Transcriptomic Profiling Reveals That HMGB1 Induces Macrophage Polarization Different from Classical M1
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Harvesting and Culturing of BMDMs
2.3. Recombinant HMGB1 Production
2.4. Cell Culture Experiments
2.5. RNA-Seq
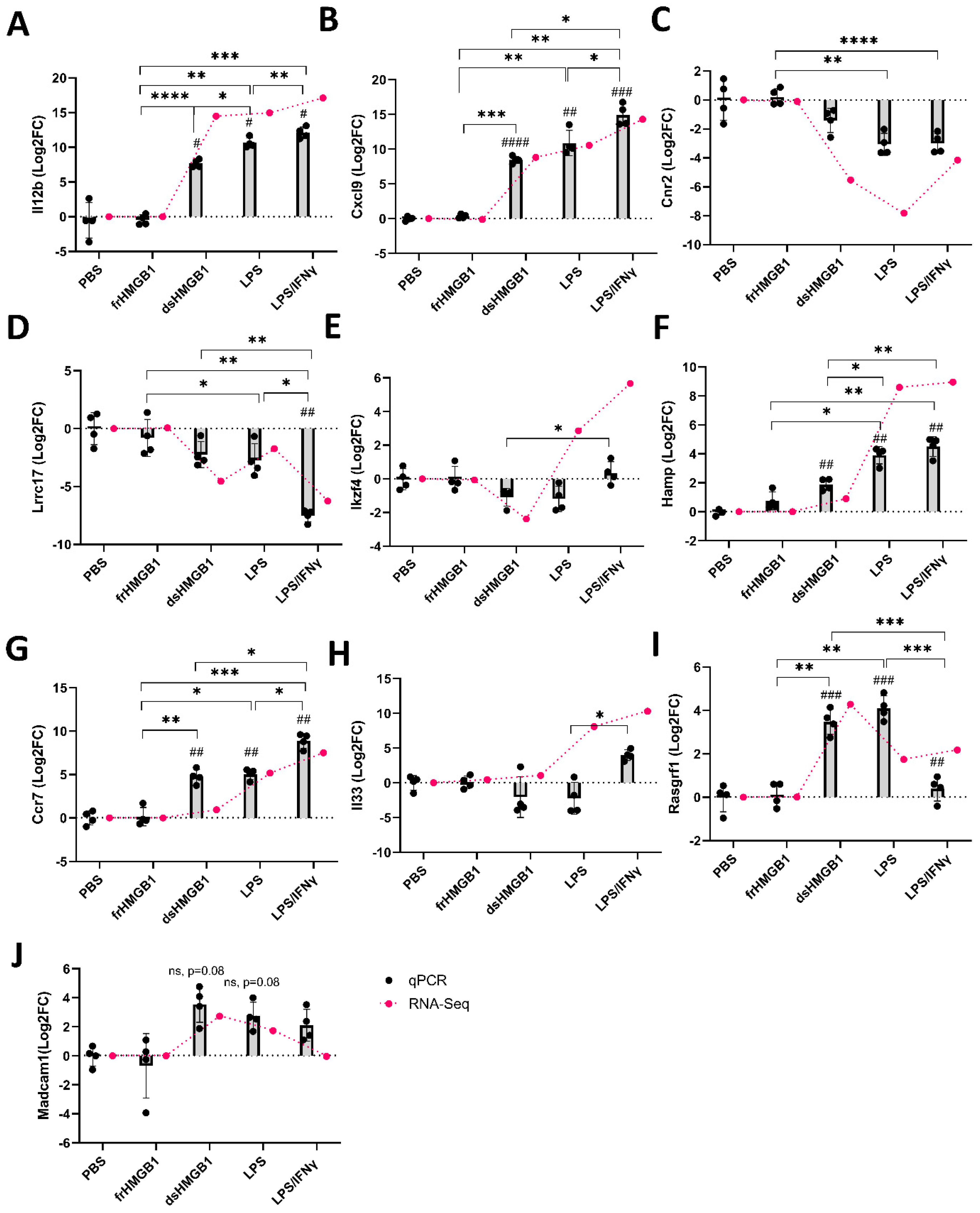
2.6. Verification of Differentially Expressed Genes Identified in RNA-Seq
2.7. Oil Red O Staining
2.8. Statistical Analysis
3. Results
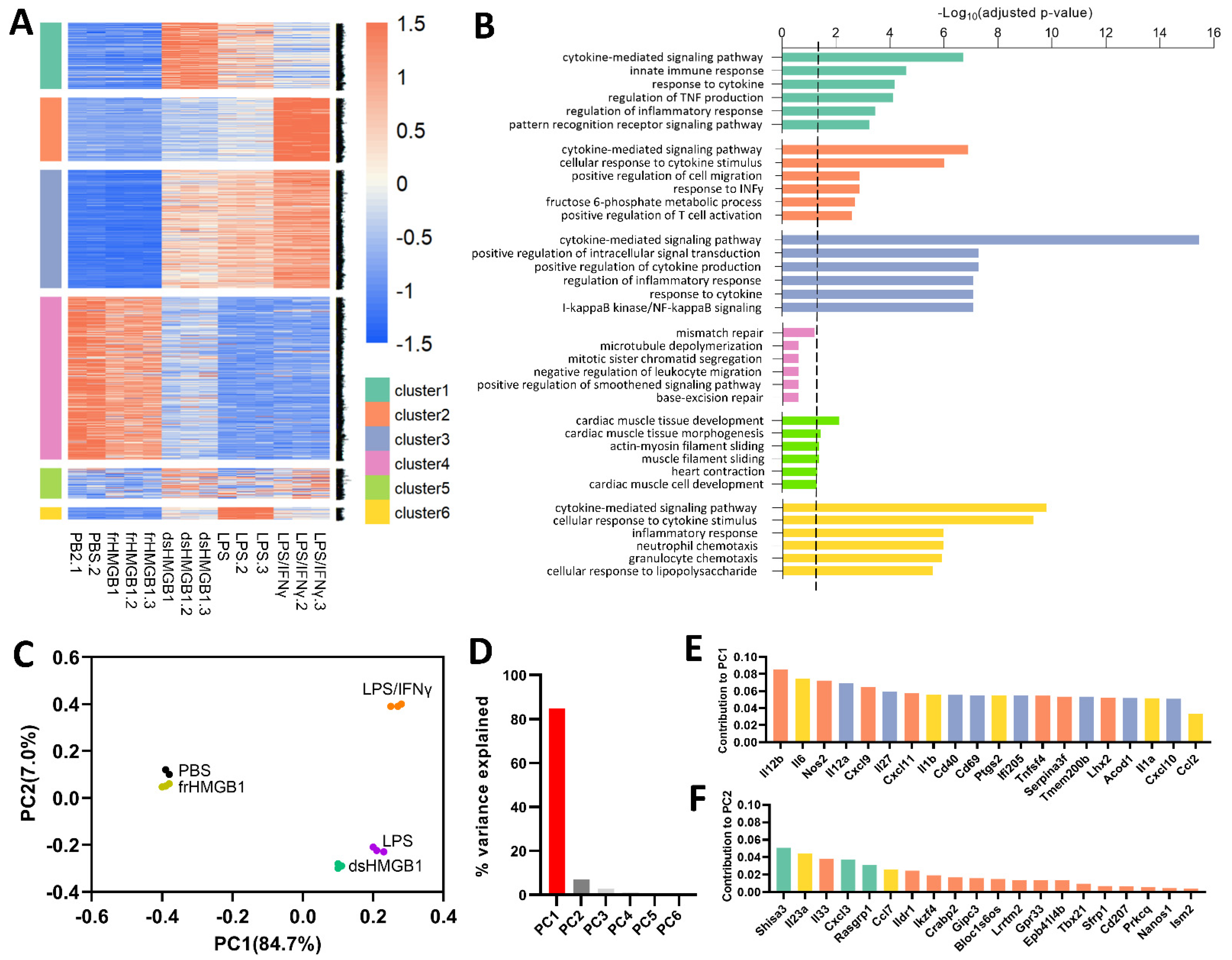
3.1. dsHMGB1-, LPS- and LPS/IFNγ-Stimulation Resulted in Similar Pro-Inflammatory Gene Expression Patterns, While frHMGB1 Overlapped with PBS Control
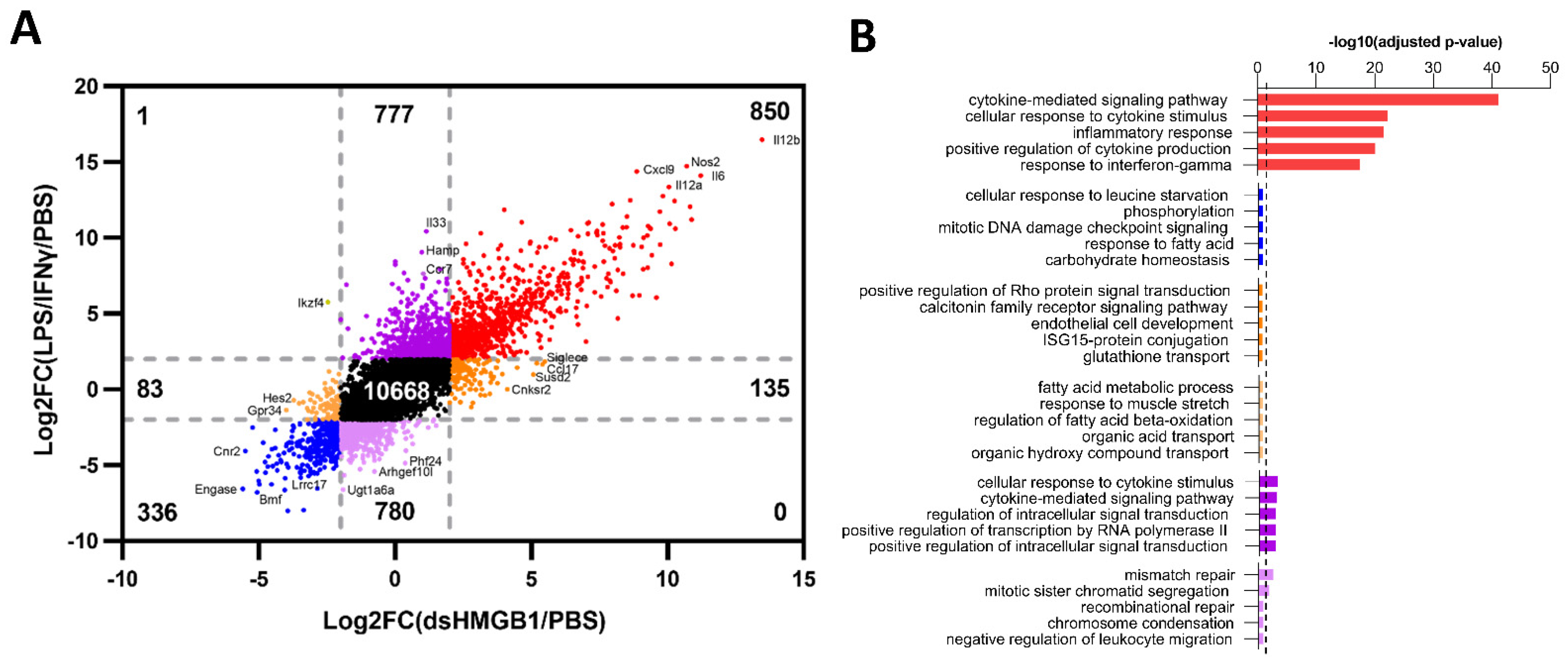
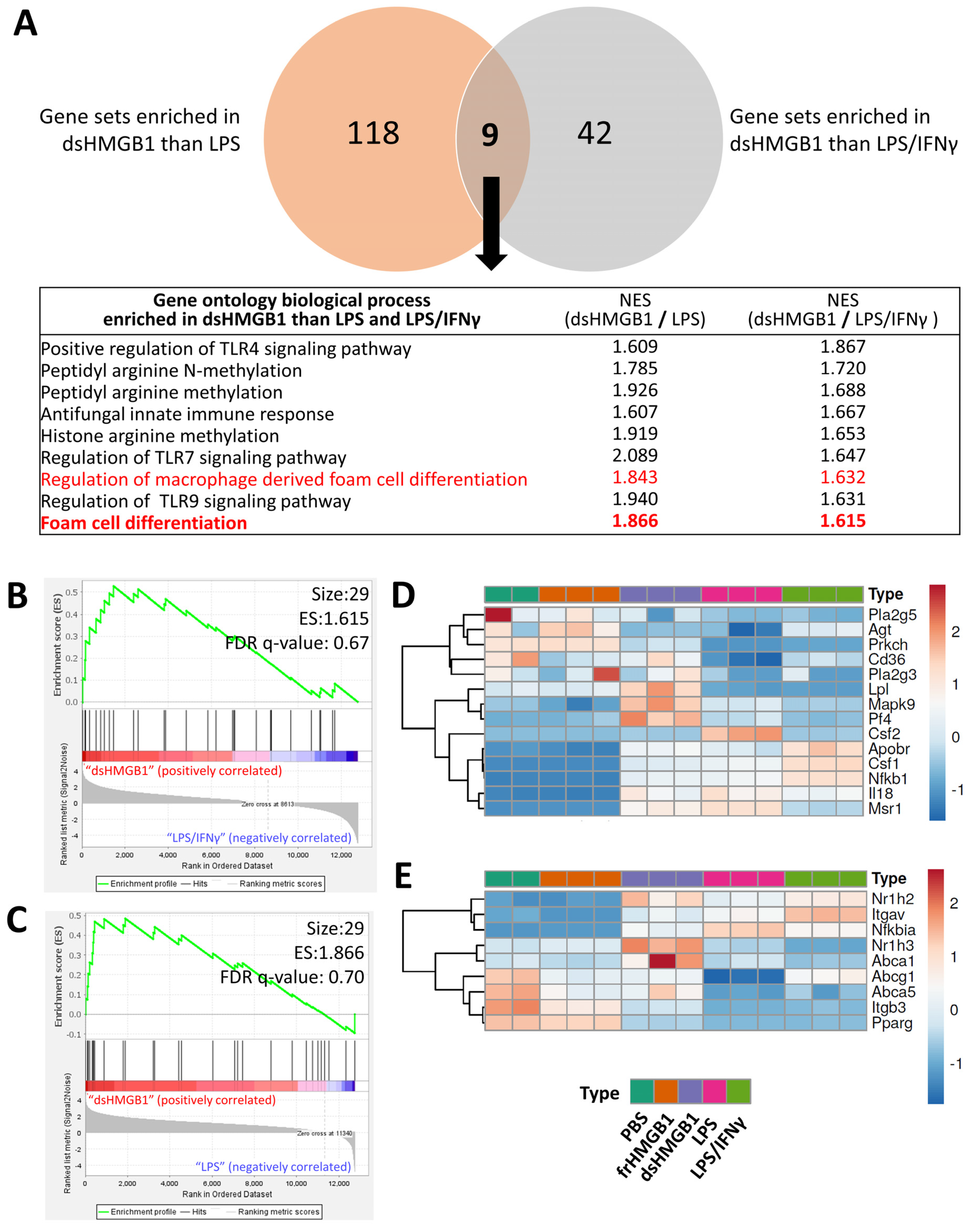
3.2. Identification of Gene Signatures in dsHMGB1- Compared to LPS/IFNγ-Treated BMDMs
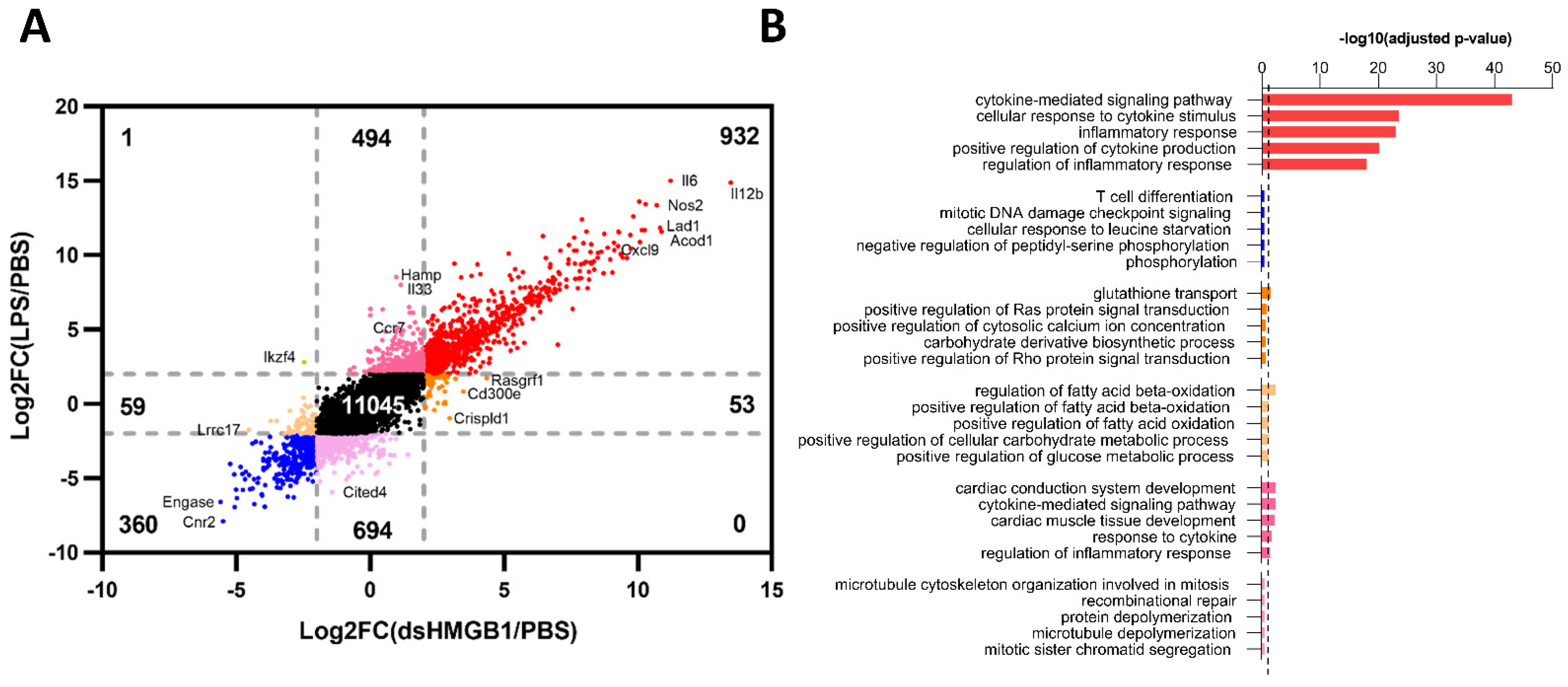
3.3. Identification of Gene Signatures in dsHMGB1- Compared to LPS-Treated BMDMs
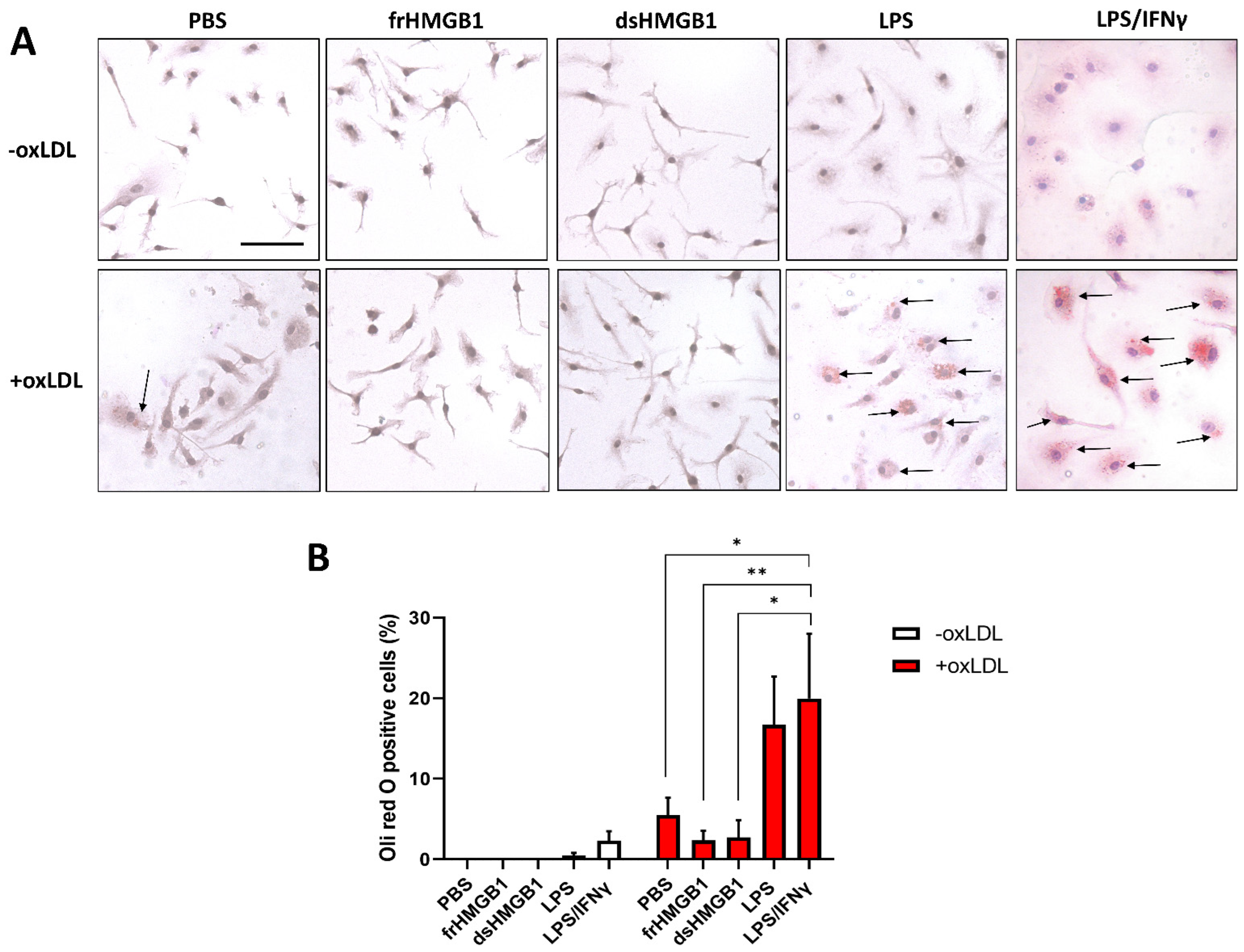
3.4. frHMGB1 and dsHMGB1 Attenuated BMDM-Derived Foam Cell Formation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hamidzadeh, K.; Christensen, S.M.; Dalby, E.; Chandrasekaran, P.; Mosser, D.M. Macrophages and the Recovery from Acute and Chronic Inflammation. Annu. Rev. Physiol. 2017, 79, 567–592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murray, P.J. Macrophage Polarization. Annu. Rev. Physiol. 2017, 79, 541–566. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, L.; Foglio, E.; Pontemezzo, E.; Germani, A.; Russo, M.A.; Limana, F. Hmgb1 and Repair: Focus on the Heart. Pharmacol. Ther. 2019, 196, 160–182. [Google Scholar] [CrossRef] [PubMed]
