1. Introduction
Space flight conditions differ to those on Earth due to cosmic ionizing radiation (IR) and the absence of gravity, known as microgravity (μG), both of which pose health risk factors to humans, causing complex DNA damage and genome instability [
1,
2,
3,
4,
5]. It is crucial to gain more insights on these factors, as astronauts will be continuously exposed to them in long-duration exploration missions, such as those to the Moon or Mars, which require humans to remain in space for days, months and even years.
Space radiation risks, arising mainly from solar energetic particles (SEPs) and galactic cosmic rays (GCRs), involve a flux comprising 2% electrons and 98% nuclei, with the nuclear component being ~87% hydrogen, ~12% helium and ~1% high atomic number and energy (HZE) particles [
6]. Despite low GCR particle flux levels, their high linear energy transfer (LET) induces intense ionization in matter [
7,
8]. During Earth-to-Mars manned missions using Hohmann transfer [
9] which will last 2–3 years [
10], astronauts will face an estimated ~1.01 Sv [
11] radiation exposure, increasing the risk of cancer, central nerve system decrements, degenerative tissue effects [
12], and irreversible chromosomal instability risks due to HZE particle exposure.
During lengthy planetary flights, astronauts face both potential radiation hazards and simultaneous exposure to microgravity (μG). Living organisms undergo physiological changes in varying gravitational conditions, including muscle atrophy, reduced bone density, immune function decline and endocrine disorders. The space environment, comprising GCRs and reduced gravity, necessitates testing for possible additive or synergistic effects. Altered DNA repair mechanisms due to gravity changes can impede cellular responses to space radiation, increasing the risk of DNA damage accumulation and tumorigenesis [
13]. Recent studies highlight that spaceflight stressors like ionizing radiation (IR) and/or microgravity disrupt the wound healing process, affecting pathways like inflammation and proliferation [
14]. Microgravity significantly impacts cell death, migration and gene expression in tumor cells, including cancer stem cells, and alters the effects of chemotherapeutic drugs [
15]. While extensive research has focused on the impact of either radiation or microgravity alone, limited studies have addressed their combined effects. Previous attempts were found to be challenging, as older clinostats had to pause rotation which simulates microgravity to irradiate the cells [
16,
17], potentially introducing additional gravitational stimuli, and thus activating specific signaling cascades.
In deep space beyond the Van Allen belts, galactic cosmic rays consist of both high-energy and low-energy radiation. To investigate the combined effects of space radiation and microgravity, considering lunar and Mars explorations and long-term stays in space in the near future, it was decided to use carbon ions as high-energy radiation and X-rays as low-energy radiation. To maintain the consistent simulated μG condition before, during and after exposure to radiation, a 3D clinostat that allows samples to be rotated and irradiated simultaneously was developed [
18] and used in this study. This clinostat was previously used for the study of the differential expression of exclusively nine cell cycle-related genes in response to X-ray or Carbon-ion (C-ion) irradiation, with and without simulated μG, in human fibroblasts [
1]. The raw transcriptomic data produced from that previous study [
1] were reanalyzed in the current work, applying a systems biology approach, to identify all differentially expressed genes between various conditions and their predominant processes they participate in and to identify a possible synergy between radiation and μG.
