UCS Chaperone Folding of the Myosin Head: A Function That Evolved before Animals and Fungi Diverged from a Common Ancestor More than a Billion Years Ago
Abstract
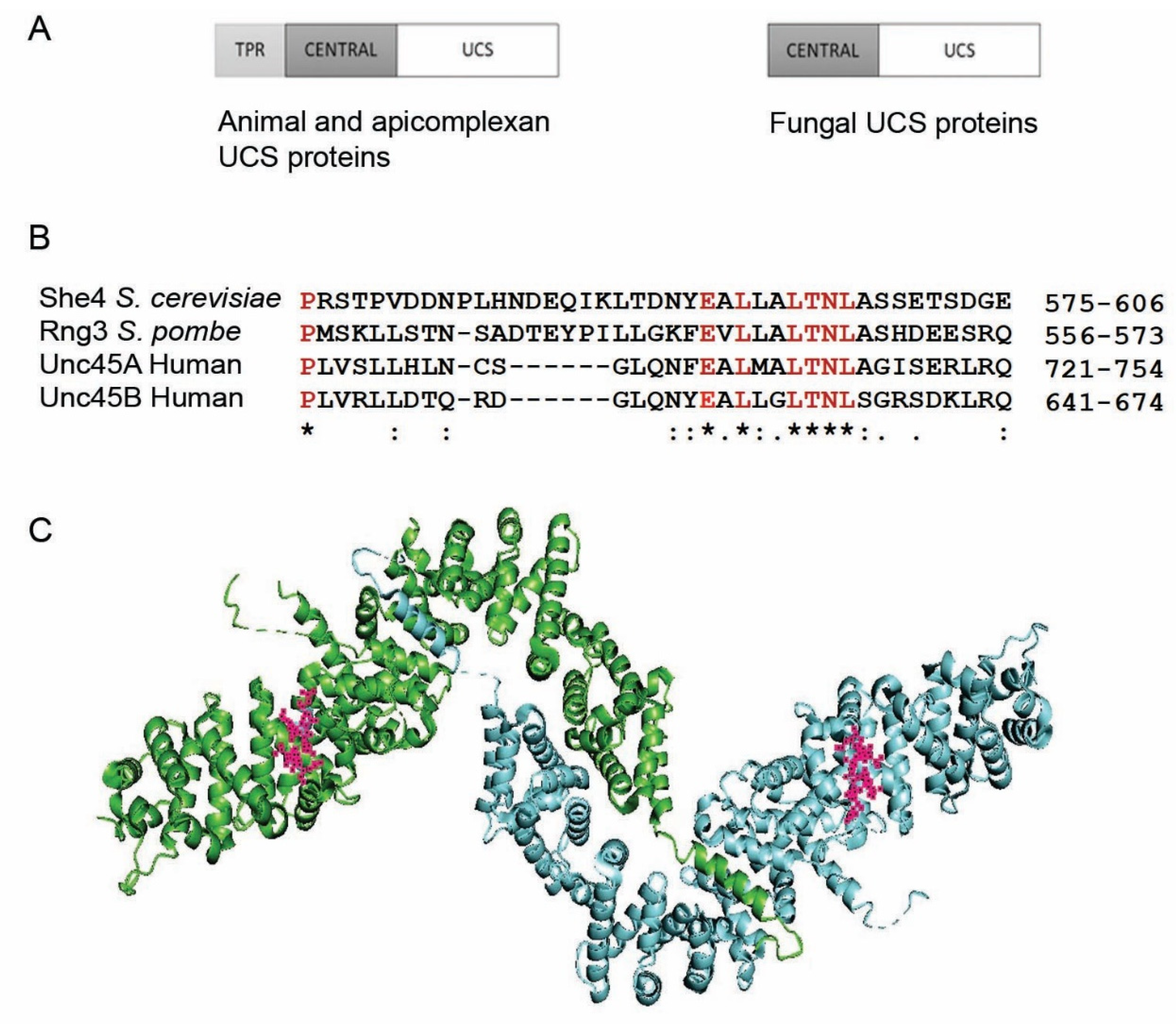
1. The UCS Protein Function
2. Vertebrate Unc45A (UNC45-GC) and Unc45B (UNC45-SM)
3. The UCS Function Evolved before Animals and Fungi Diverged from a Common Ancestor
4. Genetic Studies on the UCS Proteins of Ascomycete Fungi; UCS Function in the Absence of the TPR
5. Myosins in Fungal Growth
6. Hsp90 in UCS Protein Function
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barral, J.M.; Bauer, C.C.; Ortiz, I.; Epstein, H.F. Unc-45 mutations in Caenorhabditis elegans implicate a CRO1/She4p-like domain in myosin assembly. J. Cell Biol. 1998, 143, 1215–1225. [Google Scholar] [CrossRef] [PubMed]
- Ao, W.; Pilgrim, D. Caenorhabditis elegans UNC-45 is a component of muscle thick filaments and colocalizes with myosin heavy chain B, but not myosin heavy chain A. J. Cell Biol. 2000, 148, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Kachur, T.; Ao, W.; Berger, J.; Pilgrim, D. Maternal UNC-45 is involved in cytokinesis and colocalizes with non-muscle myosin in the early Caenorhabditis elegans embryo. J. Cell Sci. 2004, 117, 5313–5321. [Google Scholar] [CrossRef] [PubMed]
- Barral, J.M.; Hutagalung, A.H.; Brinker, A.; Hartl, F.U.; Epstein, H.F. Role of the myosin assembly protein UNC-45 as a molecular chaperone for myosin. Science 2002, 295, 669–671. [Google Scholar] [CrossRef] [PubMed]
- Landsverk, M.L.; Li, S.; Hutagalung, A.H.; Najafov, A.; Hoppe, T.; Barral, J.M.; Epstein, H.F. The UNC-45 chaperone mediates sarcomere assembly through myosin degradation in Caenorhabditis elegans. J. Cell Biol. 2007, 177, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Gazda, L.; Pokrzywa, W.; Hellerschmied, D.; Lowe, T.; Forne, I.; Mueller-Planitz, F.; Hoppe, T.; Clausen, T. The myosin chaperone UNC-45 is organized in tandem modules to support myofilament formation in C. elegans. Cell 2013, 152, 183–195. [Google Scholar] [CrossRef]
- Lee, C.F.; Melkani, G.C.; Yu, Q.; Suggs, J.A.; Kronert, W.A.; Suzuki, Y.; Hipolito, L.; Price, M.G.; Epstein, H.F.; Bernstein, S.I. Drosophila UNC-45 accumulates in embryonic blastoderm and in muscles, and is essential for muscle myosin stability. J. Cell Sci. 2011, 124, 699–705. [Google Scholar] [CrossRef]
- Melkani, G.C.; Bodmer, R.; Ocorr, K.; Bernstein, S.I. The UNC-45 chaperone is critical for establishing myosin-based myofibrillar organization and cardiac contractility in the Drosophila heart model. PLoS ONE 2011, 6, e22579. [Google Scholar] [CrossRef]
- Lee, C.F.; Melkani, G.C.; Bernstein, S.I. The UNC-45 myosin chaperone: From worms to flies to vertebrates. Int. Rev. Cell Mol. Biol. 2014, 313, 103–144. [Google Scholar]
- Karunendiran, A.; Nguyen, C.T.; Barzda, V.; Stewart, B.A. Disruption of Drosophila larval muscle structure and function by UNC45 knockdown. BMC Mol. Cell Biol. 2021, 22, 38. [Google Scholar] [CrossRef]
