A Gold Nanoparticle-Based Cortisol Aptasensor for Non-Invasive Detection of Fish Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Artificial Seawater (ASW)
2.3. AuNP Synthesis
2.4. Aptamer Affinity Characterization Using Bare AuNPs’ Aggregation Profile
2.5. Co-Conjugation of Aptamer and Blocker on AuNPs (Au-Apt-CALNN) and Stability Test
2.6. Cortisol Detection Using Au-Apt-CALNN
2.7. Dynamic Light Scattering Characterization
2.8. Fish Tank Water Samples
2.9. High Performance Liquid Chromatography (HPLC) for Analysis of Cortisol in Fish Tank Water
3. Results and Discussion
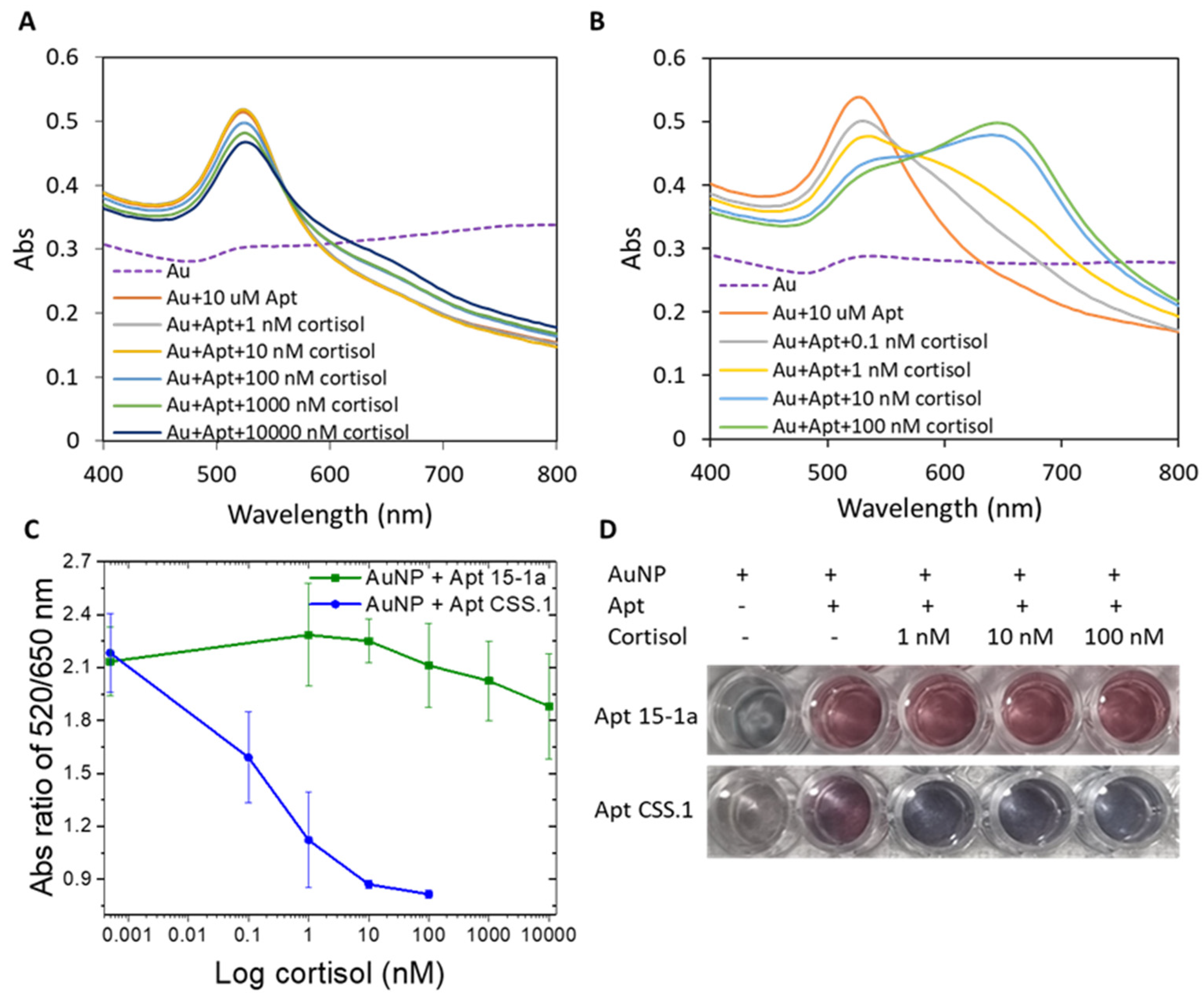
3.1. Confirmation of Aptamer–Cortisol Binding Using Bare AuNPs
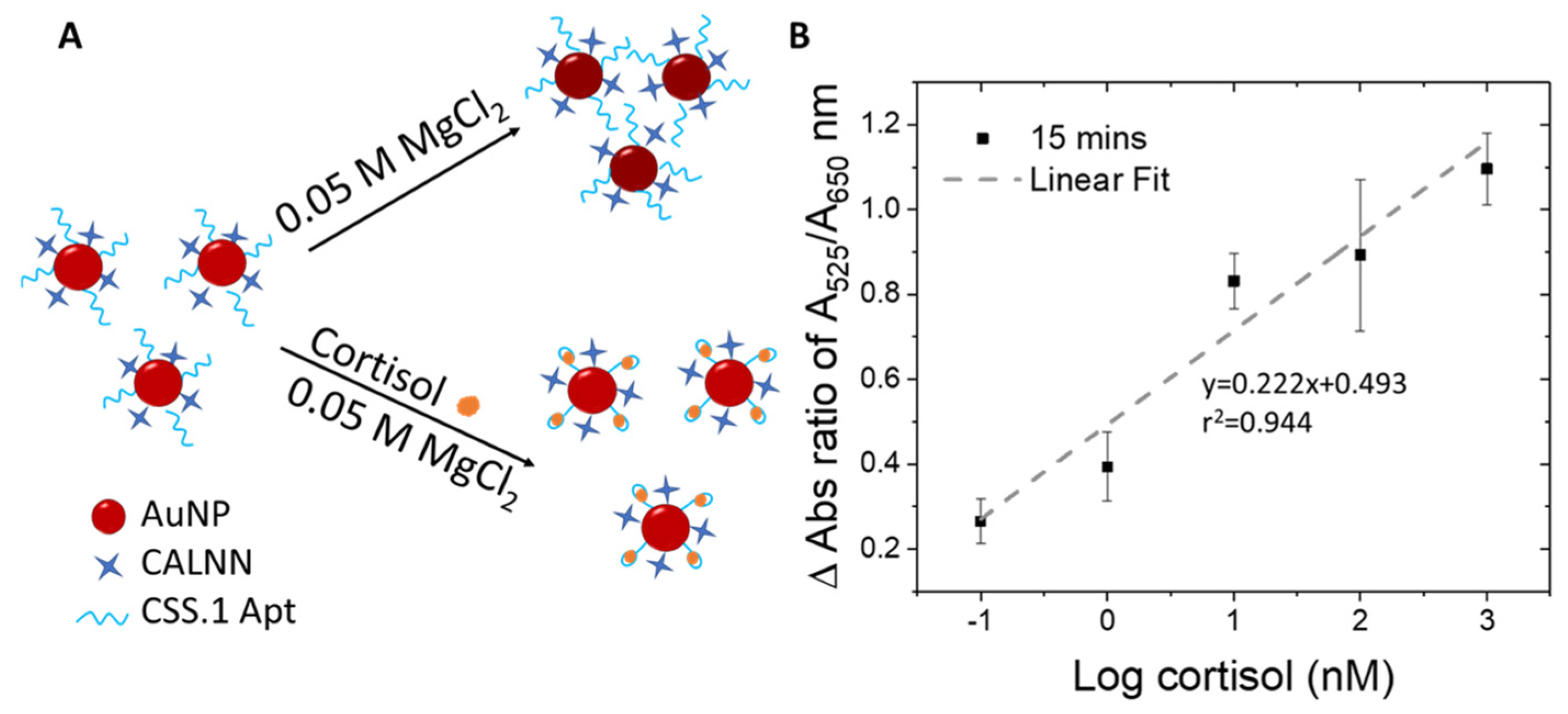
3.2. Co-Conjugation of Aptamer and Blocker Molecules with AuNPs and Particle Stability Test
3.3. Cortisol Detection Using Au-Apt-CALNN Conjugate
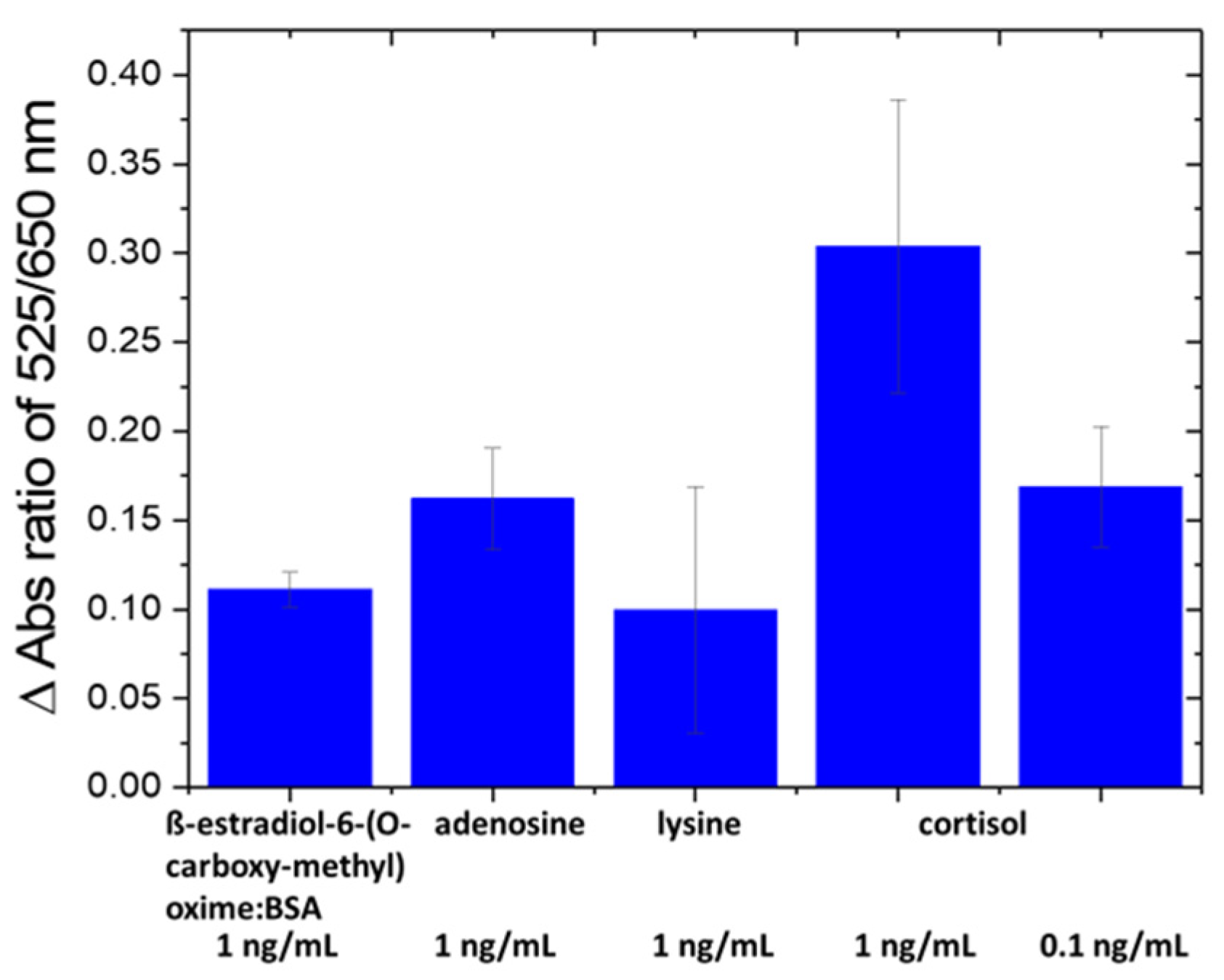
3.4. Selectivity Test and Real Sample Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Santonocito, R.; Puglisi, R.; Cavallaro, A.; Pappalardo, A.; Trusso Sfrazzetto, G. Cortisol Sensing by Optical Sensors. Analyst 2024, 149, 989–1001. [Google Scholar] [CrossRef] [PubMed]
- Su, T.; Mi, Z.; Xia, Y.; Jin, D.; Xu, Q.; Hu, X.; Shu, Y. A Wearable Sweat Electrochemical Aptasensor Based on the Ni–Co MOF Nanosheet-Decorated CNTs/PU Film for Monitoring of Stress Biomarker. Talanta 2023, 260, 124620. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.-K.; Kim, J.-W.; Kim, J.-M.; Kim, M.-G. High Sensitive and Broad-Range Detection of Cortisol in Human Saliva Using a Trap Lateral Flow Immunoassay (TrapLFI) Sensor. Analyst 2018, 143, 3883–3889. [Google Scholar] [CrossRef] [PubMed]
- Zea, M.; Bellagambi, F.G.; Halima, H.B.; Zine, N.; Jaffrezic-Renault, N.; Villa, R.; Gabriel, G.; Errachid, A. Electrochemical Sensors for Cortisol Detections: Almost There. TrAC Trends Anal. Chem. 2020, 132, 116058. [Google Scholar] [CrossRef]
- Arya, S.K.; Chornokur, G.; Venugopal, M.; Bhansali, S. Antibody Functionalized Interdigitated μ-Electrode (IDμE) Based Impedimetric Cortisol Biosensor. Analyst 2010, 135, 1941–1946. [Google Scholar] [CrossRef] [PubMed]