- Scaffidi, P.; Misteli, T.; Bianchi, M.E. Release of Chromatin Protein Hmgb1 by Necrotic Cells Triggers Inflammation. Nature 2002, 418, 191–195. [Google Scholar] [CrossRef]
- Kang, R.; Zhang, Q.; Hou, W.; Yan, Z.; Chen, R.; Bonaroti, J.; Bansal, P.; Billiar, T.R.; Tsung, A.; Wang, Q.; et al. Intracellular Hmgb1 Inhibits Inflammatory Nucleosome Release and Limits Acute Pancreatitis in Mice. Gastroenterology 2014, 146, 1097–1107.e8. [Google Scholar] [CrossRef] [Green Version]
- Tirone, M.; Tran, N.L.; Ceriotti, C.; Gorzanelli, A.; Canepari, M.; Bottinelli, R.; Raucci, A.; Di Maggio, S.; Santiago, C.; Mellado, M.; et al. High Mobility Group Box 1 Orchestrates Tissue Regeneration via Cxcr4. J. Exp. Med. 2018, 215, 303–318. [Google Scholar] [CrossRef]
- Yang, H.; Wang, H.; Ju, Z.; Ragab, A.A.; Lundbäck, P.; Long, W.; Valdés-Ferrer, S.I.; He, M.; Pribis, J.P.; Li, J.; et al. Md-2 Is Required for Disulfide Hmgb1-Dependent Tlr4 Signaling. J. Exp. Med. 2015, 212, 5–14. [Google Scholar] [CrossRef] [Green Version]
- Hreggvidsdóttir, H.S.; Lundberg, A.M.; Aveberger, A.-C.; Klevenvall, L.; Andersson, U.; Harris, H.E. High Mobility Group Box Protein 1 (Hmgb1)-Partner Molecule Complexes Enhance Cytokine Production by Signaling through the Partner Molecule Receptor. Mol. Med. Camb. Mass 2012, 18, 224–230. [Google Scholar] [CrossRef]
- Liu, F.-Y.; Fan, D.; Yang, Z.; Tang, N.; Guo, Z.; Ma, S.-Q.; Ma, Z.-G.; Wu, H.-M.; Deng, W.; Tang, Q.-Z. Tlr9 Is Essential for Hmgb1-Mediated Post-Myocardial Infarction Tissue Repair through Affecting Apoptosis, Cardiac Healing, and Angiogenesis. Cell Death Dis. 2019, 10, 480. [Google Scholar] [CrossRef]