- Lee, C.F.; Hauenstein, A.V.; Gasper, W.C.; Sankaran, B.; Bernstein, S.I.; Huxford, T. Crystal Structure of Drosophila Unc-45, a Putative Myosin Chaperone. Biophys. J. 2010, 98, 34. [Google Scholar] [CrossRef][Green Version]
- Ni, W.; Hutagalung, A.H.; Li, S.; Epstein, H.F. The myosin-binding UCS domain but not the Hsp90-binding TPR domain of the UNC-45 chaperone is essential for function in Caenorhabditis elegans. J. Cell Sci. 2011, 124, 3164–3173. [Google Scholar] [CrossRef][Green Version]
- Bernick, E.P.; Zhang, P.J.; Du, S. Knockdown and overexpression of Unc-45b result in defective myofibril organization in skeletal muscles of zebrafish embryos. BMC Cell Biol. 2010, 11, 70. [Google Scholar] [CrossRef] [PubMed]
- Hellerschmied, D.; Lehner, A.; Franicevic, N.; Arnese, R.; Johnson, C.; Vogel, A.; Meinhart, A.; Kurzbauer, R.; Deszcz, L.; Gazda, L.; et al. Molecular features of the UNC-45 chaperone critical for binding and folding muscle myosin. Nat. Commun. 2019, 10, 4781. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, P.; Bujalowski, P.J.; Epstein, H.F.; Boehning, D.F.; Barral, J.M.; Oberhauser, A.F. Chaperone-mediated reversible inhibition of the sarcomeric myosin power stroke. FEBS Lett. 2014, 588, 3977–3981. [Google Scholar] [CrossRef]
- Bujalowski, P.J.; Nicholls, P.; Garza, E.; Oberhauser, A.F. The central domain of UNC-45 chaperone inhibits the myosin power stroke. FEBS Open Biol. 2018, 8, 41–48. [Google Scholar] [CrossRef]
- Shi, H.; Blobel, G. UNC-45/CRO1/She4p (UCS) protein forms elongated dimer and joins two myosin heads near their actin binding region. Proc. Natl. Acad. Sci. USA 2010, 107, 21382–21387. [Google Scholar] [CrossRef]
- Moncrief, T.; Matheny, C.J.; Gaziova, I.; Miller, J.M.; Qadota, H.; Benian, G.M.; Oberhauser, A.F. Mutations in conserved residues of the myosin chaperone UNC-45 result in both reduced stability and chaperoning activity. Protein Sci. 2021, 30, 2221–2232. [Google Scholar] [CrossRef]
- Hutagalung, A.H.; Landsverk, M.L.; Price, M.G.; Epstein, H.F. The UCS family of myosin chaperones. J. Cell Sci. 2002, 115, 3983–3990. [Google Scholar] [CrossRef]
- Price, M.G.; Landsverk, M.L.; Barral, J.M.; Epstein, H.F. Two mammalian UNC-45 isoforms are related to distinct cytoskeletal and muscle-specific functions. J. Cell Sci. 2002, 115, 4013–4023. [Google Scholar] [CrossRef]
- Esteve, C.; Francescatto, L.; Tan, P.L.; Bourchany, A.; de Leusse, C.; Marinier, E.; Blanchard, A.; Bourgeois, P.; Brochier-Armanet, C.; Bruel, A.L.; et al. Loss-of-Function Mutations in UNC45A Cause a Syndrome Associating Cholestasis, Diarrhea, Impaired Hearing, and Bone Fragility. Am. J. Hum. Genet. 2018, 102, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Faivre, L.; Esteve, C.; Francescatto, L.; Tan, P.L.; Bourchany, A.; Delafoulhouze, C.; Marinier, E.; Bourgeois, P.; Brochier-Armanet, C.; Bruel, A.; et al. Description Osteo-Oto-Hepato-Enteric (O2HE) syndrome, a new recessive autosomal syndrome secondary to loss of function mutations in the UNC45A gene. Eur. J. Hum. Genet. EJHG 2019, 27, 795–796. [Google Scholar]