- Grau, J.; Moreno-Guzmán, M.; Arruza, L.; López, M.Á.; Escarpa, A.; Chisvert, A. Analysis of Microsamples by Miniaturized Magnetic-Based Pipette Tip Microextraction: Determination of Free Cortisol in Serum and Urine from Very Low Birth Weight Preterm Newborns. Analyst 2023, 148, 1050–1057. [Google Scholar] [CrossRef] [PubMed]
- Jia, M.; Chew, W.M.; Feinstein, Y.; Skeath, P.; Sternberg, E.M. Quantification of Cortisol in Human Eccrine Sweat by Liquid Chromatography–Tandem Mass Spectrometry. Analyst 2016, 141, 2053–2060. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Mohamed Salleh, N.A.b.; Aung, K.M.M.; Su, X.; Sutarlie, L. Cortisol Biosensors: From Sensing Principles to Applications. World Sci. Annu. Rev. Funct. Mater. 2023, 01, 2330001. [Google Scholar] [CrossRef]
- Sadoul, B.; Geffroy, B. Measuring Cortisol, the Major Stress Hormone in Fishes. J. Fish Biol. 2019, 94, 540–555. [Google Scholar] [CrossRef]
- Chmelíková, E.; Bolechová, P.; Chaloupková, H.; Svobodová, I.; Jovičić, M.; Sedmíková, M. Salivary Cortisol as a Marker of Acute Stress in Dogs: A Review. Domest. Anim. Endocrinol. 2020, 72, 106428. [Google Scholar] [CrossRef]
- Moore, T.J.; Sharma, B. Direct Surface Enhanced Raman Spectroscopic Detection of Cortisol at Physiological Concentrations. Anal. Chem. 2020, 92, 2052–2057. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.R.; Aung, K.M.M.; Salleh, N.A.b.M.; Tan, J.Y.A.; Chua, K.X.; Doblado, G.J.; Chua, K.L.; Tham, V.; Lin, J.J.; Chaganty, V. Dynamics of Endogenous and Water Cortisol Release in Asian Seabass Lates Calcarifer after Acute Stress in a Farm Scale Recirculating Aquaculture System. Aquac. Rep. 2024, 37, 102223. [Google Scholar] [CrossRef]
- Weng, X.; Fu, Z.; Zhang, C.; Jiang, W.; Jiang, H. A PorTable 3D Microfluidic Origami Biosensor for Cortisol Detection in Human Sweat. Anal. Chem. 2022, 94, 3526–3534. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Chang, H.; Yang, G.; Xu, Z.; Li, J.; Gu, Z.; Li, J. An Integrated Wearable Sticker Based on Extended-Gate AlGaN/GaN High Electron Mobility Transistors for Real-Time Cortisol Detection in Human Sweat. Analyst 2024, 149, 958–967. [Google Scholar] [CrossRef] [PubMed]
