Is Cancer Reversible? Rethinking Carcinogenesis Models—A New Epistemological Tool
Abstract
1. Introduction
“The capacity of embryonal carcinoma cells to form normally functioning adult tissues demonstrates that conversion to neoplasia did not involve structural changes in the genome, but rather a change in gene expression”.[6]
2. Tumor Reversion—An Experimental Model Overview
- (a)
- Cancer cells display relevant plasticity, and their fate is not “irreversibly” determined.
- (b)
- It is possible to inhibit the phenotypic expression of the malignant characteristics of cancer cells mostly through epigenetic processes, although other mechanisms are likely to participate.
- (c)
- Depending on the tumor type and stage, some context-dependent conditions/constraints (such as those pertaining to the microenvironment of specific embryogenesis stages) can induce a phenotypic reversion of malignant cancer cells.
- (d)
- Gene mutations do not play a “causative” role as the somatic mutation theory (SMT) posits, albeit they can be associated throughout the process of cancer development.
3. Critical Aspects of Gene-Centric Models
“Most oncologists believe that insertion of viral information into the genome, or mutation (a structural change in the genome), is the underlying mechanism of carcinogenesis. On the basis of our experiences with spontaneously occurring embryonal carcinoma, and because all of the phenotypic traits of malignant cells appear to be encoded in the genome of normal cells, I favor the idea that the production of a neoplasm is probably similar to the production of any normal tissue (...) The mechanism of tissue genesis involves cell division, differentiation, and organization. In other words, I believe that carcinogenesis is an epigenetic event, similar to postembryonic differentiation”.[83]
“If mutation proves to be the causative event, then our discovery that malignant cells can differentiate to benign cells implies that the process of differentiation is capable of regulating the mutation that causes cancer. If expression of an oncogene is the cause of cancer, then what we have shown is that the process of differentiation represses the oncogene”.[84]
4. The Role of the Microenvironment
4.1. Essentiality of the Microenvironment in Biological Models
4.2. The Microenvironment as a Target
4.3. The Integration of the Microenvironment within Biological Models
5. The Systemic Approach
5.1. Limits of the Pragmatic Approach
5.2. From Entities to Relations
5.3. Bottom-Up and Top-Down Causation
5.4. The Morphogenetic Field Concept
5.5. Conceptual Tools for Describing a Biological Network
6. Interpreting Tumor Reversion Processes within the Systemic Framework
6.1. Genotype and Phenotype
6.2. The Systemic View of Pathology
6.3. Tumor Reversion as a Reorganization/Transition Process
7. Conclusions: Challenges and the Way to Move Forward
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Askanazy, M. Die Teratome nach ihrem Bau, ihrem Verlauf, ihrer Genese und im Vergleich zum experimentellen Teratoid. Verh. Der Dtsch. Pathol. Ges. 1907, 11, 39–82. [Google Scholar]
- Stevens, L.C.; Little, C.C. Spontaneous testicular teratomas in an inbred strain of mice. Proc. Natl. Acad. Sci. USA 1954, 40, 1080–1087. [Google Scholar] [CrossRef] [PubMed]
- Pierce, G.B.; Dixon, F.J., Jr. Testicular teratomas. I. Demonstration of teratogenesis by metamorphosis of multipotential cells. Cancer 1959, 12, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Pierce, G.B.; Verney, E.L. An in vitro and in vivo study of differentiation in teratocarcinomas. Cancer 1961, 14, 1017–1029. [Google Scholar] [CrossRef] [PubMed]
- Brinster, R.L. The effect of cells transferred into the mouse blastocyst on subsequent development. J. Exp. Med. 1974, 140, 1049–1056. [Google Scholar] [CrossRef] [PubMed]
- Mintz, B.; Illmensee, K. Normal genetically mosaic mice produced from malignant teratocarcinoma cells. Proc. Natl. Acad. Sci. USA 1975, 72, 3585–3589. [Google Scholar] [CrossRef]
- Dolberg, D.S.; Bissell, M.J. Inability of Rous sarcoma virus to cause sarcomas in the avian embryo. Nature 1984, 309, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Pierce, G.B.; Pantazis, C.G.; Caldwell, J.E.; Wells, R.S. Specificity of the control of tumor formation by the blastocyst. Cancer Res. 1982, 42, 1082–1087. [Google Scholar] [PubMed]
- Gerschenson, M.; Graves, K.; Carson, S.D.; Wells, R.S.; Pierce, G.B. Regulation of melanoma by the embryonic skin. Proc. Natl. Acad. Sci. USA 1986, 83, 7303–7310. [Google Scholar] [CrossRef]
- Lee, L.M.; Seftor, E.A.; Bonde, G.; Cornell, R.A.; Hendrix, M.J. The fate of human malignant melanoma cells transplanted into zebrafish embryos: Assessment of migration and cell division in the absence of tumor formation. Dev. Dyn. 2005, 233, 1560–1570. [Google Scholar] [CrossRef] [PubMed]
- Cucina, A.; Biava, P.M.; D’anselmi, F.; Coluccia, P.; Conti, F.; Di Clemente, R.; Miccheli, A.; Frati, L.; Gulino, A.; Bizzarri, M. Zebrafish embryo proteins induce apoptosis in human colon cancer cells (Caco2). Apoptosis 2006, 11, 1617–1628. [Google Scholar] [CrossRef] [PubMed]
- Proietti, S.; Cucina, A.; Pensotti, A.; Biava, P.M.; Minini, M.; Monti, N.; Catizone, A.; Ricci, G.; Leonetti, E.; Harrath, A.H.; et al. Active Fraction from Embryo Fish Extracts Induces Reversion of the Malignant Invasive Phenotype in Breast Cancer through Down-regulation of TCTP and Modulation of E-cadherin/β-catenin Pathway. Int. J. Mol. Sci. 2019, 20, 2151. [Google Scholar] [CrossRef]
- Lichtenberg, F.R. Has Medical Innovation Reduced Cancer Mortality? NBER Working Paper No. w15880. Available online: https://ssrn.com/abstract=1586687 (accessed on 12 April 2010).
- Hanahan, D. Rethinking the war on cancer. Lancet 2014, 383, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Proietti, S.; Cucina, A.; Pensotti, A.; Fuso, A.; Marchese, C.; Nicolini, A.; Bizzarri, M. Tumor reversion and embryo morphogenetic factors. Semin. Cancer Biol. 2020, 79, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Braun, A.C. A Demonstration of the Recovery of the Crown-Gall Tumor Cell with the Use of Complex Tumors of Single-Cell Origin. Proc. Natl. Acad. Sci. USA 1959, 45, 932–938. [Google Scholar] [CrossRef]
- Macpherson, I. Reversion in Hamster Cells Transformed by Rous Sarcoma Virus. Science 1965, 148, 1731–1733. [Google Scholar] [CrossRef] [PubMed]
- Rose, S.M.; Wallingford, H.M. Transformation of renal tumors of frogs to normal tissues in regenerating limbs of salamanders. Science 1948, 107, 457. [Google Scholar] [PubMed]
- Rose, S.M. Epidermal dedifferentiation during blastema formation in regenerating limbs of Triturus viridescens. J. Exp. Zoöl. 1948, 108, 337–361. [Google Scholar] [CrossRef] [PubMed]