- Donkervoort, S.; Kutzner, C.E.; Hu, Y.; Lornage, X.; Rendu, J.; Stojkovic, T.; Baets, J.; Neuhaus, S.B.; Tanboon, J.; Maroofian, R.; et al. Pathogenic Variants in the Myosin Chaperone UNC-45B Cause Progressive Myopathy with Eccentric Cores. Am. J. Hum. Genet. 2020, 107, 1078–1095. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.J.; Pham, V.N.; Vogel, A.M.; Weinstein, B.M.; Roman, B.L. Loss of unc45a precipitates arteriovenous shunting in the aortic arches. Dev. Biol. 2008, 318, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Janiesch, P.C.; Kim, J.; Mouysset, J.; Barikbin, R.; Lochmuller, H.; Cassata, G.; Krause, S.; Hoppe, T. The ubiquitin-selective chaperone CDC-48/p97 links myosin assembly to human myopathy. Nat. Cell Biol. 2007, 9, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Comyn, S.A.; Pilgrim, D. Lack of developmental redundancy between Unc45 proteins in zebrafish muscle development. PLoS ONE 2012, 7, e48861. [Google Scholar] [CrossRef]
- Etard, C.; Roostalu, U.; Strahle, U. Shuttling of the chaperones Unc45b and Hsp90a between the A band and the Z line of the myofibril. J. Cell Biol. 2008, 180, 1163–1175. [Google Scholar] [CrossRef]
- Geach, T.J.; Zimmerman, L.B. Paralysis and delayed Z-disc formation in the Xenopus tropicalis unc45b mutant dicky ticker. BMC Dev. Biol. 2010, 1, 75. [Google Scholar] [CrossRef]
- Hansen, L.; Comyn, S.; Mang, Y.; Lind-Thomsen, A.; Myhre, L.; Jean, F.; Eiberg, H.; Tommerup, N.; Rosenberg, T.; Pilgrim, D. The myosin chaperone UNC45B is involved in lens development and autosomal dominant juvenile cataract. Eur. J. Hum. Genet. 2014, 22, 1290–1297. [Google Scholar] [CrossRef]
- Bazzaro, M.; Santillan, A.; Lin, Z.; Tang, T.; Lee, M.K.; Bristow, R.E.; Ie, M.S.; Roden, R.B. Myosin II co-chaperone general cell UNC-45 overexpression is associated with ovarian cancer, rapid proliferation, and motility. Am. J. Pathol. 2007, 171, 1640–1649. [Google Scholar] [CrossRef]
- Habicht, J.; Mooneyham, A.; Hoshino, A.; Shetty, M.; Zhang, X.; Emmings, E.; Yang, Q.; Coombes, C.; Gardner, M.K.; Bazzaro, M. UNC-45A breaks the microtubule lattice independently of its effects on non-muscle myosin II. J. Cell Sci. 2021, 134, jcs248815. [Google Scholar] [PubMed]
- Hoffman, C.S.; Wood, V.; Fantes, P.A. An Ancient Yeast for Young Geneticists: A Primer on the Schizosaccharomyces pombe Model System. Genetics 2015, 201, 403–423. [Google Scholar] [CrossRef] [PubMed]
- Escalante, S.G.; Brightmore, J.A.; Piper, P.W.; Millson, S.H. UCS protein function is partially restored in the Saccharomyces cerevisiae she4 mutant with expression of the human UNC45-GC, but not UNC45-SM. Cell Stress Chaperones 2018, 23, 609–615. [Google Scholar] [CrossRef]
- Hartman, M.A.; Spudich, J.A. The myosin superfamily at a glance. J. Cell Sci. 2012, 125, 1627–1632. [Google Scholar] [CrossRef]
