36 ± 1.14 c |
| 0.1% | 63.57 ± 1.85 b,c | 1.59 ± 0.76 b | 24.08 ± 1.25 e | |
| 0.2% | 59.03 ± 1.87 a | 1.37 ± 1.09 a | 24.60 ± 1.28 g | |
| Sample | PV [meq O2/kg] | ||||
|---|---|---|---|---|---|
| Storage Time (Months) | |||||
| 0 | 1 | 2 | 3 | ||
| Control | 1.06 ± 0.02 B,a | 1.13 ± 0.01 B,b | 1.26 ± 0.02 A,c | 1.66 ± 0.03 C,d | |
| BHA | 0.02% | 0.99 ± 0.04 A,a | 1.03 ± 0.02 A,b | 1.25 ± 0.04 A,c | 1.43 ± 0.06 A,d |
| Hyssop Extract | 0.02% | 1.47 ± 0.12 C,a | 2.11 ± 0.09 C,d | 1.94 ± 0.06 B,c | 1.49 ± 0.08 B,b |
| 0.1% | 2.05 ± 0.03 D,a | 3.52 ± 0.11 E,d | 2.98 ± 0.10 C,c | 2.18 ± 0.12 D,b | |
| 0.2% | 3.57 ± 0.07 G,a | 4.98 ± 0.17 H,d | 4.23 ± 0.21 F,c | 3.73 ± 0.16 H,b | |
| Lemon Balm Extract | 0.02% | 3.87 ± 0.25 I,a | 5.12 ± 0.15 I,d | 4.43 ± 0.22 G,c | 3.95 ± 0.14 J,b |
| 0.1% | 3.42 ± 0.17 F,b | 4.78 ± 0.14 H,d | 3.82 ± 0.17 E,c | 3.39 ± 0.14 F,a | |
| 0.2% | 2.78 ± 0.09 E,b | 3.94 ± 0.07 F,d | 2.98 ± 0.11 C,c | 2.76 ± 0.12 E,a | |
| Nettle Extract | 0.02% | 3.67 ± 0.11 H,d | 3.45 ± 0.08 D,c | 3.33 ± 0.13 D,a | 3.43 ± 0.17 G,b |
| 0.1% | 4.98 ± 0.29 J,d | 4.75 ± 0.13 G,c | 4.53 ± 0.16 H,a | 4.61 ± 0.21 J,b | |
| 0.2% | 6.13 ± 0.21 K,b | 5.89 ± 0.17 J,a | 6.28 ± 0.21 J,c | 6.73 ± 0.26 K,d | |
| Sample | p-AnV | ||||
|---|---|---|---|---|---|
| Storage Time (Months) | |||||
| 0 | 1 | 2 | 3 | ||
| Control | 5.32 ± 0.22 I,a | 5.40 ± 0.19 I,b | 5.93 ± 0.32 I,c | 6.73 ± 0.39 J,d | |
| BHA | 0.02% | 2.81 ± 0.08 A,a | 2.96 ± 0.11 A,b | 3.46 ± 0.21 A,c | 3.94 ± 0.05 A,d |
| Hyssop Extract | 0.02% | 5.18 ± 0.16 H,a | 5.35 ± 0.49 H,b | 5.78 ± 0.36 H,d | 5.77 ± 0.28 F,c |
| 0.1% | 4.60 ± 0.07 G,a | 4.64 ± 0.21 F,b | 4.95 ± 0.21 F,c | 5.45 ± 0.22 C,d | |
| 0.2% | 4.18 ± 0.11 C,a | 4.25 ± 0.18 D,b | 4.38 ± 0.19 C,c | 5.63 ± 0.26 E,d | |
| Lemon Balm Extract | 0.02% | 5.63 ± 0.21 J,a | 5.68 ± 0.45 J,b | 7.04 ± 0.52 K,c | 7.89 ± 0.44 K,d |
| 0.1% | 5.77 ± 0.14 K,a | 5.79 ± 0.34 K,b | 6.89 ± 0.57 J,c | 6.98 ± 0.34 I,d | |
| 0.2% | 3.63 ± 0.10 B,a | 4.09 ± 0.27 B,b | 4.46 ± 0.71 D,c | 5.05 ± 0.32 B,d | |
| Nettle Extract | 0.02% | 4.19 ± 0.13 D,a | 4.21 ± 0.18 C,b | 4.36 ± 0.16 B,c | 5.55 ± 0.27 D,d |
| 0.1% | 4.59 ± 0.19 F,a | 4.88 ± 0.13 G,b | 5.07 ± 0.19 G,c | 5.86 ± 0.25 G,d | |
| 0.2% | 4.27 ± 0.21 E,a | 4.29 ± 0.17 E,a | 4.50 ± 0.16 E,b | 6.02 ± 0.29 H,c | |
| Fatty Acids | Area [%] | RT [min] |
|---|---|---|
| C12:0 (Lauric acid) | 0.06 ± 0.01 a | 8.065 |
