Methylation and Acetylation Enhanced the Antidiabetic Activity of Some Selected Flavonoids: In Vitro, Molecular Modelling and Structure Activity Relationship-Based Study
Abstract
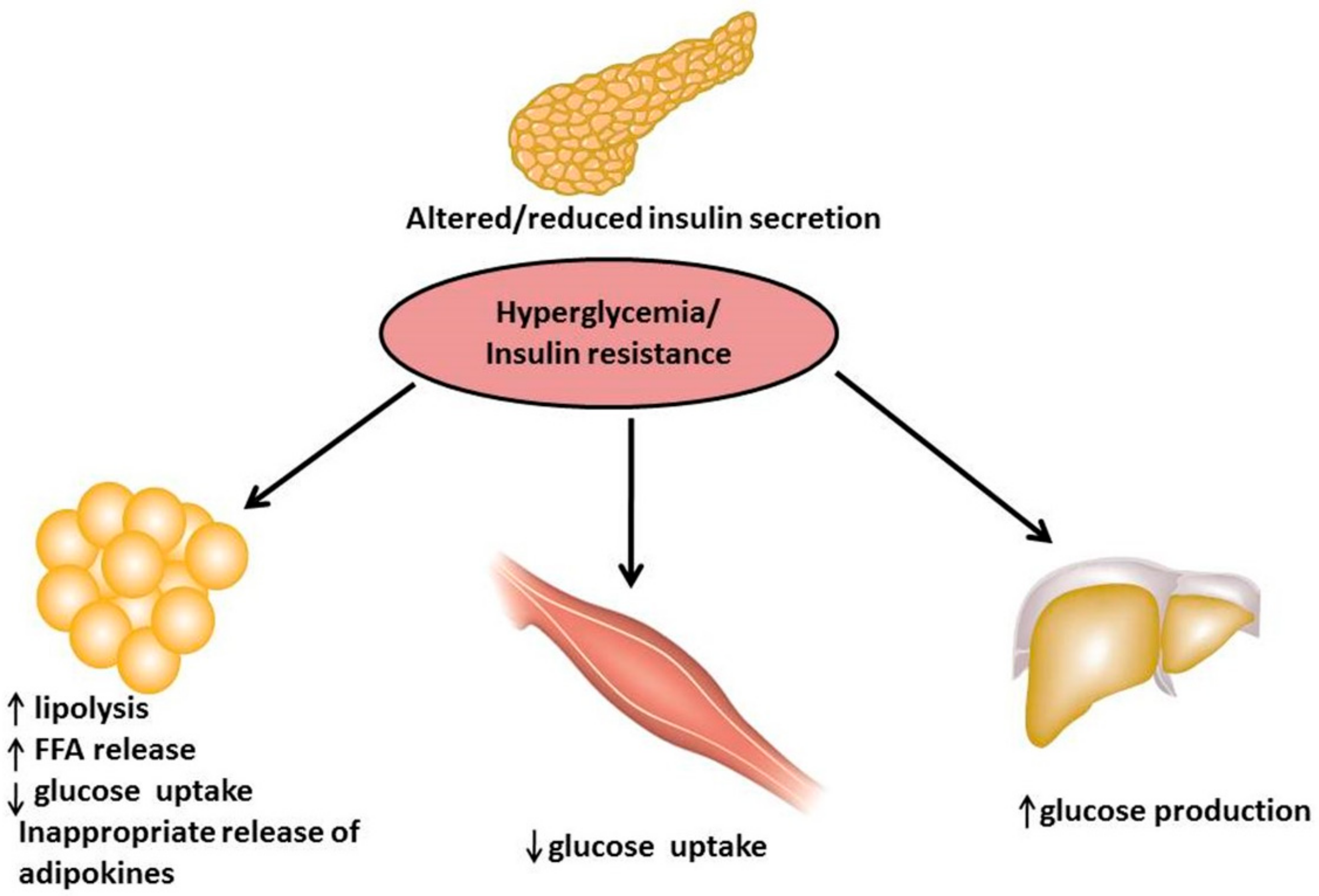
1. Introduction
2. Materials and Methods
2.1. Chemicals, Reagents and Cells
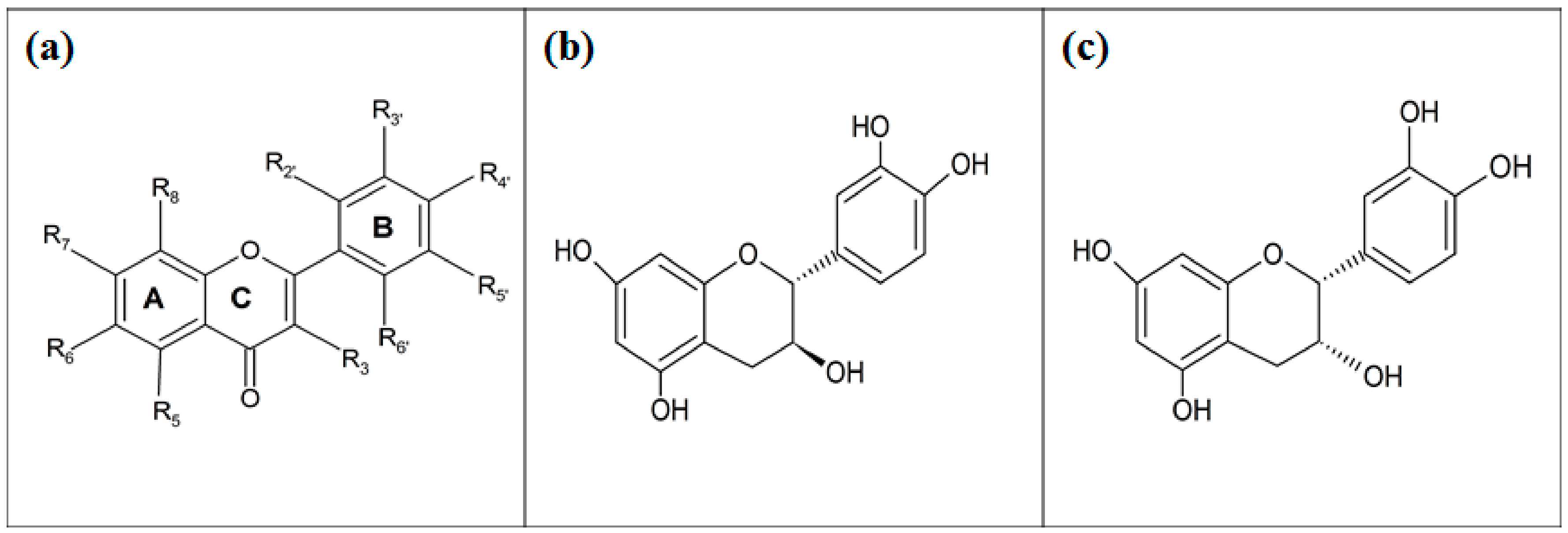
2.2. Compounds’ Isolation and Derivatisation
2.3. In Vitro Antidiabetic Evaluation
2.3.1. Cell Viability Assay (RIN-5F Pancreatic Cells and 3T3-L1 Adipocyte Cells)
2.3.2. Insulin Secretion Assay
2.3.3. Adipogenesis Assay
2.3.4. Fluorescence Glucose Uptake Measurement Assay
2.4. Molecular Antidiabetic Evaluation
Leptin, Adiponectin, Tumor Necrosis Factor-A (Tnf-A) and Retinol Binding Protein-4 (Rbp-4) Assays
2.5. Western Blotting Assay
2.6. In-Silico Molecular Docking of Glucose Transporter 1 (Glut 1)
2.7. Statistical Analysis
3. Results
3.1. In Vitro Antidiabetic Evaluation
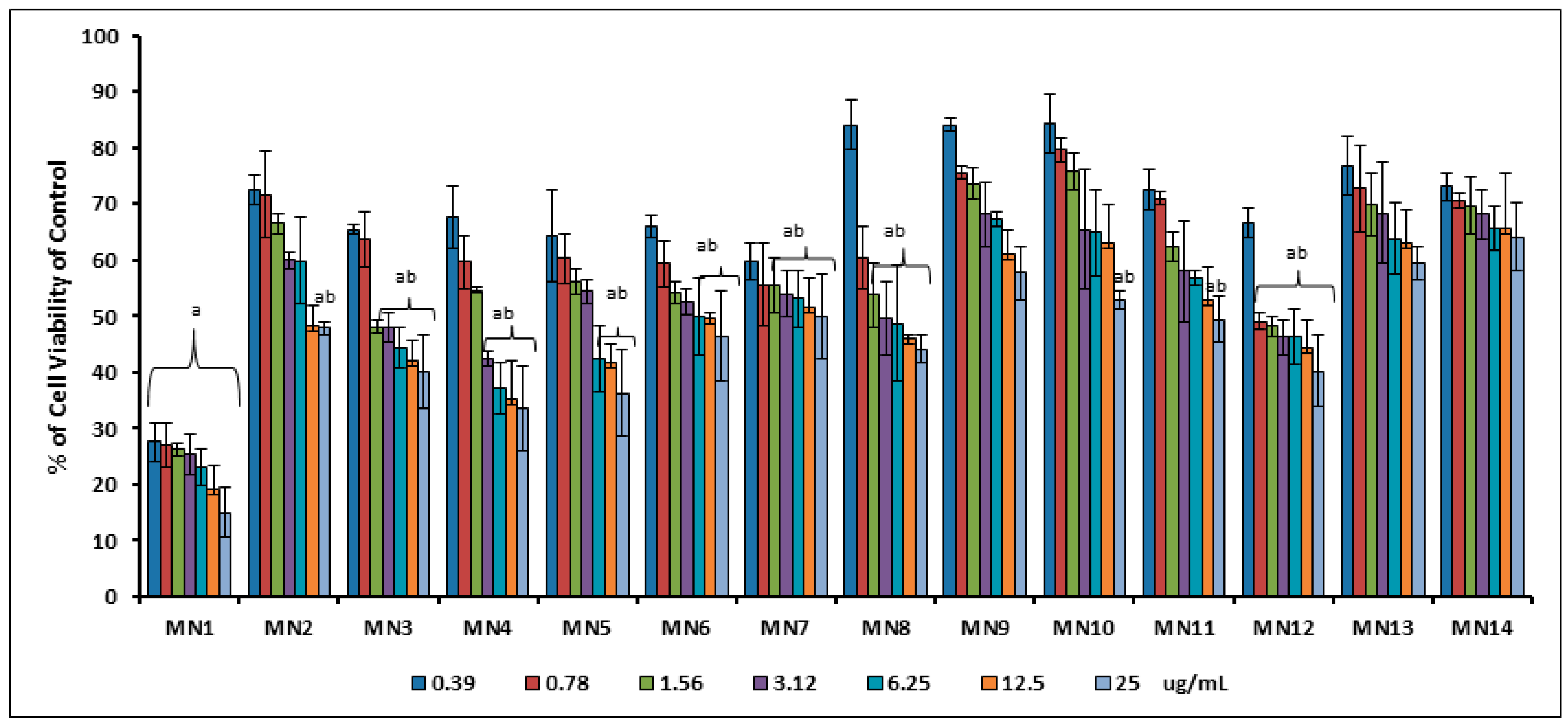
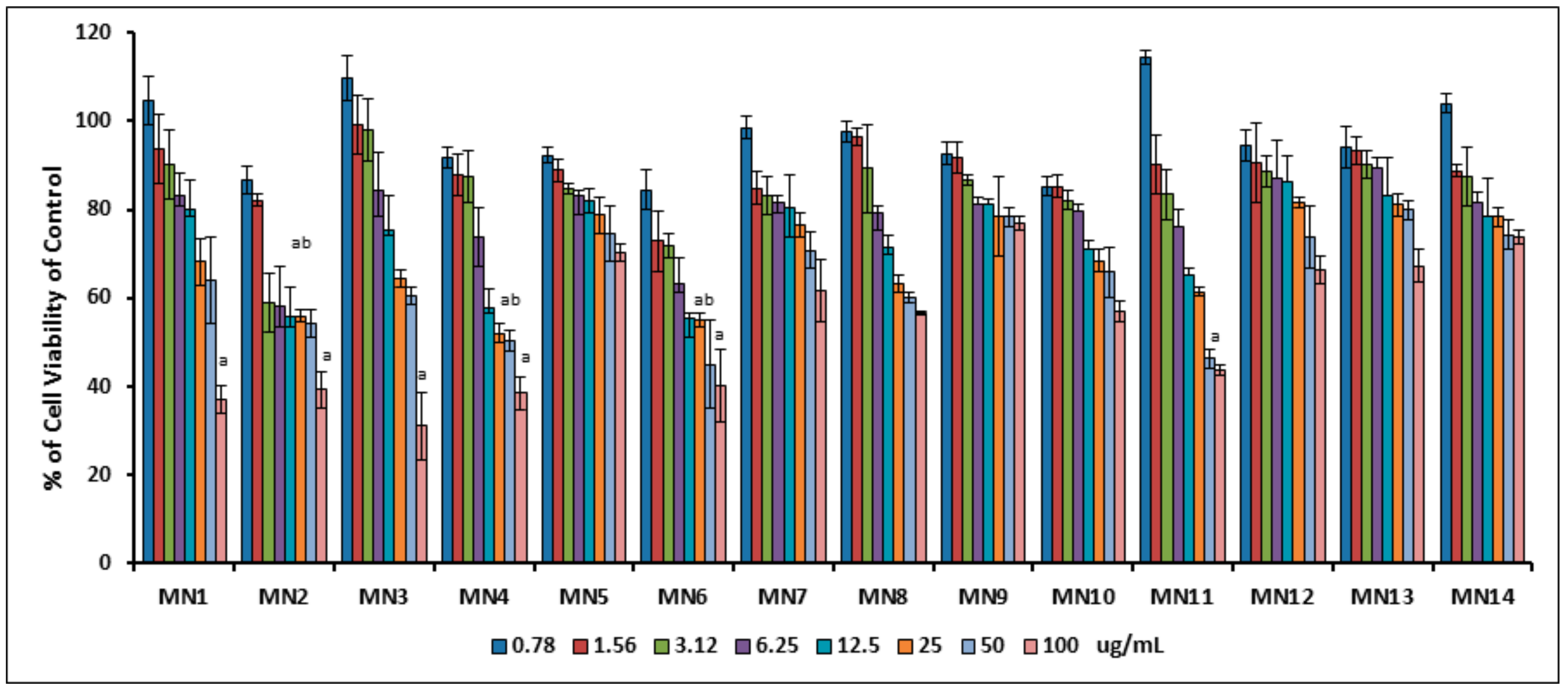
3.1.1. Viability Study of RIN-5F Pancreatic Cells
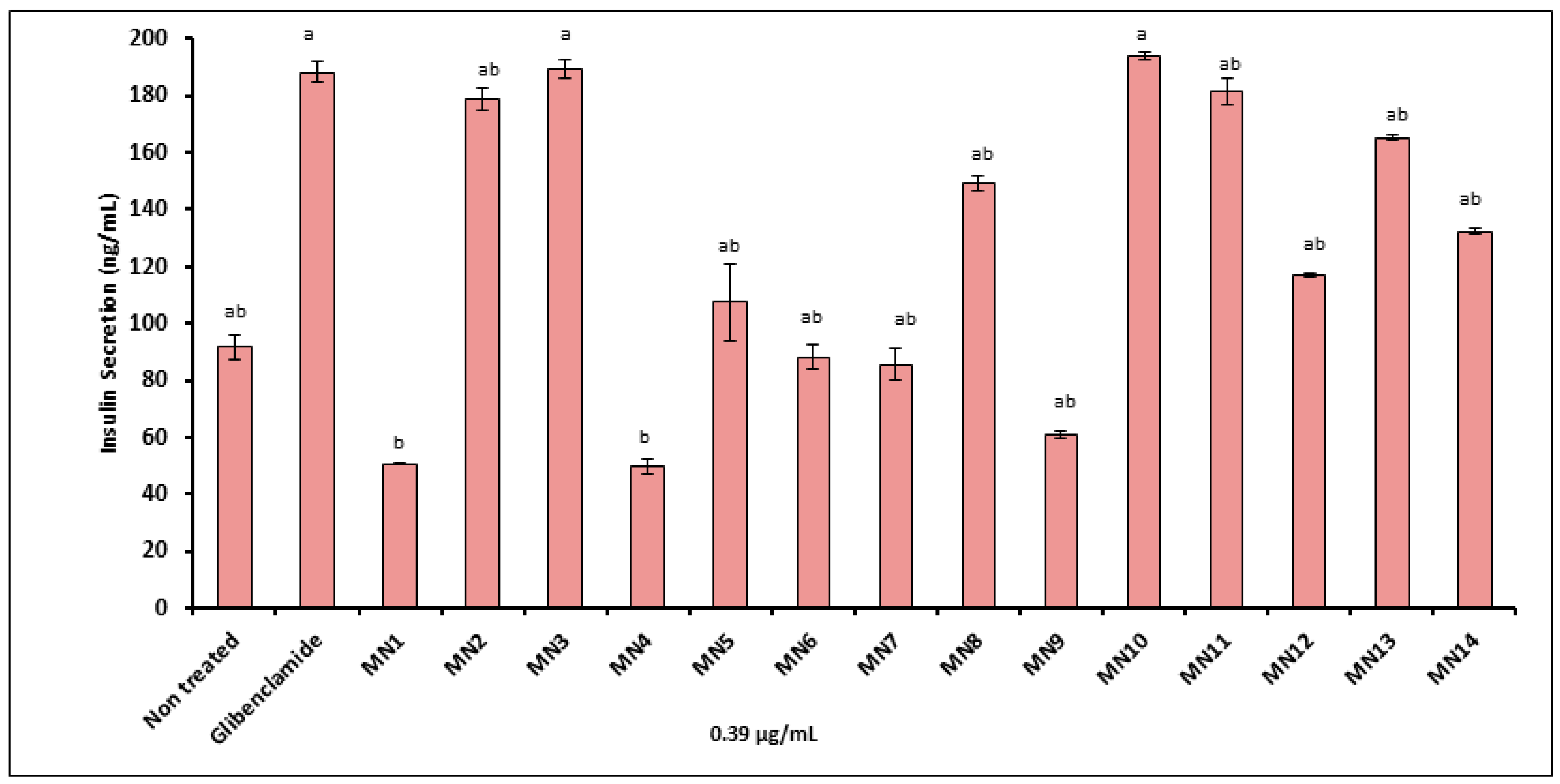
3.1.2. Insulin Secretion Assay (Rin-5F Pancreatic Cells)
3.1.3. Viability Study of 3T3-L1 Preadipocyte Cells
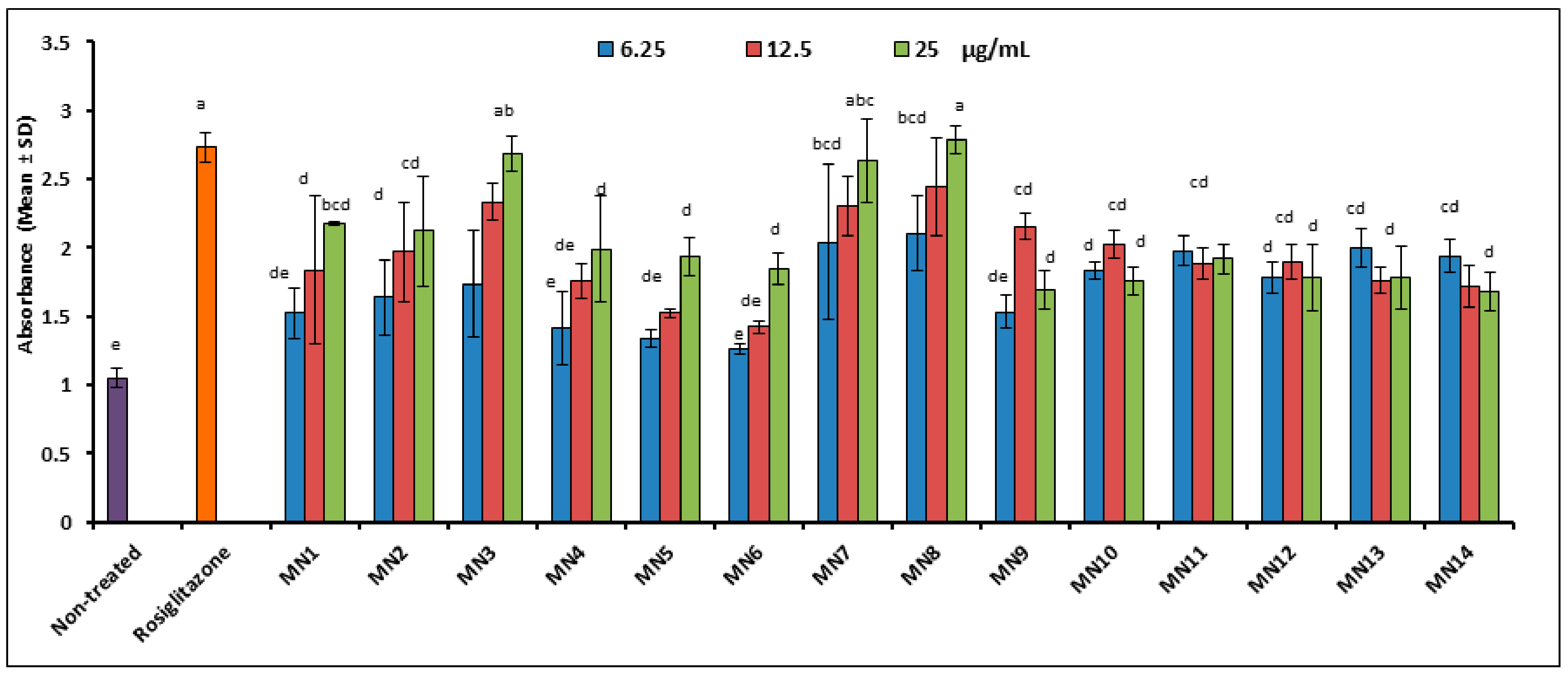
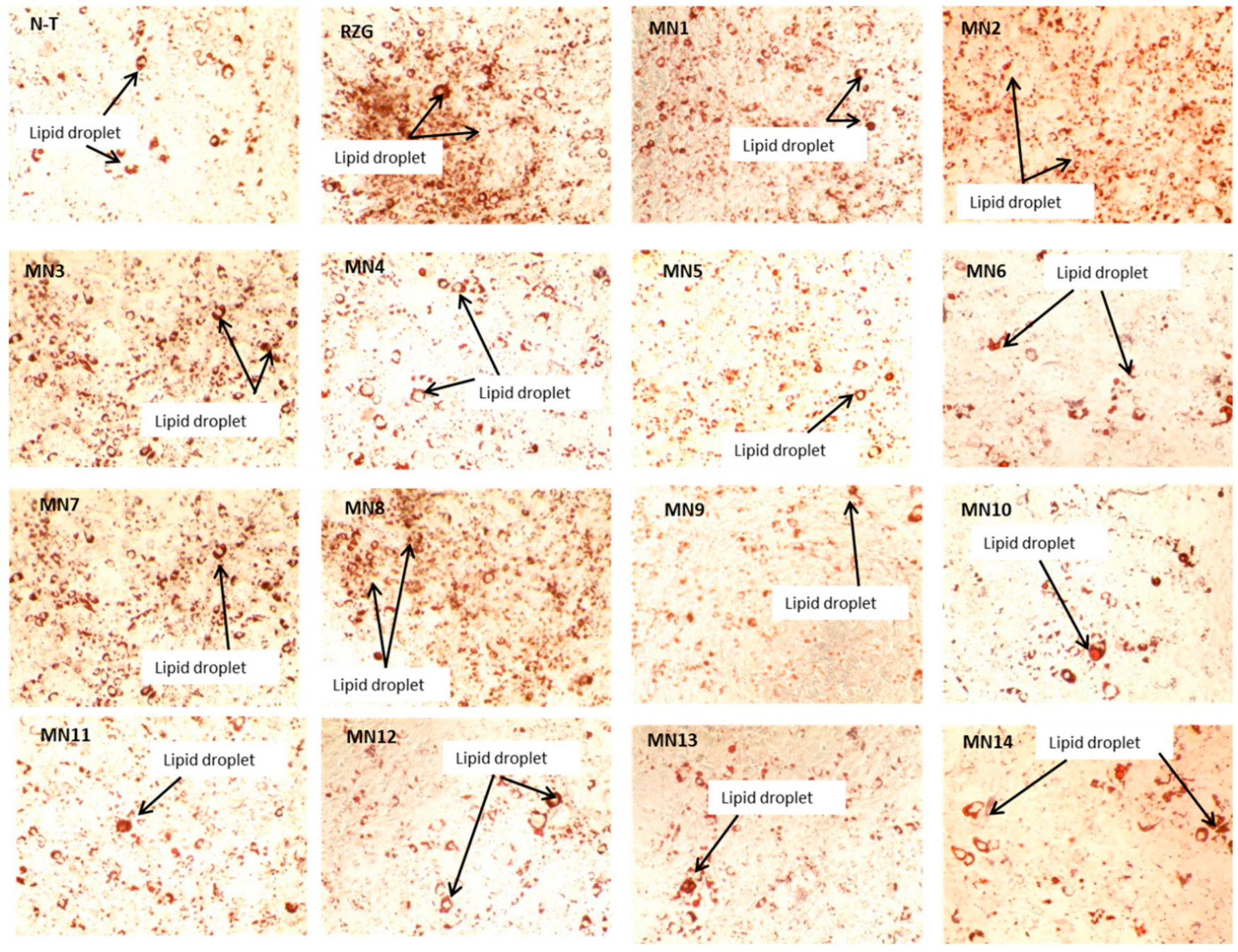
3.1.4. Adipogenesis (Oil Red O Staining)
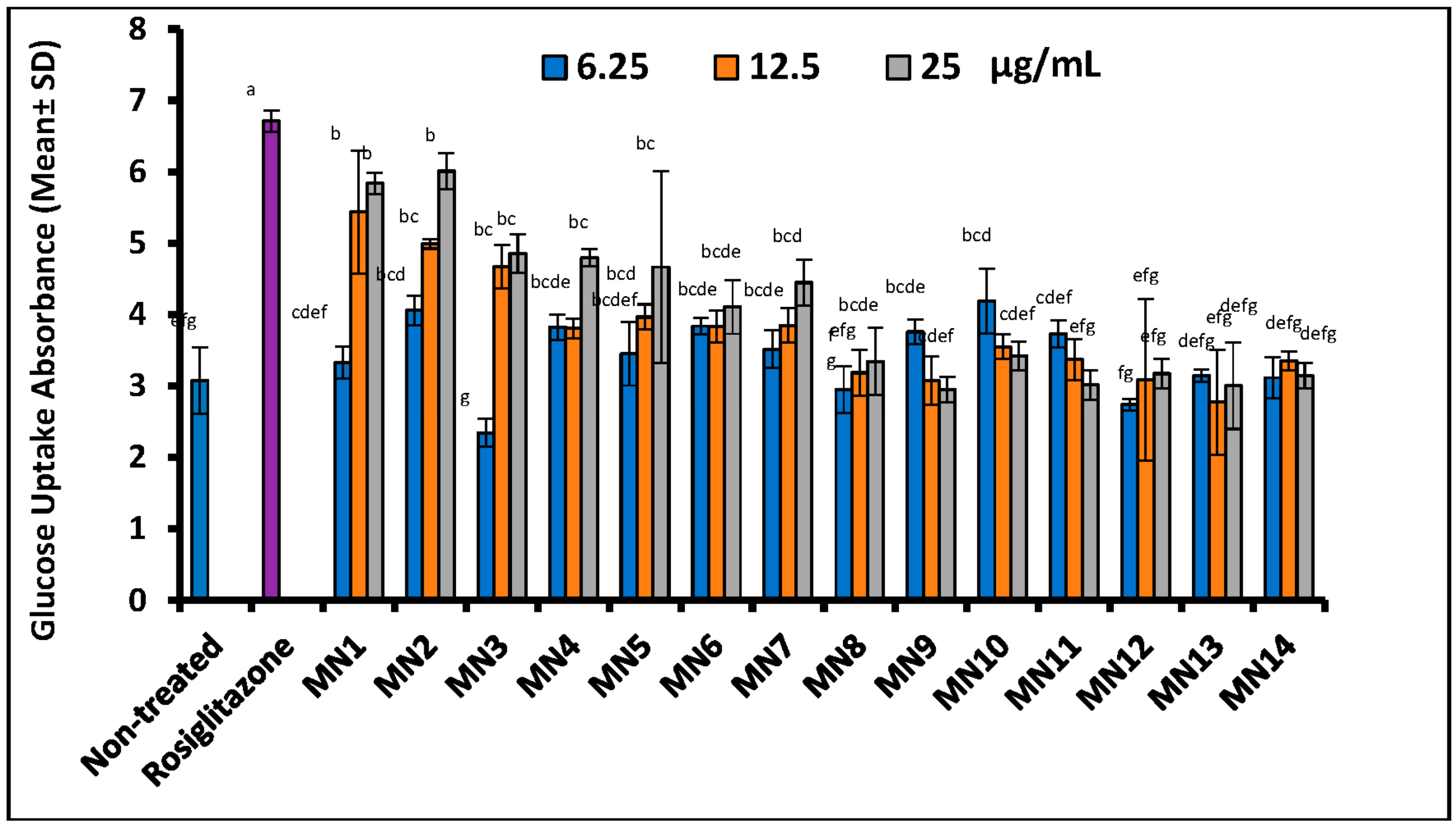
3.1.5. Fluorescent Glucose [2-NDBG] Uptake Measurement
3.2. Molecular Antidiabetic Evaluation
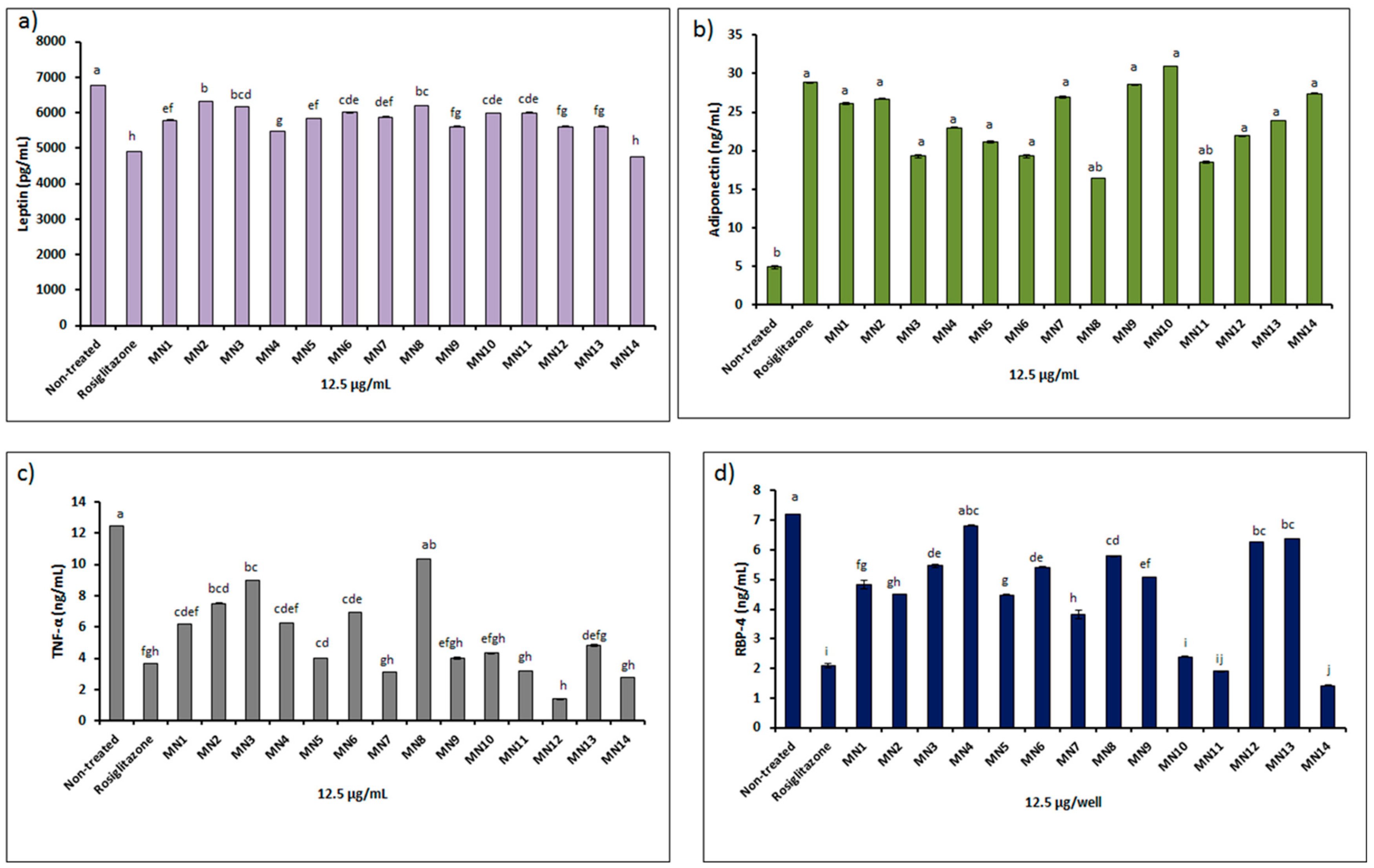
3.2.1. Measurement of Adipokines (Leptin, Adiponectin, Tumor Necrosis Factor-A (TNF-A), Retinol Binding Protein-4 (RBP-4)) of 3T3-L1 Adipocyte Cells Upon Flavonoids Treatment
- For adiponectin measurement, at 12 μg/mL, except for acetate of norwogonin (MN8) and kaempferol (MN11), result of the tested flavonoids showed statistically increased in adiponectin concentration (ng/mL) as compared to the non-treated group (p < 0.05). Hypolaetin (MN10), showed the highest concentration, followed by isoscutellarein (MN9), norwogonin (MN7), (-)-epicatechin (MN14), methyl ether of wogonin (MN2), wogonin (MN1), (+)-catechin (MN13), techtochrysin (MN4), quercetin (MN12), 8-hydroxy-7-methoxyflavone (MN5), acetate of wogonin (MN3) and chrysin (MN6), respectively.
