Effect of Hydrogen Oxide-Induced Oxidative Stress on Bone Formation in the Early Embryonic Development Stage of Chicken
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
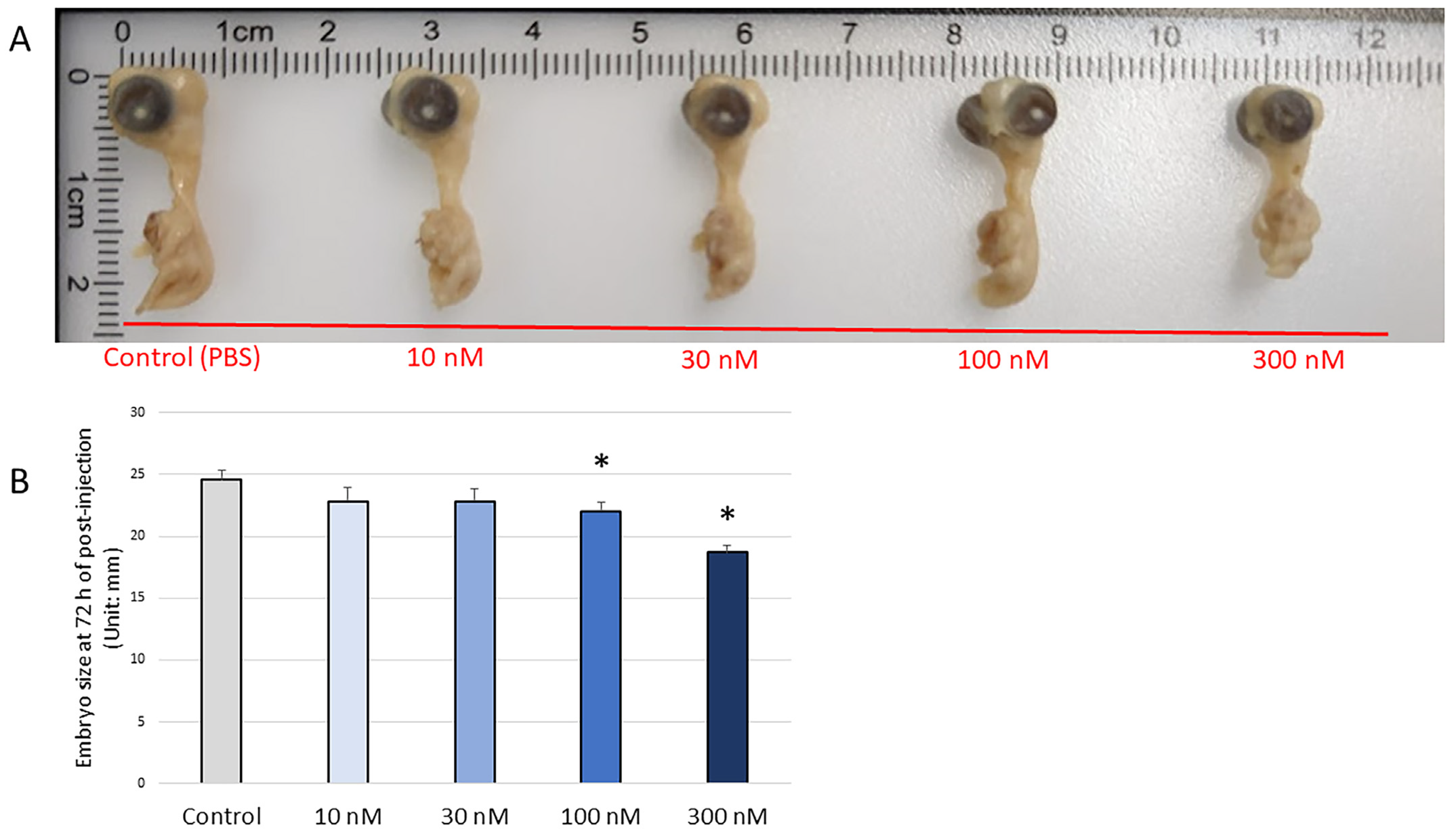
2.2. H2O2 Microinjection
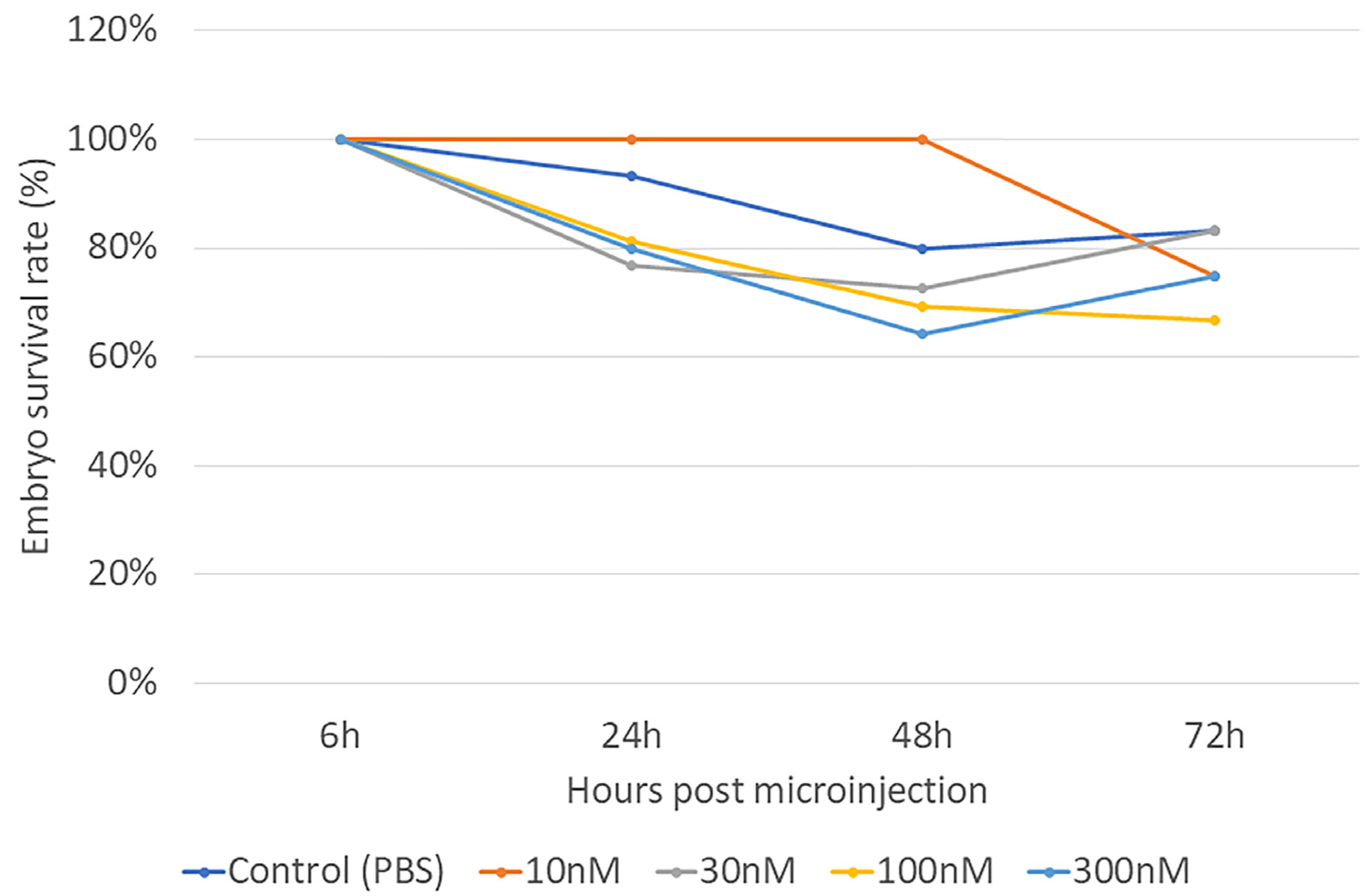
2.3. Embryonic Survival Rate and Embryo Length Measurements
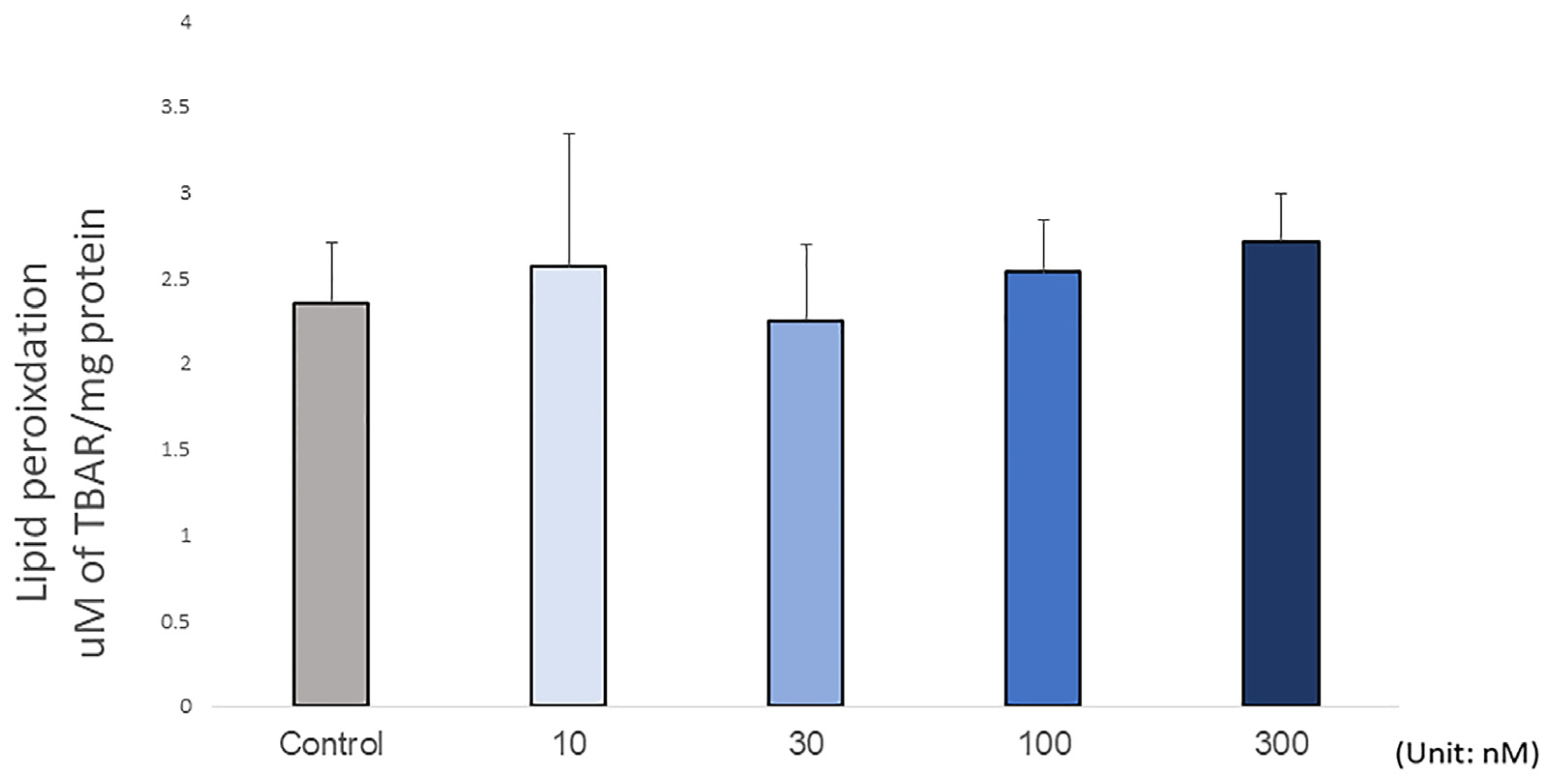
2.4. Lipid Peroxidation and Antioxidant Status Assay
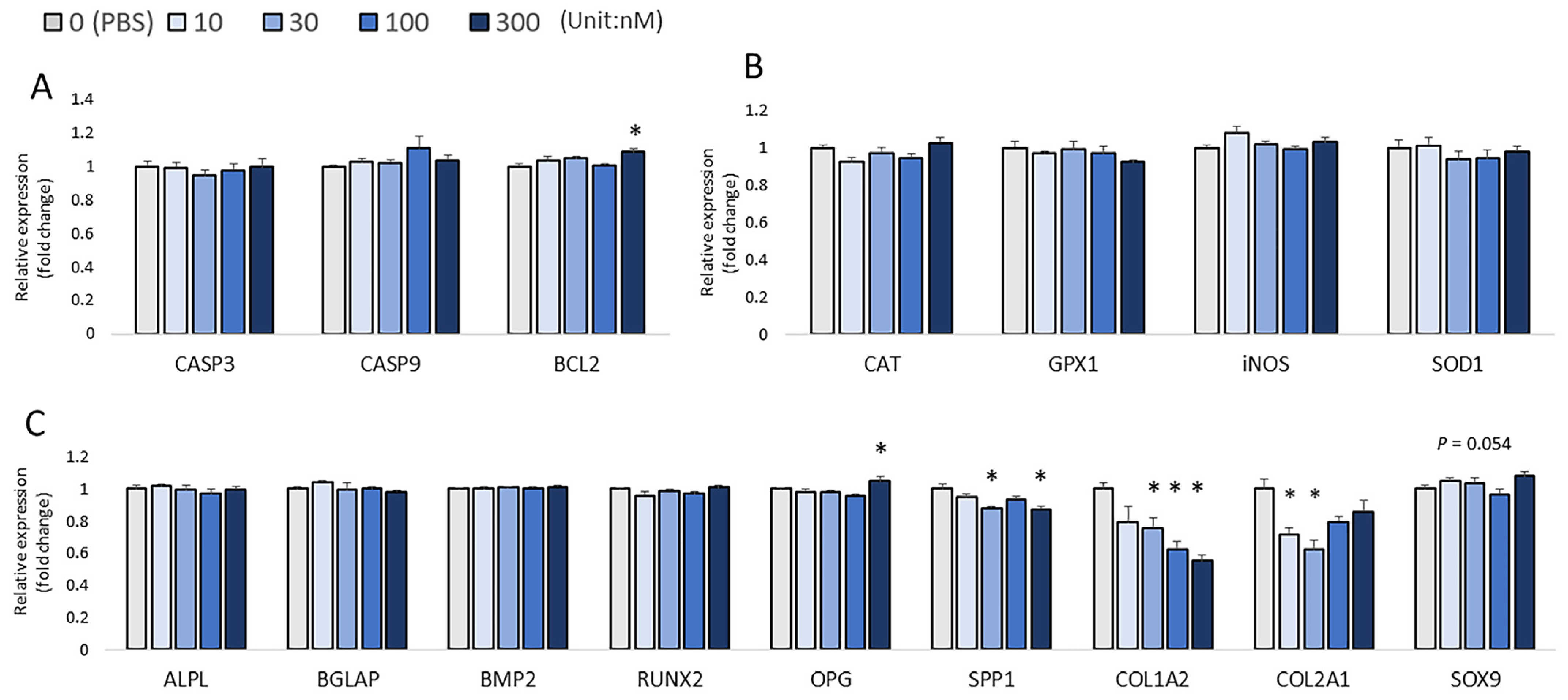
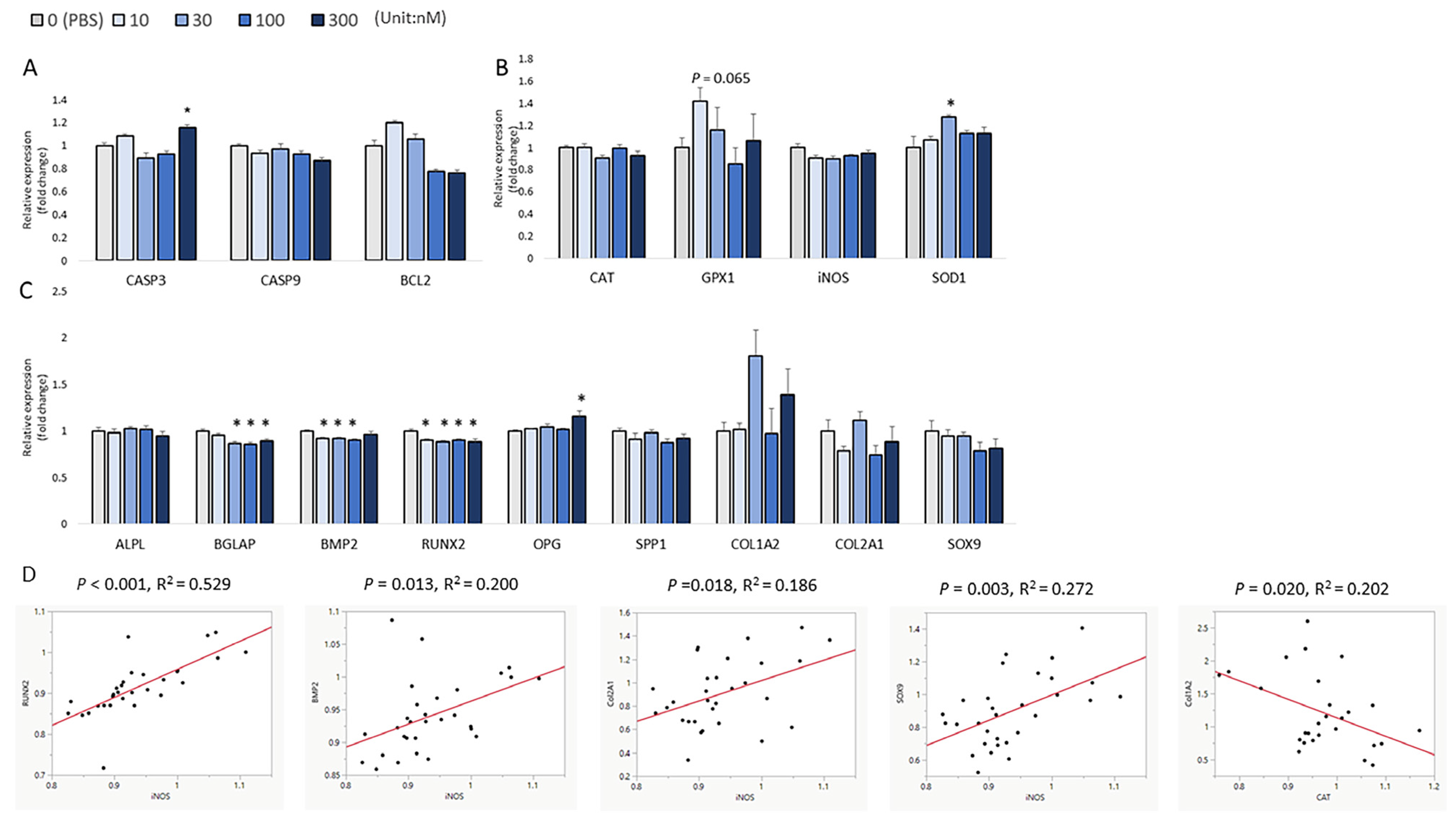
2.5. RNA Isolation, cDNA Synthesis, and Real-Time Polymerase Chain Reaction (qRT-PCR) Analysis
2.6. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mishra, B.; Jha, R. Oxidative Stress in the Poultry Gut: Potential Challenges and Interventions. Front. Vet. Sci. 2019, 6, 60. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.T.; Lin, W.C.; Lee, T.T. Potential crosstalk of oxidative stress and immune response in poultry through phytochemicals—A review. Asian-Australas J. Anim. Sci. 2019, 32, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Covarrubias, L.; Hernandez-Garcia, D.; Schnabel, D.; Salas-Vidal, E.; Castro-Obregon, S. Function of reactive oxygen species during animal development: Passive or active? Dev. Biol. 2008, 320, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Dennery, P.A. Oxidative stress in development: Nature or nurture? Free Radic. Biol. Med. 2010, 49, 1147–1151. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.P.; Al-Hasan, Y. Impact of oxidative stress in fetal programming. J. Pregnancy 2012, 2012, 582748. [Google Scholar] [CrossRef]
- Dennery, P.A. Role of redox in fetal development and neonatal diseases. Antioxid. Redox Signal. 2004, 6, 147–153. [Google Scholar] [CrossRef]
- Prater, M.R.; Laudermilch, C.L.; Liang, C.; Holladay, S.D. Placental oxidative stress alters expression of murine osteogenic genes and impairs fetal skeletal formation. Placenta 2008, 29, 802–808. [Google Scholar] [CrossRef]
- Domazetovic, V.; Marcucci, G.; Iantomasi, T.; Brandi, M.L.; Vincenzini, M.T. Oxidative stress in bone remodeling: Role of antioxidants. Clin. Cases Miner. Bone Metab. 2017, 14, 209–216. [Google Scholar] [CrossRef]