- Gersch, M. Zellentartung und Zellwucherung bei wirbellosen Tieren. Arch. Geschwulst-Forsch. 1951, 3, 1–18. [Google Scholar]
- Seilern-Aspang, F.; Kratochwil, K. Induction and Differentiation of an Epithelial Tumour in the Newt (Triturus cristatus). Development 1962, 10, 337–356. [Google Scholar] [CrossRef]
- Coleman, W.B.; Wennerberg, A.E.; Smith, G.J.; Grisham, J.W. Regulation of the differentiation of diploid and some aneuploid rat liver epithelial (stemlike) cells by the hepatic microenvironment. Am. J. Pathol. 1993, 142, 1373–1382. [Google Scholar]
- Podesta, A.N.; Mullins, J.; Pierce, G.B.; Sells, R.S. The neurula state mouse embryos in control of neuroblastomas. Proc. Natl. Acad. Sci. USA 1984, 81, 7608–7611. [Google Scholar] [CrossRef]
- Gootwine, E.; Webb, C.G.; Sachs, L. Participation of myeloid leukaemia cells injected into embryos in haemato poietic differentiation in adult mice. Nature 1982, 299, 63–65. [Google Scholar] [CrossRef] [PubMed]
- DeCosse, J.J.; Gossens, C.L.; Kuzma, J.F.; Unsworth, B.R. Breast cancer: Induction of differentiation by embryonic tissue. Science 1973, 181, 1057–1058. [Google Scholar] [CrossRef]
- Biava, P.M.; Fiorito, A.; Negro, C.; Mariani, M. Effects of treatment with embryonic and uterine tissue homogenates on Lewis lung carcinoma development. Cancer Lett. 1988, 41, 265–270. [Google Scholar] [PubMed]
- Biava, P.M.; Bonsignorio, D.; Hosha, M. Cell proliferation curves of different human tumor lines after in vitro treatment with Zebrafish embryonic extracts. J. Tumor Marker Oncol. 2001, 16, 195–201. [Google Scholar]
- Biava, P.M.; Bonsignorio, D. Cancer and cell differentiation: A model to explain malignancy. J. Tumor Marker Oncol. 2002, 17, 47–53. [Google Scholar]
- Breitman, T.R.; E Selonick, S.; Collins, S.J. Induction of differentiation of the human promyelocytic leukemia cell line (HL-60) by retinoic acid. Proc. Natl. Acad. Sci. USA 1980, 77, 2936–2940. [Google Scholar] [CrossRef]
- Rohdenburg, G.L. Fluctuations in the growth of malignant tumors in man, with especial reference to spontaneous regression. J. Cancer Res. 1918, 3, 192–221. [Google Scholar]
- Everson, T.C.; Cole, W.H. Spontaneous Regression of Cancer; W.B. Saunders: Philadelphia, PA, USA, 1966. [Google Scholar]
- Challis, G.B.; Stam, H.J. The Spontaneous Regression of Cancer. A Review of Cases from 1900 to 1987. Acta Oncol. 1990, 29, 545–549. [Google Scholar] [CrossRef] [PubMed]
- O’Regan, B.; Hirschberg, C. Spontaneous Regression. An Annotated Bibliography; Institute of Noetic Science: Sausalito, CA, USA, 1993. [Google Scholar]
- Papac, R.J. Spontaneous regression of cancer: Possible mechanisms. Vivo 1998, 12, 571–578. [Google Scholar]
- Livraghi, T.; Meloni, F.; Frosi, A.; Lazzaroni, S.; Bizzarri, M.; Frati, L.; Biava, P.M. Treatment with Stem Cell Differentiation Stage Factors of Intermediate-Advanced Hepatocellular Carcinoma: An Open Randomized Clinical Trial. Oncol. Res. Featur. Preclin. Clin. Cancer Ther. 2005, 15, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Hendrix, M.J.C.; Seftor, E.A.; Seftor, R.E.B.; Kasemeier-Kulesa, J.; Kulesa, P.M.; Postovit, L.-M. Reprogramming metastatic tumour cells with embryonic microenvironments. Nat. Rev. Cancer 2007, 7, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Postovit, L.-M.; Margaryan, N.V.; Seftor, E.A.; Kirschmann, D.A.; Lipavsky, A.; Wheaton, W.W.; Abbott, D.E.; Seftor, R.E.B.; Hendrix, M.J.C. Human embryonic stem cell microenvironment suppresses the tumorigenic phenotype of aggressive cancer cells. Proc. Natl. Acad. Sci. USA 2008, 105, 4329–4334. [Google Scholar] [CrossRef] [PubMed]
- Giuffrida, D.; Rogers, I.M.; Nagy, A.; Calogero, A.E.; Brown, T.J.; Casper, R.F. Human embryonic stem cells secrete soluble factors that inhibit cancer cell growth. Cell Prolif. 2009, 42, 788–798. [Google Scholar] [CrossRef] [PubMed]
- Costa, F.F.; A Seftor, E.; Bischof, J.M.; A Kirschmann, D.; Strizzi, L.; Arndt, K.; Bonaldo, M.D.F.; Soares, M.B.; Hendrix, M.J. Epigenetically reprogramming metastatic tumor cells with an embryonic microenvironment. Epigenomics 2009, 1, 387–398. [Google Scholar] [CrossRef]
- Allegrucci, C.; Rushton, M.D.; E Dixon, J.; Sottile, V.; Shah, M.; Kumari, R.; Watson, S.; Alberio, R.; Johnson, A.D. Epigenetic reprogramming of breast cancer cells with oocyte extracts. Mol. Cancer 2011, 10, 7. [Google Scholar] [CrossRef]
- Huang, S. Reconciling Non-Genetic Plasticity with Somatic Evolution in Cancer. Trends Cancer 2021, 7, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Versteeg, R. Tumours outside the mutation box. Nature 2014, 506, 438–439. [Google Scholar] [CrossRef]
- Weinberg, R.A. Coming Full Circle—From Endless Complexity to Simplicity and Back Again. Cell 2014, 157, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Monti, N.; Verna, R.; Piombarolo, A.; Querqui, A.; Bizzarri, M.; Fedeli, V. Paradoxical Behavior of Oncogenes Undermines the Somatic Mutation Theory. Biomolecules 2022, 12, 662. [Google Scholar] [CrossRef]
- Baker, S.G.; Kramer, B.S. Paradoxes in carcinogenesis: New opportunities for research directions. BMC Cancer 2007, 7, 151. [Google Scholar] [CrossRef]
- Marcum, J.A. Metaphysical presuppositions and scientific practices: Reductionism and organicism in cancer research. Int. Stud. Philos. Sci. 2005, 19, 31–45. [Google Scholar] [CrossRef]
- Ashby, J.; Purchase, I. Reflections on the declining ability of the Salmonella assay to detect rodent carcinogens as positive. Mutat. Res. Toxicol. 1988, 205, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Lijinsky, W. Non-genotoxic environmental carcinogens. Environ. Carcinog. Rev. 1990, 8, 45–87. [Google Scholar] [CrossRef]
- Greenman, C.; Stephens, P.; Smith, R.; Dalgliesh, G.L.; Hunter, C.; Bignell, G.; Davies, H.; Teague, J.; Butler, A.; Stevwns, C.; et al. Patterns of somatic mutation in human cancer genomes. Nature 2007, 446, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Imielinski, M.; Berger, A.H.; Hammerman, P.S.; Hernandez, B.; Pugh, T.J.; Hodis, E.; Cho, J.; Suh, J.; Capelletti, M.; Sivachenko, A.; et al. Mapping the hallmarks of lung adenocarcinoma with massively parallel sequencing. Cell 2012, 150, 1107–1120. [Google Scholar] [CrossRef]
- Lawrence, M.S.; Stojanov, P.; Polak, P.; Kryukov, G.V.; Cibulskis, K.; Sivachenko, A.; Carter, S.L.; Stewart, C.; Mermel, C.H.; Roberts, S.A.; et al. Mutational heterogene- ity in cancer and the search for new cancer associated genes. Nature 2013, 499, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Konishi, N.; Hiasa, Y.; Matsuda, H.; Tao, M.; Tsuzuki, T.; Hayashi, I.; Kitahori, Y.; Shiraishi, T.; Yatana, R.; Shimazaki, J. Intratumor cellular heterogeneity and alterations in ras oncogene and p53 tumor suppressor gene in human prostate carcinoma. Am. J. Pathol. 1995, 147, 112–122. [Google Scholar]