- For tumor necrosis factor, i.e., α (TNF-α), at 12 μg/mL, except for acetate of norwogonin (MN8), the result of the tested flavonoids displayed statistically decreased in TNF-α concentration (ng/mL) as compared to the non-treated group (p < 0.05). Quercetin (MN12) showed the lowest concentration of TNF-α, followed by (-)-epicatechin (MN14), norwogonin (MN7), kaempferol (MN11), (+)-catechin (MN13), wogonin (MN1), techtochrysin (MN4), chrysin (MN6), methyl ether of wogonin (MN2) and the least concentration was shown by the acetate of wogonin (MN3), respectively.
- Subsequently, for retinol binding protein IV (RBP-IV), at 12 μg/mL, except for techtochrysin (MN4), result of the tested flavonoids exhibited statistically reduced in RBP-IV concentration (ng/mL) as compared to the non-treated group (p < 0.05). (-)-epicatechin (MN14) showed the lowest concentration, followed by kaempferol (MN11), hypolaetin (MN10), norwogonin (MN7), 8-hydroxy-7-methoxyflavone (MN5), methyl ether of wogonin (MN2), wogonin (MN1), isoscutellarein (MN9), chrysin (MN6), acetate of norwogonin (MN3), acetate of norwogonin (MN8), quercetin (MN12), and the least concentration was displayed by (+)-catechin (MN13), respectively.
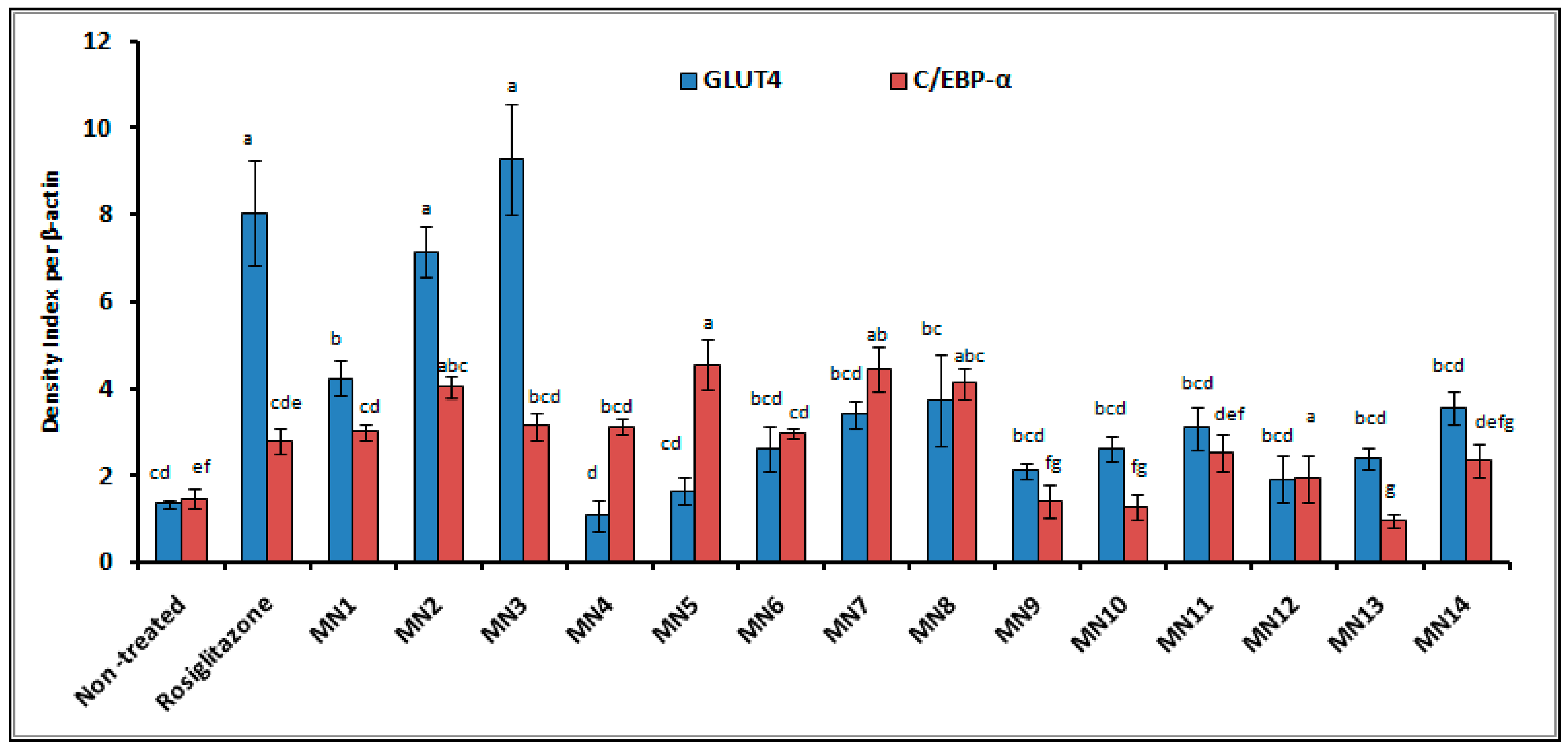
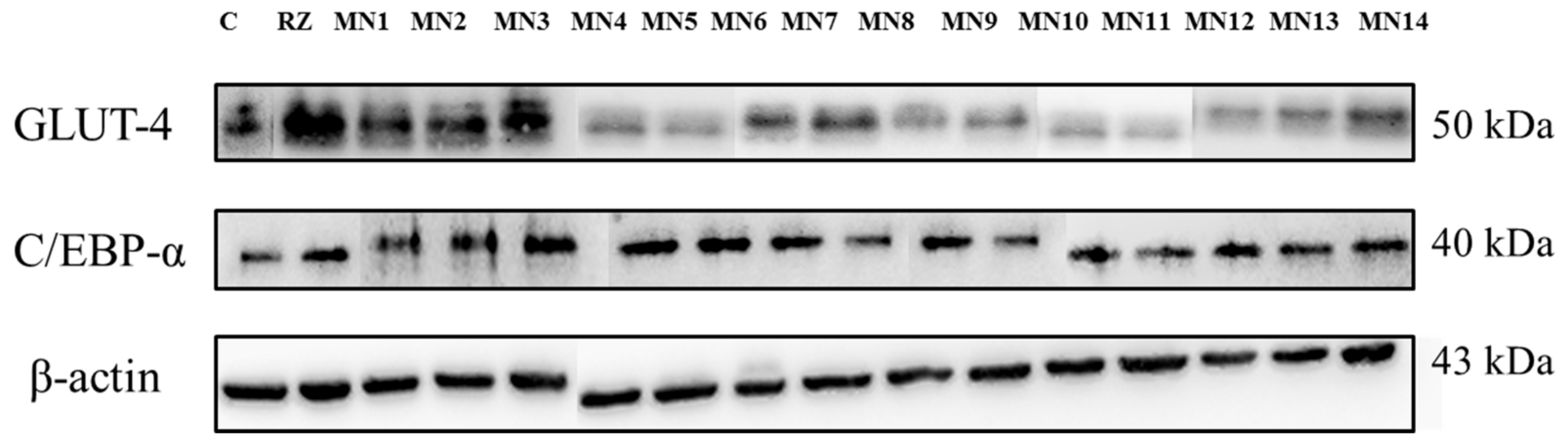
3.2.2. Western Blotting
3.3. In Silico Molecular Docking (GLUT 1)
4. Discussion
4.1. In Vitro Antidiabetic Evaluation on RIN-5F Pancreatic Cells and 3T3-L1 Adipocytes
4.2. In Vitro Molecular Antidiabetic Evaluation
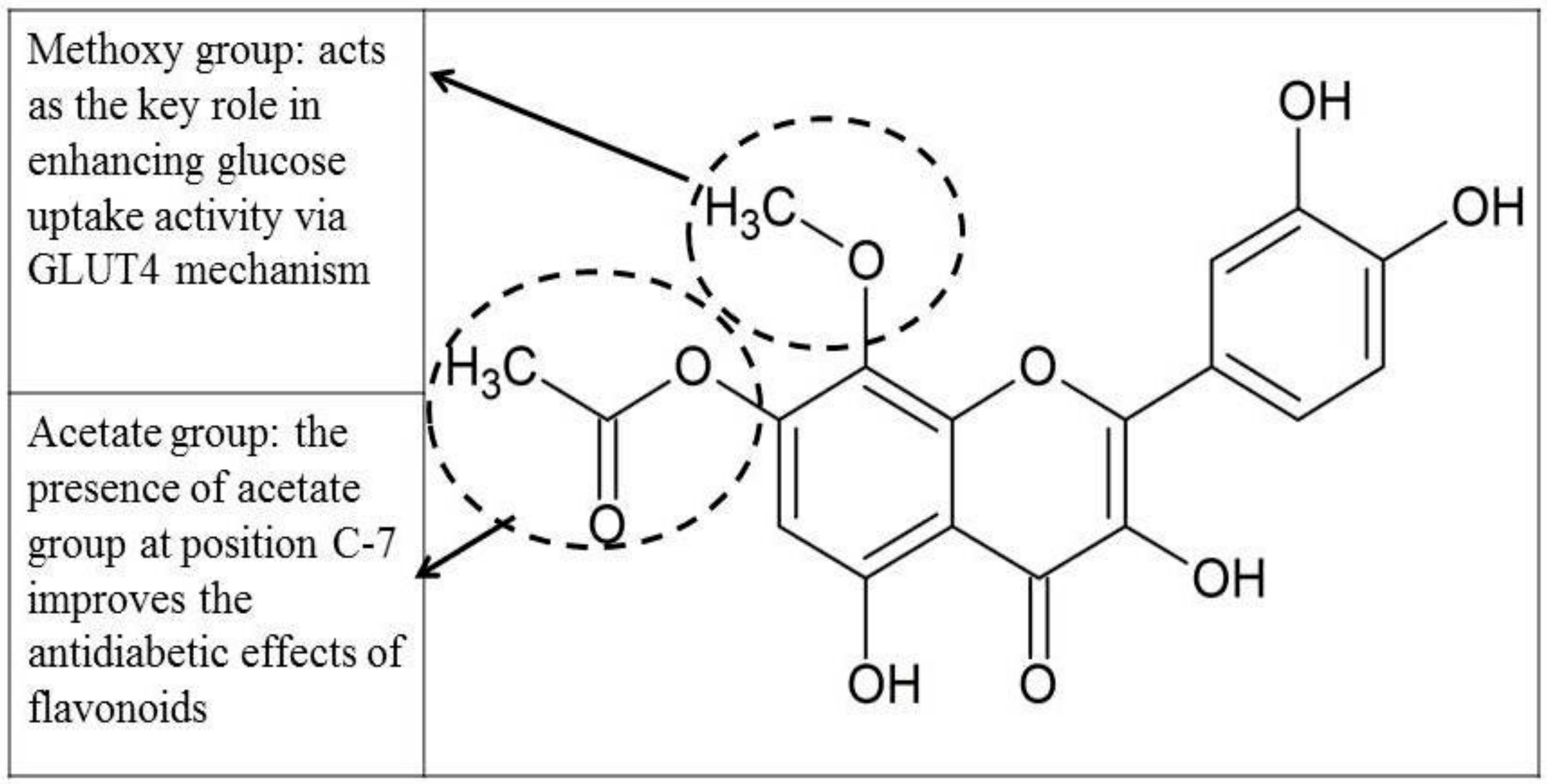
4.3. In Silico Molecular Docking Using GLUT 1
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Diabetes Association. 10. Microvascular complications and foot care: Standards of medical care in diabetes-2018. Diabetes Care 2018, 41, S105–S118. [Google Scholar] [CrossRef] [PubMed]
- Riddy, D.M.; Delerive, P.; Summers, R.; Sexton, P.M.; Langmead, C.J. G protein–coupled receptors targeting insulin resistance, obesity, and type 2 diabetes mellitus. Pharmacol. Rev. 2018, 70, 39–67. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.G.; Halter, J.B. The pathophysiology of hyperglycemia in order adults: The clinical consideration. Diabetes Care 2017, 40, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, M. The pathobiology of diabetic complications: A unifying mechanism. Diabetes 2005, 54, 1615–1625. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Leung, L.K.; Su, Y.; Chen, R.; Zhang, Z.; Huang, Y.; Chen, Z.Y. Theaflavins in black tea and catechins in green tea are equally effective antioxidants. J. Nutr. 2001, 131, 2248–2251. [Google Scholar] [CrossRef] [PubMed]