- Aucott, H.; Sowinska, A.; Harris, H.E.; Lundback, P. Ligation of Free Hmgb1 to Tlr2 in the Absence of Ligand Is Negatively Regulated by the C-Terminal Tail Domain. Mol. Med. Camb. Mass 2018, 24, 19. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.; Kwak, M.; Kim, S.; Shin, J. The Role of High Mobility Group Box 1 in Innate Immunity. Yonsei Med. J. 2014, 55, 1165–1176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Bloom, O.; Zhang, M.; Vishnubhakat, J.M.; Ombrellino, M.; Che, J.; Frazier, A.; Yang, H.; Ivanova, S.; Borovikova, L.; et al. Hmg-1 as a Late Mediator of Endotoxin Lethality in Mice. Science 1999, 285, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, R.S.; Bruchfeld, A.; Yang, L.; Qureshi, A.R.; Gallowitsch-Puerta, M.; Patel, N.B.; Huston, B.J.; Chavan, S.; Rosas-Ballina, M.; Gregersen, P.K.; et al. Cholinergic Anti-Inflammatory Pathway Activity and High Mobility Group Box-1 (Hmgb1) Serum Levels in Patients with Rheumatoid Arthritis. Mol. Med. 2007, 13, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Schierbeck, H.; Pullerits, R.; Pruunsild, C.; Fischer, M.; Holzinger, D.; Laestadius, Å.; Sundberg, E.; Harris, H.E. Hmgb1 Levels Are Increased in Patients with Juvenile Idiopathic Arthritis, Correlate with Early Onset of Disease, and Are Independent of Disease Duration. J. Rheumatol. 2013, 40, 1604–1613. [Google Scholar] [CrossRef] [Green Version]
- Liu, T.; Son, M.; Diamond, B. Hmgb1 in Systemic Lupus Erythematosus. Front. Immunol. 2020, 11, 1057. [Google Scholar] [CrossRef]
- Aulin, C.; Harris, H.; Larsson, S.; Struglics, A. Increased Levels of the Alarmin High Mobility Group Box 1 Is Detected in Synovial Fluid after Acute Knee Injury. Osteoarthr. Cartil. 2019, 27, S106–S107. [Google Scholar] [CrossRef] [Green Version]