- Amorim, M.J.; Mata, J. Rng3, a member of the UCS family of myosin co-chaperones, associates with myosin heavy chains cotranslationally. EMBO Rep. 2009, 10, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.C.Y.; Naqvi, N.I.; Iino, Y.; Yamamoto, M.; Balasubramanian, M.K. Fission yeast Rng3p: An UCS-domain protein that mediates myosin II assembly during cytokinesis. J. Cell Sci. 2000, 113, 2421–2432. [Google Scholar] [CrossRef]
- Lord, M.; Pollard, T.D. UCS protein Rng3p activates actin filament gliding by fission yeast myosin-II. J. Cell Biol. 2004, 167, 315–325. [Google Scholar] [CrossRef]
- Mishra, M.; D’souza, V.M.; Chang, K.C.; Huang, Y.; Balasubramanian, M.K. Hsp90 protein in fission yeast Swo1p and UCS protein Rng3p facilitate myosin II assembly and function. Eukaryot. Cell 2005, 4, 567–576. [Google Scholar] [CrossRef]
- Lord, M.; Sladewski, T.E.; Pollard, T.D. Yeast UCS proteins promote actomyosin interactions and limit myosin turnover in cells. Proc. Natl. Acad. Sci. USA 2008, 105, 8014–8019. [Google Scholar] [CrossRef]
- Stark, B.C.; James, M.L.; Pollard, L.W.; Sirotkin, V.; Lord, M. UCS protein Rng3p is essential for myosin-II motor activity during cytokinesis in fission yeast. PLoS ONE 2013, 8, e79593. [Google Scholar]
- Lord, M.; Laves, E.; Pollard, T.D. Cytokinesis depends on the motor domains of myosin-II in fission yeast but not in budding yeast. Mol. Biol. Cell 2005, 16, 5346–5355. [Google Scholar] [CrossRef] [PubMed]
- Wesche, S.; Arnold, M.; Jansen, R.-P. The UCS Domain Protein She4p Binds to Myosin Motor Domains and Is Essential for Class I and Class V Myosin Function. Curr. Biol. 2003, 13, 715–724. [Google Scholar] [CrossRef]
- Wendland, B.; McCaffery, J.M.; Xiao, Q.; Emr, S.D. A novel fluorescence-activated cell sorter-based screen for yeast endocytosis mutants identifies a yeast homologue of mammalian eps15. J. Cell Biol. 1996, 135, 1485–1500. [Google Scholar] [CrossRef]
- Goodson, H.V.; Anderson, B.L.; Warrick, H.M.; Pon, L.A.; Spudich, J.A. Synthetic lethality screen identifies a novel yeast myosin I gene (MYO5): Myosin I proteins are required for polarization of the actin cytoskeleton. J. Cell Biol. 1996, 133, 1277–1291. [Google Scholar] [CrossRef]
- Toi, H.; Fujimura-Kamada, K.; Irie, K.; Takai, Y.; Todo, S.; Tanaka, K. She4p/Dim1p interacts with the motor domain of unconventional myosins in the budding yeast, Saccharomyces cerevisiae. Mol. Biol. Cell 2003, 14, 2237–2249. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Escalante, S.; Piper, P.W.; Millson, S.H. Mutation of the Ser18 phosphorylation site on the sole Saccharomyces cerevisiae UCS protein, She4, can compromise high-temperature survival. Cell Stress Chaperones 2017, 22, 135–141. [Google Scholar] [CrossRef]
- Long, R.M.; Singer, R.H.; Meng, X.; Gonzalez, I.; Nasmyth, K.; Jansen, R.P. Mating type switching in yeast controlled by asymmetric localization of ASH1 mRNA. Science 1997, 277, 383–387. [Google Scholar] [CrossRef]