- De Souza, V.T.; de Franco, E.P.; de Araújo, M.E.; Messias, M.C.; Priviero, F.; Frankland Sawaya, A.C.; de Oliveira, C.P. Characterization of the antioxidant activity of aglycone and glycosylated derivatives of hesperetin: An in-vitro and in-vitro study. J. Mol. Recognit. 2016, 29, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Z.; Chen, C.Z.; Min, H.; Ai-Zhi, W.; Zeren, D.B.; Suolang, Q.K. Structure-activity relationships of antioxidant activity in vitro about flavonoids isolated from Pyrethrum tatsienense. J. Intercult. Ethnopharmacol. 2014, 3, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Sarian, M.N.; Ahmed, Q.U.; Mat So’ad, S.Z.; Alhassan, A.M.; Murugesu, S.; Perumal, V.; Syed Mohamad, S.N.A.; Khatib, A.; Latip, J. Antioxidant and antidiabetic effects of flavonoids: A structure-activity relationship based study. Biomed. Res. Int. 2017, 2017, 8386065. [Google Scholar] [CrossRef] [PubMed]
- Zeka, K.; Ruparelia, K.; Arroo, R.R.J.; Budriesi, R.; Micucci, M. Flavonoids and their metabolites: Prevention in cardiovascular diseases and diabetes. Diseases 2017, 5, 19. [Google Scholar] [CrossRef]
- Soares, J.M.D.; Leal, A.E.B.P.; Silva, J.C.; Almeida, J.R.G.S.; Oliveira, H.P. Influence of flavonoids on mechanism of modulation of insulin secretion. Pharmacogn. Mag. 2017, 13, 639–646. [Google Scholar] [PubMed]
- Testa, R.; Bonfigli, A.R.; Genovese, S.; De Nigris, V.; Ceriello, A. The possible role of flavonoids in the prevention of diabetic complications. Nutrients 2016, 8, 310. [Google Scholar] [CrossRef] [PubMed]
- Vinayagam, R.; Xu, B. Antidiabetic properties of dietary flavonoids: A cellular mechanism review. Nutr. Metab. 2015, 12, 60. [Google Scholar] [CrossRef] [PubMed]
- Nicolle, E.; Souard, F.; Faure, P.; Boumendjel, A. Flavonoids as promising lead compounds in type 2 diabetes mellitus: Molecules of interest and structure-activity relationship. Curr. Med. Chem. 2011, 18, 2661–2672. [Google Scholar] [CrossRef] [PubMed]