2. Materials and Methods
1BR-hTERT human fibroblast cells were cultured in CO
2-independent medium (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% (
v/
v) fetal bovine serum (MP Biomedicals, Santa Ana, CA, USA), 200 mM L-glutamine (Thermo Fisher Scientific, Waltham, MA, USA), and penicillin–streptomycin mixed solution (Nacalai Tesque, Kyoto, Kyoto, Japan) at 37 °C under 1G or simulated μG for 48 h in total. The samples were collected 0 (sham irradiated), 3 or 24 h after X-ray or C-ion irradiation at 1 Gy. X-ray irradiation was performed using an X-ray generator (200 kV, 14.6 mA, aluminum filter (0.3 mm thick), MultiRad225, Faxitron Bioptics, LLC, Tucson, AZ, USA) equipped with a high-speed shutter. C-ion irradiation was performed using a synchrotron (Gunma University Heavy Ion Medical Center (GHMC), Maebashi, Gunma, Japan) and respiratory gating signals with a dose-averaged LET of 50 keV/μm at the center of the 6 cm spread-out Bragg peak of the beam with energy of 290 MeV/n. The dose rate was approximately 0.03 Gy/min for both X-ray and C-ion irradiation under the simulated μG or 1G conditions. Simulated μG was accomplished using a three-dimensional clinostat [
18,
19]. This device can manipulate the effect of gravity through the 3D rotation of two orthogonal axes and by continuously changing the direction of gravity. The X:Y ratios of clino-rotation were set at 11:13 rpm and 66°/s:78°/s using a special controller to maintain suitable conditions, which means that it does not use random speed and random direction. The rotation angle between the
Z-axis of the 3D clinostat (i.e., the axis of radiation exposure) and the normal line of the sample holder on the 3D clinostat, θ, was kept at less than 12°, assuming the X:Y ratio of the clino-rotation was 11:13 rpm. Because different research groups are performing simulated microgravity experiments under various conditions with different types of simulators and cell line, we think that it is important to carefully consider the experimental conditions and provide details such as simulator limitations in order to avoid misinterpretation of the results [
20]. Among such limitations, we used adherent human fibroblasts in a thin culture vessel completely filled with medium (without bubbles) to minimize stress on the cells as much as possible. It is not necessary to change the medium under our simulated microgravity conditions until the sampling. From our previous data of cell growth, which did not differ significantly between rotating and standing conditions after 48 h of culture, we believe that it is unlikely that cells are subjected to shear stress under our experimental conditions using our system. The samples were irradiated when in horizontal position, without pausing the rotation, for 0.2 s every minute. In total, 12 conditions were studied (
Table 1) in triplicate [
1].
2.1. RNA-Sequencing
RNA from each of the 36 samples, was extracted using TRIzol™ Reagent (Thermo Fisher Scientific, Waltham, MA, USA) and its quality was assessed using the RNA 6000 Pico Kit (Agilent Technologies, Santa Clara, CA, USA). rRNA was depleted using the NEBNext
® rRNA Depletion Kit (New England Biolabs, Ipswich, MA, USA). Then, RNA-Seq library was prepared using NEBNext
® Ultra™ Directional RNA Library Prep Kit for Illumina
® (New England Biolabs, Ipswich, MA, USA). Paired-end sequencing (2 × 36 bp) was performed with NextSeq 500 (Illumina, San Diego, CA, USA) at Tsukuba i-Laboratory LLP (Tsukuba, Ibaraki, Japan) [
1]. Four FASTQ files were produced from each sample.
2.2. Differentially Expressed Gene Analysis
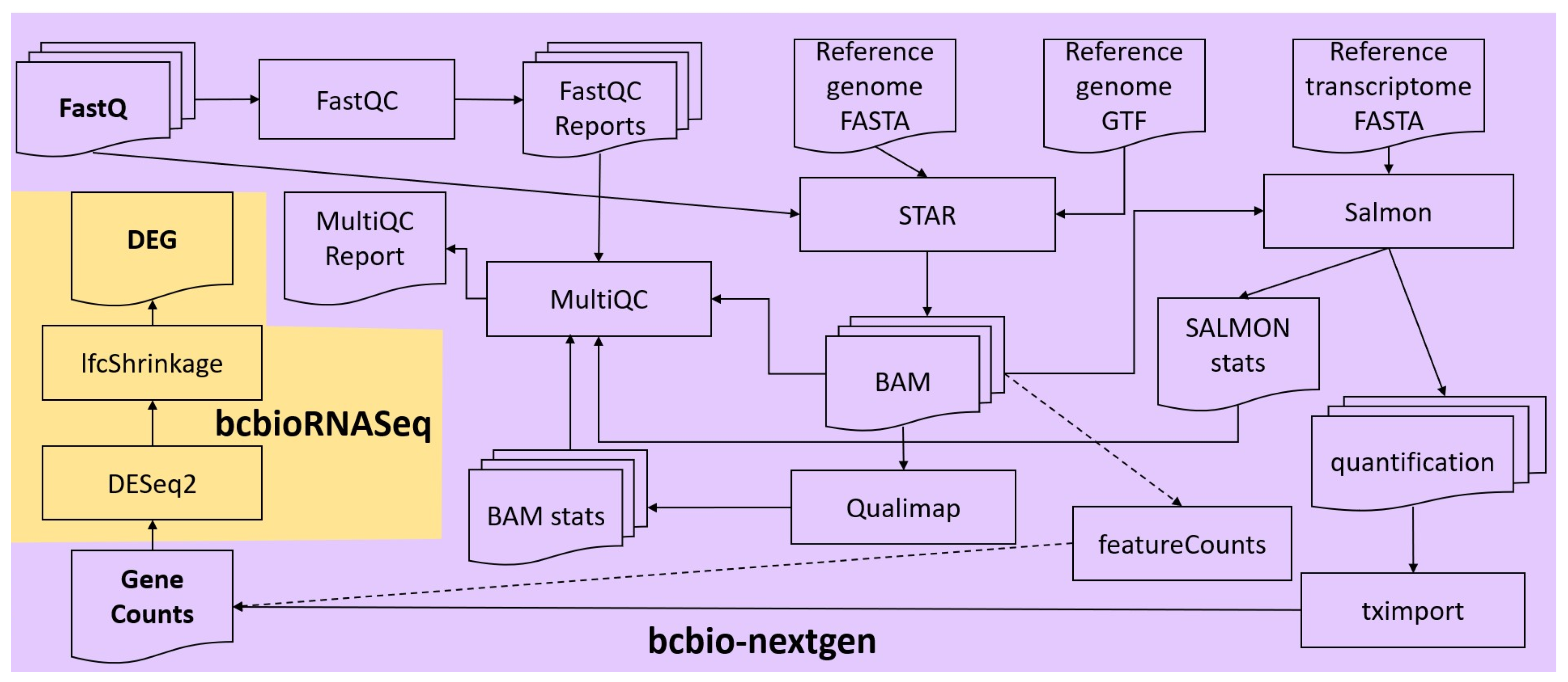