| C14:0 (Myristic acid) | 0.23 ± 0.02 a | 8.421 |
| C16:0 (Palmitic acid) | 21.01 ± 0.04 d | 8.862 |
| C16:1 (Palmitoleic acid) | 0.08 ± 0.01 a | 8.934 |
| C17:0 (Margaric acid) | 0.05 ± 0.01 a | 9.605 |
| C18:0 (Stearic acid) | 3.36 ± 0.02 b | 10.388 |
| C18:1, n-9c (Oleic acid) | 29.18 ± 0.03 e | 10.453 |
| C18:1, n-9t (Elaidic acid) | 0.49 ± 0.01 a | 10.771 |
| C18:2, n-6 (Linoleic acid) | 40.52 ± 0.05 f | 11.239 |
| C18:3, n-3 (α-Linolenic acid) | 4.68 ± 0.03 c | 11.881 |
| C20:0 (Arachidic acid) | 0.22 ± 0.01 a | 12.464 |
| C20:1 (Gondoic acid) | 0.12 ± 0.01 a | 12.668 |
| ∑ SFA | 24.93 ± 0.04 | |
| ∑ MUFA | 29.87 ± 0.06 | |
| ∑ PUFA | 45.20 ± 0.08 |
| Sample | Storage Time (Months) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | ||||||
| K232 | K268 | K232 | K268 | K232 | K268 | K232 | K268 | ||
| Control | 1.44 ± 0.02 C,a | 0.78 ± 0.01 D,E,a | 1.89 ± 0.03 G,b | 0.79 ± 0.01 E,a,b | 2.03 ± 0.02 I,c | 0.81 ± 0.01 E,b,c | 2.98 ± 0.03 I,d | 0.82 ± 0.02 D,c | |
| BHA | 0.02% | 1.09 ± 0.01 A,a | 0.60 ± 0.02 A,a | 1.24 ± 0.02 A,b | 0.65 ± 0.01 A,b | 1.47 ± 0.02 A,c | 0.67 ±0,01 A,c | 1.78 ± 0.03 A,d | 0.69 ± 0.02 A,d |
| Hyssop Extract | 0.02% | 1.42 ± 0.00 B,a | 0.77 ± 0.02 C,D,E,a | 1.84 ± 0.02 F,b | 0.82 ± 0.01 F,b | 2.09 ± 0.02 J,c | 0.84 ± 0.01 F,c | 2.49 ± 0.02 D,d | 0.88 ± 0.02 E,d |
| 0.1% | 1.53 ± 0.01 G,a | 0.75 ± 0.02 C,a | 1.69 ± 0.02 C,D,b | 0.76 ± 0.01 C,D,a,b | 1.86 ± 0.01 F,c | 0.77 ± 0.01 C,D,a,b | 2.62 ± 0.03 F,d | 0.78 ± 0.02 B,C,b | |
| 0.2% | 1.55 ± 0.02 I,a | 0.76 ± 0.02 C,D,a | 1.78 ± 0.01 E,b | 0.78 ± 0.02 D,E,a,b | 1.94 ± 0.02 G,c | 0.79 ± 0.01 D,E,b | 2.78 ± 0.03 G,d | 0.81 ± 0.02 D,c | |
| Lemon Balm Extract | 0.02% | 1.63 ± 0.01 J,a | 0.82 ± 0.01 F,a | 1.75 ± 0.01 E,b | 0.83 ± 0.02 F,G,a | 1.88 ± 0.02 F,c | 0.84 ± 0.02 F,a | 2.93 ± 0.03 H,d | 0.88 ± 0.01 E,b |
| 0.1% | 1.49 ± 0.02 D,a | 0.83 ± 0.02 F,a | 1.67 ± 0.01 C,b | 0.85 ± 0.01 G,a,b | 1.79 ± 0.02 E,c | 0.87 ± 0.02 G,b | 2.55 ± 0.02 E,d | 0.87 ± 0.01 E,b | |
| 0.2% | 1.50 ± 0.00 E,a | 0.79 ± 0.02 E,a | 1.91 ± 0.01 G,b | 0.79 ± 0.02 E,a | 1.98 ± 0.02 H,c | 0.80 ± 0.02 E,a | 2.51 ± 0.03 D,d | 0.82 ± 0.01 D,b | |
| Nettle Extract | 0.02% | 1.64 ± 0.01 K,a | 0.78 ± 0.02 D,E,a | 1.69 ± 0.02 C,D,b | 0.78 ± 0.02 D,E,a | 1.70 ± 0.01 C,b | 0.79 ± 0.02 D,E,a | 3.29 ± 0.03 J,c | 0.80 ± 0.02 C,D,a |