- Kyriakis, E.; Stravodimos, G.A.; Kantsadi, A.L.; Chatzileontiadou, D.S.M.; Skamnaki, V.T.; Leonidas, D.D. Natural flavonoids as antidiabetic agents. The binding of gallic and ellagic acids to glycogen phosphorylase b. FEBS Lett. 2015, 589, 1787–1794. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Kogami, Y.; Nakamura, S.; Sugiyama, T.; Yoshikawa, T. Structural requirements of flavonoids for the 3T3-L1 cells. Bioorg. Med. Chem. 2011, 19, 2835–2841. [Google Scholar] [CrossRef] [PubMed]
- Alhassan, M.A.; Ahmed, Q.U.; Latip, J.; Shah, S.A.A. A new sulphated flavone and other phytoconstituents from the leaves of Tetracera indica Merr. and their alpha-glucosidase inhibitory activity. Nat. Prod. Res. 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Subash-Babu, P.; Ignachimuthu, S.; Alshatwi, A.A. Nymphayol increases glucose-stimulated insulin secretion by RIN-5F cells and GLUT4-mediated insulin sensitization in type 2 diabetic rat liver. Chem. Biol. Interact. 2015, 226, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid calorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Park, J.H.; Kim, R.Y.; Park, E. Antidiabetic activity of fruits and vegetables commonly consumed in Korea: Inhibitory potential against α glucosidase and insulin like action in-vitro. Food Sci. Biotechnol. 2012, 21, 1187–1193. [Google Scholar] [CrossRef]
- Zhu, H.J.; Ding, H.H.; Deng, J.Y.; Pan, H.; Wang, L.J.; Li, N.S.; Gong, F.Y. Inhibition of preadipocyte differentiation and adipogenesis by zinc-α2-glycoprotein treatment in 3T3-L1 cells. J. Diebetes Investig. 2013, 4, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.M.; Ahmed, Q.U.; Siti Zaiton, M.S.; Latip, J.; Taher, M.; Tengku, M.F.S.; Sarian, M.N.; Alhassan, M.A.; Zakaria, Z.A. Flavonoids from Tetracera indica Merr. induce adipogenesis and exert glucose uptake activities in 3T3-L1 adipocyte cells. BMC Complement. Altern. Med. 2017, 17, 431. [Google Scholar] [CrossRef]
- Nouroozi, R.V.; Noroozi, M.V.; Ahmadizadeh, M. Determination of protein concentration using bradford microplate protein quantification assay. IEJM 2015, 4, 11–17. [Google Scholar]
- Mardian, E.B.; Bradley, R.M.; Aristizabal- Henao, J.J.; Marvyn, P.M.; Moes, K.A.; Bombardier, E.; Tupling, A.R.; Stark, K.D.; Duncan, R.E. Agpat4/Lpaatδ deficiency highlights the molecular heterogeneity of epididymal and perirenal white adipose depots. J. Lipid Res. 2017, 58, 2037–2050. [Google Scholar] [CrossRef] [PubMed]