- Baisse, B.; Bouzourene, H.; Saraga, E.P.; Bosman, F.T.; Benhattar, J. Intratumor genetic heterogeneity in advanced human colorectal adenocarcinoma. Int. J. Cancer 2001, 93, 346–352. [Google Scholar] [CrossRef]
- van Elsas, A.; Zerp, S.; van der Flier, S.; Krüse-Wolters, M.; Vacca, A.; Ruiter, D.J.; Schrier, P. Analysis of N-ras Mutations in Human Cutaneous Melanoma: Tumor Heterogeneity Detected by Polymerase Chain Reaction/Single-Stranded Conformation Polymorphism Analysis. Recent Results Cancer Res. 1995, 139, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Sauter, G.; Moch, H.; Moore, D.; Carroll, P.; Kerschmann, R.; Chew, K.; Mihatsch, M.J.; Gudat, F.; Waldman, F. Heterogeneity of erbB-2 gene amplification in bladder cancer. Cancer Res. 1993, 53 (Suppl. 10), 2199–2203. [Google Scholar] [PubMed]
- Szöllösi, J.; Balázs, M.; Feuerstein, B.G.; Benz, C.C.; Waldman, F.M. erBB-2 (her2/neu) gene copy number, p185her-2 overexpression and intratumor heterogeneity in human breast cancer. Cancer Res. 1995, 55, 5400–5407. [Google Scholar] [PubMed]
- Park, S.H.; Maeda, T.; Mohapatra, G.; Waldman, F.M.; Davis, R.L.; Feuerstein, B.G. Heterogeneity, polyploidy, aneusomy, and 9p deletion in human glioblastoma multiforme. Cancer Genet. Cytogenet. 1995, 83, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Washington, C.; Dalbègue, F.; Abreo, F.; Taubenberger, J.K.; Lichy, J.H. Loss of Heterozygosity in Fibrocystic Change of the Breast: Genetic Relationship Between Benign Proliferative Lesions and Associated Carcinomas. Am. J. Pathol. 2000, 157, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Paez, J.G.; Jänne, P.A.; Lee, J.C.; Tracy, S.; Greulich, H.; Gabriel, S.; Herman, P.; Kaye, F.J.; Lindemanm, N.; Boggon, T.J.; et al. EGFR mutations in lung cancer: Correlation with clinical response to gefitinib therapy. Science 2004, 304, 1497–1500. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhou, W.; Velculescu, V.E.; Kern, S.E.; Hruban, R.H.; Hamilton, S.R.; Vogelstein, B.; Kinzler, K.W. Gene expression profiles in normal and cancer cells. Science 1997, 276, 1268–1272. [Google Scholar] [CrossRef]
- Chanock, S.J.; Thomas, G. The devil is in the DNA. Nat. Genet. 2007, 39, 283–284. [Google Scholar] [CrossRef] [PubMed]
- Martincorena, I.; Roshan, A.; Gerstung, M.; Ellis, P.; Van Loo, P.; McLaren, S.; Wedge, D.C.; Fullam, A.; Alexandrov, L.B.; Tobio, J.M.; et al. High burden and pervasive positive selection of somatic mutations in normal human skin. Science 2015, 348, 880–886. [Google Scholar] [CrossRef]
- Kan, Z.; Jaiswal, B.S.; Stinson, J.; Janakiraman, V.; Bhatt, D.; Stern, H.M.; Yue, P.; Haverty, P.M.; Bourgon, R.; Zheng, J.; et al. Diverse somatic mutation patterns and pathway alterations in human cancers. Nature 2010, 466, 869–873. [Google Scholar] [CrossRef]
- Baker, S.G. A Cancer Theory Kerfuffle Can Lead to New Lines of Research. Gynecol. Oncol. 2014, 107, dju405. [Google Scholar] [CrossRef] [PubMed]
- Duesberg, P.; Li, R.; Rasnick, D. Aneuploidy Approaching a Perfect Score in Predicting and Preventing Cancer: Highlights from a Conference Held in Oakland, CA in January, 2004. Cell Cycle 2004, 3, 821–826. [Google Scholar] [CrossRef] [PubMed]
- Prehn, R.T. Cancers beget mutations versus mutations beget cancers. Cancer Res. 1994, 54, 5296–5300. [Google Scholar] [PubMed]
- Prehn, R.T. The role of mutation in the new cancer paradigm. Cancer Cell Int. 2005, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, D.J. Biological atomism and cell theory. Stud. Hist. Philos. Sci. Part C Stud. Hist. Philos. Biol. Biomed. Sci. 2010, 41, 202–211. [Google Scholar] [CrossRef]
- Vogelstein, B.; Papadopoulos, N.; Velculescu, V.E.; Zhou, S.; Diaz, L.A.; Kinzler, K.W. Cancer genome landscapes. Science 2013, 339, 1546–1558. [Google Scholar] [CrossRef]
- Garraway, L.A.; Lander, E.S. Lessons from the cancer genome. Cell 2013, 153, 17–37. [Google Scholar] [CrossRef]
- Bertolaso, M. Philosophy of Cancer—A Dynamic and Relational View; Springer: Berlin/Heidelberg, Germany. [CrossRef]
- Kamb, A.; Wee, S.; Lengauer, C. Why is cancer drug discovery so difficult? Nat. Rev. Drug Discov. 2007, 6, 115–120. [Google Scholar] [CrossRef]
- Cassidy, J.W.; Bruna, A. Chapter 4—Tumor Heterogeneity. In Patient Derived Tumor Xenograft Models; Uthamanthil, R., Tinkey, P., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 37–55. [Google Scholar] [CrossRef]
- Seoane, J. Cancer: Division hierarchy leads to cell heterogeneity. Nature 2017, 549, 164–166. [Google Scholar] [CrossRef]
- Li, R.X.; Zeng, R. Dynamic proteomics for investigating the response of individual cancer cells under drug action. Expert Rev. Proteom. 2009, 6, 19–21. [Google Scholar] [CrossRef]
- Bhang, H.E.C.; Ruddy, D.A.; Krishnamurthy Radhakrishna, V.; Caushi, J.X.; Zhao, R.; Hims, M.M.; Singh, A.P.; Kao, I.; Rakiec, D.; Shaw, P.; et al. Studying clonal dynamics in response to cancer therapy using high-complexity barcoding. Nat. Med. 2015, 21, 440–448. [Google Scholar] [CrossRef]
- Ness, R.B. Fear of Failure: Why American Science Is not Winning the War on Cancer. Ann. Epidemiol. 2010, 20, 89–91. [Google Scholar] [CrossRef]
- Miklos, G.L.G. The Human Cancer Genome Project—One more misstep in the war on cancer. Nat. Biotechnol. 2005, 23, 535–537. [Google Scholar] [CrossRef]
- Leppert, J.; Patel, C. Beyond the genome. Nature 2015, 518, 273. [Google Scholar]
- Kaelin, W.G. The concept of synthetic lethality in the context of anticancer therapy. Nature Rev. Cancer. 2005, 5, 689–698. [Google Scholar] [CrossRef]
- Joyner, M.J.; Paneth, N. Promises, promises, and precision medicine. J. Clin. Investig. 2019, 129, 946–948. [Google Scholar] [CrossRef] [PubMed]
- Fojo, T.; Mailankody, S.; Lo, A. Unintended consequences of expensive cancer therapeutics—The pursuit of marginal indications and a me-too mentality that stifles innovation and creativity: The John Conley Lecture. JAMA Otolaryngol. Head Neck Surg. 2014, 140, 1225–1236. [Google Scholar] [CrossRef] [PubMed]
- Pierce, G.B.; Johnson, L.D. Differentiation and cancer. In Vitro 1971, 7, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Pierce, G.B. The cancer cell and its control by the embryo. Rous-Whipple Award lecture. Am. J. Pathol. 1983, 113, 115–124. [Google Scholar] [PubMed]
- Noble, D. Claude Bernard, the first systems biologist, and the future of physiology. Exp. Physiol. 2007, 93, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Soto, A.M.; Sonnenschein, C. The cancer puzzle: Welcome to organicism. Prog. Biophys. Mol. Biol. 2021, 165, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Dennett, D.C. Darwin’s Dangerous Idea; Simon & Schuster: New York, NY, USA, 1995. [Google Scholar]