- Berteaux-Lecellier, V.; Zickler, D.; Debuchy, R.; Panvier-Adoutte, A.; Thompson-Coffe, C.; Picard, M. A homologue of the yeast SHE4 gene is essential for the transition between the syncytial and cellular stages during sexual reproduction of the fungus Podospora anserina. EMBO J. 1998, 17, 1248–1258. [Google Scholar] [CrossRef]
- Steinberg, G. On the move: Endosomes in fungal growth and pathogenicity. Nat. Rev. Microbiol. 2007, 5, 309–316. [Google Scholar] [CrossRef]
- Govindan, B.; Bowser, R.; Novick, P. The role of Myo2, a yeast class V myosin, in vesicular transport. J. Cell Biol. 1995, 128, 1055–1068. [Google Scholar] [CrossRef]
- Johnston, G.C.; Prendergast, J.A.; Singer, R.A. The Saccharomyces cerevisiae MYO2 gene encodes an essential myosin for vectorial transport of vesicles. J. Cell Biol. 1991, 113, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Schott, D.H.; Collins, R.N.; Bretscher, A. Secretory vesicle transport velocity in living cells depends on the myosin-V lever arm length. J. Cell Biol. 2002, 156, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Motegi, F.; Arai, R.; Mabuchi, I. Identification of two type V myosins in fission yeast, one of which functions in polarized cell growth and moves rapidly in the cell. Mol. Biol. Cell 2001, 12, 1367–1380. [Google Scholar] [CrossRef] [PubMed]
- Mulvihill, D.P.; Edwards, S.R.; Hyams, J.S. A critical role for the type V myosin, Myo52, in septum deposition and cell fission during cytokinesis in Schizosaccharomyces pombe. Cell Motil. Cytoskelet. 2006, 63, 149–161. [Google Scholar] [CrossRef]
- Win, T.Z.; Gachet, Y.; Mulvihill, D.P.; May, K.M.; Hyams, J.S. Two type V myosins with non-overlapping functions in the fission yeast Schizosaccharomyces pombe: Myo52 is concerned with growth polarity and cytokinesis, Myo51 is a component of the cytokinetic actin ring. J. Cell Sci. 2001, 114, 69–79. [Google Scholar] [CrossRef]
- Geli, M.I.; Riezman, H. Role of type I myosins in receptor-mediated endocytosis in yeast. Science 1996, 272, 533–535. [Google Scholar] [CrossRef]
- Renshaw, H.; Juvvadi, P.R.; Cole, D.C.; Steinbach, W.J. The class V myosin interactome of the human pathogen Aspergillus fumigatus reveals novel interactions with COPII vesicle transport proteins. Biochem. Biophys. Res. Commun. 2020, 527, 232–237. [Google Scholar] [CrossRef]
- Weber, I.; Gruber, C.; Steinberg, G. A class-V myosin required for mating, hyphal growth, and pathogenicity in the dimorphic plant pathogen Ustilago maydis. Plant. Cell 2003, 15, 2826–2842. [Google Scholar] [CrossRef]
- McGoldrick, C.A.; Gruver, C.; May, G.S. myoA of Aspergillus nidulans encodes an essential myosin I required for secretion and polarized growth. J. Cell Biol. 1995, 128, 577–587. [Google Scholar] [CrossRef]
- Osherov, N.; Yamashita, R.A.; Chung, Y.S.; May, G.S. Structural requirements for in vivo myosin I function in Aspergillus nidulans. J. Biol. Chem. 1998, 273, 27017–27025. [Google Scholar] [CrossRef][Green Version]
- Yamashita, R.A.; May, G.S. Constitutive activation of endocytosis by mutation of myoA, the myosin I gene of Aspergillus nidulans. J. Biol. Chem. 1998, 273, 14644–14648. [Google Scholar] [CrossRef] [PubMed]
