The HER2 S310F Mutant Can Form an Active Heterodimer with the EGFR, Which Can Be Inhibited by Cetuximab but Not by Trastuzumab as well as Pertuzumab
Abstract
1. Introduction
2. Materials and Methods
2.1. Expression and Purification of Recombinant Fusion Proteins
2.2. ELISA
2.3. Cell Culture
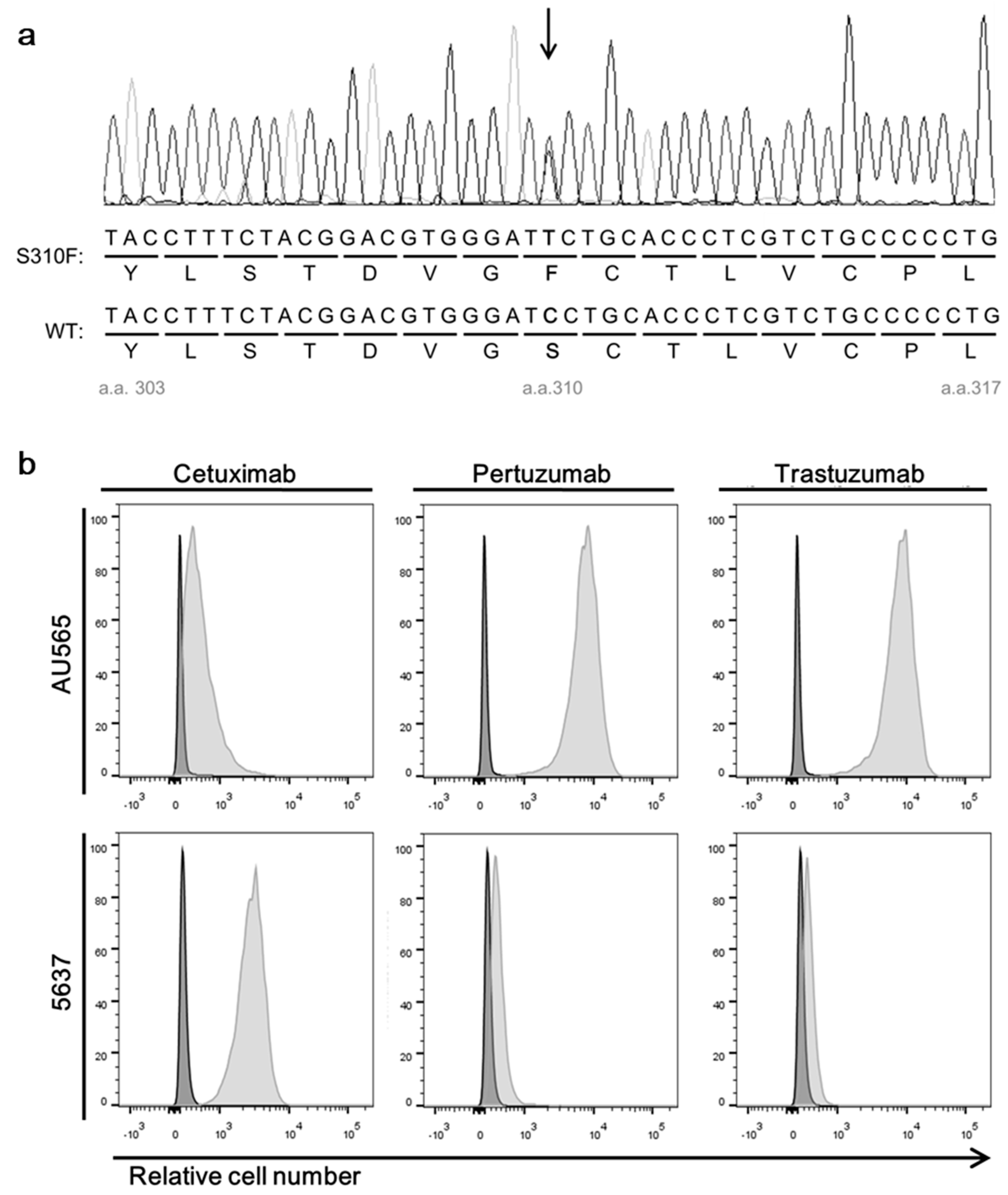
2.4. Sequencing of Amplified HER2 Gene Fragments of 5637 Cell
2.5. Flow Cytometry Analysis
2.6. Immunoprecipitation and Immunoblot Analyses
2.7. Cell Viability Assay and Immunoblotting
2.8. Single-Molecular Interaction Analysis Using Total Internal Reflection Fluorescence (TIRF) Microscopy
3. Results
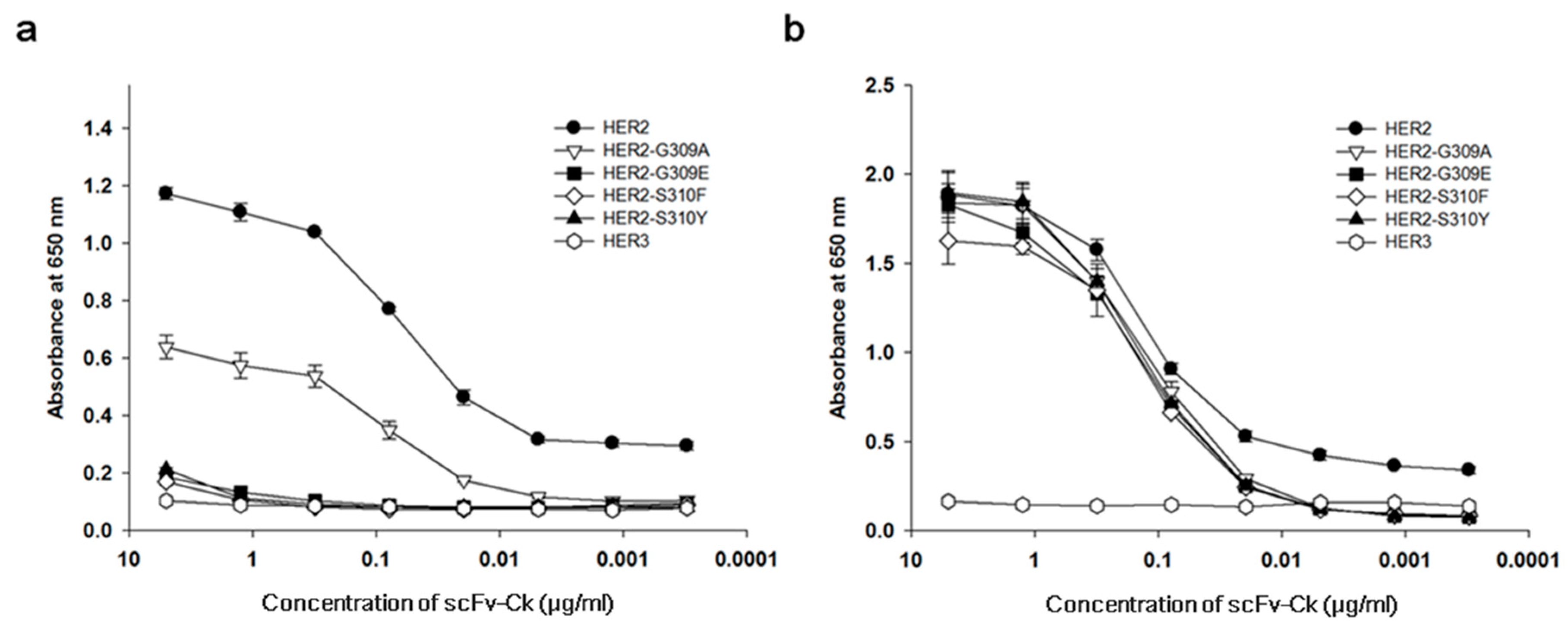
3.1. The Recombinant S310F Mutant Is Not Reactive to Pertuzumab but Binds to Trastuzumab
3.2. A Bladder Cancer Cell Line, 5637, Expresses Both Wild-Type HER2 and S310F Mutant
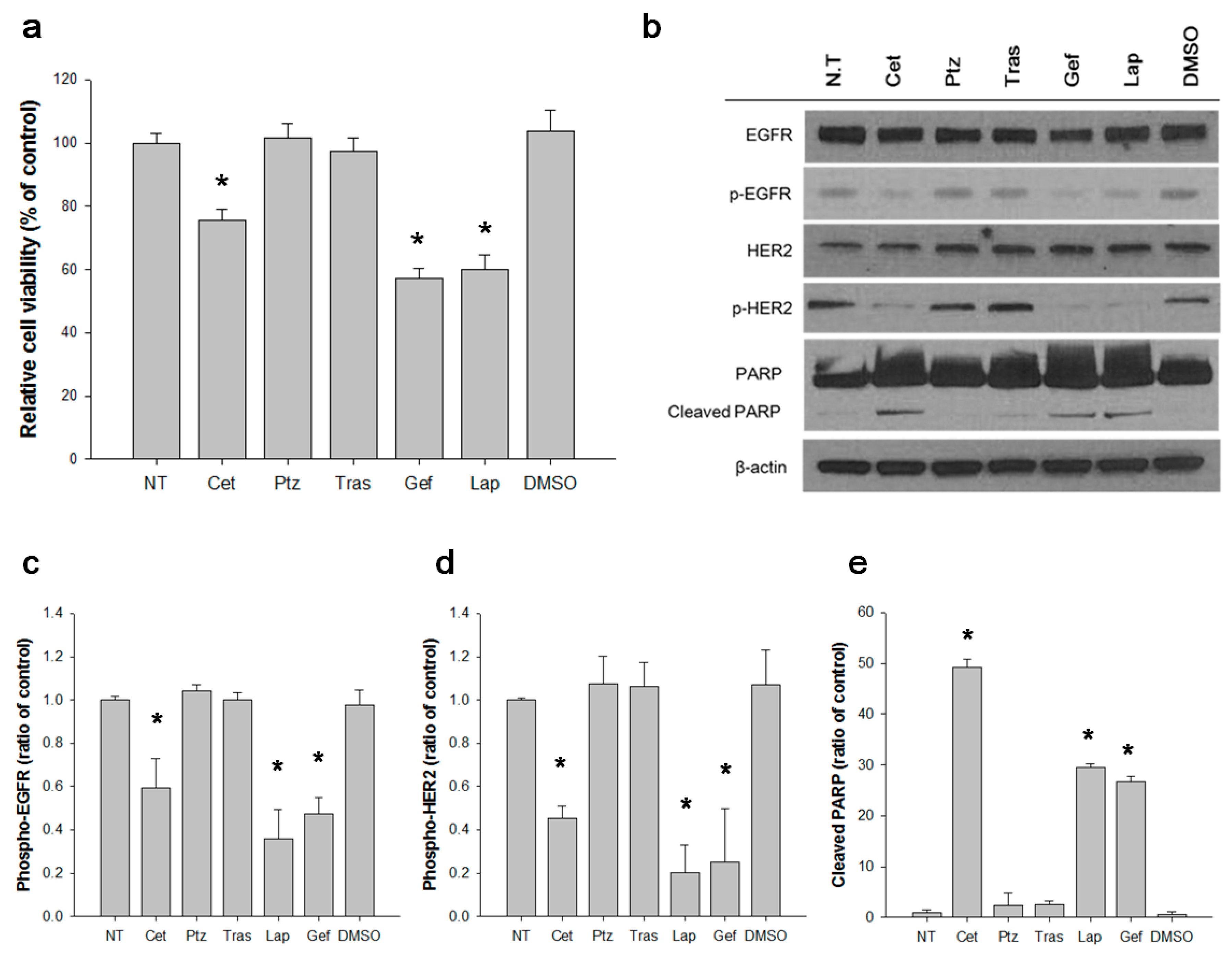
3.3. EGFR Activates S310F HER2 Mutant in 5637 Cell
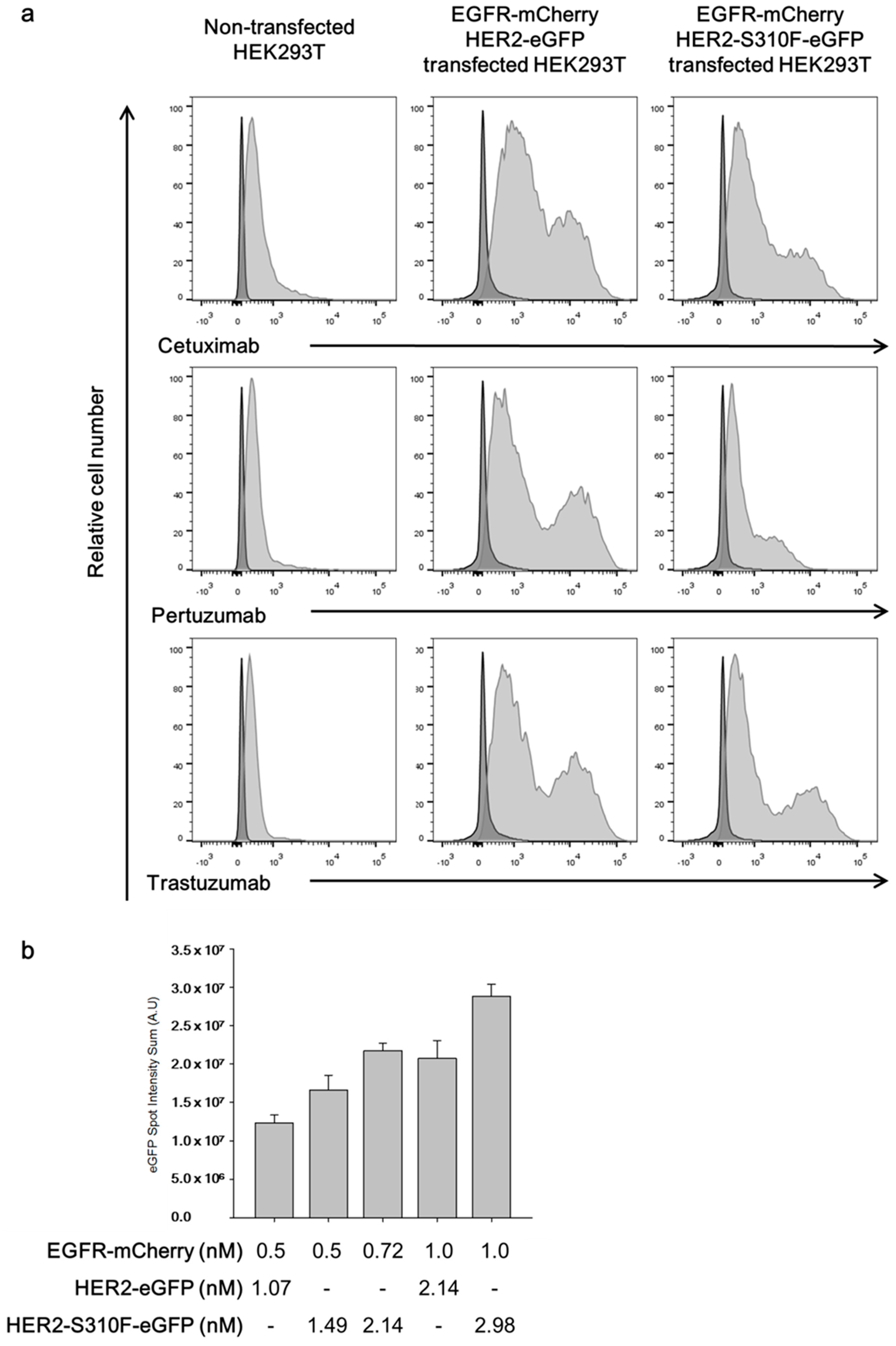
3.4. Single-Molecular Interaction Analysis Demonstrated That the S310F HER2 Mutant Formed Heterodimers with the EGFR
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shepard, H.M.; Brdlik, C.M.; Schreiber, H. Signal integration: A framework for understanding the efficacy of therapeutics targeting the human EGFR family. J. Clin. Invest. 2008, 118, 3574–3581. [Google Scholar] [CrossRef] [PubMed]
- Yarden, Y.; Sliwkowski, M.X. Untangling the ErbB signalling network. Nat. Rev. Mol. Cell Biol. 2001, 2, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Lemmon, M.A.; Schlessinger, J.; Ferguson, K.M. The EGFR family: Not so prototypical receptor tyrosine kinases. Cold Spring Harb. Perspect. Biol. 2014, 6, a020768. [Google Scholar] [CrossRef] [PubMed]
- Yarden, Y.; Pines, G. The ERBB network: At last, cancer therapy meets systems biology. Nat. Rev. Cancer 2012, 12, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Moasser, M.M. The oncogene HER2: Its signaling and transforming functions and its role in human cancer pathogenesis. Oncogene 2007, 26, 6469–6487. [Google Scholar] [CrossRef] [PubMed]
- Slamon, D.J.; Clark, G.M.; Wong, S.G.; Levin, W.J.; Ullrich, A.; McGuire, W.L. Human breast cancer: Correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 1987, 235, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, R.; Narasanna, A.; Wang, S.E.; Liu, S.; Chakrabarty, A.; Balko, J.M.; Gonzalez-Angulo, A.M.; Mills, G.B.; Penuel, E.; Winslow, J.; et al. Trastuzumab has preferential activity against breast cancers driven by HER2 homodimers. Cancer Res. 2011, 71, 1871–1882. [Google Scholar] [CrossRef]
- Cho, H.S.; Mason, K.; Ramyar, K.X.; Stanley, A.M.; Gabelli, S.B.; Denney, D.W., Jr.; Leahy, D.J. Structure of the extracellular region of HER2 alone and in complex with the Herceptin Fab. Nature 2003, 421, 756–760. [Google Scholar] [CrossRef]
- Franklin, M.C.; Carey, K.D.; Vajdos, F.F.; Leahy, D.J.; de Vos, A.M.; Sliwkowski, M.X. Insights into ErbB signaling from the structure of the ErbB2-pertuzumab complex. Cancer Cell 2004, 5, 317–328. [Google Scholar] [CrossRef]