- Paget, S. The distribution of secondary growths in cancer of the breast. Cancer Metastasis Rev. 1989, 133, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Langley, R.R.; Fidler, I.J. The seed and soil hypothesis revisited-The role of tumor-stroma interactions in metastasis to different organs. Int. J. Cancer 2011, 128, 2527–2535. [Google Scholar] [CrossRef] [PubMed]
- Bayir, E.; Sahinler, M.; Celtikoglu, M.M.; Sendemir, A. Biomaterials for Organ and Tissue Regeneration; Elsevier: Amsterdam, The Netherlands, 2020; pp. 709–752. [Google Scholar] [CrossRef]
- Chang, H.H.; Hemberg, M.; Barahona, M.; Ingber, D.E.; Huang, S. Transcriptome-wide noise controls lineage choice in mammalian progenitor cells. Nature 2008, 453, 544–547. [Google Scholar] [CrossRef]
- Oakley, E.J.; Van Zant, G. Unraveling the complex regulation of stem cells: Implications for aging and cancer. Leukemia 2007, 21, 612–621. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, M.; Piras, V.; Choi, S.; Akira, S.; Tomita, M.; Giuliani, A.; Selvarajoo, K. Emergent genome-wide control in wild-type and genetically mutated lipopolysaccharides-stimulated macrophages. PLoS ONE 2009, 4, e4905. [Google Scholar] [CrossRef]
- Barabási, A.-L.; Oltvai, Z.N. Network biology: Understanding the cell’s functional organization. Nat. Rev. Genet. 2004, 5, 101–113. [Google Scholar] [CrossRef]
- Pearson, H. Surviving a knockout blow. Nature 2002, 415, 8–9. [Google Scholar] [CrossRef]
- Bizzarri, M.; Cucina, A.; Conti, F.; D’anselmi, F. Beyond the Oncogene Paradigm: Understanding Complexity in Cancerogenesis. Acta Biotheor. 2008, 56, 173–196. [Google Scholar] [CrossRef]
- Scannell, J.W.; Bosley, J. When quality beats quantity: Decision theory, drug discovery and the reproducibility crisis. PLoS ONE 2016, 11, e0147215. [Google Scholar] [CrossRef]
- Bizzarri, M. Do new anticancer drugs really work? A serious concern, Organisms. J. Biol. Sciences. 2017, 1, 9–10. [Google Scholar]
- Leung, C.T.; Brugge, J.S. Outgrowth of single oncogene-expressing cells from suppressive epithelial environments. Nature 2012, 482, 410–413. [Google Scholar] [CrossRef]
- Maffini, M.V.; Soto, A.M.; Calabro, J.M.; Ucci, A.A.; Sonnenschein, C. The stroma as a crucial target in rat mammary gland carcinogenesis. J. Cell Sci. 2004, 117, 1495–1502. [Google Scholar] [CrossRef]
- Rana, B.; Mischoulon, D.; Xie, Y.; Bucher, N.L.; Farmer, S.R. Cell-extracellular matrix interactions can regulate the switch between growth and differentiation in rat hepatocytes: Reciprocal expression of C/EBP alpha and immediate-early growth response transcription factors. Mol. Cell. Biol. 1994, 14, 5858–5869. [Google Scholar] [CrossRef]
- Huang, S.; Chen, C.S.; Ingber, D.E. Control of cyclin D1, p27Kip1, and cell cycle progression in human capillary endothe- lial cells by cell shape and cytoskeletal tension. Mol. Biol. Cell 1998, 9, 3179–3193. [Google Scholar] [CrossRef] [PubMed]
- Dike, L.E.; Ingber, D.E. Integrin-dependent induction of early growth response genes in capillary endothelial cells. J. Cell Sci. 1996, 109, 2855–2863. [Google Scholar] [CrossRef] [PubMed]
- Roskelley, C.D.; Desprez, P.Y.; Bissell, M.J. Extracellular matrix-dependent tissue-specific gene expression in mammary epithelial cells requires both physical and biochemical signal transduction. Proc. Natl. Acad. Sci. USA 1994, 91, 12378–12382. [Google Scholar] [CrossRef]
- Boudreau, N.; Myers, C.; Bissell, M.J. From laminin to lamin: Regulation of tissue-specific gene expression by the ECM. Trends Cell Biol. 1995, 5, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.S.; Mrksich, M.; Huang, S.; Whitesides, G.M.; Ingber, D.E. Geometric Control of Cell Life and Death. Science 1997, 276, 1425–1428. [Google Scholar] [CrossRef]
- Paszek, M.J.; Zahir, N.; Johnson, K.R.; Lakins, J.N.; Rozenberg, G.I.; Gefen, A.; Reinhart-King, C.A.; Margulies, S.S.; Dembo, M.; Boettiger, D.; et al. Tensional home- ostasis and the malignant phenotype. Cancer Cell 2005, 8, 241–254. [Google Scholar] [CrossRef]
- Colpaert, C.G.; Vermeulen, P.B.; Fox, S.; Harris, A.; Dirix, L.Y.; Van Marck, E.A. The Presence of a Fibrotic Focus in Invasive Breast Carcinoma Correlates with the Expression of Carbonic Anhydrase IX and is a Marker of Hypoxia and Poor Prognosis. Breast Cancer Res. Treat. 2003, 81, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.J.; Boyd, N.F. Mammographic density. Potential mechanisms of breast cancer risk associated with mammographic density: Hypotheses based on epidemiological evidence. Breast Cancer Res. 2008, 10 (Suppl. 1), 201. [Google Scholar] [CrossRef] [PubMed]
- Meredith, J.E., Jr.; Fazeli, B.; Schwartz, M.A. Theextracellular matrix as a cell survival factor. Mol. Biol. Cell 1993, 4, 953–961. [Google Scholar] [CrossRef]
- Boudreau, N.; Sympson, C.J.; Werb, Z.; Bissell, M.J. Suppression of ICE and Apoptosis in Mammary Epithelial Cells by Extracellular Matrix. Science 1995, 267, 891–893. [Google Scholar] [CrossRef] [PubMed]
- Vosseler, S.; Mirancea, N.; Bohlen, P.; Mueller, M.M.; Fusenig, N.E. Angiogenesis inhibition by vascular endothelial growth factor receptor-2 blockade reduces stromal matrix metalloproteinase expression, normalizes stromal tissue, and reverts epithelial tumor phenotype in surface heterotransplants. Cancer Res. 2005, 65, 1294–1305. [Google Scholar] [CrossRef] [PubMed]
- Sternlicht, M.D.; Lochter, A.; Sympson, C.J.; Huey, B.; Rougier, J.-P.; Gray, J.W.; Pinkel, D.; Bissell, M.J.; Werb, Z. The Stromal Proteinase MMP3/Stromelysin-1 Promotes Mammary Carcinogenesis. Cell 1999, 98, 137–146. [Google Scholar] [CrossRef]
- Wiseman, B.S.; Werb, Z. Stromal Effects on Mammary Gland Development and Breast Cancer. Science 2002, 296, 1046–1049. [Google Scholar] [CrossRef]
- Bissell, M.J.; LaBarge, M.A. Context, tissue plasticity, and cancer: Are tumor stem cells also regulated by the microenvironment? Cancer Cell 2005, 7, 17–23. [Google Scholar] [CrossRef]
- Huang, S. Back to the biology in systems biology: What can we learn from biomolecular networks? Brief. Funct. Genom. 2004, 2, 279–297. [Google Scholar] [CrossRef]
- Craver, C.F. The explanatory power of network models. Philos. Sci. 2016, 83, 698–709. [Google Scholar] [CrossRef]
- Bizzarri, M.; Cucina, A. Tumor and the microenvironment: A chance to reframe the paradigm of carcinogenesis? BioMed Res. Int. 2014, 2014, 934038. [Google Scholar] [CrossRef] [PubMed]
- Noble, D. Modelling the heart e from genes to cells to the whole organ. Science 2002, 295, 1678–1682. [Google Scholar] [CrossRef] [PubMed]