- Surai, P.F. Natural Antioxidants in Avian Nutrition and Reproduction; Nottingham University Press: Nottingham, UK, 2002. [Google Scholar]
- Haussmann, M.F.; Longenecker, A.S.; Marchetto, N.M.; Juliano, S.A.; Bowden, R.M. Embryonic exposure to corticosterone modifies the juvenile stress response, oxidative stress and telomere length. Proc. Biol. Sci. 2012, 279, 1447–1456. [Google Scholar] [CrossRef]
- Dennery, P.A. Effects of oxidative stress on embryonic development. Birth Defects Res. C Embryo Today Rev. 2007, 81, 155–162. [Google Scholar] [CrossRef]
- Ebeid, T.A.; Twfeek, F.A.; Assar, M.H.; Bealish, A.M.; Abd El-Karim, R.E.; Ragab, M. Influence of pre-storage incubation on hatchability traits, thyroid hormones, antioxidative status and immunity of newly hatched chicks at two chicken breeder flock ages. Animal 2017, 11, 1966–1974. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, K.Y.; Bai, S.P.; Wang, J.P.; Zeng, Q.F.; Peng, H.W.; Xuan, Y.; Su, Z.W.; Ding, X.M. The impacts of egg storage time and maternal dietary vitamin E on the growth performance and antioxidant capacity of progeny chicks. Poult. Sci. 2021, 100, 101142. [Google Scholar] [CrossRef] [PubMed]
- Gou, Z.; Fan, Q.; Li, L.; Wang, Y.; Lin, X.; Cui, X.; Ye, J.; Ding, F.; Cheng, Z.; Abouelezz, K.; et al. High dietary copper induces oxidative stress and leads to decreased egg quality and reproductive performance of Chinese Yellow broiler breeder hens. Poult. Sci. 2021, 100, 100779. [Google Scholar] [CrossRef]
- Reijrink, I.A.; Meijerhof, R.; Kemp, B.; Graat, E.A.; van den Brand, H. Influence of prestorage incubation on embryonic development, hatchability, and chick quality. Poult. Sci. 2009, 88, 2649–2660. [Google Scholar] [CrossRef] [PubMed]
- Tona, K.; Bamelis, F.; De Ketelaere, B.; Bruggeman, V.; Moraes, V.M.; Buyse, J.; Onagbesan, O.; Decuypere, E. Effects of egg storage time on spread of hatch, chick quality, and chick juvenile growth. Poult. Sci. 2003, 82, 736–741. [Google Scholar] [CrossRef] [PubMed]