- ImageJ. Available online: https://imagej.en.softonic.com/download (accessed on 30 November 2017).
- AutoDock Tools. Available online: http://www.scripps.edu (accessed on 3 September 2017).
- Discovery Studio Visualizer. Available online: http://www.accelrys.com (accessed on 3 September 2017).
- RCSB Protein Data Bank. Available online: http://www.rcsb.org (accessed on 3 September 2017).
- Alhassan, A.M.; Ahmed, Q.U.; Latip, J.; Shah, S.A.A.; Khan, A.Y.F.; Sarian, M.N.; Wahab, R.A.; Taher, M.; Abdullahi, M.I.; Khatib, A. Phytoconstituents from Vernonia glaberrima Welw. Ex O. Hoffm. leaves and their cytotoxic activities on a panel of human cancer cell lines. S. Afr. J. Bot. 2018, 116, 16–24. [Google Scholar] [CrossRef]
- Szollosi, A.; Nenquin, M.; Bryan, L.A.; Bryan, J.; Henquin, J.C. Glucose stimulates Ca2+ influx and insulin secretion in 2-week-old β-cells lacking ATP-sensitive K+ channels. J. Biol. Chem. 2006, 282, 1747–1756. [Google Scholar] [CrossRef] [PubMed]
- Röder, P.V.; Wu, B.; Liu, Y.; Han, W. Pancreatic regulation of glucose homeostasis. Exp. Mol. Med. 2016, 48, e219. [Google Scholar] [CrossRef] [PubMed]
- Mann, E.; Bellin, M.D. Secretion of insulin in response to diet and hormones. Pancreapedia 2016. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, Y.; Zhu, J.; Li, B.; Li, Z.; Zhu, W.; Shi, J.; Jia, Q.; Li, Y. Recent progress in natural products as DPP-IV inhibitors. Future Med. Chem. 2015, 7, 1079–1089. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, J.; Zhang, P.; Gao, Z.; Zhao, Y.; Qiao, X.; Chen, C. PI4KIIα regulates insulin secretion and glucose homeostasis via a PKD-dependent pathway. Biophys. Rep. 2018, 4, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.L.; Randall, M.D.; Chan, S.L.F. The complex effecys of cannabinoids on insulin secretion from rat isolated islets of Langerhans. Eur. J. Pharmacol. 2013, 1, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakasam, B.; Vareed, S.K.; Olson, L.K.; Nair, M.G. Insulin secretion by bioactive anthocyanins and anthocyanidins present in fruits. J. Agric. Food Chem. 2005, 53, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Stafeev, I.S.; Vorotnikov, A.V.; Ratner, E.I.; Menshikov, M.Y.; Parfyonova, Y.V. Latent inflammation and insulin resistance in adipose tissue. Int. J. Endocrinol. 