- Werner, E. How central is the genome. Science 2007, 317, 753–754. [Google Scholar] [CrossRef]
- Longo, G.; Miquel, P.A.; Sonnenschein, C.; Soto, A.M. Is information a proper observable for biological organization? Prog. Biophys. Mol. Biol. 2012, 109, 108–114. [Google Scholar] [CrossRef]
- Newman, S.A. Developmental mechanisms: Putting genes in their place. J. Biosci. 2002, 27, 97–104. [Google Scholar] [CrossRef]
- Calvert, J. Systems Biology, Interdisciplinarity and Disciplinary Identity. In Collaboration in the New Life Sciences; Parker, J.N., Vermeulen, N., Penders, B., Eds.; Ashgate: Farnham, Surrey, UK, 2010; pp. 219–224. [Google Scholar]
- Mazzocchi, F Complexity in biology. EMBO Rep. 2008, 9, 10–14. [CrossRef]
- Bizzarri, M.; Giuliani, A.; Minini, M.; Monti, N.; Cucina, A. Constraints Shape Cell Function and Morphology by Canalizing the Developmental Path along the Waddington’s Landscape. BioEssays 2020, 42, 1900108. [Google Scholar] [CrossRef]
- Boogerd, F.C.; Bruggeman, F.J.; Hofmeyer, J.-H.S.; Westerhoff, H.V. (Eds.) Systems Biology: Philosophical Foundations; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- O’Malley, M.A.; Dupre, J. Fundamental issues in systems biology. BioEssays 2005, 27, 1270–1276. [Google Scholar] [CrossRef]
- Macheras, P.; Iliadis, A. Modeling in Biopharmaceutics, Pharmacokinetics and Pharmacodynamics: Homogeneous and Heterogeneous Approaches; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Melham, T. Modelling, abstraction, and computation in systems biology: A view from computer science. Prog. Biophys. Mol. Biol. 2012, 111, 129–136. [Google Scholar] [CrossRef]
- Green, S.; Şerban, M.; Scholl, R.; Jones, N.; Brigandt, I.; Bechtel, W. Network analyses in systems biology: New strategies for dealing with biological complexity. Synthese 2017, 195, 1751–1777. [Google Scholar] [CrossRef]
- Bertolaso, M.; Giuliani, A.; De Gara, L. Systems biology reveals biology of systems. Complexity 2010, 16, 10–16. [Google Scholar] [CrossRef]
- Kuhn, T. Theory of Scientific Revolution; University of Chicago Press: Chicago, IL, USA, 1962; ISBN 978022645811. [Google Scholar]
- Mazzocchi, F. Could Big Data be the end of theory in science? EMBO Rep. 2015, 16, 1250–1255. [Google Scholar] [CrossRef]
- Karpatne, A.; Atluri, G.; Faghmous, J.H.; Steinbach, M.; Banerjee, A.; Ganguly, A.; Shekhar, S.; Samatova, N.; Kumar, V. Theory-guided data science: A new paradigm for scientific discovery from data. IEEE Trans. Knowl. Data Eng. 2017, 29, 2318–2331. [Google Scholar] [CrossRef]
- Claude, C.S.; Longo, G. The deluge of spurious correlations in big data. Found. Sci. 2016, 22, 595–612. [Google Scholar] [CrossRef]
- Joyce, A.R.; Palsson, B. The model organism as a system: Integrating ‘omics’ data sets. Nat. Rev. Mol. Cell Biol. 2006, 7, 198–210. [Google Scholar] [CrossRef] [PubMed]
- Assmus, H.E.; Herwig, R.; Cho, K.-H.; Wolkenhauer, O. Dynamics of biological systems: Role of systems biology in medical research. Expert Rev. Mol. Diagn. 2006, 6, 891–902. [Google Scholar] [CrossRef] [PubMed]
- Coveney, P.V.; Dougherty, E.R.; Highfield, R.R. Big data need big theory too. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2016, 374, 20160153. [Google Scholar] [CrossRef] [PubMed]
- Auffray, C.; Nottale, L. Scale relativity theory and integrative systems biology: 1: Founding principles and scale laws. Prog. Biophys. Mol. Biol. 2008, 97, 79–114. [Google Scholar] [CrossRef] [PubMed]
- Bertolaso, M.; Ratti, E. Conceptual Challenges in the Theoretical Foundations of Systems Biology. Syst. Biol. 2017, 1702, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Boogerd, F.C.; Bruggeman, F.J.; Richardson, R.C.; Stephan, A.; Westerhoff, H.V. Emergence and its place in nature: A case study of biochemical networks. Synthese 2005, 145, 131–164. [Google Scholar] [CrossRef]
- Bizzarri, M.; Palombo, A.; Cucina, A. Theoretical aspects of Systems Biology. Prog. Biophys. Mol. Biol. 2013, 112, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Szent-Gyorgyi, C. Bioenergetics; Academic Press: New York, NY, USA, 1957. [Google Scholar]
- Adey, W.R. Physiological signaling across cell membranes and cooperative influence of extremely-low frequency electromagnetic fields. In Biological Coherence and Response to External Stimuli; Fröhlich, H., Ed.; Springer: New York, NY, USA, 1988; pp. 148–170. [Google Scholar]
- Klink, O.; Hanke, W.; de Lima, V.M.F. Gravitational Influence on an Oscillating Chemical Reaction. Microgravity Sci. Technol. 2011, 23, 403–408. [Google Scholar] [CrossRef]
- Saetzler, K.; Sonnenschein, C.; Soto, A. Systems biology beyond networks: Generating order from disorder through self-organization. Semin. Cancer Biol. 2011, 21, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, A.; Filippi, S.; Bertolaso, M. Why network approach can promote a new way of thinking in biology. In Frontiers in Genetics (Systems Biology); Wu, X., Westerhoff, H., De Meyts, P., Kitano, H., Eds.; Frontiers Media SA: Lausanne, Switzerland, 2014; Volume 5, pp. 1–5. [Google Scholar]
- Bailly, F.; Longo, G. Biological organization and anti-entropy. J. Biol. Syst. 2009, 17, 63–96. [Google Scholar] [CrossRef]
- Elowitz, M.B.; Levine, A.J.; Siggia, E.D.; Swain, P.S. Stochastic gene expression in a single cell. Science 2002, 297, 1183–1186. [Google Scholar] [CrossRef]
- Laughlin, R.B.; Pines, D.; Schmalian, J.; Stojkovic, B.P.; Wolynes, P. The middle way. Proc. Natl. Acad. Sci. USA 2000, 97, 32–37. [Google Scholar] [CrossRef]
- Noble, D. Biophysics and systems biology. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2010, 368, 1125. [Google Scholar] [CrossRef]
- Barabasi, A.L. Network medicine from obesity to the “diseasome”. N. Engl. J. Med. 2007, 357, 404–407. [Google Scholar] [CrossRef]
- Soto, A.M.; Sonnenschein, C.; Miquel, P.A. On physicalism and Downward Causation in Developmental and Cancer Biology. Acta Biotheor. 2008, 56, 257–274. [Google Scholar] [CrossRef]
- Simon, H.A. The Organization of Complex Systems. In Boston Studies in the Philosophy of Science; Springer: Dordrecht, The Natherland, 1977; Volume 54. [Google Scholar] [CrossRef]
- Strange, K. The end of “naive reductionism”: Rise of systems biology or renaissance of physiology? Am. J. Physiol., Cell. Physiol. 2004, 288, C968–C974. [Google Scholar] [CrossRef]
- Bizzarri, M.; Naimark, O.; Nieto-Villar, J.; Fedeli, V.; Giuliani, A. Complexity in Biological Organization: Deconstruction (and Subsequent Restating) of Key Concepts. Entropy 2020, 22, 885. [Google Scholar] [CrossRef] [PubMed]
- Bizzarri, M.; Monici, M.; van Loon, J.J. How microgravity affects the biology of living systems. Biomed. Res. Int. 2015, 2015, 863075. [Google Scholar] [CrossRef]