- Agus, D.B.; Akita, R.W.; Fox, W.D.; Lewis, G.D.; Higgins, B.; Pisacane, P.I.; Lofgren, J.A.; Tindell, C.; Evans, D.P.; Maiese, K.; et al. Targeting ligand-activated ErbB2 signaling inhibits breast and prostate tumor growth. Cancer Cell 2002, 2, 127–137. [Google Scholar] [CrossRef]
- Mann, M.; Sheng, H.; Shao, J.; Williams, C.S.; Pisacane, P.I.; Sliwkowski, M.X.; DuBois, R.N. Targeting cyclooxygenase 2 and HER-2/neu pathways inhibits colorectal carcinoma growth. Gastroenterology 2001, 120, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Lua, W.H.; Gan, S.K.; Lane, D.P.; Verma, C.S. A search for synergy in the binding kinetics of Trastuzumab and Pertuzumab whole and F(ab) to Her2. NPJ Breast Cancer 2015, 1, 15012. [Google Scholar] [CrossRef] [PubMed]
- Burris, H.A., 3rd; Rugo, H.S.; Vukelja, S.J.; Vogel, C.L.; Borson, R.A.; Limentani, S.; Tan-Chiu, E.; Krop, I.E.; Michaelson, R.A.; Girish, S.; et al. Phase II study of the antibody drug conjugate trastuzumab-DM1 for the treatment of human epidermal growth factor receptor 2 (HER2)-positive breast cancer after prior HER2-directed therapy. J. Clin. Oncol. 2011, 29, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Goss, P.E.; Smith, I.E.; O’Shaughnessy, J.; Ejlertsen, B.; Kaufmann, M.; Boyle, F.; Buzdar, A.U.; Fumoleau, P.; Gradishar, W.; Martin, M.; et al. Adjuvant lapatinib for women with early-stage HER2-positive breast cancer: A randomised, controlled, phase 3 trial. Lancet Oncol. 2013, 14, 88–96. [Google Scholar] [CrossRef]
- Kelly, K.; Altorki, N.K.; Eberhardt, W.E.; O’Brien, M.E.; Spigel, D.R.; Crino, L.; Tsai, C.M.; Kim, J.H.; Cho, E.K.; Hoffman, P.C.; et al. Adjuvant Erlotinib Versus Placebo in Patients With Stage IB-IIIA Non-Small-Cell Lung Cancer (RADIANT): A Randomized, Double-Blind, Phase III Trial. J. Clin. Oncol. 2015, 33, 4007–4014. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.; Delaloge, S.; Holmes, F.A.; Moy, B.; Iwata, H.; Harvey, V.J.; Robert, N.J.; Silovski, T.; Gokmen, E.; von Minckwitz, G.; et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): A multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016, 17, 367–377. [Google Scholar] [CrossRef]
- Connell, C.M.; Doherty, G.J. Activating HER2 mutations as emerging targets in multiple solid cancers. ESMO Open 2017, 2, e000279. [Google Scholar] [CrossRef]
- Kiss, B.; Wyatt, A.W.; Douglas, J.; Skuginna, V.; Mo, F.; Anderson, S.; Rotzer, D.; Fleischmann, A. Her2 alterations in muscle-invasive bladder cancer: Patient selection beyond protein expression for targeted therapy. Sci. Rep. 2017, 7, 42713. [Google Scholar] [CrossRef]
- Chumsri, S.; Weidler, J.; Ali, S.; Balasubramanian, S.; Wallweber, G.; DeFazio-Eli, L.; Chenna, A.; Huang, W.; DeRidder, A.; Goicocheal, L.; et al. Prolonged Response to Trastuzumab in a Patient With HER2-Nonamplified Breast Cancer With Elevated HER2 Dimerization Harboring an ERBB2 S310F Mutation. J. Natl. Compr. Cancer Netw. 2015, 13, 1066–1070. [Google Scholar] [CrossRef]
- Greulich, H.; Kaplan, B.; Mertins, P.; Chen, T.H.; Tanaka, K.E.; Yun, C.H.; Zhang, X.; Lee, S.H.; Cho, J.; Ambrogio, L.; et al. Functional analysis of receptor tyrosine kinase mutations in lung cancer identifies oncogenic extracellular domain mutations of ERBB2. Proc. Natl. Acad. Sci. USA 2012, 109, 14476–14481. [Google Scholar] [CrossRef]