2017, 2017, 5076732. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, L.; Li, Y.; Wuang, L.; Liu, Y.; Pang, N.; Luo, Y.; He, J.; Zhang, L.; Chen, N.; et al. Transplantation of normal adipose tissue improves blood flow and reduces inflammation in high fat fed mice with hindlimb ischemia. Front. Physiol. 2018, 9, 197. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Yang, Y.; Zhou, L.; Jiang, B.; Jin, H.; Chen, M. Ginsenoside Rb1 stimulates glucose uptake through insulin-like signaling pathway in 3T3-L1 adipocytes. J. Endocrinol. 2008, 198, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Marín-Juez, R.; Rovira, M.; Crespo, D.; Vaart, M.; Spaink, H.P.; Planas, J.V. GLUT2-mediated glucose uptake and availability are required for embryonic brain development in zebrafish. J. Cereb. Blood Flow Metab. 2015, 35, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Johnston, K.; Sharp, P.; Clifford, M.; Morgan, L. Dietary polyphenols decrease glucose uptake by human intestinal Caco-2 cells. FEBS Lett. 2005, 579, 1653–1657. [Google Scholar] [CrossRef] [PubMed]
- Nomura, M.; Takahashi, T.; Nagata, N.; Tsutsumi, K.; Kobayashi, S.; Akiba, T.; Yokogawa, K.; Moritani, S.; Miyamoto, K. Inhibitory mechanisms of flavonoids on insulin-stimulated glucose uptake in MC3T3-G2/PA6 adipose cells. Biol. Pharm. Bull. 2008, 31, 1403–1409. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Abe, D.; Sekiya, K. Nobiletin enhances differentiation and lipolysis of 3T3-L1 adipocytes. Biochem. Biophys. Res. Commun. 2007, 357, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Dunmore, S.J.; Brown, J.E. The role of adipokines in beta cell failure of type 2 diabetes. J. Endocrinol. 2012, 237, 278–290. [Google Scholar]
- Moller, D.E. Potential role of TNF-alpha in the pathogenesis of insulin resistance and type 2 diabetes. Trends Endocrinol. Metab. 2000, 11, 212–217. [Google Scholar] [CrossRef]
- Graham, T.E.; Yang, Q.; Bluher, M.; Hammarstedt, A.; Ciaraldi, T.P.; Henry, R.R.; Wason, C.J.; Oberbach, A.; Jansson, P.A.; Smith, U.; et al. Retinol-binding protein 4 and insulin resistance in lean, obese, and diabetic subjects. N. Engl. J. Med. 2006, 354, 2552–2563. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Graham, T.E.; Mody, N.; Preitner, F.; Peroni, O.D.; Zabolotny, J.M.; Kotani, K.; Quadro, L.; Kahn, B.B. Serum retinol binding protein 4 contributes to insulin resistance in obesity and type 2 diabetes. Nature 2005, 463, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Czech, M.P. The GLUT4 glucose transporter. Cell Metab. 2007, 5, 237–252. [Google Scholar] [CrossRef] [PubMed]