- Drack, M.; Wolkenhauser, O. System approaches of Weiss and Bertalanffy and their relevance for Systems Biology today. Semin. Cancer Biol. 2011, 21, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Waddington, C.H. Cancer and the theory of organizers. Nature 1935, 135, 606–608. [Google Scholar] [CrossRef]
- Needham, J. New advances in the chemistry and biology of organized growth. Proc. R. Soc. Lond. B. Biol. Sci. 1936, 29, 1577–1626. [Google Scholar] [CrossRef]
- Thompson, D.A. On Growth and Form; The University Press: Cambridge, UK, 1917. [Google Scholar]
- Rossenbloich, B. Outline of a concept for organismic systems biology. Semin. Cancer Biol. 2001, 21, 156–164. [Google Scholar] [CrossRef]
- Goodwin, B.C. How the Leopard Changed Its Spots—The Evolution of Complexity; Charles Scribner’s Sons: New York, NY, USA, 1994. [Google Scholar]
- Gilbert, S.F.; Sarkar, S. Embracing complexity: Organicism for the 21st century. Dev. Dyn. 2000, 219, 1–9. [Google Scholar] [CrossRef]
- Gilbert, S.F.; Barresi, M.J.F. Developmental Biology, 11th ed.; Sinauer Associates Inc: Sunderland, MA, USA, 2016. [Google Scholar]
- Tyler, S.E.B. The Work Surfaces of Morphogenesis: The Role of the Morphogenetic Field. Biol. Theory 2014, 9, 194–208. [Google Scholar] [CrossRef]
- Løvtrup, S.; Løvtrup, M. The morphogenesis of molluscan shells: A mathematical account using biological parameters. J. Morphol. 1988, 197, 53–62. [Google Scholar] [CrossRef]
- Bissell, M.J. The differentiated state of normal and malignant cells or how to define a “normal” cell in culture. Int. Rev. Cytol. 1981, 70, 27–100. [Google Scholar] [PubMed]
- Clegg, J.S. Intracellular water and the cytomatrix: Some methods of study and current views. J. Cell Biol. 1984, 99, 167s–171s. [Google Scholar] [CrossRef]
- Butcher, D.T.; Alliston, T.; Weaver, V.M. A tense situation: Forcing tumour progression. Nat. Rev. Cancer 2009, 9, 108–122. [Google Scholar] [CrossRef]
- Kirson, E.D.; Dbalý, V.; Tovarys, F.; Vymazal, J.; Soustiel, J.F.; Itzhaki, A.; Mordechovich, D.; Steinberg-Shapira, S.; Gurvich, Z.; Schneiderman, R.; et al. Alternating electric fields arrest cell proliferation in animal tumour models and human brain tumours. Proc. Natl. Acad. Sci. USA 2007, 104, 10152–10157. [Google Scholar] [CrossRef] [PubMed]
- Hammond, T.G.; Benes, E.; O’Reilly, K.C.; Wolf, D.A.; Linnehan, R.M.; Taher, A.; Kaysen, J.H.; Allen, P.L.; Goodwin, T.J. Mechanical culture conditions affect gene expression: Gravity-induced changes on the space shuttle. Physiol. Genom. 2000, 3, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Bissell, M.J.; Barcellos-Hoff, M.H. The influence of extra- cellular matrix on gene expression: Is structure the message? J. Cell Sci. 1987, 1987 (Suppl. 8), 327–343. [Google Scholar] [CrossRef] [PubMed]
- Van den Hoff, A. Stromal involvement in malignant growth. Adv. Cancer Res. 1988, 50, 159–196. [Google Scholar] [CrossRef]
- Waddington, C.H. The Strategy of the Genes; George Allen & Unwin: London, UK, 1957. [Google Scholar]
- Guo, Y.; Eichler, G.S.; Feng, Y.; Ingber, D.E.; Huang, S. Towards a holistic, yet gene-centered analysis of gene expression profiles: A case study of human lung cancers. J. Biomed. Biotechnol. 2006, 2006, 69141. [Google Scholar] [CrossRef]
- DiNicola, S.; D’Anselmi, F.; Pasqualato, A.; Proietti, S.; Lisi, E.; Cucina, A.; Bizzarri, M. A Systems Biology Approach to Cancer: Fractals, Attractors, and Nonlinear Dynamics. OMICS A J. Integr. Biol. 2011, 15, 93–104. [Google Scholar] [CrossRef]
- Vidal, M.; Cusick, M.E.; Barabási, A.-L. Interactome Networks and Human Disease. Cell 2011, 144, 986–998. [Google Scholar] [CrossRef]
- Uetz, P.; Grigoriev, A. The yeast interactome. In Encyclopedia of Genetics, Genomics, Proteomics and Bioinformatics; Jorde, L.B., Little, P.F.R., Dunn, M.J., Subramaniam, S., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2005; Volume 5, pp. 2033–2051. [Google Scholar]
- Costanzo, M.; Baryshnikova, A.; Bellay, J.; Kim, Y.; Spear, E.D.; Sevier, C.S.; Ding, H.; Koh, J.Y.; Toufighi, K.; Mostafavi, S.; et al. The genetic landscape of a cell. Science 2010, 327, 425–431. [Google Scholar] [CrossRef]
- Welch, G.R. The ‘fuzzy’ interactome. Trends Biochem. Sci. 2009, 34, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Csermely, P.; Korcsmáros, T.; Kiss, H.J.; London, G.; Nussinov, R. Structure and dynamics of molecular networks: A novel paradigm of drug discovery: A comprehensive review. Pharmacol. Ther. 2013, 138, 333–408. [Google Scholar] [CrossRef] [PubMed]
- Ghoshal, G.; Barabási, A.L. Ranking stability and super-stable nodes in complex networks. Nat. Commun. 2011, 2, 394. [Google Scholar] [CrossRef]
- Bizzarri, M.; Giuliani, A.; Pensotti, A.; Ratti, E.; Bertolaso, M. Co-emergence and Collapse: The Mesoscopic Approach for Conceptualizing and Investigating the Functional Integration of Organisms. Front. Physiol. 2019, 10, 924. [Google Scholar] [CrossRef] [PubMed]
- Kitano, H. Systems Biology: A Brief Overview. Science 2002, 295, 1662–1664. [Google Scholar] [CrossRef]
- Ma’ayan, A. Introduction to Network Analysis in Systems Biology. Sci. Signal. 2011, 4, tr5. [Google Scholar] [CrossRef]
- Yoshida, Z. Non Linear Science. In The Challenge of Complex Systems; Springer: Berlin, Heidelberg, 2010. [Google Scholar]
- De Canete, J.F.; Galindo, C.; Garcia-Moral, I. System Engineering and Automation: An Interactive Educational Approach; Springer: Berlin/Heidelberg, Germany, 2011; p. 46. ISBN 978-3642202292. [Google Scholar]
- Huang, S.; Ingber, D.E. A Non-Genetic Basis for Cancer Progression and Metastasis: Self-Organizing Attractors in Cell Regulatory Networks. Breast Dis. 2007, 26, 27–54. [Google Scholar] [CrossRef]
- Binney, J.; Dowrick, N.J.; Fisher, A.J.; Newman, M.E.J. The Theory of Critical Phenomena: An Introduction to the Renormalization Group; Oxford University Press: New York, NY, USA, 1992. [Google Scholar]
- Prigogine, I.; Glansdorff, P. Thermodynamic Theory of Structure, Stability and Fluctuations; Wiley: New York, NY, USA, 1971. [Google Scholar]
- Westerhoff, H.V.; Palsson, B.O. The evolution of molecular biology into systems biology. Nat. Biotechnol. 2004, 22, 1249–1252. [Google Scholar] [CrossRef]
- Schrodinger, E. What is Life? In What Is Life? and Mind and Matter; Cambridge University Press: London, UK, 1946. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. The Hallmarks of Cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Huang, S. Genetic and non-genetic instability in tumor progression: Link between the fitness landscape and the epigenetic landscape of cancer cells. Cancer Metastasis Rev. 2013, 32, 423–448. [Google Scholar] [CrossRef] [PubMed]