- Ding, L.; Getz, G.; Wheeler, D.A.; Mardis, E.R.; McLellan, M.D.; Cibulskis, K.; Sougnez, C.; Greulich, H.; Muzny, D.M.; Morgan, M.B.; et al. Somatic mutations affect key pathways in lung adenocarcinoma. Nature 2008, 455, 1069–1075. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Ali, S.M.; Saad, S.; Chan, C.A.; Miller, V.A.; Halmos, B. Successful treatment of a patient with Li-Fraumeni syndrome and metastatic lung adenocarcinoma harboring synchronous EGFR L858R and ERBB2 extracellular domain S310F mutations with the pan-HER inhibitor afatinib. Cancer Biol. Ther. 2014, 15, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Eng, J.; Hsu, M.; Chaft, J.E.; Kris, M.G.; Arcila, M.E.; Li, B.T. Outcomes of chemotherapies and HER2 directed therapies in advanced HER2-mutant lung cancers. Lung Cancer 2016, 99, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Kan, Z.; Jaiswal, B.S.; Stinson, J.; Janakiraman, V.; Bhatt, D.; Stern, H.M.; Yue, P.; Haverty, P.M.; Bourgon, R.; Zheng, J.; et al. Diverse somatic mutation patterns and pathway alterations in human cancers. Nature 2010, 466, 869–873. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.P.; Roth, A.; Goya, R.; Oloumi, A.; Ha, G.; Zhao, Y.; Turashvili, G.; Ding, J.; Tse, K.; Haffari, G.; et al. The clonal and mutational evolution spectrum of primary triple-negative breast cancers. Nature 2012, 486, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Banerji, S.; Cibulskis, K.; Rangel-Escareno, C.; Brown, K.K.; Carter, S.L.; Frederick, A.M.; Lawrence, M.S.; Sivachenko, A.Y.; Sougnez, C.; Zou, L.; et al. Sequence analysis of mutations and translocations across breast cancer subtypes. Nature 2012, 486, 405–409. [Google Scholar] [CrossRef]
- Bose, R.; Kavuri, S.M.; Searleman, A.C.; Shen, W.; Shen, D.; Koboldt, D.C.; Monsey, J.; Goel, N.; Aronson, A.B.; Li, S.; et al. Activating HER2 mutations in HER2 gene amplification negative breast cancer. Cancer Discov. 2013, 3, 224–237. [Google Scholar] [CrossRef]
- Ross, J.S.; Wang, K.; Sheehan, C.E.; Boguniewicz, A.B.; Otto, G.; Downing, S.R.; Sun, J.; He, J.; Curran, J.A.; Ali, S.; et al. Relapsed classic E-cadherin (CDH1)-mutated invasive lobular breast cancer shows a high frequency of HER2 (ERBB2) gene mutations. Clin. Cancer Res. 2013, 19, 2668–2676. [Google Scholar] [CrossRef]
- Ali, S.M.; Alpaugh, R.K.; Downing, S.R.; Stephens, P.J.; Yu, J.Q.; Wu, H.; Buell, J.K.; Miller, V.A.; Lipson, D.; Palmer, G.A.; et al. Response of an ERBB2-mutated inflammatory breast carcinoma to human epidermal growth factor receptor 2-targeted therapy. J. Clin. Oncol. 2014, 32, e88-91. [Google Scholar] [CrossRef]
- Endo, Y.; Dong, Y.; Kondo, N.; Yoshimoto, N.; Asano, T.; Hato, Y.; Nishimoto, M.; Kato, H.; Takahashi, S.; Nakanishi, R.; et al. HER2 mutation status in Japanese HER2-positive breast cancer patients. Breast Cancer 2016, 23, 902–907. [Google Scholar] [CrossRef]