- Saleh, K.M.M.; Tarkhan, A.H.; Al-Zghoul, M.B. Embryonic Thermal Manipulation Affects the Antioxidant Response to Post-Hatch Thermal Exposure in Broiler Chickens. Animals 2020, 10, 126. [Google Scholar] [CrossRef]
- Hartcher, K.M.; Lum, H.K. Genetic selection of broilers and welfare consequences: A review. World’s Poult. Sci. J. 2019, 76, 154–167. [Google Scholar] [CrossRef]
- Rath, N.C.; Durairaj, V. Avian bone physiology and poultry bone disorders. In Sturkie’s Avian Physiology; Elsevier: Amsterdam, The Netherlands, 2022; pp. 529–543. [Google Scholar]
- Heidinger, B.J.; Blount, J.D.; Boner, W.; Griffiths, K.; Metcalfe, N.B.; Monaghan, P. Telomere length in early life predicts lifespan. Proc. Natl. Acad. Sci. USA 2012, 109, 1743–1748. [Google Scholar] [CrossRef]
- Paradowska, M.; Dunislawska, A.; Siwek, M.; Slawinska, A. Avian Cell Culture Models to Study Immunomodulatory Properties of Bioactive Products. Animals 2022, 12, 670. [Google Scholar] [CrossRef]
- Korn, M.J.; Cramer, K.S. Windowing chicken eggs for developmental studies. J. Vis. Exp. 2007, 8, 306. [Google Scholar] [CrossRef]
- Korhonen, A.; Hemminki, K.; Vainio, H. Embryotoxic effects of eight organic peroxides and hydrogen peroxide on three-day chicken embryos. Environ. Res. 1984, 33, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, J.T.; Kengar, S. Influence of hydrogen peroxide induced oxidative stress on survival rate of early chick embryo development. Int. J. Bioassays 2016, 5, 4603–4606. [Google Scholar] [CrossRef]
- Newton, P.T.; Li, L.; Zhou, B.; Schweingruber, C.; Hovorakova, M.; Xie, M.; Sun, X.; Sandhow, L.; Artemov, A.V.; Ivashkin, E.; et al. A radical switch in clonality reveals a stem cell niche in the epiphyseal growth plate. Nature 2019, 567, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Ben-Yair, R.; Kalcheim, C. Single cell transfection in chick embryos. J. Vis. Exp. 2010, 43, e2133. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; White, D.L.; Marshall, B.; Kim, W.K. Role of 25-Hydroxyvitamin D3 and 1,25-Dihydroxyvitamin D3 in Chicken Embryo Osteogenesis, Adipogenesis, Myogenesis, and Vitamin D3 Metabolism. Front. Physiol. 2021, 12, 637629. [Google Scholar] [CrossRef]