- Vogelgesang, A.; May, V.E.L.; Grunwald, U.; Bakkeboe, M.; Langner, S.; Wallaschofski, H.; Kessler, C.; Bröker, B.M.; Dressel, A. Functional Status of Peripheral Blood T-Cells in Ischemic Stroke Patients. PLoS ONE 2010, 5, e8718. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.; Wang, H.; Andersson, U. Targeting Inflammation Driven by Hmgb1. Front. Immunol. 2020, 11, 484. [Google Scholar] [CrossRef] [Green Version]
- Lyu, J.; Xie, D.; Bhatia, T.N.; Leak, R.K.; Hu, X.; Jiang, X. Microglial/Macrophage Polarization and Function in Brain Injury and Repair after Stroke. CNS Neurosci. Ther. 2021, 27, 515–527. [Google Scholar] [CrossRef]
- Fujiwara, N.; Kobayashi, K. Macrophages in Inflammation. Curr. Drug Targets-Inflamm. Allergy 2005, 4, 281–286. [Google Scholar] [CrossRef]
- Oishi, Y.; Manabe, I. Macrophages in Inflammation, Repair and Regeneration. Int. Immunol. 2018, 30, 511–528. [Google Scholar] [CrossRef] [PubMed]
- Salo, H.; Qu, H.; Mitsiou, D.; Aucott, H.; Han, J.; Zhang, X.; Aulin, C.; Harris, H.E. Disulfide and Fully Reduced Hmgb1 Induce Different Macrophage Polarization and Migration Patterns. Biomolecules 2021, 11, 800. [Google Scholar] [CrossRef] [PubMed]
- Weischenfeldt, J.; Porse, B. Bone Marrow-Derived Macrophages (Bmm): Isolation and Applications. Cold Spring Harb. Protoc. 2008, 2008, pdb-prot5080. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, H.; Hreggvidsdottir, H.S.; Palmblad, K.; Wang, H.; Ochani, M.; Li, J.; Lu, B.; Chavan, S.; Rosas-Ballina, M.; Al-Abed, Y.; et al. A Critical Cysteine Is Required for Hmgb1 Binding to Toll-Like Receptor 4 and Activation of Macrophage Cytokine Release. Proc. Natl. Acad. Sci. USA 2010, 107, 11942–11947. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. Star: Ultrafast Universal Rna-Seq Aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated Estimation of Fold Change and Dispersion for Rna-Seq Data with Deseq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [Green Version]