FASTQ files of each sample were concatenated, and the integrity of the resulting files was checked, using in-house scripts. Quality control and alignment of their reads were carried out via the RNA-seq workflow from the bcbio-nextgen bioinformatics framework (version 1.2.9) [
21] (
Figure 1). To ensure that the library generation and sequencing quality were suitable for further analysis, FastQC [
22] was used to examine the raw reads for quality issues. Then, raw reads were aligned to the GRCh38 (hg38) version of the human reference genome (FASTA and GTF files) with the splice-aware aligner STAR (version 2.6.1d) in two-pass mode (the first pass discovers new splice junctions and inserts them into the junction database, and the second pass calls junctions and calculates their counts) [
23]. Moreover, Salmon (version 1.7.0) was run in alignment-based mode, using the genome alignments from STAR (BAM files) and the reference transcriptome FASTA file, to generate abundance estimates for known splice isoforms [
24]. Bcbio assessed the complexity and quality of the RNA-seq data by quantifying ribosomal RNA (rRNA) content and the genomic context of alignments in known transcripts and introns using a combination of custom tools and Qualimap [
25]. MultiQC [
26] was then used for quality control and assurance analysis of the resulting BAM files by comparing to metrics gathered from bcbio-nextgen, samtools [
27], Salmon, STAR, Qualimap, and FastQC. Next, we quantitated reads by assigning them to genes (features) annotated in Ensembl (release 105) and counting them with the featureCounts tool [
28] or preferably tximport [
29]. Gene counts were processed for DGEA, using DESeq2 (version 1.38.2) [
30] default settings through the bcbioRNASeq R package (version 0.5.1) [
31] (
Figure 1). Moreover, log fold change shrinkage for visualization and ranking was performed calling the lfcShrink function of the DESeq2 package, replacing
p-values with s-values produced by the apeglm estimation method [
32]. S-value was proposed as a statistic giving the aggregate false sign rate for tests with equal or lower s-value than the one considered [
33]. Exported lists containing statistically significant differentially expressed genes (DEGs) include metrics such as Log
2 Fold Change (Log
2FC) and s-values for each gene. The lists were further annotated by bcbioRNASeq to include HGNC [
34] gene symbols and names. The threshold for statistical significance was set at s-value < 0.001, as suggested [
35]. Using this method, the statistically significant differentially expressed genes between various pairs of biological conditions (
Table 2) were identified.
2.3. Biological Term Enrichment Analysis
Gene term enrichment analyses were performed to identify the prevalent biological processes and pathways over- and under-expressed genes of each DEG analysis participate in using WebGestalt [
36]. The over-representation analysis (ORA) method [
37] was employed, applying BH [
38] multiple test adjustment. The threshold for statistical significance was set at a false discovery rate (FDR) < 0.05. The functional databases that were used for biological term enrichment were Biological Process, Cellular Component and Molecular Function from Gene Ontology [
39], KEGG [
40] and Transcription Factor Targeting and miRNA Targeting networks from MSigDB [
41]. In order to identify and depict overlapping genes or biological terms between comparisons of conditions, Venn diagrams were produced using a webtool developed by the Bioinformatics and Evolutionary Genomics group of Ghent University at
https://bioinformatics.psb.ugent.be/webtools/Venn/ (accessed on 7 January 2022).