- Thiese, M.S.; Ronna, B.; Ott, U. P value interpretations and considerations. J. Thorac. Dis. 2016, 8, E928–E931. [Google Scholar] [CrossRef] [PubMed]
- Serdar, C.C.; Cihan, M.; Yucel, D.; Serdar, M.A. Sample size, power and effect size revisited: Simplified and practical approaches in pre-clinical, clinical and laboratory studies. Biochem. Med. 2021, 31, 010502. [Google Scholar] [CrossRef]
- Surai, P.F.; Fisinin, V.I.; Karadas, F. Antioxidant systems in chick embryo development. Part 1. Vitamin E, carotenoids and selenium. Anim. Nutr. 2016, 2, 1–11. [Google Scholar] [CrossRef]
- Jassim, E.W.; Grossman, M.; Koops, W.J.; Luykx, R.A. Multiphasic analysis of embryonic mortality in chickens. Poult. Sci. 1996, 75, 464–471. [Google Scholar] [CrossRef]
- Tzahor, E.; Kempf, H.; Mootoosamy, R.C.; Poon, A.C.; Abzhanov, A.; Tabin, C.J.; Dietrich, S.; Lassar, A.B. Antagonists of Wnt and BMP signaling promote the formation of vertebrate head muscle. Genes Dev. 2003, 17, 3087–3099. [Google Scholar] [CrossRef]
- Sato, Y. Dorsal aorta formation: Separate origins, lateral-to-medial migration, and remodeling. Dev. Growth Differ. 2013, 55, 113–129. [Google Scholar] [CrossRef] [PubMed]
- Deeming, D.C. The Role of Egg Turning during Incubation. Avian Biol. Res. 2009, 2, 67–71. [Google Scholar] [CrossRef]
- Mousseau, T. The adaptive significance of maternal effects. Trends Ecol. Evol. 1998, 13, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Deeming, D.C.; Pike, T.W. Embryonic growth and antioxidant provision in avian eggs. Biol. Lett. 2013, 9, 20130757. [Google Scholar] [CrossRef] [PubMed]
- Parolini, M.; Possenti, C.D.; Karadas, F.; Colombo, G.; Romano, M.; Caprioli, M.; Dalle-Donne, I.; Rubolini, D.; Milzani, A.; Saino, N. Yolk vitamin E positively affects prenatal growth but not oxidative status in yellow-legged gull embryos. Curr. Zool. 2018, 64, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Pomari, E.; Stefanon, B.; Colitti, M. Effect of plant extracts on H2O2-induced inflammatory gene expression in macrophages. J. Inflamm. Res. 2014, 7, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Dell’Orco, M.; Milani, P.; Arrigoni, L.; Pansarasa, O.; Sardone, V.; Maffioli, E.; Polveraccio, F.; Bordoni, M.; Diamanti, L.; Ceroni, M.; et al. Hydrogen peroxide-mediated induction of SOD1 gene transcription is independent from Nrf2 in a cellular model of neurodegeneration. Biochim. Biophys. Acta 2016, 1859, 315–323. [Google Scholar] [CrossRef]