- Sharef, O.A.; Feely, J.; Kavanagh, P.V.; Scott, K.R.; Sharma, S.C. An HPLC Method for the Determination of the Free Cortisol/Cortisone Ratio in Human Urine. Biomed. Chromatogr. 2007, 21, 1201–1206. [Google Scholar] [CrossRef] [PubMed]
- Turpeinen, U.; Markkanen, H.; Välimäki, M.; Stenman, U.-H. Determination of Urinary Free Cortisol by HPLC. Clin. Chem. 1997, 43, 1386–1391. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Ohnuki, H.; Ota, S.; Murata, M.; Yoshiura, Y.; Endo, H. New Approach for Monitoring Fish Stress: A Novel Enzyme-Functionalized Label-Free Immunosensor System for Detecting Cortisol Levels in Fish. Biosens. Bioelectron. 2017, 93, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Barry, T.P.; Lapp, A.F.; Kayes, T.B.; Malison, J.A. Validation of a Microtitre Plate ELISA for Measuring Cortisol in Fish and Comparison of Stress Responses of Rainbow trout (Oncorhynchus mykiss) and Lake trout (Salvelinus namaycush). Aquaculture 1993, 117, 351–363. [Google Scholar] [CrossRef]
- Apilux, A.; Rengpipat, S.; Suwanjang, W.; Chailapakul, O. Paper-Based Immunosensor with Competitive Assay for Cortisol Detection. J. Pharm. Biomed. Anal. 2020, 178, 112925. [Google Scholar] [CrossRef]
- Zhang, Y.; Lai, Q.; Chen, W.; Zhang, C.; Mo, L.; Liu, Z. Recent Advance in Cortisol Immunosensing Technologies and Devices. Chemosensors 2023, 11, 90. [Google Scholar] [CrossRef]
- Stevens, R.C.; Soelberg, S.D.; Near, S.; Furlong, C.E. Detection of Cortisol in Saliva with a Flow-Filtered, Portable Surface Plasmon Resonance Biosensor System. Anal. Chem. 2008, 80, 6747–6751. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Yang, J.; Hur, H.; Oh, S.; Lee, H.H. Highly Sensitive Colorimetric Assay of Cortisol Using Cortisol Antibody and Aptamer Sandwich Assay. Biosensors 2021, 11, 163. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.W.H.; Bhuiyan, W.T.; Pang, S.; Warren, B.; Makris, K.; Coleman, S.; Hassan, S.; Niu, X. A Portable Droplet Microfluidic Device for Cortisol Measurements Using a Competitive Heterogeneous Assay. Analyst 2021, 146, 4535–4544. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.K.; Chung, S.; Sveiven, M.; Hall, D.A. Cortisol Detection in Undiluted Human Serum Using a Sensitive Electrochemical Structure-Switching Aptamer over an Antifouling Nanocomposite Layer. ACS Omega 2021, 6, 27888–27897. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yu, H.; Alkhamis, O.; Moliver, J.; Xiao, Y. Tuning Biosensor Cross-Reactivity Using Aptamer Mixtures. Anal. Chem. 2020, 92, 5041–5047. [Google Scholar] [CrossRef] [PubMed]
- Sen, D.; Lazenby, R.A. Selective Aptamer Modification of Au Surfaces in a Microelectrode Sensor Array for Simultaneous Detection of Multiple Analytes. Anal. Chem. 2023, 95, 6828–6835. [Google Scholar] [CrossRef]
- Pallares, R.M.; Thanh, N.T.K.; Su, X. Sensing of Circulating Cancer Biomarkers with Metal Nanoparticles. Nanoscale 2019, 11, 22152–22171. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Neoh, K.G.; Kang, E.-T.; Choe, W.-S.; Su, X. Affinity Analysis of DNA Aptamer–Peptide Interactions Using Gold Nanoparticles. Anal. Biochem. 2012, 421, 725–731. [Google Scholar] [CrossRef]
- Dalirirad, S.; Han, D.; Steckl, A.J. Aptamer-Based Lateral Flow Biosensor for Rapid Detection of Salivary Cortisol. ACS Omega 2020, 5, 32890–32898. [Google Scholar] [CrossRef]
- Wu, T.; Ding, L.; Zhang, Y.; Fang, W. A Simple Cortisol Biosensor Based on AuNPs-DNA Aptamer Conjugate. IEEE Sens. J. 2022, 22, 12485–12492. [Google Scholar] [CrossRef]
- Dalirirad, S.; Steckl, A.J. Aptamer-Based Lateral Flow Assay for Point of Care Cortisol Detection in Sweat. Sens. Actuators B Chem. 2019, 283, 79–86. [Google Scholar] [CrossRef]
- Jo, S.; Lee, W.; Park, J.; Kim, W.; Kim, W.; Lee, G.; Lee, H.-J.; Hong, J.; Park, J. Localized Surface Plasmon Resonance Aptasensor for the Highly Sensitive Direct Detection of Cortisol in Human Saliva. Sens. Actuators B Chem. 2020, 304, 127424. [Google Scholar] [CrossRef]