- Jasra, S.; Opyrchal, M.; Norton, L.; Mehta, R. A Rare Case of S310F Somatic ERBB2 Mutation in a HER2-Nonamplified Breast Cancer. Clin. Breast Cancer 2017, 17, e37–e41. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.S.; Wang, K.; Gay, L.M.; Al-Rohil, R.N.; Nazeer, T.; Sheehan, C.E.; Jennings, T.A.; Otto, G.A.; Donahue, A.; He, J.; et al. A high frequency of activating extracellular domain ERBB2 (HER2) mutation in micropapillary urothelial carcinoma. Clin. Cancer Res. 2014, 20, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Vornicova, O.; Hershkovitz, D.; Yablonski-Peretz, T.; Ben-Itzhak, O.; Keidar, Z.; Bar-Sela, G. Treatment of metastatic extramammary Paget’s disease associated with adnexal adenocarcinoma, with anti-HER2 drugs based on genomic alteration ERBB2 S310F. Oncologist 2014, 19, 1006–1007. [Google Scholar] [CrossRef] [PubMed]
- Barretina, J.; Caponigro, G.; Stransky, N.; Venkatesan, K.; Margolin, A.A.; Kim, S.; Wilson, C.J.; Lehar, J.; Kryukov, G.V.; Sonkin, D.; et al. The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature 2012, 483, 603–607. [Google Scholar] [CrossRef]
- Wang, T.; Xu, Y.; Sheng, S.; Yuan, H.; Ouyang, T.; Li, J.; Wang, T.; Fan, Z.; Fan, T.; Lin, B.; et al. HER2 somatic mutations are associated with poor survival in HER2-negative breast cancers. Cancer Sci. 2017, 108, 671–677. [Google Scholar] [CrossRef]
- Muzny, D.M.; Bainbridge, M.N.; Chang, K.; Dinh, H.H.; Drummond, J.A.; Fowler, G.; Kvar, C.L.; Lewis, L.R. Comprehensive molecular characterization of human colon and rectal cancer. Nature 2012, 487, 330–337. [Google Scholar] [CrossRef]
- Han, S.W.; Kim, H.P.; Shin, J.Y.; Jeong, E.G.; Lee, W.C.; Lee, K.H.; Won, J.K.; Kim, T.Y.; Oh, D.Y.; Im, S.A.; et al. Targeted sequencing of cancer-related genes in colorectal cancer using next-generation sequencing. PLoS ONE 2013, 8, e64271. [Google Scholar] [CrossRef]
- Wang, K.; Kan, J.; Yuen, S.T.; Shi, S.T.; Chu, K.M.; Law, S.; Chan, T.L.; Kan, Z.; Chan, A.S.; Tsui, W.Y.; et al. Exome sequencing identifies frequent mutation of ARID1A in molecular subtypes of gastric cancer. Nat. Genet. 2011, 43, 1219–1223. [Google Scholar] [CrossRef]
- Petrelli, F.; Tomasello, G.; Barni, S.; Lonati, V.; Passalacqua, R.; Ghidini, M. Clinical and pathological characterization of HER2 mutations in human breast cancer: A systematic review of the literature. Breast Cancer Res. Treat. 2017, 166, 339–349. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, H.; Chung, J. An antibody reactive to the Gly63-Lys68 epitope of NT-proBNP exhibits O-glycosylation-independent binding. Exp. Mol. Med. 2014, 46, e114. [Google Scholar] [CrossRef]
- Park, S.; Lee, D.H.; Park, J.G.; Lee, Y.T.; Chung, J. A sensitive enzyme immunoassay for measuring cotinine in passive smokers. Clin. Chim. Acta 2010, 411, 1238–1242. [Google Scholar] [CrossRef] [PubMed]
- Takeshi, K.; Hitomi, S.; Yasuto, A.; Shun’ichiro, T. In situ delivery and production system of trastuzumab scFv with Bifidobacterium. Biochem. Biophy. Res. Commun. 2017, 493, 306–312. [Google Scholar]