- Jing, Y.; Jing, J.; Ye, L.; Liu, X.; Harris, S.E.; Hinton, R.J.; Feng, J.Q. Chondrogenesis and osteogenesis are one continuous developmental and lineage defined biological process. Sci. Rep. 2017, 7, 10020. [Google Scholar] [CrossRef]
- Bruderer, M.; Richards, R.G.; Alini, M.; Stoddart, M.J. Role and regulation of RUNX2 in osteogenesis. Eur. Cell Mater. 2014, 28, 269–286. [Google Scholar] [CrossRef]
- Pines, M.; Reshef, R. Poultry bone development and bone disorders. In Sturkie’s Avian Physiology; Elsevier: Amsterdam, The Netherlands, 2015; pp. 367–377. [Google Scholar]
- Gaur, T.; Lengner, C.J.; Hovhannisyan, H.; Bhat, R.A.; Bodine, P.V.; Komm, B.S.; Javed, A.; van Wijnen, A.J.; Stein, J.L.; Stein, G.S.; et al. Canonical WNT signaling promotes osteogenesis by directly stimulating Runx2 gene expression. J. Biol. Chem. 2005, 280, 33132–33140. [Google Scholar] [CrossRef]
- Montero, J.A.; Lorda-Diez, C.I.; Francisco-Morcillo, J.; Chimal-Monroy, J.; Garcia-Porrero, J.A.; Hurle, J.M. Sox9 Expression in Amniotes: Species-Specific Differences in the Formation of Digits. Front. Cell Dev. Biol. 2017, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Javed, A.; Chen, H.; Ghori, F.Y. Genetic and transcriptional control of bone formation. Oral Maxillofac. Surg. Clin. N. Am. 2010, 22, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Garnero, P. The contribution of collagen crosslinks to bone strength. Bonekey Rep. 2012, 1, 182. [Google Scholar] [CrossRef]
- Karna, E.; Szoka, L.; Huynh, T.Y.L.; Palka, J.A. Proline-dependent regulation of collagen metabolism. Cell Mol. Life Sci. 2020, 77, 1911–1918. [Google Scholar] [CrossRef]
- Merlino, G.; McKeon, C.; de Crombrugghe, B.; Pastan, I. Regulation of the expression of genes encoding types I, II, and III collagen during chick embryonic development. J. Biol. Chem. 1983, 258, 10041–10048. [Google Scholar] [CrossRef] [PubMed]