2.4. Protein–Protein Interaction Network Analysis
STRING [
42]-based protein–protein interaction (PPI) network analyses were performed for the DEGs and PPI networks were constructed for each DEG analysis from all comparisons in order to discover their functional associations. The average local clustering coefficient [
43] served as a measure of how connected the produced PPI networks were and PPI enrichment
p-values provide the probability to obtain the observed number of edges by chance. To identify the hub genes of the PPI networks, i.e., the genes with the highest degree of connectivity, the interactions of each gene were counted and genes with the highest number of edges were pinpointed.
4. Discussion
In this space radiation-related work and through extensive transcriptomic analysis, using two distinct radiations (X-rays or C-ions), several key and original findings were retrieved. First, comparisons between 24 h post-irradiation vs. sham irradiation (0 h) identified more DEGs than comparisons between 3 h post-radiation vs. 0 h. More specifically, and based on biological term enrichments, processes related to response to DNA damage stimulus were identified 3 h post-irradiation, and suppressed 24 h post-irradiation [
49]. Signal transduction by p53 and its downstream gene
CDKN1A (p21) class mediator were also identified mainly in early response, as previously shown [
50].
CDKN1A was found to be over-expressed 3 and 24 h after irradiation with 1 Gy of X-ray and 1 Gy of C-ion, as it was previously found [
1]. FOXO4 (Forkhead Box O4) was identified as a transcription factor inducing genes in response to irradiation [
4]. FOXOs are transcription factors that play a crucial role in cell fate decision and are involved in the promotion of apoptosis [
51].
CDKN1A,
MDM2,
PURPL,
PTCHD4,
TP53INP1,
PAPPA and
BTG2 were found to be up-regulated post-radiation in 1G and simulated μG condition, having the lowest