- Kuleshov, M.V.; Jones, M.R.; Rouillard, A.D.; Fernandez, N.F.; Duan, Q.; Wang, Z.; Koplev, S.; Jenkins, S.L.; Jagodnik, K.M.; Lachmann, A.; et al. Enrichr: A Comprehensive Gene Set Enrichment Analysis Web Server 2016 Update. Nucleic Acids Res. 2016, 44, W90–W97. [Google Scholar] [CrossRef] [Green Version]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene Set Enrichment Analysis: A Knowledge-Based Approach for Interpreting Genome-Wide Expression Profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [Green Version]
- Martinez, F.O.; Gordon, S. The M1 and M2 Paradigm of Macrophage Activation: Time for Reassessment. F1000prime Rep. 2014, 6, 13. [Google Scholar] [CrossRef] [Green Version]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. Nf-Κb Signaling in Inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [Green Version]
- Abreu, J.R.; De Launay, D.; E Sanders, M.; Grabiec, A.M.; Sande, M.G.; Tak, P.P.; A Reedquist, K. The Ras Guanine Nucleotide Exchange Factor Rasgrf1 Promotes Matrix Metalloproteinase-3 Production in Rheumatoid Arthritis Synovial Tissue. Arthritis Res. Ther. 2009, 11, R121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schippers, A.; Leuker, C.; Pabst, O.; Kochut, A.; Prochnow, B.; Gruber, A.D.; Leung, E.; Krissansen, G.W.; Wagner, N.; Müller, W. Mucosal Addressin Cell-Adhesion Molecule-1 Controls Plasma-Cell Migration and Function in the Small Intestine of Mice. Gastroenterology 2009, 137, 924–933. [Google Scholar] [CrossRef]