- Siwik, D.A.; Pagano, P.J.; Colucci, W.S. Oxidative stress regulates collagen synthesis and matrix metalloproteinase activity in cardiac fibroblasts. Am. J. Physiol. Cell Physiol. 2001, 280, C53–C60. [Google Scholar] [CrossRef] [PubMed]
- Laurent, G.J. Dynamic state of collagen: Pathways of collagen degradation in vivo and their possible role in regulation of collagen mass. Am. J. Physiol. 1987, 252, C1–C9. [Google Scholar] [CrossRef] [PubMed]
- Wardale, R.J.; Duance, V.C. Collagen expression in chicken tibial dyschondroplasia. J. Cell Sci. 1996, 109 Pt 5, 1119–1131. [Google Scholar] [CrossRef]
- Yoshino, T.; Murai, H.; Saito, D. Hedgehog-BMP signalling establishes dorsoventral patterning in lateral plate mesoderm to trigger gonadogenesis in chicken embryos. Nat. Commun. 2016, 7, 12561. [Google Scholar] [CrossRef]
- Bandyopadhyay, A.; Tsuji, K.; Cox, K.; Harfe, B.D.; Rosen, V.; Tabin, C.J. Genetic analysis of the roles of BMP2, BMP4, and BMP7 in limb patterning and skeletogenesis. PLoS Genet. 2006, 2, e216. [Google Scholar] [CrossRef]
- Komori, T. Regulation of Proliferation, Differentiation and Functions of Osteoblasts by Runx2. Int. J. Mol. Sci. 2019, 20, 1694. [Google Scholar] [CrossRef] [PubMed]
- Barreto, C.; Wilsman, N.J. Hypertrophic chondrocyte volume and growth rates in avian growth plates. Res. Vet. Sci. 1994, 56, 53–61. [Google Scholar] [CrossRef]
- Li, J.; Dong, S. The Signaling Pathways Involved in Chondrocyte Differentiation and Hypertrophic Differentiation. Stem. Cells Int. 2016, 2016, 2470351. [Google Scholar] [CrossRef] [PubMed]
- An, S.Y.; Lee, H.J.; Lee, S.C.; Heo, J.S. Supplement of nitric oxide through calcium carbonate-based nanoparticles contributes osteogenic differentiation of mouse embryonic stem cells. Tissue Cell 2020, 66, 101390. [Google Scholar] [CrossRef]
- Amin, A.R.; Abramson, S.B. The role of nitric oxide in articular cartilage breakdown in osteoarthritis. Curr. Opin. Rheumatol. 1998, 10, 263–268. [Google Scholar] [CrossRef]