p-values, as well as the highest Log
2FoldChanges.
MDM2 was found to be over-expressed after irradiation with 1 Gy of X-ray and 1 Gy of C-ion, as previously found [
1].
TP53INP1 was found to be up-regulated in human irradiated fibroblasts and is associated with the regulation of apoptosis [
52,
53].
MKI67 and
CCNB1 had a lower expression in response to radiation, especially 24 h post-irradiation in 1G or simulated μG conditions [
1]. Histone-clustered genes (
H2AC13,
H3C2,
H3C10,
H3C8 and
H4C3) were found to be down-regulated genes post-radiation in 1G and simulated μG condition, as radiation suppressed the expression of various histone-clustered genes [
54].
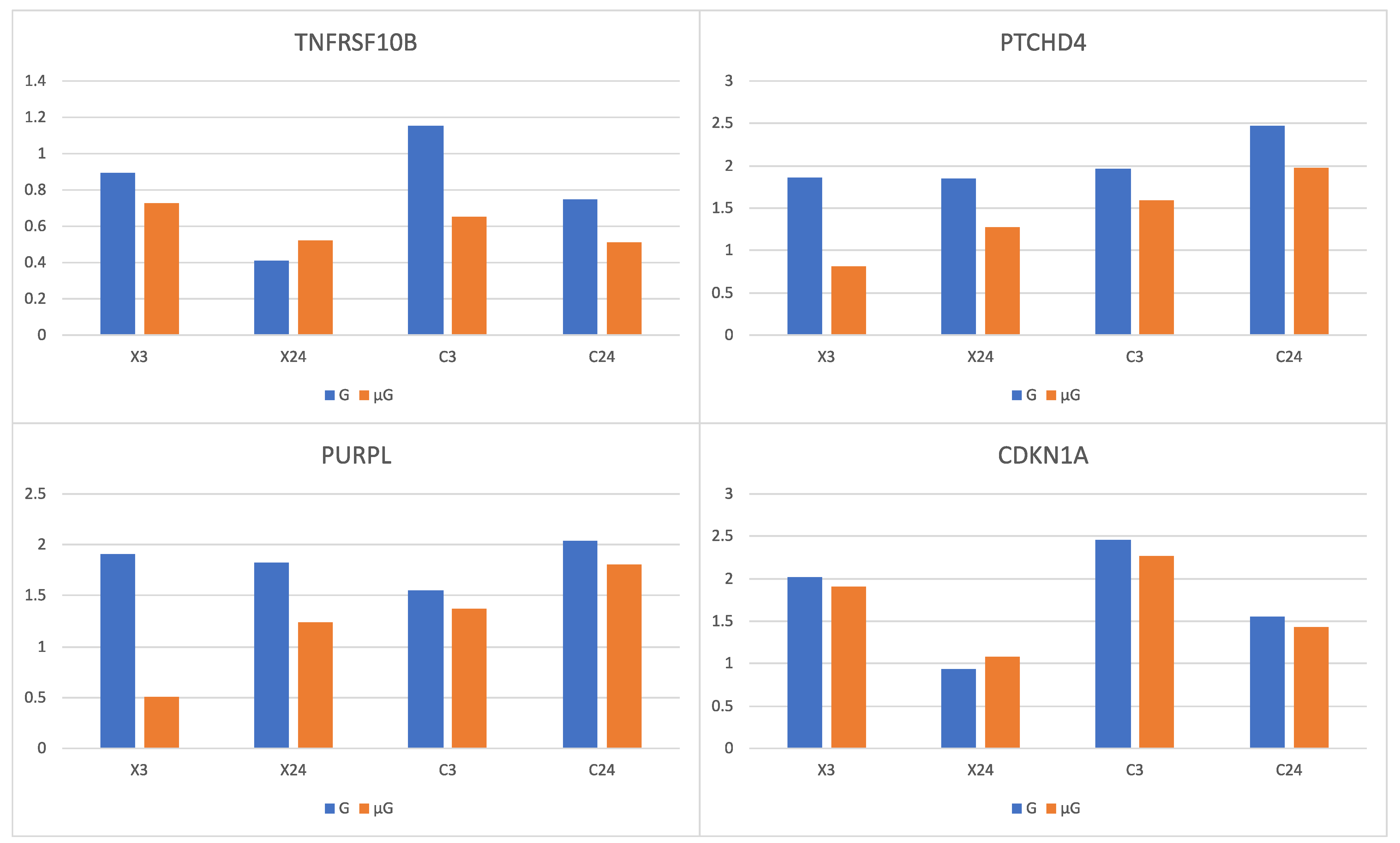
At a second level, the combined effect of C-ion or X-ray and simulated μG resulted in the up-regulation of
TNFRSF10B,
PTCHD4 and
PURPL. Their expression increased with irradiation alone, but their expression increase was lower when irradiation was combined with simulated μG than when it was combined with 1G. The expression of
CDKN1A was found to behave in a similar way, but its lower increase was less statistically significant (
Figure 5). It has been reported that the combined effect of C-ion radiation and simulated μG results in a lower increase of the expression of
CDKN1A, compared to the effect of C-ion radiation treatment alone [
1]. The expression of
TNFRSF10B significantly increased in irradiated germline stem cells (GSCs) [
55]. TNFSF10-TNFRSF10B pathway was found to be involved in radiation-induced apoptosis. The combined effect of radiation and simulated μG, may reduce the role of the TNFSF10-TNFRSF10B pathway that is crucial in the regulation of response to radiation, suppressing the process of apoptosis, meaning unrepaired cells not to undergo apoptosis, potentially causing the duplication of damaged cells.
PURPL is a long non-coding RNA (lncRNA) [
56].
PURPL has been reported to be up-regulated after DNA damage [
44].