| 0.1% | 1.54 ± 0.02 H,a | 0.75 ± 0.01 C,a | 1.71 ± 0.01 D,b | 0.75 ± 0.02 B,C,a | 1.76 ± 0.02 D,c | 0.76 ± 0.02 B,C,a | 2.44 ± 0.02 C,d | 0.76 ± 0.02 B,a | |
| 0.2% | 1.51 ± 0.01 F,a | 0.72 ± 0.02 B,a | 1.55 ± 0.01 B,b | 0.73 ± 0.02 B,a | 1.57 ± 0.02 B,c | 0.74 ± 0.01 B,a | 2.08 ± 0.01 B,d | 0.78 ± 0.01 B,C,b | |
| Sample | TEAC (µmol/g fat) | ||||
|---|---|---|---|---|---|
| Storage Time (months) | |||||
| 0 | 1 | 2 | 3 | ||
| Control | 0.91 ± 0.09 B,d | 0.82 ± 0.07 A,c | 0.78 ± 0.06 B,b | 0.71 ± 0.05 B,a | |
| BHA | 0.02% | 1.52 ± 0.14 F,d | 1.47 ± 0.15 G,c | 1.40 ± 0.12 I,b | 1.32 ± 0.10 I,a |
| Hyssop Extract | 0.02% | 1.49 ± 0.13 F,d | 1.39 ± 0.12 F,c | 1.12 ± 0.10 D,b | 1.06 ± 0.09 F,a |
| 0.1% | 0.85 ± 0.07 A,d | 0.81 ± 0.07 A,c | 0.68 ± 0.07 A,b | 0.54 ± 0.04 A,a | |
| 0.2% | 1.56 ± 0.16 G,d | 1.16 ± 0.12 B,c | 1.15 ± 0.12 E,b | 0.96 ± 0.07 D,a | |
| Lemon Balm Extract | 0.02% | 1.31 ± 0.12 D,d | 1.18 ± 0.10 B,c | 0.90 ± 0.07 C,b | 0.82 ± 0.06 C,a |
| 0.1% | 1.15 ± 0.11 C,b | 1.25 ± 0.11 C,d | 1.17 ± 0.12 E,c | 1.01 ± 0.09 E,a | |
| 0.2% | 1.86 ± 0.16 H,d | 1.35 ± 0.12 E,c | 1.15 ± 0.11 E,b | 1.11 ± 0.10 G,a | |
| Nettle Extract | 0.02% | 1.56 ± 0.14 G,d | 1.30 ± 0.12 D,c | 1.22 ± 0.10 F,b | 1.18 ± 0.11 H,a |
| 0.1% | 1.84 ± 0.16 H,b | 1.31 ± 0.11 D,a | 1.30 ± 0.12 H,a | 1.03 ± 0.08 E,a | |
| 0.2% | 1.36 ± 0.12 E,d | 1.33 ± 0.12 D,e | 1.27 ± 0.12 G,b | 0.84 ± 0.06 C,a | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kozlowska, M.; Zbikowska, A.; Marciniak-Lukasiak, K.; Kowalska, M. Herbal Extracts Incorporated into Shortbread Cookies: Impact on Color and Fat Quality of the Cookies. Biomolecules 2019, 9, 858. https://doi.org/10.3390/biom9120858
Kozlowska M, Zbikowska A, Marciniak-Lukasiak K, Kowalska M. Herbal Extracts Incorporated into Shortbread Cookies: Impact on Color and Fat Quality of the Cookies. Biomolecules. 2019; 9(12):858. https://doi.org/10.3390/biom9120858
Chicago/Turabian StyleKozlowska, Mariola, Anna Zbikowska, Katarzyna Marciniak-Lukasiak, and Malgorzata Kowalska. 2019. "Herbal Extracts Incorporated into Shortbread Cookies: Impact on Color and Fat Quality of the Cookies" Biomolecules 9, no. 12: 858. https://doi.org/10.3390/biom9120858
APA StyleKozlowska, M., Zbikowska, A., Marciniak-Lukasiak, K., & Kowalska, M. (2019). Herbal Extracts Incorporated into Shortbread Cookies: Impact on Color and Fat Quality of the Cookies. Biomolecules, 9(12), 858. https://doi.org/10.3390/biom9120858