- Mitchell, J.S.; Lowe, T.E.; Ingram, J.R. Rapid Ultrasensitive Measurement of Salivary Cortisol Using Nano-Linker Chemistry Coupled with Surface Plasmon Resonance Detection. Analyst 2009, 134, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Ran, J.; Luo, D.; Liu, B. A Dual-Mode Biosensor for Salivary Cortisol with Antibody-Aptamer Sandwich Pattern and Enzyme Catalytic Amplification. J. Solid State Electrochem. 2023, 27, 399–408. [Google Scholar] [CrossRef]
- Liu, X.; Zhao, R.; Mao, W.; Feng, H.; Liu, X.; Wong, D.K.Y. Detection of Cortisol at a Gold Nanoparticle|Protein G–DTBP-Scaffold Modified Electrochemical Immunosensor. Analyst 2011, 136, 5204–5210. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Dighe, K.; Wang, Z.; Srivastava, I.; Schwartz-Duval, A.S.; Misra, S.K.; Pan, D. Electrochemical-Digital Immunosensor with Enhanced Sensitivity for Detecting Human Salivary Glucocorticoid Hormone. Analyst 2019, 144, 1448–1457. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.A.; Chávez, J.L.; Chushak, Y.; Chapleau, R.R.; Hagen, J.; Kelley-Loughnane, N. Tunable Stringency Aptamer Selection and Gold Nanoparticle Assay for Detection of Cortisol. Anal. Bioanal. Chem. 2014, 406, 4637–4647. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Ding, Y.; Zhang, C.; Liu, J. Comparing Two Cortisol Aptamers for Label-Free Fluorescent and Colorimetric Biosensors. Sens. Diagn. 2022, 1, 541–549. [Google Scholar] [CrossRef]
- Zulkeflee Sabri, M.; Abdul Hamid, A.A.; Sayed Hitam, S.M.; Abdul Rahim, M.Z. In-Silico Study of Single-Strand DNA Aptamers against Cortisol as a Potential Biomarker Receptor in Therapeutics and Diagnostics. Mater. Today Proc. 2020, 31, A90–A97. [Google Scholar] [CrossRef]
- Yang, K.-A.; Chun, H.; Zhang, Y.; Pecic, S.; Nakatsuka, N.; Andrews, A.M.; Worgall, T.S.; Stojanovic, M.N. High-Affinity Nucleic-Acid-Based Receptors for Steroids. ACS Chem. Biol. 2017, 12, 3103–3112. [Google Scholar] [CrossRef]