PURPL expression is anti-correlated with that of
TP53. It was suggested that its anti-apoptotic action is due to its regulation of the
TP53 gene [
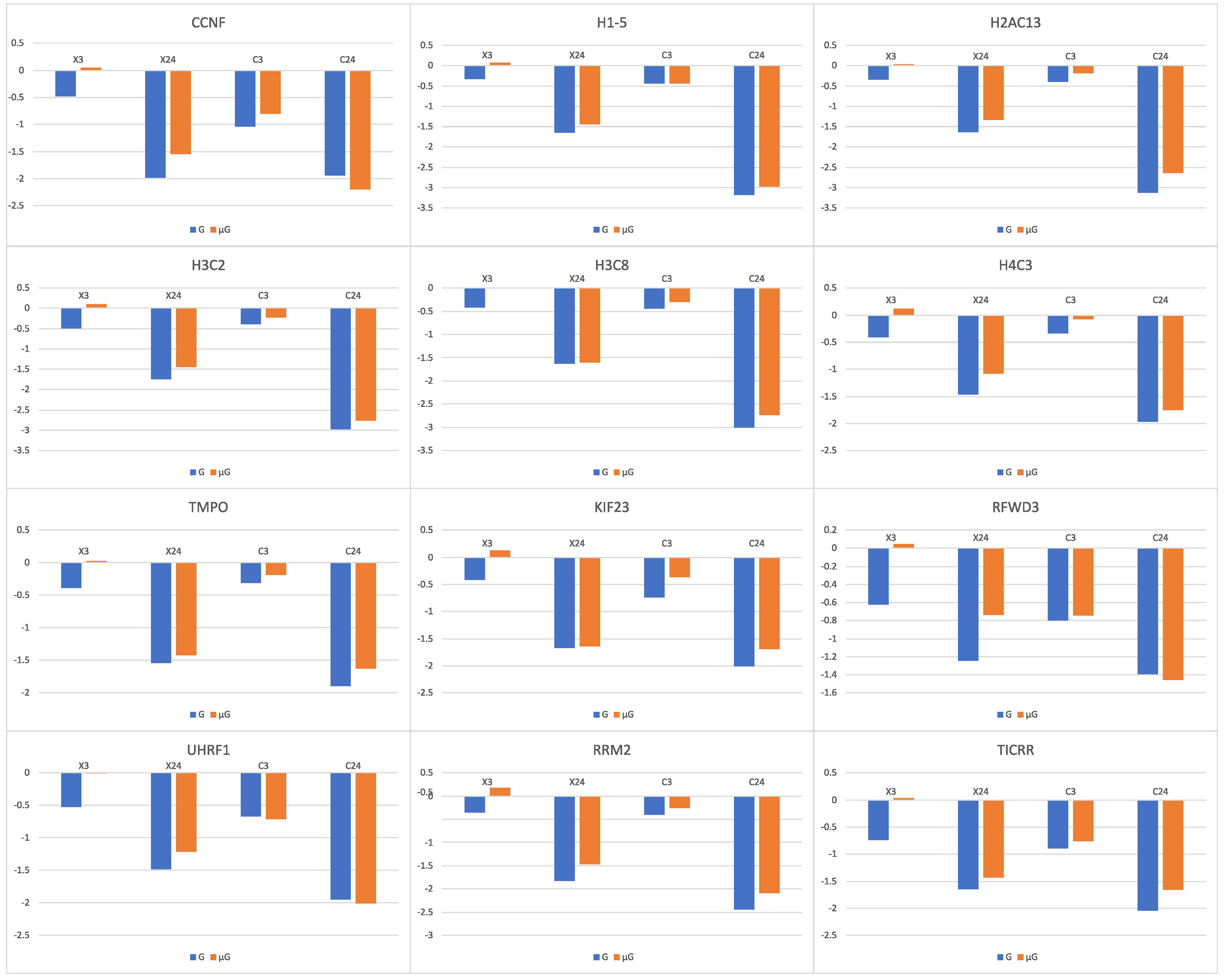
56]. It was found that in the 12 genes (
CCNF,
H1-5,
H2AC13,
H3C2,
H3C8,
H4C3,
TMPO,
KIF23,
RFWD3,
UHRF1,
RRM2,
TICRR) whose expression was decreased with irradiation alone, simulated μG in combination with irradiation resulted in a lower gene expression decrease (
Figure 6). The lowered transcriptional response to irradiation under simulated μG of genes that are related to cell apoptosis and DNA damage repair may explain the observed increase in chromosome aberrations in cells that were exposed simultaneously to radiation and simulated μG [
2].
On a parallel direction of application of our accumulated data presented, a significant number of genes found to be affected by the combined effect of microgravity and radiation, are related to the following pathways: apoptosis, DNA damage response and repair, and cell cycle. All these pathways can be considered as potential drug targets for resistant tumors. Apoptosis-related genes
BLOC1S2,
EDA2R,
TP53INP1,
MDM2,
CDKN1A,
FAS,
BCL2L1, repair-related genes
BRCA1,
POLQ,
BLM,
H2AFX or cell cycle-related genes
MKI67,
CDT1,
CDC6,
MSH6,
TERT and other genes highly impacted by the microgravity environment open the possibility for targeting by microgravity conditions. Altered cancer cell gene expression after the application of microgravity can be used as a roadmap for the fight against cancer even without any other drug or chemical treatment [
57]. For example, the down-regulation of cell growth or DNA repair genes can automatically sensitize cells to chemo- or radiotherapy. Another option would be to use a combination of microgravity with immunotherapy drugs. Last but not least, the results showing the ability of microgravity to especially impact cancer stem cells [
15] underlines its possible use as a feasible tool in cancer therapy.