- Rosen, P.; Nawroth, P.P.; King, G.; Moller, W.; Tritschler, H.J.; Packer, L. The role of oxidative stress in the onset and progression of diabetes and its complications: A summary of a Congress Series sponsored by UNESCO MCBN, the American Diabetes Association and the German Diabetes Society. Diabetes Metab. Res. Rev. 2001, 17, 189–212. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, K.; Finer-Moore, J.S.; Pedersem, B.P.; Caboni, L.; Waight, A.; Hillig, R.C.; Bringmann, P.; Heisler, I.; Müller, T.; Siebeneicher, H.; et al. Mechanism of inhibition of human glucose transporter GLUT1 is conserved between cytochalasin B and phenylalanine amides. Proc. Natl. Acad. Sci. USA 2016, 113, 4711–4716. [Google Scholar] [CrossRef] [PubMed]
- Santer, R.; Klepper, J. Disorders of Glucose Transport. In born Metabolic Diseases: Diagnosis and Treatment; Saudubray, J.M., van den Berghe, G., Walter, J.H., Eds.; Springer: New York, NY, USA, 2011; pp. 175–181. [Google Scholar]
- Mishra, R.K.; Wei, C.; Hresco, R.C.; Bajpai, R.; Heitmeier, M.; Matulis, S.M.; Nooka, A.K.; Rosen, S.T.; Hruz, P.W.; Schiltz, G.E.; et al. In silico modeling-based identification of glucose transporter 4 (GLUT4)-selective inhibitors for cancer therapy. J. Biol. Chem. 2015, 290, 14441–14445. [Google Scholar] [CrossRef] [PubMed]
- Mohan, S.S.; Perry, J.J.; Poulose, N.; Nair, B.G.; Anilkumar, G. Homology modeling of GLUT4, an insulin regulated facilitated glucose transporter and docking studies with ATP and its inhibitors. J. Biomol. Struct. Dyn. 2009, 26, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Tripos. Version 1.3, Tripos; Tripos International: St. Louis, MO, USA, 2011. [Google Scholar]
- Strobel, P.; Allard, C.; Perez-Acle, T.; Calderon, R.; Aldunate, R.; Leighton, F. Myricetin, quercetin and catechin-gallate inhibit glucose uptake in isolated rat adipocytes. Biochem. J. 2005, 386, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.; Kim, H.P.; Park, H. Structure and antiinflammatory activity relationships of wogonin derivatives. Arch. Pharmacal. Res. 2005, 28, 877–884. [Google Scholar] [CrossRef]