- Oberholzer, U.; TIouk, L.; Thomas, D.Y.; Whiteway, M. Functional characterization of myosin I tail regions in Candida albicans. Eukaryot. Cell 2004, 3, 1272–1286. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Osherov, N.; Yamashita, R.; Brzeska, H.; Korn, E.D.; May, G.S. Myosin I mutants with only 1% of wild-type actin-activated MgATPase activity retain essential in vivo function(s). Proc. Natl. Acad. Sci. USA 2001, 98, 9122–9127. [Google Scholar] [CrossRef]
- Liu, L.; Srikakulam, R.; Winkelmann, D.A. Unc45 activates Hsp90-dependent folding of the myosin motor domain. J. Biol. Chem. 2008, 283, 13185–13193. [Google Scholar] [CrossRef] [PubMed]
- Srikakulam, R.; Liu, L.; Winkelmann, D.A. Unc45b forms a cytosolic complex with Hsp90 and targets the unfolded myosin motor domain. PLoS ONE 2008, 3, e2137. [Google Scholar] [CrossRef]
- Subbarao Sreedhar, A.; Kalmár, É.; Csermely, P.; Shen, Y.-F. Hsp90 isoforms: Functions, expression and clinical importance. FEBS Lett. 2004, 562, 11–15. [Google Scholar] [CrossRef]
- Etard, C.; Behra, M.; Fischer, N.; Hutcheson, D.; Geisler, R.; Strahle, U. The UCS factor Steif/Unc-45b interacts with the heat shock protein Hsp90a during myofibrillogenesis. Dev. Biol. 2007, 308, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Du, S.J.; Li, H.; Bian, Y.; Zhong, Y. Heat-shock protein 90alpha1 is required for organized myofibril assembly in skeletal muscles of zebrafish embryos. Proc. Natl. Acad. Sci. USA 2008, 105, 554–559. [Google Scholar] [CrossRef]
- Krone, P.H.; Evans, T.G.; Blechinger, S.R. Heat shock gene expression and function during zebrafish embryogenesis. Semin. Cell Dev. Biol. 2003, 14, 267–274. [Google Scholar] [CrossRef]
- Grad, I.; Cederroth, C.R.; Walicki, J.; Grey, C.; Barluenga, S.; Winssinger, N.; de Massy, B.; Nef, S.; Picard, D. The molecular chaperone Hsp90α is required for meiotic progression of spermatocytes beyond pachytene in the mouse. PLoS ONE 2010, 5, e15770. [Google Scholar] [CrossRef]
- Etard, C.; Roostalu, U.; Strähle, U. Lack of Apobec2-related proteins causes a dystrophic muscle phenotype in zebrafish embryos. J. Cell Biol. 2010, 189, 527–539. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Millson, S.H.; Truman, A.W.; Wolfram, F.; King, V.; Panaretou, B.; Prodromou, C.; Pearl, L.H.; Piper, P.W. Investigating the protein-protein interactions of the yeast Hsp90 chaperone system by two-hybrid analysis: Potential uses and limitations of this approach. Cell Stress Chaperones 2004, 9, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Millson, S.H.; Truman, A.W.; King, V.; Prodromou, C.; Pearl, L.H.; Piper, P.W. A two-hybrid screen of the yeast proteome for Hsp90 interactors uncovers a novel Hsp90 chaperone requirement in the activity of a stress-activated mitogen-activated protein kinase, Slt2p (Mpk1p). Eukaryot. Cell 2005, 4, 849–860. [Google Scholar] [CrossRef] [PubMed]