- Prante, M.; Segal, E.; Scheper, T.; Bahnemann, J.; Walter, J. Aptasensors for Point-of-Care Detection of Small Molecules. Biosensors 2020, 10, 108. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Zandieh, M.; Zheng, J.; Liu, J. Reversible and Irreversible HAuCl4 Binding to DNA for Seeded Gold Nanoparticle Growth and Opposite DNA and Aptamers Colorimetric Sensing Outcomes. Part. Part. Syst. Charact. 2022, 39, 2200121. [Google Scholar] [CrossRef]
- Murillo-Gurrea, D.P.; Coloso, R.M.; Borlongan, I.G.; Serrano, A.E. Lysine and Arginine Requirements of Juvenile Asian Sea Bass (Lates calcarifer). J. Appl. Ichthyol. 2001, 17, 49–53. [Google Scholar]
- Lim, L.H.; Regina, L.Z.L.; Soh, Y.T.; Teo, S.S. Determination of Levels of Phosphate, Ammonia and Chlorine from Indoor and Outdoor Nano Tank System. Int. J. Aquac. 2018, 8, 145–150. [Google Scholar]
- Kovács, B.D.; de Jesus Gregersen, K.J.; Rüppel, F.; von Danwitz, A.; Pedersen, L.F. Evaluating Protein Skimmer Performance in a Commercial Seawater Recirculating Aquaculture System (RAS). Aquac. Eng. 2023, 103, 102369. [Google Scholar] [CrossRef]
- Zhao, W.; Chiuman, W.; Lam, J.C.F.; McManus, S.A.; Chen, W.; Cui, Y.; Pelton, R.; Brook, M.A.; Li, Y. DNA Aptamer Folding on Gold Nanoparticles: From Colloid Chemistry to Biosensors. J. Am. Chem. Soc. 2008, 130, 3610–3618. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Tang, L.; Gao, F.; Li, Y.; Liu, J.; Zheng, J. DNA-Encoded Bimetallic Au-Pt Dumbbell Nanozyme for High-Performance Detection and Eradication of Escherichia Coli O157:H7. Biosens. Bioelectron. 2021, 187, 113327. [Google Scholar] [CrossRef]
- Odhiambo, E.; Angienda, P.O.; Okoth, P.; Onyango, D. Stocking Density Induced Stress on Plasma Cortisol and Whole Blood Glucose Concentration in Nile Tilapia Fish (Oreochromis Niloticus) of Lake Victoria, Kenya. Int. J. Zool. 2020, 2020, 9395268. [Google Scholar] [CrossRef]
- Nguyen, T.V. Preparation of Artificial Sea Water (ASW) for Culturing Marine Bacteria. Available online: https://www.researchgate.net/profile/Thao-Nguyen-43/publication/323971616_Preparation_of_Artificial_Sea_Water_ASW_for_Culturing_Marine_Bacteria/links/5ab5896d45851515f59a7a5b/Preparation-of-Artificial-Sea-Water-ASW-for-Culturing-Marine-Bacteria.pdf (accessed on 18 December 2021).