- Reuveni, E.; Giuliani, A. Emergent properties of gene evolution: Species as attractors in phenotypic space. Phys. A Stat. Mech. its Appl. 2012, 391, 1172–1178. [Google Scholar] [CrossRef]
- Guerroui, S.; Deschatrette, J.; Wolfrom, C. Prolonged perturbation of the oscillations of hepatoma Fao cell proliferation by a single small dose of methotrexate. Pathol. Biol. 2005, 53, 290–294. [Google Scholar] [CrossRef]
- Zhou, J.X.; Aliyu, M.D.S.; Aurell, E.; Huang, S. Quasi-potential landscape in complex multi-stable systems. J. R. Soc. Interface 2012, 9, 3539–3553. [Google Scholar] [CrossRef] [PubMed]
- Pisco, A.O.; Huang, S. Non-genetic cancer cell plasticity and therapy-induced stemness in tumour relapse: ‘What does not kill me strengthens me’. Br. J. Cancer 2015, 112, 1725–1732. [Google Scholar] [CrossRef]
- Shen, S.; Clairambault, J. Cell plasticity in cancer cell populations. F1000Research 2020, 9, 635. [Google Scholar] [CrossRef] [PubMed]
- Chuang, H.-Y.; Hofree, M.; and Ideker, T. A decade of systems biology. Annu. Rev. Cell Dev. Biol. 2010, 26, 721–744. [Google Scholar] [CrossRef] [PubMed]
- Marr, C.; Zhou, J.X.; Huang, S. Single-cell gene expression profiling and cell state dynamics: Collecting data, correlating data points and connecting the dots. Curr. Opin. Biotechnol. 2016, 39, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Kell, D.B. Systems biology, metabolic modelling and metabolomics in drug discovery and development. Drug Discov. Today 2006, 11, 1085–1092. [Google Scholar] [CrossRef]
- Huang, S. The molecular and mathematical basis of Waddington’s epigenetic landscape: A framework for post-Darwinian biology? Bioessays 2011, 34, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Aldridge, S.; Teichmann, S.A. Single cell transcriptomics comes of age. Nat. Commun. 2020, 11, 1–4. [Google Scholar] [CrossRef]
- Kapuy, O.; He, E.; López-Avilés, S.; Uhlmann, F.; Tyson, J.J.; Novák, B. System-level feedbacks control cell cycle progression. FEBS Lett. 2009, 583, 3992–3998. [Google Scholar] [CrossRef] [PubMed]
- Haken, H. Synergetics. Phys. Bull. 1977, 20, 197. [Google Scholar] [CrossRef]
- Venegas, J.G.; Winkler, T.; Musch, G.; Vidal Melo, M.F.; Layfield, D.; Tgavalekos, N.; Fishman, A.J.; Collahan, R.J.; Bellani, G.; Scott Harris, R. Self-organized patchiness in asthma as a prelude to catastrophic shifts. Nature 2005, 434, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, G.; Tsumoto, K.; Tsuji, S.; Aihara, K. Bifurcation analysis on a hybrid systems model of intermittent hormonal therapy for prostate cancer. Phys. D Nonlinear Phenom. 2008, 237, 2616–2627. [Google Scholar] [CrossRef]
- Liu, X.; Chang, X.; Liu, R.; Yu, X.; Chen, L.; Aihara, K. Quantifying critical states of complex diseases using single-sample dynamic network biomarkers. PLoS Comput. Biol. 2017, 13, e1005633. [Google Scholar] [CrossRef]
- Chen, L.; Liu, R.; Liu, Z.-P.; Li, M.; Aihara, K. Detecting early-warning signals for sudden deterioration of complex diseases by dynamical network biomarkers. Sci. Rep. 2012, 2, 1–8. [Google Scholar] [CrossRef]
- Goodacre, R.; Vaidyanathan, S.; Dunn, W.B.; Harrigan, G.G.; Kell, D.B. Metabolomics by numbers: Acquiring and understanding global metabolite data. Trends Biotechnol. 2004, 22, 245–252. [Google Scholar] [CrossRef]
- Gu, Y.; Lu, C.; Zha, Q.; Kong, H.; Lu, X.; Lu, A.; Xu, G. Plasma metabonomics study of rheumatoid arthritis and its Chinese medicine subtypes by using liquid chromatography and gas chromatography coupled with mass spectrometry. Mol. Biosyst. 2012, 8, 1535–1543. [Google Scholar] [CrossRef]
- Harrigan, G.G.; Goodacre, R. Metabolic Profiling: Its Role in Biomarker Discovery and Gene Function Analysis; Kluwer Academic Publishers: Boston, MA, USA, 2003. [Google Scholar]
- Urbanczyk-Wochniak, E.; Luedemann, A.; Kopka, J.; Selbig, J.; Roessner-Tunali, U.; Willmitzer, L.; Fernie, A.R. Parallel analysis of transcript and metabolic profiles: A new approach in systems biology. EMBO Rep. 2003, 4, 989–993. [Google Scholar] [CrossRef]
- Loscalzo, J.; Kohane, I.; Barabasi, A. Human disease classification in the postgenomic era: A complex systems approach to human pathobiology. Mol. Syst. Biol. 2007, 3, 124. [Google Scholar] [CrossRef] [PubMed]
- Müller, G.B.; Newman, S.A. (Eds.) Origination of Organismal Form: Beyond the Gene in Developmental and Evolutionary Biology; MIT Press: Cambridge, MA, USA, 2003. [Google Scholar]
- Soto, A.M.; Sonnenschein, C. Emergentism as a default: Cancer as a problem of tissue organization. J. Biosci. 2005, 30, 103–118. [Google Scholar] [CrossRef]
- Kenny, P.A.; Bissell, M.J. Tumor reversion: Correction of malignant behavior by microenvironmental cues. Int. J. Cancer 2003, 107, 688–695. [Google Scholar] [CrossRef] [PubMed]
- Kolodkin, A.; Boogerd, F.C.; Plant, N.; Bruggeman, F.J.; Goncharuk, V.; Lunshof, J.; Moreno-Sanchez, R.; Yilmaz, N.; Bakker, B.M.; Snoep, J.L.; et al. Emergence of the silicon human and network targeting drugs. Eur. J. Pharm. Sci. 2012, 46, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Amson, R.; Karp, J.E.; Telerman, A. Lessons from tumor reversion for cancer treatment. Curr. Opin. Oncol. 2013, 25, 59–65. [Google Scholar] [CrossRef]
- Pisanu, M.E.; Noto, A.; De Vitis, C.; Masiello, M.G.; Coluccia, P.; Proietti, S.; Giovagnoli, M.R.; Ricci, A.; Giarnieri, E.; Cucina, A.; et al. Lung Cancer Stem Cell Lose Their Stemness Default State after Exposure to Microgravity. BioMed Res. Int. 2014, 2014, 470253. [Google Scholar] [CrossRef]
- Tripathi, A.; Kashyap, A.; Tripathi, G.; Yadav, J.; Bibban, R.; Aggarwal, N.; Thakur, K.; Chhokar, A.; Jadli, M.; Sah, A.K.; et al. Tumor reversion: A dream or a reality. Biomark. Res. 2021, 9, 31. [Google Scholar] [CrossRef] [PubMed]
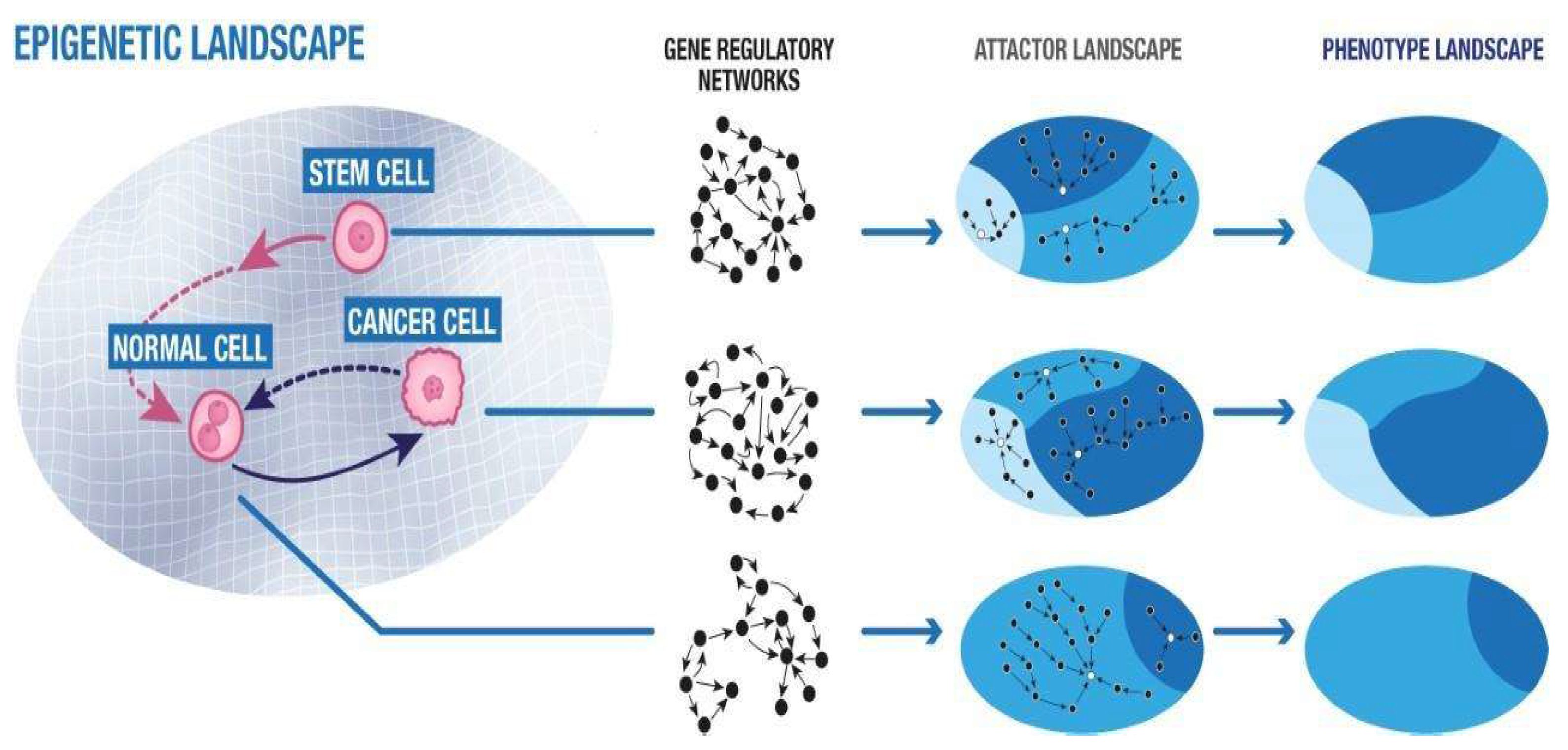
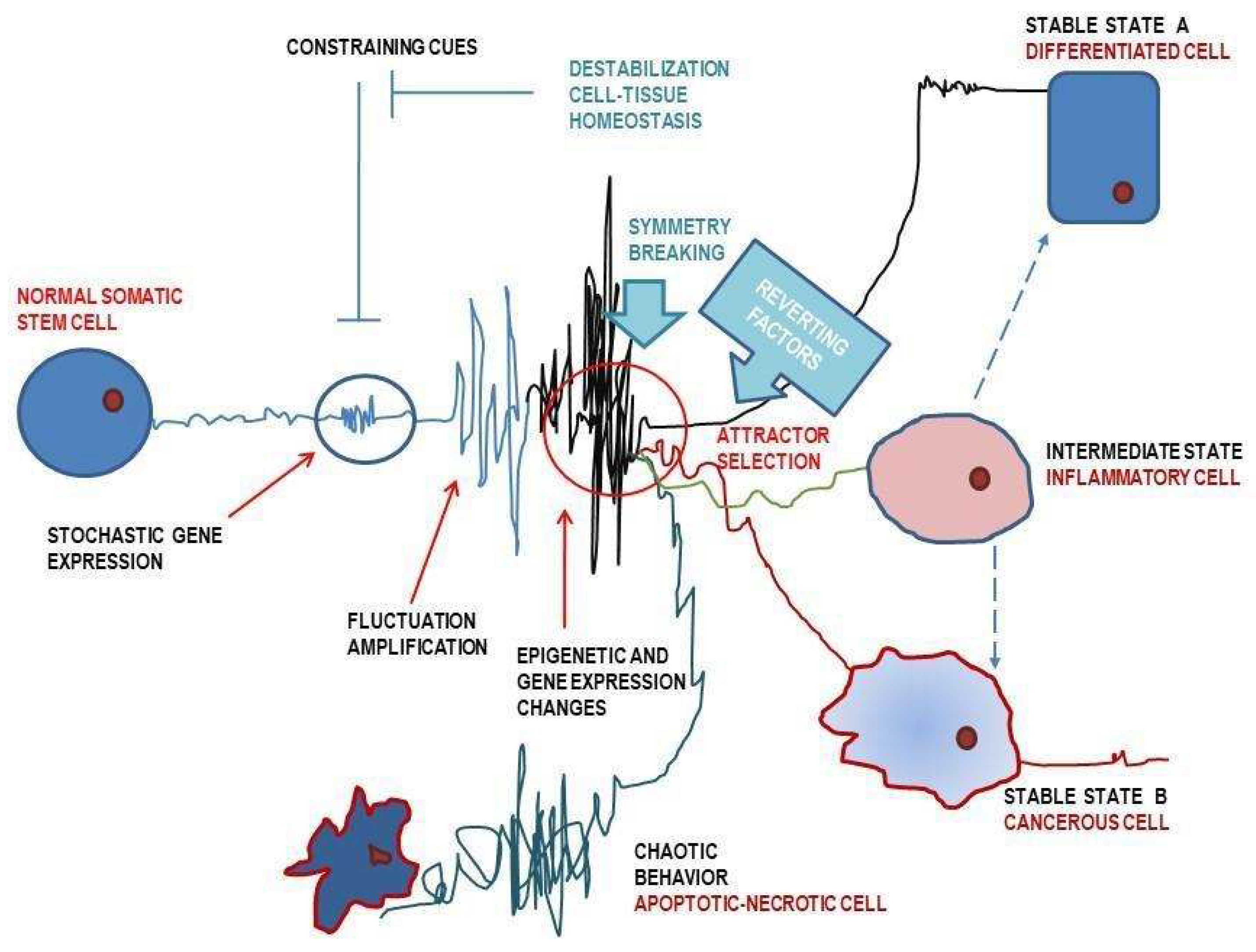
| Tumor Model | Experimental Methodology | Results | Comments | Ref. |
|---|---|---|---|---|
| Ovarian teratocarcinoma | Clinical observation | Spontaneous regression, differentiation of cancer cells into normal tissue | Askanazy, 1907 [1] | |
| Plant teratoma clonal cells | Succession of grafts on healthy tobacco plant | Disappearance of the teratoma and plant generation with seeds capable of giving life to a new plant | The results led the author to introduce the concept of pluripotentiality of cancer cells | Braun, 1959 [16] |
| Murine embryonic tumor cells | Transplantation on mice | Differentiation of cells | Hypothesis on the role of the tissue context in determining the fate of cancer cells | Pierce, 1961 [4] |