- Bujalowski, P.J.; Nicholls, P.; Barral, J.M.; Oberhauser, A.F. Thermally-induced structural changes in an armadillo repeat protein suggest a novel thermosensor mechanism in a molecular chaperone. FEBS Lett. 2015, 589, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Hellerschmied, D.; Roessler, M.; Lehner, A.; Gazda, L.; Stejskal, K.; Imre, R.; Mechtler, K.; Dammermann, A.; Clausen, T. UFD-2 is an adaptor-assisted E3 ligase targeting unfolded proteins. Nat. Commun. 2018, 9, 484. [Google Scholar] [CrossRef]
- Wang, D.Y.; Kumar, S.; Hedges, S.B. Divergence time estimates for the early history of animal phyla and the origin of plants, animals and fungi. Proc. Biol. Sci. 1999, 266, 163–171. [Google Scholar] [CrossRef]
- Bookwalter, C.S.; Tay, C.L.; McCrorie, R.; Previs, M.J.; Lu, H.; Krementsova, E.B.; Fagnant, P.M.; Baum, J.; Trybus, K.M. Reconstitution of the core of the malaria parasite glideosome with recombinant Plasmodium class XIV myosin A and Plasmodium actin. J. Biol. Chem. 2017, 292, 19290–19303. [Google Scholar] [CrossRef]
- Berbee, M.L.; James, T.Y.; Strullu-Derrien, C. Early diverging fungi: Diversity and impact at the dawn of terrestrial life. Annu. Rev. Microbiol. 2017, 71, 41–60. [Google Scholar] [CrossRef]
- Brunet, T.; Albert, M.; Roman, W.; Coyle, M.C.; Spitzer, D.C.; King, N. A flagellate-to-amoeboid switch in the closest living relatives of animals. eLife 2021, 10, e61037. [Google Scholar] [CrossRef]
- Patel, P.K.; Free, S.J. The Genetics and Biochemistry of Cell Wall Structure and Synthesis in Neurospora crassa, a Model Filamentous Fungus. Front. Microbiol. 2019, 10, 2294. [Google Scholar] [CrossRef]
- Frumkin, A.; Dror, S.; Pokrzywa, W.; Bar-Lavan, Y.; Karady, I.; Hoppe, T.; Ben-Zvi, A. Challenging muscle homeostasis uncovers novel chaperone interactions in Caenorhabditis elegans. Front. Mol. Biosci. 2017, 1, 21. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piper, P.W.; Scott, J.E.; Millson, S.H. UCS Chaperone Folding of the Myosin Head: A Function That Evolved before Animals and Fungi Diverged from a Common Ancestor More than a Billion Years Ago. Biomolecules 2022, 12, 1028. https://doi.org/10.3390/biom12081028
Piper PW, Scott JE, Millson SH. UCS Chaperone Folding of the Myosin Head: A Function That Evolved before Animals and Fungi Diverged from a Common Ancestor More than a Billion Years Ago. Biomolecules. 2022; 12(8):1028. https://doi.org/10.3390/biom12081028
Chicago/Turabian StylePiper, Peter William, Julia Elizabeth Scott, and Stefan Heber Millson. 2022. "UCS Chaperone Folding of the Myosin Head: A Function That Evolved before Animals and Fungi Diverged from a Common Ancestor More than a Billion Years Ago" Biomolecules 12, no. 8: 1028. https://doi.org/10.3390/biom12081028
APA StylePiper, P. W., Scott, J. E., & Millson, S. H. (2022). UCS Chaperone Folding of the Myosin Head: A Function That Evolved before Animals and Fungi Diverged from a Common Ancestor More than a Billion Years Ago. Biomolecules, 12(8), 1028. https://doi.org/10.3390/biom12081028