- Kuhbandner, K.; Hammer, A.; Haase, S.; Terbrack, E.; Hoffmann, A.; Schippers, A.; Wagner, N.; Hussain, R.Z.; Miller-Little, W.A.; Koh, A.Y.; et al. Madcam-1-Mediated Intestinal Lymphocyte Homing Is Critical for the Development of Active Experimental Autoimmune Encephalomyelitis. Front. Immunol. 2019, 10, 903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wunderer, F.; Traeger, L.; Sigurslid, H.H.; Meybohm, P.; Bloch, D.B.; Malhotra, R. The Role of Hepcidin and Iron Homeostasis in Atherosclerosis. Pharmacol. Res. 2020, 153, 104664. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Liang, M.C.; Soong, T.W. Nitric Oxide, Iron and Neurodegeneration. Front. Neurosci. 2019, 13, 114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, M.; Tang, Y.; Li, W.; Wang, X.; Zhang, R.; Zhang, X.; Zhao, X.; Liu, J.; Tang, C.; Liu, Z.; et al. The Endotoxin Delivery Protein Hmgb1 Mediates Caspase-11-Dependent Lethality in Sepsis. Immunity 2018, 49, 740–753.e7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, D.Y.; Teyssier, C.; Strahl, B.D.; Stallcup, M.R. Role of Protein Methylation in Regulation of Transcription. Endocr. Rev. 2005, 26, 147–170. [Google Scholar] [CrossRef]
- Wang, Y.; Dubland, J.A.; Allahverdian, S.; Asonye, E.; Sahin, B.; Jaw, J.E.; Sin, D.D.; Seidman, M.A.; Leeper, N.J.; Francis, G.A. Smooth Muscle Cells Contribute the Majority of Foam Cells in Apoe (Apolipoprotein E)-Deficient Mouse Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 876–887. [Google Scholar] [CrossRef]
- Owsiany, K.M.; Alencar, G.F.; Owens, G.K. Revealing the Origins of Foam Cells in Atherosclerotic Lesions. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 836–838. [Google Scholar] [CrossRef]