- Akbari, V.; Sadeghi, H.M.M.; Jafrian-Dehkordi, A.; Abedi, D.; Chou, C.P. Functional expression of a single-chain antibody fragment against human epidermal growth factor receptor 2 (HER2) in Escherichia coli. J. Ind. Microbiol. Biotechnol. 2014, 41, 947–956. [Google Scholar] [CrossRef]
- Judith, N.; Christoph, S.; Hannes, B.; Grit, H.T.; Rolf, F.; Georg, M.; Rainer, F.; Stefan, B. Novel EGFR-specific immunotoxins based on panitumumab and cetuximab show in vitro and ex vivo activity against different tumor entities. J. Cancer Res. Clin. Oncol. 2015, 141, 2079–2095. [Google Scholar] [CrossRef]
- Baldi, L.; Muller, N.; Picasso, S.; Jacquet, R.; Girard, P.; Thanh, H.P.; Derow, E.; Wurm, F.M. Transient gene expression in suspension HEK-293 cells: Application to large-scale protein production. Biotechnol. Prog. 2005, 21, 148–153. [Google Scholar] [CrossRef]
- Yoon, S.; Kim, Y.H.; Kang, S.H.; Kim, S.K.; Lee, H.K.; Kim, H.; Chung, J.; Kim, I.H. Bispecific Her2 x cotinine antibody in combination with cotinine-(histidine)2-iodine for the pre-targeting of Her2-positive breast cancer xenografts. J. Cancer Res. Clin. Oncol. 2014, 140, 227–233. [Google Scholar] [CrossRef]
- Kim, H.; Hong, S.H.; Kim, J.Y.; Kim, I.C.; Park, Y.W.; Lee, S.J.; Song, S.W.; Kim, J.J. Preclinical development of a humanized neutralizing antibody targeting HGF. Exp. Mol. Med. 2017, 49, e309. [Google Scholar] [CrossRef]
- Lee, S.; Yoon, I.H.; Yoon, A.; Cook-Mills, J.M.; Park, C.G.; Chung, J. An antibody to the sixth Ig-like domain of VCAM-1 inhibits leukocyte transendothelial migration without affecting adhesion. J. Immunol. 2012, 189, 4592–4601. [Google Scholar] [CrossRef]
- Lee, H.W.; Ryu, J.Y.; Yoo, J.; Choi, B.; Kim, K.; Yoon, T.Y. Real-time single-molecule coimmunoprecipitation of weak protein-protein interactions. Nat. Protoc. 2013, 8, 2045–2060. [Google Scholar] [CrossRef]
- Roy, R.; Hohng, S.; Ha, T. A practical guide to single-molecule FRET. Nat. Methods 2008, 5, 507–516. [Google Scholar] [CrossRef]
- Selvin, P.R.; Ha, T. Single-Molecule Techniques: A Labatory Manual; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2008. [Google Scholar]
- Strausberg, R.L. Cancer Genome Anatomy Project; Wiley Online Library: New York, New York, USA, 2006. [Google Scholar] [CrossRef]
- Shigematsu, H.; Takahashi, T.; Nomura, M.; Majmudar, K.; Suzuki, M.; Lee, H.; Wistuba, I.I.; Fong, K.M.; Toyooka, S.; Shimizu, N.; et al. Somatic mutations of the HER2 kinase domain in lung adenocarcinomas. Cancer Res. 2005, 65, 1642–1646. [Google Scholar] [CrossRef] [PubMed]
- The Cancer Genome Atlas Research Network; Weinstein, J.N.; Akbani, R.; Broom, B.M.; Wang, W.; Verhaak, R.G.W.; Lerner, S.; Morgan, M.; Creighton, C.J.; Kwiatkowski, D.J.; et al. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature 2014, 507, 315–322. [Google Scholar] [CrossRef]
- Wen, W.; Chen, W.S.; Xiao, N.; Bender, R.; Ghazalpour, A.; Tan, Z.; Swensen, J.; Millis, S.Z.; Basu, G.; Gatalica, Z.; et al. Mutations in the Kinase Domain of the HER2/ERBB2 Gene Identified in a Wide Variety of Human Cancers. J. Mol. Diagn. 2015, 17, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Zuo, W.J.; Jiang, Y.Z.; Wang, Y.J.; Xu, X.E.; Hu, X.; Liu, G.Y.; Wu, J.; Di, G.H.; Yu, K.D.; Shao, Z.M. Dual Characteristics of Novel HER2 Kinase Domain Mutations in Response to HER2-Targeted Therapies in Human Breast Cancer. Clin. Cancer Res. 2016, 22, 4859–4869. [Google Scholar] [CrossRef] [PubMed]