- Armour, K.J.; Armour, K.E.; Van’t Hof, R.J.; Reid, D.M.; Wei, X.-Q.; Liew, F.Y.; Ralston, S.H. Activation of the inducible nitric oxide synthase pathway contributes to inflammation-induced osteoporosis by suppressing bone formation and causing osteoblast apoptosis. Arthritis Rheum. 2001, 44, 2790–2796. [Google Scholar] [CrossRef]
- Ocarino, N.M.; Boeloni, J.N.; Goes, A.M.; Silva, J.F.; Marubayashi, U.; Serakides, R. Osteogenic differentiation of mesenchymal stem cells from osteopenic rats subjected to physical activity with and without nitric oxide synthase inhibition. Nitric Oxide 2008, 19, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Corino, C.; Rossi, R. Antioxidants in Animal Nutrition. Antioxidants 2021, 10, 1877. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lazarenko, O.P.; Blackburn, M.L.; Badger, T.M.; Ronis, M.J.; Chen, J.R. Blueberry consumption prevents loss of collagen in bone matrix and inhibits senescence pathways in osteoblastic cells. Age 2013, 35, 807–820. [Google Scholar] [CrossRef]
- Surai, P.F. Effect of selenium and vitamin E content of the maternal diet on the antioxidant system of the yolk and the developing chick. Br. Poult. Sci. 2000, 41, 235–243. [Google Scholar] [CrossRef]
- Li, K.; Jiang, L.; Wang, J.; Xia, L.; Zhao, R.; Cai, C.; Wang, P.; Zhan, X.; Wang, Y. Maternal dietary supplementation with different sources of selenium on antioxidant status and mortality of chicken embryo in a model of diquat-induced acute oxidative stress. Anim. Feed Sci. Technol. 2020, 261, 114369. [Google Scholar] [CrossRef]



| Gene 1 | Primer Sequence (5′-3′) | Product Length (bp) | Annealing Temperature (°C) | Accession |
|---|---|---|---|---|
| GAPDH | F-GCTAAGGCTGTGGGGAAAGT R-TCAGCAGCAGCCTTCACTAC | 161 | 55 | NM_204305.1 |
| HMBS | F-GGCTGGGAGAATCGCATAGG R-TCCTGCAGGGCAGATACCAT | 131 | 59 | XM_004947916.3 |
| ACTB | F-CAACACAGTGCTGTCTGGTGGTA R-ATCGTACTCCTGCTTGCTGATCC | 205 | 61 | NM_205518.1 |
| ALPL | F-CGACCACTCACACGTCTTCA R-CGATCTTATAGCCAGGGCCG | 140 | 60 | NM_205360.1 |
| RUNX2 | F-ACTTTGACAATAACTGTCCT R-GACCCCTACTCTCATACTGG | 192 | 60 | XM_015285081.2 |
| BGLAP | F-GGATGCTCGCAGTGCTAAAG R-CTCACACACCTCTCGTTGGG | 142 | 57 | NM_205387.3 |
| SPP1 | F-GCCCAACATCAGAGCGTAGA R-ACGGGTGACCTCGTTGTTTT | 204 | 57 | NM_204535.4 |