| Hamster sarcoma cells (induced by Rous sarcoma virus) | Succession of cell cultures and re-platings | Transformation of 19% of cells, which return to orienting themselves in an orderly manner, as in healthy tissues | The author uses the term “reversion” to describe results | Macpherson, 1965 [17] |
| Murine testicular teratocarcinoma cells (black mice) | Injection into murine blastocyst implanted in albino female mice | Development of healthy mice | One of the mice had black tufts, characteristic traits of the genome of teratocarcinoma cells | Brinster, 1974 [5] |
| Embryonic carcinoma cells from black mice | Blastocyst injection implanted in brown female mice | Normal fetal development; normal newborn mice feature hybrid traits between black and brown mice | Authors use the term “reversion” to describe their findings | Mintz and Illmensee, 1975 [6] |
| Lucke renal tumor cells (tumor of viral origin) | Planting on regenerative salamander limbs | Stopping of tumor growth and subsequent differentiation of cells | Failed to determine whether the differentiated cells came from cancer cells or healthy tissue | Rose and Wa llingford, 1948 [18,19] |
| Spontaneous tumors in animals | Observations on the rate of onset | Reduced occurrence of tumors in animals with high regenerative capacities | Gersch, 1951 [20] | |
| Triton-induced epithelial tumors | Monitoring the spontaneous evolution of tumors | Tendency to tumor regression in anatomical areas with high regenerative potential | Results confirmed by histological analysis | Seilern-Aspangand Kratochwil, 1962 [21] |
| Liver cancer cells (marked with dye) | Injected into liver tissue | Reduction of malignancy and, in some cases, differentiation of cancer cells | Use of the term “partial reversion” to describe the phenotypic change of cells | Coleman, 1993 [22] |
| Neuroblastoma cells | Injected into 8 ½ day old murine blastocyst | Differentiation of tumor cells | Podesta, 1984 [23] | |
| Leukemia cells | Injected into 10-day-old murine blastocyst | Correct hematopoietic maturation | Gootwine, 1982 [24] | |
| Rous sarcoma virus | Injected into chicken embryos | No tumor development | If the virus is injected into adult chickens, then sarcoma develops | Dolberg and Bissell, 1984 [7] |
| Mouse melanoma cells | Implanted into embryos in murine uterus | Cell differentiation and normal embryonic development | Differentiation occurs when cells are implanted into a 14-day embryo | Gerschenson, 1986 [9] |
| Murine breast adenocarcinoma cells | Exposure to diffusible substances of murine embryonic mesenchyme | Differentiation of tumor cells | DeCosse, 1973 [25] | |
| Primary murine lung cancer | Administration (in vivo) of homogenates of pregnant murine uteri | Suppression of tumor development | Biava, 1988 [26] | |
| Glioblastoma, melanoma, renal adenocarcinoma, breast cancer, and lymphoblastic leukemia cells | Exposure to embryonic extracts of zebrafish taken before gastrulation | Reduction of cell proliferation rates | Biava, 2001; 2002 [27,28] | |
| Human melanoma cells | Implanted in zebrafish embryos in the early stages of development | Suppression of malignant tumor phenotype and birth of healthy fish | Lee, Hendrix, 2005 [10] | |
| Leukemia cells | Retinoic acid administration | Differentiation of leukemia cells into granulocytes, subsequently digested by macrophages | Today, acute promyelocytic leukemia is treated in most cases with differential treatments based on retinoic acid | Breitman, 1980 [29] |
| Various types of human tumors | Clinical remarks | Spontaneous regressions of tumors | Several cases of spontaneous regression of tumors have been analyzed, confirmed, and classified | Rohdenburg, 1918; Everson and Cole, 1966; Challis and Stam 1990; O’Regan and Hirshberg, 1993; Papac, 1998 [30,31,32,33,34] |
| Advanced hepatocarcinoma (179 cases) | In vivo administration of extracts of zebrafish embryos | 20% of cancer regressions, of which 2.5% total stabilization progression in 16% of cases | Partial or complete disappearance of liver cancer in terminally ill patients | Livraghi, 2005 [35] |
| Melanoma cells and breast cancer cells | Exposed to embryonic stem cell factors | Reversal of the malignant phenotype and activation of apoptotic processes (nodal signal inhibition was also observed) | If cells are exposed to factors extracted from umbilical cord and bone marrow stem cells, then no phenotypic reversion is observed | Henrix, 2007; Postovit, 2008 [36,37] |
| Ovarian, prostate, and breast cancer cells | Microenvironmental exposure of human embryonic stem cells | Reversion of malignant phenotype block of cancer cells in phase G1 | These results led researchers to hypothesize an inhibitory action on the cell cycle by factors extracted from embryonic stem cells | Giuffrida, 2009 [38] |
| Melanoma cells | Microenvironmental exposure of human embryonic stem cells | Reversion of malignant phenotype | The study identified some mRNAs involved in these cellular reprogramming processes. | Costa, 2009 [39] |
| Breast cancer cells | Exposure to salamander, frog, and mouse embryonic cell extracts | Stable reversal of malignant phenotype (confirmed with subsequent implantation of reprogrammed cells in immunosuppressed mice) | Re-expression of some cancer suppressor genes has been observed; mouse embryonic cells did not give results | Allegrucci, 2011 [40] |
| Breast cancer cells | Exposure to embryonic extracts of zebrafish taken at different times of embryogenesis | Reduction of invasiveness, migration, and proliferation parameters; action on cytoskeleton and TCTP downregulation | An activation method of reversion was identified, implying the down-regulation of TCTP by exposing the cells to a specific embryonic microenvironment composition that corresponds to a specific phase of embryogenesis | Proietti and Bizzarri, 2019 [12] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pensotti, A.; Bertolaso, M.; Bizzarri, M. Is Cancer Reversible? Rethinking Carcinogenesis Models—A New Epistemological Tool. Biomolecules 2023, 13, 733. https://doi.org/10.3390/biom13050733
Pensotti A, Bertolaso M, Bizzarri M. Is Cancer Reversible? Rethinking Carcinogenesis Models—A New Epistemological Tool. Biomolecules. 2023; 13(5):733. https://doi.org/10.3390/biom13050733
Chicago/Turabian StylePensotti, Andrea, Marta Bertolaso, and Mariano Bizzarri. 2023. "Is Cancer Reversible? Rethinking Carcinogenesis Models—A New Epistemological Tool" Biomolecules 13, no. 5: 733. https://doi.org/10.3390/biom13050733
APA StylePensotti, A., Bertolaso, M., & Bizzarri, M. (2023). Is Cancer Reversible? Rethinking Carcinogenesis Models—A New Epistemological Tool. Biomolecules, 13(5), 733. https://doi.org/10.3390/biom13050733