- Kavuri, S.M.; Jain, N.; Galimi, F.; Cottino, F.; Leto, S.M.; Migliardi, G.; Searleman, A.C.; Shen, W.; Monsey, J.; Trusolino, L.; et al. HER2 activating mutations are targets for colorectal cancer treatment. Cancer Discov. 2015, 5, 832–841. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Jiang, W.; Ye, S.; He, T.; Pei, X.; Li, J.; Chan, D.W.; Ngan, H.Y.S.; Li, F.; Tao, P.; et al. ERBB2 mutation: A promising target in non-squamous cervical cancer. Gynecol. Oncol. 2018, 148, 311–316. [Google Scholar] [CrossRef]
- Endo, Y.; Dong, Y.; Yoshimoto, N.; Asano, T.; Hato, Y.; Yamashita, H.; Sato, S.; Takahashi, S.; Fujii, Y.; Toyama, T. HER2 mutation status in Japanese HER2-negative breast cancer patients. Jpn. J. Clin. Oncol. 2014, 44, 619–623. [Google Scholar] [CrossRef]
- Weigelt, B.; Reis-Filho, J.S. Activating mutations in HER2: Neu opportunities and neu challenges. Cancer Discov. 2013, 3, 145–147. [Google Scholar] [CrossRef]
- Kancha, R.K.; von Bubnoff, N.; Bartosch, N.; Peschel, C.; Engh, R.A.; Duyster, J. Differential sensitivity of ERBB2 kinase domain mutations towards lapatinib. PLoS ONE 2011, 6, e26760. [Google Scholar] [CrossRef]
- Park, C.K.; Hur, J.Y.; Choi, C.M.; Kim, T.O.; Cho, H.J.; Shin, H.J.; Lim, J.H.; Choi, Y.D.; Kim, Y.C.; Oh, I.J. Efficacy of Afatinib in a Previously-Treated Patient with Non-Small Cell Lung Cancer Harboring HER2 Mutation: Case Report. J. Korean Med. Sci. 2018, 33, e7. [Google Scholar] [CrossRef]
- Gharib, E.; Salmanipour, R.; Nazemalhosseini, M.E.; Yaghoob, T.M.; Sarlak, S.; Malekzade-Moghani, M.; Nasrabadi, P.N.; Meiary, M.A.; Asadzadeh, A.H.; AZali, M.R. HER2+ mCRC patients with exon 20 R784G substitution mutation do not respond to the cetuximab therapy. J. Cell Physiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Zabransky, D.J.; Yankaskas, C.L.; Cochran, R.L.; Wong, H.Y.; Croessmann, S.; Chu, D.; Kavuri, S.M.; Red Brewer, M.; Rosen, D.M.; Dalton, W.B.; et al. HER2 missense mutations have distinct effects on oncogenic signaling and migration. Proc. Natl. Acad. Sci. USA 2015, 112, E6205–E6214. [Google Scholar] [CrossRef] [PubMed]
- Garrett, T.P.; McKern, N.M.; Lou, M.; Elleman, T.C.; Adams, T.E.; Lovrecz, G.O.; Kofler, M.; Jorissen, R.N.; Nice, E.C.; Burgess, A.W.; et al. The crystal structure of a truncated ErbB2 ectodomain reveals an active conformation, poised to interact with other ErbB receptors. Mol. Cell. 2003, 11, 495–505. [Google Scholar] [CrossRef]
- Kumagai, T.; Katsumata, M.; Hasegawa, A.; Furuuchi, K.; Funakoshi, T.; Kawase, I.; Greene, M.I. Role of extracellular subdomains of p185c-neu and the epidermal growth factor receptor in ligand-independent association and transactivation. Proc. Natl. Acad. Sci. USA 2003, 100, 9220–9225. [Google Scholar] [CrossRef]
- Junttila, T.T.; Akita, R.W.; Parsons, K.; Fields, C.; Lewis Phillips, G.D.; Friedman, L.S.; Sampath, D.; Sliwkowski, M.X. Ligand-independent HER2/HER3/PI3K complex is disrupted by trastuzumab and is effectively inhibited by the PI3K inhibitor GDC-0941. Cancer Cell 2009, 15, 429–440. [Google Scholar] [CrossRef]