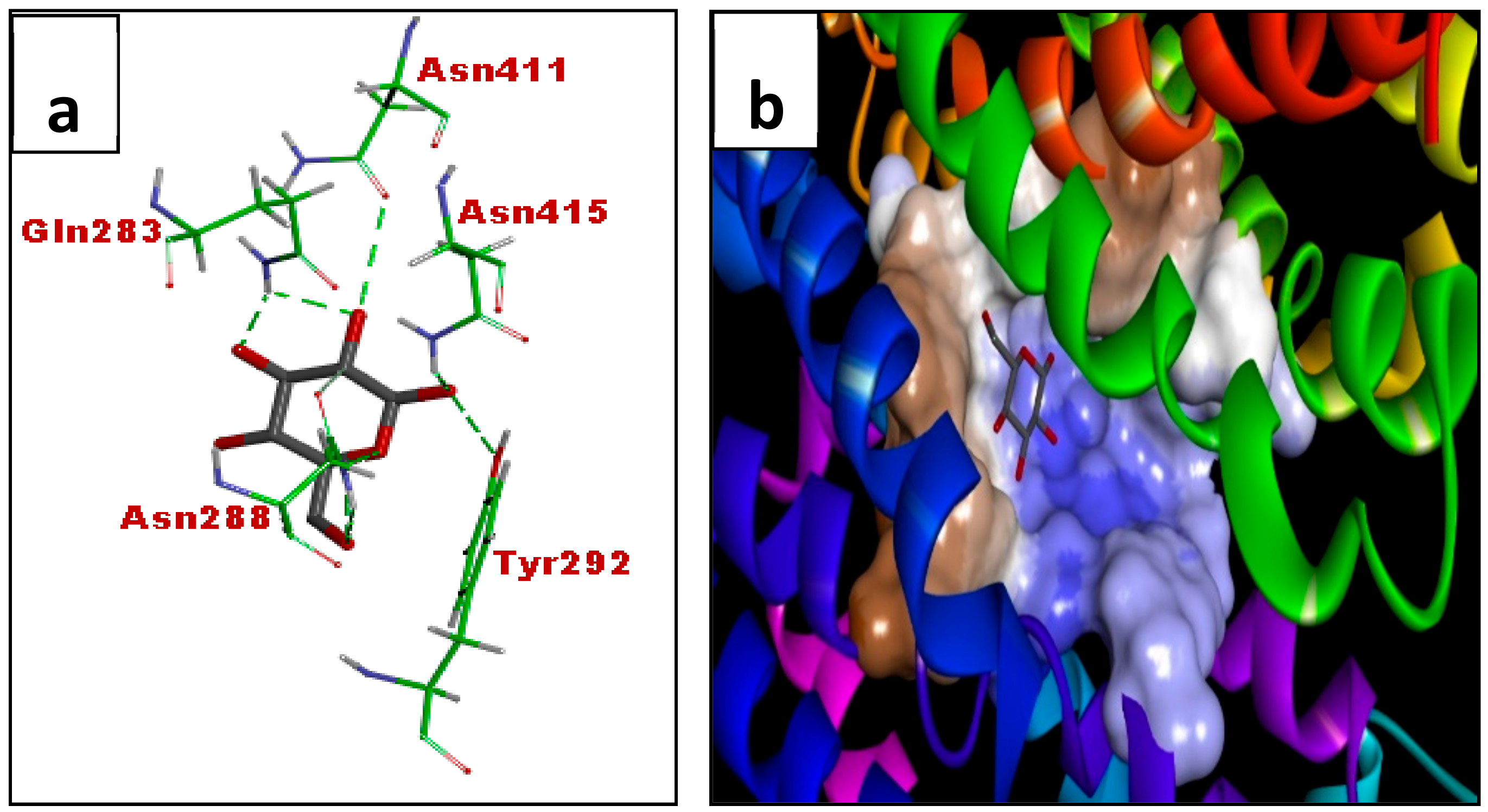
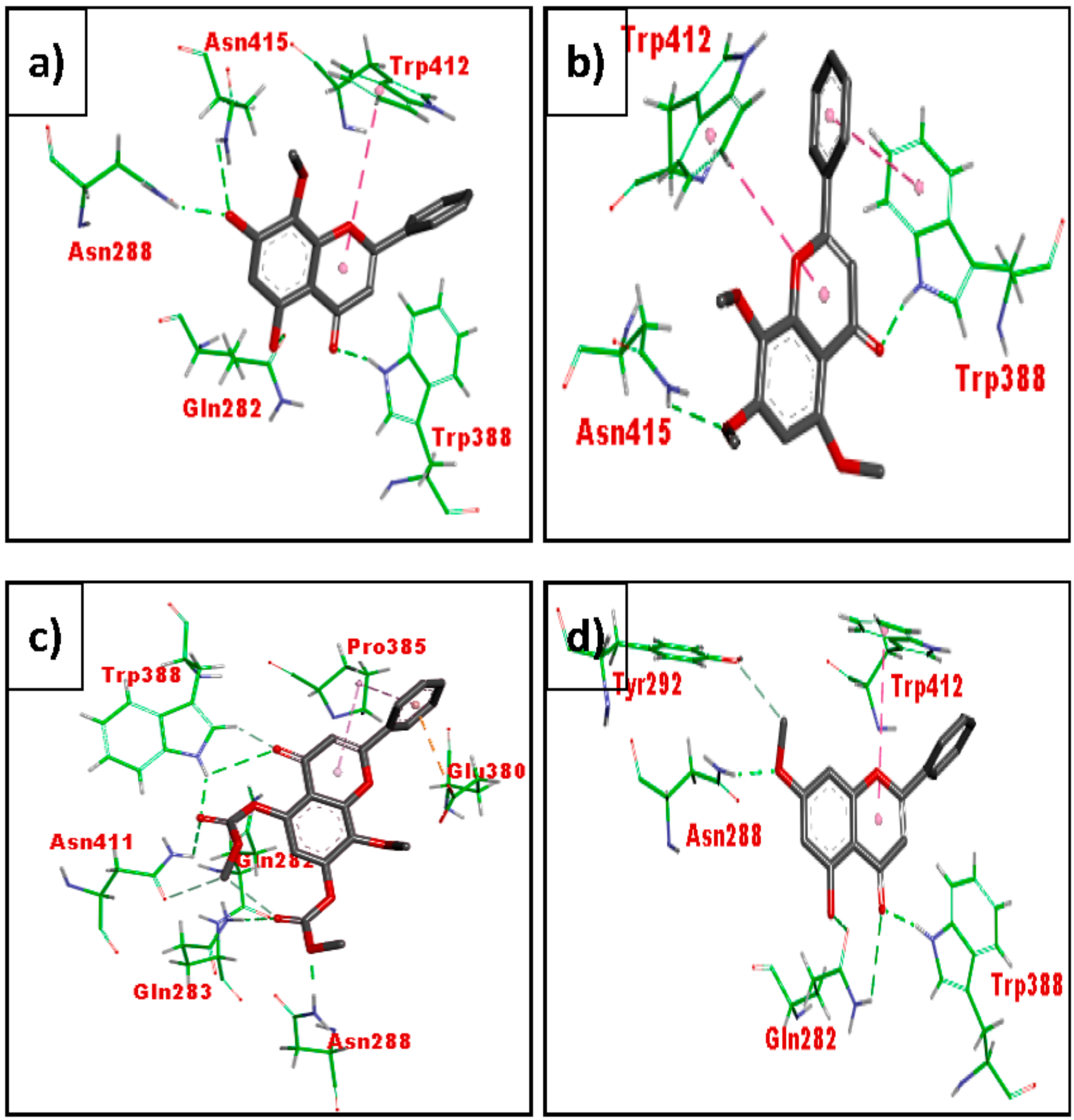
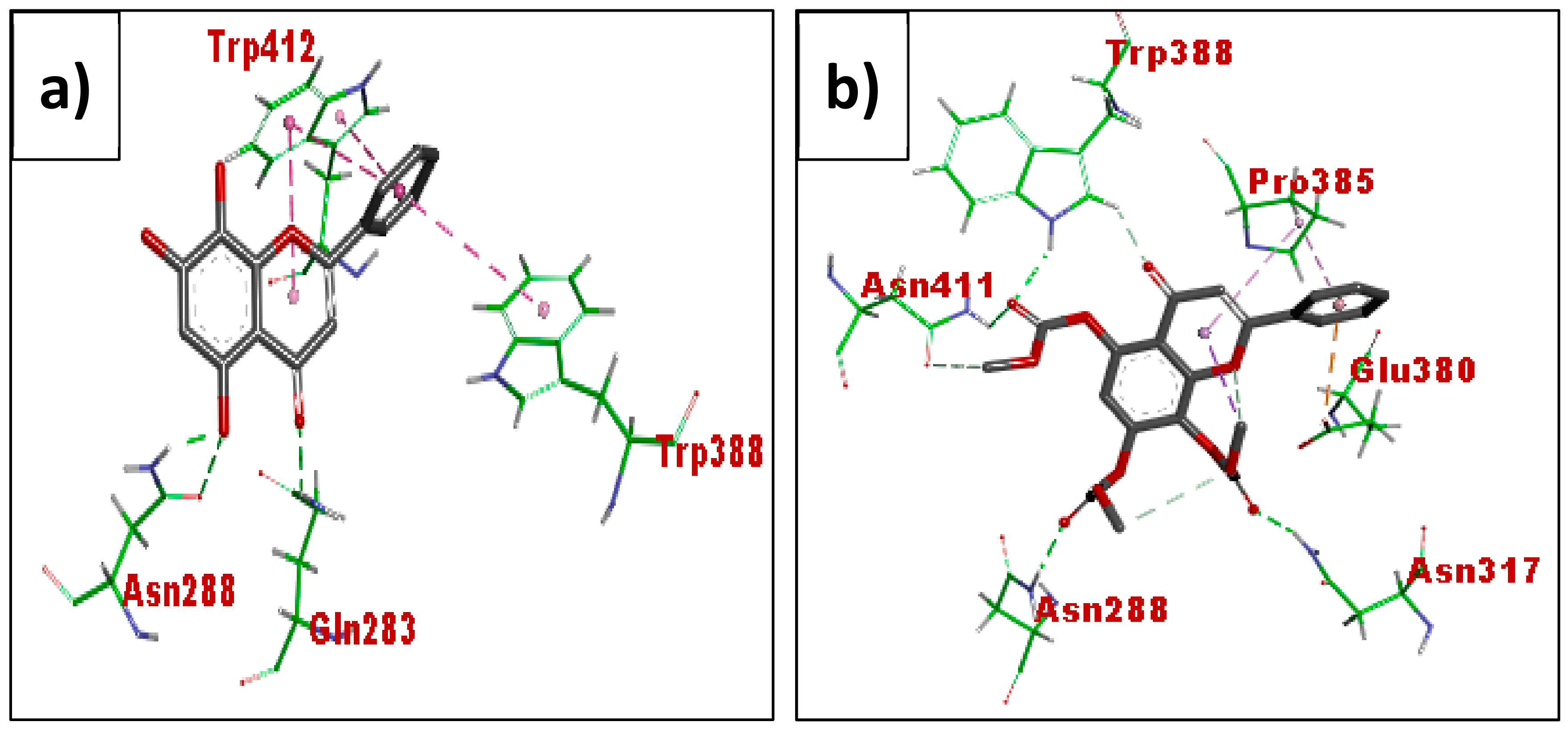
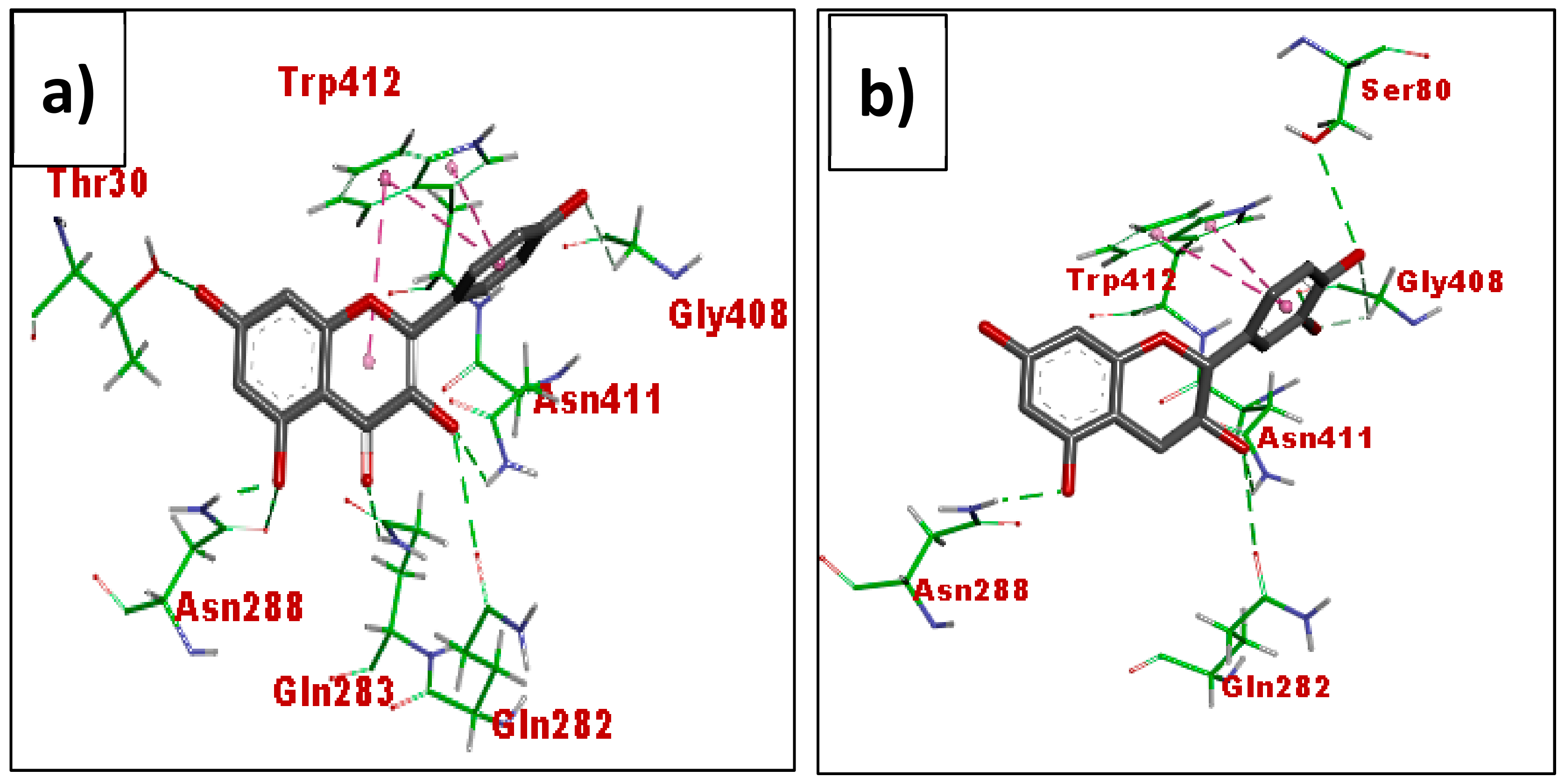
| Code | Ligand | Free Binding Energy (kcal/mol) |
|---|---|---|
| Control | Cytochalasin B (co-crystallize ligand) | −10.75 |
| MN1 | Wogonin | −7.07 |
| MN2 | Methyl ether of wogonin | −6.97 |
| MN3 | Acetate of wogonin | −7.84 |
| MN4 | Techtochrysin | −6.8 |
| MN7 | Norwogonin | −7.17 |
| MN8 | Acetate of norwogonin | −7.45 |
| MN11 | Kaempferol | 7.63 |
| MN14 | (-)-Epicatechin | 7.86 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed, Q.U.; Sarian, M.N.; Mat So'ad, S.Z.; Latip, J.; Arief Ichwan, S.J.; Hussein, N.N.; Taher, M.; Alhassan, A.M.; Hamidon, H.; Fakurazi, S. Methylation and Acetylation Enhanced the Antidiabetic Activity of Some Selected Flavonoids: In Vitro, Molecular Modelling and Structure Activity Relationship-Based Study. Biomolecules 2018, 8, 149. https://doi.org/10.3390/biom8040149
Ahmed QU, Sarian MN, Mat So'ad SZ, Latip J, Arief Ichwan SJ, Hussein NN, Taher M, Alhassan AM, Hamidon H, Fakurazi S. Methylation and Acetylation Enhanced the Antidiabetic Activity of Some Selected Flavonoids: In Vitro, Molecular Modelling and Structure Activity Relationship-Based Study. Biomolecules. 2018; 8(4):149. https://doi.org/10.3390/biom8040149
Chicago/Turabian StyleAhmed, Qamar Uddin, Murni Nazira Sarian, Siti Zaiton Mat So'ad, Jalifah Latip, Solachuddin Jauhari Arief Ichwan, Nurlaili Najmie Hussein, Muhammad Taher, Alhassan Muhammad Alhassan, Hanisuhana Hamidon, and Sharida Fakurazi. 2018. "Methylation and Acetylation Enhanced the Antidiabetic Activity of Some Selected Flavonoids: In Vitro, Molecular Modelling and Structure Activity Relationship-Based Study" Biomolecules 8, no. 4: 149. https://doi.org/10.3390/biom8040149
APA StyleAhmed, Q. U., Sarian, M. N., Mat So'ad, S. Z., Latip, J., Arief Ichwan, S. J., Hussein, N. N., Taher, M., Alhassan, A. M., Hamidon, H., & Fakurazi, S. (2018). Methylation and Acetylation Enhanced the Antidiabetic Activity of Some Selected Flavonoids: In Vitro, Molecular Modelling and Structure Activity Relationship-Based Study. Biomolecules, 8(4), 149. https://doi.org/10.3390/biom8040149