Comparing sham-irradiated cells under simulated μG and 1G, enrichment analyses in statistically significant down-regulated genes, highlighted terms related to the response to oxygen levels, muscle contraction and regulation of blood circulation. Based on their log
2 fold changes,
TTN and
MSTN were identified as the most prominent down-regulated genes.
TTN encodes for the lengthiest human protein, Titin, which controls sarcomere elasticity and contraction and is linked to the development of muscle atrophy [
58]. As μG induces skeletal muscle atrophy [
59,
60],
TTN under-expression under μG could be responsible for muscle mass loss in space flights. It has been found that an effect of μG is also the reduced human ventilatory response to hypoxia [
61].
Biological processes related to the defense response (immune effector process and type I interferon signaling pathway) were highlighted in genes that were only differentially expressed under C-ion (high-LET) radiation but not in genes that were differentially expressed under X-ray (low-LET) radiation. Type I interferons are components of the early immune response. Strong associations between response to radiation and immune system and inflammation have been suggested in the past [
62,
63]. Although DNA repair was identified as an over-represented term in both aforementioned gene subsets, DDR terms such as double-strand break repair, double-strand break repair via the less-accurate non-homologous end joining (NHEJ) and non-recombinational repair were found exclusively in the genes that were differentially expressed under high-LET radiation [
64]. The increased complexity of damage is often associated with a lethality increase compared to low-LET radiations [
2]. Based also on our data for X-ray and C-ion-irradiated human G2-phase cells, it is suggested that classical NHEJ will make an initial attempt to repair the DSBs [
65,
66]. Examining up-regulated genes due to X-ray (low-LET) radiation, biological processes related to angiogenesis were discovered. Late down-regulated genes were found to be related to DNA damage repair, as it was previously shown [
67]. Last but not least, relating to the biological effects of IR, the generation of oxidative stress is expected [
68]. The lysyl oxidase (LOX) gene family contains five members:
LOX;
LOXL1;
LOXL2;
LOXL3; and
LOXL4 [
69], all of which were found over-expressed as a response to radiation according to our results [
70,
71]. Hydrogen peroxide is a side product of this catalytic reaction. LOX proteins are expressed in fibroblasts [
72]. Aberrant expression is involved in tumor invasion and metastasis [
73]. Thus, lysyl oxidases provide targets for pharmacological and therapeutic intervention. Another cytochrome c oxidase subunit which is up-regulated in our experiments is cytochrome c oxidase subunit 7C (
COX7C). Cytochrome c oxidase is related to the regulation of oxidative phosphorylation [
74]. Other oxidases that we discovered to be over-expressed due to IR include Acyl-CoA oxidase 2 (
ACOX2), Aldehyde oxidase 1 (
AOX1) and Quiescin sulfhydryl oxidase 1 (
QSOX1), all involved in the regulation of reactive oxygen species (ROS) homeostasis [
75,
76,
77,
78,
79,
80,
81,
82,
83]. In addition, Glutathione peroxidase 1 (
GPX1), a major antioxidant enzyme [
84], was also found to be over-expressed as a response to IR in our analysis.
Conclusively, as key gene signatures, we have identified that PLK1, BRCA1, CCNB1, AURKB, CDK1, CHEK1, RAD51, CCNA2 and TOP2A are hub genes in protein interaction networks for DEGs from the comparison between X-ray or C-ion-irradiated cells collected 24 h post-irradiation and sham-irradiated ones under 1G condition. Also, BRCA1 and RAD51 are associated with damage repair of DNA breaks. We note that PLK1, CCNB1, AURKB, CDK1, CHEK1, CCNA2 and TOP2A play a critical role in the process of mitosis.
Our current findings suggest that human cells exposed to microgravity may significantly change their response to a genuine stressor like radiation. Alterations in biological responses to space-related radiations are known and the challenge that we aimed to address in this study was to bring into surface key processes impacted by the specific types of radiations and irradiation methodologies probing the exposure of human cells to space radiations. Therefore, the field is open for the use of these results in the development of new tools and methodologies to overcome tumor resistance beyond the current use in space missions.