- Cookson, F.B. The Origin of Foam Cells in Atherosclerosis. Br. J. Exp. Pathol. 1971, 52, 62–69. [Google Scholar]
- Yu, X.H.; Fu, Y.C.; Zhang, D.W.; Yin, K.; Tang, C.K. Foam Cells in Atherosclerosis. Clin. Chim. Acta 2013, 424, 245–252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, R.; Wu, W.; Li, W.; Huang, S.; Li, Z.; Liu, R.; Shan, Z.; Zhang, C.; Wang, S. Activation of Nlrp3 Inflammasome Promotes Foam Cell Formation in Vascular Smooth Muscle Cells and Atherogenesis via Hmgb1. J. Am. Heart Assoc. 2018, 7, e008596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, H.; Chen, Z.; Chen, J.-Z.; Pei, L.-G.; Xie, J.; Wei, Z.-H.; Kang, L.-N.; Wang, L.; Xu, B. High Mobility Group B-1 (Hmgb-1) Promotes Apoptosis of Macrophage-Derived Foam Cells by Inducing Endoplasmic Reticulum Stress. Cell. Physiol. Biochem. 2018, 48, 1019–1029. [Google Scholar] [CrossRef]
- Pirillo, A.; Norata, G.D.; Catapano, A.L. Lox-1, Oxldl, and Atherosclerosis. Mediat. Inflamm. 2013, 2013, 152786. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, A.-W.; Huang, C.-Y.; Shih, C.-M.; Lin, Y.-W.; Tsao, N.-W.; Chen, Y.H.; Chang, Y.-J.; Chang, N.-C.; Li, C.-Y.; Fong, T.-H.; et al. Ursolic Acid Attenuates Hmgb1-Induced Lox-1 Expression in Vascular Endothelial Cells in Vitro and Inhibits Atherogenesis in Hypercholesterolemic Mice in Vivo. Immunol. Endocr. Metab. Agents Med. Chem. Under Re-Organ. 2012, 12, 317–329. [Google Scholar] [CrossRef]
- Liu, L.; Lu, Y.; Martinez, J.; Bi, Y.; Lian, G.; Wang, T.; Milasta, S.; Wang, J.; Yang, M.; Liu, G.; et al. Proinflammatory Signal Suppresses Proliferation and Shifts Macrophage Metabolism from Myc-Dependent to Hif1α-Dependent. Proc. Natl. Acad. Sci. USA 2016, 113, 1564–1569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McNelis, J.C.; Olefsky, J.M. Macrophages, Immunity, and Metabolic Disease. Immunity 2014, 41, 36–48. [Google Scholar] [CrossRef] [Green Version]