- Sutarlie, L.; Aung, K.M.M.; Lim, M.G.L.; Lukman, S.; Cheung, E.; Su, X. Studying Protein–DNA Complexes Using Gold Nanoparticles by Exploiting Particle Aggregation, Refractive Index Change, and Fluorescence Quenching and Enhancement Principles. Plasmonics 2014, 9, 753–763. [Google Scholar] [CrossRef]
- Ney, L.J.; Felmingham, K.L.; Bruno, R.; Matthews, A.; Nichols, D.S. Chloroform-Based Liquid-Liquid Extraction and LC–MS/MS Quantification of Endocannabinoids, Cortisol and Progesterone in Human Hair. J. Pharm. Biomed. Anal. 2021, 201, 114103. [Google Scholar] [CrossRef]
- Viljoen, F.P.; Du Preez, J.L.; Wessels, J.C.; Aucamp, M.E.; Meyer, L.C.R.; Pohlin, F. An HPLC-DAD Validated Method for the Detection and Quantification of Cortisol, Corticosterone and Melatonin in Plasma Samples of Two Different Animal Species. Die Pharm. Int. J. Pharm. Sci. 2019, 74, 206–211. [Google Scholar]
- Semwal, V.; Højgaard, J.; Møller, E.; Bang, O.; Janting, J. Study on Cortisol Sensing Principle Based on Fluorophore and Aptamer Competitive Assay on Polymer Optical Fiber. Photonics 2023, 10, 840. [Google Scholar] [CrossRef]
- Sanghavi, B.J.; Moore, J.A.; Chávez, J.L.; Hagen, J.A.; Kelley-Loughnane, N.; Chou, C.-F.; Swami, N.S. Aptamer-Functionalized Nanoparticles for Surface Immobilization-Free Electrochemical Detection of Cortisol in a Microfluidic Device. Biosens. Bioelectron. 2016, 78, 244–252. [Google Scholar] [CrossRef]
- An, J.E.; Kim, K.H.; Park, S.J.; Seo, S.E.; Kim, J.; Ha, S.; Bae, J.; Kwon, O.S. Wearable Cortisol Aptasensor for Simple and Rapid Real-Time Monitoring. ACS Sens. 2022, 7, 99–108. [Google Scholar] [CrossRef]
- Huang, Z.; Chen, H.; Ye, H.; Chen, Z.; Jaffrezic-Renault, N.; Guo, Z. An Ultrasensitive Aptamer-Antibody Sandwich Cortisol Sensor for the Noninvasive Monitoring of Stress State. Biosens. Bioelectron. 2021, 190, 113451. [Google Scholar] [CrossRef]
- Jia, Y.; Yan, X.; Guo, X.; Zhou, G.; Liu, P.; Li, Z. One Step Preparation of Peptide-Coated Gold Nanoparticles with Tunable Size. Materials 2019, 12, 2107. [Google Scholar] [CrossRef]
- Zhou, G.; Liu, Y.; Luo, M.; Li, X.; Xu, Q.; Ji, X.; He, Z. Controlled Assembly of Gold Nanoparticles through Antibody Recognition: Study and Utilizing the Effect of Particle Size on Interparticle Distance. Langmuir 2013, 29, 4697–4702. [Google Scholar] [CrossRef]
- Lévy, R.; Thanh, N.T.K.; Doty, R.C.; Hussain, I.; Nichols, R.J.; Schiffrin, D.J.; Brust, M.; Fernig, D.G. Rational and Combinatorial Design of Peptide Capping Ligands for Gold Nanoparticles. J. Am. Chem. Soc. 2004, 126, 10076–10084. [Google Scholar] [CrossRef]