| BMP2 | F-TCAGCTCAGGCCGTTGTTAG R-GTCATTCCACCCCACGTCAT | 163 | 57 | XM_025148488.1 |
| OPG | F-ACGCTTGTGCTCTTGGACAT R-CAGCGTAGTACTGGTCTGGG | 193 | 60 | NM_001033641.1 |
| COL1A2 | F-CTGGTGAAAGCGGTGCTGTT R-CACCAGTGTCACCTCTCAGAC | 222 | 60 | NM_001079714.2 |
| COL2A1 | F-GGACCAGCAAGACGAAAGAC R-TGTAGGCGATGCTGTTCTTG | 189 | 59 | NM_204426.2 |
| SOX9 | F-AGGAAGCTGGCTGACCAGTA R-CGTTCTTCACCGACTTCCTC | 193 | 61 | XM_046929245.1 |
| SOD1 | F-ATTACCGGCTTGTCTGATGG R-CCTCCCTTTGCAGTCACATT | 173 | 58 | NM_205064.1 |
| CAT | F-ACTGCAAGGCGAAAGTGTTT R-GGCTATGGATGAAGGATGGA | 222 | 60 | NM_001031215.1 |
| iNOS | F-CCTGTACTGAAGGTGGCTATTGG R-AGGCCTGTGAGAGTGTGCAA | 66 | 58 | NM_204961.2 |
| GPX1 | F-AACCAATTCGGGCACCAG R-CCGTTCACCTCGCACTTCTC | 122 | 60 | NM_001277853.2 |
| CASP3 | F-TGGTATTGAAGCAGACAGTGGA R-GGAGTAGTAGCCTGGAGCAGTAGA | 103 | 60 | XM_015276122.2 |
| CASP9 | F-ATTCCTTTCCAGGCTCCATC R-CACTCACCTTGTCCCTCCAG | 130 | 60 | XM_046931415.1 |
| BCL2 | F-GAGTTCGGCGGCGTGATGTG R-TTCAGGTACTCGGTCATCCAGGTG | 92 | 63 | XM_046910476.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tompkins, Y.; Liu, G.; Marshall, B.; Sharma, M.K.; Kim, W.K. Effect of Hydrogen Oxide-Induced Oxidative Stress on Bone Formation in the Early Embryonic Development Stage of Chicken. Biomolecules 2023, 13, 154. https://doi.org/10.3390/biom13010154
Tompkins Y, Liu G, Marshall B, Sharma MK, Kim WK. Effect of Hydrogen Oxide-Induced Oxidative Stress on Bone Formation in the Early Embryonic Development Stage of Chicken. Biomolecules. 2023; 13(1):154. https://doi.org/10.3390/biom13010154
Chicago/Turabian StyleTompkins, Yuguo, Guanchen Liu, Brett Marshall, Milan Kumar Sharma, and Woo Kyun Kim. 2023. "Effect of Hydrogen Oxide-Induced Oxidative Stress on Bone Formation in the Early Embryonic Development Stage of Chicken" Biomolecules 13, no. 1: 154. https://doi.org/10.3390/biom13010154
APA StyleTompkins, Y., Liu, G., Marshall, B., Sharma, M. K., & Kim, W. K. (2023). Effect of Hydrogen Oxide-Induced Oxidative Stress on Bone Formation in the Early Embryonic Development Stage of Chicken. Biomolecules, 13(1), 154. https://doi.org/10.3390/biom13010154