- Hubert, P.; Roncarati, P.; Demoulin, S.; Pilard, C.; Ancion, M.; Reynders, C.; Lerho, T.; Bruyere, D.; Lebeau, A.; Radermecker, C.; et al. Extracellular Hmgb1 Blockade Inhibits Tumor Growth through Profoundly Remodeling Immune Microenvironment and Enhances Checkpoint Inhibitor-Based Immunotherapy. J. Immunother. Cancer 2021, 9, e001966. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, H.; Heinbäck, R.; Salo, H.; Ewing, E.; Espinosa, A.; Aulin, C.; Erlandsson Harris, H. Transcriptomic Profiling Reveals That HMGB1 Induces Macrophage Polarization Different from Classical M1. Biomolecules 2022, 12, 779. https://doi.org/10.3390/biom12060779
Qu H, Heinbäck R, Salo H, Ewing E, Espinosa A, Aulin C, Erlandsson Harris H. Transcriptomic Profiling Reveals That HMGB1 Induces Macrophage Polarization Different from Classical M1. Biomolecules. 2022; 12(6):779. https://doi.org/10.3390/biom12060779
Chicago/Turabian StyleQu, Heshuang, Rebecka Heinbäck, Henna Salo, Ewoud Ewing, Alexander Espinosa, Cecilia Aulin, and Helena Erlandsson Harris. 2022. "Transcriptomic Profiling Reveals That HMGB1 Induces Macrophage Polarization Different from Classical M1" Biomolecules 12, no. 6: 779. https://doi.org/10.3390/biom12060779
APA StyleQu, H., Heinbäck, R., Salo, H., Ewing, E., Espinosa, A., Aulin, C., & Erlandsson Harris, H. (2022). Transcriptomic Profiling Reveals That HMGB1 Induces Macrophage Polarization Different from Classical M1. Biomolecules, 12(6), 779. https://doi.org/10.3390/biom12060779