- Hu, S.; Huang, P.-J.J.; Wang, J.; Liu, J. Dissecting the Effect of Salt for More Sensitive Label-Free Colorimetric Detection of DNA Using Gold Nanoparticles. Anal. Chem. 2020, 92, 13354–13360. [Google Scholar] [CrossRef]
- Bao, X.; Huo, G.; Li, L.; Cao, X.; Liu, Y.; Lakshmipriya, T.; Chen, Y.; Hariri, F.; Gopinath, S.C. Coordinated dispersion and aggregation of gold nanorod in aptamer-mediated gestational hypertension analysis. J. Anal. Methods Chem. 2019, 2019, 5676159. [Google Scholar] [CrossRef]
- Alsager, O.A.; Kumar, S.; Zhu, B.; Travas-Sejdic, J.; McNatty, K.P.; Hodgkiss, J.M. Ultrasensitive colorimetric detection of 17β-estradiol: The effect of shortening DNA aptamer sequences. Anal. Chem. 2015, 87, 4201–4209. [Google Scholar] [CrossRef]
- Sadhu, N.; Sharma, S.R.K.; Joseph, S.; Dube, P.; Philipose, K.K. Chronic Stress Due to High Stocking Density in Open Sea Cage Farming Induces Variation in Biochemical and Immunological Functions in Asian seabass (Lates calcarifer, Bloch). Fish Physiol. Biochem. 2014, 40, 1105–1113. [Google Scholar] [CrossRef]
- Li, D.; Liu, Z.; Xie, C. Effect of Stocking Density on Growth and Serum Concentrations of Thyroid Hormones and Cortisol in Amur Sturgeon, Acipenser Schrenckii. Fish Physiol. Biochem. 2012, 38, 511–520. [Google Scholar] [CrossRef]

| Aptamer (Apt) | Sequence 5′-3′ |
|---|---|
| 15-1a | ATGGGCAATGCGGGGTGGAGAATGGTTGCCGCACTTCGGC |
| CSS.1 | GACGACGCCCGCATGTTCCATGGATAGTCTTGACTAGTCGTC |
| In Water (nm) | In PBS (nm) | In PBS and MgCl2 (nm) | |
|---|---|---|---|
| Au | 15.2 ± 0.1 | - | - |
| Au-Apt | 22.5 ± 0.1 | - | - |
| Au-Apt-CALNN | 27.8 ± 0.7 | 22.9 ± 0.5 | 49.2 ± 13.8 |
| Au-Apt-CALNN + cortisol * | - | 25.4 ± 0.4 | 40.2 ± 1.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanaka, Y.; Salleh, N.A.b.M.; Tan, M.R.; Vij, S.; Wee, C.L.; Sutarlie, L.; Su, X. A Gold Nanoparticle-Based Cortisol Aptasensor for Non-Invasive Detection of Fish Stress. Biomolecules 2024, 14, 818. https://doi.org/10.3390/biom14070818
Tanaka Y, Salleh NAbM, Tan MR, Vij S, Wee CL, Sutarlie L, Su X. A Gold Nanoparticle-Based Cortisol Aptasensor for Non-Invasive Detection of Fish Stress. Biomolecules. 2024; 14(7):818. https://doi.org/10.3390/biom14070818
Chicago/Turabian StyleTanaka, Yuki, Nur Asinah binte Mohamed Salleh, Marie Ruoyun Tan, Shubha Vij, Caroline Lei Wee, Laura Sutarlie, and Xiaodi Su. 2024. "A Gold Nanoparticle-Based Cortisol Aptasensor for Non-Invasive Detection of Fish Stress" Biomolecules 14, no. 7: 818. https://doi.org/10.3390/biom14070818
APA StyleTanaka, Y., Salleh, N. A. b. M., Tan, M. R., Vij, S., Wee, C. L., Sutarlie, L., & Su, X. (2024). A Gold Nanoparticle-Based Cortisol Aptasensor for Non-Invasive Detection of Fish Stress. Biomolecules, 14(7), 818. https://doi.org/10.3390/biom14070818