- Maadi, H.; Nami, B.; Tong, J.; Li, G.; Wang, Z. The effects of trastuzumab on HER2-mediated cell signaling in CHO cells expressing human HER2. BMC Cancer 2018, 18, 238. [Google Scholar] [CrossRef]
- Laux, I.; Jain, A.; Singh, S.; Agus, D.B. Epidermal growth factor receptor dimerization status determines skin toxicity to HER-kinase targeted therapies. Br. J. Cancer 2006, 94, 85–92. [Google Scholar] [CrossRef][Green Version]
- Arkhipov, A.; Shan, Y.; Kim, E.T.; Dror, R.O.; Shaw, D.E. Her2 activation mechanism reflects evolutionary preservation of asymmetric ectodomain dimers in the human EGFR family. Elife 2013, 2, e00708. [Google Scholar] [CrossRef]
| Protein Change | Pfam Domain [8] | Tumor Type | Impact | HER2 | Phosphorylation | Reference |
|---|---|---|---|---|---|---|
| L12R | - | Breast Cancer | ND | Negative | ND * | [35] |
| A20T | Lung cancer | ND | ND | ND | [24] | |
| L49H | Lung cancer | ND | Negative | ND | [21] | |
| L49H | Glioblastoma | ND | ND | ND | [20] | |
| E139D | Receptor L | Breast Cancer | ND | Negative | ND | [35] |
| E139G | Breast Cancer | ND | Negative | ND | [35] | |
| R157W | Bladder cancer (MPUC) | ND | Negative | ND | [32] | |
| T216S | Furin-like | Lung cancer | ND | Negative | ND | [21] |
| T216S | Glioblastoma | ND | ND | High | [20] | |
| I263T | Colorectal cancer | ND | Negative | ND | [36] | |
| G309A | Breast Cancer | Activating | Negative | ND | [27] | |
| G309E | Breast Cancer | Activating | Positive | ND | [24] | |
| S310F | Gastric cancer | Activating | ND | High | [38] | |
| S310F(2) | Breast Cancer | Negative | High | [26] | ||
| S310F | Lung cancer | Negative | High | [20] | ||
| S310F | Breast Cancer | Negative | High | [25] | ||
| S310F(2) | Colorectal cancer | Negative | High | [37] | ||
| S310F | Breast Cancer | Negative | High | [28] | ||
| S310F | Lung cancer | Positive | High | [22] | ||
| S310F | Breast Cancer | Negative | High | [29] | ||
| S310F(4) | Bladder cancer (MPUC) | Negative | High | [32] | ||
| S310F | Adnexal cancer | Negative | High | [33] | ||
| S310F | Breast Cancer | Negative | High | [29] | ||
| S310F(2) | Breast Cancer | Positive | High | [30] | ||
| S310F | Lung cancer | ND | High | [23] | ||
| S310F | Breast Cancer | Negative | High | [31] | ||
| S310F(4) | Breast Cancer | Positive | ND | [35] | ||
| S310Y | Lung cancer | Activating | ND | High | [24] | |
| S310Y | Lung cancer | ND | High | [20] | ||
| S310Y | Colorectal cancer | Positive | High | [21] | ||
| S310Y | Bladder cancer (MPUC) | Negative | High | [20] | ||
| C311R and E321G | Lung cancer | ND | Negative | ND | [21] | |
| C311R | Glioblastoma | ND | ND | Low | [20] | |
| N319D | Lung cancer | ND | Negative | ND | [21] | |
| N319D | Glioblastoma | ND | ND | ND | [20] | |
| E321G | Glioblastoma | ND | ND | Low | [20] | |
| D326G and C334S | Lung cancer | ND | Negative | ND | [21] | |
| D326G | Glioblastoma | ND | ND | Low | [20] | |
| C334S | Glioblastoma | ND | ND | ND | [20] | |
| A466T | Receptor L | Colorectal cancer | ND | Negative | ND | [36] |
| A466V | Breast Cancer | Activating | Negative | High | [35] | |
| C515R | Growth factor receptor | Breast Cancer | Activating | Negative | High | [35] |
| T526A | Breast Cancer | Activating | Negative | High | [35] |
| Cell Line | Protein Change | Pfam Domain [8] | Lineage | Impact | Phosphorylation |
|---|---|---|---|---|---|
| SW1271 | S22N | - | Lung | ND * | ND |
| HEC59 | R100W | Receptor L | Endometrium | ND | ND |
| HEC108 | T166M | Endometrium | ND | ND | |
| SBC1 | Q178H | Lung | ND | ND | |
| MOLT16 | R217H | Furin-like | Haematopoietic and lymphoid tissue | ND | ND |
| NALM6 | R226H | Haematopoietic and lymphoid tissue | ND | ND | |
| KM12 | P230L | Large intestine | ND | ND | |
| DSH1 | D277H | Urinary | ND | ND | |
| 5637 | S310F | Urinary | Activating | High | |
| DSH1 | Urinary | ||||
| OACM51 | Oesophagus | ||||
| HRT18 | L313I | Intestine | ND | ND | |
| NCIH2110 | N319Y | Lung | ND | ND | |
| HCC1359 | T328S | Lung | ND | ND | |
| NCIH1563 | S335C | Lung | ND | ND | |
| Jurkat | S335I | - | Haematopoietic and lymphoid tissue | ND | ND |
| OC314 | A386T | Receptor L | Ovary | ND | ND |
| OC316 | A386T | Ovary | ND | ND | |
| NCIN87 | F425L | Stomach | ND | ND | |
| NCIN87 | L436V | Stomach | ND | ND | |
| SET2 | T444S | Haematopoietic and lymphoid tissue | ND | ND | |
| M059J | W452S | Central nervous system | ND | ND | |
| ISTSL1 | R499Q | - | Lung | ND | ND |
| SUPB8 | R499W | - | Haematopoietic and lymphoid tissue | ND | ND |
| Jurkat | T479M | Receptor L | Haematopoietic and lymphoid tissue | ND | ND |
| RL952 | G518V | Growth factor receptor | Endometrium | ND | ND |
| NCIH1793 | V541M | Lung | ND | ND | |
| NCIH740 | R558M | Lung | ND | ND | |
| Karpas45 | A586G | Haematopoietic and lymphoid tissue | ND | ND |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, J.W.; Kim, S.; Ha, S.; Choi, B.; Kim, S.; Im, S.-A.; Yoon, T.-Y.; Chung, J. The HER2 S310F Mutant Can Form an Active Heterodimer with the EGFR, Which Can Be Inhibited by Cetuximab but Not by Trastuzumab as well as Pertuzumab. Biomolecules 2019, 9, 629. https://doi.org/10.3390/biom9100629
Shin JW, Kim S, Ha S, Choi B, Kim S, Im S-A, Yoon T-Y, Chung J. The HER2 S310F Mutant Can Form an Active Heterodimer with the EGFR, Which Can Be Inhibited by Cetuximab but Not by Trastuzumab as well as Pertuzumab. Biomolecules. 2019; 9(10):629. https://doi.org/10.3390/biom9100629
Chicago/Turabian StyleShin, Jung Won, Soohyun Kim, Suji Ha, Byungsan Choi, Seongyeong Kim, Seock-Ah Im, Tae-Young Yoon, and Junho Chung. 2019. "The HER2 S310F Mutant Can Form an Active Heterodimer with the EGFR, Which Can Be Inhibited by Cetuximab but Not by Trastuzumab as well as Pertuzumab" Biomolecules 9, no. 10: 629. https://doi.org/10.3390/biom9100629
APA StyleShin, J. W., Kim, S., Ha, S., Choi, B., Kim, S., Im, S.-A., Yoon, T.-Y., & Chung, J. (2019). The HER2 S310F Mutant Can Form an Active Heterodimer with the EGFR, Which Can Be Inhibited by Cetuximab but Not by Trastuzumab as well as Pertuzumab. Biomolecules, 9(10), 629. https://doi.org/10.3390/biom9100629





